Abstract
Each year, over one million hip replacement surgeries are performed worldwide, with an increasing number of patients presenting with complex anatomical cases.Despite advancements, current mass-produced hip implants often fail to provide a precise fit for patients with unique or atypical anatomies, resulting in complications, extended recovery times, and reduced implant longevity. These challenges highlight the need for personalized solutions that address individual anatomical variations more effectively. This review investigates whether 3D-printed hip implants offer superior anatomical fit, surgical outcomes, and patient satisfaction compared to conventional implants, particularly in cases with complex anatomical deformities. By leveraging 3D printing, the aim is to enhance the fit, function, and overall success of hip implants, particularly for complex cases.This review includes studies that utilize patient-specific imaging data, computer-aided design (CAD), and additive manufacturing.These techniques will allow for the creation of implants that closely match the patient’s anatomy, improving surgical outcomes and reducing the risk of complications.Custom 3D-printed hip implants are designed to achieve improved anatomical conformity, potentially leading to shorter recovery times and reduced post-surgical complications compared to traditional implants, particularly in anatomically complex cases. The evidence suggests that these personalized implants will lead to better long-term patient outcomes and satisfaction.In conclusion, this review highlights the capability of 3D printing technology that can revolutionize hip replacement surgeries for complex anatomical cases by offering customized, efficient, and more effective solutions than conventional implants.
Introduction
Hip replacement surgeries are critical in restoring mobility and improving the quality of life for millions of individuals worldwide1. Despite advancements in surgical techniques and implant design2, traditional mass-produced hip implants often fail to accommodate the unique anatomical structures of patients with complex cases3. This mismatch can lead to complications such as improper fit, prolonged recovery times, and increased rates of revision surgeries4. As a result, the need for innovative approaches to address these challenges has become increasingly urgent5.
Radiological Standards of Normal Hip Anatomy
The clinical demand for customized implant solutions is driven by the diverse anatomical variations seen in patients, particularly those with unique or complex hip structures6. To understand the need for customization, it is essential to first define what constitutes a normal hip joint. A typical hip joint is classified based on four key anatomical markers that determine its stability, function, and alignment. One of the primary indicators of a normal hip joint is joint space width (JSW), which represents the space between the femoral head and the acetabulum. This measurement is critical in assessing cartilage health, with normal values showing a narrower JSW at the superomedial site compared to the apical site7. Studies have shown that an asymmetric or significantly reduced JSW can indicate joint abnormalities8. Another key parameter is the acetabular angle (AA), which measures the slope of the acetabular roof. Normal hips exhibit an acetabular angle within a specific range, while dysplastic hips tend to have significantly higher values, indicating a steeper slope and reduced coverage of the femoral head9. Proper acetabular orientation ensures balanced load distribution and stability10.
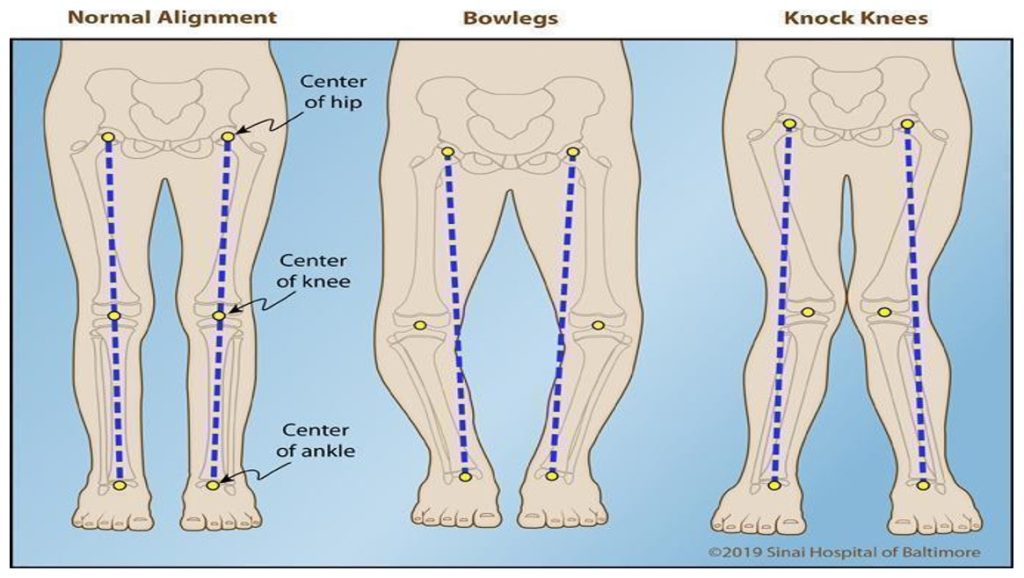
The center-edge angle (CEA) is another crucial factor in defining a normal hip joint. This measurement assesses the degree of femoral head coverage by the acetabulum. A normal hip has a CEA within a defined range, whereas dysplastic hips show significantly lower values, leading to instability and increased risk of dislocation11. Lastly, femoral head coverage evaluates how well the acetabulum covers the femoral head. In normal hips, a higher coverage percentage ensures proper articulation and load distribution. In contrast, dysplastic hips demonstrate lower coverage, which correlates with superior migration of the femoral head and increased joint stress12. These four parameters—joint space width, acetabular angle, center-edge angle, and femoral head coverage—serve as critical radiographic markers that physicians use to classify a hip joint as normal. Understanding these anatomical standards is essential in designing customized hip implants that accommodate individual patient variations, ultimately improving surgical outcomes and long-term mobility13. As shown in Figure 1, variations such as genu varum or genu valgum can significantly affect lower limb alignment, altering joint loading and stability. Recognizing these differences underscores the importance of patient-specific implant design to achieve proper biomechanical performance.
Common Structural Abnormalities in Hip Joints
Abnormal hip structures encompass deviations from standard hip anatomy, leading to compromised joint function and potential musculoskeletal complications. One of the most significant abnormalities is femoral antetorsion, which refers to the inward twisting of the femur. This condition affects the alignment between the femoral neck and the condyles, and when it deviates from the normal range, either through excessive or reduced torsion, it disrupts hip biomechanics14. Studies have linked elevated femoral antetorsion to increased lateral retropatellar cartilage degeneration, especially in individuals with genu valgum (knock-knees)15. This abnormality can contribute to anterior knee pain, instability, and long-term functional impairments16. These structural differences can be clearly seen in Figure 1, which compares leg alignment in cases of genu varum (bowlegs), genu valgum (knock knees), and normal hips.17.
Another critical deviation from normal hip anatomy is abnormal pelvic and acetabular tilt. Pelvic tilt is defined as the orientation of the pelvis in the sagittal plane, influencing how the acetabulum covers the femoral head. When pelvic tilt is abnormal, it alters acetabular version and inclination, which can significantly impact hip stability. A decrease in pelvic tilt reduces acetabular anteversion, leading to insufficient femoral head coverage. This reduction in coverage is a primary contributor to hip instability and can increase the risk of impingement and osteoarthritis over time18. Research has demonstrated that variations in pelvic tilt significantly influence acetabular orientation and subsequently affect hip joint mechanics19.
Additionally, acetabular dysplasia is another common abnormality that affects hip stability and function. Acetabular dysplasia occurs when the acetabulum is too shallow, failing to provide adequate coverage of the femoral head. This lack of coverage leads to increased joint instability and a higher risk of osteoarthritis due to abnormal load distribution20. The condition is often diagnosed using radiographic markers, including the center-edge angle and acetabular depth. A reduced center-edge angle (< 25°) is a key diagnostic criterion for acetabular dysplasia and indicates a greater risk of hip dislocation and labral tears21. Studies have confirmed a strong correlation between acetabular dysplasia and the early onset of degenerative hip disease22 showing insufficient femoral head coverage in dysplastic hips and proper alignment in healthy hips, as seen in Figure 217.
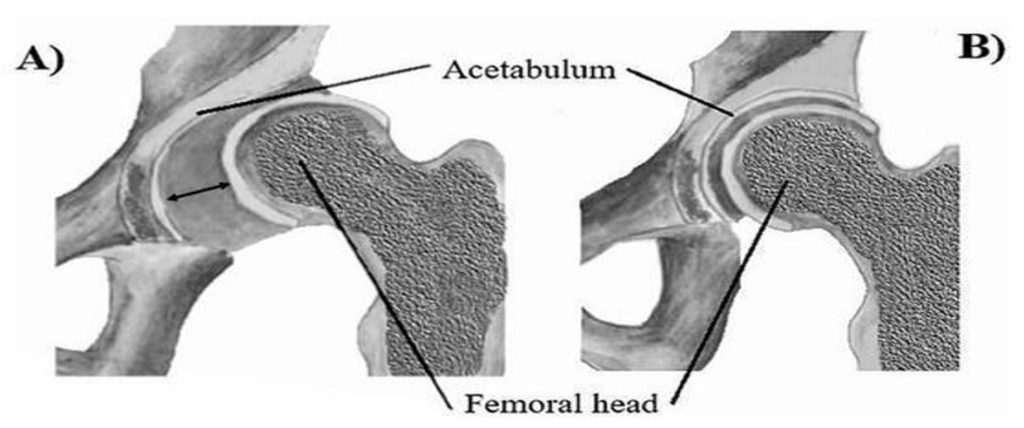
Origins of Structural Hip Deformities
Abnormal hip structures can arise from various factors, including congenital defects, traumatic injuries, and degenerative diseases. One of the most common congenital causes is developmental dysplasia of the hip (DDH), a condition in which the acetabulum is too shallow to properly house the femoral head, resulting in hip instability and an increased risk of dislocation23. Studies indicate that untreated DDH significantly increases the likelihood of early-onset osteoarthritis, often necessitating surgical intervention later in life24. Research suggests that individuals diagnosed with DDH who do not receive early treatment have a higher probability of experiencing joint degeneration and functional impairment, reinforcing the importance of early detection and intervention25.
Traumatic injuries are another major cause of hip abnormalities. Fractures and dislocations resulting from falls, motor vehicle accidents, or high-impact sports can permanently alter hip joint anatomy, leading to malalignment or irregular femoral head positioning26. These structural deficiencies often compromise joint function and accelerate cartilage degeneration. Post-traumatic osteoarthritis, which develops as a consequence of hip trauma, has been identified as a leading indication for hip replacement surgery, accounting for 10–15% of total hip arthroplasty (THA) cases worldwide27. The long-term consequences of hip trauma often necessitate surgical intervention, as non-surgical treatments are generally insufficient in preventing progressive joint deterioration28. Degenerative conditions, particularly osteoarthritis, play a significant role in the development of abnormal hip structures. Osteoarthritis is a chronic, progressive disease characterized by cartilage degradation and joint space narrowing, leading to impaired biomechanics and chronic pain29. Studies estimate that hip osteoarthritis affects 10–25% of adults over the age of 55, making it one of the leading causes of disability and reduced mobility in older populations30. The degenerative process associated with osteoarthritis is often irreversible, with many patients ultimately requiring surgical procedures such as total hip arthroplasty to restore function and alleviate pain31. Individuals with structural hip abnormalities are significantly more likely to require surgical intervention than those with normal joint anatomy. When conservative treatments such as physical therapy, pain management, and lifestyle modifications fail to provide sufficient relief, surgical correction becomes necessary to restore joint stability and prevent further degeneration32. In cases of developmental dysplasia of the hip, surgical intervention has been shown to produce favorable outcomes when performed at the appropriate stage of disease progression. A study evaluating delayed surgical treatment in children with DDH found that 94% of hips achieved excellent or good results, demonstrating the necessity and effectiveness of operative management in these cases33.
Despite the success of surgical interventions, patients with abnormal hip structures face unique challenges that can complicate outcomes. Research assessing open reduction (OR) procedures for infantile DDH identified a 7% risk of redislocation, a 44% risk of proximal femoral growth disturbance, and a 55% risk of residual acetabular dysplasia34, highlighting the complexities associated with surgical correction. Additionally, hip replacement surgery in patients with underlying structural abnormalities is associated with a higher rate of implant failure, particularly in younger individuals or those with severe joint deformities35.
3D Printing Applications in Orthopedic Surgery
Surgical procedures for hip abnormalities present a unique set of challenges due to the variability in joint morphology. Standard hip implants are designed for patients with typical anatomical parameters, meaning they may not adequately accommodate deviations such as acetabular dysplasia, femoral anteversion abnormalities, or excessive pelvic tilt36. As a result, improper implant fit can lead to chronic pain, limited range of motion, and mechanical instability37.
To address these challenges, researchers have increasingly turned to customized implant solutions designed to accommodate individual anatomical differences. Advances in 3D printing technology have made it possible to create patient-specific hip implants tailored to unique bone structures38. These innovations not only improve implant fit but also reduce the risk of mechanical complications and revision surgery39. By integrating patient-specific imaging data with computer-aided design (CAD) and additive manufacturing techniques, 3D-printed implants offer a promising alternative for patients with complex hip abnormalities40.
The objective of this study is to evaluate whether 3D-printed hip implants offer improved anatomical fit, reduced revision surgery rates, and greater patient satisfaction compared to conventional implants in complex anatomical cases. To guide this review, we pose the following research question: Do 3D-printed hip implants improve clinical outcomes—such as surgical fit, patient-reported satisfaction, and revision rates—more effectively than traditional mass-produced implants in patients with atypical hip anatomy? A systematic review was conducted using PubMed to analyze research on 3D printing’s application in orthopedic care, focusing on topics such as implant design methodologies, revision outcomes, and material innovations41. Unlike previous reviews, this study emphasizes the integration of revision surgery data and explores how 3D printing technology can address limitations associated with traditional implants42. It also examines advancements in personalized surgical planning to provide a comprehensive understanding of the current progress in the field43. While earlier research has highlighted the general advantages of 3D printing in orthopedics44, there are still gaps in understanding the long-term outcomes, regulatory challenges, cost-effectiveness, and scalability of these solutions45. This research seeks to bridge these gaps by building on prior literature and presenting an in-depth analysis of how 3D printing can transform orthopedic care. With the potential to significantly improve surgical outcomes and address the limitations of standard implants, 3D printing represents a shift in addressing the needs of patients with abnormal or complex anatomical structures46.
Systematic Review Methodology
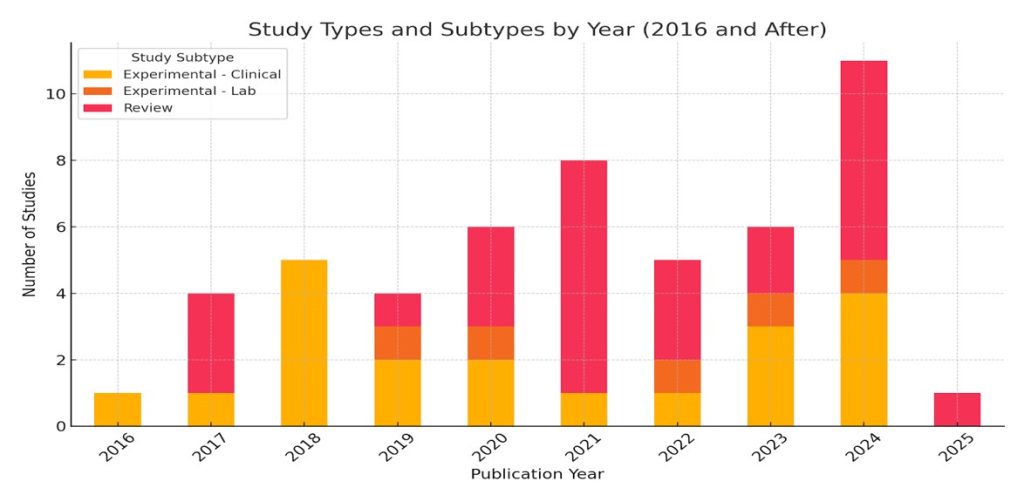
A systematic review of the literature was conducted using PubMed on January 1, 2025. The search terms included “3D Printing,” “Additive Manufacturing,” “Hip Implant,” and “Hip Prosthesis,” while excluding knee-related studies. The review included full-text, peer-reviewed articles published after 2016, written in English, and focused specifically on 3D-printed hip prosthetics that presented either quantitative or qualitative outcome data. The 2016 threshold was selected to ensure technological relevance, as earlier studies typically reflected prototype-level applications and lacked the clinical maturity of more recent work. By excluding pre-2016 publications, the review concentrated on studies aligned with current additive manufacturing capabilities and surgical practices. Editorials, commentaries, and studies not directly related to hip implants were excluded. The review did not include gray literature or non-peer-reviewed materials. Duplicate records were removed using Zotero’s built-in tools. A formal PRISMA diagram was not included in this version of the review, and study quality was not assessed using standardized scoring tools such as the Newcastle-Ottawa Scale or Cochrane Risk of Bias Tool. These are acknowledged as limitations, and future iterations of this study will aim to incorporate these components to enhance rigor and reproducibility.47).Additionally, this review used a narrative synthesis approach and did not include pooled statistical analysis or meta-analysis due to heterogeneity in study designs, patient populations, and outcome reporting. While the narrative approach was chosen to accommodate this heterogeneity, it is important to note that the differences in patient demographics, surgical techniques, follow-up durations, and assessment tools limit the comparability of outcomes. Thus, any observed trends should be interpreted with caution and not viewed as universally generalizable. As a result, direct comparisons of revision rates, surgical time, or complication incidence were not included. Future reviews could benefit from incorporating effect sizes, confidence intervals, or standardized outcome metrics to provide a more robust quantitative synthesis. Studies that did not specifically focus on 3D-printed hip prosthetics or lacked sufficient outcome data were also excluded48). Data from the selected studies were systematically extracted and organized using Zotero, a third-party reference management system (Zotero.org)49). The extracted information included study focus, categorizing research into technical advancements, surgical applications, and theoretical reviews, as well as impact assessment, distinguishing studies that highlighted the benefits of 3D printing from those discussing its challenges and limitations50). Additional data points included publication year, allowing for a comparative analysis of studies published before and after 2020 to track advancements in technology, patient outcomes, and material selection51). Where available, details on patient satisfaction rates, post-surgical revision rates, success rates stratified by patient age, and material composition of prosthetics (e.g., metallic or polymer-based) were extracted to provide a comprehensive evaluation of 3D-printed hip prosthetic development over time52). Figure 3 summarizes the systematic literature screening and selection process, visually illustrating how the final 63 studies were identified and categorized by type and publication year. This diagram reinforces the transparency and scope of the review methodology.
Data Synthesized in Methods
To synthesize the extracted data, studies were categorized into distinct groups based on their primary research focus. Technical research included studies investigating material properties, mechanical strength, and fabrication techniques of 3D-printed implants53). Surgical and clinical applications encompassed research evaluating the use of 3D printing in real-world surgical procedures and patient treatments54). Review and theoretical papers consisted of literature reviews, meta-analyses, and discussions on the broader advancements of 3D printing55). Finally, studies that provided a general overview of the technology’s potential and future applications were classified under general discussion56). In addition to study focus, research was further analyzed based on its impact assessment. Studies emphasizing improvements in patient outcomes, surgical precision, and prosthetic effectiveness were categorized under success and benefits of 3D printing, while those highlighting complications, risks, or barriers to widespread adoption were classified under challenges and limitations57). Articles that presented a balanced discussion of both advantages and drawbacks were grouped under general discussion58). A comparative analysis was conducted by grouping studies published before 2020 and 2020 and after to assess differences in outcomes, methodologies, and technological advancements59). While material-specific data was limited, studies discussing metallic implants, such as titanium, and polymer-based prosthetics were examined to evaluate trends in material selection60). Finally, where applicable, research on patient satisfaction, revision rates, and procedural success was analyzed to assess the performance of 3D-printed prosthetics across different patient demographics61). The meta-analysis of publication trends and study subtypes is illustrated in Figure 3. The visual shows an increase in clinical and in vivo studies after 2020, reflecting the growing emphasis on real-world testing and clinical validation of 3D-printed implants.
Results
Publication Types and Research Focus
A total of 26 Review articles and 25 Experimental studies were analyzed in this research. The Experimental studies were further classified into Clinical and Lab studies. Among the experimental studies, 20 were Clinical and 5 were Lab-based.
The analysis revealed a significant division between review and experimental studies. A total of 26 review articles were identified, which predominantly provided theoretical insights into the potential of 3D printing for hip implants, focusing on its benefits, challenges, and possibilities for future applications62,63,64,65. In contrast, 25 experimental studies provided empirical data, with the majority—20 studies—focused on clinical applications, testing the real-world effectiveness of 3D-printed implants in patient surgeries66,67,68,69,65. The remaining 5 studies were lab-based, focused on material testing, and were simulation research70.
Material Types and Effectiveness
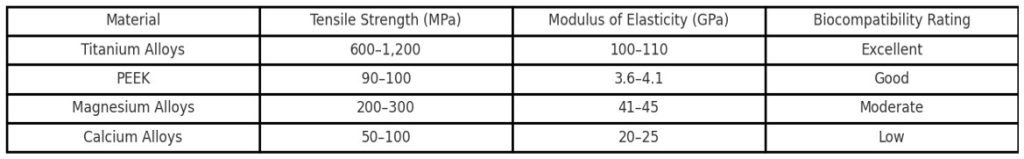
The selection of biomaterials significantly influences the clinical and mechanical success of 3D-printed hip implants. This section compares established options like titanium and cobalt-chromium alloys with emerging alternatives such as polyetheretherketone (PEEK) and biodegradable metals, focusing on differences in biocompatibility, mechanical strength, osseointegration, and imaging compatibility. Titanium alloys—particularly Ti-6Al-7Nb—were the most frequently used materials in the studies reviewed, due to their high mechanical strength (~900 MPa), corrosion resistance, and excellent biocompatibility. The selection of biomaterials significantly influences the clinical and mechanical success of 3D-printed hip implants. This section compares established options like titanium and cobalt-chromium alloys with emerging alternatives such as polyetheretherketone (PEEK) and biodegradable metals, focusing on differences in biocompatibility, mechanical strength, osseointegration, and imaging compatibility. Titanium alloys—particularly Ti-6Al-7Nb—were the most frequently used materials in the studies reviewed, due to their high mechanical strength (~900 MPa), corrosion resistance, and excellent biocompatibility.Several clinical studies have investigated revision surgery outcomes following the use of 3D-printed custom hip implants. In a pediatric cohort, one study noted zero revision surgeries over a 12-month follow-up period when using 3D-printed femoral guides during osteotomy (J. Cao, C. Gao, J. H. Sun, et al. Effect of 3D printing technology in proximal femoral osteotomy in children with developmental dysplasia of the hip. Dis Markers 2022, 1291996 (2022). Another retrospective review involving 34 patients who received titanium 3D-printed acetabular implants for revision hip arthroplasty reported only two reoperations, with most patients experiencing improved Harris Hip Scores( J. Guan, Z. Wang, Z. Zhang, et al. 3D printing application in bone defect area measurement on patients with developmental dysplasia of the hip. Acta Ortop Bras 30, e233064 (2022). Despite promising trends, the follow-up periods in many studies remain short (typically under two years), limiting the ability to evaluate long-term durability. Additionally, stratified outcomes by age, implant location, and implant material were inconsistently reported across studies.
Clinical findings also highlighted promising results, such as lower revision surgery rates and improved patient outcomes in select cases. Cobalt-chromium (Co-Cr) alloys offer even higher wear resistance and compressive strength (~1000 MPa), but their increased stiffness can lead to stress shielding and reduced bone remodeling. Polyetheretherketone (PEEK), a high-performance polymer, is radiolucent—allowing for artifact-free imaging—and provides moderate tensile strength (~100 MPa) and fatigue resistance. Figure 4 provides an example of a hip implant incorporating a PEEK acetabular cup and femoral head, demonstrating how this material can be used in custom designs to improve imaging clarity and wear resistance.However, PEEK lacks osseointegration, which limits its long-term fixation. Biodegradable metals such as magnesium and calcium-based alloys offer the potential for resorption after healing, but face challenges including rapid corrosion, hydrogen gas release, and low load-bearing capacity.71
Cobalt-Chromium alloys have traditionally been used in hip implant components, particularly for femoral heads and acetabular liners. Although less frequently used in 3D-printed models, several studies noted their continued use due to exceptional wear resistance and durability in high-friction environments72.
Emerging materials such as polyether ether ketone (PEEK) and biodegradable metals also appeared in multiple publications, signaling innovation in the field. PEEK is valued for its high biocompatibility, radiolucency, and ease of manufacturing. However, it does not yet match titanium alloys in terms of survivorship or structural stability under long-term stress73. Biodegradable metals—including magnesium and calcium—were explored as temporary scaffolds. One study reported that these materials provide structural support during the early healing phase and safely degrade over time, reducing the need for follow-up surgeries73. PEEK components, such as the one shown in Figure 4, are designed for high wear resistance and improved imaging compatibility. Though not yet widely adopted for load-bearing orthopedic use, they remain promising for specific applications in hip implants74. Figure 5 further summarizes comparative mechanical and biological properties of common materials, showing titanium’s superior tensile strength and biocompatibility.

Implant Design and Clinical Fit
Several clinical studies directly compared standard implants to 3D-printed custom implants. One study found that 3D-printed implants resulted in significantly improved conformity with patient-specific anatomy, reducing intraoperative modifications and improving initial fit75. Another study concluded that the use of patient-matched implants led to shorter operating times, fewer complications, and better patient mobility in follow-ups76. Results from multiple trials showed high patient satisfaction scores specifically with 3D-printed custom implants, which researchers attributed to improved post-surgical comfort, enhanced range of motion, and a more natural anatomical fit77.
3D Printing Technology Types
Selective laser melting (SLM) and electron beam melting (EBM) were the dominant 3D printing technologies used for hip implant production78,79,80. These techniques allow for precise fabrication of dense, load-bearing components with complex geometries. In contrast, methods like fused deposition modeling (FDM) and stereolithography (SLA) were primarily used for surgical planning models. They were not suitable for permanent orthopedic implants due to lower material strength81.
Drawbacks and Gaps Identified in the Literature
Despite promising clinical and lab results, multiple studies noted limitations in the current body of evidence. One comprehensive review highlighted the lack of long-term follow-up studies tracking 3D-printed implant survivorship and functional outcomes74. Several studies emphasized that costs associated with design, material processing, and equipment remain barriers to widespread adoption82. In addition, one lab study found that certain additive manufacturing processes could lead to surface roughness and porosity, potentially impairing bone ingrowth83. Lastly, without standardized guidelines for design and manufacturing, inconsistencies across providers could undermine the reliability of custom 3D-printed solutions84.
Discussion
Real-World Application of 3D Printing in Orthopedics
3D printing in orthopedics is evolving from a theoretical concept to a real-world application. The increasing number of experimental studies demonstrates that 3D printing is transitioning toward mainstream use in orthopedic surgeries85. The shift from review articles to empirical and clinical studies, especially after 2016, reflects growing confidence in 3D printing as a viable tool in patient care86. This trend highlights a broader shift from innovation to implementation. The number of publications on 3D-printed hip implants has steadily increased since 2016, with significant growth observed in 2020 and 2023. These spikes in publications indicate advancements in both the technology and its application in clinical trials, reinforcing confidence in the real-world viability of this technology. This steady shift from review-based research to more experimental and clinical studies marks a significant transition from theoretical exploration to practical, real-world applications of 3D printing in orthopedic surgery.87.This increase in publication volume is indicative of both technological advancements and expanded clinical applications, highlighting the growing confidence in 3D printing’s real-world potential.
Titanium Alloys are the Most Effective Material for 3D-Printed Hip Implants
Titanium alloys, especially Ti-6Al-7Nb, are consistently reported as the most effective materials for 3D-printed hip implants. These materials are favored for their superior mechanical strength, resistance to corrosion, and compatibility with human tissue88. They show high survivorship under long-term stress and are associated with reduced revision rates89. Based on this research, titanium remains the most reliable material for load-bearing, custom hip replacements86. The mechanical performance and biocompatibility of materials used in 3D-printed hip implants are summarized in Figure 5, which draws on peer-reviewed data comparing titanium alloys, PEEK, and biodegradable metals90,91,85,87,92.
Emerging Materials for Hip Implants
Although titanium dominates, alternative materials such as PEEK and biodegradable metals are gaining attention. PEEK is appreciated for its radiolucency and manufacturing flexibility. PEEK has yet to match titanium’s performance under high mechanical loads63. Biodegradable metals like magnesium and calcium offer innovative temporary solutions that support healing before safely degrading the body63 Although this sounds promising, these options require more long-term validation in clinical settings. While titanium remains the gold standard, further biomechanical testing and comparative clinical studies are needed to validate the safety, durability, and effectiveness of emerging materials before they can see widespread use.
High Patient Satisfaction with Customized Implants
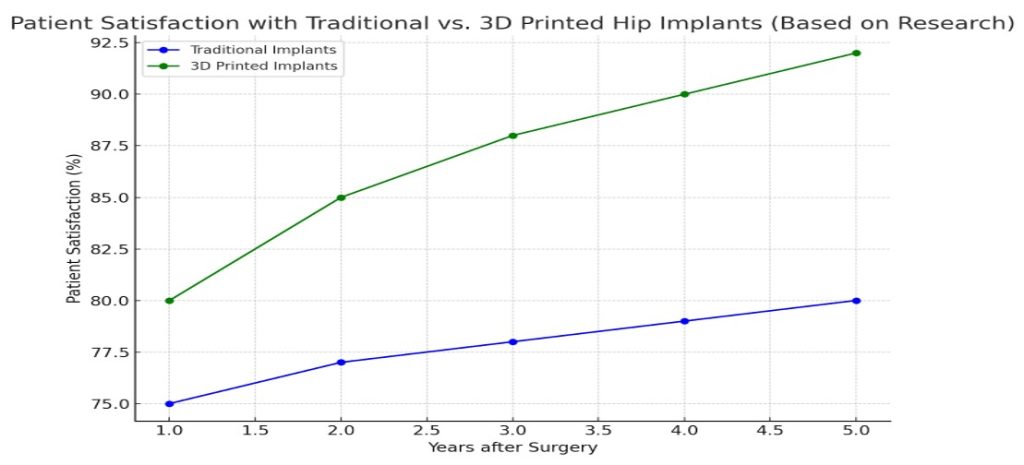
The data suggests that high patient satisfaction rates observed in some studies involving 3D-printed hip implants may be influenced by multiple factors, including implant customization, surgical expertise, and careful patient selection. These factors, along with the potential for improved anatomical fit, likely contribute to the positive outcomes reported93. A key factor contributing to the high satisfaction rates was the customization of the implants, which were specifically designed to match the unique anatomical features of each patient94. Figure 6 visually compares patient satisfaction rates for conventional versus 3D-printed hip implants over a five-year period, highlighting the higher satisfaction reported in several studies. This figure illustrates how customization may contribute to improved outcomes, while recognizing that these results are likely influenced by multiple factors. Custom implants allowed for a better fit, reducing post-surgical discomfort and enhancing overall function, making them a more appealing option for patients compared to traditional implants. The personalized approach to surgery, made possible by 3D printing, was frequently cited as a major factor in improving patient outcomes95. Some studies employed standardized clinical outcome measures to evaluate recovery and implant performance. For example, St. John et al. reported an average Harris Hip Score improvement from 62.4 to 89.1 after surgery with 3D-printed acetabular cups (R. St John, S. Spicer, M. Hadaya, H. Brancaccio, S. Park, S. McMillan. Comparing functional outcomes between 3D printed acetabular cups and traditional prosthetic implants in hip arthroplasty: a systematic review and meta analysis. Arch Orthop Trauma Surg 145, 99 (2025).. This validated metric demonstrated improved joint function, mobility, and pain reduction, supporting the clinical efficacy of custom implants. However, across the broader literature, standardized outcome reporting remains inconsistent, limiting the ability to perform meta-analysis or directly compare treatment approaches.
Lower Revision Rates with Custom Implants
Some studies report that 3D-printed hip implants may be associated with lower revision rates compared to traditional implants. However, these outcomes are likely influenced by multiple factors, including improved anatomical matching, surgical technique, and patient selection. Additional high-quality studies are needed to confirm whether customization alone significantly reduces revision rates97. Multiple clinical studies reported that the customization inherent in 3D printing technology provided a better fit, leading to fewer post-surgical complications such as dislocation or loosening72. As a result, patients who received 3D-printed implants required fewer follow-up surgeries, contributing to better long-term outcomes and reduced healthcare costs74. While these findings are promising, they remain preliminary; further longitudinal and multicenter trials are necessary to validate whether 3D-printed implants consistently reduce revision rates across diverse patient populations.
Technological Developments in 3D Printing
Recent advancements in 3D printing technologies—particularly in resolution, fabrication techniques, and material compatibility—have significantly enhanced the precision and efficiency of custom implant production98. These improvements are especially impactful for titanium-based implants, which remain the gold standard for 3D-printed hip replacements due to their superior mechanical strength and biocompatibility. The accuracy of modern 3D printers enables the creation of detailed, patient-specific designs that closely match complex anatomical structures, improving implant fit and biomechanical function. Additionally, faster production speeds have reduced the turnaround time from digital modeling to surgical implantation, making personalized titanium implants increasingly viable in clinical settings. These technological developments are directly linked to better surgical outcomes and lower complication rates, reinforcing the expanding role of 3D printing in orthopedic practice99.
Technical, Regulatory, and Infrastructure Barriers
Despite these advances, several challenges remain. Studies noted issues such as surface porosity, residual stress, and microstructural inconsistencies in 3D-printed implants, which may compromise long-term durability99.Additionally, the lack of standardized global regulatory frameworks and quality control measures continues to hinder widespread adoption100.Even in well-resourced facilities, integrating in-house 3D printing capabilities requires significant investments in equipment, skilled personnel, and regulatory compliance infrastructure101. These limitations restrict access to the technology, particularly in lower-resourced clinical environments. Regulatory and ethical considerations surrounding 3D-printed hip implants remain underexplored in current literature. Although this review briefly mentions the lack of standardized guidelines, it does not fully address how these gaps affect device approval pathways, patient safety protocols, or legal liability in different healthcare systems. For instance, implantable devices like 3D-printed hip prostheses are subject to rigorous oversight by agencies such as the FDA and EMA, which require evidence of long-term safety, biocompatibility, and manufacturing consistency. The absence of globally harmonized standards for additive manufacturing processes and materials increases the risk of inconsistent device quality and delays in regulatory approval, ultimately impacting patient outcomes and the scalability of these technologies.(U.S. Food and Drug Administration. Technical Considerations for Additive Manufactured Medical Devices: Guidance for Industry and Food and Drug Administration Staff. 2017. ) Failure to elaborate on these dimensions may understate the true challenges of clinical translation.
Data Gaps and Review Limitations
The majority of studies reviewed had short follow-up periods—often under five years—limiting the ability to draw conclusions about long-term survivorship and delayed complications102. Moreover, this meta-analysis included only 63 open-access articles from PubMed. Relevant studies published behind institutional paywalls or within cost-required databases could not be reviewed, potentially omitting key findings. Future analyses with broader access and longer-term data may offer a more comprehensive evaluation of the field’s maturity and risks. Additionally, this review did not systematically compare 3D-printed hip implants with conventional implants using matched cohorts or randomized controlled trials. This limits the ability to draw definitive conclusions about comparative clinical effectiveness. Future studies should aim to include controlled comparisons to strengthen the evidence base for clinical decision-making.This review also did not include a systematic quantitative evaluation of biomechanical testing data, such as stress–strain relationships or fatigue testing results, for 3D-printed hip implant materials. Instead, these mechanical properties were summarized qualitatively based on the reported ranges where available. This limits the depth of the material comparisons and the ability to draw standardized conclusions about mechanical durability. Future reviews should systematically compile and compare biomechanical performance data to strengthen the evidence base for selecting optimal implant materials.
Future Directions for Personalized Orthopedic Solutions
Although this review provides a comprehensive overview of current evidence, it did not formally assess study design strength. As a result, all included studies were treated equally, which may limit the reliability of the conclusions. As the field of orthopedic 3D printing continues to advance, future research should aim to expand the clinical applicability of custom hip implants by addressing the specific challenges identified in this review. Studies should explore broader patient demographics, including pediatric and elderly populations, as well as individuals with rare or highly complex hip deformities, to assess the adaptability of 3D-printed implants across varied anatomical cases. Given that titanium alloys emerged as the most effective material in this study, future materials research could investigate methods to further enhance titanium’s performance or develop hybrid materials that combine strength, biocompatibility, and imaging compatibility.
Cost-effectiveness and scalability also remain crucial. Future investigations should assess how custom implant production can be made economically viable across different healthcare systems, particularly in under-resourced settings. This includes analyzing the cost-benefit ratio of reduced revision surgeries and improved long-term outcomes compared to traditional implants.
Moreover, integrating artificial intelligence (AI) into the preoperative planning process could optimize surgical outcomes by automating implant design based on patient-specific imaging. When combined with additive manufacturing, AI-driven workflows may significantly reduce planning time, increase implant accuracy, and standardize custom care. Moreover, integrating artificial intelligence (AI) into the preoperative planning process could optimize surgical outcomes by automating implant design based on patient-specific imaging. When combined with additive manufacturing, AI-driven workflows may significantly reduce planning time, increase implant accuracy, and standardize custom care. Despite the promise of 3D-printed implants, several socioeconomic and logistical barriers hinder their widespread clinical adoption. One major limitation is the high production cost, particularly for custom-made implants that require personalized imaging and design workflows. These costs are often not reimbursed under traditional healthcare billing systems, which limits accessibility for patients in both public and private hospitals. Additionally, hospital infrastructure plays a key role; many institutions lack access to the specialized software, additive manufacturing equipment, or sterilization protocols necessary for in-house 3D printing. Furthermore, advanced imaging requirements, such as high-resolution CT scans, may be unavailable in rural or low-resource settings, limiting patient eligibility. In low- and middle-income countries, the challenges are even more pronounced due to budget constraints, a shortage of trained orthopedic surgeons familiar with new technologies, and a lack of centralized production hubs. Addressing these real-world barriers is essential to realizing the full translational potential of 3D printing in orthopedics. In parallel, regulatory and ethical considerations must be addressed to ensure safe and equitable deployment of 3D-printed implants. In the United States, the FDA has published guidance on additive manufactured medical devices, outlining technical considerations such as device design, material characterization, and post-processing requirements (U.S. Food and Drug Administration. Technical Considerations for Additive Manufactured Medical Devices: Guidance for Industry and Food and Drug Administration Staff. 2017.). However, the lack of harmonized international standards continues to pose challenges, particularly for cross-border clinical implementation and manufacturing consistency. In the European Union, CE marking requirements for 3D-printed implants remain under refinement, with significant variability depending on the device classification and intended use103. These regulatory gaps can delay clinical translation, raise liability concerns, and complicate surgeon and hospital adoption of novel implant designs.
In conclusion, 3D printing holds substantial promise in advancing personalized orthopedic care, especially in anatomically complex hip replacement cases. However, while early studies suggest favorable outcomes, the evidence base is still maturing. Many of the reviewed studies feature small sample sizes, limited follow-up durations, and a lack of randomized controlled trials, which limits the generalizability of findings. Therefore, although the technology is transformative in concept, its widespread clinical adoption should proceed with cautious optimism. Future research must include rigorous trial designs, long-term evaluations, and broader patient demographics to validate the safety, efficacy, and cost-effectiveness of 3D-printed hip implants across diverse clinical settings. However, despite these advantages, 3D-printed implants may introduce novel mechanical risks. For example, layer delamination can result from incomplete fusion between successive layers during the additive manufacturing process, potentially reducing implant strength. Residual stresses, often induced by uneven cooling, can lead to microcracks or fatigue fractures. Additionally, surface irregularities inherent to some 3D printing methods may impair osseointegration or increase wear. These unique failure modes warrant further study and quality assurance measures to ensure safe long-term use104.
References
- Jabil. 3D printing in healthcare makes it personal. (2025). [↩]
- R. St John, S. Spicer, M. Hadaya, H. Brancaccio, S. Park, S. McMillan. Comparing functional outcomes between 3D printed acetabular cups and traditional prosthetic implants in hip arthroplasty: a systematic review and meta analysis. Arch Orthop Trauma Surg 145, 99 (2025). [↩]
- J. Su, X. Xing, Y. Lin, et al. In vitro physicochemical and biological properties of titanium alloy, zirconia, polyetheretherketone, and carbon fiber-reinforced polyetheretherketone. Front Mater 9 (2022). [↩]
- E. Marin, A. Lanzutti. Biomedical applications of titanium alloys: a comprehensive review. Materials (Basel) 17, 114 (2023). [↩]
- C. Zhou. Application of 3D printing technology and related materials in total hip arthroplasty. In: Second International Conference on Biological Engineering and Medical Science (ICBioMed 2022). Vol 12611. SPIE; pp. 354–359 (2023). [↩]
- S. Goreninskii, I. Akimchenko, A. Vorobyev, et al. Single-step method for the immobilization of hydroxyapatite on 3D-printed porous polyetherketoneketone implants. arXiv preprint arXiv:2501.08133 (2024). [↩]
- W. Liang, C. Zhou, H. Zhang, et al. Recent advances in 3D printing of biodegradable metals for orthopaedic applications. J Biol Eng 17, 56 (2023). [↩]
- Additive Manufacturing Material. What are the industrial applications of hip additive manufacturing. (2023). [↩]
- M. Meng, J. Wang, H. Huang, X. Liu, J. Zhang, Z. Li. 3D printing metal implants in orthopedic surgery: methods, applications and future prospects. J Orthop Transl 42, 94–112 (2023). [↩]
- K. L. Welton, M. J. Kraeutler, T. Garabekyan, O. Mei-Dan. Radiographic parameters of adult hip dysplasia. Orthop J Sports Med 11, 23259671231152868 (2023). [↩]
- Y. J. Kim. Hip osteoarthritis: bench to bedside perspective. Adv Exp Med Biol 1402, 125–133 (2023). [↩]
- S. N. Kiani, A. L. Gornitzky, T. H. Matheney, et al. A prospective, multicenter study of developmental dysplasia of the hip: what can patients expect after open reduction? J Pediatr Orthop 43, 279–285 (2023). [↩]
- S. Konan, C. P. Duncan. Total hip arthroplasty in patients with neuromuscular imbalance. Bone Joint J 100-B, 17–21 (2018). [↩]
- G. D. Dumont. Hip instability: current concepts and treatment options. Clin Sports Med 35, 435–447 (2016). [↩]
- Ö. Çiçekli, M. Doğan. Evaluation of surgical outcome in advanced age patients with developmental hip dysplasia. Int J Surg 52, 44–49 (2018). [↩]
- J. C. Clohisy, M. A. Dobson, J. F. Robison, et al. Radiographic structural abnormalities associated with premature, natural hip-joint failure. J Bone Joint Surg Am 93 Suppl 2, 3–9 (2011). [↩]
- S. B. Goodman. Comparison of radiographic parameters for analysis of normal and dysplastic hips in the adult. Contemp Orthop 20, 505–511 (1990). [↩] [↩]
- D. Kosuge, N. Yamada, S. Azegami, P. Achan, M. Ramachandran. Management of developmental dysplasia of the hip in young adults: current concepts. Bone Joint J 95-B, 732–737 (2013). [↩]
- W. Dandachli, S. Ul Islam, R. Richards, M. Hall-Craggs, J. Witt. The influence of pelvic tilt on acetabular orientation and cover: a three-dimensional computerised tomography analysis. Hip Int 23, 87–92 (2013). [↩]
- R. Glenister, S. Sharma. Anatomy, bony pelvis and lower limb, hip. In: StatPearls. StatPearls Publishing; (2025). [↩]
- M. A. Roussot, S. Salih, G. Grammatopoulos, J. D. Witt. What is the pelvic tilt in acetabular dysplasia and does it change following peri-acetabular osteotomy? J Hip Preserv Surg 7, 777–785 (2021). [↩]
- A. Flury, A. Hoch, O. Andronic, B. Fritz, F. B. Imhoff, S. F. Fucentese. Increased femoral antetorsion correlates with higher degrees of lateral retropatellar cartilage degeneration, further accentuated in genu valgum. Knee Surg Sports Traumatol Arthrosc 29, 1760–1768 (2021). [↩]
- B. Goker, A. Sancak, M. Arac, S. Shott, J. A. Block. The radiographic joint space width in clinically normal hips: effects of age, gender and physical parameters. Osteoarthritis Cartilage 11, 328–334 (2003). [↩]
- N. Fredensborg, B. E. Nilsson. The joint space in normal hip radiographs. Radiology 126, 325–326 (1978). [↩]
- D. Tönnis. Normal values of the hip joint for the evaluation of X-rays in children and adults. Clin Orthop Relat Res 119, 39–47 (1976). [↩]
- M. Lequesne, J. Malghem, E. Dion. The normal hip joint space: variations in width, shape, and architecture on 223 pelvic radiographs. Ann Rheum Dis 63, 1145–1151 (2004). [↩]
- M. Carmona, C. Tzioupis, S. LiArno, A. Faizan, J. N. Argenson, M. Ollivier. Upper femur anatomy depends on age and gender… J Arthroplasty 34, 2487–2493 (2019). [↩]
- C. A. Makarewich, M. B. Anderson, J. M. Gililland, C. E. Pelt, C. L. Peters. Ten-year survivorship of primary total hip arthroplasty in patients 30 years of age or younger. Bone Joint J 100-B, 867–874 (2018). [↩]
- L. E. Bayliss, D. Culliford, A. P. Monk, et al. The effect of patient age at intervention on risk of implant revision after total replacement of the hip or knee: a population-based cohort study. Lancet 389, 1424–1430 (2017). [↩]
- M. G. Wiesli, M. Özcan. High-performance polymers and their potential application as medical and oral implant materials: a review. Implant Dent 24, 448–457 (2015). [↩]
- M. Kaur, K. Singh. Review on titanium and titanium based alloys as biomaterials for orthopaedic applications. Mater Sci Eng C Mater Biol Appl 102, 844–862 (2019). [↩]
- G. Marongiu, L. Leinardi, S. M. Antuofermo, et al. Proximal femoral defect classifications in revision total hip arthroplasty from X-rays imaging to advanced 3D imaging: a narrative review. Ann Jt 9, 18 (2024). [↩]
- H. Zhang, J. Z. Guan, Z. Zhang, et al. Restoring rotation center in total hip arthroplasty for developmental dysplasia of the hip with the assistance of 3D printing technology: a pilot study. Orthop Surg 14, 119–128 (2022). [↩]
- G. Facco, L. Greco, M. Mandolini, et al. Assessing 3-D printing in hip replacement surgical planning. Radiol Technol 93, 246–254 (2022). [↩]
- Y. Liu, F. Wang, J. Ying, et al. Biomechanical analysis and clinical observation of 3D-printed acetabular prosthesis for the acetabular reconstruction of total hip arthroplasty in Crowe III hip dysplasia. Front Bioeng Biotechnol 11, 1219745 (2023). [↩]
- Y. Chen, B. Zhang. Medicine (Baltimore) 102, e36832 (2023). [↩]
- M. Cimerman, A. Kristan, M. Jug, M. Tomaževič. Int Orthop 45, 1057–1064 (2021). [↩]
- X. Hu, Y. Chen, W. Cai, M. Cheng, W. Yan, W. Huang. Bioengineering (Basel) 9, 400 (2022). [↩]
- H. Zhang, Y. Liu, Q. Dong, J. Guan, J. Zhou. Medicine (Baltimore) 99, e22578 (2020). [↩]
- S. Arabnejad, B. Johnston, M. Tanzer, D. Pasini. J Orthop Res 35, 1774–1783 (2017). [↩]
- C. Xiao, S. Zhang, Z. Gao, C. Tu. Technol Health Care 31, 283–291 (2023). [↩]
- Z. Qu, J. Yue, N. Song, S. Li. Int J Surg 110, 6748–6762 (2024). [↩]
- T. D. Lerch, T. Kaim, V. Grob, et al. J Child Orthop 18, 162–170 (2024). [↩]
- C. Si, B. Bai, W. Cong, L. Zhang, R. Guan. Jt Dis Relat Surg 35, 521–528 (2024). [↩]
- I. Aguado-Maestro, C. Simón-Pérez, M. García-Alonso, J. J. Ailagas-De Las Heras, E. Paredes-Herrero. J Clin Med 13, 599 (2024). [↩]
- R. Zhang, J. Lin, F. Chen, W. Liu, M. Chen. J Orthop Surg Res 16, 495 (2021). [↩]
- L. Wan, G. Wu, P. Cao, K. Li, J. Li, S. Zhang. Exp Ther Med 18, 659–663 (2019). [↩]
- A. Sharma, K. S. Kirkland, R. M. Holloway, S. G. Parekh. Foot Ankle Spec 14, 361–365 (2021). [↩]
- Y. Liu, Z. Zhang, W. Wang, et al. Front Surg 10, 1237075 (2023). [↩]
- Z. Cai, C. Piao, T. Zhang, L. Li, L. Xiang. J Orthop Surg Res 16, 256 (2021). [↩]
- G. Anzillotti, E. Guazzoni, P. Conte, et al. J Clin Med 13, 474 (2024). [↩]
- X. Geng, Y. Li, F. Li, et al. J Orthop Surg Res 15, 383 (2020). [↩]
- A. D. M. Paish, H. N. Nikolov, I. D. Welch, et al. J Orthop Res 38, 1627–1636 (2020). [↩]
- J. Cao, C. Gao, J. H. Sun, et al. Dis Markers 2022, 1291996 (2022). [↩]
- W. Barakeh, O. Zein, M. Hemdanieh, B. Sleem, M. Nassereddine. Cureus 16, e60201 (2024). [↩]
- J. Henckel, T. J. Holme, W. Radford, J. A. Skinner, A. J. Hart. J Am Acad Orthop Surg 26, e342–e348 (2018). [↩]
- J. Guan, Z. Wang, Z. Zhang, et al. Acta Ortop Bras 30, e233064 (2022). [↩]
- R. Z. Xia, Z. J. Zhai, Y. Y. Chang, H. W. Li. Orthop Surg 11, 533–544 (2019). [↩]
- A. J. Hughes, C. DeBuitleir, P. Soden, et al. Adv Orthop 2017, 8925050 (2017). [↩]
- L. Yan, P. Wang, H. Zhou. Indian J Orthop 54, 856–862 (2020). [↩]
- S. Feng, Y. J. Sun, Q. R. Zhu, S. F. Shi, Y. S. Zhang, F. Yuan. BMC Med Educ 24, 986 (2024). [↩]
- G. D. Dumont. Hip instability: current concepts and treatment options. Clin Sports Med 35, 435–447 (2016). [↩]
- M. Kaur, K. Singh. Review on titanium and titanium based alloys as biomaterials for orthopaedic applications. Mater Sci Eng C Mater Biol Appl 102, 844–862 (2019). [↩] [↩] [↩]
- L. Wan, G. Wu, P. Cao, K. Li, J. Li, S. Zhang. Curative effect and prognosis of 3D printing titanium alloy trabecular cup and pad in revision of acetabular defect of hip joint. Exp Ther Med 18, 659–663 (2019). [↩]
- R. Z. Xia, Z. J. Zhai, Y. Y. Chang, H. W. Li. Clinical applications of 3-dimensional printing technology in hip joint. Orthop Surg 11, 533–544 (2019). [↩] [↩]
- R. Glenister, S. Sharma. Anatomy, bony pelvis and lower limb, hip. In: StatPearls. StatPearls Publishing; (2025). [↩]
- M. Cimerman, A. Kristan, M. Jug, M. Tomaževič. Fractures of the acetabulum: from yesterday to tomorrow. Int Orthop 45, 1057–1064 (2021). [↩]
- S. Arabnejad, B. Johnston, M. Tanzer, D. Pasini. Fully porous 3D printed titanium femoral stem to reduce stress-shielding following total hip arthroplasty. J Orthop Res 35, 1774–1783 (2017). [↩]
- A. Sharma, K. S. Kirkland, R. M. Holloway, S. G. Parekh. Incorporating 3D printing into your practice: lessons learned. Foot Ankle Spec 14, 361–365 (2021). [↩]
- Y. Liu, Z. Zhang, W. Wang, et al. Artificial intelligence planning and 3D printing augmented modules in the treatment of a complicated hip joint revision: a case report. Front Surg 10, 1237075 (2023). [↩]
- J. Cao, C. Gao, J. H. Sun, et al. Effect of 3D printing technology in proximal femoral osteotomy in children with developmental dysplasia of the hip. Dis Markers 2022, 1291996 (2022). W. Barakeh, O. Zein, M. Hemdanieh, B. Sleem, M. Nassereddine. Enhancing hip arthroplasty outcomes: the multifaceted advantages, limitations, and future directions of 3D printing technology. Cureus 16, e60201 (2024). J. Guan, Z. Wang, Z. Zhang, et al. 3D printing application in bone defect area measurement on patients with developmental dysplasia of the hip. Acta Ortop Bras 30, e233064 (2022). [↩]
- J. Guan, Z. Wang, Z. Zhang, et al. 3D printing application in bone defect area measurement on patients with developmental dysplasia of the hip. Acta Ortop Bras 30, e233064 (2022). [↩] [↩]
- J. Cao, C. Gao, J. H. Sun, et al. [↩] [↩]
- R. Z. Xia, Z. J. Zhai, Y. Y. Chang, H. W. Li. Clinical applications of 3-dimensional printing technology in hip joint. Orthop Surg 11, 533–544 (2019). [↩] [↩] [↩]
- J. Cao, C. Gao, J. H. Sun, et al. Effect of 3D printing technology in proximal femoral osteotomy in children with developmental dysplasia of the hip. Dis Markers 2022, 1291996 (2022). [↩]
- W. Barakeh, O. Zein, M. Hemdanieh, B. Sleem, M. Nassereddine. Enhancing hip arthroplasty outcomes: the multifaceted advantages, limitations, and future directions of 3D printing technology. Cureus 16, e60201 (2024). [↩]
- J. Henckel, T. J. Holme, W. Radford, J. A. Skinner, A. J. Hart. 3D-printed patient-specific guides for hip arthroplasty. J Am Acad Orthop Surg 26, e342–e348 (2018). [↩]
- M. Carmona, C. Tzioupis, S. LiArno, A. Faizan, J. N. Argenson, M. Ollivier. Upper femur anatomy depends on age and gender: a three-dimensional computed tomography comparative bone morphometric analysis of 628 healthy patients’ hips. J Arthroplasty 34, 2487–2493 (2019). [↩]
- L. E. Bayliss, D. Culliford, A. P. Monk, et al. The effect of patient age at intervention on risk of implant revision after total replacement of the hip or knee: a population-based cohort study. Lancet 389, 1424–1430 (2017). [↩]
- G. Facco, L. Greco, M. Mandolini, et al. Assessing 3-D printing in hip replacement surgical planning. Radiol Technol 93, 246–254 (2022). [↩]
- Y. Chen, B. Zhang. 3D printing-assisted total hip arthroplasty and internal fixation for the treatment of fresh acetabular fracture and femoral head necrosis: a case report. Medicine (Baltimore) 102, e36832 (2023). [↩]
- A. J. Hughes, C. DeBuitleir, P. Soden, et al. 3D printing aids acetabular reconstruction in complex revision hip arthroplasty. Adv Orthop 2017, 8925050 (2017). [↩]
- L. Yan, P. Wang, H. Zhou. 3D printing navigation template used in total hip arthroplasty for developmental dysplasia of the hip. Indian J Orthop 54, 856–862 (2020). [↩]
- S. Feng, Y. J. Sun, Q. R. Zhu, S. F. Shi, Y. S. Zhang, F. Yuan. Integration of 3D printing and case-based learning in clinical practice for the treatment of developmental dysplasia of the hip. BMC Med Educ 24, 986 (2024). [↩]
- M. Carmona, C. Tzioupis, S. LiArno, A. Faizan, J. N. Argenson, M. Ollivier. Upper femur anatomy depends on age and gender: a three-dimensional computed tomography comparative bone morphometric analysis of 628 healthy patients’ hips. J Arthroplasty 34, 2487–2493 (2019). [↩] [↩]
- C. A. Makarewich, M. B. Anderson, J. M. Gililland, C. E. Pelt, C. L. Peters. Ten-year survivorship of primary total hip arthroplasty in patients 30 years of age or younger. Bone Joint J 100-B, 867–874 (2018). [↩] [↩]
- L. E. Bayliss, D. Culliford, A. P. Monk, et al. The effect of patient age at intervention on risk of implant revision after total replacement of the hip or knee: a population-based cohort study. Lancet 389, 1424–1430 (2017). [↩] [↩]
- Ö. Çiçekli, M. Doğan. Evaluation of surgical outcome in advanced age patients with developmental hip dysplasia. Int J Surg 52, 44–49 (2018). [↩]
- J. C. Clohisy, M. A. Dobson, J. F. Robison, et al. Radiographic structural abnormalities associated with premature, natural hip-joint failure. J Bone Joint Surg Am 93 Suppl 2, 3–9 (2011). [↩]
- B. Goker, A. Sancak, M. Arac, S. Shott, J. A. Block. The radiographic joint space width in clinically normal hips: effects of age, gender and physical parameters. Osteoarthritis Cartilage 11, 328–334 (2003). [↩]
- M. Lequesne, J. Malghem, E. Dion. The normal hip joint space: variations in width, shape, and architecture on 223 pelvic radiographs. Ann Rheum Dis 63, 1145–1151 (2004). [↩]
- M. G. Wiesli, M. Özcan. High-performance polymers and their potential application as medical and oral implant materials: a review. Implant Dent 24, 448–457 (2015). [↩]
- J. Cao, C. Gao, J. H. Sun, et al. Effect of 3D printing technology in proximal femoral osteotomy in children with developmental dysplasia of the hip. Dis Markers 2022, 1291996 (2022). [↩]
- W. Barakeh, O. Zein, M. Hemdanieh, B. Sleem, M. Nassereddine. Enhancing hip arthroplasty outcomes: the multifaceted advantages, limitations, and future directions of 3D printing technology. Cureus 16, e60201 (2024). [↩]
- J. Henckel, T. J. Holme, W. Radford, J. A. Skinner, A. J. Hart. 3D-printed patient-specific guides for hip arthroplasty. J Am Acad Orthop Surg 26, e342–e348 (2018). [↩]
- J. Cao, C. Gao, J. H. Sun, et al. Dis Markers 2022, 1291996 (2022); W. Barakeh, O. Zein, M. Hemdanieh, B. Sleem, M. Nassereddine. Cureus 16, e60201 (2024); J. Henckel, T. J. Holme, W. Radford, J. A. Skinner, A. J. Hart. J Am Acad Orthop Surg 26, e342–e348 (2018 [↩]
- J. Henckel, T. J. Holme, W. Radford, J. A. Skinner, A. J. Hart. 3D-printed patient-specific guides for hip arthroplasty. J Am Acad Orthop Surg 26, e342–e348 (2018). [↩]
- L. E. Bayliss, D. Culliford, A. P. Monk, et al. The effect of patient age at intervention on risk of implant revision after total replacement of the hip or knee: a population-based cohort study. Lancet 389, 1424–1430 (2017). [↩]
- S. Arabnejad, B. Johnston, M. Tanzer, D. Pasini. Fully porous 3D printed titanium femoral stem to reduce stress-shielding following total hip arthroplasty. J Orthop Res 35, 1774–1783 (2017). [↩] [↩]
- C. Xiao, S. Zhang, Z. Gao, C. Tu. Custom-made 3D-printed porous metal acetabular composite component in revision hip arthroplasty with Paprosky type III acetabular defects: a case report. Technol Health Care 31, 283–291 (2023). [↩]
- H. Zhang, Y. Liu, Q. Dong, J. Guan, J. Zhou. Novel 3D printed integral customized acetabular prosthesis for anatomical rotation center restoration in hip arthroplasty for developmental dysplasia of the hip Crowe type III: a case report. Medicine (Baltimore) 99, e22578 (2020). [↩]
- X. Hu, Y. Chen, W. Cai, M. Cheng, W. Yan, W. Huang. Computer-aided design and 3D printing of hemipelvic endoprosthesis for personalized limb-salvage reconstruction after periacetabular tumor resection. Bioengineering (Basel) 9, 400 (2022 [↩]
- O. Okolie, I. Stachurek, B. Kandasubramanian, J. Njuguna. 3D printing for hip implant applications: a review. Polymers (Basel) 12, 2682 (2020). [↩]
- O. Okolie, I. Stachurek, B. Kandasubramanian, J. Njuguna. Polymers (Basel) 12, 2682 (2020 [↩]