Abstract
Oral cavity squamous cell carcinoma (OCSCC) is a form of oral cancer with prevalence rates that are rapidly increasing, representing approximately 54,000 annual cases in the US. Current treatment includes chemotherapy, radiotherapy, and surgery, techniques that are invasive procedures with limited efficacy. This review examines recent advancements in OCSCC therapy, comparing conventional treatments with emerging approaches such as monoclonal antibodies, cell-based therapy, tumor-treating fields TTFields, and nanotechnology. Relevant studies were identified through a systematic literature search of PubMed and Google Scholar. Reported survival data suggest that patients receiving immunotherapy reported survival outcomes in immunotherapy studies are in fact encouraging, although differences in study design and follow-up limit direct comparison with conventional therapies. Surgical effectiveness also varies by margin status, with higher mortality observed in patients with positive margins. This review will address current treatments for oral cancer as well as their limitations in terms of survival and recurrence rates, then evaluate novel therapies and their role in OCSCC. By exploring these developments, this paper shows the potential in these new therapies to overcome the limitations of traditional treatment. Although evidence for novel therapies in OCSCC remains limited, early results suggest immunotherapy and nanotechnology-based strategies having the potential to improve survival outcomes and reduce recurrence. Continued research, larger clinical trials, and development of predictive biomarkers will be essential to validate their role in advancing the standard of care for OCSCC.
Keywords: Oral Cavity Squamous Cell Carcinoma (OCSCC), Immunotherapy, Overall Survival Rate, Treatment
Introduction
Oral cavity squamous cell carcinoma (OCSCC) arises in the oral cavity from squamous cells. Squamous cells are flat, scale-like cells that appear under the surface of skin, making them highly vulnerable to external factors. They are found in the floor of the mouth, hard palate, soft palate, tonsil (important for immune defense)1, lip, alveolar, buccal, and gum2. The floor of the mouth consists of mylohyoid muscles which elevate the hyoid-laryngeal complex when swallowing3. The roof is formed by the hard palate and posteriorly by the soft palate, which serve vital functions of digestion, speech articulation, and respiration3. The alveolar, gum and buccal work to anchor and protect the roots of the teeth.
In the US, oral cancer represents almost 3% of cancers with approximately 54,000 annual cases4. Mainly, oral cancer affects adults over the age of 40, with age groups over 70 being the most at risk5. In a study consisting of 603 patients diagnosed with oral cancer, 97.7% were older than 40 years6. Despite this, there has been a rise in oral cancer cases in younger demographics7, which can potentially be due to certain carcinogens such as environmental factors such as tobacco8, UV radiation9, and HPV10.
Historically, African American males are at a higher risk of death to oral cancer compared to white American males, yet white American males having higher overall incidence rates11. Recent studies have illustrated further shifts in racial disparity trends, with total incidence rates substantially declining in African American males and total incidence rates increasing for White males12. Additionally, there are no statistically significant differences in mortality for Hispanics vs whites or Asians vs whites13. To explore the racial disparities within cancer development, further studies should examine a wide range of socio-demographic variables in order to determine factors in racial and gender oral cancer incidence.
Meanwhile, there are prevalent gender disparities, with men holding a significantly higher incidence rate than women14. Still, oral cancer remains one of the most common tumors, with incidence rates expected to rise 65% in 2025 as estimated by GLOBOCAN15.
Current treatment for oral cancer patients such as chemotherapy, radiotherapy, and surgical therapy is considered invasive and often ineffective. Recurrence rates are at about 20% in oral cancer patients16 highlighting the need for more effective treatment to reduce recurrence rates.
A paper published by Douglas Hanahan and Robert A. Weinberg explores the hallmarks of cancers, to which there are six – these hallmarks represent the defining characteristics of cancer, specifically how malignant cells can grow at such a rapid pace. Cancer cells can sustain proliferative signaling without growth signals, evade growth suppressors, evade apoptosis, enable replicative immortality, induce angiogenesis, and activate invasion and metastasis17. This is applicable to the development of cancer treatment as it provides a framework in what treatments need to address. These hallmarks are the characteristics that distinguish cancer cells from healthy cells, allowing researchers to advance personalized therapies.
This review aims to evaluate current standard-of-care treatments for oral cavity squamous cell carcinoma and critically examine emerging therapeutic approaches, including immunotherapy, tumor-treating fields, and nanotechnology-based strategies. Despite advances in oncology, survival outcomes for OCSCC remain poor, and many novel therapies lack clear clinical integration. A key gap in the literature is the limited synthesis of how these emerging treatments compare conceptually to conventional modalities, particularly in terms of invasiveness, toxicity, and potential impact on recurrence. By consolidating existing evidence and highlighting translational limitations, this review seeks to clarify the potential clinical role of emerging therapies and identify priorities for future research.
Developments of Oral Cancer
The incidence of cancer is influenced by multiple factors that contribute to disease development. Factors such as smoking, sex differences, and demographic factors increase the risk of developing cancer18.
Cancer develops when a change, or mutation, occurs in a person’s DNA. This change or mutation can deregulate the cell cycle, causing uncontrolled cell division, leading to the formation of a tumor. In mitosis, there are key cell checkpoints that regulate the cell cycle to ensure safe cell division. TP53, which encodes the p53 protein, is a significant tumor suppressor gene that aids DNA repair and apoptosis. When the gene TP53 is mutated, p53 can no longer perform these functions, thereby damaged cells will continue dividing, leading to cancer growth19.
Oral cancer develops through mutations in tumor suppressor genes such as TP53, which drive genomic damage and malignant transformation. TP53 is the most common mutation in oral cancer patients, occurring in 60% of patient samples20. Furthermore, upregulation of immune checkpoint proteins p53, CK17, and PD-L1 further facilitate immune evasion within the tumor microenvironment21.
Studies have shown tobacco to be a key cause in the overexpression of mutated p53 protein in the oral cavity, which is linked to the presence of oral cavity squamous cell carcinoma (OCSCC), in which overexpression leads to dysfunction of vital cell regulators8. The damage to the DNA ultimately results in uncontrolled cell growth in the oral cavity, leading to tumor development. Exposure of the lip to the sun also increases the risk of oral cancer4. The mouth has higher susceptibility to cancer when exposed to UV radiation as opposed to bodily skin. This hypothesis was further explored in a study showing a decreased rate of repair and apoptosis in oral cells to skin cells when exposed to the same UV conditions9.
About 50% of those diagnosed with oral cancer will only survive more than five years22. The reason this cancer had such high mortality rates is its ability to metastasize and high rates of tumor infiltration. Over time, the cancer cell will break away from the original tumor and spread to distant tissues and other parts of the body. In distinct steps, this new tumor cell will travel to surrounding areas and establish new, secondary tumors. The epithelial–mesenchymal transition plays a key role in metastasis. During this pathway, transcriptional regulators aid the cell in invasion, dissociation, and cell migration23. As a result of high rates of metastasis, a study of 216 patients have shown recurrence to affect 89% of patients in the 5th year of follow up16.
Stages
Cancer is developed through several stages which determine the size and pervasiveness of the cancer. Identifying the stage of cancer is vital to the type of treatment a patient requires and how severe the cancer may be. The first stage is the least severe and the easiest to treat. This is because the cancer is usually small and localized, meaning it’s contained to only the organ it first developed in. In stage two, the cancer is still localized however larger and more developed. Depending on the type of cancer, it’s possible for the cancer to have spread to nearby lymph nodes. Stage three indicates that the cancer has spread outside of the original organ and into the surrounding tissues. The cancer has not spread to other distant parts of the body yet may consist of multiple tumors. Stage three cancer has multiple subcategories, that are determined by the tumor’s size, relative location, and the number of tumors. Stage four of cancer, also known as metastatic cancer, is the last and most advanced stage of cancer. This occurs when the cancer is spread to other distant parts of the body. By this point, the survival rate has significantly decreased, and doctors can only attempt to manage the symptoms and control the cancer rather than cure the patient24.
Early diagnosis is of the utmost importance for cancer patients, as when cancer develops and spreads in a patient, it becomes harder to treat. Specifically, oral cancer is harder to detect and diagnose, with 50% of OCSCCs being diagnosed at an advanced tumor stage25.
Tumor Microenvironment
The tumor microenvironment (TME) plays a crucial role in oral cancer development by stimulating cancer cells. The TME consists of surrounding immune cells, signaling molecules, fibroblasts, bone marrow-derived inflammatory cells, the extracellular matrix, and blood vessels26. With rapidly evolving research, scientists have discovered the significant role the TME plays with the goal of advancing treatment to become more effective. Hypoxia is closely linked to tumorigenesis and a significant contributor in the TME of oral cavity. Hypoxic conditions are caused by significantly dropped oxygen levels, due to the high concentration of cancer cells27. Once these oxygen levels fall, the tissue site is more prone to increased tumor aggression, invasion, enhanced metastatic potential, resistance to therapy (chemo and radio), and overall reduced survival rates28. Hypoxic conditions in the tumor tissue recruits and activates immunosuppressive cells – myeloid-derived suppressor cells29 and tumor-associated macrophages30 – which suppress immune responses (immune suppression)31. Additionally, hypoxic conditions lead to the activation of hypoxia-inducible factors (HIF), specifically HIF-1ɑ, which positively upregulates PD-L1 levels, constituting as a mechanism for tumor cell immune escape, ultimately supporting the development of tumor survival and proliferation32. Specifically, in the oral cavity, the hypoxia-inducible factor will cause the direct upregulation of the antigenic factors such as vascular endothelial growth factor (VEGF). In fact, VEGF mRNA expression levels increased by 10-50 times in reduced oxygen level environments33. VEGF will bind to vascular endothelial growth factor receptors, further advancing tumor angiogenesis by activating downstream signaling pathways and changing vascular permeability34.
The tumor microenvironment in the oral cavity includes immunosuppressive cells, tumor-associated macrophages, regulator T cells, and cancer-associated fibroblasts, which promote tumor growth through resistance to immune treatment and proliferating cell cycles. Once these cancer cells establish, they secrete exosomes to drive immunosuppressive macrophages, and in turn drive cell proliferation through metabolic reprogramming35. In fact, exosomes are highly abundant in human saliva36, supporting the theory of oral cavity being especially prone to tumor growth. These exosomes contain vital micro RNAs such as miR-24-3p, miR-512-3p, miR-412-3p, and miR-31 that are associated with patients diagnosed with OCSCC37. Involved with gene expression, miRNAs are non-coding RNAs that bind to mRNAs and block translation. Depending on location and abundance, miRNAs may act as signaling molecules to mediate cell-cell communication40. This upregulated expression of immunosuppressive cells in the tumor microenvironment promotes cell proliferation and tube formation on tumors, and adding additional barriers to non-specific cancer therapy.
The tumor microenvironment plays a critical role in cancer progression, and the inability of current treatments to effectively target it contributes to high recurrence rates and poor survival outcomes. However, novel therapies such as immune checkpoint inhibitors are able to modify the TME, altering its physical state to promote immune infiltration38. Future research should continue to explore how cancer therapies could bypass the physical barrier that is the tumor microenvironment, whilst working to shrink/eliminate tumor cells.
Methods
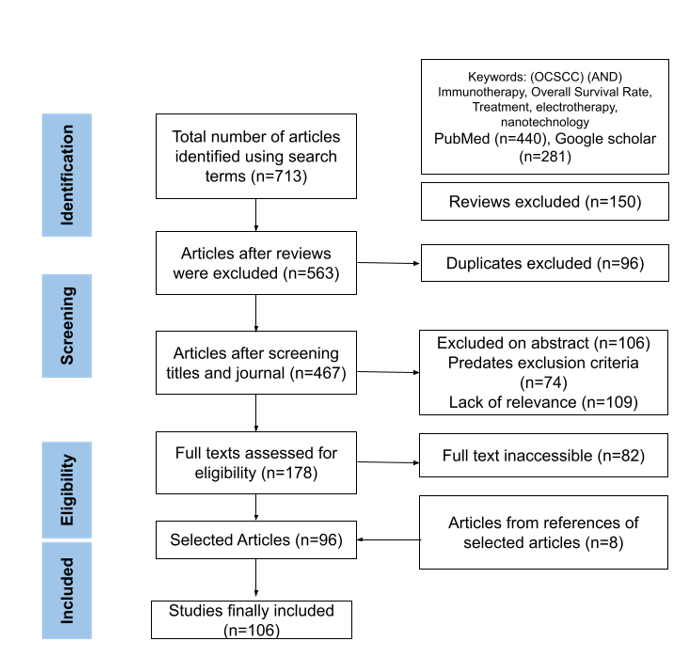
This review was conducted as a systematic literature search to explore recent advancements in oral cancer treatments, including T-cell therapy, tumor-infiltrating lymphocytes (TILs), immune checkpoint inhibitors, proton radiation, and tumor-treating fields (TTFields). The selection process was carried out using PubMed and Google Scholar from dates no earlier than 2000. Only peer-reviewed original research articles and systematic reviews published in English were considered while non peer reviewed articles with limited generalizability were excluded.
Survival data was extracted from published clinical studies of chemotherapy, radiotherapy, and immunotherapy in OCSCC or head and neck cancers. Reported survival percentages at defined follow-up points were compiled and presented in a table (Table 1). Data extraction was targeted for results of clinical trials such as overall survival rates and recurrence rates. The screening process included only papers containing clinically accurate and relevant information, while duplicate articles retrieved from independent searches were removed.
The following keywords and Boolean combinations were applied: “oral cancer” AND (“T-cell therapy” OR “tumor-infiltrating lymphocytes” OR “immune checkpoint inhibitors” OR “proton therapy” OR “tumor-treating fields”). Only peer-reviewed publications written in English were considered.
Treatment Options
Cancer treatment is constantly developing as researchers are finding new ways to apply advanced technology to therapy. A recent study has demonstrated 89% of oral cancer patients experiencing disease recurrence16. The need for developing effective treatment for oral cancer is urgent, so this section explores new technologies that aim to improve outcomes.
Current Treatment
This section explores the common and current treatment directed towards cancer patients. It will discuss chemotherapy, the most common cancer therapy, radiotherapy, and then surgical treatment. Addressing the challenges will encourage a new outlook on cancer treatment, searching for less invasive, yet more effective procedures.
Cancer treatment is rapidly evolving, constantly becoming more advanced to successfully treat patients. Specifically, scientists are researching more targeted therapies that would be more effective in treating cancer. Current treatment, surgical, chemotherapy, and radiotherapy, have the inability to target the tumor microenvironment (TME), which plays crucial roles in tumorigenesis. The TME contains cancer associated fibroblasts and stromal tissues; key components that progress cancer development in patients39. To address this, scientists have been developing immunotherapy techniques including small molecule inhibitors and monoclonal antibodies. Not only does this type of therapy offer higher survival rates, but due to its personalization, patients experience less side effects than they would in generalized treatment.
Chemotherapy
Chemotherapy is the most common oral cancer therapy, with overall usage increasing by 54.7% from 2008-202040. Chemotherapy works by targeting rapidly dividing cells, effectively killing cancer cells but also damaging other rapidly dividing cells in the body, including immune cells. Immunosuppression has become a serious side effect, increasing a patient’s risk of infection41. Depending on the stage of cancer, chemotherapy is often utilized to either treat the patient’s cancer, prevent recurrence, or relieve symptoms. Doctors will prescribe treatment, which could be in the form of pills, injections, or intravenous therapy. The purpose of chemotherapy is to stop the growth of these cancer cells by disrupting their cell cycle. However, current chemotherapy leaves patients vulnerable to a multitude of risks, one of them being organ dysfunction. A patient’s organs such as their liver or lungs react negatively to many chemotherapy drugs.
Chemotherapy agents are categorized by their mechanism of action in which they target different phases of cancer development. Antimetabolites, a type of immune suppressant, reduces the growth of cancer cells by interfering with cell division42. Common antimetabolites used to treat carcinoma include Methotrexate, 5-Fluorouracil43, and Capecitabine44. Platinum-based agents target and damage cancer cell DNA during replication, with agents including Cisplatin45 and Carboplatin46. Finally, plant alkaloids are naturally occurring compounds that interfere with microtubules and cell division. Paclitaxel and Docetaxel are common plant alkaloid agents that are used in treatment for head and neck cancers47.
Radiotherapy
Radiation therapy is another highly developed cancer treatment, using high energy beams to target and kill cancer cells. These beams are designed to destroy the cancer’s genetic material and prevent it from growing. Patients are placed under a linear accelerator and despite its precision, it is possible for healthy cells to be damaged under the radiation. However, non-cancerous cells can more effectively repair themselves. Radiation therapy may place patients under the risk of side effects due to damaged surrounding cells, including neurologic symptoms48. In a review paper exploring the relationship between cancer stem cells (CSCs) and tumor relapse, researchers have discovered a positive correlation in how CSCs respond to radiotherapy. CSCs in the oral cavity are significantly more likely to survive radiotherapy than that of non-stem cells. In fact, radiotherapy activates cell proliferation in said CSCs, heavily contributing to tumor relapse and metastasis. Post-radiotherapy, these “awakened” CSCs have radiation tolerance, DNA repair ability, and self-renewal properties49. Therefore, OCSCC patients treated with radiotherapy have possibly worse survival outcomes because of the response of CSC’s to radiotherapy. Further research is required to fully comprehend this phenomenon and prepare strategies of defense against this.
Interstitial Radiotherapy (Brachytherapy) is a traditional approach to OCSCC patients, and an alternative to external beam radiotherapy (EBRT)50.
Brachytherapy administers radiotherapy to patients through the placement of radioactive sources adjacent to the tumor. It’s highly used in the oral cavity as it’s able to precisely deliver a concentrated dose to the tumor area51. In some cases of treating patients with oral cancer, post operative brachytherapy is applied as an adjuvant to radiotherapy, for cases unable to tolerate surgery, or to address recurrent cases50. EBRT administers radiotherapy from outside the body, positioned and shaped to target regions of tumor and lymph nodes52.
Due to EBRT’s lack of precision, recurrence rates and toxicity become associated problems, which is why researchers emphasize the use of intensity-modulated radiotherapy (IMRT). IMRT allows for concave dose distributions and shows substantial reduction of osteoradionecrosis in post-radiotreated patients53,54.
Surgical therapy
Surgical therapy is most effective with a solid tumor, if the cancer is localized. Often, chemotherapy is administered before surgical procedures to cause shrinkage of the tumor. During this procedure, surgeons will make a large cut inside the oral cavity, or smaller cuts and use a laparoscope. Either way, the purpose is to remove the tumor inside the body, as well as any surrounding tissue and lymph nodes55. However, there are many known risks associated with surgical therapies. Depending on the location of the cancer, doctors can have difficulty resecting the cancer. During stages 3 and 4 of cancer, the tumor is spread beyond the location of origin, complicating surgical decision making regarding accurate resection margins, leading to a high risk of ineffective removal and therefore higher recurrence rates.
Within resecting the primary oral tumor, doctors aim to remove an adequate margin of healthy tissue while still preserving functions of the oral cavity. Peroral resection removes the tumor without external incisions and is most used for small tumors that occur on the tongue, floor of mouth, or buccal mucosa56. For larger, deeper tumors, doctors perform external incisions to expose deeper parts of the oral cavity, such as the lower cheek, visor flap, mandibulotomy, and upper cheek flap56.
Immunotherapy
Cancer treatment research is being rapidly developed as scientists make breakthroughs in treatment and diagnosis. Many new promising therapies, such as targeted therapies, have great potential to minimize side effects, and reduce the invasiveness of cancer treatment.
Immunotherapy is a rapidly developing, novel therapy that has shown immense potential in improving cancer treatments in clinical trials. Immunotherapy utilizes the body’s own immune system in containing tumorigenesis and tumor progression57. Researchers are exploring the connection between tumor-infiltration lymphocytes in cancer tissues to further enhance one’s defensive mechanism against cancer. This section will cover advancements in monotherapies such as tumor-infiltration B-lymphocytes, immune checkpoint inhibitors, and cell-based therapy, and their role in treating OCSCC patients.
Importantly, different types of immunotherapy have different types of toxicity that is induced by the same immunologic mechanisms responsible for the treatment’s therapeutic effects58. Clinical trials of patients with breast cancer have revealed immune checkpoint inhibitors to directly increase pathological response rate as they are of immune-based origin59. Immunotherapy toxicity most commonly involves the skin, endocrine glands, gastrointestinal system, and liver, along with most organ systems58. These toxicities require highly specific treatment management due to prolonged duration and fatal, yet rare, severity59.
Acquired resistance to immunotherapy is an additional clinical challenge that limits cancer remission. During the tumor immune cycle, tumor cells and the TME are modified following immune cell interaction, developing new characteristics that later prevent the tumor from effectively responding to immunotherapy60. Patients may experience temporary, partial, or even complete clinical response after immunotherapy treatment, however will ultimately experience clinical progression of the disease61.
Monotherapy
Tumor-infiltration lymphocytes (TILs) are immune cells that enter tumors and act as a natural defense against cancer. These cells recognize tumor antigens and can activate both T and B cell memory responses, helping the body identify and interact with, to destroy cancerous cells62.
TIL therapy is a type of adoptive cell therapy that builds on this natural immune response. After the resection of a tumor, surgeons extract the TILs from the tumor lesion where these immune cells are grown in a lab setting. They are expanded ex vivo then reinfused into the patient’s immune system63,64.
This approach has shown promising results in clinical trials, particularly in metastatic melanoma, and researchers are now exploring its potential use in other cancers, including oral cavity squamous cell carcinoma (OCSCC)65.
In the OCSCC, there are significant levels of tumor-infiltrating B-lymphocytes (TIL-Bs) that contain high expression of CD45. In a study exploring the prospective of TIL-Bs acting as biomarkers, researchers took samples of 222 OCSCC patients and classified each as TIL-rich or TIL poor. Individuals who were classified as TIL-rich were diagnosed at earlier stages and had improved survival66. Thus, these TIL-Bs cells help with early diagnosis, but they also correlate with less aggressive tumor development. Similarly, a retrospective analysis of 278 patients showed that increased CD4+ and CD8+ TIL levels correlated with reduced recurrence and higher survival rates. These findings suggest that TILs could serve not only as potential therapeutic agents, but also as valuable prognostic biomarkers for OCSCC67.
Immune checkpoint inhibitors
Immune checkpoint inhibition is a novel, yet extensively studied area of immunotherapy, representing a major advancement in cancer treatment. They work by targeting specific proteins that normally act as brakes on the immune system, allowing cancer cells to avoid detection and destruction. Two key checkpoint proteins are CTLA-4 and PD-168, which are mainly found on T cells, but are also expressed by other immune cells, such as B lymphocytes and fibroblasts66,69. Under normal conditions, these checkpoints help maintain self-tolerance and prevent the immune system from attacking healthy tissue. However, many cancers, including oral cavity squamous cell carcinoma (OCSCC), upregulate checkpoint proteins to suppress immune responses70. Exploring immune checkpoint inhibitors in oral cancer is of importance as it addresses the limitations of current treatment.
Checkpoint inhibitor drugs block PD-1 or PD-L1, restoring the ability of T cells to recognize and kill tumor cells by promoting apoptosis71. This approach has shown strong results in several cancers and is now being explored in head and neck malignancies, including OCSCC. Currently, the Food and Drug Administration has approved pembrolizumab, a PD-1 inhibitor as treatment for head and neck cancers72. For cancers generally, the US FDA has approved three different categories of immune checkpoint inhibitors. PD-1 inhibitors, PDL-1 inhibitors, and CTLA-4 inhibitor73. Especially, hypoxic conditions, PD-L1 is upregulated due to overexpression of HIF-α32. For immune checkpoint inhibitors, patients would only benefit if they express PD-L1 in their tumor as these therapies work by blocking PD-L1 activity that suppresses T-cell activity.
By reactivating the immune response, checkpoint inhibitors offer a less invasive and more targeted alternative to chemotherapy and surgery, potentially improving survival and reducing recurrence rates.
Cell-Based Therapy
T-cell therapy represents a treatment for cancer patients that utilizes one’s own immune system against tumorigenesis. The T cell receptor (TCR) acts as an antigen sensor that detects proteins expressed by cancer – neoantigens, cancer-germline antigens, and viral oncoproteins – by binding to epitopes74, leading to the elimination of tumor cells. In a recent study exploring the effects of bispecific T-cell engagers (BiTEs), which are engineered antibodies that link T cells directly to tumor cells, the researchers took samples from patients diagnosed with melanoma, gastric, lung, pancreatic, and liver carcinomas. The findings indicate that BiTEs are especially effective for tumors exhibiting high PD-L1 expression, linking their targeted activity to the enhancement of T-cell function and subsequent anti-tumor effects75. Moreover, this study showed the effectiveness of BiTEs elevating oncolytic response both in vitro and in vivo. T-cell therapy has risen to become an effective cancer treatment as it can detect intracellular antigens derived from the full proteome of cancer cells. In contrast, antibodies are limited to identifying only surface-level epitopes76. Meaning, T-cell therapy offers a broader and more comprehensive immune response, allowing it to target cancer cells that would otherwise evade traditional antibody-based treatments.
Still, researchers face limitations with T-cell therapy, including low efficacy in solid tumors, which contain immune suppressors that block T-cell activity77 and are present in epithelial cancers such as OCSCC. The tumor-associated extracellular matrix that is a part of the microenvironment creates an obstinate barrier for T cells targeting cancer cells. Not only does the dense extracellular matrix physically impede T-cell movement into tumor parenchyma, but also creates high pressure that restricts cell migration78,79 . For these reasons, the tumor builds immune resistance rather than being destroyed74,80. To improve T-cell infiltration, researchers have remolded matrix-degrading enzymes that are able to cleave the extracellular matrix, facilitating T-cell entry81. Additionally, studies have discovered that modifying CAR-T cells to secrete heparinase enzyme would destroy the tumor matrix82. To maximize the advantages of cell-based therapy, more research on enzymatic remodeling as well as a comprehensive understanding of the tumor environment needs to be pursued. Although clinical data in oral cancer is limited, findings suggest overall efficacy in patients with high expression of PD-L1.
Emerging Therapies
A prominent concern in cancer treatment today is the invasiveness of therapies. Research has found therapies such as chemotherapy and surgical therapy to be unnecessarily invasive through the introduction of foreign drugs and/or tools into the patient’s body. Invasive treatment induces anoxia, bleeding, exudate, offensive odors, and dyspnea which significantly compromise a patient’s quality of life, delay healing, and increase the risk of infection. Especially in OCSCC patients, mastication (chewing), oral mucositis, deglutition (swallowing)83,84, maintenance of oral competency (keeping mouth closed), and articulation of speech85 are seen in most cases when treated with therapies such as surgical therapy, chemotherapy, and radiotherapy86.
Tumor Treating Fields
Recent studies have proposed tumor treating fields (TTFields), a type of electrotherapy as a non-invasive cancer treatment alternative. Intermittent electric fields are delivered to the tumor site from transducer arrays that are placed on the patient’s skin. The transducer arrays are connected to a portable device which allows the treatment to be delivered from home and during possible daily activities86. The electric currents exerted by TTFields employ physical force onto the cancer cell, disrupting its cell cycle, forcing it to cause cancer cell death. TTFields are commonly addressed to glioblastoma patients86 due to the complex spinal system of the encephalon. However, research has demonstrated the versatile nature of TTFields, allowing it to be applied as a monotherapy, or adjuvant with existing therapies. While TTFields have demonstrated clinical benefit in glioblastoma and are under investigation in other solid tumors, their application in OCSCC remains exploratory and has not yet been validated in clinical trials. TTFields send electric currents through skin, meaning, can be placed on the neck, face, or jaw. This novel approach represents a promising innovation in oncology, offering a targeted and noninvasive method to minimize unnecessary damage as well as improve overall prognosis87. If clinical trials expand, TTFields hold potential to revolutionize cancer care.
Nanotechnology
Nanotechnology serves as a rapidly revolutionizing cancer treatment technology that shows positive trends in oral cancer patients. Researchers define nanotechnology as the manipulation of matter on the molecular and atomic levels using nanorobotics, nanomaterials and biotechnology. Nano dentistry has emerged as a form of medicine that maintains near-perfect oral health88. In nanomedicine, nanoscale materials are applied to diagnostic techniques, targeted drug therapies, genomics, proteomics, artificial biotics, intermediate microbial diagnosis, and enhancement of physiological functions89,90,91. These techniques are applied through dentistry in a multitude of ways due to their versatile nature.
In general cancer therapy, nanotherapy has shown efficient results in drug delivery, due to their ability of deep tissue penetration, increasing drug permeability and retention92. Types of nanoparticles include Doxil, Eligard, Abraxane, Genexol-PM, and Onivyde. Doxil is a liposome coated with polyethylene glycol that administers the drug Doxorubicin, designed to target drug release to primary tumors while decreasing drug exposure to healthy tissue93. In preclinical models, Doxil has demonstrated less systemic toxicity than free doxorubicin, as well as versatility in multiple cancer types. Next, Eligard carries the drug Leuprolide acetate and functions to sustain hormone suppression of testosterone, mainly for prostate cancer patients94. Genexol-PM is another nanoparticle designed to carry Paciltaxel, designed to improve drug solubility and reduce toxic sensitivity while facilitating high doses95. Collectively, these nanoparticle techniques demonstrate how engineered drug carriers can facilitate cancer treatment. By enhancing tumor-specific accumulation and reducing systemic toxicity, nanotechnology-based approaches offer significant potential for a wider range of malignancies, including OCSCC.
In OCSCC therapy, doctors apply nanoanaesthesia, brachy therapy, and nanovectors therapy, valued for their preciseness and high efficacy.
Nanotechnology in active treatment includes brachy therapy, nano vectors for gene therapy, and nonviral gene delivery services.
Traditional brachy therapy via nanotechnology enhances targeted radiation therapy through immaculate precision and controlled release, minimizing invasiveness and disturbance to surrounding tissue96. Nano vectors, engineered to carry genetic material, deliver corrective genes to silence oncogenes or insert tumor-suppressor genes directly into the cancer cell97.
As previously mentioned, invasiveness is a vital component of conventional therapies such as chemotherapy, significantly worsening the patient’s quality of life as they suffer additional attacks on their immune systems. Targeted therapies via nanotechnology acknowledges these limitations and provides tangible solutions.
Outcome of Treatment Regimes
| S no | Studied by | Therapy | Type of Cancer/lesion | Outcome/Status |
| 1 | Babar et al.98 | Concurrent cisplatin + RT | 181 patients with oral cavity cell carcinoma | 51% overall survival over 3 year period |
| 2 | Yamazaki et al.99 | Reirradiation therapy | 32 patients with oral cavity cell carcinoma | 34.6% overall survival over 2 year period |
| 3 | Falco et al.100 | Immunotherapy | 40 Patients with Head and Neck cell carcinoma | 76% overall survival of 12 months |
| 4 | Liu et al.101 | Carmelizmub | 34 patients with locally advanced oral cavity cell carcinoma | 88.2% overall survival over 32 months |
| 5 | Timar et al.102 | Leukocyte Interleukin Injection | 39 patients diagnosed with moderately advanced oral cavity cell carcinoma | Overall response rate, 42% |
| 6 | Saba et al.103 | Nivolumab | 361 patients diagnosed with recurrent/metastatic squamous cell carcinoma of the head and neck of the oral cavity, oropharynx, hypo-pharynx, or larynx | Higher median overall survival in nivolumab patients and overall Response Rate versus control group ie. 12.8% Vs 6.6% |
| 7 | Gross et al.104 | Cemiplimab(neoadjuvant PD-1 inhibition) | 20 locoregionally advanced squamous cell carcinoma of the head and neck | 12-month disease-free survival and overall survival rates were 89.5% and 95% respectively |
Table 1: Overall survival of patients stratified by type of therapy received. Survival was assessed over a follow-up period of 1 to 3 years and compiled from published clinical studies (see Methods for details). Concurrent cisplatin + RT 3 years survival of 181 patients98, Reirradiation therapy of 2 years survival of 32 patients99, Immune checkpoint inhibitors 12 months overall survival status of 40 patients100, Camrelizumab therapy 32 months survival of 34 patients101. Leukocyte interleukin injection is administered as a neoadjuvant immunotherapy, shown to alter the composition of tumor-infiltrating mononuclear cells, increase CD4+:CD8+ ratio and tumor stroma to epithelial ratio102. Patients treated with nivolumab, PD-1 checkpoint inhibitor classified under immunotherapy drugs, experienced higher overall survival, response rate, and regulation of PD-1103. Patients treated with Cemiplimab, and neoadjuvant PD-1 inhibition immunotherapy drug, had 12 month disease-free survival104.
| Margin Status | N of patients | Hazard Ratio for OS (95% Confidence Interval) |
| Positive Margin | 3237 | 2.27 (1.43–3.58) |
| Close Margin | 2627 | 1.41 (1.06–1.87) |
HR = 1, meaning no difference in risk. HR > 1, meaning higher risk of death in the study group compared to those with clear margins. HR < 1, meaning lower risk of death in the study group compared to those with clear margin. Surgical margins being defined as the edges of the tissue removed during surgery105.
Hazard Ratio equaling 1 means there is no difference in risk between patients in the control group and patients in the study group. HR > 1, meaning higher risk of death in the study group compared to those with clear margins. HR < 1, meaning lower risk of death in the study group compared to those with clear margin. Surgical margins being defined as the edges of the tissue removed during surgery105 . Patients with positive margins, defined as margins being less than 1mm, have about 2.3 times higher risk of death compared to those with clear margins, margins of greater than 5mm. Patients with close margins, margins of 1-5mm, have about 1.4 times higher risk of death compared to clear margins. This illustrates the associated risks of surgical treatment, as full removal of cancer cells may be challenging, especially after metastasis and invasion. Even with a close margin patients are 41% more at risk of death compared to patients with completely clear margins.
Discussion
There are approximately 54,000 annual cases of OCSCC patients, representing about 4.6% of global cancer-related deaths. These statistics demonstrate serious challenges in the field of cancer therapy, invoking the need for advancements in treatment, such as developing targeted therapies such as immunotherapy, nano therapy, and electrotherapy. As shown in clinical data, overall survival rate is higher in patients treated with immunotherapy techniques, including immune checkpoint inhibitors, compared to that of chemotherapy and conventional radiotherapy.
Oral cancer has become a critical disease today, however, advancements in therapies are constantly being developed to address current limitations, reduce recurrence rates, and increase overall survival. The nature of the OCSCC invokes life-threatening conditions to the patients, including the inability to speak, eat, or breathe properly. To solve the widespread concern of invasive and ineffective treatment for OCSCC patients, this paper systematically reviews advancements in treatment that offer new approaches. Advancements in immunotherapy have been rapidly developing, with promising OCSCC techniques in T-cell, TIL, and checkpoint therapy. These therapies show strong defenses against tumorigenesis, resulting in less aggressive cancer stages and lower recurrence rates. Table 1 summarizes various studies focusing on different methods of therapy, with immunotherapy administration showing improved overall status.
These conventional therapies have associated risks, along with lower survival rates and higher recurrence rates. In surgical treatment, a clear margin represents full removal of the tumor and patients without clear margins have a significantly increased risk of death. This illustrates the need for alternative treatments, even neoadjuvant techniques. Moreover, chemotherapy and radiotherapy pose higher risks of invasiveness, systemic toxicity, and immunosuppression, as these types of drugs lack the ability to specifically target and destroy only cancer cells. More recently, immunotherapy has shown promising results in monotherapy and cell-based therapy. By utilizing a patient’s own immune system and manipulating it to enhance the body’s defense mechanisms, immunotherapy acknowledges the invasive nature of cancer therapies. TTFields in radiotherapy and the versatile techniques of nanotechnology offer therapeutic strategies that can enhance treatment efficacy through targeted drug delivery and localized tumor disruption, ultimately improving quality of life for OCSCC patients while addressing the limitations of conventional therapy.
However, patient access to these novel therapies are limited due to cost barriers. CAR T-cell administration averages around $454 000 in an academic hospital setting106, similar to administering nanotechnology, TTFields, and other types of immunotherapy. Disproportionately, access to healthcare is limited in low-income communities, or even minority-concentrated areas. Factors such as discrimination, hospital closures, or distance, influence treatment options for an individual. Despite high success rates, advanced treatment remains inaccessible, responding to socioeconomic and even geographic gaps.
In terms of clinical application, patients of different situations benefit most from distinct therapies. Early-stage OCSCC patients would most benefit from surgery with nanotechnology-enhanced diagnostics, considering the effective nanoanesthesia and incredibly high preciseness. Those with advanced-stage OCSCC, or recurrence, immunotherapy would be applied, as the enhancement of the body’s own immune system is most effective in full removal of the tumor, minimizing recurrence rates and toxicity. For most therapies, especially immune checkpoint inhibitors, patients with high PD-L1 or CTLA-4 expression should be addressed, considering the specific target of these therapies to these proteins. In all, these novel therapies provide targeted, non-invasive techniques to improve overall survival.
It’s important to acknowledge the oral cavity’s tumor microenvironment, and its vital role in the development of tumorigenesis. Emerging therapies must target the TME to create the most effective treatment for patients. Especially in hypoxic environments, the oral cavity is more susceptible to tumor development as lower oxygen levels facilitate the activation of downstream signaling pathways in the TME.
To acknowledge the limitations of this literature review, new clinical data on oral cancer patients treated with emerging therapies aren’t as prevalent. As a result, there was heterogeneity among the studies, and reliance on head and neck cancer data was not always specific to OCSCC. Table 1 highlights the overall survival rate of four therapies under different confounding conditions.
Future research should focus on the development and integration of novel adjuvant therapies to further improve survival outcomes, as well as working on making cancer treatment available to wider demographics. The application of nanotechnology-based therapies has shown potential in removing OCSCC and should be further explored in how it can be used in conjunction with immunotherapy, where destruction of cancer cells can become even more targeted. Tumor-infiltration lymphocytes is an additional therapy that is extremely novel in oral cancer, representing effectiveness in high mutational burdens that would offer durable and personalized alternatives in cases where surgical margins are suboptimal. Even adjuvant therapies of conventional treatment have promising effects on survival outcomes. Research on immunotherapy and radiotherapy, or radiotherapy in combination with surgical therapy should be further explored to confirm effectiveness of such adjuvant therapies. Taken together, this literature review underscores the urgent call for advancements in oral cavity squamous cell carcinoma therapies to provide effective treatments to the hundreds of thousands that suffer from this disease. The use of emerging therapies, immunotherapy, electrotherapy, nanotechnology, have the potential to reduce recurrence and improve survival rates, as continued research will be essential in establishing these as standard care in OCSCC.
Acknowledgments
I would like to express gratitude to Dr. Anna Clay, University of Cambridge, for her invaluable guidance in this research process.
References
- K. G. Masters, S. Lasrado. Anatomy, head and neck: tonsils. in StatPearls StatPearls Publishing, Treasure Island (FL), 2025 [↩]
- T. Wong, D. Wiesenfeld. Oral cancer. Australian Dental Journal. Vol. 63, 2018 https://doi.org/10.1111/adj.12594 [↩]
- P. Kamrani, N. M. Sadiq. Anatomy, head and neck, oral cavity (mouth). in StatPearls StatPearls Publishing, Treasure Island (FL), 2025 [↩] [↩]
- Oral cancer | nidcr. [↩] [↩]
- D. J. Weatherspoon, A. Chattopadhyay, S. Boroumand, I. Garcia. Oral cavity and oropharyngeal cancer incidence trends and disparities in the united states: 2000-2010. Cancer Epidemiology. Vol. 39, pg. 497–504, 2015 https://doi.org/10.1016/j.canep.2015.04.007 [↩]
- X.-X. Bai, J. Zhang, L. Wei. Analysis of primary oral and oropharyngeal squamous cell carcinoma in inhabitants of beijing, china—a 10-year continuous single-center study. BMC Oral Health. Vol. 20, pg. 208, 2020 https://doi.org/10.1186/s12903-020-01192-6 [↩]
- Oral cancers on the rise in young people | university hospitals. [↩]
- B. Chakrobarty, J. G. Roy, S. Majumdar, D. Uppala. Relationship among tobacco habits, human papilloma virus (hpv) infection, p53 polymorphism/mutation and the risk of oral squamous cell carcinoma. Journal of Oral and Maxillofacial Pathology: JOMFP. Vol. 18, pg. 211–216, 2014 https://doi.org/10.4103/0973-029X.140752 [↩] [↩]
- A. Agrawal, E. Shindell, F. Jordan, L. Baeva, J. Pfefer, D. E. Godar. UV radiation increases carcinogenic risks for oral tissues compared to skin. Photochemistry and Photobiology. Vol. 89, pg. 1193–1198, 2013 https://doi.org/10.1111/php.12140 [↩] [↩]
- G. D’Souza, A. R. Kreimer, R. Viscidi, M. Pawlita, C. Fakhry, W. M. Koch, W. H. Westra, M. L. Gillison. Case-control study of human papillomavirus and oropharyngeal cancer. The New England Journal of Medicine. Vol. 356, pg. 1944–1956, 2007 https://doi.org/10.1056/NEJMoa065497 [↩]
- G. R. Thomas. Racial disparity in head and neck cancer. Cancer. Vol. 127, pg. 2612–2613, 2021 https://doi.org/10.1002/cncr.33555 [↩]
- L. M. Brown, D. P. Check, S. S. Devesa. Oral cavity and pharynx cancer incidence trends by subsite in the united states: changing gender patterns. Journal of Oncology. Vol. 2012, pg. 649498, 2012 https://doi.org/10.1155/2012/649498 [↩]
- A. J. Yu, J. S. Choi, M. S. Swanson, N. C. Kokot, T. N. Brown, G. Yan, U. K. Sinha. Association of race/ethnicity, stage, and survival in oral cavity squamous cell carcinoma: a seer study. OTO Open. Vol. 3, pg. 2473974X19891126, 2019 https://doi.org/10.1177/2473974X19891126 [↩]
- Oral cancer incidence (new cases) by age, race, and sex | nidcr. [↩]
- Cancer today. https://gco.iarc.who.int/today/ [↩]
- S. Blatt, M. Krüger, K. Sagheb, M. Barth, P. W. Kämmerer, B. Al-Nawas, K. Sagheb. Tumor recurrence and follow-up intervals in oral squamous cell carcinoma. Journal of Clinical Medicine. Vol. 11, pg. 7061, 2022 https://doi.org/10.3390/jcm11237061 [↩] [↩] [↩]
- D. Hanahan, R. A. Weinberg. The hallmarks of cancer. Cell. Vol. 100, pg. 57–70, 2000 https://doi.org/10.1016/s0092-8674(00)81683-9 [↩]
- N. D. Freedman, C. C. Abnet, M. F. Leitzmann, A. R. Hollenbeck, A. Schatzkin. Prospective investigation of the cigarette smoking-head and neck cancer association by sex. Cancer. Vol. 110, pg. 1593–1601, 2007 https://doi.org/10.1002/cncr.22957 [↩]
- H. Hamada, Y. Tashima, Y. Kisaka, K. Iwamoto, T. Hanai, Y. Eguchi, M. Okamoto. Sophisticated framework between cell cycle arrest and apoptosis induction based on p53 dynamics. PLOS ONE. Vol. 4, pg. e4795, 2009 https://doi.org/10.1371/journal.pone.0004795 [↩]
- D. L. Faden, S. T. Arron, C. M. Heaton, J. DeRisi, A. P. South, S. J. Wang. Targeted next-generation sequencing of tp53 in oral tongue carcinoma from non-smokers. Journal of Otolaryngology – Head & Neck Surgery = Le Journal D’oto-Rhino-Laryngologie Et De Chirurgie Cervico-Faciale. Vol. 45, pg. 47, 2016 https://doi.org/10.1186/s40463-016-0160-4 [↩]
- I. Tojyo, Y. Shintani, T. Nakanishi, K. Okamoto, Y. Hiraishi, S. Fujita, M. Enaka, F. Sato, Y. Muragaki. PD-l1 expression correlated with p53 expression in oral squamous cell carcinoma. Maxillofacial Plastic and Reconstructive Surgery. Vol. 41, pg. 56, 2019 https://doi.org/10.1186/s40902-019-0239-8 [↩]
- Oral cancer prevention – mississippi state department of health. [↩]
- E. Casas, J. Kim, A. Bendesky, L. Ohno-Machado, C. J. Wolfe, J. Yang. Snail2 is an essential mediator of twist1-induced epithelial mesenchymal transition and metastasis. Cancer Research. Vol. 71, pg. 245–254, 2011 https://doi.org/10.1158/0008-5472.CAN-10-2330 [↩]
- Stages of cancer | cancer research uk. [↩]
- Surveillance, epidemiology, and end results program. SEER https://seer.cancer.gov/index.html [↩]
- B. Arneth. Tumor microenvironment. Medicina. Vol. 56, pg. 15, 2019 https://doi.org/10.3390/medicina56010015 [↩]
- E. E. Verwer, R. Boellaard, A. A. van der Veldt. Positron emission tomography to assess hypoxia and perfusion in lung cancer. World Journal of Clinical Oncology. Vol. 5, pg. 824–844, 2014 https://doi.org/10.5306/wjco.v5.i5.824 [↩]
- M. Z. Noman, G. Desantis, B. Janji, M. Hasmim, S. Karray, P. Dessen, V. Bronte, S. Chouaib. PD-l1 is a novel direct target of hif-1α, and its blockade under hypoxia enhanced mdsc-mediated t cell activation. The Journal of Experimental Medicine. Vol. 211, pg. 781–790, 2014 https://doi.org/10.1084/jem.20131916 [↩]
- C. A. Corzo, T. Condamine, L. Lu, M. J. Cotter, J.-I. Youn, P. Cheng, H.-I. Cho, E. Celis, D. G. Quiceno, T. Padhya, T. V. McCaffrey, J. C. McCaffrey, D. I. Gabrilovich. HIF-1α regulates function and differentiation of myeloid-derived suppressor cells in the tumor microenvironment. The Journal of Experimental Medicine. Vol. 207, pg. 2439–2453, 2010 https://doi.org/10.1084/jem.20100587 [↩]
- A. Casazza, D. Laoui, M. Wenes, S. Rizzolio, N. Bassani, M. Mambretti, S. Deschoemaeker, J. A. Van Ginderachter, L. Tamagnone, M. Mazzone. Impeding macrophage entry into hypoxic tumor areas by sema3a/nrp1 signaling blockade inhibits angiogenesis and restores antitumor immunity. Cancer Cell. Vol. 24, pg. 695–709, 2013 https://doi.org/10.1016/j.ccr.2013.11.007 [↩]
- A. Facciabene, X. Peng, I. S. Hagemann, K. Balint, A. Barchetti, L.-P. Wang, P. A. Gimotty, C. B. Gilks, P. Lal, L. Zhang, G. Coukos. Tumour hypoxia promotes tolerance and angiogenesis via ccl28 and t(reg) cells. Nature. Vol. 475, pg. 226–230, 2011 https://doi.org/10.1038/nature10169 [↩]
- Y.-L. Chang, C.-Y. Yang, M.-W. Lin, C.-T. Wu, P.-C. Yang. High co-expression of pd-l1 and hif-1α correlates with tumour necrosis in pulmonary pleomorphic carcinoma. European Journal of Cancer (Oxford, England: 1990). Vol. 60, pg. 125–135, 2016 https://doi.org/10.1016/j.ejca.2016.03.012 [↩] [↩]
- S. Niklander, M. J. Bordagaray, A. Fernández, M. Hernández. Vascular endothelial growth factor: a translational view in oral non-communicable diseases. Biomolecules. Vol. 11, pg. 85, 2021 https://doi.org/10.3390/biom11010085 [↩]
- M. Huang, B. Huang, G. Li, S. Zeng. Apatinib affect vegf-mediated cell proliferation, migration, invasion via blocking vegfr2/raf/mek/erk and pi3k/akt pathways in cholangiocarcinoma cell. BMC Gastroenterology. Vol. 18, pg. 169, 2018 https://doi.org/10.1186/s12876-018-0870-3 [↩]
- S. M. Morrissey, F. Zhang, C. Ding, D. E. Montoya-Durango, X. Hu, C. Yang, Z. Wang, F. Yuan, M. Fox, H.-G. Zhang, H. Guo, D. Tieri, M. Kong, C. T. Watson, R. A. Mitchell, X. Zhang, K. M. McMasters, J. Huang, J. Yan. Tumor-derived exosomes drive immunosuppressive macrophages in a pre-metastatic niche through glycolytic dominant metabolic reprogramming. Cell Metabolism. Vol. 33, pg. 2040-2058.e10, 2021 https://doi.org/10.1016/j.cmet.2021.09.002 [↩]
- M. Gonzalez-Begne, B. Lu, X. Han, F. K. Hagen, A. R. Hand, J. E. Melvin, J. R. Yates. Proteomic analysis of human parotid gland exosomes by multidimensional protein identification technology (mudpit). Journal of Proteome Research. Vol. 8, pg. 1304–1314, 2009 https://doi.org/10.1021/pr800658c [↩]
- C. Gai, F. Camussi, R. Broccoletti, A. Gambino, M. Cabras, L. Molinaro, S. Carossa, G. Camussi, P. G. Arduino. Salivary extracellular vesicle-associated mirnas as potential biomarkers in oral squamous cell carcinoma. BMC Cancer. Vol. 18, pg. 439, 2018 https://doi.org/10.1186/s12885-018-4364-z [↩]
- Z. Ji, X. Wang, J. Xin, L. Ma, D. Zuo, H. Li, L. Su, X. Lv, S. Ge, L. Zhang, Y. Liu, Y. Zhang, T. Ding, T. Deng, Y. Ba, R. Liu. Multiomics reveals tumor microenvironment remodeling in locally advanced gastric and gastroesophageal junction cancer following neoadjuvant immunotherapy and chemotherapy. Journal for Immunotherapy of Cancer. Vol. 12, pg. e010041, 2024 https://doi.org/10.1136/jitc-2024-010041 [↩]
- Y.-T. Liu, H.-M. Liu, J.-G. Ren, W. Zhang, X.-X. Wang, Z.-L. Yu, Q.-Y. Fu, X.-P. Xiong, J. Jia, B. Liu, G. Chen. Immune-featured stromal niches associate with response to neoadjuvant immunotherapy in oral squamous cell carcinoma. Cell Reports. Medicine. Vol. 6, pg. 102024, 2025 https://doi.org/10.1016/j.xcrm.2025.102024 [↩]
- A. Moreira, C. Bernardo, C. Ramos, P. Aguiar, F. Alves da Costa. National trends in the use of oral chemotherapy over 13 years. Frontiers in Pharmacology. Vol. 13, pg. 909948, 2022 https://doi.org/10.3389/fphar.2022.909948 [↩]
- M. T. Amjad, A. Chidharla, A. Kasi. Cancer chemotherapy. in StatPearls StatPearls Publishing, Treasure Island (FL), 2025 [↩]
- M. Kawami, R. Harabayashi, M. Miyamoto, R. Harada, R. Yumoto, M. Takano. Methotrexate-induced epithelial-mesenchymal transition in the alveolar epithelial cell line a549. Lung. Vol. 194, pg. 923–930, 2016 https://doi.org/10.1007/s00408-016-9935-7 [↩]
- T. Sakabe, H. Tsuchiya, K. Kanki, J. Azumi, K. Gonda, Y. Mizuta, D. Yamada, H. Wada, K. Shomori, H. Nagano, G. Shiota. Identification of the genes chemosensitizing hepatocellular carcinoma cells to interferon-α/5-fluorouracil and their clinical significance. PloS One. Vol. 8, pg. e56197, 2013 https://doi.org/10.1371/journal.pone.0056197 [↩]
- E. Munzone, M. M. Regan, S. Cinieri, E. Montagna, L. Orlando, R. Shi, E. Campadelli, L. Gianni, M. Palleschi, F. Petrelli, C. Bengala, D. Generali, E. Collovà, F. Puglisi, E. Cretella, C. Zamagni, C. Chini, B. Ruepp, S. Loi, M. Colleoni, International Breast Cancer Study Group (IBCSG). Efficacy of metronomic oral vinorelbine, cyclophosphamide, and capecitabine vs weekly intravenous paclitaxel in patients with estrogen receptor-positive, erbb2-negative metastatic breast cancer: final results from the phase 2 meteora-ii randomized clinical trial. JAMA Oncology. Vol. 9, pg. 1267–1272, 2023 https://doi.org/10.1001/jamaoncol.2023.2150 [↩]
- M. Bostan, G. G. Petrică-Matei, G. Ion, N. Radu, M. Mihăilă, R. Hainăroşie, L. I. Braşoveanu, V. Roman, C. Constantin, M. T. Neagu. Cisplatin effect on head and neck squamous cell carcinoma cells is modulated by erk1/2 protein kinases. Experimental and Therapeutic Medicine. Vol. 18, pg. 5041–5051, 2019 https://doi.org/10.3892/etm.2019.8139 [↩]
- A. J. Rosenberg, A. Juloori, M. J. Jelinek, N. Agrawal, J. F. Cursio, N. Cipriani, M. W. Lingen, E. Izumchenko, R. Katipally, J. Chin, D. Ginat, O. Pasternak-Wise, Z. Gooi, E. Blair, A. T. Pearson, D. J. Haraf, E. E. Vokes. Neoadjuvant nivolumab plus chemotherapy followed by response-stratified chemoradiation therapy in hpv-negative head and neck cancer: the depend phase 2 nonrandomized clinical trial. JAMA Oncology. Vol. 11, pg. 492–501, 2025 https://doi.org/10.1001/jamaoncol.2025.0081 [↩]
- H. Zang, G. Qian, J. Arbiser, T. K. Owonikoko, S. S. Ramalingam, S. Fan, S.-Y. Sun. Overcoming acquired resistance of egfr-mutant nsclc cells to the third generation egfr inhibitor, osimertinib, with the natural product honokiol. Molecular Oncology. Vol. 14, pg. 882–895, 2020 https://doi.org/10.1002/1878-0261.12645 [↩]
- N. Cayuela, E. Jaramillo-Jiménez, E. Càmara, C. Majós, N. Vidal, A. Lucas, M. Gil-Gil, F. Graus, J. Bruna, M. Simó. Cognitive and brain structural changes in long-term oligodendroglial tumor survivors. Neuro-Oncology. Vol. 21, pg. 1470–1479, 2019 https://doi.org/10.1093/neuonc/noz130 [↩]
- Y. Liu, M. Yang, J. Luo, H. Zhou. Radiotherapy targeting cancer stem cells ‘awakens’ them to induce tumour relapse and metastasis in oral cancer. International Journal of Oral Science. Vol. 12, pg. 19, 2020 https://doi.org/10.1038/s41368-020-00087-0 [↩]
- M. Lapeyre, M. A. Bollet, S. Racadot, L. Geoffrois, M.-C. Kaminsky, S. Hoffstetter, G. Dolivet, B. Toussaint, E. Luporsi, D. Peiffert. Postoperative brachytherapy alone and combined postoperative radiotherapy and brachytherapy boost for squamous cell carcinoma of the oral cavity, with positive or close margins. Head & Neck. Vol. 26, pg. 216–223, 2004 https://doi.org/10.1002/hed.10377 [↩] [↩]
- T.-W. Leung, V. Y.-W. Wong, K.-H. Kwan, T.-Y. Ng, C.-M. Wong, S. Y. Tung, L.-C. Leung, S.-K. O. High dose rate brachytherapy for early stage oral tongue cancer. Head & Neck. Vol. 24, pg. 274–281, 2002 https://doi.org/10.1002/hed.10021 [↩]
- U. Selek, A. S. Garden, W. H. Morrison, A. K. El-Naggar, D. I. Rosenthal, K. K. Ang. Radiation therapy for early-stage carcinoma of the oropharynx. International Journal of Radiation Oncology, Biology, Physics. Vol. 59, pg. 743–751, 2004 https://doi.org/10.1016/j.ijrobp.2003.12.002 [↩]
- B. O’Sullivan, R. B. Rumble, P. Warde, Members of the IMRT Indications Expert Panel. Intensity-modulated radiotherapy in the treatment of head and neck cancer. Clinical Oncology (Royal College of Radiologists (Great Britain). Vol. 24, pg. 474–487, 2012 https://doi.org/10.1016/j.clon.2012.05.006 [↩]
- G. Studer, S. P. Studer, R. A. Zwahlen, P. Huguenin, K. W. Grätz, U. M. Lütolf, C. Glanzmann. Osteoradionecrosis of the mandible: minimized risk profile following intensity-modulated radiation therapy (imrt). Strahlentherapie Und Onkologie: Organ Der Deutschen Rontgengesellschaft … [et Al]. Vol. 182, pg. 283–288, 2006 https://doi.org/10.1007/s00066-006-1477-0 [↩]
- Surgery for cancer – nci. [↩]
- J. P. Shah. Surgical approaches to the oral cavity primary and neck. International Journal of Radiation Oncology*Biology*Physics. Vol. 69, pg. S15–S18, 2007 https://doi.org/10.1016/j.ijrobp.2007.03.069 [↩] [↩]
- E. A. Stadtmauer, J. A. Fraietta, M. M. Davis, A. D. Cohen, K. L. Weber, E. Lancaster, P. A. Mangan, I. Kulikovskaya, M. Gupta, F. Chen, L. Tian, V. E. Gonzalez, J. Xu, I. Jung, J. J. Melenhorst, G. Plesa, J. Shea, T. Matlawski, A. Cervini, A. L. Gaymon, S. Desjardins, A. Lamontagne, J. Salas-Mckee, A. Fesnak, D. L. Siegel, B. L. Levine, J. K. Jadlowsky, R. M. Young, A. Chew, W.-T. Hwang, E. O. Hexner, B. M. Carreno, C. L. Nobles, F. D. Bushman, K. R. Parker, Y. Qi, A. T. Satpathy, H. Y. Chang, Y. Zhao, S. F. Lacey, C. H. June. CRISPR-engineered t cells in patients with refractory cancer. Science (New York, N.Y.). Vol. 367, pg. eaba7365, 2020 https://doi.org/10.1126/science.aba7365 [↩]
- I. Puzanov, A. Diab, K. Abdallah, C. O. Bingham, C. Brogdon, R. Dadu, L. Hamad, S. Kim, M. E. Lacouture, N. R. LeBoeuf, D. Lenihan, C. Onofrei, V. Shannon, R. Sharma, A. W. Silk, D. Skondra, M. E. Suarez-Almazor, Y. Wang, K. Wiley, H. L. Kaufman, M. S. Ernstoff, Society for Immunotherapy of Cancer Toxicity Management Working Group. Managing toxicities associated with immune checkpoint inhibitors: consensus recommendations from the society for immunotherapy of cancer (sitc) toxicity management working group. Journal for Immunotherapy of Cancer. Vol. 5, pg. 95, 2017 https://doi.org/10.1186/s40425-017-0300-z [↩] [↩]
- E. A. Mittendorf, H. Zhang, C. H. Barrios, S. Saji, K. H. Jung, R. Hegg, A. Koehler, J. Sohn, H. Iwata, M. L. Telli, C. Ferrario, K. Punie, F. Penault-Llorca, S. Patel, A. N. Duc, M. Liste-Hermoso, V. Maiya, L. Molinero, S. Y. Chui, N. Harbeck. Neoadjuvant atezolizumab in combination with sequential nab-paclitaxel and anthracycline-based chemotherapy versus placebo and chemotherapy in patients with early-stage triple-negative breast cancer (impassion031): a randomised, double-blind, phase 3 trial. Lancet (London, England). Vol. 396, pg. 1090–1100, 2020 https://doi.org/10.1016/S0140-6736(20)31953-X [↩] [↩]
- D. S. Shin, J. M. Zaretsky, H. Escuin-Ordinas, A. Garcia-Diaz, S. Hu-Lieskovan, A. Kalbasi, C. S. Grasso, W. Hugo, S. Sandoval, D. Y. Torrejon, N. Palaskas, G. A. Rodriguez, G. Parisi, A. Azhdam, B. Chmielowski, G. Cherry, E. Seja, B. Berent-Maoz, I. P. Shintaku, D. T. Le, D. M. Pardoll, L. A. Diaz, P. C. Tumeh, T. G. Graeber, R. S. Lo, B. Comin-Anduix, A. Ribas. Primary resistance to pd-1 blockade mediated by jak1/2 mutations. Cancer Discovery. Vol. 7, pg. 188–201, 2017 https://doi.org/10.1158/2159-8290.CD-16-1223 [↩]
- J. M. Zaretsky, A. Garcia-Diaz, D. S. Shin, H. Escuin-Ordinas, W. Hugo, S. Hu-Lieskovan, D. Y. Torrejon, G. Abril-Rodriguez, S. Sandoval, L. Barthly, J. Saco, B. Homet Moreno, R. Mezzadra, B. Chmielowski, K. Ruchalski, I. P. Shintaku, P. J. Sanchez, C. Puig-Saus, G. Cherry, E. Seja, X. Kong, J. Pang, B. Berent-Maoz, B. Comin-Anduix, T. G. Graeber, P. C. Tumeh, T. N. M. Schumacher, R. S. Lo, A. Ribas. Mutations associated with acquired resistance to pd-1 blockade in melanoma. The New England Journal of Medicine. Vol. 375, pg. 819–829, 2016 https://doi.org/10.1056/NEJMoa1604958 [↩]
- D. C. Deniger, A. Pasetto, P. F. Robbins, J. J. Gartner, T. D. Prickett, B. C. Paria, P. Malekzadeh, L. Jia, R. Yossef, M. M. Langhan, J. R. Wunderlich, D. N. Danforth, R. P. T. Somerville, S. A. Rosenberg. T-cell responses to tp53 ‘hotspot’ mutations and unique neoantigens expressed by human ovarian cancers. Clinical Cancer Research: An Official Journal of the American Association for Cancer Research. Vol. 24, pg. 5562–5573, 2018 https://doi.org/10.1158/1078-0432.CCR-18-0573 [↩]
- J. E. Mullinax, M. Hall, M. Beatty, A. M. Weber, Z. Sannasardo, T. Svrdlin, J. Hensel, M. Bui, A. Richards, R. J. Gonzalez, C. A. Cox, L. Kelley, J. J. Mulé, A. A. Sarnaik, S. Pilon-Thomas. Expanded tumor-infiltrating lymphocytes from soft tissue sarcoma have tumor-specific function. Journal of Immunotherapy. Vol. 44, pg. 63, 2021 https://doi.org/10.1097/CJI.0000000000000355 [↩]
- D. J. Einstein, B. Halbert, T. Denize, S. Matar, D. J. West, M. Gupta, E. Andrianopoulos, V. Seery, C. Herman, K. Onimus, A. Wells, B. Bunch, S. Signoretti, A. Natarajan, A. Veerapathran, D. F. McDermott. Generation and characterization of ex vivo expanded tumor-infiltrating lymphocytes from renal cell carcinoma tumors for adoptive cell therapy. Journal of Immunotherapy. Vol. 47, pg. 361–368, 2024 https://doi.org/10.1097/CJI.0000000000000533 [↩]
- G. T. Wolf, S. Liu, E. Bellile, M. Sartor, L. Rozek, D. Thomas, A. Nguyen, K. Zarins, J. B. McHugh, INSPIRE Trial Clinical Investigators. Tumor infiltrating lymphocytes after neoadjuvant irx-2 immunotherapy in oral squamous cell carcinoma: interim findings from the inspire trial. Oral Oncology. Vol. 111, pg. 104928, 2020 https://doi.org/10.1016/j.oraloncology.2020.104928 [↩]
- I. H. Nauta, D. N. L. M. Nijenhuis, S. H. Ganzevles, P. I. Raaff, J. Kloosterman, E. Bloemena, R. H. Brakenhoff, C. R. Leemans, R. Van De Ven. Richness for tumor-infiltrating b-cells in the oral cancer tumor microenvironment is a prognostic factor in early-stage disease and improves outcome in advanced-stage disease. Cancers. Vol. 17, pg. 113, 2025 https://doi.org/10.3390/cancers17010113 [↩] [↩]
- P. Balermpas, F. Rödel, C. Rödel, M. Krause, A. Linge, F. Lohaus, M. Baumann, I. Tinhofer, V. Budach, E. Gkika, M. Stuschke, M. Avlar, A.-L. Grosu, A. Abdollahi, J. Debus, C. Bayer, S. Stangl, C. Belka, S. Pigorsch, G. Multhoff, S. E. Combs, D. Mönnich, D. Zips, E. Fokas. CD8+ tumour-infiltrating lymphocytes in relation to hpv status and clinical outcome in patients with head and neck cancer after postoperative chemoradiotherapy: a multicentre study of the german cancer consortium radiation oncology group (dktk-rog). International Journal of Cancer. Vol. 138, pg. 171–181, 2016 https://doi.org/10.1002/ijc.29683 [↩]
- K. L. Good-Jacobson, C. G. Szumilas, L. Chen, A. H. Sharpe, M. M. Tomayko, M. J. Shlomchik. PD-1 regulates germinal center b cell survival and the formation and affinity of long-lived plasma cells. Nature Immunology. Vol. 11, pg. 535–542, 2010 https://doi.org/10.1038/ni.1877 [↩]
- D. Quandt, H. Hoff, M. Rudolph, S. Fillatreau, M. C. Brunner-Weinzierl. A new role of ctla-4 on b cells in thymus-dependent immune responses in vivo. Journal of Immunology (Baltimore, Md.: 1950). Vol. 179, pg. 7316–7324, 2007 https://doi.org/10.4049/jimmunol.179.11.7316 [↩]
- J. Ries, L. Trumet, A. Hahn, L. Kunater, R. Lutz, C. Geppert, M. Kesting, M. Weber. The immune checkpoint btla in oral cancer: expression analysis and its correlation to other immune modulators. International Journal of Molecular Sciences. Vol. 25, pg. 6601, 2024 https://doi.org/10.3390/ijms25126601 [↩]
- Z. Xu, C. J. Vandenberg, E. Lieschke, L. Di Rago, C. L. Scott, I. J. Majewski. CHK2 inhibition provides a strategy to suppress hematologic toxicity from parp inhibitors. Molecular Cancer Research: MCR. Vol. 19, pg. 1350–1360, 2021 https://doi.org/10.1158/1541-7786.MCR-20-0791 [↩]
- FDA approves neoadjuvant and adjuvant pembrolizumab for resectable locally advanced head and neck squamous cell carcinoma | fda. [↩]
- Y. Shiravand, F. Khodadadi, S. M. A. Kashani, S. R. Hosseini-Fard, S. Hosseini, H. Sadeghirad, R. Ladwa, K. O’Byrne, A. Kulasinghe. Immune checkpoint inhibitors in cancer therapy. Current Oncology (Toronto, Ont.). Vol. 29, pg. 3044–3060, 2022 https://doi.org/10.3390/curroncol29050247 [↩]
- Q. Ye, D.-G. Song, M. Poussin, T. Yamamoto, A. Best, C. Li, G. Coukos, D. J. Powell. CD137 accurately identifies and enriches for naturally occurring tumor-reactive t cells in tumor. Clinical Cancer Research: An Official Journal of the American Association for Cancer Research. Vol. 20, pg. 44–55, 2014 https://doi.org/10.1158/1078-0432.CCR-13-0945 [↩] [↩]
- J. Jin, R. Wang, J. Yang, H. Hu, D. Wang, L. Cai, Z. Fang, S. Dong, S. Hu, Y. Wang, B. Liu. Bispecific antibody expressed by an oncolytic herpes simplex virus type 2 can transform heterologous t cells into uniform tumor killer cells. Human Gene Therapy. Vol. 33, pg. 649–663, 2022 https://doi.org/10.1089/hum.2021.277 [↩]
- Z. L. Chang, M. H. Lorenzini, X. Chen, U. Tran, N. J. Bangayan, Y. Y. Chen. Rewiring t-cell responses to soluble factors with chimeric antigen receptors. Nature Chemical Biology. Vol. 14, pg. 317–324, 2018 https://doi.org/10.1038/nchembio.2565 [↩]
- G. Wang, X. Lu, P. Dey, P. Deng, C. C. Wu, S. Jiang, Z. Fang, K. Zhao, R. Konaparthi, S. Hua, J. Zhang, E. M. Li-Ning-Tapia, A. Kapoor, C.-J. Wu, N. B. Patel, Z. Guo, V. Ramamoorthy, T. N. Tieu, T. Heffernan, D. Zhao, X. Shang, S. Khadka, P. Hou, B. Hu, E.-J. Jin, W. Yao, X. Pan, Z. Ding, Y. Shi, L. Li, Q. Chang, P. Troncoso, C. J. Logothetis, M. J. McArthur, L. Chin, Y. A. Wang, R. A. DePinho. Targeting yap-dependent mdsc infiltration impairs tumor progression. Cancer Discovery. Vol. 6, pg. 80–95, 2016 https://doi.org/10.1158/2159-8290.CD-15-0224 [↩]
- H. Salmon, K. Franciszkiewicz, D. Damotte, M.-C. Dieu-Nosjean, P. Validire, A. Trautmann, F. Mami-Chouaib, E. Donnadieu. Matrix architecture defines the preferential localization and migration of t cells into the stroma of human lung tumors. The Journal of Clinical Investigation. Vol. 122, pg. 899–910, 2012 https://doi.org/10.1172/JCI45817 [↩]
- D. E. Kuczek, A. M. H. Larsen, M.-L. Thorseth, M. Carretta, A. Kalvisa, M. S. Siersbæk, A. M. C. Simões, A. Roslind, L. H. Engelholm, E. Noessner, M. Donia, I. M. Svane, P. thor Straten, L. Grøntved, D. H. Madsen. Collagen density regulates the activity of tumor-infiltrating t cells. Journal for ImmunoTherapy of Cancer. Vol. 7, pg. 68, 2019 https://doi.org/10.1186/s40425-019-0556-6 [↩]
- M. J. Pont, T. Hill, G. O. Cole, J. J. Abbott, J. Kelliher, A. I. Salter, M. Hudecek, M. L. Comstock, A. Rajan, B. K. R. Patel, J. M. Voutsinas, Q. Wu, L. Liu, A. J. Cowan, B. L. Wood, D. J. Green, S. R. Riddell. γ-secretase inhibition increases efficacy of bcma-specific chimeric antigen receptor t cells in multiple myeloma. Blood. Vol. 134, pg. 1585–1597, 2019 https://doi.org/10.1182/blood.2019000050 [↩]
- G. Dobra, E. Gyukity-Sebestyén, M. Bukva, M. Harmati, V. Nagy, Z. Szabó, T. Pankotai, Á. Klekner, K. Buzás. MMP-9 as prognostic marker for brain tumours: a comparative study on serum-derived small extracellular vesicles. Cancers. Vol. 15, pg. 712, 2023 https://doi.org/10.3390/cancers15030712 [↩]
- I. Caruana, B. Savoldo, V. Hoyos, G. Weber, H. Liu, E. S. Kim, M. M. Ittmann, D. Marchetti, G. Dotti. Heparanase promotes tumor infiltration and antitumor activity of car-redirected t lymphocytes. Nature Medicine. Vol. 21, pg. 524–529, 2015 https://doi.org/10.1038/nm.3833 [↩]
- M. Palmieri, D. J. S. Sarmento, A. P. Falcão, V. A. O. Martins, T. B. Brandão, K. Morais-Faria, A. C. P. Ribeiro, B. Hasséus, D. Giglio, P. H. Braz-Silva. Frequency and evolution of acute oral complications in patients undergoing radiochemotherapy treatment for head and neck squamous cell carcinoma. Ear, Nose, & Throat Journal. Vol. 100, pg. 449S-455S, 2021 https://doi.org/10.1177/0145561319879245 [↩]
- M.-M. Suarez-Cunqueiro, A. Schramm, R. Schoen, J. Seoane-Lestón, X.-L. Otero-Cepeda, K.-H. Bormann, H. Kokemueller, M. Metzger, P. Diz-Dios, N.-C. Gellrich. Speech and swallowing impairment after treatment for oral and oropharyngeal cancer. Archives of Otolaryngology–Head & Neck Surgery. Vol. 134, pg. 1299–1304, 2008 https://doi.org/10.1001/archotol.134.12.1299 [↩]
- M. Riemann, C. Knipfer, M. Rohde, W. Adler, M. Schuster, E. Noeth, N. Oetter, N. Shams, F.-W. Neukam, F. Stelzle. Oral squamous cell carcinoma of the tongue: prospective and objective speech evaluation of patients undergoing surgical therapy. Head & Neck. Vol. 38, pg. 993–1001, 2016 https://doi.org/10.1002/hed.23994 [↩]
- E. D. Kirson, Z. Gurvich, R. Schneiderman, E. Dekel, A. Itzhaki, Y. Wasserman, R. Schatzberger, Y. Palti. Disruption of cancer cell replication by alternating electric fields. Cancer Research. Vol. 64, pg. 3288–3295, 2004 https://doi.org/10.1158/0008-5472.can-04-0083 [↩] [↩] [↩]
- T. Leal, R. Kotecha, R. Ramlau, L. Zhang, J. Milanowski, M. Cobo, J. Roubec, L. Petruzelka, L. Havel, S. Kalmadi, J. Ward, Z. Andric, T. Berghmans, D. E. Gerber, G. Kloecker, R. Panikkar, J. Aerts, A. Delmonte, M. Pless, R. Greil, C. Rolfo, W. Akerley, M. Eaton, M. Iqbal, C. Langer. Tumor treating fields therapy with standard systemic therapy versus standard systemic therapy alone in metastatic non-small-cell lung cancer following progression on or after platinum-based therapy (lunar): a randomised, open-label, pivotal phase 3 study. The Lancet Oncology. Vol. 24, pg. 1002–1017, 2023 https://doi.org/10.1016/S1470-2045(23)00344-3 [↩]
- L. a. M. J. Crins, N. J. M. Opdam, C. M. Kreulen, E. M. Bronkhorst, B. a. M. M. Sterenborg, M. C. D. N. J. M. Huysmans, B. a. C. Loomans. Randomized controlled trial on the performance of direct and indirect composite restorations in patients with severe tooth wear. Dental Materials: Official Publication of the Academy of Dental Materials. Vol. 37, pg. 1645–1654, 2021 https://doi.org/10.1016/j.dental.2021.08.018 [↩]
- J. Hu, F. Safir, K. Chang, S. Dagli, H. B. Balch, J. M. Abendroth, J. Dixon, P. Moradifar, V. Dolia, M. K. Sahoo, B. A. Pinsky, S. S. Jeffrey, M. Lawrence, J. A. Dionne. Rapid genetic screening with high quality factor metasurfaces. Nature Communications. Vol. 14, pg. 4486, 2023 https://doi.org/10.1038/s41467-023-39721-w [↩]
- E. Catalano. Magnetic biophysical characterization of biomimetic polyeth-ylenimine-coated nanoparticles on in vitro silico model. Preprint at http://arxiv.org/abs/2208.02838 2022 https://doi.org/10.48550/arXiv.2208.02838 [↩]
- O. Yavas, M. Svedendahl, P. Dobosz, V. Sanz, R. Quidant. On-a-chip biosensing based on all-dielectric nanoresonators. Nano Letters. Vol. 17, pg. 4421–4426, 2017 https://doi.org/10.1021/acs.nanolett.7b01518 [↩]
- W.-K. Shin, J. Cho, A. G. Kannan, Y.-S. Lee, D.-W. Kim. Cross-linked composite gel polymer electrolyte using mesoporous methacrylate-functionalized sio2 nanoparticles for lithium-ion polymer batteries. Scientific Reports. Vol. 6, pg. 26332, 2016 https://doi.org/10.1038/srep26332 [↩]
- H. Chen, Z. Yang, C. Ding, L. Chu, Y. Zhang, K. Terry, H. Liu, Q. Shen, J. Zhou. Fragment-based drug design and identification of hjc0123, a novel orally bioavailable stat3 inhibitor for cancer therapy. European Journal of Medicinal Chemistry. Vol. 62, pg. 498–507, 2013 https://doi.org/10.1016/j.ejmech.2013.01.023 [↩]
- M. Shim, W. J. Bang, C. Y. Oh, Y. S. Lee, J. S. Cho. Effectiveness of three different luteinizing hormone-releasing hormone agonists in the chemical castration of patients with prostate cancer: goserelin versus triptorelin versus leuprolide. Investigative and Clinical Urology. Vol. 60, pg. 244–250, 2019 https://doi.org/10.4111/icu.2019.60.4.244 [↩]
- Z. Liu, D. Wang, G. Li, M. Yi, Z. Zhang, G. Zhong, L. Xu, R. Jiang, Y. Zheng, L. Huang, Y. Peng, L. Liang, J. Li, Y. Liu, J. Lai, X. Lv, Y. Xu, Q. Liu, Z. Wang, Z. Liu, Q. Yang, L. Nie, J. Lei, X. Huang, Z. Liu, W. Jiang. Neoadjuvant with low-dose radiotherapy, tislelizumab, albumin-bound paclitaxel, and cisplatin for resectable locally advanced head and neck squamous cell carcinoma: phase ii single-arm trial. Nature Communications. Vol. 16, pg. 4608, 2025 https://doi.org/10.1038/s41467-025-59865-1 [↩]
- R. Kumar, J. Belz, S. Markovic, T. Jadhav, W. Fowle, M. Niedre, R. Cormack, M. G. Makrigiorgos, S. Sridhar. Nanoparticle-based brachytherapy spacers for delivery of localized combined chemoradiation therapy. International Journal of Radiation Oncology, Biology, Physics. Vol. 91, pg. 393–400, 2015 https://doi.org/10.1016/j.ijrobp.2014.10.041 [↩]
- N. Kong, W. Tao, X. Ling, J. Wang, Y. Xiao, S. Shi, X. Ji, A. Shajii, S. T. Gan, N. Y. Kim, D. G. Duda, T. Xie, O. C. Farokhzad, J. Shi. Synthetic mrna nanoparticle-mediated restoration of p53 tumor suppressor sensitizes p53-deficient cancers to mtor inhibition. Science Translational Medicine. Vol. 11, pg. eaaw1565, 2019 https://doi.org/10.1126/scitranslmed.aaw1565 [↩]
- A. Babar, N. M. Woody, A. I. Ghanem, J. Tsai, N. E. Dunlap, M. Schymick, H. Y. Liu, B. B. Burkey, E. D. Lamarre, J. A. Ku, J. Scharpf, B. L. Prendes, N. P. Joshi, J. J. Caudell, F. Siddiqui, S. V. Porceddu, N. Lee, L. Schwartzman, S. A. Koyfman, D. J. Adelstein, J. L. Geiger. Outcomes of post-operative treatment with concurrent chemoradiotherapy (crt) in high-risk resected oral cavity squamous cell carcinoma (ocscc): a multi-institutional collaboration. Current Oncology (Toronto, Ont.). Vol. 28, pg. 2409–2419, 2021 https://doi.org/10.3390/curroncol28040221 [↩] [↩]
- H. Yamazaki, G. Suzuki, N. Aibe, H. Shiomi, R. Oh, K. Yoshida, S. Nakamura, K. Konishi, M. Ogita. Reirradiation for local recurrence of oral, pharyngeal, and laryngeal cancers: a multi-institutional study. Scientific Reports. Vol. 13, pg. 3062, 2023 https://doi.org/10.1038/s41598-023-29459-2 [↩] [↩]
- A. H. Falco. Immunotherapy in advanced head and neck squamous cell carcinoma: a retrospective evaluation in latin america. Biomedical Journal of Scientific & Technical Research. Vol. 17, 2019 https://doi.org/10.26717/BJSTR.2019.17.002983 [↩] [↩]
- H.-M. Liu, X.-P. Xiong, Z.-L. Yu, Z. Shao, G.-L. Chen, Y.-T. Liu, X.-X. Wang, Q.-Y. Fu, X.-X. Cheng, J. Li, J.-L. Zhang, B. Li, H.-Y. Gong, Y.-H. Zhong, W. Zhang, J. Jia, B. Liu, G. Chen. Neoadjuvant immunotherapy with or without chemotherapy in locally advanced oral squamous cell carcinoma: randomized, two-arm, phase 2 trial. Cell Reports. Medicine. Vol. 6, pg. 101930, 2025 https://doi.org/10.1016/j.xcrm.2025.101930 [↩] [↩]
- J. Tímár, A. Ladányi, C. Forster-Horváth, J. Lukits, B. Döme, E. Remenár, M. Godény, M. Kásler, B. Bencsik, G. Répássy, G. Szabó, N. Velich, Z. Suba, J. Elo, Z. Balatoni, K. Pócza, B. Zemplén, P. Chretien, E. Talor. Neoadjuvant immunotherapy of oral squamous cell carcinoma modulates intratumoral cd4/cd8 ratio and tumor microenvironment: a multicenter phase ii clinical trial. Journal of Clinical Oncology: Official Journal of the American Society of Clinical Oncology. Vol. 23, pg. 3421–3432, 2005 https://doi.org/10.1200/JCO.2005.06.005 [↩] [↩]
- N. F. Saba, G. Blumenschein, J. Guigay, L. Licitra, J. Fayette, K. J. Harrington, N. Kiyota, M. L. Gillison, R. L. Ferris, V. Jayaprakash, L. Li, P. Brossart. Nivolumab versus investigator’s choice in patients with recurrent or metastatic squamous cell carcinoma of the head and neck: efficacy and safety in checkmate 141 by age. Oral Oncology. Vol. 96, pg. 7–14, 2019 https://doi.org/10.1016/j.oraloncology.2019.06.017 [↩] [↩]
- N. Gross, R. Ferrarotto, P. Nagarajan, D. Bell, A. El-Naggar, J. M. Johnson, Y. Yuan, B. S. Glisson, M. Wong, D. Rosenthal, B. Esmaeli, M. R. Migden, J. Wargo, R. S. Weber, J. Myers. LBA74 – phase ii study of neoadjuvant cemiplimab prior to surgery in patients with stage iii/iv (m0) cutaneous squamous cell carcinoma of the head and neck (cscc-hn). Annals of Oncology. Vol. 30, pg. v910, 2019 https://doi.org/10.1093/annonc/mdz394.071 [↩] [↩]
- D. K. Zanoni, J. C. Migliacci, B. Xu, N. Katabi, P. H. Montero, I. Ganly, J. P. Shah, R. J. Wong, R. A. Ghossein, S. G. Patel. A proposal to redefine close surgical margins in squamous cell carcinoma of the oral tongue. JAMA Otolaryngology– Head & Neck Surgery. Vol. 143, pg. 555–560, 2017 https://doi.org/10.1001/jamaoto.2016.4238 [↩] [↩]
- G. H. Lyman, A. Nguyen, S. Snyder, M. Gitlin, K. C. Chung. Economic evaluation of chimeric antigen receptor t-cell therapy by site of care among patients with relapsed or refractory large b-cell lymphoma. JAMA Network Open. Vol. 3, pg. e202072, 2020 https://doi.org/10.1001/jamanetworkopen.2020.2072 [↩]