Abstract
Introduction: Childhood apraxia of speech is a rare motor-speech disorder linked to development disorders such as ASD and Down syndrome. This paper explores the relationship between sequential processing deficits in both fine motor rhythmic movement and speech articulation, exploring whether rhythmic motor intervention can improve CAS outcomes through three questions: (1) What are the anatomical and functional connections between motor and speech in children with CAS compared to children with typical development? (2) How does imitation and repetitive practice influence speech production? (3) How does rhythmic motor movement intervention influence speech production? Method: Records were searched on PubMed and Science Direct using booleans operators, filtered by publishing date, peer review, study and sample type, and relevance. Results: (1) Findings indicate that deficits in the cerebellum, Broca’s area, basal ganglia, thalamus, primary motor cortex, SMA, articulate fasciculus, and impairment in the deep cerebellar nuclei underlie disruption in the feedforward circuit. (2) Motor therapies produce long-term speech improvements with neuroimaging evidence suggesting associated cortical thinning. (3) Parallels between rhythmic fine motor control and oral articulation may be mediated by mirror neurons activating the feedforward system. Music therapy paired with traditional intervention may provide rhythmic cues that improve articulation. Discussion: Generalizability is limited by CAS comorbidities, broad age ranges, low prevalence, and restricted regions of interest and invite speculation. Future research should longitudinally track neuroanatomy and pursue larger sample sizes. Machine learning also presents opportunities for detecting larger and more subtle multivariate patterns that focused ROI analysis cannot.
Introduction
Childhood Apraxia of Speech (CAS) is a widely unknown motor-speech disorder interfering in transcriptional development of the speech plan-production pathway1. Essentially while the speech muscles prove strong, the brain is unable to carry out accurate results during speech articulation and production due to deficits in motor planning and programming. This disconnect occurs from microscopic abnormal development in multiple coordination control sections of the cerebral cortex2,3. CAS is a highly comorbid, rare, heterogenetic disorder often accompanied by other neurodevelopmental speech and neurological disorders including Autism Spectrum Disorder (ASD), intellectual disorders, epilepsy, dysarthria etc. and appearing in about 0.1% of children1,4.
Three common defining symptoms of CAS are (1) inconsistent errors in vowel and consonant acquisition, (2) inappropriate prosody, and (3) disrupted coarticulatory transitions between sounds5. More specifically, CAS is described as having reduced speech time and sequencing due to processing deficits in the internal speech model2. The DIVA model explains cerebellum and cerebral cortex impairment that result in the motor programming deficiencies of CAS6.
By far, the pathogenic FOXP2 gene variant proves to be the most common biomarker of CAS after a groundbreaking study found a rare missense mutation in the KE family7. FOXP2 is a gene that codes transcription factors involved in modulating synaptic plasticity, neural development, axon guidance in speech-language pathways8. Recently, more studies regarding the genetic basis of CAS have uncovered about 30 genes implicated in CAS and accompanying neurodevelopmental disorders including the highly pathogenic SETBP1 loss-of-function variant1,9. Yet, specific etiology for CAS still requires more research. Due to its wide phenotypic spectrum, CAS exhibits a broad range of multifactor symptoms that overlaps with many other speech-language-disorders, therefore there is no single test that confirms diagnosis. Instead, multiple factors including multiple types of SLP tests and motor deficit symptoms are required.
Current long-term speech therapy for speech disorders CAS focuses on early intervention based on visual and auditory feedback, specifically imitation to improve speech motor coordination5. Within the past two decades, emerging research regarding the intricate crossovers between motor and speech processing and production pathways offers possibilities in the field of CAS therapy. This review explores a relationship between fine motor movements and speech production and its effect on speech development children with speech-language disorder. We do so through three questions. First, what are the anatomical and functional connections between motor and speech in children with CAS compared to children with typical development? Second, how does imitation and repetitive practice influence speech production? Lastly, how does rhythmic motor movement intervention influence speech production?
Method
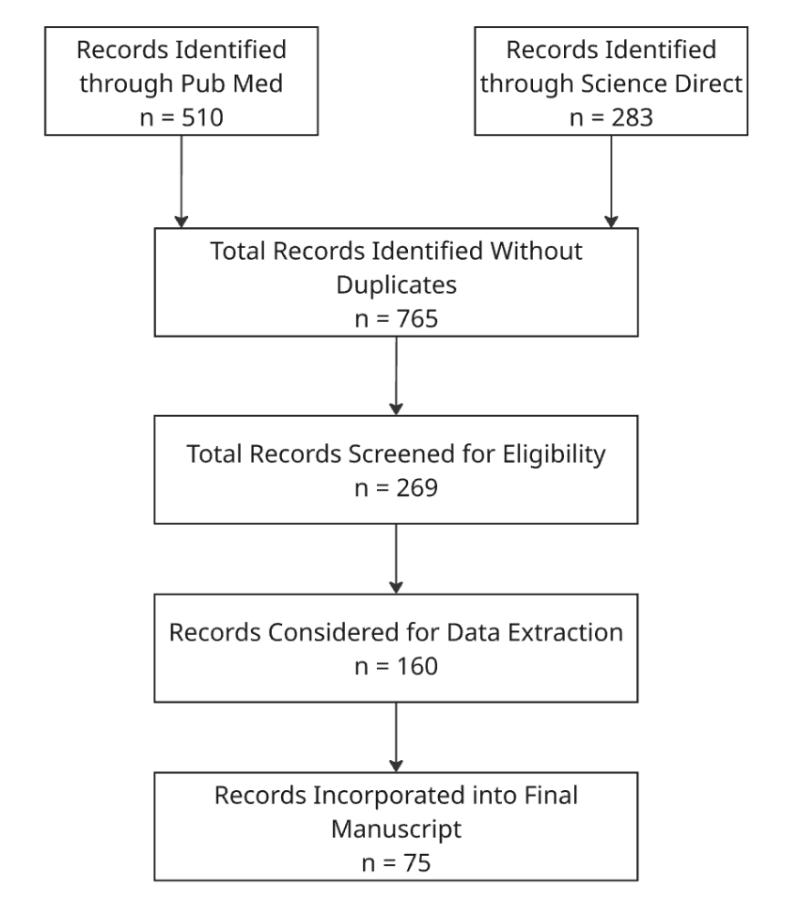
Search Strategy: Keywords and mesh terms were used to identify records included Childhood Apraxia of Speech, neuroimaging, white matter, fMRI, diffusion tensor imaging, fractional anisotropy, neuroplasticity, motor learning, imitation, mirror neurons, speech therapy outcomes, music therapy, melodic intonation, rhythmic tapping, tactile cueing, motor intervention, DTTC, NDP3, ReST, and PROMPT. Booleans “AND” and “OR” were used to ensure results were relevant to CAS or foundational information on speech-motor networks. Records were published between January 2000 and March 2026 to ensure imaging and therapeutic practices were contemporary. Any records before January 2000 are foundational references.
Inclusion and Exclusion Criteria: Each record must be published in peer-reviewed journals and be of relevance to the three questions posed. The types of studies included were reviews, meta-analysis, controlled clinical trials, case studies, imaging or computer model studies. Some foundational documents included book chapters. Sample types including age group, inherited or acquired speech disorder, and monolingualism were considered. Due to the rarity of CAS, many papers reference older age groups and various speech language impairments as well. Any claims made in regards to these papers are speculation needing more research and must be approached with limitations.
Screening Process:
Data Extraction: Data was extracted based on sample size, age range, author, year, methodology, findings, outcome measures, limitations, and relevance to the three questions.
Controlled clinical trials and meta-analysis with relatively larger sample sizes were considered more valuable. Some case studies with detailed, promising findings were considered highly valuable, although more research with larger sample sizes are required for significant claims.
Neuroanatomical Connections and Abnormalities
Sequential Processing and Memory in Speech
Speech formalization is an intricate process that can be explained through multiple models. One of the earliest models of speech, the WEAVER++ model, was used in Levelt’s theory of lexical access that indicates discrete processing stages2,10. After conceptualization, Levelt describes speech formalization to continue with lexical selection, then morphological encoding, phonological encoding, and end with phonetic encoding before the next stage: articulation10. During lexical selection, syntactic words known as “lemmas” are pulled from memory to grammatically encode arguments relating to the semantic message created during conceptualization11. During morphological and phonological encoding, the brain accesses phonological memory codes to generate each word’s syllabification, syllable by syllable, and create a phonological score11. The phonetic encoding system then generates a sequence of articulatory gestural scores and parameters such as pitch, duration, vocal movement, amplitude etc.11.
The WEAVER++ model is known to be a feed-forward model with each stage being discrete, but despite evidence of discreteness by the tip-of-the-tongue phenomena, recent evidence suggests a more parallel course where specific parts draw upon each other12. However, WEAVER++ was designed around picture-naming paradigms in typical Dutch and English speakers, limiting its application to simple word production in non-disordered speech10,12. It does not account for the sequential processing deficits in CAS. After Levelt’s initial analysis of the WEAVER++ model, various other models incorporating other aspects of speech developed, some analyzing speech errors as well. The DIVA model is specific to sequential processing errors in CAS, focusing on the impaired phonetic encoding and motor programming stage for articulation6,13,14. Specifically, results from literature generally agree that the DIVA model shows a higher reliance on the feedback system than the feedforward system which causes the slower articulation speed and neural disconnect3,6,15,16.
The feedback system, as indicated by Guenther and colleagues, has a parallel correcting mechanism unlike the feedforward system, slowing down motor programming2. Yet, during speech production, if an error is detected, the feedback system accompanies the feedforward system to produce coherent rapid articulation2. The model itself, similar to WEAVER++ exhibits some limitations compared to real-life articulation. Researchers used simple syllables in short phrases which omitted many aspects of articulation, and this model is unable to differentiate between cued and uncued speech, an important variable in daily speech. There are some neural mechanisms that are not discussed, and some limits to the biological plausibility of the model such as cellular activation patterns. A specific computational study on a sample of CAS patients clarified that overreliance on the feedback system also leads to increase in variability of vowel (p < 0.01) and consonant production (p < 0.05) depending on placement rather than type of sound, leading to carry-over articulation6. This study also relied on computer simulations, measuring only articulation during sound production, had less focus on phonological development, a crucial component of the feedforward and feedback system, and showed inaccurate increased change in carry-over due to neuromotor constraints6. In reality, speech is more complex. Despite these limitations, somatosensory and auditory influence on timing disruptions and inaccurate articulation is proposed as a reason for increased feedback and error correction6. Although due to low prevalence, finding participants with idiopathic CAS is a large drawback for studies like these, the DIVA model provides close comparisons to fMRI activity in CAS from other studies2.
Sensory information is crucial to the feedback loop. Speech perception strengthens connections between the speech-sound map that hosts sound sequences and sensory encoders which draw from past auditory information to form accurate phonetic codes2. Studies have tested the influence of auditory sensory information on the feedforward system, supporting the hypothesis made by Guenther and colleagues15,16. A masking study with 30 children showed the 9 CAS children with significant differences in voice onset time, vowel duration, vowel space area, and speech intensity compared to typical development children due to heavy reliance on auditory feedback16. Although the results align with studies on sensory reliance in CAS, there are multiple limitations including the incredibly small sample size and no set diagnostic plan for recruitment in CAS. The 2-pronged criteria for participant eligibility including a Goldman–Fristoe Test of Articulation 2 rating and past diagnosis, while identified CAS status, doesn’t necessarily account for difference in comorbidity. For the VOT tests, while the effect size was large (ŋ2 = 0.22), the power (p = 0.6) was relatively low indicating the possibility of false positives. Such limitations are common considering CAS’s low prevalence, comorbidity, and cross-linguistic differences.
Beside the lack of sensory input, working memory deficits could also be a reason for sequential processing dysregulation.
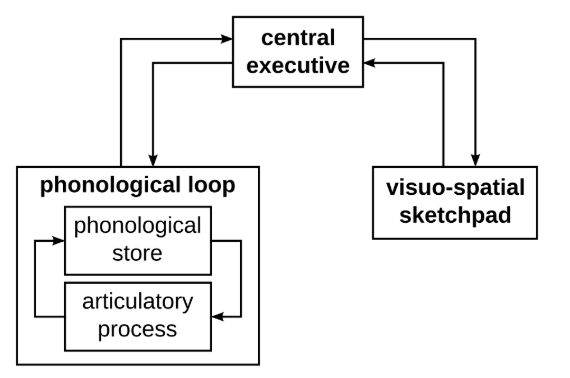
The phonological loop is the part of working memory that stores auditory information for immediate use. There are 2 major parts of the phonological loop as shown in Figure 1: the phonological store and the articulatory control process. The phonological store remembers the temporal order of sounds while the articulatory control process acts like an ‘inner voice’, repeating information to prevent memory loss. Researchers suggest the phonological loop to be impaired, causing mis-sequences during phonetic encoding and carryover downstream in later transcoding processes3,17,18,19,20.
This theory is clearly depicted in a comparative study including 40 CAS participants with a mean age of 11 years20. In each syllable repetition task testing memory, encoding, and transcoding, the CAS group performed lowest compared to the typical, speech delay, and language impairment groups. Effect size for encoding compared to the typical group’s performance was large (d = 0.92) while transcoding was very large (d = 1.36) and memory extremely large (d = 2.77). A higher proportion of idiopathic CAS exhibited better transcoding results suggesting that, although the difference was not significant, accompanying neurological disorders can influence motor programming and results should be cautiously generalized. Shriberg and colleagues suggest that the addition of syllables by the CAS group could be due to increased processing demands, similar to the other groups. This study is a highly comprehensive view at how memory, encoding, and transcoding go together. The relatively large sample size and differentiation between idiopathic and comorbid CAS provides a clearer look at sequential processing deficits, and future studies should consider this.
Cerebellum
The cerebellum has long been understood as a balance, posture, and movement coordinating hub in the brain. Emerging research suggests a larger role in emotional, cognitive, and linguistic processing. A clear example is the theory known as dysmetria of thought, a theory first introduced by Schmahmann outlining the cerebellum’s role in detecting and correcting cognitive or emotional mismatches between intended and actual outcomes21.
In 1998, Wolpert, Miall, and Kawato first established the idea of a dual internal model system, the “Smith model”, operating in the cerebellum for motor programming and planning: an inverse model that generates motor commands through prediction, and a forward model that incorporates sensory information from an efference copy from the cerebrum to correct mismatches22. Through an on-line model of motor control, the cerebellum is able to use predicted movement from an efference copy delivered by the motor cortex and sensory input to rapidly compute sensory prediction errors (sPE)23. Then, the sPE is relayed to designated subcortical structures to correct for them23.
A recent review on the model shows how the cerebellum’s anatomy also provides evidence for the internal model. First, mossy fibers carry the sensory input from the efference copy to granule cells and then parallel fibers through the pontine nuclei, initiating an expansion of information, while climbing fibers carry error signals to Purkinje cells23. Simultaneously, some information is transferred to the deep cerebellar nuclei (DCN), which later incorporates the compressed output from the Purkinje cells and relays it back to the cerebral cortex through the dentate nucleus23. The Purkinje cells use the information to not only compute an sPE, but also modify its synaptic weights to improve future predictions23. While this model is extensively supported throughout literature, the exact mechanism by which the efference copy and sPE is delivered is still unclear. Yet, this model supports the idea of a cerebellar deficiency in the DIVA model and CAS encoding-output mismatch.
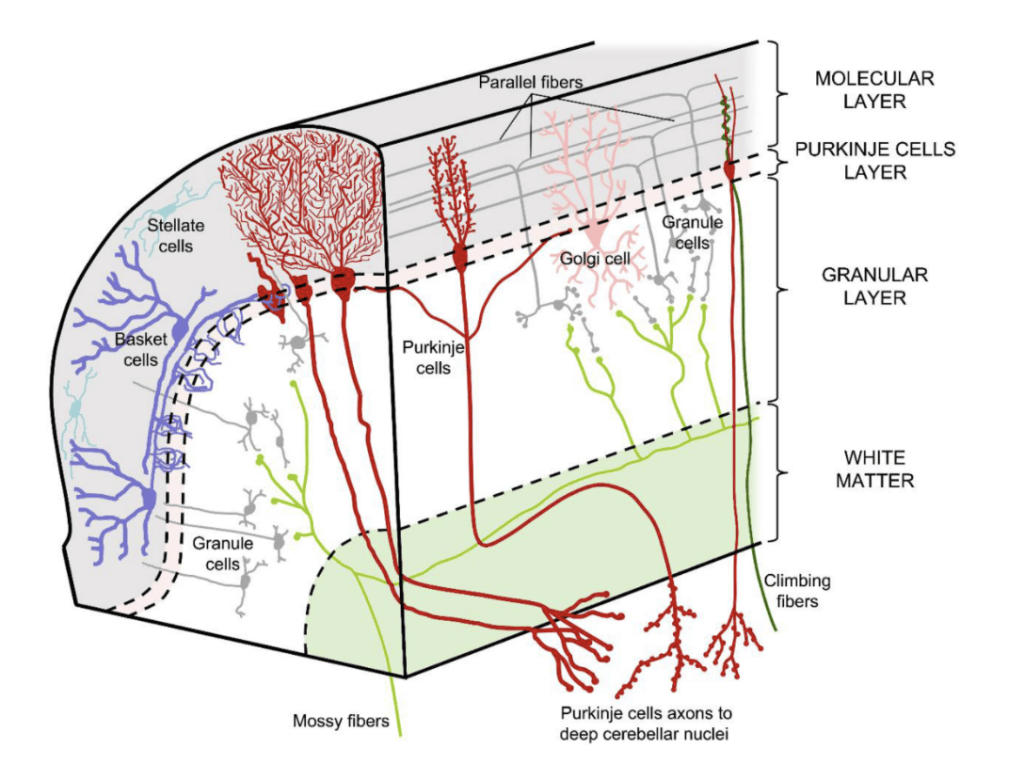
Direct gross motor evidence regarding internal model impairment in CAS is limited, but converging evidence from cerebellar lesion studies in children and ataxic adults indicate compromise of the DCN, the comparator hub, causes both gross and fine motor impairment24,25. This literature focuses on non-oral motor movements including visual, gross, and hand motor movements; therefore, relations to CAS are purely inferential. Early research indicates age-related differences, yet recent studies suggest DCN impairment affects feedforward indiscriminately regardless of age, therefore more research focusing on children is required24,26. Handedness and region impairment affecting fine oral movements should also be considered.
Feedforward control increases with age, an impaired process in CAS27. A recent large cross-sectional study indicated a reliance on the feedback system before adolescence28. Younger children performed better on the Push ball task (5–6yo p = 0.04), allowing for both feedforward and feedback, than the Launch ball task, allowing for just feedforward control, compared to older children who were more precise, accurate, and faster (13-14 yo p = 0.76)28. Although the study was conducted online under 2 hrs to limit inattention, age differences could influence the rate of attention between younger age groups. There is also the high possibility of false positives considering a Bonferroni test was used with over 200 trials across the 4 blocks in both tasks. Contradictory to other visuospatial studies, researchers found no significant effect from sex in the younger age group29.
From birth to 5 years, the cerebellum highly resembles the topography, functional organizations, and lateralization of the adult brain, although functional networks generally concentrate into a more focal distribution at 36 months, much younger than originally thought30. But, only after 24 months does connectivity between higher-order networks strengthen beyond that of primary-order networks30. Research regarding higher-order networks’ connectivity in the cerebellum, specifically the deep cerebral nuclei, is limited. Future studies on the neuroanatomy of the cerebellum in children need to further address the effects of sex-based differences, nutrition, handedness, social interaction, genetics, and socioeconomic differences on development.
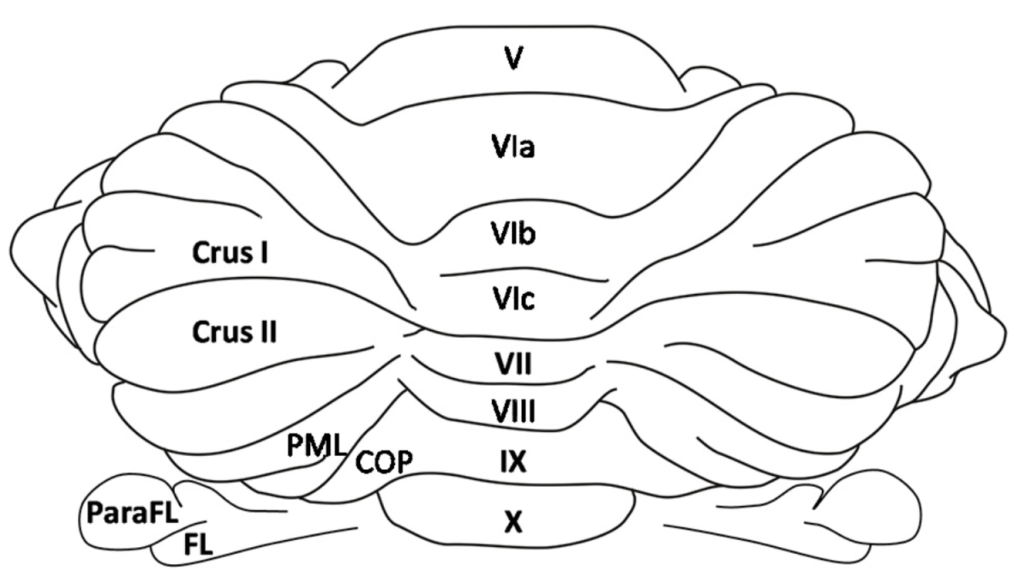
The cerebellum is topographically organized following an anterior sensorimotor region to a posterior higher-order transmodal region between which holds areas involved in working memory, specifically Lobule VI/VII which is implicated in articulatory loop rehearsal of the phonological loop, and Crus I/II31,32,33. Working memory has two main parts: the phonological storage system localized to parietal inferior regions and the articulatory control system localized to the frontal superior regions where lateral regions support covert speech and anterior regions support covert speech34,35. Lobule VIIB corrects errors and guides rehearsal in the articulatory loop during motor programming by comparing information to the phonological loop, using sensory information to predict and correct35. This is also evident in children where imaging shows lateral regions of Crus I to activate during motor programming for oral regions such as the tongue30.
Imaging evidence of cerebellar deficits in CAS is very limited. However, evidence from FOXP2 mutations in rodents and the KE family implicated in verbal dyspraxia indicate reduced gray matter volumes in Crus I, Lobule IV–VI, and Lobule VIIb-VIIIb36. Specifically, the study indicated a consistent 20% volume reduction in Purkinje cells over 3 periods of time in bilateral Crus I (p >0.1)36. Among the affected members were children from 9 years old, although the mean age ranged between 15 – 34 years old. Few studies suggest general white matter deficits in the cerebellum; however, other studies comparing CAS to other neurocognitive disorders indicate no significant deficits in the cerebellum37.
Together these findings suggest the cerebellum’s role to be more pronounced in motor planning, programming, and phonological rehearsal. The specific deficits including working memory, the internal model, and sensory input are debated among researchers. Current neuroimaging evidence from pediatric populations with CAS also remains too sparse to draw conclusions from. More research on cerebellar-focused imaging in CAS is required with ROIs at Lobules VI-VIII and Crus I.
Functional Connectivity
Beyond the cerebellum, many cortical areas are involved in the feedforward model as discussed earlier. Hickok and Poppel distinguished the speech pathway into two distinct tracts, the ventral for speech preparation and dorsal stream for sound articulation mapping38. The dorsal stream is known to be the sensorimotor planning tract for speech and language compared to the ventral stream which controls semantic programming and comprehension38. Within the dorsal stream, the cerebello-thalamo-cortical (CTC) tract, extending from the cerebellum through the thalamus to cortical structures, is the backbone of the feedforward circuit directing movement initiation39. White matter and gray matter volume deficits in this tract have been implicated in CAS including regions like the supplementary motor area (SMA), primary cortex (M1), and Broca’s area40. There are some structures which prove more vital to the feedforward loop than others. It is important to understand not only how they function in the circuit, but also how they fail to develop properly in CAS.
The SMA is involved in higher-level processing including memory retrieval, movement execution, transformation of information, and phonetic encoding in the feedforward loop2,41. The SMA can be divided into two parts that loop together: SMA proper, involved in motor control tasks and movement execution, and anterior pre-SMA, involved in other cognitive control domains as indicated by connections to the prefrontal cortex and angular gyrus in resting-state fMRI imaging42. Converging primate and transcranial stimulation (TMS) studies indicate pre-SMA to be involved in temporal prediction and have a larger role in the go/no go decision41,42. Neither part of the SMA can function properly in sequential processing and speech articulation without the other part, but damage to the pre-SMA loses the semantic programming and phonological planning that is fed to M1 by the SMA proper41,42.
Through the frontal aslant tract, the SMA connects to Broca’s area which focuses heavily on motor programming and articulation by activating the speech sound map2. An fMRI study to test motoric rehearsal and working memory in adults found significant consistent activation in left and right inferior frontal gyrus (IFG) (p < 0.001)34. Despite each participant being right-handed, other studies have also suggested a significant activation in Broca’s area in both hemispheres regardless of handedness, although handedness may influence lateral bias43. Further evidence from other studies shows that Broca’s area is involved in sentence comprehension, syntax and semantics, and word retrieval, all aspects involved in the motor planning of speech articulation insisting on an implication in CAS oral motor planning32,37,44. This study assumed articulatory rehearsal when participants may have used different tactics like chunking and visual imagery, and used single letter strings instead of larger phonemes commonly used in speech, limiting results significantly and implicating Broca’s area in CAS oral motor planning.
M1 is primarily responsible for initiating movement2. The secondary motor cortex (M2), containing Broca’s area, also sends axonal projections to M1 and shows high activation during motor learning45. Researchers developed a map known as the motor homunculus for M1 since it is somatotopically organized according to specific body parts where the lateral regions are involved in facial movement46. It receives direct input including the initial phonetic scale from the SMA and the sensory error prediction from the cerebellum as shown in the DIVA model making it a possible location for deficiencies in CAS2.
M1 is strongly influenced by the basal ganglia, specifically the striatum and pallidum which helps in producing predetermined actions46. The striatum is composed of the putamen, caudate nucleus, and accumbens nucleus, and the pallidum is mainly composed of globus pallidus, a structure that is closely connected with the SMA and IFG46,47. Recently, research regarding the basal ganglia’s involvement in speech has been growing, yet similar to the cerebellum, there is much that is still unknown. Converging neural computational research indicates the basal ganglia helps time the initiation of movement and switch between motor programs playing a role in reinforcement based learning, and disruption to the cortico-basal ganglia circuits can lead to hypokinesia and Parkinson’s disease2,48. Lesion studies confined solely to the basal ganglia, specifically the putamen, caudate nucleus and globus pallidus have shown to be implicated in orofacial apraxia and cause general apraxia49.
Computational studies on the DIVA model and neuroimaging suggest the thalamus to have a similar role in this circuit as the basal ganglia including movement initiation and coordination, also acting as the relay center23,50. The thalamus has been shown to act as a convergence hub from the cerebellum, brainstem, and basal ganglia to control the accuracy of ongoing movements in mice50. Researchers suggest that effective somatomotor integration is dependent on the thalamus, and the cerebellum deficits can influence the firing rate of thalamic neurons and therefore the ability to interpret sensory contexts50. Despite its involvement in the feedback and feedforward circuit being clear, the thalamus’s specific involvement in articulatory programming is still vague and future studies should consider focusing ROI’s on the thalamus.
Neuroanatomical Deficiencies in CAS
CAS imaging studies indicate gray and white matter deficits, reduced FA, underactivation, and abnormalities in cortical thickness and volume in circuits that connect these major subcortical structures.
A study comparing Autism Spectrum Disorder (ASD) to CAS neuroanatomy showed specific anomalies near the frontal lobe in CAS vs. typicals37. These included higher cortical volume in the right pars triangularis (d = 0.52), a region part of Broca’s area; left supramarginal gyrus (d = 0.50), located near the end of the sylvian fissure which separates Wernicke’s area and Broca’s area and is highly involved in semantic and phonological processing; left nucleus accumbens (d = 0.97), in the basal ganglia involved in motor selection and behavior reinforcement; and left paracentral (d = 0.80), a sensory area involved in motor control in adults37. While literature, as explained earlier, shows these structures to be implicated in sensory and motor-speech pathways for speech articulation in adults, this study suggests impaired development in these regions for CAS.
This study is the first to analyze volume differences in CAS using machine learning. Yet, it showed to have a more optimal predictive power for ASD than CAS, partly because of the small sample size. Also, this study calculated an average AUC of 0.62 using a linear-kernel SVM classification. Due to the small sample size, this AUC value is unapparent, and a larger sample might make specific neuroanatomical patterns and classifications more apparent and significant. The linear model also limits classification of more subtle structural differences37. Despite the limitations, this study indicates areas like Broca’s, somatosensory cortices in the parietal lobe, and the striatum to be involved in speech in children and adults, and abnormal development in these areas can lead to speech developmental disorders like CAS. Furthermore, there is a possibility of these deficits shown in the CAS cohort to also be implicated in non-oral, fine motor movement considering these somatosensory regions, striatum, and M1 to be dictating all motor movement46.
Other studies have also suggested a heavy role of the basal ganglia in speech production47. A case study on an 8 year old male with CAS and dysarthria showed volume reduction in the left and right caudate nucleus (d = -2.161, d = -2.352 respectively), left and right globus pallidus (d = -3.056, d = -1.8), and left and right putamen (-0.647, -0.950)47. The significant difference between left and right globus pallidus even after volume correction suggests the typical left-lateralization, as shown in Parkinson’s motor and lateralized connectivity studies, is affected by CAS51. On the other hand, the putamen and caudate nucleus, a neuromarker known to be implicated in the KE family, appeared to have n significance differences to the control after volume correction47. Yet, these structures have shown to have significant deficits in other converging CAS imaging studies40,52. fMRI scans also showed activation in the left IFG during nonword repetition, but the differences in these results between the child, A-II, and typicals was not significant after volume correction47.
The results from this study also suggest the hippocampus and thalamus to be involved in speech production. Liégeois and colleagues’ novel finding still requires more research. Considering this to be a case study and the sparse literature on hippocampus development in early childhood, future studies should focus on limiting ROIs to these two cortical structures and incorporating larger sample sizes of idiopathic CAS. Also, the control group included children from metropolitan Australia which may influence results to be biased towards tighter jaw and increased facial and throat movement. These children’s socioeconomic status and education may also play a role in a more refined language. A-II also underwent surgery for a submucous cleft, and his speech resonance greatly improved. Further research needs to address these aspects.
Other more recent studies have also indicated greater grey matter and less white matter in other subcortical and cortical structures involved in the feedforward network40,52,53. A neuroimaging study on 17, 10-year-old children with idiopathic CAS indicates reduced FA in multiple intra-hemispheric regions of Broca’s area and post-central gyrus, another somatosensory subcortical structure implicated in the pathogenesis of CAS53. Another neural network connecting speech motor planning areas the left precuneus and the front aslant tract including the SMA and orbital left IFG; visuo-spatial working memory and attention areas the left precuneus and left middle frontal gyrus as well as the right superior parietal and right superior occipital gyrus; and sensorimotor prediction and error prediction areas left cuneus, left superior occipital gyrus and right cerebellum53. The last neural network included the right angular gyrus, right superior temporal gyrus and right inferior occipital gyrus, an auditory-language integration network.
This study confirms the need for bilateral hemispheric deficiencies in CAS regardless of handedness. While CAS is generally considered left-lateralized, the last neural network discussed exhibits pure right lateralization deficits. And, despite all participants being right handed, deficits were seen in both left and right hemispheres of the cerebral cortex, similar to previous studies47. Yet, handedness might influence the degree of deficiencies between hemispheric structures. Impairments in the intra-hemispheric networks such as Broca’s area and left arcuate fasciculus were implicated in a low diadochokinesis rate (p = 0.01, R = 0.57), poor expressive grammar (p = 0.02, R = 0.53), and poor lexical production (p = 0.003, R = 0.67)53. These left-lateralized impairments such as the arcuate fasciculus, a connective network between all parts of the DIVA models including the phonological loop and the auditory feedback and somatosensory feedback system, have been reported in several imaging studies specific to CAS suggesting to be a specific biomarker between 9 and 15 year-olds regardless of comorbidity40,52,53. Interestingly, recent studies have also been suggesting the involvement of the precuneus, a structure associated with episodic memory retrieval, mental imagery, and self-referential processing, aligned with this study in speech production52,53. Specifically, idiopathic CAS neuroimaging indicates greater gray matter volume in the bilateral precuneus with correlating to low diadochokinesis rates, perhaps to compensate for other connective deficits52.
Converging studies suggest a larger role of the basal ganglia in the feedforward circuit in CAS accompanied by cortical abnormalities in regions like Broca’s area, auditory and somatosensory feedback networks, and working memory networks37,40,47,52,53. While limitations like comorbidity, smaller sample sizes, and environmental influences make it difficult to draw definitive conclusions, converging results strengthens the case for a distributed neural network underlying CAS. Furthermore, the identification of biomarkers such as reduced FA in the arcuate fasciculus or volumetric deficiencies in the bilateral precuneus and basal ganglia in CAS have significant clinical implications. Evidence from these studies can inform earlier diagnosis and intervention to focus on strengthening specific oral and fine motor networks. Applied machine learning on larger sample sizes can also uncover multivariate patterns that single ROI analyses cannot. Longitudinal studies would also help to explore the overall development of these structures in CAS, possibly providing earlier biomarkers for diagnosis or tracking traits that shift with treatment.
These neuroanatomical findings directly inform the second question of this review: how does imitation and repetitive practice influence speech production in children with CAS? The distributed deficits identified across the feedforward circuit, particularly in Broca’s area, the SMA, and the arcuate fasciculus, suggest that effective intervention must target motor planning and sensory feedback integration simultaneously. Current motor therapies for CAS have been developed with precisely these networks in mind.
Current Motor Therapies for CAS
There are many therapies available for speech sound disorders. Motor interventions are commonly applied to CAS cases5. These therapies including ReST, DTTC, and PROMPT provide insights into how imitation and repetition influence speech improvement.
Rapid Syllable Transition Treatment (ReST) focuses on transitions between syllables54. The approach is centered around the idea that syllables are the smallest “chunks” of sound in the phonetic inventory, and intervention helps transition between movements for each syllable gesture to facilitate novel vocabulary acquisition54. ReST is recommended for older children with mild speech impairment.
Studies show ReST to be reliable and have significant improvement in CAS54,55,56,57. Recently, studies show significant long-term gains in speech sound production from intensive ReST therapy (p= 0.007, η2 = 0.132)57. Although this study was split between face-to-face and telehealth sessions during the onset of COVID-19, limiting randomness and statistical power, there were no significant differences between the change in percent target sounds correct for untreated phrases (p = 0.317, η2 = .025), and outcomes suggested explicit KP feedback to contribute to gains57. This study is the first to exhibit a large sample size (48 participants) of 9 – 17 years old CAS patients without accompanying developmental disorders with diverse racial backgrounds. These results align with other randomized control trials using ReST treatment on younger age groups and provide evidence for intensive treatment to be more beneficial than distributed, allowing for stable gains post treatment55,57. This study should be interpreted with caution as there was no control for maturation. Although most ReST studies with younger samples with control for maturation show similar results to these, older children develop differently and gains may have been influenced by natural speech acquisition55.
Compared to ReST, Dynamic Temporal Tactile Cueing (DTTC) uses multimodal cueing methods including visual, tactile, auditory, and proprioceptive cues to shape articulation54. The clinician starts with simple gestural oral movements and slowly increases speech rate for imitation and decreases motor planning time over treatment period54. Due to the direct oral approach to improving motor planning, DTTC is generally recommended for younger children with more severe CAS54. Converging studies on DTTC intervention for CAS suggest cueing to be beneficial for overall improvement in younger and older children58,59. A 6-week DTTC intervention study on seven idiopathic CAS children below 6 years old showed significant increase with relatively small effect sizes in word accuracy for untreated (d = 0.26) and treated words (d = 0.31) into maintenance59. Imitation of visual cues alongside feedback helped to specifically target inaccurate habits such as improper phonetic placement59. Considering the wide age gap between 2 years to 5 years in speech development, maturation could have also played a role in improvement although the child effect size was calculated to be insignificant, and it was unknown whether additional treatment was received outside of the study. Interestingly, although improvement was significant, the older children had consistent resistant speech errors perhaps due to the short timeframe of the program and increasing complexity59. Regardless, DTTC shows some promise with speech improvement for CAS. Future studies on DTTC intervention for young idiopathic CAS patients should account for real-world limitations such as age as a factor in speech improvement.
Beyond idiopathic CAS, DTTC also seems to improve speech in comorbid CAS with intellectual disability58. A longitudinal case study on a slightly older CAS patient undergoing DTTC intervention resulted in a reduced speech rate from 94 to 71 syllables per minute and overall increase in comprehensibility from 0 to 58%58. Once again, through imitation of visual cues, vocal behaviors that attracted negative attention reduced and word accuracy greatly improved58. Similar to the previous shorter study, imitation in DTTC led to increase in overall speech word accuracy suggesting the importance of replicating tactile cues to be crucial to this intervention’s success.
Prompts for Restructuring Oral Muscular Phonetic Targets (PROMPT) is more specific to CAS and has been shown to improve sequential processing through the development of motor skills needed for interactive communication using tactile kinesthetic cueing54,60. Treatment follows a Motor Speech Treatment Hierarchy which starts at a general body tone and phonation before moving to articulation including aspects like vocal frequency, prosody, and intensity54. While both ReST and DTTC are limited to speech sound disorders, PROMPT is also recommended for those with mild to severe CAS with accompanying cognitive delay54.
Recent PROMPT studies provide deeper insight into the neuroplasticity of tactile cueing, imitation, and repetition for speech articulation60,61. A PROMPT intervention study on 14 children aged 3.9–6.6 years with idiopathic CAS used direct tactile-kinesthetic cuing to the oral regions near the mouth and face to inform correct positions and movement alongside visual and auditory cues60. Clinicians encouraged repetitive imitation through visual cues to work up to single word articulation60. ROI analysis and an independent sample t-test showed significant thinning of posterior superior temporal gyrus and canonical Wernicke’s area (t(8) = 2.42, p ≤ 0.05.), regions important for sensory feedback in phonological processing, and were associated with significant gains in speech although the direct relationship was unclear60. Although neural pruning is a development trait during older childhood and adolescence, results also showed an original increased thickness in the left supramarginal gyri, a region that is still unclear regarding its effects in CAS, and its sustained cortical structure60. More research regarding the left supramarginal gyri’s role in CAS from early childhood to late adolescence is required.
This rapid neuroplasticity could be a result of imitation from direct kinesthetic and visual cues. Other studies focusing on speech intelligibility with similar sample traits (3 to 6 years with CAS) have shown that decreasing feedback overtime with longer and more intensive sessions result in significant gains in speech motor control and speech intelligibility at the sentence level62. Converging studies on young CAS patients with comorbid CAS suggest PROMPT’s unique approach to tactile cueing provides robust gains in articulatory control and perceptual, sensory feedback integration61,62,63. Another cortical study on Italian children with a mean age of 6.3 years also showed significant improvement in speech intelligibility (p = 0.039) and motor control (p = 0.057) associated with FA increases with large effect sizes in the left dorsal corticobulbar tract, specifically the motor map for the face and tongue (p = 0.043, d = 0.70-1.24)61. Despite the small sample size, this study indicates the possibility for PROMPT therapy to directly induce left lateralized cortical tract changes in speech61. More research on the motor tracts implicated in the DIVA model such as Broca’s area, the SMA, and front aslant tract, is required to make connections between speech improvement and neuroplasticity. Future research should also control for maturation in a larger sample size ranging from early childhood to adolescence in English speakers.
While DTTC, ReST, and PROMPT each demonstrate that imitation and repetitive cueing can drive neuroplastic gains in speech articulation, they remain confined to oral motor practice. This raises the third question: whether rhythmic motor movement beyond the oral system can influence speech production. Evidence from tapping studies suggests that the sequential processing deficits in CAS extend well beyond the mouth, pointing toward a shared motor timing circuit that non-oral rhythmic intervention may be able to target.
Rhythm in Speech
Children with speech sequential processing deficits are unable to accurately time oral motor movements6. These sequential processing deficits can also manifest as rhythmic motor error extending beyond oral and facial muscles to broader motor systems3,64. Tapping studies provide insights into how fine and gross motor movements are impacted by sequential processing deficits.
Research comparing diadochokinetic scores with repetitive and alternate tapping indicates similarities in prosody and rhythm where syllable and tapping durations are both coupled and controlled by a “central rate delimiter” like the cerebellum in the feedforward circuit65. Specifically, converging studies show that as syllables and keyboard tapping change from repetitive to alternating, sequential processing loads increase and performance decreases3,65,66. A tapping study on 21 7-11 year old children with speech language impairments indicated significantly different speeds and rhythms than the controls for the paced metronome task as the tempo increased from 1.5Hz (p < 0.001, η2 = .21) to 2Hz (p < 0.01, η2 = .16)67. Post-hoc Bonneferri tests also showed SLI scores to be significantly more inconsistent compared to controls67. Although the results for the paced task were significant, the effect sizes were relatively small as expected due to the simplicity of the task. Other studies showed similar results with insignificant statistical values specific to CAS children performing repetitive keyboard tapping tasks (p = 0.4126, d = 0.10)3.
Beyond repetitive tasks, children with CAS perform considerably worse on alternating tapping. A specific study indicated a significant reduction in pacing from repetitive keyboard tapping (p = 0.0545), a statistically insignificant rate, to alternative keyboard tapping (p = 0.0088)65. Similarly, the monosyllabic diadochokinetic task (p = 0.0545) had higher accuracy than the disyllabic task (p = 0.0004)65. Researchers explain this to be a correlation between tapping and speech articulation due to increased sequential processing load in the store and motor aspect of the circuit, leading to errors, delay, and overcorrection3. Yet, slower repetitive tapping rates in CAS also indicate an extreme impairment in the feedforward circuit, even when sequential processing loads are low3,67.
Emerging research suggests the possibility for motor rhythms to help develop auditory rhythmic sensitivity as they both are implicated in the same motor programming circuit66,67. To further investigate this hypothesis, future studies should focus on solely longitudinal motor rhythmic intervention through imitation and cueing to test improvement in auditory feedback mechanisms. Together, these findings offer the potential to target the shared feedforward circuit underlying both speech and non-speech motor timing in CAS through non-oral methods.
The overlap between speech timing and non-oral motor rhythm implies a shared neural mechanism coordinating both. One candidate for this mechanism is the mirror neuron system, which has been implicated in auditory-motor coupling, imitation, and sensorimotor learning, all processes central to both rhythmic intervention and speech acquisition in CAS.
Mirror Neurons in Speech-Motor Networks
The concept of the mirror neuron system provides some insight into why a motor rhythmic intervention program for CAS may work. Rizzolatti and colleagues first discovered the presence of mirror neurons in macaque monkeys when certain F5 neurons in the motor cortex fired when observing and performing specific sensorimotor actions such as grasping and placing68. Since then, research suggests an involvement of mirror neurons in the human cerebral cortex, specifically Broca’s area, for sensorimotor learning and social interaction68,69,70. Through visual cues, repetition, and imitation, Hebian-like strengthening occurs between motor and sensory neurons when watching our own actions, observing others, and even receiving accompanying auditory input69. After mirror neurons strengthen, sensory cues can trigger motor activation even without direct prior experience of the action, particularly when similar movements exist in the motor repertoire, and they also develop in relation to individual experiences such as dancing or playing the piano69.
The relevance of this system to CAS is indirect but meaningful. First, the primary locus of this system is Broca’s area which studies have shown to be consistently implicated in CAS showing reduced FA, volumetric abnormalities, and underactivation across multiple studies37,40,53. Second, the auditory-motor coupling in the mirror neuron system directly parallels the feedforward and feedback deficits in CAS. If this system is compromised, the ability to map auditory information into the speech sound map and motor programs would be impaired, contributing to the inconsistent speech patterns and poor coarticulatory transitions that define CAS. Third, studies investigating the correlation between oral imitation and fine motor movement in 2-5 year old children with severe speech impairments suggest the possibility for oral impairment to co-occur with broader gross and fine motor imitation abilities consistent with mirror neuron dysfunction71,72.
Together, these converging lines of evidence support a testable hypothesis: that rhythmic interventions using visual and auditory cues, such as tapping to a beat or musical rhythm, may engage mirror neuron circuits in Broca’s area and through auditory-motor coupling, strengthen motor representations that inform the feedforward circuit for speech. The tapping literature already demonstrates shared sequential processing demands between rhythmic motor tasks and speech articulation in CAS3,65,67. What remains to be directly tested is whether activating this shared circuit through non-oral rhythmic practice produces measurable upstream effects on speech motor programming. Future studies should examine mirror neuron activation in pediatric CAS populations during rhythmic tasks, controlling for maturation, comorbidity, handedness, and severity, to determine whether this mechanism can be directly exploited in intervention for CAS with varying severity.
If mirror neurons provide the neural bridge between rhythmic motor movement and speech, music therapy presents one of the most naturalistic contexts in which this bridge could be activated. Rhythm, melody, and imitation converge in musical interaction in ways that may simultaneously engage the auditory-motor coupling and feedforward circuits implicated in CAS.
Music Therapy
Given the mirror neuron system’s capacity to strengthen auditory-motor coupling through observation and imitation, music therapy presents itself as a naturalistic medium through which rhythmic motor representations in CAS may be targeted. To date, there is little evidence this type of intervention is successful relative to the traditional interventions like ReST, DTTC, and PROMPT. The small evidence available for this theory spans in multiple levels from neuroimaging in adult musicians to preliminary pediatric case studies and should be interpreted with that hierarchy in mind.At the neuroimaging level, studies comparing singing and instrumental performance reveal overlapping neural activity across regions implicated in both speech and fine motor control73. Regions included the cerebellum VI, CrusI-II, VIIIa, and VIIIb, SMA, caudate nucleus, middle frontal gyrus, and larynx area of the motor cortex when playing the cello versus singing in adults73. The anterior insula (aINS) and anterior cingulate cortex (ACC) regions coordinating movement and implicated in CAS were also observed to exhibit high activity during both tasks2,47,52,73. The association between speech prosody and music perception suggests the auditory-motor coupling in the mirror neuron system to be linking rhythm from music to speech. Rhythm training can enhance the perception of speech signals, syllable production, and phonological awareness. Through music intervention, children with speech language disorders may be able to strengthen motor movement and rhythmic control, indirectly helping to strengthen speech articulation. This study is only generalizable to adults with about 13.9 years of experience at playing the cello73. Children exhibit different neural structures that are undergoing maturation, and the comorbidity and severity of the speech disorder may result in slower rhythmic control development. The listen/play paradigms in this study only utilized single notes whereas in reality, speech tones and general music are much more complex. Yet, Hausen and colleagues indicated that beginning training before 7 years old may contribute to co-opt neural networks developing, especially in the larynx area of the motor cortex73. This novel finding provides evidence for the mirror neuron connection between fine motor movement and speech articulation, and the possibility for strengthening the auditory-vocal network through early music exposure and interaction.
At the younger case study level, pediatric case studies suggest that early musical exposure may similarly engage overlapping speech and motor networks in children with CAS. A 9-month long music therapy case study on a 3-year-old girl with severe CAS showed music treatment to improve verbal communication, socialization, cognitive, emotional, and motor skills/movement74. At the 5th session, although nonverbal, she attempted to verbalize sounds to visual instrument cues and displayed oral muscle movement including smiles as short single notes on the piano were used to facilitate interaction74. At the 10th session, she was imitating oral movement for vowel sounds and producing consonant sounds for the starting letter of various instruments74. By the last session, the girl displayed high vocalization of simple words and increased cognitive understanding of letters despite taking long breaks between treatment sessions74. Despite this study not indicating fidelity or reliability measures, the intervention was performed at a university clinic by a licensed clinician. The child’s parents had also reported the child’s liking for music and high energy levels which may have influenced her early interactions with the clinician and exploration. Other recent case studies using Speech–Music Therapy for Aphasia (SMTA) indicated similar results with increased vocalization and speech intelligibility74.
In contrast, at the structured intervention level, case studies on Melodic Intonation Therapy (MIT) for CAS have shown more mixed results. One case study compared 2 more emotionally sensitive children with CAS (aged 5 and 6 years respectively) regarding speech gains after 6 weeks of 40 minute at-home sessions per week. A Goldman Fristoe Test of Articulation and Khan-Lewis Phonological Analysis test before and after treatment showed a slight increase in STP scores and accuracy, specifically 7% for Participant 1 and 15% for Participant 275. Overall both participants had more accurate word productions in phonological processes after intervention in aspects including deletion of final consonants, initial voicing, final devoicing, syllable reduction, stopping of fricatives and affricatives, velar fronting and palatal fronting75. Although statistical analysis showed no significant improvement, pre and post intervention scores indicate a possibility for MIT therapy to improve outcomes for CAS. Future studies on MIT intervention for CAS should consider an intensive and longitudinal treatment with a larger sample size. They should also consider the type of instrument, music, age, severity, handedness, sex, and personality traits for each CAS participant. MRI data throughout treatment may also provide insights into the neuroplasticity in CAS from music and rhythmic intervention.
Discussion
This review addressed 3 questions: (1) What are the anatomical and functional connections between motor and speech in children with CAS compared to children with typical development? (2) How does imitation and repetitive practice influence speech production? (3) How does rhythmic motor movement intervention influence speech production?
Anatomical and Functional Connections in CAS
Converging evidence from neuroimaging studies confirms that CAS is underpinned by deficiencies throughout the feedforward circuit in motor speech programming. Abnormalities are consistently identified in the cerebellum, Broca’s area, the articulate fasciculus, SMA, M1, basal ganglia, and thalamus. Cerebellar deficits, although less directly evidenced in pediatric CAS populations, are implicated in FOXP2 mutations of Purkinje cells in Lobule VII and VIII as well as Crus I. Lesion studies specifically point to the DCN impairment as the source of feedforward dysregulation. Beyond the cerebellum, FA reduction in the articulate fasciculus that connects Broca’s area and Wernicke’s area consistently emerges as a reliable structural biomarker for CAS between 9 and 15 years of age. Volumetric reductions in the basal ganglia and thalamus, specifically the caudate nucleus, putamen, striatum, and global pallidus, alongside somatosensory and auditory feedback cortices support the DIVA model characterization as a motor planning and programming deficit with overreliance on the feedback system.
Imitation and Repetition
The evidence reviewed regarding motor interventions provides evidence that imitation and repetition positively influences speech acquisition. Across ReST, DTTC, and PROMPT, the use of visual, auditory, proprioceptive, and tactile cues consistently produces gains in word accuracy, speech intelligibility, and articulation control that continue beyond post-treatment. Intensive motor intervention also proves to influence aspects beyond behavioral. PROMPT neuroimaging studies show significant thinning of Wernicke’s area into maintenance, consistent with neural pruning accompanying consolidation of more efficient sensory feedback processing. FA reductions in the corticobulbar tract following PROMPT further suggest that repetitive imitation of sensory cues directly induces structural neural changes in the motor tracts governing face and tongue muscle movement. ReST’s RCT evidence additionally demonstrates intensive treatment produces more durable long-term gains than distributed treatment, indicating a dose-response relationship between motor planning and programming and neuroplasticity. Together, these findings indicate that imitation and repetition do not just reinforce correct surface articulation, they appear to reorganize the underlying motor-speech network in ways that generalize beyond treated targets.
Rhythmic Motor Movement Intervention
The evidence presented in this study yields a cautious but promising answer to the third question. Tapping studies confirm that sequential processing deficits are not tied to oral behavior, rather are implicated in fine motor movements as well. CAS children perform significantly worse on complex motor movements such as alternative tapping compared to typicals. This parallelism between speech timing errors and non-oral motor timing errors implies a shared central rate delimiter, likely the cerebellum, coordinating both domains through the same feedforward circuit. The implication is meaningful: if speech and non-oral rhythmic movement draw on overlapping neural resources, then targeting that shared circuit through rhythmic intervention may produce upstream effects on speech motor programming.
The mirror neuron theory might offer a plausible mechanism for this theory. Broca’s area, the main hub of mirror neurons, is consistently implicated in CAS. If this region were impaired, the ability to map observed rhythmic patterns to motor programs would be compromised, consistent with the inconsistent error patterns and poor coarticulatory transitions defining CAS. Conversely, rhythmic interventions using visual and auditory cues may engage these same circuits, and through auditory-motor coupling strengthen motor representations that feed into the feedforward system for speech. This hypothesis is supported indirectly by the tapping literature, by mirror neuron imitation studies in children with severe speech impairments, and by the established role of Hebbian strengthening in sensorimotor learning.
Music therapy offers the most developed application of this principle to date, though the evidence base remains stratified and should be interpreted accordingly. At the neuroimaging level, overlapping activation across speech and fine motor networks, including the cerebellum, SMA, caudate nucleus, and laryngeal region of the motor cortex, provides mechanistic support for music as a vehicle for engaging CAS-relevant circuits. The identification of a critical period before age 7 sensitive to musical training forming co-opt neural networks in oral regions such as the larynx in the motor cortex is a particularly significant finding for early intervention. At the case study level, preliminary pediatric evidence suggests that early musical engagement can facilitate vocalization, imitation, and phonological awareness in children with severe CAS, though the absence of fidelity measures and controlled designs limits the conclusions that can be drawn. At the structured intervention level, MIT results remain statistically mixed, most likely reflecting the early state of the research rather than a true ceiling on the approach’s potential.
Overall, rhythmic motor movement intervention for CAS should currently be understood as a theoretically well-grounded but empirically immature approach. The convergence of tapping research, mirror neuron theory, and preliminary music therapy findings is sufficient to justify dedicated controlled investigation, but not yet sufficient to recommend rhythmic intervention as a standalone clinical tool alongside established approaches like DTTC and PROMPT.
Limitations
Significant constrictions limit the conclusions across this review. Many of them lie in aspects of CAS that make it difficult to perform experimental studies and invite speculation. The low prevalence of CAS, about 0.1% of children, makes recruitment of sample sizes larger than 30 extremely difficult, limiting statistical power and generalizability. CAS is also highly comorbid co-occurring with ASD, dysarthria, intellectual disability, and other developmental disorders, and most studies are unable to control for this factor. The result is that findings from comorbid populations are routinely, if cautiously, extended to idiopathic CAS without sufficient empirical basis.
Age differences across small samples also presents a significant challenge. Speech-motor development both behaviorally and anatomically differ between 3 year olds and 9 year olds into adolescence. Across development, maturation is rarely controlled and could account for treatment improvement in longitudinal studies, especially in younger studies. Severity variation is similarly inconsistent across recruitment criteria, making it difficult to determine whether findings generalize across the mild-to-severe spectrum of the disorder. The assumption of left lateralization in CAS is also increasingly challenged by bilateral deficit patterns observed across multiple neuroimaging studies, yet many studies continue to focus ROIs predominantly on left hemisphere structures, potentially missing relevant right hemisphere abnormalities. Finally, the majority of neuroimaging studies reviewed are cross-sectional, providing snapshots of neural architecture rather than insight into how these structures develop or respond to intervention over time.
Future Directions
Several research priorities emerge clearly from this review. Longitudinal neuroimaging studies tracking the development of key structures, particularly the arcuate fasciculus, bilateral precuneus, thalamus, and cerebellar Crus I and Lobules VI–VIII, from early childhood through adolescence would provide critical insight into the developmental trajectory of CAS and enable identification of earlier structural biomarkers for diagnosis. The thalamus and precuneus in particular remain understudied relative to their apparent involvement in the feedforward circuit and call for dedicated ROI analysis in future work.
Machine learning applied to larger neuroimaging datasets presents a significant opportunity to uncover multivariate structural patterns that single ROI analyses cannot detect. The preliminary application of linear SVM classification to CAS neuroanatomy has shown limited discriminative power partly due to sample size constraints. Larger datasets and non-linear classification approaches may reveal more diagnostically meaningful patterns. Dedicated empirical studies on mirror neuron activation in pediatric CAS populations during rhythmic tasks are also needed to move the rhythmic intervention hypothesis from theoretical framework to testable clinical model. Such studies should systematically control for maturation, comorbidity, handedness, severity, and sex-based differences. Finally, controlled rhythmic and music-based intervention trials with concurrent neuroimaging and larger, more carefully characterized samples would establish whether the auditory-motor coupling mechanisms identified in adult musicians and preliminary pediatric case studies can be reliably recruited for speech gains in CAS.
References
- Morgan, A. T., Amor, D. J., St John, M. D., Scheffer, I. E., & Hildebrand, M. S. (2024). Genetic architecture of childhood speech disorder: a review. Molecular Psychiatry, 29(5), 1281–1292. https://doi.org/10.1038/s41380-024-02409-8. [↩] [↩] [↩]
- Guenther, F. H., Ghosh, S. S., & Tourville, J. A. (2005). Neural modeling and imaging of the cortical interactions underlying syllable production. Brain and Language, 96(3), 280–301. https://doi.org/10.1016/j.bandl.2005.06.001 [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- Peter, B., Button, L., Stoel-Gammon, C., Chapman, K., & Raskind, W. H. (2013). Deficits in sequential processing manifest in motor and linguistic tasks in a multigenerational family with childhood apraxia of speech. Clinical Linguistics & Phonetics, 27(3), 163–191. https://doi.org/10.3109/02699206.2012.736011 [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- Shriberg, L. D., Kwiatkowski, J., & Mabie, H. L. (2019). Estimates of the prevalence of motor speech disorders in children with idiopathic speech delay. Clinical Linguistics & Phonetics, 33(8), 679–706. https://doi.org/10.1080/02699206.2019.1595731. [↩]
- Morgan, A. T., Murray, E., & Liégeois, F. J. (2018). Interventions for childhood apraxia of speech. Cochrane Database of Systematic Reviews, 2019(8), CD006278. https://doi.org/10.1002/14651858.cd006278.pub3 [↩] [↩] [↩]
- Terband, H., Maassen, B., Guenther, F., & Brumberg, J. (Eds.). (2010). Computational Neural Modeling of Speech Motor Control in Childhood Apraxia of Speech (CAS): Vol. Volume 52. Journal of Speech, Language, and Hearing Research. https://doi.org/10.1044/1092-4388(2009/07-0283 [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- Hoogman, M., Guadalupe, T., Zwiers, M. P., Klarenbeek, P., Francks, C., & Fisher, S. E. (2014). Assessing the effects of common variation in the FOXP2 gene on human brain structure. Frontiers in Human Neuroscience, 8. https://doi.org/10.3389/fnhum.2014.00473. [↩]
- Vernes, S. C., Spiteri, E., Nicod, J., Groszer, M., Taylor, J. M., Davies, K. E., Geschwind, D. H., & Fisher, S. E. (2007). High-Throughput analysis of promoter occupancy reveals direct neural targets of FOXP2, a gene mutated in speech and language disorders. The American Journal of Human Genetics, 81(6), 1232–1250. https://doi.org/10.1086/522238 [↩]
- Kaspi, A., Hildebrand, M. S., Jackson, V. E., Braden, R., Van Reyk, O., Howell, T., Debono, S., Lauretta, M., Morison, L., Coleman, M. J., Webster, R., Coman, D., Goel, H., Wallis, M., Dabscheck, G., Downie, L., Baker, E. K., Parry-Fielder, B., Ballard, K., . . . Morgan, A. T. (2022). Genetic aetiologies for childhood speech disorder: novel pathways co-expressed during brain development. Molecular Psychiatry. https://doi.org/10.1038/s41380-022-01764-8. [↩]
- Levelt, W. J. M., Roelofs, A., & Meyer, A. S. (1999). A theory of lexical access in speech production. Behavioral and Brain Sciences, 22(1), 1–38. doi:10.1017/S0140525X99001776 [↩] [↩] [↩]
- Levelt, W. J. M. (n.d.). Producing spoken language: a blueprint of the speaker. https://www.mpi.nl/world/materials/publications/levelt/Levelt_Producing_spoken_language_1999.pdf [↩] [↩] [↩]
- Strijkers, K., Costa, A., & Pulvermüller, F. (2017). The cortical dynamics of speaking: Lexical and phonological knowledge simultaneously recruit the frontal and temporal cortex within 200 ms. NeuroImage, 163, 206–219. https://doi.org/10.1016/j.neuroimage.2017.09.041 [↩] [↩]
- Terband, H., Namasivayam, A., Maas, E., Van Brenk, F., Mailend, M., Diepeveen, S., Van Lieshout, P., & Maassen, B. (2019). Assessment of Childhood Apraxia of Speech: A Review/Tutorial of Objective Measurement Techniques. Journal of Speech Language and Hearing Research, 62(8S), 2999–3032. https://doi.org/10.1044/2019_jslhr-s-csmc7-19-0214 [↩]
- Button, L., Peter, B., Stoel-Gammon, C., & Raskind, W. H. (2013). Associations among measures of sequential processing in motor and linguistics tasks in adults with and without a family history of childhood apraxia of speech: A replication study. Clinical Linguistics & Phonetics, 27(3), 192–212. https://doi.org/10.3109/02699206.2012.744097 [↩]
- Maas, E., Mailend, M., & Guenther, F. H. (2015). Feedforward and feedback control in Apraxia of speech: Effects of noise masking on vowel production. Journal of Speech Language and Hearing Research, 58(2), 185–200. https://doi.org/10.1044/2014_jslhr-s-13-0300 [↩] [↩]
- Iuzzini-Seigel, J., Hogan, T. P., Guarino, A. J., & Green, J. R. (2015). Reliance on auditory feedback in children with childhood apraxia of speech. Journal of Communication Disorders, 54, 32–42. https://doi.org/10.1016/j.jcomdis.2015.01.002 [↩] [↩] [↩]
- Peter, B. (2017). The role of short-term memory impairment in nonword repetition, real word repetition, and nonword decoding: A case study. Clinical Linguistics & Phonetics, 32(4), 347–352. https://doi.org/10.1080/02699206.2017.1375561 [↩]
- Schulze, K., Vargha-Khadem, F., & Mishkin, M. (2017). Phonological working memory and FOXP2. Neuropsychologia, 108, 147–152. https://doi.org/10.1016/j.neuropsychologia.2017.11.027 [↩]
- Preston, J. L., Molfese, P. J., Gumkowski, N., Sorcinelli, A., Harwood, V., Irwin, J. R., & Landi, N. (2014). Neurophysiology of speech Differences in Childhood Apraxia of speech. Developmental Neuropsychology, 39(5), 385–403. https://doi.org/10.1080/87565641.2014.939181 [↩]
- Shriberg, L. D., Lohmeier, H. L., Strand, E. A., & Jakielski, K. J. (2012). Encoding, memory, and transcoding deficits in Childhood Apraxia of Speech. Clinical Linguistics & Phonetics, 26(5), 445–482. https://doi.org/10.3109/02699206.2012.655841 [↩] [↩]
- Schmahmann, J. D. (1998). Dysmetria of thought: clinical consequences of cerebellar dysfunction on cognition and affect. Trends in Cognitive Sciences, 2(9), 362–371. https://doi.org/10.1016/s1364-6613(98)01218-2 [↩]
- Wolpert, D., Miall, R. C., & Kawato, M. (1998). Internal models in the cerebellum. Trends in Cognitive Sciences, Vol. 2(No. 9). https://wolpertlab.neuroscience.columbia.edu/sites/wolpertlab.neuroscience.columbia.edu/files/content/papers/WolMiaKaw98.pdf [↩]
- Welniarz, Q., Worbe, Y., & Gallea, C. (2021). The Forward Model: a unifying theory for the role of the cerebellum in motor control and sense of agency. Frontiers in Systems Neuroscience, 15, 644059. https://doi.org/10.3389/fnsys.2021.644059 [↩] [↩] [↩] [↩] [↩] [↩]
- Konczak, J., Schoch, B., Dimitrova, A., Gizewski, E., & Timmann, D. (2005). Functional recovery of children and adolescents after cerebellar tumour resection. Brain, 128(6), 1428–1441. https://doi.org/10.1093/brain/awh385 [↩] [↩]
- Cabaraux, P., Gandini, J., Kakei, S., Manto, M., Mitoma, H., & Tanaka, H. (2020). Dysmetria and Errors in Predictions: The role of the Internal Forward model. International Journal of Molecular Sciences, 21(18), 6900. https://doi.org/10.3390/ijms21186900 [↩]
- Miall, R., & Wolpert, D. (1996). Forward models for physiological motor control. Neural Networks, 9(8), 1265–1279. https://doi.org/10.1016/s0893-6080(96)00035-4 [↩]
- Ballard, K. J., Tourville, J. A., & Robin, D. A. (2014). Behavioral, computational, and neuroimaging studies of acquired apraxia of speech. Frontiers in Human Neuroscience, 8, 892. https://doi.org/10.3389/fnhum.2014.00892 [↩]
- Malone, L. A., Hill, N. M., Tripp, H., Zipunnikov, V., Wolpert, D. M., & Bastian, A. J. (2025). The control of movement gradually transitions from feedback control to feedforward adaptation throughout childhood. Npj Science of Learning, 10(1), 13. https://doi.org/10.1038/s41539-025-00304-7 [↩] [↩]
- Clements-Stephens, A. M., Rimrodt, S. L., & Cutting, L. E. (2008). Developmental sex differences in basic visuospatial processing: Differences in strategy use? Neuroscience Letters, 449(3), 155–160. https://doi.org/10.1016/j.neulet.2008.10.094 [↩]
- Lyu, W., Thung, K., Huynh, K. M., Wang, L., Lin, W., Ahmad, S., & Yap, P. (2025). Functional development of the human cerebellum from birth to age five. Nature Communications, 16(1), 6350. https://doi.org/10.1038/s41467-025-61465-y [↩] [↩] [↩]
- Guell, X., Schmahmann, J. D., De Gabrieli, J., & Ghosh, S. S. (2018). Functional gradients of the cerebellum. eLife, 7. https://doi.org/10.7554/elife.36652 [↩]
- Mariën, P., Ackermann, H., Adamaszek, M., Barwood, C. H. S., Beaton, A., Desmond, J., De Witte, E., Fawcett, A. J., Hertrich, I., Küper, M., Leggio, M., Marvel, C., Molinari, M., Murdoch, B. E., Nicolson, R. I., Schmahmann, J. D., Stoodley, C. J., Thürling, M., Timmann, D., . . . Ziegler, W. (2013). Consensus Paper: Language and the Cerebellum: an Ongoing Enigma. The Cerebellum, 13(3), 386–410. https://doi.org/10.1007/s12311-013-0540-5 [↩] [↩]
- Metoki, A., Wang, Y., & Olson, I. R. (2021). The Social Cerebellum: A Large-Scale Investigation of Functional and Structural Specificity and connectivity. Cerebral Cortex, 32(5), 987–1003. https://doi.org/10.1093/cercor/bhab260 [↩]
- Chen, S. A., & Desmond, J. E. (2004). Cerebrocerebellar networks during articulatory rehearsal and verbal working memory tasks. NeuroImage, 24(2), 332–338. https://doi.org/10.1016/j.neuroimage.2004.08.032 [↩] [↩]
- Marvel, C. L., & Desmond, J. E. (2010). Functional topography of the cerebellum in verbal working memory. Neuropsychology Review, 20(3), 271–279. https://doi.org/10.1007/s11065-010-9137-7 [↩] [↩]
- Argyropoulos, G. P. D., Watkins, K. E., Belton-Pagnamenta, E., Liégeois, F., Saleem, K. S., Mishkin, M., & Vargha-Khadem, F. (2018). Neocerebellar Crus I Abnormalities Associated with a Speech and Language Disorder Due to a Mutation in FOXP2. The Cerebellum, 18(3), 309–319. https://doi.org/10.1007/s12311-018-0989-3 [↩] [↩]
- Conti, E., Retico, A., Palumbo, L., Spera, G., Bosco, P., Biagi, L., Fiori, S., Tosetti, M., Cipriani, P., Cioni, G., Muratori, F., Chilosi, A., & Calderoni, S. (2020). Autism Spectrum Disorder and Childhood Apraxia of Speech: Early Language-Related Hallmarks across Structural MRI Study. Journal of Personalized Medicine, 10(4), 275. https://doi.org/10.3390/jpm10040275 [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- Hickok, G., & Poeppel, D. (2004). Dorsal and ventral streams: a framework for understanding aspects of the functional anatomy of language. Cognition, 92(1–2), 67–99. https://doi.org/10.1016/j.cognition.2003.10.011 [↩] [↩]
- Dacre, J., Colligan, M., Clarke, T., Ammer, J. J., Schiemann, J., Chamosa-Pino, V., Claudi, F., Harston, J. A., Eleftheriou, C., Pakan, J. M., Huang, C., Hantman, A. W., Rochefort, N. L., & Duguid, I. (2021). A cerebellar-thalamocortical pathway drives behavioral context-dependent movement initiation. Neuron, 109(14), 2326-2338.e8. https://doi.org/10.1016/j.neuron.2021.05.016 [↩]
- Liégeois, F. J., Turner, S. J., Mayes, A., Bonthrone, A. F., Boys, A., Smith, L., Parry-Fielder, B., Mandelstam, S., Spencer-Smith, M., Bahlo, M., Scerri, T. S., Hildebrand, M. S., Scheffer, I. E., Connelly, A., & Morgan, A. T. (2019). Dorsal language stream anomalies in an inherited speech disorder. Brain, 142(4), 966–977. https://doi.org/10.1093/brain/awz018 [↩] [↩] [↩] [↩] [↩] [↩]
- Tanji, J. (1994). The supplementary motor area in the cerebral cortex. Neuroscience Research, 19(3), 251–268. https://doi.org/10.1016/0168-0102(94)90038-8 [↩] [↩] [↩]
- Hertrich, I., Dietrich, S., & Ackermann, H. (2016). The role of the supplementary motor area for speech and language processing. Neuroscience & Biobehavioral Reviews, 68, 602–610. https://doi.org/10.1016/j.neubiorev.2016.06.030 [↩] [↩] [↩]
- Tzourio-Mazoyer, N. (2016). Intra- and inter-hemispheric connectivity supporting hemispheric specialization. In Research and perspectives in neurosciences (pp. 129–146). https://doi.org/10.1007/978-3-319-27777-6_9 [↩]
- Liégeois, F., Morgan, A., Connelly, A., & Vargha-Khadem, F. (2011). Endophenotypes of FOXP2: Dysfunction within the human articulatory network. European Journal of Paediatric Neurology, 15(4), 283–288. https://doi.org/10.1016/j.ejpn.2011.04.006 [↩]
- Lee, C., Kim, Y., & Kaang, B. (2022). The primary motor cortex: the hub of motor learning in rodents. Neuroscience, 485, 163–170. https://doi.org/10.1016/j.neuroscience.2022.01.009 [↩]
- Watson, C., Kirkcaldie, M., & Paxinos, G. (2010). Command and control—the motor systems. In Elsevier eBooks (pp. 55–74). https://doi.org/10.1016/b978-0-12-373889-9.50005-x [↩] [↩] [↩] [↩]
- Liégeois, F. J., Hildebrand, M. S., Bonthrone, A., Turner, S. J., Scheffer, I. E., Bahlo, M., Connelly, A., & Morgan, A. T. (2016). Early neuroimaging markers of FOXP2 intragenic deletion. Scientific Reports, 6(1), 35192. https://doi.org/10.1038/srep35192 [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- Rubchinsky, L. L., Kopell, N., & Sigvardt, K. A. (2003). Modeling facilitation and inhibition of competing motor programs in basal ganglia subthalamic nucleus–pallidal circuits. Proceedings of the National Academy of Sciences, 100(24), 14427–14432. https://doi.org/10.1073/pnas.2036283100 [↩]
- Pramstaller, P. P., & Marsden, C. D. (1996). The basal ganglia and apraxia. Brain, 119(1), 319–340. https://doi.org/10.1093/brain/119.1.319 [↩]
- Heck, D. H., Fox, M. B., Chapman, B. C., McAfee, S. S., & Liu, Y. (2023). Cerebellar control of thalamocortical circuits for cognitive function: A review of pathways and a proposed mechanism. Frontiers in Systems Neuroscience, 17, 1126508. https://doi.org/10.3389/fnsys.2023.1126508 [↩] [↩] [↩]
- Miranda-Domínguez, Ó., Ragothaman, A., Hermosillo, R., Feczko, E., Morris, R., Carlson-Kuhta, P., Nutt, J. G., Mancini, M., Fair, D., & Horak, F. B. (2020). Lateralized Connectivity between Globus Pallidus and Motor Cortex is Associated with Freezing of Gait in Parkinson’s Disease. Neuroscience, 443, 44–58. https://doi.org/10.1016/j.neuroscience.2020.06.036 [↩]
- Bosco, P., Podda, I., Cipriano, E., Bombonato, C., Cipriani, P., Bartoli, M., Pasquariello, R., Fiori, S., Biagi, L., & Chilosi, A. (2025). Alterations of the neural substrate in childhood apraxia of speech: new evidence from neuroimaging. Brain Communications, 7(5), fcaf302. https://doi.org/10.1093/braincomms/fcaf302 [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- Fiori, S., Guzzetta, A., Mitra, J., Pannek, K., Pasquariello, R., Cipriani, P., Tosetti, M., Cioni, G., Rose, S. E., & Chilosi, A. (2016). Neuroanatomical correlates of childhood apraxia of speech: A connectomic approach. NeuroImage Clinical, 12, 894–901. https://doi.org/10.1016/j.nicl.2016.11.003 [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- Maas, E., Gildersleeve-Neumann, C. E., Jakielski, K. J., & Stoeckel, R. (2014). Motor-Based Intervention Protocols in Treatment of Childhood Apraxia of Speech (CAS). Current Developmental Disorders Reports, 1(3), 197–206. https://doi.org/10.1007/s40474-014-0016-4 [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- Thomas, D. C., McCabe, P., & Ballard, K. J. (2014). Rapid Syllable Transitions (ReST) treatment for Childhood Apraxia of Speech: The effect of lower dose-Frequency. Journal of Communication Disorders, 51, 29–42. https://doi.org/10.1016/j.jcomdis.2014.06.004 [↩] [↩] [↩]
- Thomas, D. C., McCabe, P., Ballard, K. J., & Lincoln, M. (2016). Telehealth delivery of Rapid Syllable Transitions (ReST) treatment for childhood apraxia of speech. International Journal of Language & Communication Disorders, 51(6), 654–671. https://doi.org/10.1111/1460-6984.12238 [↩]
- Preston, J. L., Caballero, N. F., Leece, M. C., Wang, D., Herbst, B. M., & Benway, N. R. (2023). A randomized controlled trial of treatment distribution and biofeedback effects on speech production in School-Age Children with Apraxia of Speech. Journal of Speech Language and Hearing Research, 67(9S), 3414–3436. https://doi.org/10.1044/2023_jslhr-22-00622 [↩] [↩] [↩] [↩]
- Baas, B et al (2008). Treatment of Severe Childhood Apraxia in a 12 –Year-Old Male with CHARGE Association. Journal of Medical Speech-Language Pathology Vol 16 (4), 181-190 [↩] [↩] [↩] [↩]
- Grigos, M. I., Case, J., Lu, Y., & Lyu, Z. (2024). Dynamic Temporal and tactile cueing in young Children with Childhood apraxia of Speech: a Multiple Single-Case design. Journal of Speech Language and Hearing Research, 67(4), 1042–1071. https://doi.org/10.1044/2024_jslhr-23-00415 [↩] [↩] [↩] [↩]
- Kadis, D. S., Goshulak, D., Namasivayam, A., Pukonen, M., Kroll, R., De Nil, L. F., Pang, E. W., & Lerch, J. P. (2013). Cortical thickness in children receiving intensive therapy for idiopathic apraxia of speech. Brain Topography, 27(2), 240–247. https://doi.org/10.1007/s10548-013-0308-8 [↩] [↩] [↩] [↩] [↩] [↩]
- Fiori, S., Pannek, K., Podda, I., Cipriani, P., Lorenzoni, V., Franchi, B., Pasquariello, R., Guzzetta, A., Cioni, G., & Chilosi, A. (2021). Neural Changes Induced by a Speech Motor Treatment in Childhood Apraxia of Speech: A Case Series. Journal of Child Neurology, 36(11), 958–967. https://doi.org/10.1177/08830738211015800 [↩] [↩] [↩] [↩]
- Namasivayam, A. K., Pukonen, M., Goshulak, D., Yu, V. Y., Kadis, D. S., Kroll, R., Pang, E. W., & De Nil, L. F. (2013). Relationship between speech motor control and speech intelligibility in children with speech sound disorders. Journal of Communication Disorders, 46(3), 264–280. https://doi.org/10.1016/j.jcomdis.2013.02.003 [↩] [↩]
- Grigos, M., Hayden, D., & Eigen, J. (2012). Perceptual and Articulatory Changes in Speech Production Following PROMPT Treatment. J Med Speech Lang Pathol. https://pmc.ncbi.nlm.nih.gov/articles/PMC3442609/ [↩]
- Faßbender, L., Falck, J., López, F. M., Shing, Y. L., Triesch, J., & Schwarzer, G. (2025). A comparison of force adaptation in toddlers and adults during a drawer opening task. Scientific Reports, 15(1), 3699. https://doi.org/10.1038/s41598-025-87441-6 [↩]
- Peter, B., Bruce, L., Raaz, C., Williams, E., Pfeiffer, A., & Rogalsky, C. (2020). Comparing global motor characteristics in children and adults with childhood apraxia of speech to a cerebellar stroke patient: evidence for the cerebellar hypothesis in a developmental motor speech disorder. Clinical Linguistics & Phonetics, 35(4), 368–392. https://doi.org/10.1080/02699206.2020.1861103 [↩] [↩] [↩] [↩] [↩]
- Vuolo, J., Kinross, A., & DeHart, K. (2023). Manual rhythmic sequencing skills in children with childhood apraxia of Speech. Journal of Speech Language and Hearing Research, 66(10), 3773–3790. https://doi.org/10.1044/2023_jslhr-23-00141 [↩] [↩]
- Corriveau, K. H., & Goswami, U. (2008). Rhythmic motor entrainment in children with speech and language impairments: Tapping to the beat. Cortex, 45(1), 119–130. https://doi.org/10.1016/j.cortex.2007.09.008 [↩] [↩] [↩] [↩] [↩]
- Gallese, V., Fadiga, L., Fogassi, L., & Rizzolatti, G. (1996). Action recognition in the premotor cortex. Brain, 119(2), 593–609. https://doi.org/10.1093/brain/119.2.593 [↩] [↩]
- Heyes, C. (2009). Where do mirror neurons come from? Neuroscience & Biobehavioral Reviews, 34(4), 575–583. https://doi.org/10.1016/j.neubiorev.2009.11.007 [↩] [↩] [↩]
- Cook, R., Bird, G., Catmur, C., Press, C., & Heyes, C. (2014). Mirror neurons: From origin to function. Behavioral and Brain Sciences, 37(2), 177–192. https://doi.org/10.1017/s0140525x13000903 [↩]
- Iuzzini-Seigel, J. (2019). Motor performance in children with childhood apraxia of speech and speech sound disorders. Journal of Speech Language and Hearing Research, 62(9), 3220–3233. https://doi.org/10.1044/2019_jslhr-s-18-0380 [↩]
- Newmeyer, A. J., Grether, S., Grasha, C., White, J., Akers, R., Aylward, C., Ishikawa, K., & deGrauw, T. (2007). Fine Motor function and Oral-Motor Imitation skills in Preschool-Age children with Speech-Sound Disorders. Clinical Pediatrics, 46(7), 604–611. https://doi.org/10.1177/0009922807299545 [↩]
- Hausen, M., Torppa, R., Salmela, V. R., Vainio, M., & Särkämö, T. (2013). Music and speech prosody: a common rhythm. Frontiers in Psychology, 4, 566. https://doi.org/10.3389/fpsyg.2013.00566 [↩] [↩] [↩] [↩] [↩]
- Beathard, B., & Krout, R. E. (2008). A music therapy clinical case study of a girl with childhood apraxia of speech: Finding Lily’s voice. The Arts in Psychotherapy, 35(2), 107–116. https://doi.org/10.1016/j.aip.2008.01.004 [↩] [↩] [↩] [↩] [↩]
- Lagasse, B. (2012). Evaluation of melodic intonation therapy for developmental apraxia of speech. Music Therapy Perspectives, 30(1), 49–55. https://doi.org/10.1093/mtp/30.1.49 [↩] [↩]