Abstract
Crystallization conditions play a critical role in determining crystal morphology, which can influence both analytical outcomes and downstream processing in laboratory and industrial settings. In recrystallization, temperature affects cooling rate, supersaturation, and the balance between nucleation and crystal growth, yet the specific influence of recrystallization temperature on crystal length is not always examined under controlled comparative conditions. This study investigated how recrystallization temperature affects the length of salicylic acid crystals formed from aqueous solution. Salicylic acid was dissolved in water at 70 °C and recrystallized under three temperature conditions: 0 °C, 20 °C, and 35 °C. Two independent batches were prepared, with replicate beakers used for each temperature condition. Solutions were left undisturbed for 20 minutes to allow crystal formation. Crystal lengths were measured using a ruler after blotting surface moisture, and mean crystal lengths were calculated for each condition. A one-way analysis of variance (ANOVA) was performed to assess whether differences in crystal length between temperature treatments were statistically significant. Mean crystal length increased with increasing recrystallization temperature in all trials. Crystals formed at 0 °C were consistently the smallest, while those formed at 35 °C were the largest, with a greater increase observed between 20 °C and 35 °C than between 0 °C and 20 °C. ANOVA indicated that differences in crystal length among temperature conditions were statistically significant. These results demonstrate a temperature-dependent relationship between recrystallization conditions and crystal length, consistent with classical crystallization theory. The findings suggest that controlled recrystallization temperature may be used as a qualitative method to influence crystal size, with potential relevance to laboratory purification and particle size control.
Keywords: recrystallization, salicylic acid, crystal growth, nucleation, temperature effects
Introduction
When crystallized C7H6O3 (salicylic acid) is poured into water on a heat plate at 70 °C, it dissolves. This occurs because water is a polar solvent capable of dissolving polar solutes like salicylic acid1,2. Upon heating, salicylic acid dissolves in water, and the solution becomes supersaturated as it cools, creating thermodynamically unstable conditions that lead to crystallization3. The rate at which a solution cools influences crystal formation, with more rapid cooling typically resulting from a higher difference between a solution and its environment4. The cooling rate is also affected by the thermal properties of its cooling medium. Ice water, because of its higher heat capacity and thermal conductivity compared to air, removes heat more efficiently, resulting in faster cooling5. Therefore, lower cooling temperatures generally correspond to faster cooling rates, which can alter crystallization dynamics. Additionally, the speed of recrystallization affects the length of the crystals formed.
Prior literature has shown that cooling conditions, including cooling rate, can influence the behavior of crystal formation6. While this study illustrates the differences in crystalline structure at varying cooling rates, the present investigation focuses solely on crystal length rather than internal lattice properties. The observed increase in crystal length at higher cooling temperatures is consistent with classical crystallization theory, in which slower cooling leads to reduced nucleation rates, allowing a greater proportion of solute to be incorporated into fewer growing crystals7,8. Because recrystallization behavior strongly depends on solvent-solute interactions, the results of this study are limited to aqueous solutions of lab-grade salicylic acid and may not directly apply to recrystallization in other solvents9,10,11,12. Overall, this study investigates how recrystallization temperature affects the length of salicylic acid crystals formed from aqueous solution.
Controlled Variables
| Controlled Variables | How to Keep Controlled |
| Temperature measurement | Temperature of the bath for the 0 °C sample and the cooling surface for the others was measured using the same calibrated thermometer for each trial to ensure consistency in monitoring temperature. |
| Measurement tool | All crystal lengths were measured using the same ruler with 0.10 cm markings to ensure consistent measurement precision. |
| Crystallization container | Identical glass beakers were used for all trials to minimize variation in cooling rates and nucleation. |
| Cooling duration | All temperatures were allowed to crystalize for 20 minutes as soon as they were placed in their respective conditions before collecting the crystals. |
| Crystal preparation | Crystals were removed from the solution, placed on a paper towel, and gently blotted dry before measuring to maintain consistent measurement conditions. |
| Solution composition | Each trial used identical solution volumes (50 mL) and salicylic acid mass (0.20 g) to maintain a common concentration across samples. |
| Agitation | No agitation was introduced while cooling to avoid variability in nucleation and crystal growth. |
Scope (Range of Independent Variables)
- 0 °C
- 20 °C (Room temperature)
- 35 °C
Research Question
How does the recrystallization temperature (0 °C, 20 °C, 35 °C) affect the length (cm) of C7H6O3 (salicylic acid) crystals that are formed from an aqueous solution?
Hypothesis
It was hypothesized that recrystallization temperature influences the length of salicylic acid crystals formed from an aqueous solution. At a higher recrystallization temperature, slower cooling is expected to reduce nucleation rates, allowing more solute to be used for fewer growing crystals, resulting in a longer average crystal length. On the other hand, lower recrystallization temperatures are expected to promote more rapid cooling and increased nucleation, producing a higher number of shorter crystals.
Equipment
- 150 mL glass beaker
- Electric hot plate
- Rectangular plastic container (ice bath)
- Digital thermometer
- Aluminium foil
- Digital balance (±0.01 g)
- Deionized water
- Salicylic acid (lab-grade)
- Glass stirrer
- Thermal insulated gloves
- Goggles
- Volumetric pipette
- Ruler
- Digital stopwatch (±0.01 seconds)
- Laboratory tweezers/ Forceps
- Paper towels
Safety
- Salicylic acid can cause skin and eye irritation. Therefore, safety goggles must be worn across the duration of the experiment
- Thermal insulated gloves were used when working with the electric hot plates and the hot beakers to protect from burns.
- Glassware was handled with caution to avoid cuts on the skin or eyes from glass breakage.
- Long contact with ice can cause cold-related skin injuries; insulated gloves were used while handling ice baths.
Environmental and Ethical
- All the salicylic acid solution and solids were safely disposed of according to institutional laboratory safety guidelines.
- All the results of this study were collected and reported without fabrication, falsification, or omission.
Procedure
- Measure 50 mL of deionized water with a volumetric pipette and transfer it into a 150 mL glass beaker.
- Place a piece of aluminium foil on a digital balance, zero out the scale, and measure 0.20 g of lab-grade salicylic acid.
- Add the measured salicylic acid to the deionized water.
- Place the beaker on a hot plate set to 70 °C.
- Stir gently with a glass stirring rod until the solution becomes homogenous and clear, avoiding boiling.
Cooling and Recrystallization Conditions
- Prepare two beakers for each temperature.
- 0 °C set
- Prepare an ice-water bath in a rectangular plastic container, making sure to use an identical amount of ice water (300 mL measured using container markings)
- Using thermal insulated gloves, remove the beaker from the hot plate, and place it into the ice bath, making sure no water enters the beaker.
- Monitor the temperature of the ice bath using a digital thermometer.
- Leave the solution undisturbed, and wait 20 minutes for crystallization to occur.
- 20 °C set
- Remove the beaker from the hot plate and place it on a laboratory table in a room maintained at 20 °C.
- Monitor the temperature of the solution using a digital thermometer.
- Leave the solution undisturbed, and wait 20 minutes for crystallization to occur.
- 35 °C set
- Set a second hot plate to 35 °C.
- Transfer the beaker from the 70 °C hot plate to the 35 °C hot plate using thermal insulated gloves.
- Leave the solution undisturbed, and wait 20 minutes for crystallization to occur
Crystal Collection and Measurement
- After crystallization, collect the crystals using lab tweezers.
- Collect 5 unbroken crystals from each beaker, resulting in 10 crystals per temperature condition.
- Place crystals on a paper towel, and gently blot them to remove surface moisture.
- Measure the length of each crystal using a ruler.
- Calculate average crystal length for each temperature.
- Discard and replace crystals that were broken or show visibly anomalous lengths.
Raw Data
| Temp 1 (Ice water 0 °C ± 0.01) lengths ± 0.05 cm | Temp 2 (Room temperature 20 °C ± 0.01) lengths ± 0.05 cm | Temp 3 (35 °C ± 0.01) lengths ± 0.05 cm | |
| Crystal 1 | 0.56 cm | 0.74 cm | 1.21 cm |
| Crystal 2 | 0.62 cm | 0.79 cm | 1.23 cm |
| Crystal 3 | 0.67 cm | 0.81 cm | 1.28 cm |
| Crystal 4 | 0.58 cm | 0.82 cm | 1.26 cm |
| Crystal 5 | 0.63 cm | 0.76 cm | 1.29 cm |
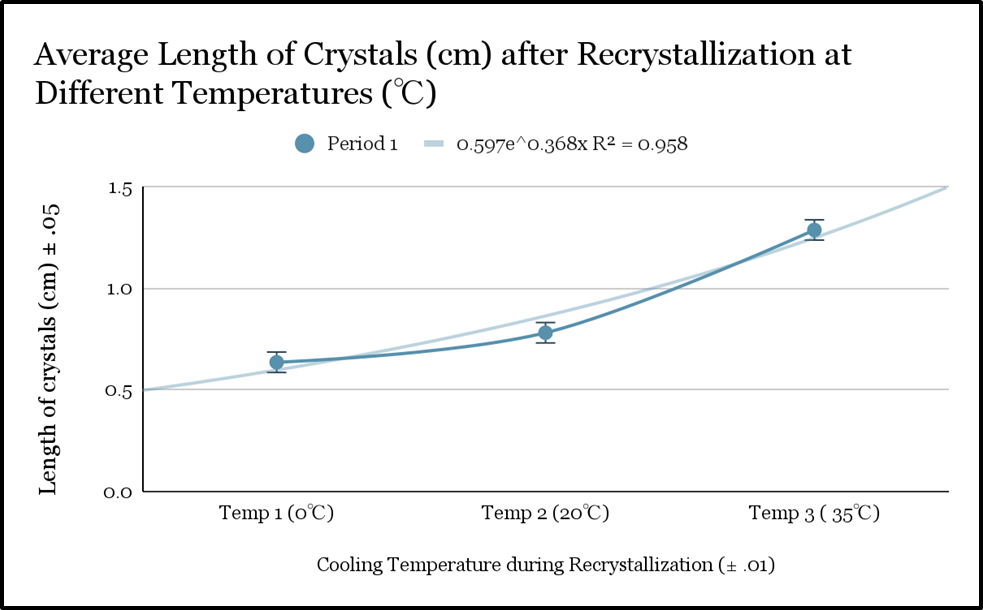
| Average (Mean) | 0.61 cm | 0.78 cm | 1.25 cm |
| Temp 1 (Ice water 0 °C ± 0.01) lengths ± 0.05 cm | Temp 2 (Room temperature 20 °C ± 0.01) lengths ±0.05 cm | Temp 3 (35°C ± 0.01) lengths ± 0.05 cm | |
| Crystal 1 | 0.63 cm | 0.79 cm | 1.25 cm |
| Crystal 2 | 0.52 cm | 0.81 cm | 1.26 cm |
| Crystal 3 | 0.59 cm | 0.82 cm | 1.28 cm |
| Crystal 4 | 0.67 cm | 0.77 cm | 1.27 cm |
| Crystal 5 | 0.68 cm | 0.78 cm | 1.24 cm |
| Average (Mean) | 0.62 cm 65 | 0.79 cm | 1.26 cm |
Processed Data
| Temp 1 (Ice water 0 °C ± 0.01) | Temp 2 (Room temperature 20 °C ± 0.01) | Temp 3 (35 °C ± 0.01) | |
| First Batch Averages | 0.61 cm | 0.78 cm | 1.25 cm |
| Second Batch Averages | 0.62 cm | 0.79 cm | 1.26 cm |
| Average (Mean) ±0.02 cm | 0.62 cm | 0.78 cm | 1.25 cm |
Uncertainty Analysis
Crystal length was measured using a ruler with a precision of ±0.05 cm. The uncertainty of the mean crystal length was measured using the relation:
uncertainty of mean = (instrument uncertainty) ÷ (√n)
Where n is the number of individual crystal length measurements used to calculate the mean. Because 10 crystals were measured for each temperature condition, the uncertainty of the mean was:
(0.05 cm) ÷ (√10) = 0.016 cm
Therefore, average crystal lengths are reported with an uncertainty of ±0.02 cm.
An ANOVA test was conducted using a TI-84 calculator with beaker means as replicates. A one-way analysis of variance (ANOVA) was used because it is appropriate for testing differences in mean response across more than two independent treatment groups under a single factor, in this case recrystallization temperature13. A statistically significant effect of recrystallization temperature was observed on mean crystal length, F(2, 3) = 4396, p = <0.05. The very large F-Value reflects substantial between-group differences relative to small within-group variance. However, results should be interpreted cautiously due to the limited number of replicates. Here, the F-value represents the ratio of variance between temperature treatments to variance within replicates, while the p-value indicates the probability of observing this degree of difference assuming no true effect of temperature.
The ANOVA assumes approximate normality within groups, homogeneity of variance, and independence of observations. Every beaker was treated like an independent replicate, and variance within temperature groups was small and comparable. Because of the limited sample size, normality couldn’t be rigorously assessed, and results should be interpreted cautiously.
Results: The Measured Data
This investigation explored the effect of recrystallization temperature on the length of salicylic acid crystals. Mean crystal length increased with increasing temperature in all trials, with the smallest crystals forming at 0 °C and the largest at 35 °C. Across the two batches the average crystal lengths increased from 0.63-0.64 cm at 0 °C to 1.28-1.29 cm at 35 °C. The magnitude of the increase was observed to be greater between 20 °C and 35 °C than between 0 °C and 20 °C. Statistical analysis using one-way ANOVA indicated that the differences in crystal length between temperature conditions were statistically significant. These results show a consistent increase in mean crystal length along with increasing recrystallization temperature.
Discussion: Interpretation, Comparison to Expectations/Literature
The observed differences in crystal length across the different recrystallization temperatures can be interpreted in the context of classical crystallization behavior, specifically the balance between nucleation and crystal growth14,15,16,17. In recrystallization under higher temperature conditions, heat was removed more gradually from the solution, which likely lessened the number of nucleation events and allowed a greater proportion of dissolved salicylic acid to be incorporated into a smaller number of forming crystals. In contrast, rapid cooling in the ice-water bath likely promoted a greater nucleation density, distributing solute across more crystals, limiting individual crystal growth18,19,20. While nucleation and supersaturation were not measured directly, this qualitative structure is consistent with established crystallization theory and offers a plausible explanation for the observed trend.
Although adjustments were made to standardize timing and temperature monitoring, the experiment does not completely isolate recrystallization temperature from closely related variables such as cooling rate and internal solution temperature. All samples were timed immediately upon placement into their respective conditions; however, differences in cooling dynamics between conditions may still result in variation in effective growth time. Temperature was monitored externally for all conditions (cooling bath for 0 °C and surface environment for 20 °C and 35 °C); however, this approach does not directly measure internal solution temperature, which may introduce minor variability in cooling profiles. As a result, the cooling profiles of the solutions were not fully equivalent across conditions, and the observed changes in crystal length likely reflect a combined influence of cooling rate, effective growth time, and final temperature rather than the temperature alone21,22. These adjustments improve comparability across conditions, although complete isolation of variables would require direct control of internal cooling rates.
Other factors that could affect crystal growth were controlled where possible but remain potential sources of variability. Solutions were not agitated during recrystallization, identical solute masses and solvent volumes were used, and lab-grade salicylic acid was utilized to reduce effects from impurity. Crystals were blotted to dry surface moisture before measurement, but were not dried to constant mass, and lengths were measured without the use of magnification. Despite uncertainty propagation showing that ruler precisions had a minimal impact relative to observed differences in mean crystal length, the use of optical magnification or calipers would improve measurement precision while also reducing subjectivity in crystal selection23.
Despite the limitations, the consistency of the trend throughout replicate beakers suggests that recrystallization conditions likely contributed to the determination of crystal length within the scope of this study, as reproducibility across independent experimental units strengthens causal inferences in lab systems24. Treating beakers as experimental replicates rather than individual crystals strengthens the reliability of conclusions through reducing pseudoreplication and highlighting reproducibility at the same level25. Future investigations could further clarify the fundamental mechanisms by standardizing cooling profiles across all conditions, directly measuring internal solution temperature for all trials, extending the period of crystallization, and quantifying nucleation density. Such improvements would allow the effects of recrystallization temperature, cooling rate, and growth time to be more clearly separated.
Within the limitations of this experimental design, the observed relationship between recrystallization temperature and crystal length may have practical relevance in contexts where particle size affects downstream processing 26,27,28. In industrial or pharmaceutical work, recrystallization is often used to influence crystal size, which can impact drying time, milling efficiency, and handling of powdered materials. For compounds like salicylic acid, temperature-controlled recrystallization may serve as a qualitative method for influencing crystal size before further processing29,30,31. Despite being preliminary, the implications of this study can be extended through the examination of mechanical or functional properties.
References
- Khouri, Sa’ib J., et al. “Salicylic Acid Solubility and Thermodynamic Dissociation Constant at Various Temperatures in Water.” Journal of Solution Chemistry, vol. 53, 2024. [↩]
- Nordström, Fredrik L., and Åke C. Rasmuson. “Solubility and Melting Properties of Salicylic Acid.” Journal of Chemical and Engineering Data, vol. 51, no. 5, 2006, pp. 1668–1671. [↩]
- Mullin, John W. Crystallization. 4th ed., Butterworth-Heinemann, 2001 [↩]
- Myerson, Allan S. Handbook of Industrial Crystallization. 3rd ed., Cambridge University Press, 2019 [↩]
- Incropera, Frank P., et al. Fundamentals of Heat and Mass Transfer. 7th ed., Wiley, 2011 [↩]
- Schelling, M. P. M., et al. “3D Visualization Reveals the Cooling Rate Dependent Crystallization Near a Wall in Dense Microgel Systems.” Soft Matter, vol. 20, no. 32, 2024 [↩]
- Cashmore, David B., et al. “Control of Nucleation and Crystal Size by Cooling Rate.” Crystal Growth & Design, vol. 18, no. 3, 2018, pp. 1542–1550 [↩]
- Kakkar, Shubhangi, Krishnaraj Renuka Devi, and Åke C. Rasmuson. “Molecular Clustering of Fenoxycarb and Salicylic Acid in Organic Solvents and Relation to Crystal Nucleation.” Crystal Growth & Design, vol. 22, no. 5, 2022, pp. 2824–2836. [↩]
- Davey, Roger J., and John W. Mullin. “The Role of Solvent in Crystal Growth and Morphology.” Journal of Crystal Growth, vol. 58, no. 2, 1982, pp. 304–312 [↩]
- Perry, Amelia R., and Patrick R. Unwin. “Quantitative Plane-Resolved Crystal Growth and Dissolution Kinetics by Coupling In Situ Optical Microscopy and Diffusion Models: The Case of Salicylic Acid in Aqueous Solution.” Crystal Growth & Design, vol. 13, no. 2, 2013, pp. 614–622. [↩]
- Jia, Lijun, Michael Svärd, and Åke C. Rasmuson. “Crystal Growth of Salicylic Acid in Organic Solvents.” Crystal Growth & Design, vol. 17, no. 6, 2017, pp. 2964–2974. [↩]
- Nartowski, Karol P., et al. “On the Origin of Surface Imposed Anisotropic Growth of Salicylic and Acetylsalicylic Acids Crystals during Droplet Evaporation.” PLOS ONE, vol. 10, no. 2, 2015, e0118993 [↩]
- Kao, Lillian S., and Charles E. Green. “Analysis of Variance: Is There a Difference in Means and What Does It Mean?” Journal of Surgical Research, vol. 144, no. 1, 2007, pp. 158–170 [↩]
- Ouda, M., et al. “Competition Between Nucleation and Growth During Crystallization.” Journal of Crystal Growth, vol. 401, 2014, pp. 53–60 [↩]
- Vekilov, Peter G. “Nucleation.” Crystal Growth & Design, vol. 10, no. 12, 2010, pp. 5007–5019. [↩]
- Vetter, Thomas, et al. “Modeling Nucleation, Growth, and Ostwald Ripening in Crystallization Processes: A Comparison between Population Balance and Kinetic Rate Equation.” Crystal Growth & Design, vol. 13, no. 11, 2013, pp. 4890–4905. [↩]
- Kadam, Somnath S., et al. “A New View on the Metastable Zone Width During Cooling Crystallization.” Chemical Engineering Science, vol. 72, 2012, pp. 10–19. [↩]
- Hudson, Steven P., et al. “Effect of Cooling Rate on Nucleation Density.” Crystal Growth & Design, vol. 12, no. 8, 2012, pp. 3871–3877 [↩]
- Lanje, Archana S., et al. “Determination of Metastable Zone Width, Induction Period and Primary Nucleation Kinetics for Cooling Crystallization of L-Asparagine Monohydrate.” Journal of Crystal Growth, vol. 408, 2014, pp. 85–92. [↩]
- Li, Sitian, et al. “Size-Independent Nucleation and Growth Model of Potassium Sulfate from Supersaturated Solution Produced by Stirred Crystallization.” Processes, vol. 12, no. 1, 2024. [↩]
- Zhang, Y., et al. “Decoupling Cooling Rate and Final Temperature in Crystallization.” Chemical Engineering Science, vol. 98, 2013, pp. 109–117 [↩]
- Yang, Huaiyu, and Alastair J. Florence. “Relating Induction Time and Metastable Zone Width.” CrystEngComm, vol. 19, no. 28, 2017, pp. 3966–3978. [↩]
- Allen, Thomas. Particle Size Measurement. 5th ed., Chapman and Hall, 1997 [↩]
- Montgomery, Douglas C. Design and Analysis of Experiments. 9th ed., Wiley, 2017 [↩]
- Hurlbert, Stuart H. “Pseudoreplication and the Design of Ecological Experiments.” Ecology, vol. 65, no. 1, 1984, pp. 187–211 [↩]
- Alvarez, A. J., et al. “Crystallization as a Tool for Particle Size Control.” Organic Process Research & Development, vol. 15, no. 5, 2011, pp. 1011–1020 [↩]
- Sun, Jiao, et al. “Effect of Particle Size on Solubility, Dissolution Rate, and Oral Bioavailability: Evaluation Using Coenzyme Q10 as Naked Nanocrystals.” International Journal of Nanomedicine, vol. 7, 2012, pp. 5733–5744. [↩]
- Hintz, R. J., and K. C. Johnson. “The Effect of Particle-Size Distribution on Dissolution Rate and Oral Absorption.” International Journal of Pharmaceutics, vol. 51, no. 1, 1989, pp. 9–17. [↩]
- Beckmann, Wolfgang. Crystallization: Basic Concepts and Industrial Applications. Wiley-VCH, 2013 [↩]
- Blagden, N., et al. “Crystal Engineering of Active Pharmaceutical Ingredients to Improve Solubility and Dissolution Rates.” Advanced Drug Delivery Reviews, vol. 59, no. 7, 2007, pp. 617–630. [↩]
- Huremovic, Melita, et al. “Crystallization and Morphological Characteristics of Acetyl-Salicylic Acid (Aspirin) Synthesized from Substrates of Different Source.” Journal of Chemical, Biological and Physical Sciences, vol. 7, no. 1, 2016, pp. 231–246. [↩]