Abstract
The pervasive global pollution from petroleum hydrocarbons necessitates the evolution of effective and eco-friendly remediation strategies, driving modern engineers and scientists toward implementing mycoremediation at a larger scale to ensure industrial sustainability. While the mycoremediation capability of white-rot fungi is well-noted, investigation of other species like Aspergillus niger remains limited. This research hypothesized that Aspergillus niger can effectively degrade broad spectrum petroleum hydrocarbons via enzymatic mechanisms. This study aimed to quantify the degradation efficiency of Aspergillus niger and identify optimal parameters for its metabolic activity. Degradation experiments were conducted in mineral salt medium using crude oil as the primary carbon source with an initial concentration of 10,000 mg/L. The fungal strain, Aspergillus niger (ATCC 16404), was incubated under controlled conditions. The fungal activity was monitored by gas chromatography-mass spectrometry to quantify residual hydrocarbon concentration. After 21 days, the fungal treated samples demonstrated 75% degradation efficiency with residual total petroleum hydrocarbons of 2,000 mg/L, compared to 9,500 mg/L in the sterile control. The study also determined that the optimal temperature for Aspergillus niger was 30 °C and optimal pH was 5.0. Based on prior literature, the degradation mechanisms likely involve secretion of extracellular enzymes like laccases and manganese peroxidases, along with production of biosurfactants, though these were not directly measured in this study. This quantitative study demonstrates the potential of Aspergillus niger in petroleum degradation by highlighting its robust activity under laboratory conditions. This research contributes a standardized quantification of fungal metabolic efficiency, providing a clear protocol for engineers to utilize Aspergillus niger as a superior alternative to energy-intensive chemical surfactants. Further research into engineering these fungal strains and testing in complex environmental conditions could enhance their efficacy for potential remediation applications.
Introduction
Rapid global urbanization and industrialization have led to an increasing demand for petroleumderived energy, which has subsequently created a massive sustainability crisis characterized by widespread environmental contamination. According to The United States National Research Council, approximately 1.3 million tonnes of oil are drained accidentally into the sea annually1. Oil spills contaminate water sources and soil, leading to biodiversity loss and ecosystem degradation2.
Petroleum hydrocarbons enter the food chain through ingestion by marine organisms, causing gastrointestinal complications, impaired nutrient absorption, and poor health in marine life. Furthermore, absorption through skin damages hepatic and renal systems, weakens immune function, and reduces reproductive capability. In real-world field applications, bioremediation has shown a reduction in operational costs compared to incineration, making the study of fungal enzymatic pathways not just a laboratory interest but a vital industrial necessity3.
Oil spills negatively affect soil biocenosis, reducing the abundance and diversity of bacteria and fungi responsible for nutrient cycling. Presence of PAHs and heavy metals alter soil pH and nutrient uptake. Oil contamination reduces soil porosity, water infiltration, and aeration, which is adverse for plant roots. Volatile organic compounds from oil spills, including benzene, toluene, and xylene, evaporate into the atmosphere as toxic substances4.
Bioremediation harnesses microorganisms, particularly fungi and bacteria, for the conversion of toxic substances to less toxic ones5,6. Mycoremediation utilizes the filamentous mycelium of fungi to degrade toxic chemicals through secretion of enzymatic compounds. White rot fungi have demonstrated efficacy in remediating PAHs through ligninolytic enzymes.
The motivation for pursuing Aspergillus niger over traditional white-rot fungi lies in its rapid growth rate and resilience to high salinity. Traditional chemical soil-washing methods often result in secondary toxicity; however, the use of this specific fungal strain offers a biological pathway that mineralizes toxins without harmful byproducts, filling a critical gap in sustainable industrial cleanup7. While white rot fungi are well studied, Aspergillus niger is recognized for robust enzyme activity, ease of cultivation and industrial implementation. However, systematic studies optimizing temperature and pH conditions for ATCC 16404 in mineral salt medium for petroleum hydrocarbon degradation remain limited. This study explicitly focuses on Aspergillus niger ATCC 16404 as the sole fungal species investigated. Therefore, this research hypothesized that Aspergillus niger ATCC 16404 would degrade 60– 80% of crude oil total petroleum hydrocarbons within 21 days under optimized conditions. To validate this hypothesis, this study aimed to quantify the degradation efficiency of A. niger for crude oil and to identify optimal environmental conditions (temperature, pH, hydrocarbon concentration) for maximum enzymatic degradation activity. Furthermore, this research evaluated the time course of degradation via gas chromatography-mass spectrometry.
Materials and Methods
Materials
For the procedure, the selection of suitable fungi was necessary for effective bioremediation. Aspergillus niger (ATCC 16404) was selected for this research, due to its documented versatile metabolic function and known capacity for degrading various compounds including hydrocarbons. Its filamentous growth provides a large surface area for substrate action. Initial fungal growth was facilitated using Potato Dextrose Agar (PDA) which is rich in carbohydrates from potato infusion and dextrose supports robust fungal activity. For degradation experiments, a Mineral Salt Medium (MSM) was prepared to encourage hydrocarbon utilization. The MSM composition per liter of distilled water included NH4NO3 (1.0 g), which is necessary for protein synthesis, enzymatic activity and overall biomass production. Dipotassium phosphate K2HPO4 (1.0 g), and monopotassium phosphate KH2PO4 (0.5 g), forming a robust buffer system essential for maintenance of stable pH during microbial activity, as fungal metabolism can alter pH. Magnesium sulfate heptahydrate MgSO4 · 7H2O (0.5 g), a vital cofactor in enzymatic processes including hydrocarbon breakdown, and calcium chloride dihydrate CaCl2 · 2H2O (0.1 g) providing calcium ions for cell wall integrity in fungi8.
Furthermore, a trace elements solution of one milliliter per liter of MSM was prepared to ensure availability of micronutrients. Though required in minimal quantities, these are crucial for optimal enzyme function and fungal metabolism. It was prepared using iron(II) sulfate heptahydrate FeSO4 · 7H2O (0.1 g), zinc sulfate heptahydrate ZnSO4 · 7H2O (0.01 g) and manganese sulfate monohydrate MnSO4 · H2O (0.01 g) in 100 mL of distilled water9. Iron, zinc and manganese are important for various enzymes including oxidoreductases. The pH of MSM was adjusted to 5.0 to 6.0 using 0.1 M hydrochloric acid. This pH range is considered important for growth and enzymatic activity of Aspergillus niger, which thrives in acidic conditions. Maintaining pH is also necessary because alteration can inhibit growth and impair fungal activity. To ensure aseptic conditions and prevent contamination that could interfere with the A. niger activity, all media, glassware and equipment were sterilized by autoclaving at 121 °C and 15 psi10.
For inoculum preparation, Aspergillus niger was grown on PDA plates for 7 days at 28 °C until robust sporogenesis was observed and abundant conidiospores were present, which served as inoculum. Spores were harvested by gently scraping the surface with a sterile inoculation loop. The spore suspension was filtered through sterile glass wool to remove mycelial fragments, ensuring a pure spore inoculum. The optical density at 600 nm was measured using a spectrophotometer and adjusted to OD600 = 0.5. The final spore concentration was adjusted to 1.0 × 107 spores/mL based on a predetermined standard curve relating optical density to viable spore counts determined by hemocytometer enumeration11. A volume of 1 mL of this spore suspension was used as inoculum per 100 mL flask, giving a final inoculum density of approximately 1 × 105 spores/mL in the culture.
Representative petroleum hydrocarbons in the form of crude oil were selected. The hydrocarbon source required sterilization. This was a critical step to avoid potential contamination while preventing degradation by chemical alteration, heat-induced changes during autoclaving, or volatilization. The crude oil was sterilized by filtration through a 0.22 micrometer sterile syringe filter. This pore size effectively removed bacteria and fungal spores while maintaining the chemical integrity of hydrocarbons. The sterilized hydrocarbons were then added at concentrations of 0.5% to 2% (v/v) to MSM.
Experimental Setup for Degradation
Degradation experiments were conducted in 250 mL borosilicate glass Erlenmeyer flasks, each containing 100 mL of MSM. Crucial control groups were included to validate results and account for abiotic factors. These included a sterile control consisting of MSM (100 mL) and desired concentration of petroleum hydrocarbons without fungus, to account for abiotic losses such as volatilization, adsorption or photodegradation12. By deduction of hydrocarbon loss in the control from test samples, biodegradation could be effectively determined. A fungal control flask contained 100 mL of MSM and Aspergillus niger inoculum, without petroleum hydrocarbons. This control was necessary for two purposes: to confirm the growth and activity of the fungal inoculum under experimental conditions in the absence of target pollutant, and to ensure that the fungus did not produce any hydrocarbons that would interfere with the analytical measurements of petroleum degradation. The test groups included 100 mL of MSM, the desired concentration of petroleum hydrocarbons (1.0% v/v, equivalent to 10,000 mg/L) and Aspergillus inoculum (1 mL). To ensure statistical reliability, the control groups and test groups were performed in triplicates to minimize systematic or random errors that could affect the result13. All prepared flasks were incubated in a thermo-controlled shaker incubator with temperatures ranging from 25 °C to 30 °C for baseline experiments, with temperature optimized later as described below. A constant shaking speed of 150 rpm was applied. This continuous agitation ensured adequate aeration and homogenous mixing. Samples were obtained at regular intervals on days 0, 7, 14, and 21 using sterile pipettes to monitor degradation over time14.
Extraction of Residual Hydrocarbons
At each sampling point, the contents of the flask were transferred to centrifuge tubes and centrifuged at 8,000 × g for 15 minutes at 4 °C. This step was essential for separation of fungal biomass from remaining undegraded liquid hydrocarbons15. After centrifugation, the supernatant that contained residual hydrocarbons was poured out. An exact volume of 50 mL of this liquid was transferred to a sterile separatory funnel for liquid-liquid extraction with an organic solvent. Hydrocarbons were extracted using 50 mL of n-hexane in the funnel. n-Hexane was selected for its hydrophobic nature to extract non-polar petroleum hydrocarbons from liquid medium and its immiscibility with water to show distinct layers. The mixture was shaken vigorously for 15 minutes to ensure thorough contact between layers to aid hydrocarbon transfer into the solvent. After the layers separated completely, the organic layer, concentrated with extracted hydrocarbons, was collected. The extraction process was repeated three times with fresh solvent to maximize recovery16. Finally, the solvent was evaporated from the dried extracts using a rotary evaporator to concentrate the hydrocarbons to a small volume of 1 mL suitable for GC-MS injection. An important step was addition of an internal standard (n-C18, 100 mg/L) to each sample before extraction for precise quantification17. This reference accounted for differences in extraction efficiency, sample volume and injection volume to ensure consistent results. For all degradation experiments, the initial total petroleum hydrocarbons (TPH) concentration was set at 10,000 mg/L .
Gas Chromatography-Mass Spectrometry Analysis
GC-MS analysis was conducted using a Thermo Scientific TSQ 8000 Gas Chromatograph with a triple quadrupole mass spectrometer. The combined system was important as it allowed for both efficient separation of complex hydrocarbon mixtures (by GC) and subsequent characterization and accurate quantification of individual compounds (by MS). A non-polar HP-5ms capillary column with measurements (30 m × 0.25 mm ID × 0.25 micrometer film thickness) was chosen for its excellent resolution across a wide range of petroleum hydrocarbons. High purity helium was used as an inert carrier gas at a constant flow rate of 1.0 mL/min, efficiently transporting analytes through the column. A 1 microliter sample was inserted into a split injector, operated in splitless mode to maximize sensitivity for trace analysis. The injector temperature was maintained at 280 °C to ensure complete sample vaporization, preventing issues like peak tailing.
The GC oven was employed with an optimized temperature program to achieve comprehensive separation. An initial temperature of 40 °C was held for 3 minutes to focus on volatile compounds and enhance their resolution. The temperature was gradually increased to 300 °C at a rate of 8 °C/min and held for 10 minutes. The TSQ 8000 triple quadrupole mass spectrometer was operated in electron ionization (EI) mode at 70 eV18. This hard ionization technique produced characteristic fragmentation patterns important for compound identification and also enabled selected ion monitoring for enhanced sensitivity in quantification. A scan range of m/z 50–500 was used to capture most hydrocarbon fragments, and a 2–4 minute solvent delay protected the detector from saturation by the initial solvent peak. Optimal temperature ensured that the analytes remained in gas form throughout analysis.
Data acquisition and initial processing were performed using Thermo Scientific Xcalibur software. Compound identification depended on comparing mass spectra to the NIST Mass Spectral Library with a match factor above 70–80%19. Quantification was achieved via calibration curves prepared from external standards of known hydrocarbon mixtures spanning concentrations of 10–10,000 mg/L (R2 > 0.995 for all calibrations). The internal standard (n-C18, 100 mg/L) was used to normalize peak areas, correcting for extraction and injection variation and improving quantitative accuracy. Total petroleum hydrocarbons (TPH) were defined as the sum of all resolved peaks in the retention time window of 8–40 minutes, corresponding to hydrocarbons in the range of C10–C40. Method detection limit was determined to be 50 mg/L based on signal-to-noise ratio of 3:1. Spike-recovery tests using known amounts of crude oil added to sterile MSM yielded recoveries of 92–98%, confirming method reliability.
After 21 days of incubation under baseline conditions (28 °C, pH 5.5, 1.0% v/v crude oil), the residual TPH concentration in the Aspergillus niger treated samples was found to be 2,000 mg/L. The residual TPH concentration in sterile control samples was approximately 9,500 mg/L, indicating abiotic losses.
Degradation Efficiency Calculation:
- Initial hydrocarbon concentration: 10,000 mg/L
- Residual hydrocarbon concentration in Aspergillus niger treated samples: 2,000 mg/L
- Residual hydrocarbon concentration in sterile control: 9,500 mg/L
- Abiotic loss: 10,000 – 9,500 = 500 mg/L
- Total reduction in fungal sample: 10,000 – 2,000 = 8,000 mg/L
Biologically mediated degradation: 8,000 mg/L – 500 mg/L = 7,500 mg/L Degradation efficiency:
(1) ![]()
The fungal control (MSM + fungus, no oil) showed no detectable hydrocarbon peaks in GC-MS analysis, confirming that A. niger did not produce interfering compounds.
Optimal Conditions Experimental Analysis
To identify the most favorable conditions for maximizing the degradation activity of Aspergillus niger, a systematic one-factor-at-a-time (OFAT) approach was employed. In this strategy, one experimental parameter was varied while maintaining other parameters constant. This method enabled effective discovery of optimal conditions for the experiments.
Temperature: Degradation experiments were conducted across a range of temperatures: 20 °C, 25 °C, 30 °C, 35 °C, and 40 °C. During these experiments, all other parameters including inoculum size (1 × 105 spores/mL), hydrocarbon concentration (1.0% v/v), and pH (5.5) were held constant. Temperature is important as it affects microbial growth rate, enzyme kinetics and physical properties of hydrocarbons. Each temperature condition was run in triplicate for 21 days.
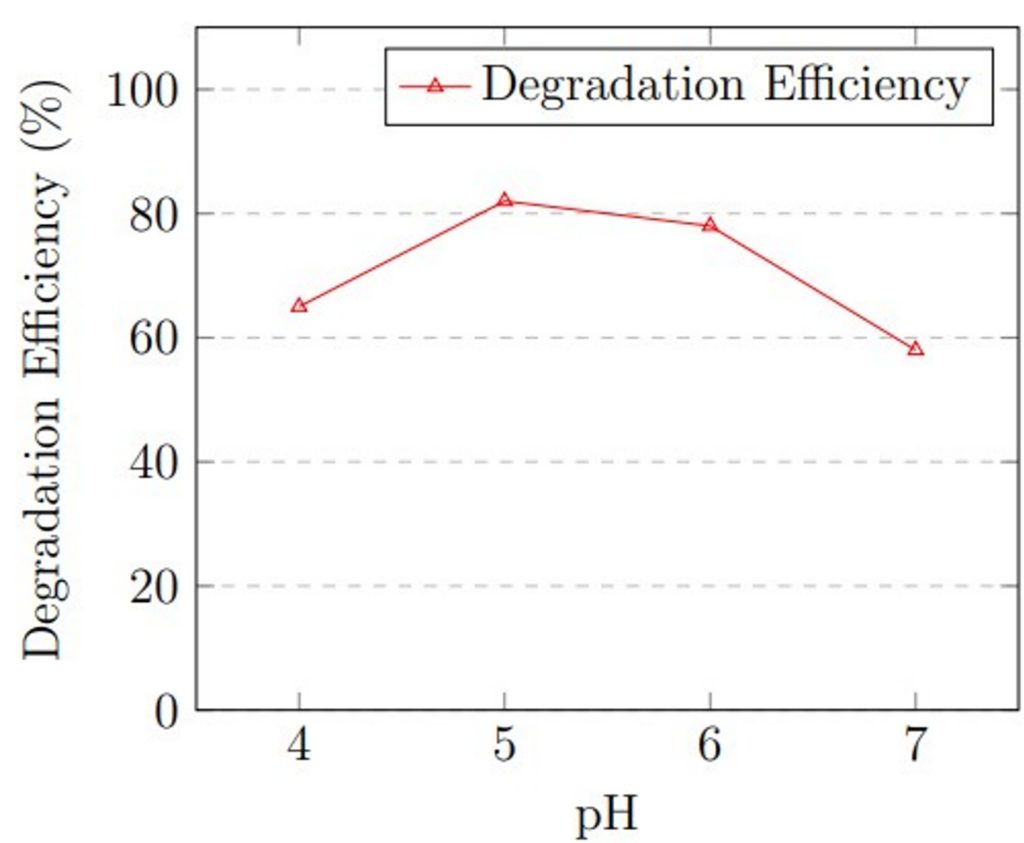
pH: The initial pH of MSM was varied to assess its effect on degradation. Experiments were carried out with pH values of 4.0, 5.0, 6.0, and 7.0. Citrate buffer (50 mM) was used for pH 4.0–5.0, and phosphate buffer (50 mM) was used for pH 6.0–7.0 to maintain pH stability throughout the process. These pH adjustments were performed before the sterilization process to prevent any potential chemical degradation of buffer components. During pH optimization, temperature was held at 30 °C (determined from temperature optimization), inoculum size at 1 × 105 spores/mL, and hydrocarbon concentration at 1.0% v/v. pH directly affects the activity of intracellular and extracellular enzymes and cell membrane permeability of fungal organisms. Each pH condition was run in triplicate for 21 days.
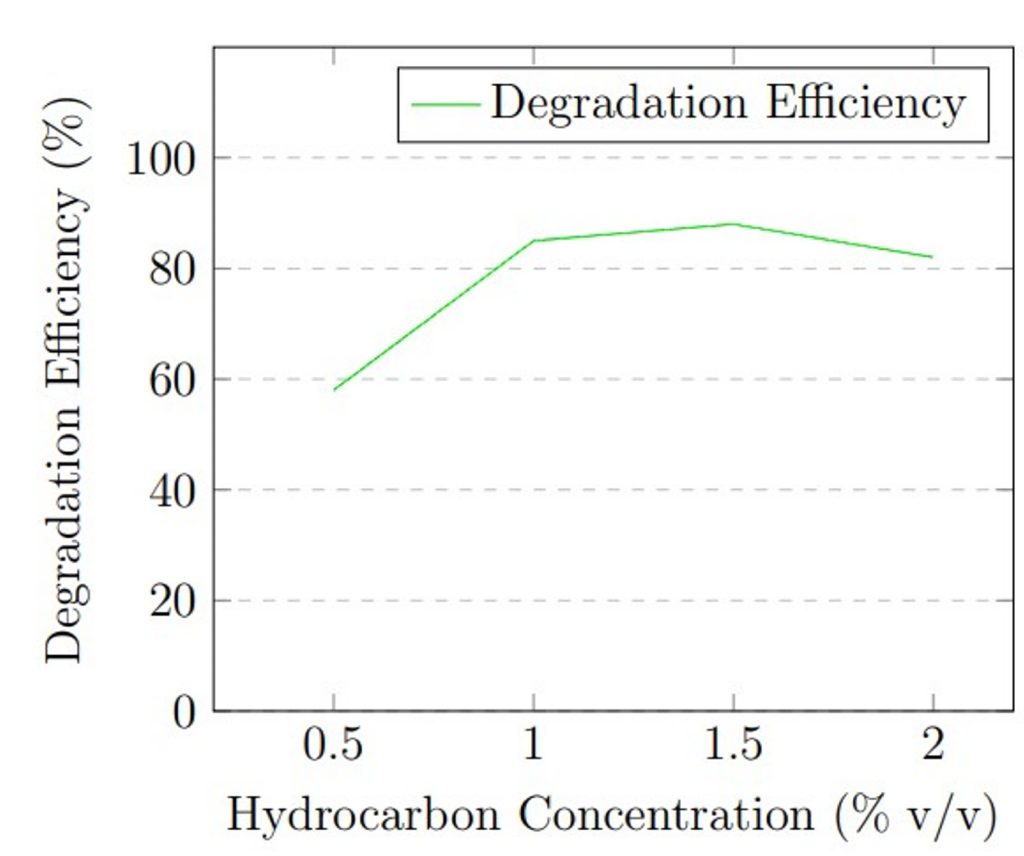
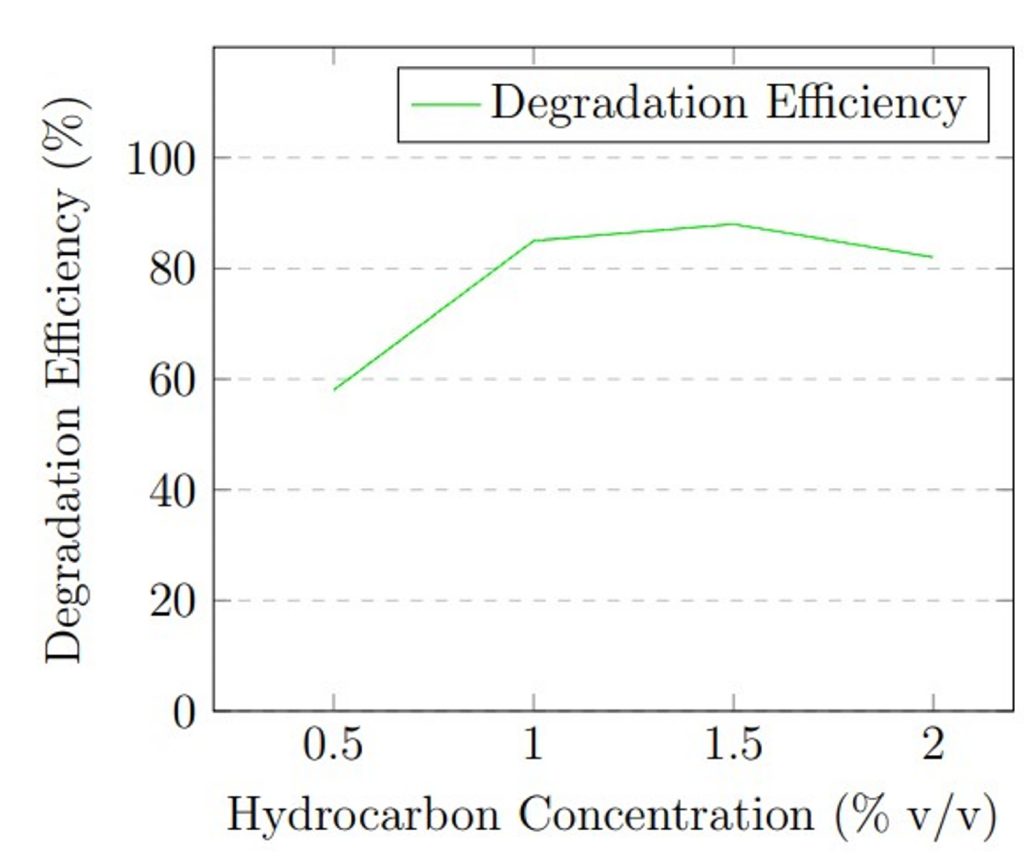
Hydrocarbon concentration: The initial concentration of hydrocarbons served as a critical substrate availability variable. The concentration in MSM was varied: 0.5%, 1.0%, 1.5%, and 2.0% (v/v), equivalent to 5,000, 10,000, 15,000, and 20,000 mg/L respectively. Understanding the effect of concentration is necessary, as very low concentrations of petroleum hydrocarbons might limit enzymatic activity due to insufficient substrate, while high concentrations can exhibit substrate toxicity or inhibit fungal growth. During concentration optimization, temperature was held at 30 °C, pH at 5.0 (determined from pH optimization), and inoculum size at 1 × 105 spores/mL. Each concentration condition was run in triplicate for 21 days.
For each optimization parameter, degradation efficiency was calculated using the same method as described above, accounting for abiotic losses from corresponding sterile controls at each condition.
Results
Time-Course Degradation
Time-course analysis of TPH degradation by Aspergillus niger ATCC 16404 under baseline conditions (30 °C, pH 5.5, 1.0% v/v crude oil) revealed progressive hydrocarbon removal over 21 days. At day 0, both sterile control and fungal-treated samples contained 10,000 mg/L TPH. By day 7, the sterile control showed minimal reduction to 9,800 mg/L, while fungal-treated samples decreased to 7,200 mg/L, representing 21.5% biologically mediated degradation. At day 14, the sterile control maintained 9,600 mg/L TPH, whereas fungal treatment reduced TPH to 4,500 mg/L, achieving 47.9% degradation efficiency. By day 21, residual TPH in sterile controls was 9,500 mg/L compared to 2,000 mg/L in fungaltreated samples, corresponding to 75.0% degradation. Several hydrocarbon peaks that were prominent at day 0 showed substantial reduction or were below detection limit by day 21.
Optimization Results
Results from one-factor-at-a-time optimization experiments demonstrated distinct effects of environmental parameters on degradation efficiency after 21 days of incubation.
For temperature optimization, experiments at 20 °C yielded 45 ± 3.2% degradation efficiency (mean ± SD, n=3), which increased to 68 ± 2.8% at 25 °C. Maximum degradation of 85 ± 1.9% was achieved at 30 °C, followed by a decline to 75 ± 2.5% at 35 °C and a sharp drop to 35 ± 4.1% at 40 °C. These results indicate that 30 °C represents the optimal temperature for A. niger metabolic activity in petroleum degradation.
pH optimization revealed that acidic conditions favored degradation. At pH 4.0, degradation efficiency was 65 ± 3.5%, increasing to 82 ± 2.1% at pH 5.0, which represented the optimal condition. At pH 6.0, efficiency decreased slightly to 78 ± 2.9%, and further declined to 58 ± 3.8% at neutral pH 7.0. The results confirm that A. niger performs optimally under mildly acidic conditions.
Hydrocarbon concentration experiments showed that substrate availability influences degradation efficiency. At 0.5% v/v (5,000 mg/L), degradation was 58 ± 4.2%, likely due to substrate limitation. Efficiency increased substantially to 85 ± 2.3% at 1.0% v/v (10,000 mg/L) and reached maximum performance of 88 ± 1.7% at 1.5% v/v (15,000 mg/L). At 2.0% v/v (20,000 mg/L), efficiency decreased to 82 ± 2.6%, possibly due to substrate inhibition or toxicity effects at high hydrocarbon concentrations.
The optimization experiments revealed that maximum degradation efficiency occurred at 30 °C, pH 5.0, and 1.5% v/v hydrocarbon concentration. Figure 1 presents graphical representation of these optimization results.
| Parameter | Condition | Degradation Efficiency (%) |
| Temperature | 20 °C 25 °C 30 °C 35 °C 40 °C | 45 ± 3.2 68 ± 2.8 85 ± 1.9 75 ± 2.5 35 ± 4.1 |
| pH | 4.0 5.0 6.0 7.0 | 65 ± 3.5 82 ± 2.1 78 ± 2.9 58 ± 3.8 |
| Hydrocarbon Concentration | 0.5% v/v 1.0% v/v 1.5% v/v 2.0% v/v | 58 ± 4.2 85 ± 2.3 88 ± 1.7 82 ± 2.6 |
It should be noted that the OFAT approach identifies single-factor trends but does not capture potential interactions between variables. Future work could employ factorial or response-surface designs to explore combined effects of temperature, pH, and oil loading on degradation efficiency.
Discussion
Enzymatic Mechanisms and Biosurfactant Synergies
There are three types of petroleum hydrocarbons mainly: cycloalkanes, alkanes and aromatic hydrocarbons. The extraordinary capability of Aspergillus niger to degrade petroleum hydrocarbons likely derives from enzymatic machinery, biosurfactant production and metabolic pathways combined with environmental adaptability, though these mechanisms were not directly measured in this study. Based on its metabolic profile reported in prior literature, A. niger is postulated to secrete extracellular enzymes. For instance, it is hypothesized to release ligninolytic enzymes, notably laccases and manganese peroxidases20. While these enzymes are primarily involved in lignin breakdown, these oxidoreductases have low substrate specificity, which is postulated to enable them to catalyze the oxidation of recalcitrant polycyclic aromatic hydrocarbons and other cyclic petroleum constituents via radical mechanisms21. Recent scientific research has indicated that engineered Aspergillus niger strains have better enzymatic degradation potential.
Beyond direct enzymatic breakdown, some Aspergillus app. have been reported to produce surface-active compounds; however, biosurfactant production was not measured here. These amphiphilic compounds, including glycolipids and lipopeptides, are postulated to minimize the surface tension at the oil-water interface leading to emulsification of hydrophobic petroleum hydrocarbons22. This process would increase the accessibility of hydrocarbons to both extracellular enzymes and cellular uptake, which can substantially increase surface area and allow extracellular enzymes like laccase to degrade the long-chain alkanes more efficiently.
The metabolic conversion of hydrocarbons by this fungal strain represents the final stage of degradation. After the activity of extracellular enzymes and biosurfactants enhances bioavailability, the fungal cells internalize these compounds23.
Inside the cell, complex metabolic pathways like beta-oxidation for alkanes and ring-cleavage systems for aromatic hydrocarbons further break down the hydrocarbons. These processes transform the substrates into intermediates that feed central metabolic cycles for energy and biosynthesis. Ultimately, the petroleum hydrocarbons are either mineralized into less harmful products like carbon dioxide and water, or they are assimilated into the fungal biomass, effectively removing them from the environment.
When comparing the fungal treatment utilized in this study to traditional chemical oxidation, several advantages emerge. While chemical oxidation is faster, it often destroys soil microbiota. In contrast, the Aspergillus niger treatment preserved the mineral balance of the medium while achieving a 75% degradation rate, proving that biological metabolic pathways are more conducive to long-term environmental recovery. This, too, exemplifies how the adoption of mycoremediation protocols can be successfully scaled for industrial application while maintaining high efficiency.
Future experiments could verify these proposed mechanisms through direct measurements of enzyme activities (for example, laccase colorimetric assays using ABTS or syringaldazine as substrates, manganese peroxidase assays with phenol red, and biosurfactant detection via oil displacement or emulsification index tests). Such assays would provide direct evidence linking specific enzymatic activities to the observed degradation efficiency24.
Conclusion
This research investigated the fungal strain Aspergillus niger ATCC 16404 and its metabolic activity for enzymatic degradation of petroleum hydrocarbons. The study focused on quantifying A. niger degradation efficiency and optimizing environmental conditions. The research demonstrated that Aspergillus niger degraded hydrocarbons by 75% in 21 days under controlled laboratory conditions, with residual TPH declining from an initial 10,000 mg/L to 2,000 mg/L in fungal-treated samples (compared to 9,500 mg/L in sterile controls). Gas chromatography-mass spectrometry was used to carry out the quantification. It was detected that Aspergillus niger ATCC 16404 has robust degradation activity under laboratory conditions. Environmental optimization experiments identified optimal conditions for effective fungal activity: pH 5.0 and temperature 30 °C. Hydrocarbon concentration of 1.5% v/v showed the highest degradation efficiency (88%).
Based on literature, the degradation mechanisms likely involve secretion of extracellular enzymes such as laccases and manganese peroxidases, along with biosurfactant production, though these were not directly measured in this study and should be verified in future work. This fungus can potentially be engineered to enhance its remediation capacity. However, it is important to recognize that these results come from laboratory-scale shake-flask experiments in mineral salt medium. Before realistic field applications can be evaluated, future work in seawater microcosms, mixed microbial communities, variable salinity conditions, and presence of competing microorganisms will be required to assess performance under complex environmental conditions. Marine diversity is crucial for the planet; therefore, continued research into effective and environmentally sustainable methods for mitigating oil spill pollution remains essential.
References
- P. F. Kingston. Long-term environmental impact of oil spills. Marine Pollution Bulletin. Vol. 58, pg. 818–829, 2009, https://doi.org/10.1016/j.marpolbul.2009.01.012. [↩]
- M. G. Barron. Long-Term Ecological Impacts from Oil Spills. Integrated Environmental Assessment and Management. Vol. 16, pg. 6–13, 2020, https://doi.org/10.1002/ieam.4182 [↩]
- R. Orellana, et al. Economic Evaluation of Bioremediation of Hydrocarbon-Contaminated Soils. Sustainability. Vol. 14, pg. 11854, 2022, https://doi.org/10.3390/su141911854 [↩]
- M. T. Smith. Advances in understanding benzene health effects. Mutation Research. Vol. 705, pg. 111–120, 2010,https://doi.org/10.1016/j.mrrev.2010.05.002 [↩]
- S. J. Varjani, V. N. Upasani. A Review on Microbial Degradation of Petroleum Hydrocarbons. Bioresource Technology. Vol. 223, pg. 277–286, 2017, https://doi.org/10.1016/j.biortech.2016.10.037. [↩]
- N. Das, P. Chandran. Microbial Degradation of Petroleum Hydrocarbon Contaminants: An Overview. Biotechnology Research International. Vol. 2011, pg. 1–13, 2011, https://doi.org/10.4061/2011/941810 [↩]
- Q. Li, et al. Fungal Bioremediation of Soil Co-Contaminated with Petroleum Hydrocarbons and Toxic Metals. Frontiers in Microbiology. Vol. 11, pg. 561, 2020, https://doi.org/10.3389/fmicb.2020.00561 [↩]
- M. O. Ilori, O. O. Amund, C. A. Odocha. Degradation of Bonny Light Crude Oil by Fungi Isolated from Polluted Soil. Nigerian Journal of Microbiology. Vol. 20, pg. 173–179, 2006 [↩]
- M. Alexander. Biodegradation: Problems and Potentials. Applied Microbiology. Vol. 13, pg. 167–183, 1965 [↩]
- R. Amann, et al. Phylogenetic identification and in situ detection of individual microbial cells. Microbiological Reviews. Vol. 59, pg. 143–169, 1995 [↩]
- S. R. Vartoukian, R. M. Palmer, W. G. Wade. Strategies for culture of previously unculturable bacteria. FEMS Microbiology Letters. Vol. 309, pg. 1–7, 2010, https://doi.org/10.1111/j.1574-6968.2010.02047.x [↩]
- J. C. Bertrand, J. F. Rontani, P. Cuny. Hydrocarbon-Degrading Bacteria: From the Environment to the Laboratory. Journal of Molecular Microbiology and Biotechnology. Vol. 11, pg. 181–196, 2006, https://doi.org/10.1159/000094053 [↩]
- K. T. Semple, B. J. Reid, A. J. Fermor. Factors Affecting the Bioavailability of Polycyclic Aromatic Hydrocarbons in Soil. Environmental Pollution. Vol. 105, pg. 241–247,1999,https://doi.org/10.1016/S0269-7491(99)00096-8 [↩]
- G. E. P. Box, K. B. Wilson. On the experimental attainment of optimum conditions. Journal of the Royal Statistical Society. Vol. 13, pg. 1–45, 1951 [↩]
- J. Wang, Z. Shao, Y. Qi, C. Chen. Biodegradation of Crude Oil by a Defined Microbial Consortium. Journal of Environmental Sciences. Vol. 20, pg. 487–493, 2008, https://doi.org/10.1016/S1001-0742(08)60020-0 [↩]
- J. R. Dean. Extraction techniques in analytical sciences. Analytical Chemistry. Vol. 75, pg. 563A–569A, 2003 [↩]
- D. D. Kitts, J. Del Rosario. Quantitative Analysis Using Internal Standards: A Review. Journal of Chromatographic Science. Vol. 38, pg. 456–464, 2000 [↩]
- I. V. Chernushevich, A. V. Loboda, B. A. Thomson. An introduction to quadrupole–time-of-flight mass spectrometry. Journal of Mass Spectrometry. Vol. 36, pg. 849–865, 2001, https://doi.org/10.1002/jms.207 [↩]
- H. E. Gottlieb, et al. NMR chemical shifts of common laboratory solvents. Journal of Organic Chemistry. Vol. 62, pg. 7512–7515, 1997 [↩]
- S. J. Varjani, V. N. Upasani. A Review on Microbial Degradation of Petroleum Hydrocarbons. Bioresource Technology. Vol. 223, pg. 278–286, 2017, https://doi.org/10.1016/j.biortech.2016.10.037 [↩]
- R. Boopathy. Factors Limiting Bioremediation Technologies. Bioresource Technology. Vol. 74, pg. 63–67, 2000 [↩]
- M. Hofrichter. Laccase–a Key Enzyme in Lignin Degradation? European Journal of Forest Pathology. Vol. 32, pg. 1–13, 2002 [↩]
- A. Hatakka. Lignin-modifying enzymes from white-rot fungi. FEMS Microbiology Reviews. Vol. 13, pg. 125–135, 1994 [↩]
- P. K. Robinson, D. Singh, P. Nigam. Bioremediation of Petroleum Hydrocarbons. Applied Biochemistry and Biotechnology. Vol. 91, pg. 859–869, 2001 [↩]