Abstract
There are many treatment options for children that struggle with pediatric cancer such as chemotherapy, immunotherapy, surgery, and radiation. While radiation is highly effective in destroying malignant tumors and cancer cells, it causes long term endocrine and developmental complications that can emerge years after treatment. These effects consist of endocrine disorders, delayed puberty, insufficient growth and development, thyroid dysfunction, and metabolism issues. At a molecular level, Radiotherapy damages crucial molecular pathways such as the PI3K/AKT/mTOR pathway,which plays an important role in cell survival, protein synthesis, and development. The primary aim of this literature review is to display a study-oriented overview of the effects and complications of radiation therapy on pediatric cancer survivors, and evaluate the prevention strategies to reduce long term harm.Current treatments include hormone replacement therapy, fertility preservation, and advanced radiation techniques such as proton therapy and IMRT to minimize the damage caused by radiation. Additionally, prevention, and early monitoring, and radioprotectants add protection against the rays. Future strategies and research opportunities include non-radiative treatments such as immunotherapy, involving CAR-T cell therapy, and targeted inhibitors that provide a more exact treatment. These potential advancements help destroy the cancer cells while also preventing the effects on pediatric cancer survivors through their adult lives.
Keywords: Pediatric cancer, radiation therapy, endocrine dysfunction, hormone imbalance, delayed development, thyroid dysfunction, hormone replacement therapy, CAR-T cell therapy, PI3K/AKT/mTOR pathway, proton therapy
Introduction
Pediatric cancer affects 1 in 285 children in the United States each year1. Pediatric cancer is a group of diseases where abnormal cells grow uncontrollably in children, disrupting normal development and health. The major cancers that affect children are leukemia, brain tumors, lymphoma, and thyroid cancer. Out of all pediatric cancer patients, 30% have leukemia, 25% have brain tumors, 15% have lymphoma, and the remaining patients have other types such as thyroid cancer1. These cancers all cause many effects on the patients and survivors through the damage they cause to the endocrine, which is the system of glands that regulates and secrete hormones into the bloodstream. In recent years, the pediatric cancer survival rate has increased to 85% due to treatment methods such as chemotherapy, immunotherapy, targeted therapy, surgery, and radiation1. Radiation therapy is one of the most commonly used treatments for pediatric cancer patients, where high-energy rays or particles are used to damage the DNA of cancer cells, stopping them from growing and dividing. Radiation is administered daily for 1-8 weeks, depending on the severity of the cancer. The high energy x-rays and gamma rays destroy the cancer cells, but they also damage and have other effects on crucial hormones of the human body. The dosage of radiotherapy can vary, but the usual amount is about 23.4-54 Gray (Gy) for children with brain tumors2. Depending on age and cancer type, radiation and dosage can vary drastically. There are 3 main types of radiation therapy, conventional photon, intensity modulated (IMRT), and proton therapy. Conventional photon radiation therapy delivers a broader entrance and exit dose, making it very accessible, but requiring higher radiation exposure to surrounding organs. IMRT uses regulated photon beams to improve tumor targeting and reduce damage, although it is very complex. Proton therapy uses the Bragg peak, a maximum in the curve of dose that occurs as charged particles travel through matter, to minimize exit dose and limit radiation, but its cost restricts widespread use3 Radiation is widely used, and it has been effective in helping childhood cancer patients survive, but it also has major effects on the pediatric patients.
| Technique | How dose is administered | Advantage | Limitation |
| Conventional photon RT | Broader entrance/exit dose | Very easily accessible | Higher dose necessary |
| IMRT | Regulated photon beams | Better targeting and spares nearby organs | Complexity |
| Proton therapy | Bragg peak reduces exit dose | Lower dose to surrounding organs | cost/access |
Radiation for treating childhood cancer patients is very effective, specifically in brain tumors, due to the specific location. For patients with medulloblastoma, a type of brain tumor, radiation combined with surgery was 80% effective in terminating the cancer (p<0.001 in large scale trials)4. This number demonstrates the change that radiation has ensued for thousands of childhood cancer survivors, yet this is accompanied by many other effects. Due to the dosage of radiotherapy depending on the cancer severity and age of the patient, the effects can grow larger or be less prevalent. The endocrine system is still developing during adolescence, proving to be crucial to the development of children. While radiation remains a prevalent treatment, it damages the developing endocrine tissues in children, leaving them uniquely vulnerable compared to adults5. Additionally, it causes major changes in hormones in the body, such as the growth hormone, T3 and T4, which regulate glucose and sugar levels in the thyroid and GnRH, which triggers puberty to start6. These are all essential hormones to the development of adolescents, and if these are damaged or destroyed, the body cannot function properly and there are many negative effects. These include a delayed growth development, later puberty, hypothyroidism, diabetes, endocrine disorders, amongst others. This paper focuses on the effects of radiation on pediatric cancer survivors, displaying why children are vulnerable, clinical trials being conducted, as well as molecular pathways and future directions to solve these problems.
Methods
This study was conducted as a literature review that examines the endocrine, metabolic, and developmental effects in pediatric cancer survivors. There are 27 studies included through PubMed and Google Scholar between 2000 and 2025 with a focus on 2020-2025,using the keywords pediatric cancer, radiation therapy, endocrine dysfunction, hormone imbalance, delayed development, thyroid dysfunction, hormone replacement therapy, CAR-T cell therapy, PI3K/AKT/mTOR pathway, proton therapy. The inclusion criteria included: primary peer-reviewed research studies involving pediatric patients, human clinical studies and translational animal models, and studies reporting endocrine effects following radiation therapy. The exclusion criteria included: non peer reviewed sources, opinion articles, adult only studies without pediatric relevance, and literature reviews. Articles were screened in order by title, abstract, and full text review. Numerical findings were used including sample size, radiation dose, and reported p-values. A PRISMA style screening was used to ensure correct selection and eliminate any bias.
Discussion
Hypothalamic-pituitary and Endocrine Disorders Caused by Radiation
The hypothalamic-pituitary (HP) axes are the regulatory systems that coordinate hormone secretion throughout the body. HP disorders are often caused by damage to the pituitary and hypothalamus glands, which are sensitive to radiation. The HP axis regulates growth hormone, gonadotropins, and thyroid stimulating hormone, making it essential to the development of adolescents. When a child undergoes radiation therapy to treat cancer, the radiation harms the glands and therefore causes hormone imbalance in childhood cancer survivors6. The glands are usually the most sensitive in the brain, and brain tumors treated by radiation in pediatric cancer patients are the most dangerous to hormone development. In addition to HP disorder, patients experience an endocrine disorder, where their glands do not produce adequate amounts of specific hormones, and cause more health issues such as diabetes and delayed growth. Endocrine disorders affect nearly 50% of all pediatric cancer survivors, and it can develop decades after the patients undergo radiation treatment7.On an organ level, the hypothalamus is a section of the brain that stores the hormones for growth and puberty amongst many other crucial things. On a molecular level, damage to hormone-producing glands from chemotherapy or radiation can alter gene expression and protein synthesis, reducing hormone availability in the body. An example of this is the alteration of protein signaling networks, such as the PI3K/AKT/mTOR pathway. This signaling protein regulates hormones through the pathway of PI3K. It starts in the membrane as PI3K, and once combined with PIP2, another regulating lipid, PIP3 is formed in the cell8.PIP3 is a major growth development lipid, and it also acts as an activation site for AKT. AKT has many effects, such as promoting cell survival, protein synthesis, and ignites growth on the cellular level, which means that without AKT, cells are not as healthy and they are more susceptible to disease and delayed growth. This pathway is extremely crucial to signaling for growth and development of adolescents. When these proteins such as PI3K and AKT are disrupted by cancer cells, the pathway that develops growth and hormone regulation is altered. In this, the radiation needed to fight the cancer cells further blocks the pathway due to the waves. The PI3K pathway by radiation has been linked to reduced hormone signaling, impaired IGF-1 production in the liver, and thyroid follicular cell dysfunction, which together contribute to endocrine phenotypes such as growth hormone deficiency, hypothyroidism, and delayed puberty in pediatric cancer survivors5.The gene alteration and protein synthesis of this PI3K pathway is affected by radiation because it blocks the pathway’s ability to regulate hormones, and promote growth in children5.
As seen below in Table 3, Endocrine disorders cause various health issues such as diabetes, delayed growth, delayed puberty, and thyroid dysfunction. For HP disorders, dose ranges of 18-24 Gy are associated with GHD and puberty disruption. Additionally, in a cohort of n= 90 patients, the prevalence of GH deficiency was significantly higher in patients receiving higher doses (45-60) Gy compared to lower doses (p<0.001)7. This displays the effects that more radiation can cause on pediatric patients. Endocrine and HP disorders are major factors that affect pediatric cancer survivors, and because of the lasting and widespread effects of endocrine disorders, long-term monitoring and early intervention are essential to protect the health and development of pediatric cancer survivors7. Endocrine effects can also emerge 5-15 years after radiation exposure, heightening the need for lifelong surveillance and new treatment methods. Overall, HP dysfunction can be understood through four components: radiation-caused molecular damage to pathways, clinical manifestations such as growth hormone deficiency and delayed puberty, regular monitoring through screening, and early management using hormone replacement therapy and targeted therapy. This approach allows the endocrine dysfunctions to be identified early and reduce long-term effects.
| Radiation site | Dose ranges | Endocrine outcomes | Time period |
| HPA axis | 18-24 Gy More effects with 45-60 Gy | GHD, altered growth, puberty disruption | Years after treatment |
| medulloblastoma | CSI + boost | High chances of GHD | Emerge over 5+ years |
| Thyroid exposure | Thyroid dose varies, risk increases with higher thyroid and HPA dose | Hypothyroidism | Persists long term |
| gonads/uterus exposure | Pelvic RT | Ovarian failure, infertility, premature menopause | Early or delayed |
The Effect of Radiotherapy on Puberty
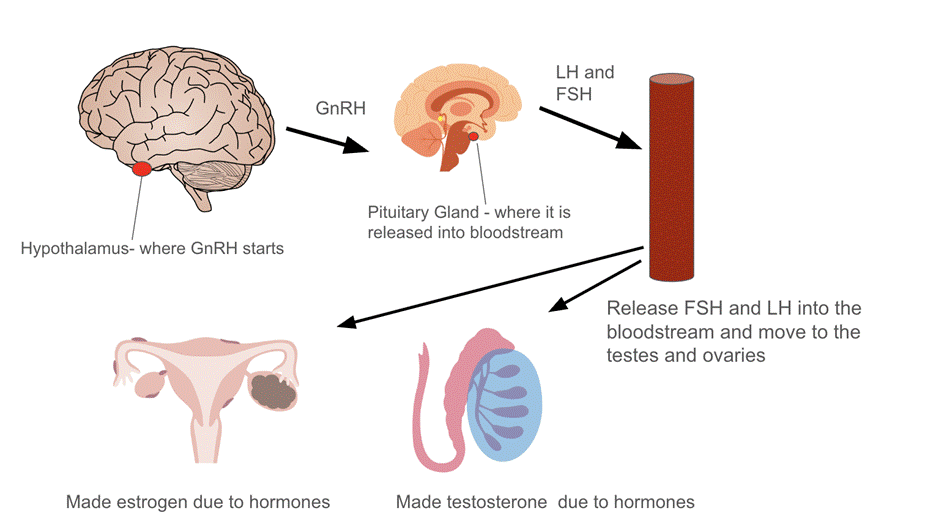
One factor of endocrine disorders in pediatric cancer survivors is delayed puberty. Although this is not life threatening, there are many side effects that can affect the patient’s physical and mental health. The hormone that controls puberty and reproduction is the gonadotropin-releasing hormone (GnRH), which initiates the process for puberty and development6. With radiation affecting the hypothalamus which stores GnRH, this hormone does not get released, therefore delaying puberty in both males and females. A clinical study of pediatric cancer survivors reported that treatment-related radiation exposure was associated with gonadal dysfunction and damaged pubertal development, as shown by the abnormal sex hormone levels. Out of a cohort of 105 survivors, for males the usual testosterone levels are 300-1000 mg/dL, where survivors had less than 300 ng/dL of testosterone. In females, the reference ranges of estradiol are 30-400 pg/mL, where survivors had lower levels than this (p<0.05)9. As shown in figure one, the GnRH hormone starts in the hypothalamus, a specific section of the brain that controls hormones and puberty, where it is stored in the preoptic area and arcuate nucleus, which is a specific part of the hypothalamus made specifically for transporting this gene9. The GnRH is transported from the hypothalamus to the pituitary gland, where it stimulates the release of luteinizing hormone (LH) and follicle-stimulating hormone (FSH), which are both essential for puberty. The LH and FSH then enter into the bloodstream and act on the ovaries and testes to regulate the sex hormone production, showing how both hormones are essential for sexual reproduction10. This spread of GnRH is molecularly how puberty is initiated, and with the radiation waves affecting the brain, this process is greatly affected. This lack of hormones is due to radiation from cancer and impaired hormone glands.
In females, many cancer survivors experienced ovarian failure and fertility. This is also due to the lack of hormones such as GnRH being released along with estrogen, which is essential for reproduction and ovary development in females. Without estrogen, females that have survived pediatric cancer and radiation therapy can face fertility and ovary failure. The cancer that mainly affects this in females is abdominal and pelvic cancer and radiotherapy. Due to this, females can experience premature ovary insufficiency (POI), ovary damage, fertility, gonadotropin deficiency, and HP axis disruption11. All of these issues with the female reproductive system are due to radiation near the abdomen and stomach, further demonstrating the later effects of pediatric cancer on women survivors11.
In male pediatric cancer survivors, patients undergo radiation which causes their testicular cells to become sensitive and even infertile as well. Testicular damage can be due to testicular disease, surgery or radiation therapy to the testes and adjacent tissues, as well as to systemic chemotherapy and or radiotherapy12. Many males also have damage to hormone production in the hypothalamus, making it difficult for growth and puberty hormones to be released, and this results in further health and reproductive issues. Testosterone production is also greatly affected due to radiation near the testes and abdominal area. Gonadyl dysfunction was the most common in males that underwent radiation therapy, which demonstrates how radiation affects males and females in similar ways12. In some cases, fertility may be permanently impaired, leading to the need for future reproductive assistance or preservation strategies. This highlights the importance of early fertility counseling and protective measures prior to cancer treatment13. Many of the issues that follow pediatric cancer from radiation therapy can appear many years post treatment, therefore highlighting the importance of long term surveillance.
In terms of treatment for hormone dysfunction and delayed puberty, as shown in Table 3, hormone replacement therapy (HRT) is widely used to restore the hormones back into the body and start puberty in both males and females14. For females, HRT is used to start breast development and treat POI. For males, HRT is used to restore testosterone levels and prevent infertility. There is another method used for males called electroejaculation, where semen samples are collected and then can be used for HRT and other treatments to preserve hormones15. This treatment is very helpful and a large advancement because it helps raise more possibilities for treatment and hormone balance. Proton radiation therapy can be used to restore ovarian function, and therefore prevent infertility and start puberty11. An additional method of preventing fertility is cryopreservation, where reproductive cells and tissue are frozen and stored before cancer treatment, making it possible for patients to have biological children after radiation treatment16. Lastly, sperm banking is a promising way for male fertility to be preserved through cancer treatment, where a patient’s sperm is collected frozen, and stored before treatment to avoid damage from radiation15.From a clinical perspective, gonadal dysfunction follows a pattern of GnRH signaling disruption, delayed puberty, monitoring through LH, FSH, and other hormone levels, and management through HRT and fertility preservation. Applying this improves early detention and allows patients to be treated faster. Together, these therapies not only address hormone deficiencies but also could support long-term physical, emotional, and reproductive health in survivors.
| Endocrine issue | Monitoring examples | Management |
| GHD | IGF-1 | GH replacement therapy |
| hypothyroidism | Thyroid stimulating hormone | Levothyroxine replacement |
| Female fertility risk | Early counseling | cryopreservation |
| Male fertility risk | Semen analysis, electroejaculation | Sperm banking |
| Reducing radiation exposure | Planning to minimise dose to HPA/ thyroid/gonads | IMRT, proton therapy, prevention |
| Radioprotective agents | Based on risk | Amifostine, palifermin |
Growth and Development Disruptions
The most common effect of pediatric cancer is growth hormone deficiency (GHD), where approximately 10-15% of all pediatric cancer survivors face GHD, depending on radiation dose and field7. One study found a mean height loss of 1.7+/- 0.2 SD in untreated patients (n=15), versus 0.6 +/- 0.2 SD in those treated with GH (n=17). A younger age at irradiation was associated with higher chances of GH deficiency (p<0.03)17. This deficiency is an additional effect of a failed pituitary gland, where patients experience slower growth, increased body fat, reduced muscle mass, and lower blood sugar levels. Growth hormone, or GH, is essential for initiating growth in children. Similar to GnRH, without this, pediatric cancer patients lack the ability to grow and develop normally17. The molecule IGF-1, or insulin growth factor, is the molecule that mediates the GH and therefore overlooks growth in the body. Without proper IGF-1, the downstream pathway is disrupted, therefore leading the growth hormone to be unmediated causing not enough or too much GH, both affecting the cancer patient’s growth18.Clinical studies report significant height deficits in pediatric patients that received more than 18-24 Gy of cranial radiation, with more risk at younger ages18. This evidence was derived from a study conducted on mice, therefore all evidence and conclusions should be interpreted with caution. This additionally displays how children are uniquely vulnerable to the effects of cancer and radiation therapy.This molecule is primarily produced in the liver, but it is also very prominent in the muscle tissue, bone, kidney and brain. Knowing this, the effects of radiation become clear on the IGF-1 production in the body.If the pediatric cancer patient is suffering from any organ-specific cancers such as kidney, liver, brain, or bone cancer, radiation therapy can damage the production of IGF-1 and therefore, there is very little hormone or molecule regulating the growth hormone. Due to this, growth is disrupted and patients develop GHD. In severe cases, the lack of IGF-1 can also impair bone mineralization, leading to brittle bones and increased fracture risk19. This disruption often requires long-term HRT to restore growth and metabolic balance. Furthermore, early action is critical, as prolonged IGF-1 deficiency during key developmental years can cause permanent stature and developmental delays.
GHD is very widespread and common, yet it is also treatable. Through growth hormone replacement therapy, the GH and IGF-1 are restored with the addition of synthetic hormones and the body can return to its normal functions17. GH replacement therapy is where synthetic hormones are injected into the patient, and the injected GH binds to the previous GH cells. After binding, the JAK-STAT pathway is triggered, which is a pathway of cells that also helps the GH promote growth, similar to the P13-K pathway20. Once this is triggered, the effects cascade down the pathway and the liver can therefore send IGF-1 into the bloodstream. Once distributed into the blood stream, adequate levels of GH and IGF-1 help bone density, fat breakdown, and growth. People undergoing growth hormone replacement therapy can see results of increased muscle, increased height, and stronger bones within 3-4 months7. Out of the patients that undergo radiation, 50% have GHD, but with the creation of GH replacement therapy, the survival rates are very high, and usually not life threatening7. Treatment is typically administered through daily injections, which require careful dosing and regular monitoring by an endocrinologist. While side effects are rare, they can include joint pain, swelling, and insulin resistance if doses are not properly regulated. Continued follow-up ensures that therapy is both safe and effective, allowing patients to achieve near-normal growth patterns. GH deficiency can be evaluated using the standardized model that could include damage to the pituitary gland, IGF-1 pathways, slowed growth and reduced bone density, monitoring through growth charts and IGF-1 measurements, and management with dosed GH replacement therapy. This framework allows patients to potentially recover as normal and also support long-term follow-up.
Thyroid Disregulation and Glucose Metabolism
The thyroid, a small gland located in the neck, produces hormones that determine metabolism as well as growth development. The main hormones that regulate metabolism in the thyroid are T3 and T4, which control the speed at which the body uses energy, therefore affecting metabolism5. The process of making these hormones takes place in follicular cells, where they produce and secrete the precursor protein thyroglobulin into the lumen, where iodide is taken from the bloodstream and brought into the cell. Another enzyme in the follicular cell, called thyroid peroxidase (TPO), binds the thyroglobulin protein with iodide, creating T3 and T4. This process happens on a cellular level and is essential for hormone production. When T3 and T4 levels increase, they alert the hypothalamus and pituitary gland to slow down or speed up, keeping hormone levels balanced21.
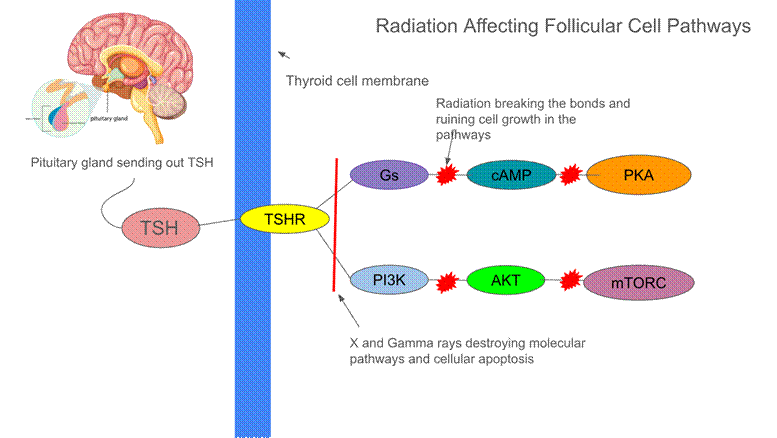
However, when cancer cells and radiation waves affect this gland, the hormones become unbalanced, disrupting metabolism in adolescents. Radiation damage to T3 and T4 hormones can cause weight gain, fatigue, and changes in lipid metabolism, as well as an increased risk of diabetes. The thyroid also regulates sugar levels by controlling how quickly cells use and produce glucose, mainly through the actions of T3 and T4. These hormones increase glucose production in the liver and promote glucose uptake and metabolism in muscles and fat, helping maintain energy balance. If this system is damaged due to radiation, sugar levels can be affected. A cancer patient’s metabolism is highly dependent on proper thyroid function and an adequate amount of T3 and T4. Due to potential damage from radiation, there can be less T4 in the body. A large cohort study of n=14,364 survivors found that for thyroid dose of 20-30 Gy, the excess relative risk for hypothyroidism was 13.4 (95% CI: 10.9-16.6), with a linear dose- response observed across all ranges of doses (p<0.001)21. Without these hormones being released into the bloodstream, thyroid issues such as hypothyroidism and diabetes become a major risk. Diabetes is caused by the body’s lack of insulin production, and this can be triggered by radiation to the thyroid. Radiation lowers thyroid hormone levels, slowing down the body’s metabolism and making it harder for cells to use insulin. Over time, the pancreas, which produces insulin, can become stressed and fail to make adequate amounts. In a study including 20,762 survivors, and 4,853 siblings as controls, survivors exposed to abdominal radiation were 2.92-fold more likely to develop diabetes than siblings (95% CI: 2.02-4.23; p<0.001 for interaction with age)2. Additionally, from a study about radiation resistance, the PI3K/AKT/mTOR signaling pathway has been linked to radiotherapy resistance. This pathway regulates apoptosis and gene transcription, with mTORC1 activity downstream of AKT playing a key role in cellular survival. This pathway helps radioresistance, and an added pathway is the TSH/TSHR/cAMP/PKA pathway, where the pituitary gland releases TSH, or thyroid stimulating hormone. The TSH binds to a TSH receptor (TSHR) in the cell membrane, which activates protein, specifically Gs22. Once this signaling pathway is activated, adenylyl cyclase produces cAMP, a messenger that stimulates thyroid hormones, and also activates protein kinase A (PKA). PKA is an enzyme that is triggered when cAMP levels increase, and it targets proteins within the thyroid cell and translates the cAMP signals in the cell to metabolism and growth. Clearly, without PKA and cAMP, metabolism and cell growth can be impacted greatly, and this is the issue caused by radiation on a molecular level23. The cAMP/PKA pathway is different from the PI3K/AKT pathway because cAMP leads to the activation of PKA, which regulates thyroid hormones, while the PI3K pathway instead mediates cell survival, and growth through signaling that does not involve cAMP.These pathways connect because they both stem from TSH and TSHR, where they then split to fulfill different goals in the cell but still protect and promote growth. When these pathways are disrupted by cancer, radioresistance diminishes, further affecting hormone regulation and metabolism. Specifically, this means T3 and T4 can be cut off or significantly decreased in the thyroid. Disruption of this pathway can lead to endocrine phenotypes such as diabetes and hypothyroidism23. Radiation can damage the cellular bonds in the pathways and affect the DNA as well. With all of these aspects ruined in the cell, the follicular cell is unable to carry out basic functions and is therefore greatly affecting the thyroid. In figure 2, the molecular level of these 2 pathways display how radiation can affect the thyroid hormones, and lead the cells to be destroyed. Because of this, children who go through cancer treatment with radiation have a higher chance of developing diabetes and metabolism issues later in life.
Besides metabolism and diabetes, the thyroid also has its own independent risks that are due to radiation. Hypothyroidism is a common risk, as stated from the Childhood Cancer Survivor Study, where out of a group of 14,364 five-year survivors of childhood cancer, 2,015 survivors had hypothyroidism due to radiation therapy21. This large number of patients portrays the effect that cancer and radiation has on thyroid independent health issues. Radiation can also cause thyroid nodules or even thyroid cancer to develop years after treatment, which is an additional risk added to hypothyroidism. Many survivors need lifelong monitoring of thyroid function to make sure hormone levels stay balanced. In some cases, patients must take daily thyroid hormone replacement to keep their body working normally. This is similar to GH replacement, where synthetic hormones are injected and binded to previous cells, therefore the hormones such as T3 and T4 can be restored back to normal. Without this replacement, the body can be deprived of T423. In a study measuring amounts of T4 before and after radiation, the results were that T4 was significantly lower in patients with radiation therapy (p=0.044)22. To minimize the effects of radiation on hypothyroidism, patients can be given synthetic thyroid hormone, specifically levothyroxine, or synthetic T4. This medicine replaces the hormone that the damaged thyroid is no longer able to produce.There are many effects of radiation and cancer on the thyroid and metabolism in childhood survivors, and these effects stay with the patients well into their adult years, therefore affecting their lives for many years post survivorship. Thyroid and metabolic dysfunction can be analysed through radiation induced cellular and molecular disruption, routine monitoring of TSH, T3, T4, and management with levothyroxine. Using this approach promotes early treatment for patients and reduces the effects of radiation therapy.
Limitations
This review is limited by differences in how radiation doses are reported, varying lengths of follow-up, and reliance on observational studies. Many of the biological explanations are derived from animal studies, which may not completely apply with children. Lastly, endocrine effects can also appear years after treatment, therefore long-term data is limited.
Conclusion
While radiation therapy significantly improves pediatric cancer survival, it introduces many long-term endocrine risks that can disrupt growth, puberty, metabolism, and fertility. There are certain limitations, such as hormone imbalance and organ failure, and the short term limitations can include fatigue, hair loss, and nausea. To manage these problems, there are different ways using hormone replacement therapy (HRT), targeted molecular therapies and specific radiation therapy. In HRT, hormones such as T3 and growth hormone can be synthetically replaced, and therefore restored back to their normal function17. In targeted molecular therapies, drugs target the pathways that are most affected by radiation, such as the PI3K/AKT/mTOR pathway. Although these treatments are very helpful to survivorship, ultimately the best cure to the effects of radiation is prevention. If the tissue and cells are protected before even starting radiation, the effects can be prevented, therefore potentially curing the cancer while also taking care of the crucial hormones and organs. This should be executed by distributing drugs into the tissue that protects against the radio waves before starting treatment. There are 3 types of radiation protection drugs, radioprotectors, which prevent damage before starting radiation, radiomitigators, given during treatment to minimize damage, or radiotherapeutics, which are given after treatment to fix damage already done24. The best strategy in terms of drugs for radiation therapy would be to distribute radioprotectors to prevent damage at all, except there are many side effects to these so they are not commonly used right now. Radiotherapeutics are the most commonly used because they prevent the side effects and are the most widely researched24. In the near future, due to clinical trials and research, radioprotectors are becoming more popular because they reduce the side effects from prevention. Examples of these drugs are Amifostine and Palifermin, both radioprotective drugs that promote growth and prevent damage such as tissue damage24. These drugs can cause side effects such as vomiting and nausea, yet they have been FDA approved to help prevent radiowave damage from X rays and gamma rays. They are distributed right before treatment, and aim to protect against the rays. Radiation damage in children can be minimized using precise techniques like proton therapy and IMRT (Intensity-Modulated Radiation Therapy), which focus treatment on tumors while sparing healthy tissues. In proton therapy, protons are used instead of X rays to kill the cancer cells, and IMRT is where there are more targeted doses of radiation distributed directly to the cancer instead of around it as well. A multi-center study reported that the 10 year cumulative incidence for in-field malignant secondary tumors after proton therapy was 0%, suggesting potential benefits over radiation therapy3. Radioprotective drugs, antioxidants, and optimized dosing schedules further reduce cellular and DNA damage. Regular monitoring of hormone levels and early intervention with hormone replacement or lifestyle adjustments can prevent long-term metabolic and endocrine complications19. With these treatments, the organs are prevented and guarded from damage right away, instead of working to fix them after radiation therapy already damaged them. With this, the effects of radiation can be fixed, and the pediatric patients can grow and develop normally while undergoing radiation therapy for cancer.
Since radiation has the ability to damage tissues, immunotherapy and targeted therapies would be a promising way to minimize the damage on hormone production in pediatric cancer patients. Current approaches focus on how to adapt radiation, but future methods could get rid of radiation altogether. CAR-T cell therapy and immune checkpoint inhibitors such as PD-1 inhibitors and PD-L1 inhibitors, have proven to be effective in clearing the cancer without the use of radiation25. These inhibitors control the T cells in the body and train them to fight off cancerous cells. CAR-T therapy is a type of immunotherapy where the patient’s own T cells are engineered to fight the cancer cells. In a clinical study about the long term follow up of this therapy, CAR-T cell therapy has demonstrated significant clinical efficacy in pediatric patients with relapsed or refractory B-call acute lymphoblastic leukemia; long term follow up data shows that this therapy can control the disease in many cases26. This is a promising way to exterminate pediatric cancer without the effects that follow. MTOR inhibitors and PI3K pathway targeting drugs are being researched to specifically block cancer growth signals without damaging surrounding tissues. These methods could help protect against cancer without the use of radiation, therefore avoiding the effects such as delayed puberty and growth.
| Approach | What it replaces |
| CAR-T cell therapy | Need for intensive chemo/radiation therapy |
| Immune checkpoint inhibitors | In some tumors may reduce reliance on radiation |
| Targeted pathway inhibitors | Broad radiation therapy need |
While these immunotherapies show great potential, they also include limitations such as relapse risk and toxicity to the body and organs. The cost and accessibility of these emerging strategies must also be a significant limitation to consider.Lastly, combination therapies are combined treatments that fight the cancer cells, which is another way of avoiding radiation or minimizing the rays admitted, and this is another potential idea for exterminating cancer27. A prospective clinical trial combining radiation with systemic immunotherapy demonstrated that a subset of patients had significant benefits, including complete tumor responses. This highlights how combination therapies can also improve treatment outcomes without singular approaches27. There are many research projects being carried out that are testing these treatments, and they display potential to help pediatric cancer patients and avoid the issues that follow radiotherapy. These innovations and ideas could help pediatric cancer patients survive and allow them to be able to live a normal life after treatment instead of facing the effects of radiotherapy well into their adult lives.
Acknowledgements:
Jenny An
References
- A. T. Meadows. Pediatric cancer survivorship: research and clinical care. Journal of Clinical Oncology. Vol. 24, 5160–5165 (2006). https://doi.org/10.1200/JCO.2006.07.3114. [↩] [↩] [↩]
- D. N. Friedman et al. Radiation dose and volume to the pancreas and subsequent risk of diabetes mellitus: a report from the Childhood Cancer Survivor Study. JNCI: Journal of the National Cancer Institute. Vol. 112, 525–532 (2020). https://doi.org/10.1093/jnci/djz152. [↩] [↩]
- A. Ruggi et al. Toxicity and clinical results after proton therapy for pediatric medulloblastoma: a multi-centric retrospective study. Cancers. Vol. 14, 2747 (2022). https://doi.org/10.3390/cancers14112747. [↩] [↩]
- A. Gajjar et al. Outcomes by clinical and molecular features in children with medulloblastoma treated with risk-adapted therapy: results of an international phase III trial (SJMB03). Journal of Clinical Oncology. Vol. 39, 822–835 (2021). https://doi.org/10.1200/JCO.20.01372. [↩]
- P. K. Duffner et al. Long-term effects of cranial irradiation on endocrine function in children with brain tumors: a prospective study. Cancer. Vol. 56, 2189–2193 (1985). https://doi.org/10.1002/1097-0142 [↩] [↩] [↩] [↩]
- W. Chemaitilly et al. Hypothalamic-pituitary axis dysfunction in survivors of childhood CNS tumors: importance of systematic follow-up and early endocrine consultation. Journal of Clinical Oncology. Vol. 34, 4315–4319 (2016). https://doi.org/10.1200/JCO.2016.70.1847. [↩] [↩] [↩]
- Z. Yavaş Abalı et al. Long-term endocrinologic follow-up of children with brain tumors and comparison of growth hormone therapy outcomes. Turkish Archives of Pediatrics. Vol. 58, 308–313 (2023). https://doi.org/10.5152/TurkArchPediatr.2023.22147. [↩] [↩] [↩] [↩] [↩] [↩]
- Y.-A. Chen et al. Antrocin sensitizes prostate cancer cells to radiotherapy through inhibiting PI3K/AKT and MAPK signaling pathways. Cancers. Vol. 11, 34 (2018). https://doi.org/10.3390/cancers11010034. [↩]
- J. Y. Yoon et al. Gonadal and sexual dysfunction in childhood cancer survivors. Cancer Research and Treatment. Vol. 49, 1057–1064 (2017). https://doi.org/10.4143/crt.2016.197. [↩] [↩]
- A. Cattoni et al. Hormonal replacement therapy in adolescents and young women with premature ovarian insufficiency. Blood Reviews. Vol. 45, 100730 (2021). https://doi.org/10.1016/j.blre.2020.100730. [↩]
- J. P. Gross et al. Proton radiotherapy to preserve fertility and endocrine function. International Journal of Radiation Oncology*Biology*Physics. Vol. 109, 84–94 (2021). https://doi.org/10.1016/j.ijrobp.2020.07.2320. [↩] [↩] [↩]
- C. A. Sklar et al. Effects of radiation on testicular function in childhood leukemia survivors. Journal of Clinical Oncology. Vol. 8, 1981–1987 (1990). https://doi.org/10.1200/JCO.1990.8.12.1981. [↩] [↩]
- J. E. Campbell et al. Fertility preservation counseling for pediatric cancer patients. Journal of Adolescent and Young Adult Oncology. Vol. 5, 58–63 (2016). https://doi.org/10.1089/jayao.2015.0040. [↩]
- A. Haavisto et al. Reproductive late effects and testosterone replacement therapy in male childhood cancer survivors. International Journal of Cancer. Vol. 154, 2121–2131 (2024). https://doi.org/10.1002/ijc.34890. [↩]
- I. Gat et al. Sperm preservation by electroejaculation in adolescent cancer patients. Pediatric Blood & Cancer. Vol. 61, 286–290 (2014). https://doi.org/10.1002/pbc.24752. [↩] [↩]
- J. Hafele et al. Fertility preservation in pediatric oncology: a retrospective study. Cancers. Vol. 17, 3615 (2025). https://doi.org/10.3390/cancers17223615. [↩]
- P. E. Clayton et al. Growth response to growth hormone therapy following craniospinal irradiation. European Journal of Pediatrics. Vol. 147, 597–601 (1988). https://doi.org/10.1007/BF00442471. [↩] [↩] [↩] [↩]
- S. Tarantini et al. IGF-1 deficiency influences vascular aging in mice. Age. Vol. 38, 239–258 (2016). https://doi.org/10.1007/s11357-016-9943-9. [↩] [↩]
- J. H. Kim et al. Diagnosis and treatment of growth hormone deficiency. Endocrinology and Metabolism. Vol. 35, 272–287 (2020). https://doi.org/10.3803/EnM.2020.35.2.272. [↩] [↩]
- Q. Hu et al. JAK/STAT pathway: extracellular signals and therapeutic regimens. Frontiers in Bioengineering and Biotechnology. Vol. 11, 1110765 (2023). https://doi.org/10.3389/fbioe.2023.1110765. [↩]
- P. D. Inskip et al. Hypothyroidism after radiation therapy for childhood cancer. Radiation Research. Vol. 190, 117 (2018). https://doi.org/10.1667/RR14888.1. [↩] [↩] [↩]
- H. J. Lee et al. Subclinical hypothyroidism in childhood cancer survivors. Yonsei Medical Journal. Vol. 57, 915–922 (2016). https://doi.org/10.3349/ymj.2016.57.4.915. [↩] [↩]
- W. Chemaitilly et al. Primary hypothyroidism in childhood cancer survivors. Cancer. Vol. 128, 606–614 (2022). https://doi.org/10.1002/cncr.33969. [↩] [↩] [↩]
- D. M. Brizel et al. Phase III randomized trial of amifostine as a radioprotector. Journal of Clinical Oncology. Vol. 18, 3339–3345 (2000). https://doi.org/10.1200/JCO.2000.18.19.3339. [↩] [↩] [↩]
- N. N. Shah et al. Long-term follow-up of CD19 CAR T-cell therapy. Journal of Clinical Oncology. Vol. 39, 1650–1659 (2021). https://doi.org/10.1200/JCO.20.02262. [↩]
- D. Sun et al. CAR-T cell therapy: a breakthrough in cancer treatment strategies. Molecular Medicine Reports. Vol. 29, 47 (2024). https://doi.org/10.3892/mmr.2024.13171. [↩]
- S. M. Hiniker et al. Radiation therapy combined with systemic immunotherapy in metastatic melanoma. International Journal of Radiation Oncology*Biology*Physics. Vol. 96, 578–588 (2016). https://doi.org/10.1016/j.ijrobp.2016.07.005. [↩] [↩]