Abstract
Radiative cooling paints can reduce surface temperatures by reflecting incoming solar radiation while emitting thermal radiation through the 8–13 μm atmospheric window. Many high-performance radiative-cooling coatings rely on engineered microstructures or specialised manufacturing processes, which can limit accessibility. This study investigates whether a low-cost, widely available additive, calcium carbonate (CaCO₃), can enhance the cooling performance of a commercial white paint under outdoor conditions. Two CaCO₃-enhanced formulations, 45% and 65% CaCO₃ by mass, were prepared and applied to aluminium panels. The 65% formulation was applied using three different methods, drip, brush, and roller, to examine whether application technique influenced thermal performance. A commercial white paint without CaCO₃ served as the primary control, and an uncoated aluminium panel provided a secondary comparison. All panels were placed side by side outdoors and exposed simultaneously to the same ambient conditions. Surface temperatures were measured at regular intervals using an infrared thermometer, and temperature differences relative to the regular white paint were analysed over three time windows: the first 300 seconds, the first hour, and extended durations. Data were collected over two weeks of outdoor testing (n = 1 panel per condition per week). The uncoated aluminium panel consistently reached the highest temperatures, peaking at approximately 10–12 °C above the regular white paint during periods of strongest heating. In contrast, CaCO₃-containing panels remained close to the control, typically within ±1 °C. No consistent separation was observed between the 45% and 65% formulations, and differences among application methods were minimal. Overall, under the outdoor conditions tested, incorporating CaCO₃ into a high-quality commercial white paint produced only modest thermal differences relative to the base formulation.
Keywords: passive radiative cooling; calcium carbonate; reflective coatings; solar reflectance; emissivity; infrared thermometry; aluminium panels
Introduction
Buildings absorb and retain significant amounts of solar heat during the day, causing indoor spaces to heat up considerably. This is especially an issue in regions that are rapidly urbanizing. To address this, people rely on energy-intensive appliances such as air conditioners, leading to higher electricity consumption and increased emissions1,2.
Passive radiative cooling is a process in which a surface maintains a lower temperature by controlling how it absorbs sunlight and emits heat. To remain cool during the day, a material must reflect most incoming solar radiation (including visible and near-infrared wavelengths). Solar energy spans roughly the ultraviolet through near-infrared region and extends well beyond visible light3,4. At the same time, the material must emit its thermal energy efficiently as infrared radiation, particularly within the 8–13 μm atmospheric window, where thermal radiation can escape through the atmosphere with relatively low absorption. When a surface combines high solar reflectance with strong thermal emission in this window, it can lose more heat than it gains and may approach (or under ideal conditions, fall below) ambient air temperature5,6.
White paint has high reflectance in the visible spectrum and is among the most effective common colors for reducing solar heating. However, “white” does not automatically mean high reflectance across the full solar spectrum, and commercial formulations vary. Paint-based radiative-cooling work has shown that composition (pigments/fillers/binders) strongly affects solar reflectance and thermal emissivity, which can limit the gains from simply adding one additional filler4,7.
Calcium carbonate (CaCO₃) is widely available, low-cost, and commonly used as a filler (e.g., chalk/limestone). CaCO₃ particles can increase scattering of incoming light; scattering is strongest when particle size is comparable to the wavelength (Mie-type scattering behaviour), which is why engineered particle distributions are used in radiative-cooling coatings8,9,10,11. For particles in the sub-micron to micron range, Mie scattering dominates when particle diameter d ≈ λ (where λ is the relevant wavelength of solar radiation, approximately 0.3–2.5 μm); Rayleigh scattering prevails for much smaller particles and is less efficient per unit mass. The CaCO₃ powder used in this study did not have engineered particle size or distribution, which reduces the ability to predict or interpret its scattering efficiency compared to studies with engineered coatings12,13.
Prior work on paint-based radiative cooling has taken place across three broad areas: high-performance engineered coatings, scalable low-cost formulations, and the optical properties of paint pigments and fillers. Landmark demonstrations by Raman et al.5 and Zhai et al.14 established that passive daytime sub-ambient cooling is achievable outdoors, motivating the development of more scalable approaches. Mandal et al.15 showed that hierarchically porous polymer coatings can achieve approximately 96 W/m² cooling power through Mie scattering from air voids alone, without particle fillers. Akbari et al.7 documented that, at the building scale, high-reflectance roof coatings reduce peak roof surface temperatures by 33–42 K and produce measurable reductions in cooling energy demand, leading to the practical motivation for developing improved white paints.
The work that is most relevant to what has been done in this study would be that of Li et al.11, who looked at low-cost, paint-based approaches to cooling. They showed that a CaCO₃-acrylic paint at 60 vol% filler achieves 95.5% solar reflectance and field-measured surface temperatures more than 1.7 °C below ambient at noon. Li et al.16 also showed that BaSO₄-acrylic composites with a broad particle-size distribution can reach 98.1% solar reflectance. Tao et al.9 and Lim et al.10 further establish CaCO₃ as a viable high-solar-reflectance filler: Tao et al. demonstrated that PVC/CaCO₃ composites can outperform PVC/TiO₂ in outdoor temperature tests, and Lim et al. showed that CaCO₃ microparticle-based devices without metal reflectors achieve 93.1 W/m² cooling power. Taken together, these studies confirm that CaCO₃ is a physically relevant scattering filler for radiative cooling; however, all high-performing implementations characterised particle-size distributions and controlled coating thickness, which the present study does not.
The optical performance of white paint depends on the scattering efficiency of its pigments, which is governed by Mie theory. Levinson et al.17 characterised the solar spectral scattering and absorption coefficients of TiO₂ and other common pigments, showing that commercial white paints have limited near-infrared reflectance despite high visible-range backscattering. Auger et al.12 demonstrated that dependent scattering between TiO₂ pigments is strongly affected by spatial dispersion and volume fraction, while Vargas et al.13 applied four-flux radiative transfer modelling to predict paint reflectance from Mie-theory parameters. These results explain why simply adding a CaCO₃ extender filler to an already optimised commercial white paint (without particle-size selection or thickness control) is likely to yield only modest improvements over the baseline formulation, as the present study confirms.
This study investigates whether adding CaCO₃ to commercial white paint measurably improves outdoor cooling performance using a low-cost method: coating aluminium panels and monitoring temperature trends outdoors. This approach is simpler than many lab studies that use engineered microstructures or tightly controlled optical measurements6,15.
This study hypothesized that incorporating CaCO₃ into white paint could reduce the surface temperature of aluminium panels exposed to sunlight relative to uncoated panels and, potentially, relative to panels coated with regular white paint. A secondary hypothesis was that higher CaCO₃ concentration would yield greater cooling (lower surface temperatures).
To test this, two CaCO₃-enhanced paints were prepared at 45% and 65% CaCO₃ by mass. The 65% formulation was applied using three application methods (drip, brush, roller) to test whether the application method affected performance. The 45% formulation and regular paint were applied using the drip method, and an uncoated panel served as the control. Surface temperatures were recorded throughout the day using an infrared thermometer. This work focuses on comparative surface-temperature behavior under outdoor conditions and does not independently characterize optical reflectance, emissivity, or coating microstructure.
The objective of this study is not to optimize radiative cooling performance, but to evaluate whether the addition of calcium carbonate produces a detectable comparative thermal effect relative to a high-quality commercial white paint under realistic outdoor conditions.
Methods
Materials
Commercial outdoor white paint, calcium carbonate (CaCO₃) powder, an infrared thermometer, six aluminium panels, distilled water, a weighing balance, beakers, and a stirring spoon were used.
Six aluminium panels were used as test surfaces. Aluminium panels were selected due to their high thermal conductivity and consistent surface properties, allowing for a clearer comparison across different coating types. The final experiment consisted of six panel types: an uncoated plain panel; a panel coated with regular commercial white outdoor paint (drip method); a panel with 45% CaCO₃ by mass (drip method); and panels with 65% CaCO₃ by mass applied by the drip, brush, and roller methods, respectively.
Two formulations of CaCO₃-enhanced paint were prepared at two concentrations by mass: 45% and 65%. Mass fraction was defined as:
![]()
where w% is the target CaCO₃ mass fraction, mCaCO3 is the mass of CaCO₃ powder, and mpaint is the mass of base paint.
To reduce clumping during mixing, the CaCO₃ powder was pre-wetted using small amounts of distilled water to form a smooth paste/slurry before addition to the paint. The paste was then added slowly to the paint while stirring continuously. The sides of the beaker were scraped during mixing to ensure the incorporation of powder adhered to the container walls. After mixing, the paint was rested briefly to allow bubbles to rise and then stirred again immediately before application. Visual inspection was used to confirm that the mixture appeared uniform (no visible dry powder pockets or large agglomerates). The CaCO₃ powder used was Calcium Carbonate Extrapure 98% (Sisco Research Laboratories, Mumbai, India; product code 67162; CAS 471-34-1; ≥98% purity). No particle size or d50 specification is provided on the manufacturer’s product datasheet for this grade; the scattering regime of the powder therefore remains uncharacterised. Mixing was performed by hand using a glass stirring rod for approximately five minutes per batch until no visible dry powder remained. 100 g of distilled water was used in total across both batches, divided in proportion to the mass of CaCO₃ powder in each batch to achieve a paste consistency comparable to the consistency before the addition of CaCO₃ to the paint.
Two separate batches were prepared: one for the 45% formulation and one for the 65% formulation. The 65% formulation was used for multiple application-method panels (drip, brush, roller).
Application methods
At first, the paint was applied using a brush. However, this approach resulted in uneven streaks and made it difficult to maintain a single, smooth layer of paint. This led to testing different application techniques to determine whether the method of application had any influence on the coating’s cooling capability. For final testing, three methods were selected: the drip method, brush application, and roller application.
Drip method: A wide tray was placed on newspaper, with a cooking tray positioned on top of it. Holding the aluminium panel at an angle, paint was poured and allowed to drip across the surface. The panel was gently tilted to distribute the paint evenly using gravity. Once most of the surface was coated, the panel was placed upright, supported against a bottle, so that excess paint could drip off into the tray below.
Brush application: A sufficient quantity of paint was poured into a tray. The brush was dipped fully into the paint and then drawn across the panel in single-direction strokes. An effort was made to minimise overlapping and maintain consistent pressure, aiming to achieve as uniform a layer as possible.
Roller application: A sufficient amount of paint was poured into a tray, and a roller was coated by rolling it through the paint. The roller was then moved gently across the panel in one direction, avoiding back-and-forth motions. Due to the roller’s soft and porous texture, only a limited amount of paint adhered to the surface, resulting in a thinner layer compared to the other methods.
Coating thickness was not measured quantitatively and may vary between panels and application methods. It should be noted that coating thickness is a critical factor in radiative cooling performance, as it affects both solar reflectance and thermal emissivity; the inability to control or measure this variable limits the interpretability of comparative results between panels and application methods.
Temperature measurements
All panels were placed side by side on a cloth sheet on an open terrace. Testing was done simultaneously to reduce variability due to environmental conditions and to ensure that all panels received similar sunlight exposure during measurement. Panels remained in the same arrangement throughout the test.
The surface temperature of the panels was measured at regular intervals to observe temperature trends over time. Temperature measurements were used to compare relative trends between panels tested simultaneously, rather than to determine absolute surface temperatures. Using an infrared thermometer, the temperature at the centre of each panel was measured and recorded; as a result, readings depend on surface emissivity and are less reliable for bare aluminium than for painted surfaces. Measuring the centre point was used to maintain consistency and reduce variation due to potential edge effects. It should be noted that without emissivity correction, IR thermometer readings cannot unambiguously separate true surface temperature changes from apparent temperature changes caused by differences in surface emissivity between coatings. Future iterations of this experiment should use contact thermocouples bonded to the rear face of each panel to obtain emissivity-independent temperature data.
Recording began at 11:30 am and continued until 11:00 pm, to capture peak sunlight hours and extend into nighttime conditions with minimal to zero sunlight. Readings were taken every 20 seconds for the first five minutes, every five minutes from five minutes to one hour, and every hour thereafter.
The independent variable was the type of panel surface/coating, and the dependent variable was the measured surface temperature.
All readings were initially recorded in tabular format on paper and then transferred to Excel for organization and inspection of trends. For analysis and plotting, temperature series were compared across panel types. In addition to plotting absolute temperature vs time, a differential temperature metric was computed relative to the regular paint panel (Normal), defined as the difference between the painted panel and ‘normal’ panel temperature.
To reflect different heating/cooling regimes visible in the time series, plots were segmented into three phases: the first 300 seconds, the first hour, and the remaining period after 1 hour.
Results
Temperature differences are reported as differences between the painted panel and the ‘Normal’ panel temperature, where Normal is the aluminium panel coated with regular commercial white paint (drip method). The temperature difference is denoted by ΔT. Thus, ΔT>0 indicates that a panel was warmer than the regular-paint control at that time, while ΔT<0 indicates it was cooler.
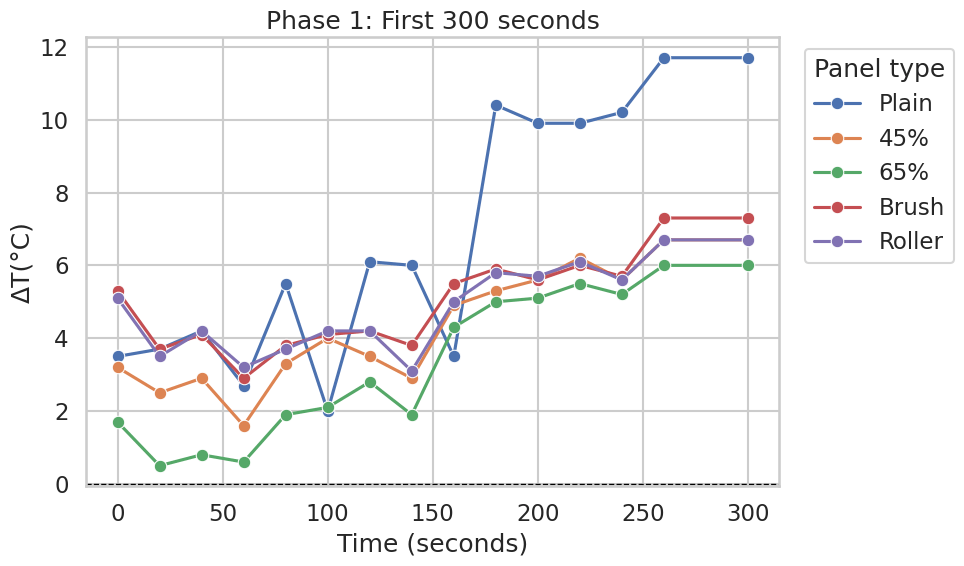
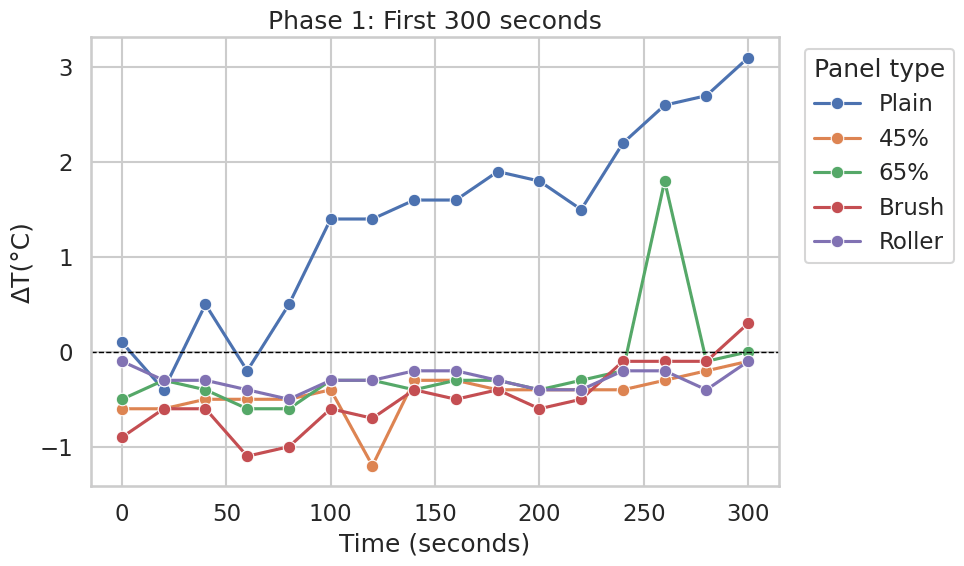
Phase 1: First 300 seconds
Across the first 5 minutes, the uncoated (Plain) panel showed the largest temperature increase relative to the Normal panel, reaching approximately +10 to +12 °C by the end of Phase 1 (Figure 2). Over the same interval, the CaCO₃-containing panels and the different application-method variants (45%, 65%, Brush, Roller) increased more gradually and remained substantially closer to the Normal panel.
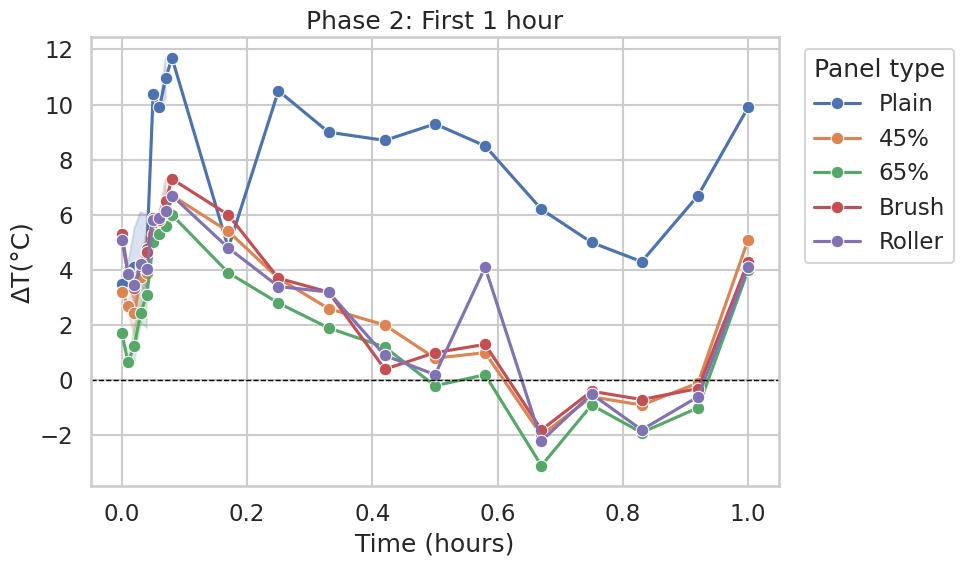
Phase 2: First 1 hour
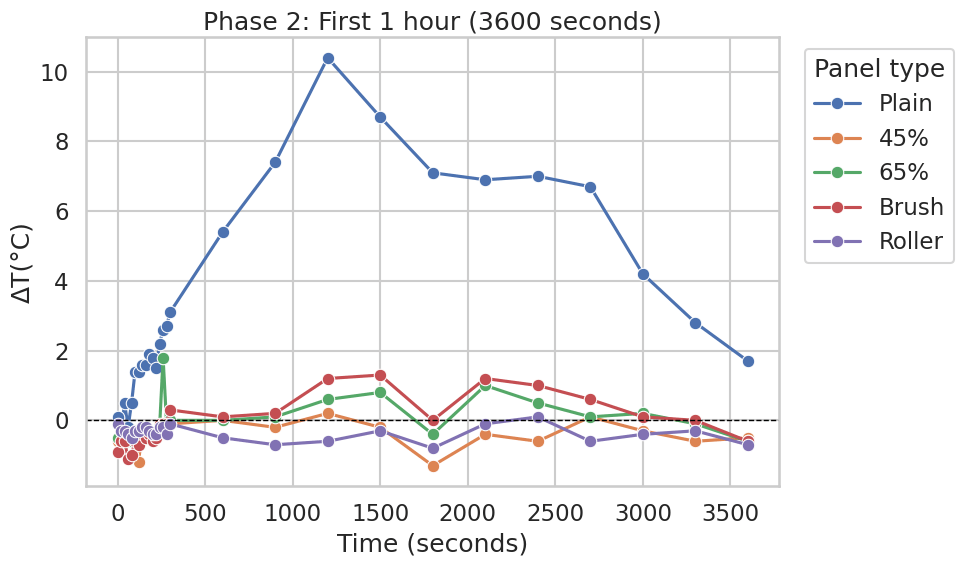
During the first hour, the Plain panel maintained the largest separation from Normal for most of the time window, peaking at approximately +10 to +12°C above Normal (Figure 4; similarly visible as a large positive excursion in Figure 6).
The CaCO₃ panels (45% and 65%) and the application-method variants (Brush, Roller) were much closer to the Normal panel during this phase. In Figure 3, these curves rise early, then trend downward toward
ΔT≈0, and briefly cross below zero later in the first hour (minimum on the order of −2 to −3°C for the coolest point). A sharp positive spike appears at the 1-hour endpoint across multiple curves in Figure 4; this feature is likely associated with the transition in sampling interval or transient outdoor conditions rather than a material-dependent thermal effect. In Figure 6 (same phase plotted in seconds), the CaCO₃ and application-method curves remain comparatively close to ΔT=0 throughout the hour (generally within about ±1°C, with brief excursions), while the Plain panel remains markedly higher for much of the phase.
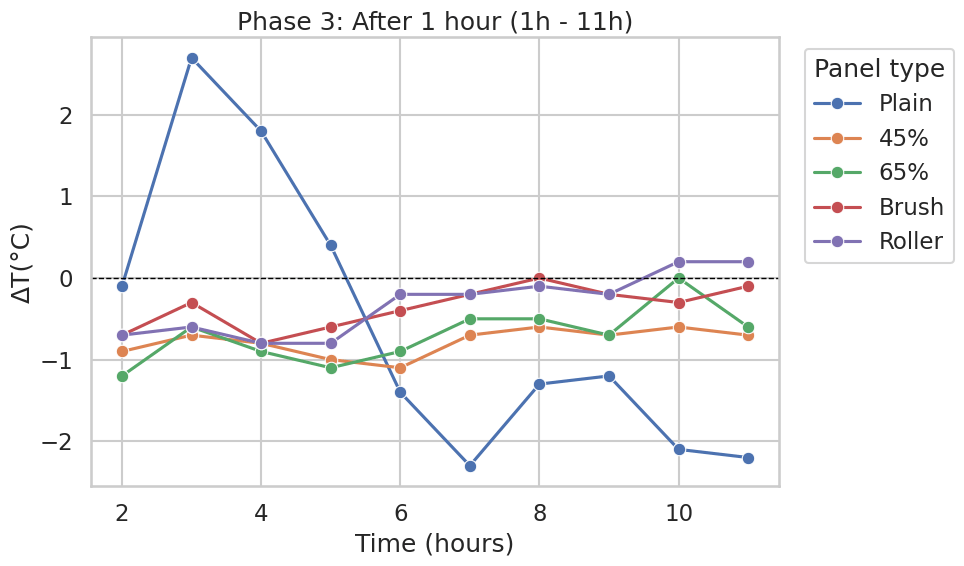
Phase 3: After 1 hour
After the first hour, the CaCO₃-containing panels and the Brush/Roller variants remained tightly grouped near the Normal panel. Differences were typically within roughly −1.2 to +0.2 °C across most hourly points. Over the same interval, the Plain panel showed the largest swing, starting a few degrees above Normal early in this window (approximately +2 to +3°C) and later falling below Normal (down to roughly −2°C by the later time points shown in Figure 6).
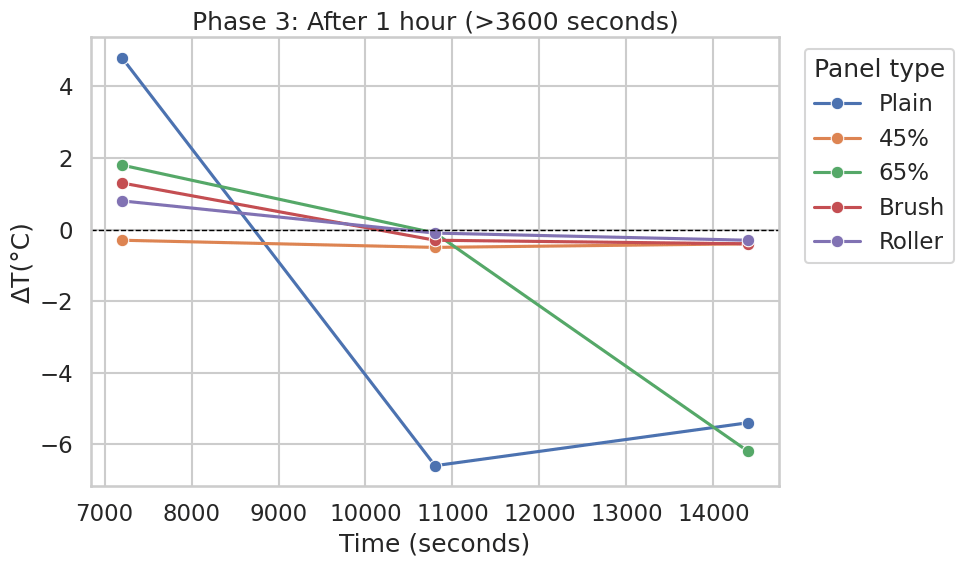
In the shorter post-hour window plotted in seconds (Figure 7), most coated panels (45%, Brush, Roller) remain close to ΔT=0 at the displayed later timestamps, while the Plain panel exhibits a pronounced drop to strongly negative ΔT values at later points in that plot (Figure 7), consistent with known infrared thermometry artefacts for low-emissivity metal surfaces. The 65% panel also shows a large negative value at the last displayed timestamp in Figure 7.
Across the plotted phases, 45% vs 65% CaCO₃ did not separate into consistently distinct trajectories; where differences appear, they are generally modest relative to the gap between the Plain panel and the coated panels (Figures 1–3 and 4–6). Similarly, the Brush and Roller application methods track closely for most of the experiment, indicating minimal observable separation between these two application techniques in the recorded temperature differences (Figures 1–3 and 4–6).
| Panel Type | Max Surface Temperature (°C) | Max ΔT vs Normal (Daytime) (°C) | Max ΔT vs Normal (Night-time) (°C) |
| Plain (Uncoated) | 48.6 | +11.8 | −2.2 |
| Normal (White Paint) | 40.5 | 0 | 0 |
| 45% CaCO₃ (Drip) | 40.1 | −0.4 | −0.8 |
| 65% CaCO₃ (Drip) | 39.1 | −1.4 | −0.6 |
| 65% CaCO₃ (Brush) | 40.3 | −0.2 | −0.7 |
| 65% CaCO₃ (Roller) | 39.8 | −0.7 | −0.8 |
Week 1 is shown as a representative dataset; Week 2 exhibited similar relative trends but differing absolute temperatures due to varying ambient conditions.
Observed temperature differences should be interpreted in the context of measurement uncertainty arising from emissivity-uncorrected infrared thermometry and the use of single panels per condition.
Discussion
Our experiment tested whether mixing calcium carbonate (CaCO₃) into white paint helps aluminium panels stay cooler outdoors compared to regular white paint and bare aluminium. Overall, all white-painted panels stayed much cooler than the uncoated panel, which is consistent with the high solar reflectance of white surfaces. The CaCO₃-enhanced paints showed only small differences compared to the normal white paint, and the 45% and 65% mixtures did not show a clear or consistent separation across the time periods analysed. Similarly, the brush, roller, and drip application methods produced very similar temperature trends, suggesting that application methods did not have a strong effect in this setup.
The relatively small difference between the CaCO₃ paints and the regular white paint is not surprising. Commercial white paints are already designed to be highly reflective, often using efficient pigments such as titanium dioxide, which limits how much additional improvement can be achieved by adding CaCO₃. In addition, because this was a low-cost, DIY preparation, the CaCO₃ may not have been perfectly dispersed within the paint. Any clumping or uneven distribution would reduce the scattering effect that CaCO₃ is expected to provide.
One unexpected observation was that the uncoated aluminium panel sometimes appeared cooler than the painted panels during periods of low sunlight or at night. The most likely explanation for this behaviour is measurement error associated with the infrared thermometer. Infrared thermometers are sensitive to surface emissivity, and bare aluminium has very low emissivity and high infrared reflectivity. As a result, the thermometer can underestimate the true temperature of the uncoated panel or pick up reflected radiation from the surroundings, making it appear artificially cool. Painted surfaces, by contrast, have higher emissivity and generally yield more reliable measurements. Therefore, this apparent nighttime cooling of the uncoated panel is best interpreted as a limitation of the measurement method rather than a real physical effect.
Importantly, during nighttime, loading is absent, and the sky radiative sink is the dominant factor driving surface cooling below ambient. This makes radiative cooling is theoretically most effective at night. The nighttime ΔT values in Table 1 (e.g., −0.6 to −0.8 °C for CaCO₃-enhanced coatings vs. the Normal control) should be presented graphically and interpreted separately from daytime data. A dedicated post-sunset cooling rate analysis is needed to assess whether CaCO₃ coatings offer any radiative advantage during periods of no solar irradiance.
Several limitations may have reduced the clarity of the results. Infrared temperature measurements depend strongly on emissivity, particularly for shiny metal surfaces18,19,20. Only one panel was tested for each condition; coating thickness was not controlled precisely, some panels were repainted between trials, and environmental factors such as wind and local ambient air temperature were not fully tracked. Together, these factors introduce variability that can obscure small performance differences between coatings. The manufacturer-specified accuracy of the infrared thermometer used (typically ±1.5–2 °C for non-contact devices) is comparable in magnitude to the CaCO₃-related ΔT differences reported here. For context, optimised radiative cooling paints in the literature report sub-ambient cooling of 5–10 °C below ambient under direct sunlight1,8, while standard commercial white paints typically remain near ambient or slightly above. The maximum daytime ΔT of −1.4 °C observed for the 65% CaCO₃ formulation relative to the Normal control in this study is small by comparison and falls within the range of measurement uncertainty given the absence of replicate panels and uncorrected IR emissivity.
If this project were extended, an important improvement would be to measure temperature in a way that does not depend on surface emissivity, such as using thermocouples or digital temperature sensors attached to the back of each panel. Recording ambient air temperature in the shade and repeating the key comparisons with multiple panels would also improve confidence in the results. With these refinements, it would be possible to more clearly determine whether CaCO₃ provides a measurable advantage over a high-quality commercial white paint under outdoor conditions. A rigorous follow-up study should incorporate backside thermocouples, randomised panel placement, continuous measurement of solar irradiance and ambient air temperature, and at least three replicate panels per condition tested across multiple days.
Overall, this study shows that while calcium carbonate is a theoretically relevant scattering additive, adding it to an already high-performance commercial white paint does not produce a strong or consistent cooling advantage under the conditions tested here. The results highlight the dominant role of existing commercial pigments, as well as the importance of coating formulation, thickness control, and measurement technique in passive radiative-cooling experiments. At the same time, this work demonstrates that simple, low-cost experimental methods can still capture meaningful temperature trends in real outdoor settings, making exploratory studies of passive cooling more accessible. Additionally, practical trade-offs associated with high CaCO₃ loadings (45–65% by mass) warrant discussion. Very high filler fractions can reduce paint film cohesion, increase porosity, and promote cracking or chalking over time, particularly under outdoor weathering conditions (UV exposure, rain, thermal cycling). These durability considerations are relevant to any proposed real-world application of CaCO₃-enhanced coatings and should be addressed in the discussion.
Acknowledgments
I would like to thank my school and parents for their constant support during this process.
References
- International Energy Agency. The future of cooling: opportunities for energy-efficient air conditioning. International Energy Agency. 2018\. [↩] [↩]
- H. Akbari, M. Pomerantz, H. Taha. Cool surfaces and shade trees to reduce energy use and improve air quality in urban areas. Solar Energy. Vol. 70, pg. 295–310, 2001, https://doi.org/10.1016/S0038-092X(00)00089-X. [↩]
- National Renewable Energy Laboratory. Solar spectra (Air Mass 1.5 / ASTM G-173-03 reference spectra). https://www.nrel.gov/grid/solar-resource/spectra-am1.5.html. 2025\. [↩]
- X. Yin, R. Yang, G. Tan, S. Fan. Terrestrial radiative cooling: using the cold universe as a renewable and sustainable energy source. Science. Vol. 370, pg. 786–791, 2020, https://doi.org/10.1126/science.abb0971. [↩] [↩]
- A. P. Raman, M. A. Anoma, L. Zhu, E. Rephaeli, S. Fan. Passive radiative cooling below ambient air temperature under direct sunlight. Nature. Vol. 515, pg. 540–544, 2014, https://doi.org/10.1038/nature13883. [↩] [↩]
- M. Chen, D. Pang, X. Chen, H. Yan, Y. Yang. Passive daytime radiative cooling: fundamentals, material designs, and applications. EcoMat. Vol. 4, pg. e12153, 2022, https://doi.org/10.1002/eom2.12153. [↩] [↩]
- H. Akbari, R. Levinson, L. Rainer. Monitoring the energy-use effects of cool roofs on California commercial buildings. Energy and Buildings. Vol. 37, pg. 1007–1016, 2005, https://doi.org/10.1016/j.enbuild.2004.11.013. [↩] [↩]
- J. Mandal, Y. Fu, A. C. Overvig, M. Jia, K. Sun, N. N. Shi, H. Zhou, X. Xiao, N. Yu, Y. Yang. Paints as a scalable and effective radiative cooling technology for buildings. Joule. Vol. 4, pg. 1350–1356, 2020, https://doi.org/10.1016/j.joule.2020.04.010. [↩] [↩]
- Y. Tao, Z. Mao, Z. Yang, J. Zhang. CaCO₃ as a new member of high solar-reflective filler on the cooling property in polymer composites. Journal of Vinyl and Additive Technology. Vol. 27, pg. 275–287, 2021, https://doi.org/10.1002/vnl.21801. [↩] [↩]
- H. Lim, D. Chae, S. Son, J. Ha, H. Lee. CaCO₃ micro particle-based radiative cooling device without metal reflector for entire day. Materials Today Communications. Vol. 32, pg. 103990, 2022, https://doi.org/10.1016/j.mtcomm.2022.103990. [↩] [↩]
- X. Li, J. Peoples, Z. Huang, Z. Zhao, J. Qiu, X. Ruan. Full daytime sub-ambient radiative cooling in commercial-like paints with high figure of merit. Cell Reports Physical Science. Vol. 1, pg. 100221, 2020, https://doi.org/10.1016/j.xcrp.2020.100221. [↩] [↩]
- J.-C. Auger, V. A. Martinez, B. Stout. Theoretical study of the scattering efficiency of rutile titanium dioxide pigments as a function of their spatial dispersion. Journal of Coatings Technology and Research. Vol. 6, pg. 89–97, 2009, https://doi.org/10.1007/s11998-008-9116-6. [↩] [↩]
- W. E. Vargas, A. Amador, G. A. Niklasson. Diffuse reflectance of TiO₂ pigmented paints: spectral dependence of the average pathlength parameter and the forward scattering ratio. Optics Communications. Vol. 261, pg. 71–78, 2006, https://doi.org/10.1016/j.optcom.2005.11.059. [↩] [↩]
- Y. Zhai, Y. Ma, S. N. David, D. Zhao, R. Lou, G. Tan, R. Yang, X. Yin. Scalable-manufactured randomized glass-polymer hybrid metamaterial for daytime radiative cooling. Science. Vol. 355, pg. 1062–1066, 2017, https://doi.org/10.1126/science.aai7899. [↩]
- J. Mandal, Y. Fu, A. C. Overvig, M. Jia, K. Sun, N. N. Shi, H. Zhou, X. Xiao, N. Yu, Y. Yang. Hierarchically porous polymer coatings for highly efficient passive daytime radiative cooling. Science. Vol. 362, pg. 315–319, 2018, https://doi.org/10.1126/science.aat9513. [↩] [↩]
- X. Li, J. Peoples, P. Yao, X. Ruan. Ultrawhite BaSO₄ paints and films for remarkable daytime subambient radiative cooling. ACS Applied Materials and Interfaces. Vol. 13, pg. 21733–21739, 2021, https://doi.org/10.1021/acsami.1c02368. [↩]
- R. Levinson, P. Berdahl, H. Akbari. Solar spectral optical properties of pigments—Part II: survey of common colorants. Solar Energy Materials and Solar Cells. Vol. 89, pg. 351–389, 2005, https://doi.org/10.1016/j.solmat.2004.11.013. [↩]
- Fluke Corporation. Emissivity: the hidden variable in infrared temperature measurement (application note). https://www.fluke.com/en-us/learn/blog/thermal-cameras/how-to-set-emissivity. 2026\. [↩]
- Omega Engineering. Infrared temperature measurement / emissivity and reflective errors (technical note). https://www.omega.com/en-us/resources/infrared-temperature-emissivity. 2026. [↩]
- Optris GmbH. Emissivity of aluminium and measurement considerations for IR thermometry (technical note). https://www.optris.com/emissivity-list. 2026\.a. [↩]