Jonah Sethna1, Andrew Li1
1 Manhasset High School, New York, USA
Abstract
Ultraviolet (UV) radiation-induced oxidative stress is a major risk factor for the development of melanoma, highlighting the need for treatments that prevent UV skin damage from the sun. While antioxidants reduce oxidative stress and promote cell regeneration, the regenerative potential of the antioxidant astaxanthin is unknown. This study investigated the effects of astaxanthin on regeneration of planaria exposed to UV radiation-induced oxidative stress. Three groups of planaria were studied: a UV-exposed group treated with astaxanthin (150µM), a UV-only group in spring water, and a non-irradiated control group in spring water. There were 10 planaria per group and the experiment was replicated three times. UV groups were exposed to UV-A light for 1 hour daily for 7 days. Following amputation below the head, planaria were observed for photoreceptor regeneration, normalized blastema growth area and blastema growth rate. One-Way ANOVA showed that there was a statistically significant difference in regeneration of photoreceptors (p=0.005), blastema growth area (p=0.0004) and blastema growth rate (p=0.0004) among the groups. In post-hoc Tukey analysis, the UV+astaxanthin group had significantly faster blastema growth rate (0.10±0.01 vs. 0.08±0.03 1/day, p=0.02), earlier photoreceptor emergence (4.30±0.74 vs. 5.20±0.77 days, p=0.046), and greater blastema growth (0.81±0.06 vs. 0.70±0.08 mm2, p=0.004) compared to the UV-only group. Findings suggest that astaxanthin may promote regeneration in UV-irradiated planaria, which warrants further research into its potential use for prevention of UV radiation-induced skin damage.
Keywords: Astaxanthin, Melanoma, Planaria, Ultraviolet Radiation
Introduction
According to the American Cancer Society, it is projected that there will be 104,960 new cases of melanoma resulting in 8,430 deaths from this aggressive form of skin cancer in the United States in 20251. Although relatively rare, melanoma accounts for the majority of deaths from all skin cancers. A major risk factor for developing melanoma is excessive exposure to ultraviolet (UV) radiation from the sun and tanning beds2. UV radiation leads to damage of skin cells by inducing oxidative stress, which leads to mutations in DNA and cell damage3. Due to melanoma’s high mortality and its link to UV-induced oxidative stress, there is an urgent need to find potential interventions that protect against such damage. Antioxidants such as astaxanthin are compounds that prevent cell damage caused by free radicals, therefore leading to decreased oxidative stress4. Astaxanthin has shown promise in reducing oxidative stress, making it a candidate for further investigation in melanoma prevention. Planaria flatworms are excellent organisms to study whether astaxanthin affects UV radiation-induced cell damage due to their regenerative properties. There is a significant gap in the present literature as astaxanthin has not been previously tested to reduce UV-induced oxidative stress and aid in regeneration of planaria. Therefore, using planaria as a model organism may further help the understanding of how astaxanthin may mitigate the negative effects of UV radiation, potentially leading to prevention of melanoma and life-saving treatments.
Melanoma is a result of DNA damage, leading to the rapid cell growth and abnormal division of melanocytes in the skin cells of the epidermis5. Melanocytes are cells in the epidermis that produce melanin, the substance that gives skin its color and protects the skin from UV radiation6. Although melanin protects skin by absorbing UV radiation, excessive UV exposure still results in DNA damage5. UV radiation directly leads to damage of DNA by forming dimers of the nucleotide base thymine7. When UV-A radiation (365 nm) in particular interacts with skin cells, it activates unstable reactive oxygen species (ROS) such as superoxide, hydrogen peroxide and hydroxyl radicals8. ROS stimulate oxidative stress, defined as the imbalance of ROS and antioxidants in the system, and cause oxidative damage to the DNA, proteins, and lipids in melanocytes by disrupting their cell membranes9.
Antioxidants have the potential to prevent and fight against the damage caused by oxidative stress that leads to melanoma. Antioxidants are compounds that prevent cell damage from ROS molecules by scavenging and donating an electron, thereby making the molecule stable and reducing the amount of oxidative stress in the body10. Antioxidants are known to have beneficial effects on stem cells and can aid in regeneration of skin and tissue11. Astaxanthin (C40H52O4), a naturally occurring carotenoid found in algae and yeast, is most commonly used as a nutritional supplement. Carotenoids are potent antioxidants that efficiently protect cells against oxidative damage by acting as an electron donor to free radicals. In addition to having anti-inflammatory properties, astaxanthin plays a vital role in protecting the skin against UV radiation-induced oxidative damage. Astaxanthin inhibits apoptosis and reduces ROS production caused by UV radiation, therefore protecting the skin cells. Astaxanthin also accumulates in the dermis layer of the skin and absorbs UV rays, protecting the skin from the sun’s rays. These qualities make astaxanthin an ideal compound to research UV radiation-induced oxidative damage, as it may positively affect stem cell regeneration of the skin that has been damaged12.
Planaria, flatworms classified in the phylum Platyhelminthes, are an effective model to study the effects of antioxidants on regeneration of UV-damaged cells because planaria have the ability to regenerate. Planaria, like humans, contain an outside layer of epidermis. The planarian epidermal layer contains similar characteristics to the human skin and is the first site of protection between the planaria and the environment. Planaria’s importance in scientific experimentation lies in their unique ability to regenerate damaged tissue and organs using their stem cells, called neoblasts13. Neoblasts are pluripotent stem cells that are the source of new cells during planarian regeneration, allowing planaria to produce and divide into all adult cell types when tissue is missing or damaged14. Wound-induced bursts of ROS act as critical signals initiating the regenerative process, and disrupting early ROS signaling impairs brain formation in planaria15. Regenerated tissue that comes from a planarian is called blastema. The stages of planarian regeneration are wound healing, blastema development, growth and differentiation16. The epidermis of the planaria turns over every 4.5 days and is regenerated by neoblasts in the skin17. If a compound such as astaxanthin is effective in remediating UV-induced oxidative stress and increasing regeneration of the planarian epidermal layer, then it may be effective in remediating UV-induced oxidative stress in the human epidermal layer. Therefore, planaria were an ideal model organism to test the antioxidant effects of astaxanthin on regeneration of cells damaged by UV light.
Studies in the literature have shown that astaxanthin reduces oxidative stress and has the potential to have beneficial effects on the planarian regeneration process. Lin et al (2024), worked with Caenorhabditis elegans and highlighted astaxanthin’s protective effects when exposed to UV-B-induced irradiation18. C. elegans were exposed to UV-B light irradiation of 200 mJ/cm2 for 4 days and after 4 days given 25, 50, and 100 μM concentrations of astaxanthin. C. elegans exposed to UV radiation solely (5.67±0.23) experienced a lower mean lifespan than the other groups that were given astaxanthin supplementation. Astaxanthin supplementation of 50 μM (7.76±0.33) showed the highest lifespan followed by the 25 μm group (6.68±0.28) and then the 100 μM group (6.18±0.28). Life was prolonged about 15.70% when C. elegans were given astaxanthin supplementation. The groups given supplementation of astaxanthin showed 5 less ROS intensity units than the sole UV-B irradiation group signaling that supplementation of astaxanthin decreased ROS in C. elegans18. This study exemplified how astaxanthin can protect against the damage caused by UV-B-induced oxidative stress, but the study did not address how astaxanthin affects regeneration.
In another study, Li et al. (2020) examined astaxanthin supplementation for UV-induced photoaging in hairless mice. Experimental groups included mice with UV exposure only, mice with UV exposure followed by astaxanthin supplementation and a control group. Mice were exposed to UV radiation for 8 weeks using a UV lamp at 1 MED intensity. Results showed UV-induced mice had an increase in skin wrinkles by 60% compared to the control group. The experimental group experienced a 50% decrease in skin wrinkles compared to the UV-induced only mice groups. Astaxanthin-treated mice had approximately 45 μM of thinner epidermis tissue (less damage) compared to the mice than were irradiated with UV only. Epidermal thickening from UV-induced oxidative stress exposure was decreased by astaxanthin supplementation with groups showing 80 μm thickness of epidermal tissue, being lower than the control group (125 μm). The astaxanthin group had a lower epidermis tissue thickness by about 35μM to the control group19. This study exemplified how astaxanthin can remediate the effects of UV irradiation in mice epidermis layers, however, there is a gap in the literature as astaxanthin has not been tested on UV-A induced planarian oxidative stress.
To address this gap, this study aimed to examine the effects of astaxanthin on regeneration of planaria exposed to UV radiation-induced oxidative stress. The alternate hypothesis was that exposure to astaxanthin after UV irradiation would be associated with a different photoreceptor regeneration, total blastema growth area, and blastema growth rate in planaria. The null hypothesis was that exposure to astaxanthin after UV irradiation would be associated with the same regeneration. This research addressed a significant gap in the literature by examining the potential of astaxanthin to reduce UV-induced oxidative stress and promote regeneration in planaria, a model organism for studying regeneration. While previous studies have studied the effect of astaxanthin on lifespan and photoaging in different animal models, none have explored regeneration in planaria exposed to UV radiation. The study may contribute to the development of treatments that mitigate damage caused by UV-induced oxidative stress and promote regeneration of cells, potentially preventing diseases such as melanoma.
Methodology
The independent variable of the experiment was the antioxidant astaxanthin. The dependent variables were planarian photoreceptor regeneration time (days), normalized blastema growth area (mm2), and blastema growth rate (1/day). UV-A light was used to induce oxidative stress in the epidermis of planaria. There were three groups in the experiment: 1) UV-exposed group treated with astaxanthin (UV+AST group), 2) UV-only group in spring water (UV group), and 3) non-irradiated control group in spring water (Control group). Immediately after amputation (Day 1), planaria were placed in petri dishes containing either spring water (Control and UV groups) or 150 μM astaxanthin solution (UV+AST group). The UV+AST group and UV group received 1 hour of UV-A irradiation (365 nm, 15 W, 10 inches above dishes) on Days 1-7 during regeneration. The control group received no UV exposure. Astaxanthin treatment for the UV+AST group was continuous throughout the 7-day regeneration period; fresh solution was replaced every 2–3 days. Measurements of photoreceptor formation, normalized blastema area, and blastema growth rate were taken on regeneration days 4–7, when blastema visibility peaks20. Housing conditions, diet, and water temperature variables were kept constant. All groups followed identical schedules and handling. Comparisons were made among the experimental and control groups. There were 10 planaria per group and the experiment was replicated three times. There was no randomization of planaria to the groups.
Planaria (Girardia tigrina)
Planaria (Giradia tigrina) were acquired from Ward’s Science. Planaria were moved individually from the shipping container to a plastic container filled with spring water (Poland Spring) using a pipette. Approximately 10 ml of spring water was used for maintenance. Spring water was maintained at room temperature. The container was loosely covered with the lid and was stored in a dimly lit area in a cabinet. The spring water was changed by preparing another container filled with fresh spring water. Planaria were then transferred to the clean water container using a pipette. The process was repeated every 2-3 days, or more frequently if needed21.
Planaria in the storage containers were fed with a pellet of egg yolk once a week. Prior to feeding, the health of the planaria was observed by looking at moving planaria without dermal lesions. Planaria on the sides of the container were guided to the bottom of the container to ensure that all had a chance to eat. The egg yolk was placed on the bottom of the container, ensuring that it stuck to the bottom. Planaria were allowed to feed for approximately 30 minutes. Feeding was halted for 5 days prior to experimentation. To prevent bacterial growth, the water underwent a triple cleanse after every feeding21.
Amputation
Prior to amputation, planaria were placed on a cool plate from the refrigerator for 3-4 minutes to immobilize them. The refrigerator was set at 5-6 °C. Planaria were amputated immediately below the head and above the pharynx with a scalpel visualized with a dissecting microscope (Fig. 1). Immediately after amputation, healthy planaria were placed in each of the petri dishes using a pipette with the final concentrations of either astaxanthin or spring water. Planarians of about uniform length were chosen (~8mm), however all blastema measurements were normalized at the end to ensure data wasn’t skewed.
Astaxanthin
Astaxanthin liquid gel capsules were purchased from Amazon. An electronic scale (Ohaus model SPX222) and graduated cylinder were used to measure the astaxanthin liquid. Each capsule contained 4 mg of pure astaxanthin. The gel capsules were punctured with a sterile needle and contents were collected using a micropipette. Using astaxanthin’s molecular weight of approximately 596.84 g/mol, 4 mg of astaxanthin were diluted in 10 ml of spring water in plastic petri dishes to achieve a solution of 670 μM. To obtain the desired concentration of approximately 150 μM, the stock solution was diluted in a 1:4 ratio with spring water. The total capsule volume was not used in calculations, as molarity was determined solely on active astaxanthin and final bath volume. This concentration aligned with the other antioxidant studies on planaria with concentrations around 150 μM22. To ensure that the astaxanthin concentration was safe for planaria, three planaria were placed in astaxanthin and were observed daily for one week. If any of the planaria died, the concentration of the astaxanthin was lowered, and the experiment was repeated until a safe concentration of astaxanthin was confirmed.
Ultraviolet Radiation Exposure
The experimental groups were exposed to 1 hour of UV-A radiation in petri dishes without lids. The control group was not exposed to UV light. UV-A radiation was sourced from a 15 watt UV-A bulb in a lightbox at approximately 365 nm, a wavelength shown to cause photophobic responses in planaria, confirming its biological relevance to their sensory physiology. It was placed at the same distance of 10 inches from the planaria in petri dishes (Fig. 2). UV irradiance was confirmed by using a UV light meter (Fig. 3). Planaria were returned to their cabinet after exposure. This procedure was repeated for seven days. The daily dose of irradiance was 2.20 J/cm^2. Distance and dose values were consistent throughout. To ensure that the duration of 1 hour was safe for planaria, three planaria exposed to UV were observed daily for one week. Some changes to the epidermal layer of the planaria were expected, but if any of the planaria showed signs of being unhealthy (severe dermal injury or decreased movement) or died, the UV light exposure was lowered, and the experiment was repeated until a safe time was confirmed. The lightbox was placed in an area where others wouldn’t be exposed to potential leaks. The lightbox was only operated when fully closed. To prevent eye damage, UV-A certified glasses were worn during experimentation23. The exposure schedule of 1 hour per day for 7 days was selected based on prior planarian oxidative stress studies that used multi-day UV exposure to induce damage without lethality shorter exposures of less than 30 minutes have been shown to produce minimal regenerative delay, whereas daily repeated UV-A doses show chronic oxidative stress. This duration thus represents a controlled yet relevant UV dosage for assessing antioxidant protection24.
Regeneration Time of Photoreceptors
The time in days for the development of the photoreceptors after amputation was measured. Photoreceptors are black dots located inside pale round eyes that help the planaria to see (Fig. 4). Planaria were observed daily for full restoration of photoreceptor structures (i.e. bilateral pigmented cups visible) under the dissecting microscope.
Normalized Total Blastema Growth Area
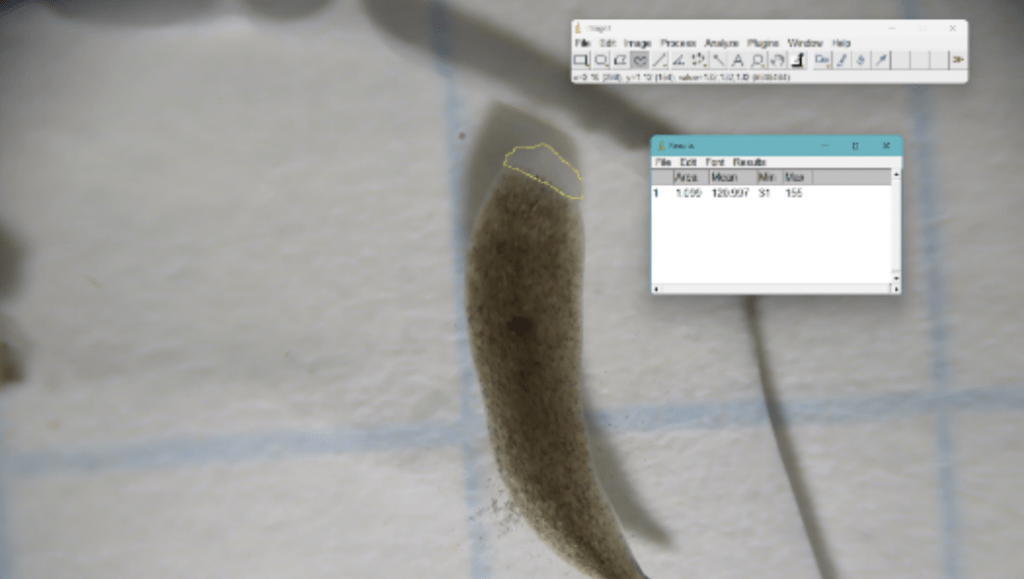
ImageJ software was used to measure the area of regeneration 7 days after amputation in all groups. Individuals using ImageJ were not blinded to group. Day 7 of regeneration was chosen as it is the point of peak blastema visibility25. 1 cm by 1 cm graph paper was used for scale. Using the software, photographs were uploaded, and the images were calibrated to scale (cm). The freehand selection tool was used to outline the regenerated area. The measure function was used to quantify the area (mm2) of regeneration. (Refer to Fig. 5.) Normalized blastema growth was calculated via Equation 125.
(1) ![]()
Normalized Blastema Growth Rate
Normalized blastema areas from days 4-7 were measured. Blastema areas were plotted on graphs in order to obtain average slopes. Slopes were averaged in order to find normalized blastema growth rate (1/day) from days 4-7, the main days of regeneration.
Statistical Analysis
Means and standard deviations were calculated for photoreceptor regeneration time, normalized blastema growth area and blastema growth rate. Bar graphs using error bars were created using Microsoft Excel to help determine outliers and to visually compare the dependent variables between the groups. Error bars visually signified standard deviation. One-way ANOVA was used to compare the means of the three different groups for individual level data. Post-hoc Tukey tests were used to determine where the differences lay between groups. Data were analyzed with IBM SPSS v.28.
Results
Regeneration Time of Photoreceptors
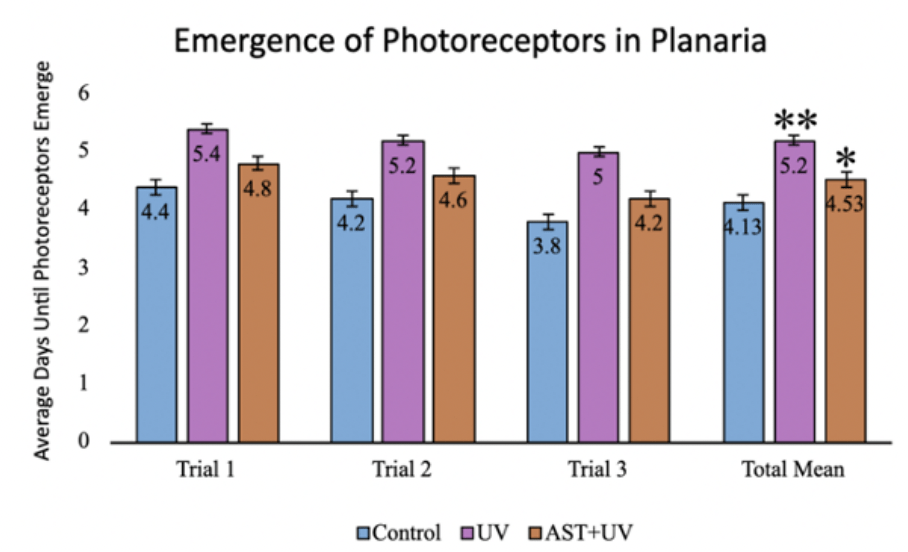
Photoreceptor emergence times (mean±standard deviation) were 4.13±1.12 days for the control group, 5.20±0.77 days for the UV group, and 4.53±0.74 days for the UV+AST group (Fig. 6). One-Way ANOVA demonstrated that the mean regeneration days of control, UV-only, and UV+AST were significantly different across groups, F=6.12, Eta squared=0.23 CI 0.03-0.39, df=2, p=0.005. Using post-hoc Tukey tests, the UV+AST group had a shorter regeneration time for photoreceptor emergence compared to the UV-only group (p=0.04, mean difference -0.8 CI -1.57—0.03). The UV-only group had impaired regeneration, as evidenced by a 1.07 day delay in photoreceptor regeneration compared to the control group. Treatment with astaxanthin mitigated these negative effects. Photoreceptor regeneration was 0.67 days faster in the UV+AST compared to the UV-only group.
Figure 6. The effect of astaxanthin on the UV-irradiated planarian emergence of photoreceptors. (control, UV, astaxanthin + UV). There were 10 planaria per group and the experiment was replicated three times. Error bars = ±1 SD. Asterisks (*) denote significance in comparison to the control group. * signals p-value <0.05, ** signals p-value <0.01. (Graph by authors, 2025)
Normalized Blastema Growth Area
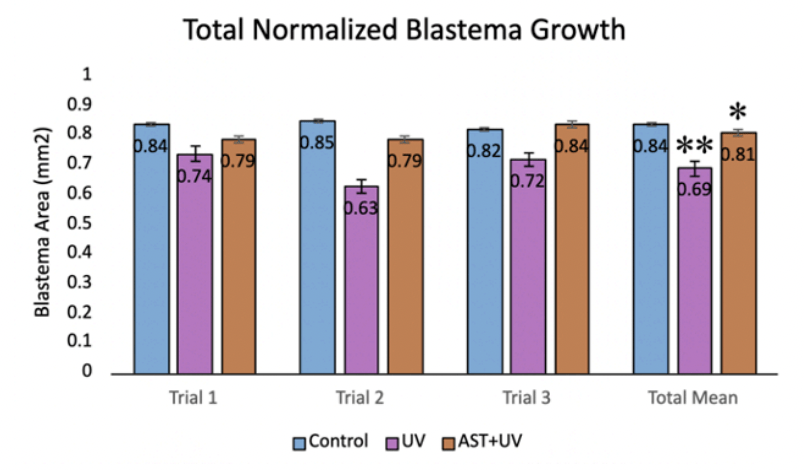
Normalized blastema growth area (mean±standard deviation) was 0.84±0.12 mm2 for the control group, 0.700±0.08 mm2 days for the UV group, and 0.81±0.06 mm2 for the UV+AST group (Fig. 7). One-Way ANOVA revealed that the mean total blastema area for the control group, UV-only group, and UV+AST group were significantly different across groups, F=9.87, df=2, Eta squared=0.32 (0.9-0.49) ,p<0.001. Using post-hoc Tukey tests, the UV-A+AST group had greater normalized blastema area compared to the UV-only group (mean difference 0.11 CI 0.03-0.19, p=0.004). The UV-only group had 17.9% lower blastema growth area compared to the control group, suggesting that UV exposure impaired regeneration. Treatment with astaxanthin mitigated these negative effects. Blastema growth was 17.3% more in the UV+AST compared to the UV-only group.
Normalized Blastema Growth Rate
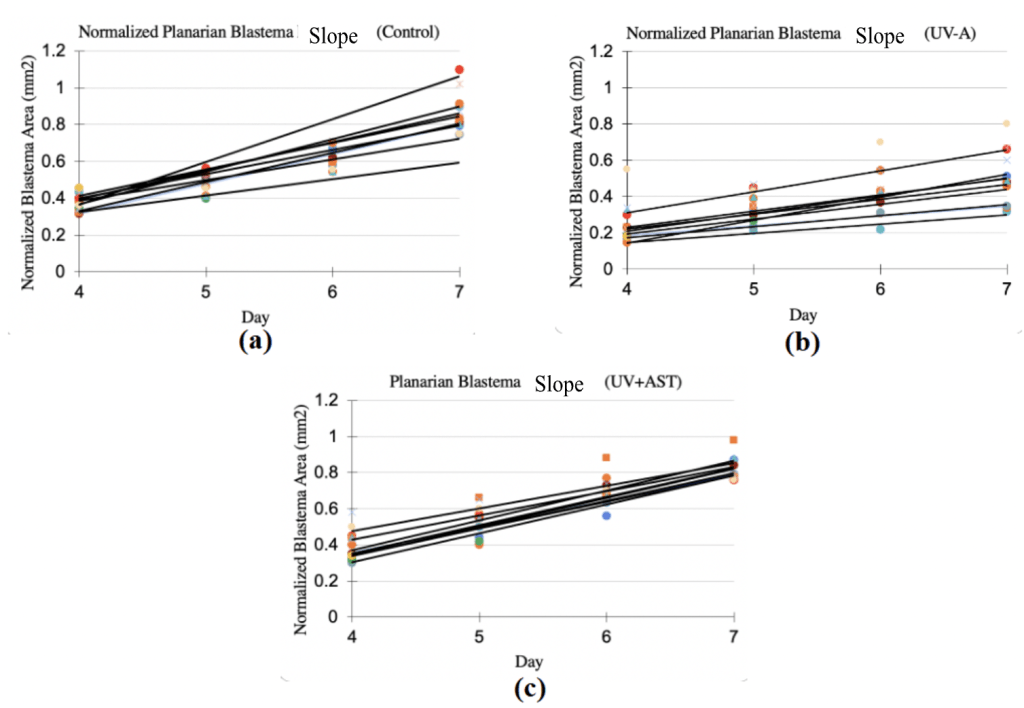
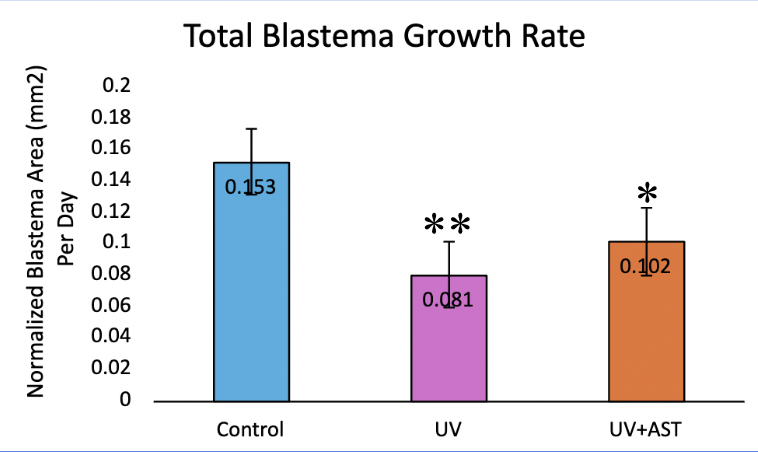
Normalized blastema growth rate (mean±standard deviation) was 0.15±0.03 1/day for the control group, 0.08±0.02 1/day for the UV group, and 0.10±0.011/day for the UV+AST group (Fig. 8). One-Way ANOVA revealed that the mean blastema growth rate for control group, UV-only group, and UV+AST group were significantly different across groups, F=8.1, df=2, Eta squared=0.28 CI 0.6-0.45 ,p=0.001. Post-hoc tests showed that the UV+AST group had a significantly faster growth rate than the UV-only group (mean difference 0.5 CI 0.02-0.08, p = 0.02). The UV-only group had a 47% slower blastema growth rate compared to the control group. Treatment with astaxanthin mitigated these negative effects. The blastema growth rate was 26% faster in the UV+AST group compared to the UV-only group. The slope of the growth rate over 4 days is shown in Fig. 8 (A-C). The UV-only group had a lower slope of growth rate compared to the AST+UV and control groups (Fig. 9).
DIiscussion
UV exposure significantly impaired planarian regeneration, evidenced by delayed photoreceptor formation, reduced blastema size, and slower regeneration rate. Notably, astaxanthin supplementation partially mitigated these effects across all measured parameters, showing clear improvement relative to the UV-only group but not a full return to control levels. For instance, blastema growth rate in the UV + AST group increased by ~26% compared to UV-only but remained approximately one-third below control. This partial rescue signals a protective, rather than completely restorative, antioxidant effect. The hypothesis is supported that astaxanthins antioxidant properties protect against UV-induced oxidative damage. These data rejected the null hypothesis that astaxanthin has no effect on regeneration in planaria exposed to UV light. Although direct ROS studies were not performed in this experiment, results of this study suggest that astaxanthin may reduce oxidative stress through its antioxidant properties, hence promoting tissue regeneration. These findings reinforce the potential role of astaxanthin in promoting tissue repair under oxidative stress.
The impaired regeneration observed in UV-exposed planaria is speculated to be mediated by oxidative stress (Fig. 6). UV-A radiation generates ROS, including superoxide, hydrogen peroxide, and hydroxyl radicals, that damage cellular DNA, proteins, and lipids25. These results are in contrast with the findings of Kalafatić et al. (2006), who reported that planaria exposed to UV light exhibited skin lesions, behavior changes, and slower motility, but had similar regeneration times as controls26. This difference in regeneration may be explained by the longer UV light exposure time in the present study (1 hour) compared to 20-30 minutes in the study by Kalafatić et al. The longer UV exposure time in this study could have caused more cell damage, leading to impaired regeneration, while the shorter times used in the study by Kalafatić et al. may not have been enough to impact regeneration. The methods of regeneration measurement were also not clearly outlined in study by Kalafatić et al, which makes comparisons between studies difficult23.
Astaxanthin effectively reversed UV-induced deficits, likely due to its potent antioxidant activity and membrane-stabilizing effects. The finding that astaxanthin promotes regeneration in planaria exposed to UV light was expected and was similar to previous studies demonstrating the beneficial effects of antioxidants on tissue repair. A study from Tsarkova et al. (2023) found that the antioxidant tameron protected stem cells from oxidative stress damage, leading to improved regeneration in planaria22. Another study by Ermakov et al. (2021) demonstrated that the antioxidant N-acetylcysteine led to greater blastema growth rate in planaria27.
There are several proposed mechanisms by which astaxanthin may exert its antioxidant properties to improve planarian regeneration. Although ROS was not studied in the present study, it is postulated that astaxanthin scavenged ROS and inhibited their production, thus mitigating the negative effects of UV exposure and allowing for greater proliferation of stem cells (Fig. 10). With greater stem cell proliferation, regeneration would be accelerated. Astaxanthin’s chemical structure with double bonds and terminal rings enables it to donate electrons to unstable ROS, neutralizing them. Since it is fat soluble, astaxanthin’s structure also protects the entire cell membrane28. Astaxanthin may also exert its antioxidant effects by influencing key cell signaling pathways affected by oxidative stress such as Wnt which plays a role in cellular differentiation and MAPK which is involved in cell growth29,30.
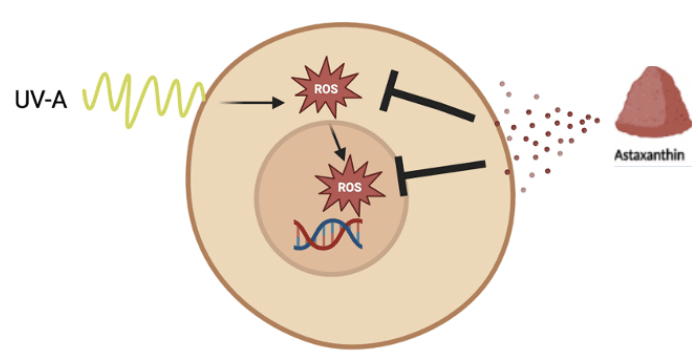
Additionally, astaxanthin may protect mitochondria from the effects of oxidative stress, which promotes longevity. In general, astaxanthin has more potent antioxidant activity compared to other well-known antioxidants such as Vitamin C, Vitamin E and beta carotene31. The study results showed that there is potential for astaxanthin to decrease oxidative stress from UV-A and may have broader implications for preventing UV damage to human skin and melanoma, which requires further research.
There were some limitations of the study to consider. There was no control group of astaxanthin only treatment to determine baseline effects of astaxanthin on regeneration. Additionally, planaria were not randomized to the groups. While effort was made to amputate the planarian just below the head in a uniform manner, there may have been variations due to the difficulty in precisely cutting the small organisms. This may have led to variability in the starting amount of tissue among planarians, making the regeneration process quicker in some planarians. Similarly, although planaria used in the experiment were chosen to be roughly around the same size, variation in the size of planaria could have introduced another potential confounder as bigger planarians generally would have more total tissue than smaller ones. However, the potential bias of these variations was likely mitigated by assigning the planaria across the different treatment groups. There may have also been observer bias when measuring the planaria growth, which was a limitation. To address this, blastema growth rate was normalized using a formula to deny the bias. Another potential source of bias was the variability in health of the planaria prior to the experiment. In order to address this, the health of planaria was observed and those with decreased movement were not included in the experiment. In addition, there may have been some degradation of astaxanthin with exposure to UV light, but the expected loss is estimated to be less than 20%.
A further limitation was that this study did not utilize an animal model of melanoma. There is currently no planarian model of melanoma, but planaria are ideal invertebrate animals for studying tissue regeneration. Finally, results may not translate to humans, as human skin has different anatomy and physiology compared to planarians. Further research using mammalian models is needed to validate the benefits of astaxanthin under oxidative stressors and to explore molecular pathways involved in its antioxidant action. Nevertheless, this study supports that astaxanthin may play a protective role against UV-induced damage in regeneration of planaria. Understanding regeneration in planaria may help provide insights into human skin, potentially leading to future treatments for melanoma.
Conclusion
In this study, planaria exposed to UV radiation showed impaired regeneration and the antioxidant astaxanthin mitigated these negative effects. Overall, the study findings rejected the null hypothesis, as expected. These results highlight the potential for astaxanthin as a preventative therapy against skin damage caused by UV light. While prior literature has shown benefits of the antioxidant properties of astaxanthin for improving lifespan and photoaging in other animal models, this study uniquely addressed a gap in our understanding of astaxanthin’s role in blastema growth in planaria exposed to UV light. Although planaria are not a direct model of human skin or melanoma, their regenerative properties make them an ideal organism for studying tissue repair. If confirmed in clinical trials in humans, astaxanthin could be a promising treatment to protect skin from UV damage and reduce the risk of melanoma.
Acknowledgments
We would like to express our sincere gratitude to our mentors, Ms. Melanie Greenwald and Ms. Alison Huenger, for their guidance, support, and encouragement throughout the course of this research.
References
- American Cancer Society. Cancer Facts and Figures 2025. Atlanta: American Cancer Society; 2025. [↩]
- The Skin Cancer Foundation. 2024. Tanning – the Skin Cancer foundation.https://www.skincancer.org/riskfactors/tanning/#:~:text=YES.,your%20risk%. [↩]
- Remigante, A., & Morabito, R. 2023. Cellular and Molecular Mechanisms in Oxidative Stress-Related Diseases 2.0/3.0. International journal of molecular sciences, 24(21), 16018. https://doi.org/10.3390/ijms242116018. [↩]
- Chung, B. Y., Park, S. H., Yun, S. Y., Yu, D. S., & Lee, Y. B. 2022. Astaxanthin Protects Ultraviolet B-Induced Oxidative Stress and Apoptosis in Human Keratinocytes via Intrinsic Apoptotic Pathway. Annals of Dermatology, 34(2), 125–131. [↩]
- Schaffer, M., Schaffer, P., & Boehm, M. 2019. Melanoma development and progression: The genetic perspective. Melanoma Research, 29(1), 1-13. [↩] [↩]
- Brenner, M., & Hearing, V. J. 2008. The protective role of melanin against UV damage in human skin. Photochemistry and Photobiology, 84(3), 539-549. [↩]
- Pfeifer, G. P. 2017. UV-induced DNA damage and DNA repair in skin. Cold Spring Harbor Perspectives in Medicine, 7(9), a026079. [↩]
- Cui, Y. Z., & Man, X. Y. 2023. Biology of melanocytes in mammals. Frontiers in Cell and Developmental Biology, 11, 1309557. [↩]
- Sies, H. 2015. Oxidative stress: A concept in redox biology and medicine. Free Radical Biology and Medicine, 89, 748-751. [↩]
- Halliwell, B., & Gutteridge, J. M. C. 2015. Free Radicals in Biology and Medicine. Oxford University Press. [↩]
- D’Orazio, J., Jarrett, S., Amaro-Ortiz, A., & Scott, T. 2013. UV radiation and the skin. International journal of molecular sciences, 14(6), 12222–12248. [↩]
- Ambati, R. R., et al. 2014. Astaxanthin: Sources, extraction, stability, biological activities and its commercial applications—A review. Marine Drugs, 12(1), 128-152. [↩]
- Reddien, P. W. 2018. The Cellular and Molecular Basis for Planarian Regeneration. Cell, 175(2), 327-340. [↩]
- Wagner, D. E., Ho, J., & Reddien, P. W. 2011. A consensus gene expression atlas of the planarian Schmidtea mediterranea. Developmental Cell, 21(3), 596-608. [↩]
- Pirotte, M., De Mulder, K., Davies, L., Rillaerts, T., Seuntjens, E., & Smeets, L. 2015. Wound-induced bursts of ROS act as critical signals initiating the regenerative process and disrupting early ROS signaling impairs brain formation in planaria. Development, 142(9),1618-1630. [↩]
- Shaban, S., El-Husseny, M. W. A., Abushouk, A. I., Salem, A. M. A., Mamdouh, M., & Abdel-Daim, M. M. 2017. Effects of antioxidant supplements on the survival and differentiation of stem cells. Oxidative Medicine and Cellular Longevity, 2017, 1–16. [↩]
- Petersen, C. P., & Reddien, P. W. 2009. A wound-induced Wnt expression program controls planarian regeneration polarity. Proceedings of the National Academy of Sciences, 106(40), 17061-17066. [↩]
- Lin, X., Shao, C., Elsherbiny, S. M., & Huang, Q. 2024. Astaxanthin attenuates UV‐irradiation aging process via activating JNK‐1/DAF‐16 in Caenorhabditis elegans. Photochemistry and Photobiology, 101(1), 133-146. [↩] [↩]
- Li, X., Matsumoto, T., Takuwa, M., Saeed Ebrahim Shaiku Ali, M., Hirabashi, T., Kondo, H., & Fujino, H. 2020. Protective Effects of Astaxanthin Supplementation against Ultraviolet-Induced Photoaging in Hairless Mice. Biomedicines, 8(2), 18. [↩]
- Oviedo, N. J., Nicolas, C. L., Adams, D. S., & Levin, M. 2010. Live imaging of planarian stem cell behavior during regeneration. Nature Protocols, 5(11), 1734-1748. [↩]
- Dean, M. R. P., & Duncan, E. M. 2020. Laboratory maintenance and propagation of freshwater planarians. Current Protocols in Microbiology, 59(1). https://doi.org/10.1002/cpmc.120. [↩] [↩]
- Tsarkova, Е. А., Filippova, K., Afanasyeva, V., Ermakova, O., Kolotova, A. A., Blagodatski, A., & Ermakov, A. M. 2023. A Study on the Planarian Model Confirms the Antioxidant Properties of Tameron against X-ray- and Menadione-Induced Oxidative Stress. Antioxidants, 12(4), 953. [↩] [↩]
- Kalafatić, M., Kovačević, G., & Franjević, D. 2006. Resistance of two planarian species to UV-irradiation. Folia Biologica, 54(3), 103–108. [↩] [↩]
- Birkholz, D. A., & Beane, W. S. 2017. A role for UV-A light in the regulation of planarian regeneration. PLoS ONE, 12(11), e0187213. [↩]
- Campillo, N., Ireland, D., Patel, Y., & Collins, E. S. 2023. A simple method for quantifying blastema growth in regenerating planarians. Current Protocols, 3(3). [↩] [↩] [↩]
- Kalafatić, M., Kovačević, G., & Franjević, D. (2006). Resistance of two planarian species to UV-irradiation. Folia Biologica, 54(3), 103–108. [↩]
- Ermakov, A. M., Kamenskikh, K. A., Ermakova, O. N., Blagodatsky, A. S., Popov, A. L., & Ivanov, V. K. 2021. Planarians as an In Vivo Experimental Model for the Study of New Radioprotective Substances. Antioxidants (Basel, Switzerland), 10(11), 1763. [↩]
- Bjørklund, G., Gasmi, A., Lenchyk, L., Shanaida, M., Zafar, S., Mujawdiya, P. K., Lysiuk, R., Antonyak, H., Noor, S., Akram, M., Smetanina, K., Piscopo, S., Upyr, T., & Peana, M. 2022. The Role of Astaxanthin as a Nutraceutical in Health and Age-Related Conditions. Molecules 27(21), 7167. [↩]
- Kim, S. H., & Kim, H. 2019. Astaxanthin Modulation of Signaling Pathways That Regulate Autophagy. Marine drugs, 17(10), 546. [↩]
- Zhu, C., Liu, G., Cui, W., Yu, Z., Chen, W., Qin, Y., Liu, J., Lu, Y., Fan, W., & Liang, W. 2023. Astaxanthin prevents osteoarthritis by blocking Rspo2-mediated Wnt/β-catenin signaling in chondrocytes and abolishing Rspo2-related inflammatory factors in macrophages. Aging, 15(12), 5775–5797. [↩]
- Hama, S., et al. 2020. Comparison of astaxanthin and other carotenoids in anti-oxidative activity using electron spin resonance and cell assay. Journal of Clinical Biochemistry and Nutrition, 67(1), 89-94. [↩]