Aditi Senthil Prasad1, Makaila Furderer2
1 North Carolina School of Science and Maths
2 University of Michigan
Abstract
Previous studies have shown that nano- and microplastics (NMPs) exhibit size and volume-dependent neurotoxicity due to their ability to cross the blood-brain barrier (BBB), where they have been theorized to induce oxidative stress and neuroinflammation, both of which play a role in neurodegeneration and neurodegenerative disorder development. Antioxidants are currently being explored as a mitigation strategy, but their protective mechanisms, especially against NMP-induced neurotoxicity, remain poorly understood. Using a literature review approach, this paper aims to further understand the various mechanisms by which NMPs might elevate oxidative stress and neuroinflammation, how antioxidants might function to alleviate these factors, and how they may reduce or protect against NMP-induced neurodegeneration. Overall, NMPs use various mechanisms and pathways to induce neuroinflammation and oxidative stress, both mutually reinforcing drivers of plastic-induced neural damage. Antioxidants demonstrate potential to mitigate NMP-induced neurotoxicity by scavenging for free radicals, supporting mitochondrial integrity, modulating inflammatory signaling, etc. However, direct evidence of antioxidant-plastic interactions in neurodegeneration remains limited, highlighting the need for targeted future research.
Keywords: Nano and microplastics, neurotoxicity, antioxidants, neuroinflammation, oxidative stress, BBB
Introduction
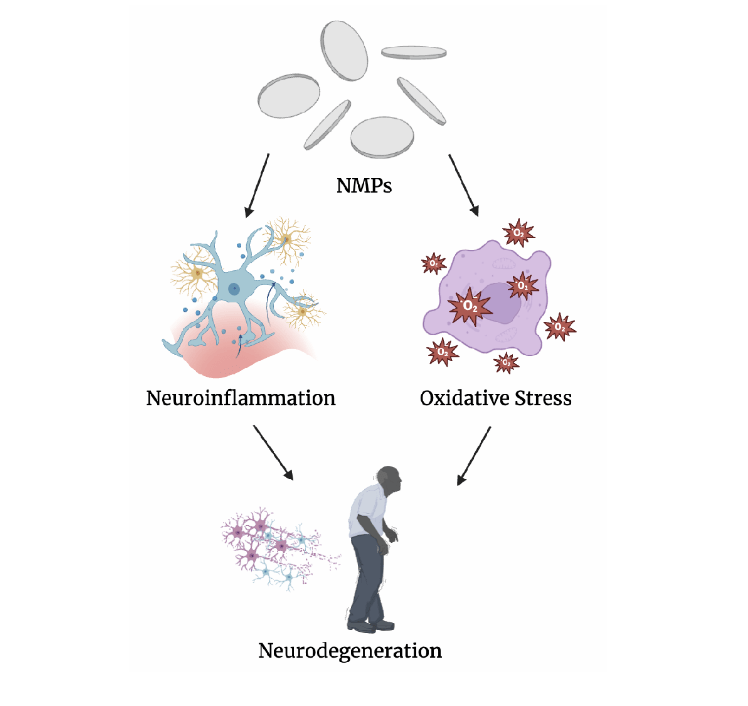
Nano- and microplastics (NMPs) are plastics typically smaller than 5mm in size and have become a major concern over the past few years. Specifically, since the COVID-19 pandemic, the widespread use of gloves and masks has led to an increase in microplastics, which have further accumulated in various ecosystems. As a rising issue, the current trend shows that by 2050, 12 billion tons of microplastics will be present in the environment. On account of the surge in pollution over recent years, human consumption of these plastics has also risen. Humans consume these plastics through contact, ingestion, and most commonly, inhalation1,2. NMPs have also shown to cross into offspring’s bodies through their mothers3,4. Because these NMPs can penetrate the BBB and induce neuroinflammation and oxidative stress, they have an extremely high neurotoxic potential5,6,7. It has also been shown the NMPs can decline mice’s cognitive ability and induce neuroinflammation in their hippocampus, causing neural damage and behavioral changes8,9. It is known that neuroinflammation and oxidative stress are both causes and progressors of neurodegeneration and neurodegenerative diseases such as Alzheimer’s and Parkinson’s disease10,11. One proposed solution for alleviation of neurodegeneration is the use of antioxidants against this neuroinflammation and oxidative stress12. This paper aims to dive into the mechanisms antioxidants use to improve blood-brain barrier health to protect the brain from the neurotoxicity and threat of neurodegenerative diseases posed by nano- and microplastics. Several research gaps exist in the research, including nanoplastics, exact correlation to neurodegeneration, the exact health effects of NMPs, and the direct correlation to antioxidants. In acknowledging these research gaps, this paper aims to synthesize current evidence on the mechanisms by which NMPs induce neuroinflammation and oxidative stress and pathways by which antioxidant pathways mitigate neuroinflammation and oxidative stress to later assess their ability to mitigate NMP-induced neurodegeneration. As shown in Figure 1, NMPs trigger a series of neurotoxic processes. After crossing the blood-brain barrier, NMPs trigger the generation of ROS species, leading to oxidative stress and also simultaneously stimulating neuroinflammation through microglial activation. Both of these processes reinforce each other cyclically. A hallmark of all neurodegenerative diseases is neuroinflammation and oxidative stress.
Methods
The literature reviewed in this paper encompasses experimental, mechanistic, and observational investigations looking into how neural systems interact with NMPs. Research on neurotoxicity pathways, such as oxidative stress, neuroinflammation, disruption of the blood-brain barrier, and neuronal dysfunction, as well as studies investigating the modulatory impact of antioxidants in reducing these effects, were prioritized. The body of literature includes in vitro neuronal models, in vivo animal studies, and select human studies, which provide insight into disease relevance and progression. Collectively, these sources offer a current understanding of both the molecular basis of NMP-induced neurotoxicity and developing neuroprotective treatments, allowing for a thorough synthesis of trends, consistencies, and gaps in the field. Key words such as neurodegenerative, oxidative stress, nano- and microplastics, neurotoxicity, neuroinflammation, antioxidants, and blood-brain barrier were used to ensure that all research papers were relevant to the topic of the literature review.
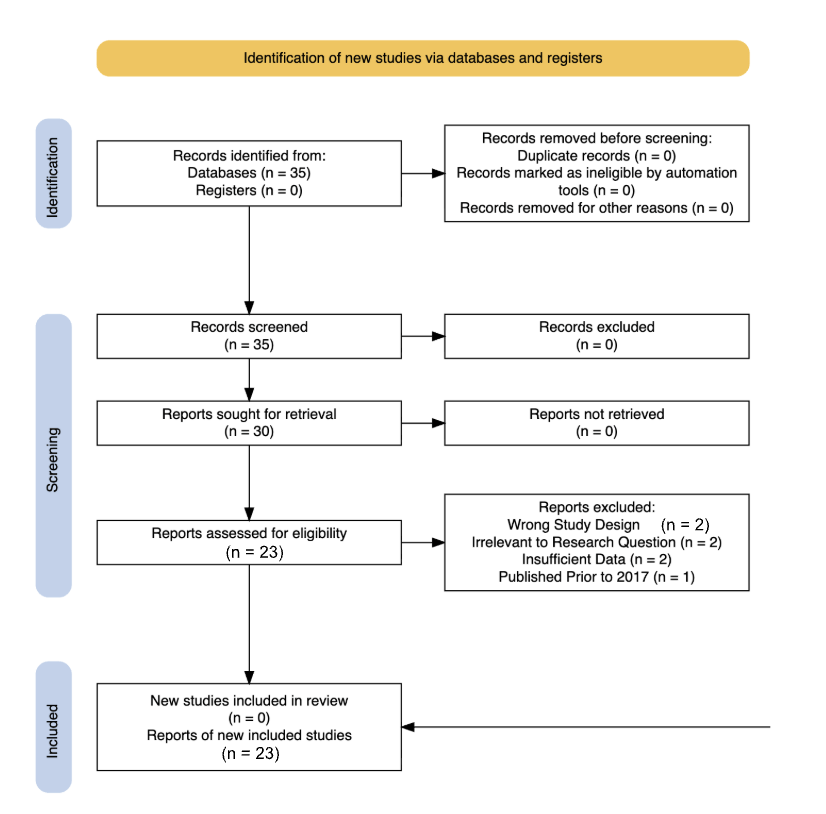
Literature for this review was identified utilizing the Google Scholar database. Searches were restricted to include only studies published between 2017 and 2024 to ensure relevance to current research on NMP neurotoxicity. Studies were included if they investigated the effects NMPs had on neural systems, neurodegenerative disease-related pathways, or antioxidant-mediated mitigation mechanisms, using in vitro, in vivo, or human-based models. A few studies focusing on NMP interactions with other biological barriers were also included to study the comparison of how NMPs interact with other barriers versus the BBB. Studies were excluded if they focused solely on environmental distribution, disregarding biological assessment, macroplastic exposure, etc. Following title and abstract screening and full-text review, approximately 23 peer-reviewed articles were selected for qualitative and quantitative data.
The information gathered from the literature was organized through a narrative synthesis approach. Notes were first taken on each source, and then recurring themes were identified across studies. Key findings were then cross-checked through other papers to ensure consistency and reliability, allowing for the integration of accurate information into the broader discussion. This process allowed for the development of a coherent narrative highlighting areas of agreement within the field while acknowledging gaps in current research. Some pre-existing papers giving background information on this topic include “The impact of microplastics on neurodegenerative diseases and underlying molecular mechanisms: A narrative review.” This paper talks about the direct impact that nanoplastics have on neurodegenerative diseases and how they affect them. The paper “Butyl benzyl phthalate as a key component of phthalate ester about cognitive impairment in NHANES elderly individuals and experimental mice” discusses the various components in different types of nano- and microplastics, which may differently affect neuroinflammatory processes. The paper, “ROS-dependent degeneration of human neurons induced by environmentally relevant levels of micro- and nanoplastics of diverse shapes and forms. Exploring MP inhalation and brain penetration,” discusses various compositions of nanoplastics that react differently to affect ROS production, which leads to oxidative stress in the brain, leading to neuroinflammation. The paper, “Polystyrene (nano)microplastics cause size-dependent neurotoxicity, oxidative damage, and other adverse effects in C. C-elegan,” adds information on the factor of size and how it can influence neurotoxicity, oxidative damage, and other effects. The paper, “Antioxidant therapy, oxidative stress, and blood-brain barrier: The road of dietary antioxidants,” adds information on how antioxidants play into neurotoxicity and BBB health. All papers were qualitatively and quantitatively analyzed and were referenced for important connections, thematic summaries, information, data sets, and conclusions.
Results
Nanoplastic Penetration
NMPs enter the human body through various pathways. Those ways include inhalation, consumption, and contact. The most common path the nano- and microplastics take is through the nasal cavity via inhalation, and then coming in contact with the olfactory bulb of the brain . In recent studies, histological examination of the olfactory bulb revealed injury and scarring, providing a proven route of nanoplastic neurotoxicity1. Not only this, but NMPs can also surpass and penetrate the blood-brain barrier, the most selective and relevant of all the barriers that protect the brain. This penetration creates a cycle by diminishing the integrity of the blood-brain barrier, allowing more harmful substances to penetrate further. A self-amplifying cycle is created through this progressive disruption of the BBB, increasing its vulnerability by permitting continuous translocation of NMP particles as well as other neurotoxic agents12.
Neurotoxicity Posed by Nanoplastics
Vivo models show that while NMP access to neural tissue is explained through these exposure pathways, the resulting neurotoxicity is driven by several intercellular and chemical mechanisms within the brain. Nanoplastics can pose neurotoxicity by breaching the BBB in multiple ways. These methods include the translocation of the BBB through endocytosis, cells engulfing the particle, and indirect transport by acting as carriers for other neurotoxic substances. Not only this, but inflammatory responses caused by nano- and microplastics also often compromise the integrity of the BBB. Once inside the brain, nano- and microplastic particles can accumulate in the neural tissues and disrupt synaptic function, triggering neuroinflammation. These plastics also partake in intercellular uptake. This includes interfering with protein folding and mitochondrial folding, which are both necessary for neural health, thus causing neuronal damage and further neuroinflammation through accumulation. One method of inducing neurotoxicity is via oxidative stress, where these nano- and microplastics activate the brain’s microglia (or the brain’s resident immune cells), which leads to the release of inflammatory cytokines and oxidative stress. This not only induces neuroinflammation and oxidative stress, but it also damages neurons, causing declining brain health.
Additionally neural models show that, NMPs can translocate through multiple biological barriers, reaching the circulatory tissues and organs, absorbing and concentrating toxic chemicals such as heavy metals and persistent organic pollutants as they move between tissues. NMPs can also bioaccumulate and serve as Trojan Horses, meaning that they carry other substances along with them13,14. Once NMPs have crossed the BBB, they can have several effects within the nervous system: disrupting functions of brain parts such as the hippocampus, cortex, and cerebellum, interacting with several neuronal components, and disrupting synaptic function and neuronal signaling. This disruption of synaptic function and neuronal signaling caused by NMPs can contribute to cognitive decline and several behavioral changes15,16. When NMPs absorb heavy metals like aluminum, lead, and mercury (all known neurotoxins), they are all transported into the brain. These harmful metals can further contribute to oxidative stress, neuroinflammation, and the formation of amyloid-beta plaques and tau tangles, which have been implicated in neurodegenerative disorder development . Similarly, NMPs can also act as carriers for persistent organic pollutants such as pesticides and flame retardants . These substances not only promote neuroinflammation but can disrupt neuronal signaling, thereby increasing the risk of Alzheimer’s disease15.
Animal models also show that NMPs not only accumulate in parts of the brain but also have been demonstrated to accumulate in the liver, lung, testes, and gastrointestinal tract17. This causes multiple pathological processes that harm the human body, such as intestinal dysfunction, gut microbial dysbiosis, metabolic disorders, oxidative stress, and hepatic or renal toxicity18. When testing microplastic injection in invertebrates, a reduction in somatic growth rates, metamorphosis, and reproductive growth was observed19.
Collectively, these various mechanisms demonstrate that NMPs do not merely induce brief neuronal injury, but establish a pro-oxidative and pro-inflammatory environment which may potentially accelerate chronic neuronal dysfunction. Such sustained disruptions to neuroimmune signaling, BBB permeability, and synaptic integrity provide a mechanistic foundation for us to speculate on the involvement of NMPs in the onset and progression of neurodegenerative diseases.
Nanoplastics and Neurodegenerative Diseases
One main concern with the neurotoxic effects NMPs have on our body is the progressive effect they may have on neurodegenerative diseases such as Alzheimer’s, Parkinson’s, and multiple sclerosis.
Findings from non-mammalian animals models show that chemical reactions from NMPs form free radicals on the surface of cells, which can then react with atmospheric oxygen and form secondary radicals such as superoxide and alkyl radicals. These radicals damage cellular structures, DNA, and proteins, and cause inflammation. This inflammation affects the central nervous system (CNS) because of its high lipid content. As the presence of NMPs activates glial cells, they begin to release pro-inflammatory messengers such as cytokines (IL-1, IL-2, IL-17, etc), which weaken the BBB, resulting in increased permeability. The increased permeability allows T cells and macrophages to infiltrate the CNS, leading to acute inflammatory diseases and infection susceptibility. This leads to chronic inflammation in the body and eventually leads to a cycle of neuronal degeneration through the overactivation of glial cells through various signaling pathways. This chronic neuronal degeneration and inflammation are what lead researchers to hypothesize NMP exposure may play a larger role in the development of neurodegenerative diseases such as Alzheimer’s, Parkinson’s, and multiple sclerosis than was previously realized. Upon neuronal inflammation, astrocytes release inflammatory mediators such as interleukin (IL-17), which also promotes the production of other pro-inflammatory cytokines and tumor necrosis factor, as well as matrix metalloproteinases from surrounding glial cells and neurons. The increased inflammation recruits more astrocytes to the site of damage, creating a positive-feedback loop and causing chronic inflammation. This sustained response to inflammation can increase existing neuronal damage. This leads to neurodegenerative diseases as elevated levels of IL-17 have been observed in Alzheimer’s, Parkinson’s, and multiple sclerosis patients. Excessive production of anti-inflammatory cytokines also causes chronic inflammation due to the resulting dysregulation of astrocytes. Increased metabolic demands compromise astrocytes’ ability to perform their regular functions, including the mediation of glutamate. Glutamate then accumulates and disrupts neuron membrane integrity, releasing systolic lactate dehydrogenase leakage and increasing ROS levels. This leads to oxidative stress and also disrupts neuronal metabolism, contributing to cognitive decline. Postmortem studies of Alzheimer’s patients have also shown overactivated astrocytes. Another type of glial cell involved in neuroinflammation is microglia. Microglia are responsible for phagocytosing infected cells through cytotoxin release and in maintaining BBB homeostasis by producing Type 1 interferons (IFNs). These are responsible for creating an innate immune response. When microglia are overstimulated, cytotoxins that damage the neighboring healthy tissues are released and contribute to the progression of neurodegeneration seen in disorders like AD and PD progression. Hallmarks of Alzheimer’s disease are the misfolded amyloid beta plaques and abnormally phosphorylated Tau proteins. Microglia respond to the misfolded proteins, releasing cytokines and chemokines.
Previous human studies have also shown that neurodegenerative diseases such as Parkinson’s disease also progress with inflammation. PD involves the loss of dopaminergic neurons in the substantia nigra and the aggregation of Lewy bodies . Lewy bodies are abnormal alpha-synuclein protein aggregations, important in neurostriatal vesicle trafficking, and have also been found to activate microglia1. Together, these inflammatory processes driven through glial cells create a cellular environment promoting neuronal damage. Major underlying mechanisms in this damage are oxidative stress and neuroinflammation, which normally amplify neurodegenerative progression.
Oxidative Stress in Neurodegenerative Diseases
Oxidative stress and neuroinflammation are both linked to nanoplastics’ effects on progressing and causing neurodegenerative diseases. Oxidative stress occurs when the balance between reactive oxygen species (ROS) formation and detoxification favors an increase in ROS levels. Oxidative stress can cause irreversible damage in cellular macromolecules, which can lead to the initiation of various diseases. While it is a well established contributor in neurodegenerative pathways.
Cellular models demonstrate that Oxidative stress is linked to altered redox regulation of cellular signaling pathways. DNA damage and the activation of ROS to the Activator Protein (AP-1) and Nuclear Factor kappa B (NF-kB) pathways of signal transduction lead to the transcription of cell growth regulation genes and initiation of cancerous conditions. A factor contributing to neurodegeneration and oxidative stress is free radicals, which are molecules that obtain unpaired electrons in the outer orbitals. They are highly reactive in the body and oxidize other atoms, or sometimes reduce other atoms.To maintain proper cell signaling, several free radical scavenging enzymes help maintain a threshold level of ROS inside the cell to prevent damage to key components in signaling pathways20. Given the central role of oxidative stress and neuroinflammation in driving neuronal damage, strategies targeting ROS accumulation have emerged as a promising therapeutic intervention.
Antioxidants’ Role in Alleviation
One method of alleviation that is still being researched is the use of antioxidants in the mitigation of the progression of neurotoxicity and neurodegenerative diseases. Neuroinflammation and oxidative stress occur in a bidirectional cycle of impact. This means that, as neuroinflammation worsens, so does oxidative stress, and while oxidative stress worsens, so does neuroinflammation. While there is a research gap in connecting neuroinflammation to antioxidants, there is research being done on theorizing antioxidants’ use in mitigating oxidative stress.
An antioxidant is a molecule that can prevent or slow the oxidation of macromolecules. Antioxidants restrain ROS production and scavenging free radicals, protecting the body from harmful effects caused by oxidative stress and free radicals, which are components leading to the onset and progression of neurodegenerative diseases. Antioxidants have a role in lowering or terminating chain reactions causing neural degeneration by removing free radicals or inhibiting other oxidation reactions by being oxidized themselves. Meaning that by decreasing one, the other could be mitigated. To effectively mitigate neurotoxicity, the body will either need to consume antioxidants for them to penetrate the BBB or will need the body to utilize the antioxidants present internally.
Antioxidants can prevent or slow the oxidation of macromolecules. Antioxidants restrain ROS production and scavenging free radicals, protecting the body from harmful effects caused by oxidative stress and free radicals, which are components leading to the onset and progression of neurodegenerative diseases.
Antioxidants utilize a series of mechanisms to minimize neurodegeneration. They first block the production of free radicals and then scavenge for oxidants. They then convert toxic free radicals into less toxic substances and block the production of secondary toxic metabolites and mediators of inflammation. Free radicals are molecules which obtain an unpaired electron in the outer orbitals. These are highly reactive atoms, removing electrons from other atoms, therefore oxidizing them. Antioxidants act as free radical scavengers and interact with and neutralize the unstable molecules to minimize oxidation, adding to the oxidative stress. They also block the chain propagation of secondary oxidants and repair injured molecules, as well as initiate the enhancement of the endogenous antioxidant defense system .
There are three major classes of antioxidant enzymes in all body cells. These include catalases, superoxide radicals (SOD), and glutathione peroxidases (GPX). Superoxide dismutases scavenge superoxide radicals and convert them into H202. Glutathione peroxidases reduce hydrogen, lipid hydroperoxides, and other organic hydroperoxides. Glutathione-s-transferases (GST) are another class of enzymatic antioxidants that catalyze the breakdown of lipid peroxides21. Glutathione peroxidase shows a high activity with hydrogen peroxide and organic hydroperoxides, while glutathione reductase catalyzes the reduction of oxidized glutathione to reduced glutathione. An increased amount of oxidized glutathione promotes an increased oxidative stress response, so decreasing the levels by converting it to the reduced form works to lower the oxidative stress experienced. Antioxidants also have different mechanisms depending on whether they are primary or secondary . While primary antioxidants scavenge free radicals directly, secondary antioxidants quench singlet oxygen, decompose peroxides, inhibit oxidative enzymes, and regenerate primary antioxidants.
A wide range of natural and cellular antioxidants– including vitamins, flavonoids, and plant-derived phenolics– play various crucial roles in protecting the body from oxidative stress and damage caused by ROS and environmental toxins such as NMPs. Protein-bound thiol and non-protein-bound thiol act as cellular reducing agents and protective agents against most inorganic pollutants. Thiol is often the first line of defense against oxidative stress, and levels can be increased through an increase in its synthesis due to slight oxidative stress. Severe oxidative stress may decrease thiol levels due to loss of the adaptive mechanism required to respond with increased synthesis. Ascorbic acid is an antioxidant found in both plants and animals and must be obtained from the diet in humans because it cannot be synthesized. Ascorbic acid can reduce and neutralize ROS. Vitamin E has been found to remove the free radical intermediates and react with lipid radicals to protect the cell membranes from oxidation. Beta-carotene is present in liver, egg yolk, milk, butter, spinach, tomato, and grains, and has shown potent antioxidant properties by protecting against a free radical attack by removing singlet oxygen. Flavonoids are another source that plays an important role in protecting against oxidative stress20. Quercetin is a natural flavonoid compound found in common foods and surpasses the BBB and reducing the progression of degenerative disorders while protecting DNA from oxidative damage. Phenolic acids are a group of acidic antioxidants that have multiple different properties. Gallic acid is a phenolic acid that prevents mitochondrial lipid peroxidation in a cell-based model using the thiobarbituric acid RS method as the endpoint. Caffeic acid has been shown to protect against oxidative brain damage induced by different prooxidants by restoring the brain levels of the endogenous antioxidants. Ferulic acid showed a remarkable capacity in a study to protect dopaminergic neurons in the substantia nigrapars compacta and nerve terminals in the striatum. Curcumin was a promising antioxidant that showed remarkable chain-breaking abilities compared to vitamin E and can scavenge forms of ROS or activities of several endogenous antioxidants22.
The findings discussed in this paper came from a wide range of study models, each contributing its own strengths and limitations. Vitro models were able to offer high mechanistic resolution and offered clarity in understanding mechanistic pathways, such as NMP and antioxidant mechanisms in their roles in neural health. However, they were extensively specific in their descriptions, failing to give a “big picture”. They were highly reductionist in the sense that they often isolated processes while failing to describe systemic interactions and broader biological outcomes. Vivo models were useful in providing greater biological relevance by describing specifics on tissue-specific models, barrier functions, and biological interactions. Vivo models utilizing invertebrates were useful for isolating specific mechanisms to study while still studying their pathological and biological characteristics. Human studies, which are highly observational, were extremely useful in giving dependable cause-and-effect characteristics of variables. They offered the highest translational relevance but were still constrained by confounding variables and did not cover much in the toxicology field due to ethical constraints.
Discussion
NMPs are an emerging issue in the modern world and could pose major risks for neural and overall health. NMPs are being overly consumed by humans (either by consumption, inhalation, or contact) due to their heavy pollution and have been found to penetrate the BBB. By doing so, NMPs could pose high levels of neurotoxicity to the brain. NMPs can induce neurotoxicity by breaching the BBB through endocytosis, enabling them to act as carriers for neurotoxic substances, trigger microglial activation and cytokine release via inflammatory pathways, disrupt protein folding and mitochondrial function, and impairing synaptic signaling as they accumulate in neural tissues. This induces neuroinflammation and oxidative stress, which both have a bidirectional relationship in worsening the other. These two factors are hallmarks of neurodegenerative diseases such as Alzheimer’s, Parkinson’s, and multiple sclerosis. These findings have led researchers to theorize that NMPs can worsen neurodegenerative diseases and even cause them. One emerging field of research is the use of antioxidants to alleviate neurotoxicity, leading researchers to wonder if they could mitigate NMP-induced neural health risks. Antioxidants could alleviate neurotoxicity because they neutralize reactive oxygen species, scavenge free radicals, block the formation of secondary toxic metabolites, and support cellular repair mechanisms, ultimately reducing oxidative stress, inflammation, and neuronal damage. They do this through a combination of enzymatic and non-enzymatic processes, such as converting superoxide radicals into less harmful molecules, protecting mitochondria and DNA, enhancing the body’s endogenous antioxidant systems, and even directly crossing the BBB to protect neural tissues from degeneration. This shows potential for antioxidants to alleviate the harm that NMPs pose to neurodegenerative diseases. With further research, a direct relationship nanoplastics have on the progression and causation of neurodegenerative diseases may be able to be confirmed.This study successfully examined various methods by which NMPs may induce neurotoxicity by crossing the BBB and other barriers and how this could lead us to presume they have a link to neurodegenerative diseases23. Existing research shows the neurotoxic effects that NMPs have on the neural system is highly dependent on the size of the plastic as well as the amount 9. The smaller the plastic, the more damage it causes as it crosses more protective barriers23. This paper also examined mechanisms by which antioxidants alleviate neurotoxicity, presenting them as a prospective tool for mitigating NMP-induced neurotoxicity. The studies cited in this review characterize the mechanisms by which NMPs act inside the brain and also review neurotoxic mechanisms in the brain. Many of these studies focus on a single specific mechanism rather than multiple mechanisms in general. Some particular focuses include BBB penetration, neuroinflammation, and oxidative stress pathways. This review acts as an aggregator of the various mechanisms NMPs may use to induce neurotoxicity. This review also integrates research on antioxidant alleviation for neurotoxicity rather than seeing oxidative stress and neuroinflammation as an endpoint. It then later evaluates whether antioxidants can be a probable competitor to be an alleviant of NMP-induced neurotoxicity in specific cases.
This review also contains inherent limitations. Due to the heterogeneity of experimental models, plastic types, and outcome measures used across the studies, precluding meta-analysis and quantitative comparison, this study is constrained as a narrative synthesis. Furthermore, as direct studies examining antioxidants’ direct relation to NMPs are scarce, conclusions regarding antioxidant efficacy must be drawn from a larger body of literature written in the more general sense of neuroinflammation and oxidative stress.
Future studies should prioritize methods to better approximate human conditions as well as physiologically relevant exposure models. Using in vitro systems, studies could better predict NMP-BBB interactions with more accurate models, allowing for more precise assessments of mechanisms, chemical interactions, and oxidative damage. In vivo studies could incorporate realistic exposure routes (such as inhalation and dietary intake), which may be key to understanding the various pathways and mechanisms used by NMPs to enter circulation and neural tissues. Studies should also directly investigate the relationship between antioxidants and NMPs in terms of whether or how the antioxidants could actually prevent NMPs from inducing toxicity as they do with ROS, or if antioxidants only target the resulting damage after NMP exposure. By examining various types of antioxidants (dietary, endogenous, etc), the exact mechanisms and characteristics of antioxidants can be pinpointed and studied to understand how their specific molecular mechanisms may be best used to counteract NMP-induced neurotoxicity.
Ultimately, antioxidants could serve as a critical defence against oxidative stress, underscoring their significance not only in maintaining cellular health but also in preventing long-term disease progression, which may be caused by NMP pollution. As we continue to explore their roles, we move closer to understanding how to protect human health in a world faced with numerous health challenges like NMP pollution.
Conclusions
In conclusion, NMPs could pose a significant threat to neural health as they breach biological barriers, accumulate in brain tissue, and trigger oxidative stress, neuroinflammation, and cellular dysfunction. Their ability to act as Trojan Horses for heavy metals and persistent organic pollutants further amplifies their neurotoxic potential, contributing to the notion that they have a link to neurodegenerative diseases1. However, the body’s antioxidant defense system plays a critical protective role in combating neurotoxicity, showing that they could mitigate neural damage posed by NMPs. Enzymatic antioxidants like SOD, catalase, and GPX, along with natural compounds such as flavonoids and phenolic acids, demonstrate powerful neuroprotective effects by neutralizing reactive oxygen species and supporting cellular repair mechanisms20. Understanding the dual role of environmental toxins and cellular defense systems not only highlights the urgency of addressing plastic pollution but also opens doors for therapeutic strategies that harness antioxidants to preserve brain health and mitigate long-term brain damage. This research advances the neurotoxicology field by addressing the possible emerging threats of NMP pollution to human health. This research investigates how NMPs affect oxidative stress, neuroinflammation, and protein interactions. By doing so, it clarifies the mechanisms through which NMPs could harm brain health and explores avenues to mitigate this toxicity. The findings contribute to our understanding of how NMP pollution impacts the brain and could help develop strategies to alleviate its effects. Additionally, the study explores how antioxidants may reduce neurodegeneration, showing them as a possible therapeutic for NMP-induced brain damage.
Acknowledgements
I would like to thank Makaila Furderer for guiding and mentoring me throughout the process of writing this paper. I would also like to thank Lumiere for the opportunity to participate in the program, along with Maheem Jawani, my program manager, for all her support.
References
- Jeong, CB., Kang, HM., Lee, MC. et al. Adverse effects of microplastics and oxidative stress-induced MAPK/Nrf2 pathway-mediated defense mechanisms in the marine copepod Paracyclopina nana. Sci Rep 7, 41323 (2017). https://doi.org/10.1038/srep41323. [↩] [↩] [↩] [↩]
- Mercè Encinas, J. Martí Clúa. Effects of microplastics and nanoplastics exposure on neurogenesis: Are thymidine analogs a good option to study such effects? International Journal of Molecular Sciences. Vol. 26, 7845, 2025, https://doi.org/10.3390/ijms26167845. [↩]
- X. Cao, W. Xie, M. Feng, J. Chen, J. Zhang, J. Luo, Y. Wang. Nanoplastic exposure mediates neurodevelopmental toxicity by activating the oxidative stress response in zebrafish (Danio rerio). ACS Omega. Vol. 9, pg. 16508–16518, 2024, https://doi.org/10.1021/acsomega.4c00231. [↩]
- F. Huang, H. You, X. Tang, Y. Su, H. Peng, H. Li, Z. Wei, J. Hua. Early‑life exposure to polypropylene nanoplastics induces neurodevelopmental toxicity in mice and human iPSC‑derived cerebral organoids. Journal of Nanobiotechnology. Vol. 23, pg. 474, 2025, https://doi.org/10.1186/s12951-025-03561-1. [↩]
- O. Baş, M. Acar, S. Çelik, B. Aydin. To what extent are orally ingested nanoplastics toxic to the hippocampus in young adult rats? Journal of Chemical Neuroanatomy. Vol. 132, pg. 102314, 2023, https://doi.org/10.1016/j.jchemneu.2023.102314. [↩]
- S. Shan, Y. Zhang, H. Zhao, T. Zeng, X. Zhao. Polystyrene nanoplastics penetrate across the blood-brain barrier and induce activation of microglia in the brain of mice. Chemosphere. Vol. 298, pg. 134261, 2022, https://doi.org/10.1016/j.chemosphere.2022.134261. [↩]
- C. Liu, Y. Zhao, W. Zhang, J. Dao, Q. Li, J. Huang, Z. Li, Y. Ma, C. Qiao, C. Cui, S. Chen, L. Yu, Y. Shen, W. Zhao. Targeted activation of ErbB4 receptor ameliorates neuronal deficits and neuroinflammation in a food‑borne polystyrene microplastic-exposed mouse model. Journal of Neuroinflammation. Vol. 22, pg. 86, 2025, https://doi.org/10.1186/s12974-025-03406-6. [↩]
- C. Lee, L. Hsu, I. Wu, Y. Wang, W. Chen, Y. Liu, L. Yang, C. Tan, Y. Luo, C. Wang, H. Chiu, T. Yang, Y. Lin, H. Chang, Y. Chiang, C. Chen, M. Lee, K. Peng, C. Huang. Exposure to polystyrene microplastics impairs hippocampus-dependent learning and memory in mice. Journal of Hazardous Materials. Vol. 430, pg. 128431, 2022, https://doi.org/10.1016/j.jhazmat.2022.128431. [↩]
- C. Wu, H.-J. Zhang, H. Ma, R. Ji, K. Pan, T. Yue, A.-J. Miao. Mechanisms underlying the size-dependent neurotoxicity of polystyrene nanoplastics in zebrafish. Environmental Science & Technology. Vol. 59, 1577–1586, 2025, https://doi.org/10.1021/acs.est.4c12148. [↩] [↩]
- N. R. Rahimi, M. Dehghani, R. Fouladi-Fard, Impact of Micro and Nanoplastics on Inflammatory and Antioxidant Gene Expression in the Gastrointestinal System. J. Environ. Health Sustain. Dev. (2025). [↩]
- H. Yu, Z. Zhao, H. Li, Y. Han, H. Li, C. Cui, Y. Hu, B. Zhang. Nanoplastics exposure exacerbates Aβ plaque deposition in Alzheimer’s disease mice by inducing microglia pyroptosis. Ecotoxicology and Environmental Safety. Vol. 299, pg. 118379, 2025, https://doi.org/10.1016/j.ecoenv.2025.118379. [↩]
- L. Lei, M. Liu, Y. Song, S. Lu, J. Hu, C. Cao, B. Xie, H. Shi, D. He, Polystyrene (nano)microplastics cause size-dependent neurotoxicity, oxidative damage, and other adverse effects in Caenorhabditis elegans. Environ. Sci.: Nano 5(8), 2009–2020 (2018). https://doi.org/10.1039/c8en00412a . [↩] [↩]
- X. Lu, Q. Luo, J. Zhao, M. Li, D. Liu. Revealing the underlying mechanisms of nanoplastics induces neuroinflammation: from transcriptomic analysis to in vivo and in vitro validation. Ecotoxicology and Environmental Safety. Vol. 298, pg. 118311, 2025, https://doi.org/10.1016/j.ecoenv.2025.118311. [↩]
- I.V. Kirstein, S. Kirmizi, A. Wichels, A. Garin-Fernandez, R. Erler, M. Löder, G. Gerdts. Dangerous hitchhikers? Evidence for potentially pathogenic Vibrio spp. on microplastic particles. Marine Environmental Research. 2016;120:1–8. doi:10.1016/j.marenvres.2016.07.004. [↩]
- Li, G., Liu, X., Sun, X., Huang, L., Kuang, W., Ou, J., Zhang, J., Zhang, Z., Li, H., Tang, H., Feng, C., Gu, L., Yang, C., Peili, W., & Wang, J. (2024). Polystyrene microplastics induce anxiety via HRAS derived PERK-NF-κB pathway. Environment international, 185, 108543. https://doi.org/10.1016/j.envint.2024.108543. [↩] [↩]
- Y. Paing, Y. Eom, G. Song, B. Kim, M. Choi, S. Hong, S. Lee. Neurotoxic effects of polystyrene nanoplastics on memory and microglial activation: Insights from in vivo and in vitro studies. Science of The Total Environment. 2024;924:171681. doi:10.1016/j.scitotenv.2024.171681. [↩]
- S. Qiang, Y. Huang, Y. Wang, L. Jiao, Y. Luo, S. Li, H. Gu, Z. Wang, Y. Zhang, X. Wu, Q. Fan. Polystyrene nanoplastics induce mitochondrial dysfunction and hepatic fibrosis via oxidative stress. SSRN Electronic Journal, Posted 14 Aug 2025, https://ssrn.com/abstract=5391979; http://dx.doi.org/10.2139/ssrn.5391979. [↩]
- Y. Jin, L. Lu, W. Tu, T. Luo, Z. Fu. Impacts of polystyrene microplastic on the gut barrier, microbiota and metabolism of mice. Science of the Total Environment. Vol. 649, pg. 308–317, 2019, https://doi.org/10.1016/j.scitotenv.2018.08.353. [↩]
- S. Y. Au, T. F. Bruce, W. C. Bridges, S. J. Klaine. Responses of Hyalella azteca to acute and chronic microplastic exposures. Environmental Toxicology and Chemistry. Vol. 34, pg. 2564–2572, 2015, doi:10.1002/etc.3093. [↩]
- Kopatz, V., Wen, K., Kovács, T., Keimowitz, A. S., Pichler, V., Widder, J., Vethaak, A. D., Hollóczki, O., & Kenner, L. (2023). Micro- and Nanoplastics Breach the Blood-Brain Barrier (BBB): Biomolecular Corona’s Role Revealed. Nanomaterials (Basel, Switzerland), 13(8), 1404. https://doi.org/10.3390/nano13081404. [↩] [↩] [↩]
- A. A. Adwas, A. S. I. Elsayed, A. E. Azab, et al., Oxidative stress and antioxidant mechanisms in the human body. J. Appl. Biotechnol. Bioeng. 6(1), 43–47 (2019). doi:10.15406/jabb.2019.06.00173. [↩]
- C. Dende, J. Meena, P. Nagarajan, V. Arun Nagaraj, A. K. Panda, G. Padmanaban. Nanocurcumin is superior to native curcumin in preventing degenerative changes in experimental cerebral malaria. Scientific Reports. Vol. 7, pg. 10062, 2017, doi:10.1038/s41598-017-10672-9. [↩]
- J. Domenech, A. Hernández, L. Rubio, R. Marcos, C. Cortés, Interactions of polystyrene nanoplastics with in vitro models of the human intestinal barrier. Arch. Toxicol. 94(9), 2997–3012 (2020). https://doi.org/10.1007/s00204-020-02805-3. [↩] [↩]