Abstract
DNA mismatch repair (MMR) is a crucial process for preserving genomic stability by correcting DNA replication errors and damage-induced errors. Loss of MMR causes an increase in microsatellite instability and accumulation of mutations that can eventually lead to carcinogenesis. Loss of MMR, which can occur sporadically and in genetic diseases like Lynch syndrome, confers a high risk of developing colorectal cancer. Fortunately, two immune checkpoint inhibitors, Pembrolizumab and Nivolumab, can be used to treat deficient MMR (dMMR) and microsatellite instability high (MSI-H) patients with colorectal cancer. These treatments have shown a higher efficacy and better response rates than conventional chemotherapies, although they do come with some possible side effects. Understanding MMR mechanisms is crucial to healthcare and further research can expand immunotherapies for not just colorectal cancers but other diseases too. This review provides a better understanding of DNA MMR, how it leads to carcinogenesis, some current immunotherapies and their mechanism of action to treat dMMR/MSI-H subtype of colorectal cancer.
Keywords: mismatch repair, Lynch syndrome, colorectal cancer, microsatellite instability, immune checkpoint, Pembrolizumab, Nivolumab.
Introduction
Preservation of genomic integrity in cells is dependent on high fidelity DNA replication and repair of damaged DNA. Even though the DNA replication process is highly accurate, nucleotide misincorporations can still occur occasionally. These can even escape DNA proofreading. In these cases, DNA mismatch repair provides a last line of defense. In addition to correcting base-base mismatches during DNA replication, DNA mismatch repair plays multiple roles in response to various DNA damage-induced mutagenic insults. Therefore, loss of MMR leads to increased mutagenesis and risk of cancer1.
There are four MMR proteins that have clinical relevance in human cancer biology. These are MLH1, PMS2, MSH2, and MSH6, which are encoded by their corresponding MMR genes, MLH1, PMS2, MSH2, and MSH6. All four of these proteins are arranged as heterodimers with MLH1 associating with PMS2 (MutLα) and MSH2 associating with MSH62’3 (MutSα). MSH2 can also associate with MSH3 (MutSβ).
MMR starts with recognition of an error in the DNA strand by MutSα or MutSβ.MutSα complex recognizes base-pair mismatches and small DNA extrusions (1-4 extrahelical residues) whileMutSβ recognizes larger DNA extrusions. Once the mismatch is recognized, theMutSα/β complex will activate either the EXOI exonuclease if strand-break is located 5’ to the mismatch or recruits MutLα (MLH1-PMS2), which contains the latent endonuclease function. Resynthesis then occurs with the help of accessory proteins such as clamp PCNA (Proliferating Cell Nuclear Antigen), clamp loader RFC (Replicating Factor C), and DNA polymeraseδ4. The resulting gap is protected by the single-strand DNA binding protein complex RPA (Replicating Protein A) after which DNA polymerase δ, assisted by the sliding clamp PCNA and the clamp loader RFC resynthesizes the DNA across the gap. If the strand break lies 3’ to the mismatch, the correction will require the help of the orientated loading of PCNA by RFC at the strand break. Therefore, theMutSα/β complex will recruitMutLα, resulting in activation of a latent endonuclease function inMutLα in the presence of DNA-loaded PCNA. The additional strand breaks catalyzed byMutLα, bracket the mismatch, and aids the hydrolysis of the nicked strand MutSα-activated EXOI. This gap is then protected and filled in the same way as the 5’ nick directed reaction4.
Mutations within the MMR genes can cause deficient MMR function, leading to increased DNA damage-induced mutagenesis which is strongly associated with development of carcinogenesis such as colorectal cancer, gastric adenocarcinoma, endometrial carcinoma, ovarian carcinoma, and more. While different cancer types are susceptible to defects in different DNA repair processes, colorectal adenocarcinoma is particularly associated with defects in the MMR pathway5’6. Loss of MMR function also underlies the hereditary disease Lynch Syndrome (LS), an autosomal dominant disorder that occurs from inheriting mutations in genes of the MMR pathway. LS accounts for 3-5 % of all colorectal cancer (CRC). LS patients have a 60-80% chance of developing colorectal cancer in their lifetimes, in addition to certain extracolonic cancers7.
The defective MMR function in Lynch syndrome leads to increased instability of microsatellites which is a hallmark of Lynch syndrome8. Microsatellites are short, repetitive DNA sequences that are prone to errors during DNA replication. Currently, there are two FDA approved medications for the treatment of metastatic colorectal cancer (mCRC) that have deficient mismatch repair (dMMR) and microsatellite instability high (MSI-H). These medications are Pembrolizumab and Nivolumab7.
This article will review ways defects in DNA mismatch repair (MMR) contribute to the development of colon cancer in dMMR-MSIH subtype, such as LS patients, and some drug therapies that can be used to treat these cancers. This is significant as colorectal cancer is one of the leading cancers worldwide and new therapeutic strategies will aid in reducing incidents of mortality. Research allows for better diagnosis and treatment plans for patients with dMMR or MSI-H colorectal cancer which can improve patient outcomes. While further research on this may benefit cancers beyond colorectal cancer, this paper focuses specifically on colorectal cancer in dMMR-MSIH subtypes, with special attention to colorectal cancer in LS patients. The reason for this is that LS provides a genetically precise model to understand the development of colorectal cancer in the context of dMMR-MSIH cancers8. This paper is limited to discussing published peer-reviewed open-access articles and does not discuss any ongoing studies.
Methods
In this paper, I have utilized many resources such as online review articles and research papers. I have chosen 26 articles based on credibility, relevance and publication date. Articles were identified by searching PubMed database using key words that include the following: DNA mismatch repair, MMR drug therapies, immune checkpoint inhibitors, Pembrolizumab, Nivolumab, Lynch syndrome, MMR proteins and their functions, colon cancer treatment and causes of colon cancer. I have excluded articles that are not available for free, are not relevant to the review topic, had duplicate/redundant information and published beyond the last 20 years to avoid any outdated information. The articles that I have chosen to include have direct relevance to the topic and provide credible evidence by ensuring it is peer reviewed.
MMR repairs errors in DNA during DNA replication
During DNA replication, the DNA template strand may not be replicated correctly, which results in errors introduced in the newly synthesized strand. MMR corrects single nucleotide misincorporation, and small insertions/deletion loops created by DNA polymerase. As described above, MMR detects mismatches in DNA introduced during its replication, and once the error is found, it recruits enzymes to excise the error contained in the newly synthesized strand and then resynthesizes the excised sequence. In eukaryotic cells, it differentiates between the DNA strand serving as a template during replication from the newly synthesized strand by recognizing the strand breaks on the newly synthesized strand and the directionality conferred by PCNA (ref for PCNA)9’10. Correcting these replication errors prevents the accumulation of mutations that may lead to tumorigenesis.
MMR repairs damage-induced mutagenic insults
MMR is one of at least 9 pathways that repair DNA damage-induced errors. MMR is neither mutually exclusive nor completely dependent on the other pathways5. MMR plays various roles in response to repair DNA damage such as methylated nucleotides, oxidative DNA lesions and helix-distorting nucleotide lesions including intrastrand crosslinks. MMR is involved in the correction of these base-base mismatches, preventing genomic instability and the suppression of numerous cancers.
Guanines, the preferred target for methylating agents, in its methylated state can be replicated by either translesion synthesis polymerase (TLS) or replicating polymerases. Methylation of guanine alters its structural tautomeric state and causes it to pair with cytosine or thymine in a newly replicated strand. During subsequent cell cycles, attempts to repair the methylated-guanine-thymine mismatchcan lead to the generation of double stranded DNA breaks leading to apoptosis or genomic deletions. Therefore, MMR-induced damage responses can protect the cell from genomic instability by inducing cell death.
Cells are also under constant threat from oxidative DNA damage induced by reactive oxygen species. Replication of oxidized guanine, the preferred base target, can lead to misincorporation of adenine (rather than cytosine), which is then excised by MMR. MMR- deficient cells therefore tend to retain more oxidative lesions than those of MMR-proficient cells.
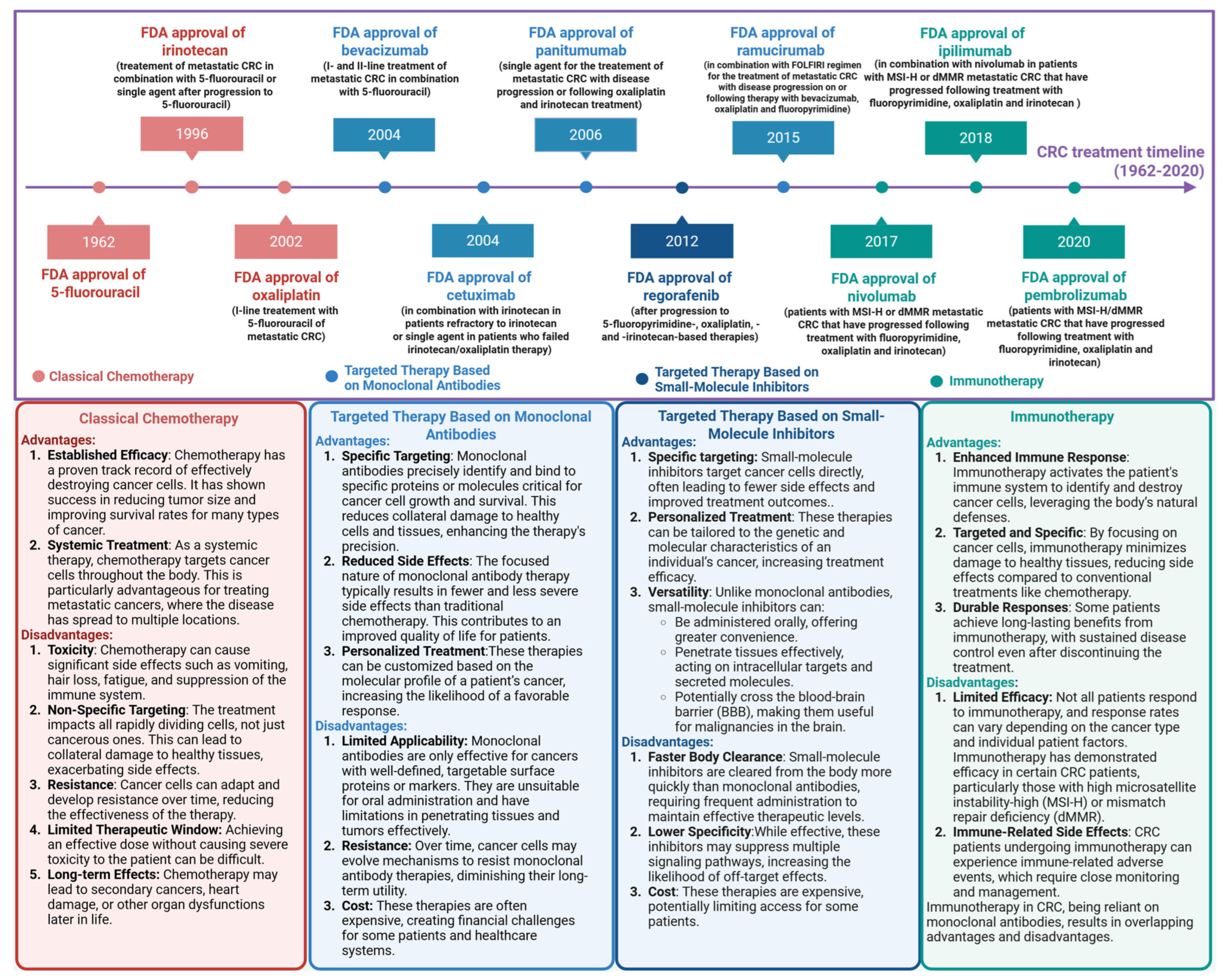
UV radiation may induce helix distorting lesions which include a variety of nucleotide lesion types, including intrastrand crosslinks and nucleotide adducts. The cell employs TLS DNA polymerases to replicate across these damaged nucleotides, which results in
DNA-damaged mutagenesis. MMR can suppress this mutagenesis when it is recruited to sites of localized UV damage. MMR selectively binds “mismatched” nucleotides opposite photolesions. It excises the nucleotide that was mis-incorporated by TLS and therefore decreases DNA damage-induced mutagenesis1. This is illustrated in Figure 1.
MMR, MSI and Tumorigenesis
Defective expression of MMR proteins leads to microsatellite instability (MSI) which causes genomic instability and increased risk of cancer5. Loss of function of MMR protein complexes also leads to mutations causing proto-oncogenes to become oncogenes and impairs tumor suppressor genes. This further leads to increased risk of developing cancer3. Deficient MMR function can occur due to a mutation within an MMR gene or because of inactivation of an otherwise intact MMR gene. These mutations may be present in germline DNA or can occur as somatic events within a tumor3.
The absence of MMR causes an increased DNA damage-induced mutagenesis and is strongly associated with the development of carcinogenesis. When intestinal cells are exposed to mutagenic compounds, this leads to an increased frequency of MMR deficiency and oncogenesis. MMR is lost in roughly 15% of colorectal cancers. Furthermore, microsatellite instability is commonly found in colorectal cancer. Microsatellite unstable tumors (like colorectal cancer) with mutations in MMR genes are characterized by increase in tumor mutation burden. Loss of canonical MMR and exposure to intentional mutagens can also cause an immune response1.
When DNA damage defects occur, they can cause acquired somatic mutations leading to the production of neoantigens, which are new abnormal proteins that appear on the cell surface. The emergence of neoantigens triggers cytotoxic T-cells which can then target and kill cancer cells. Therefore, tumors with a higher number of neoantigens have a higher immunogenicity and allow cytotoxic T cells to kill infected cells and fight cancer5.
Deficient MMR and Tumorigenesis in Lynch Syndrome
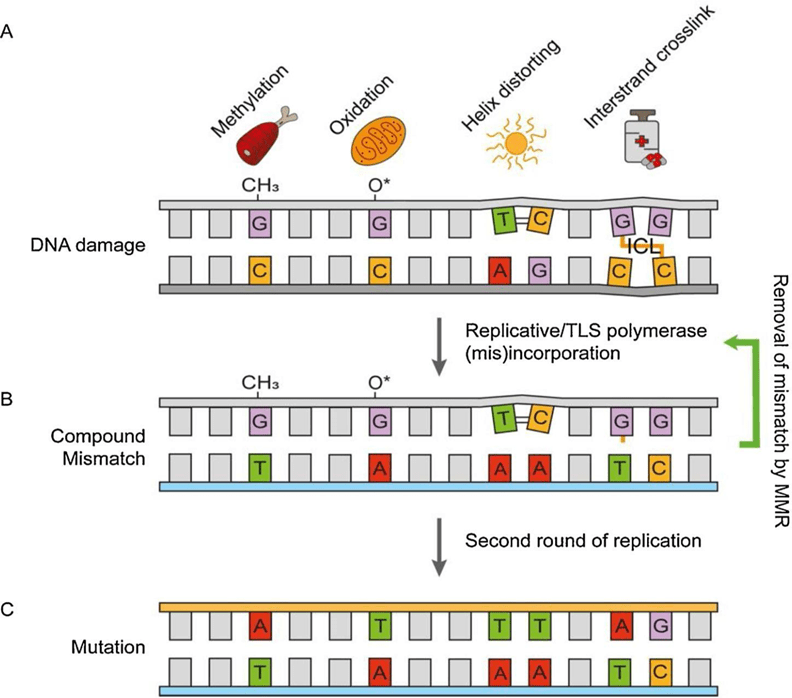
DNA MMR pathway is needed in managing genomic stability through its role in DNA repair. Inherited mutations in genes of the MMR pathway gives rise to Lynch syndrome. The majority of patients carry heterozygous mutations in MSH2 and MSH1 resulting primarily in truncated, non-functional proteins7. These autosomal dominant mutations predispose carriers to various cancers including colon cancer, endometrial cancer, and certain other cancers with a population frequency of about 1 in 279. Cancers of the uterus, ovary, urinary tract, stomach, pancreas, bile duct, small intestine, brain and skin more commonly occur in individuals with Lynch syndrome. For women with Lynch Syndrome, cumulative incidence of any cancer by age 75 ranges from 61.8 % to 81%. For men with Lynch Syndrome, cumulative incidence of any cancer by age 75 ranges from 41.7% to 71.2 %11. The process by which Lynch Syndrome patients develop cancer, is illustrated in Figure 2. The crypt cells in the intestine of Lynch Syndrome patients are all heterozygous for a deficient MMR gene. When exposed to methylating and helix distorting DNA lesions, it can lead to loss of MMR/post TLS repair. This subsequently leads to hypermutagenesis resulting in accumulation of oncogenic mutations. In addition, MMR/post TLS repair deficient cells are more tolerant of methylated and helix distorting DNA damage which lead to further expansion.
The chemotherapy that is typically used for colorectal cancer is a combination of 5-Fluororacil, Leucovorin and either oxaliplatin (FOLFOX) or Irinotecan (FOLFIRI)12. However, chemotherapy such as oxaliplatin does not work well for colon cancers that have dMMR and MSI-H. This is because functional MMR proteins recognize oxaliplatin adducts on DNA and initiate proapoptotic signals. Therefore, loss of MMR rescues cancer cells from apoptosis (cell death) upon oxaliplatin treatment5. Instead, immune therapy can be used rather than chemotherapy for colon cancer with dMMR and MSIH.
Immunotherapy Mechanisms
Immune checkpoints are proteins on immune cells that can play a key role in recognizing foreign pathogens and regulating immune responses5. Interactions between immune checkpoints on a T cell and tumor associated antigen ligands expressed on a tumor cell activate T cells to mount an immune response against the tumor. In human malignancies, the immune checkpoints and their corresponding ligands are usually upregulated. Cancer cells have evolved to evade detection (and subsequent destruction) by upregulating immune checkpoint ligands. Immune checkpoint inhibitors have been developed to prevent evasion of cancer cells from detection and destruction by cytotoxic T-cells. The two most renowned checkpoint blocking approaches are blocking cytotoxic T lymphocyte associated protein 4 (CTLA-4 or CD 152) and targeting interactions between programmed cell death-1 (PD-1 or CD279) and programmed cell death ligand-1 (PD-1 or CD 274 or B7 homolog 1)13’14.CTLA-4 is thought to regulate T-cell proliferation early in the immune response, primarily in lymph nodes. On the other hand, PD-1 suppresses T cells later in an immune response, primarily in the peripheral tissues15.
Costimulation of receptor B7-1/B7-2 and the protein CD28 provide stimulated signals that support T cell proliferation and effective differentiation (Figure 3). Structurally, the transmembrane protein CTLA-4 has significant homology to CD28 but has much higher affinity to bind with the receptors B7-1 and B7-2 molecules compared to CD28. CTLA-4 has a unique YVKM motif which binds to SHP-2 (Src homology 2 containing protein tyrosine phosphatase-2) that elicits inhibiting signals. Throughout the induction phase, the CTLA-4 on T cells obstructs T cell activation by inhibiting the formation of interaction between CD28 and B7-1/B7-2 and conveying inhibitory signals, which directly suppresses T cell activation. This means that hindering CTLA-4 activities could cause T cell response to persist13.
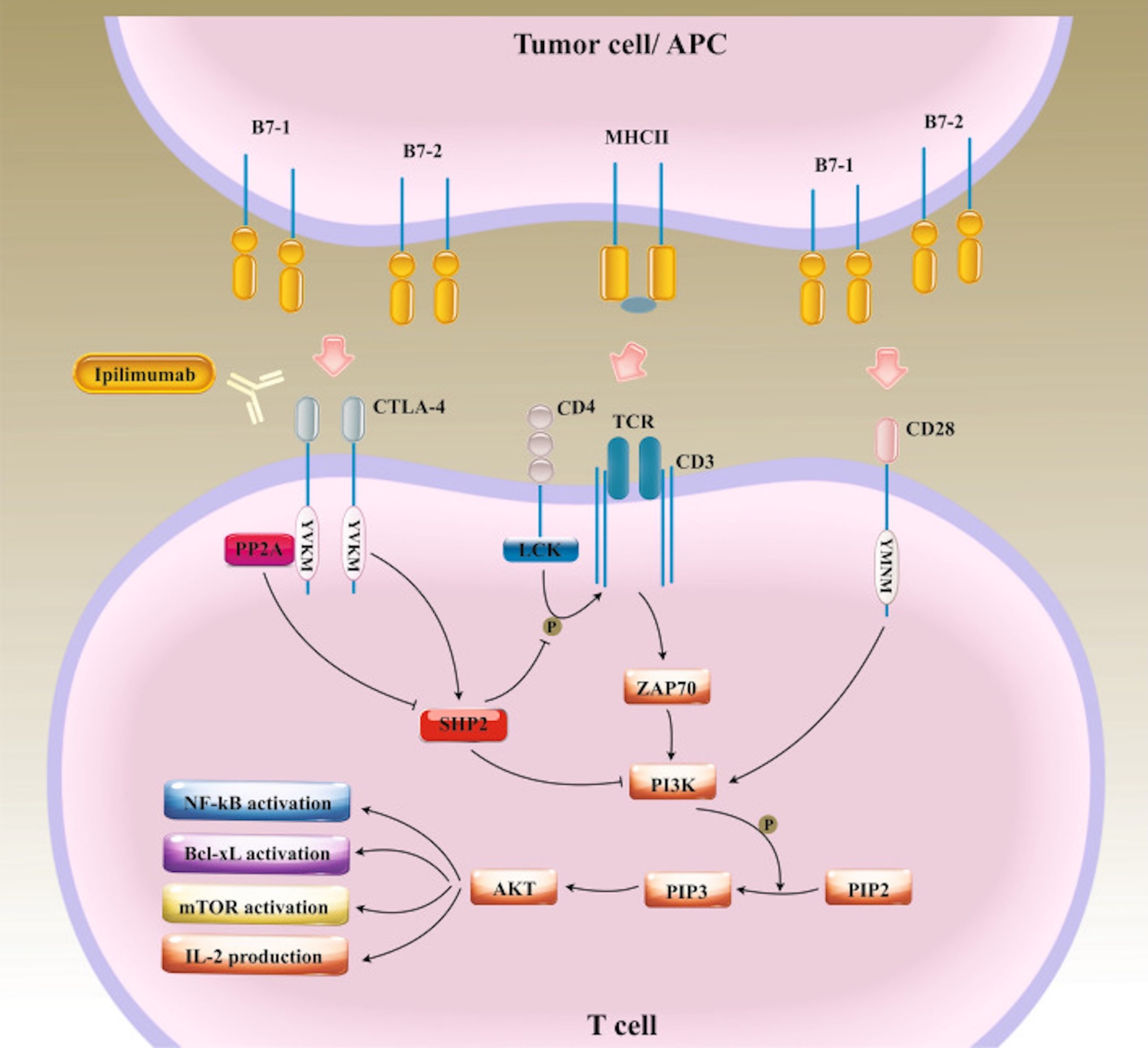
Explanation of terms: Cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), SH2 containing protein tyrosine phosphate -2 (SHP2), phosphoinoside 3 kinases. (PI3Ks), phosphatidylinosital-4, 5-bisphosphate (PIP2), phosphatidylinositol (3,4,5)-trisphosphate (PIP3), lymphocyte-specific protein tyrosine kinase (LCK), T cell receptor (TCR), nuclear factor-kB (NF-kB), mammalian target of rapamycin (mTOR), B-cell lymphoma-extra large (Bcl-xL), major histocompatibility complex class II (MHCII), Interleukin-2 (IL-2). This figure is adapted from Naimi 202213.
PD-1 expressed on T cells can bind PD-L1 and PD-L2 ligands expressed on the surface of various cancer cells. Upon ligand binding and activation, PD-1 cytoplasmic tail containing ITIM and ITSM motifs recruit SHP2 and suppress downstream signaling axes which lead to tumor cell evasion from T cell immunosurveillance13. (Figure 4).
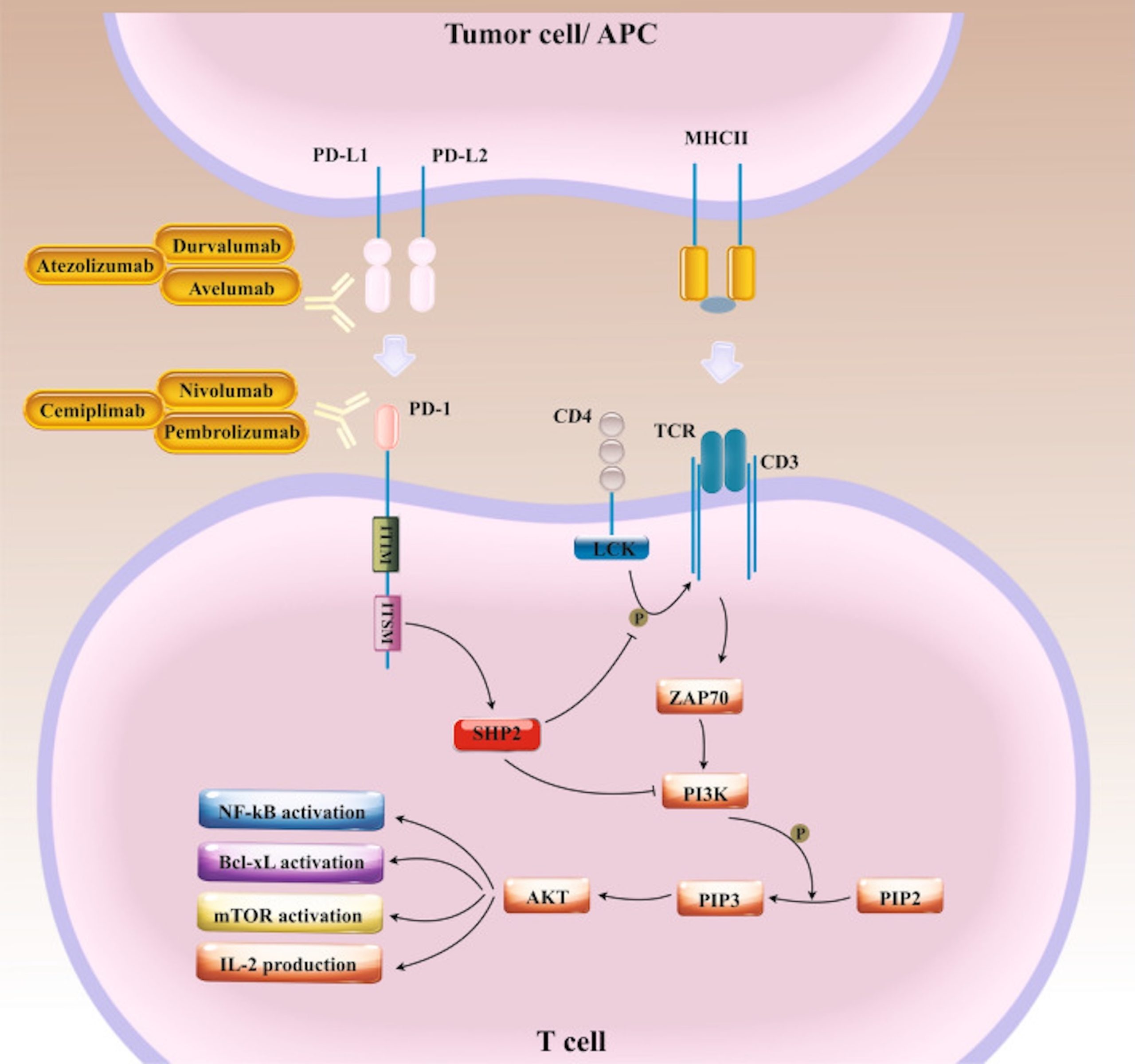
Role of Immunotherapies in Treatment for Colorectal Cancer
Colorectal cancer treatment advancements have led to transitioning from traditional modalities such as surgery, chemotherapy and radiation therapy to contemporary approaches like targeted therapies and immunotherapy. The understanding and awareness of the historical progression of colorectal cancer therapies can help us with future therapeutic innovations in addition to making informed decisions about current treatment16.
Immunotherapies are a significant advancement in colorectal cancer. It provides targeted therapy that is designed specifically to disrupt molecular pathways and processes critical to cancer cell survival and proliferation, unlike conventional chemotherapy that negatively affects both cancers and normal cells16’17’18. Pembrolizumab and Nivolumab are two programmed cell death 1 (PD-1) blocking antibodies that have been approved for treatment of mCRC that is mismatch repair deficient and microsatellite instability high (dMMR-MSI-H)19. Furthermore, Pembrolizumab and Nivolumab can used alone or in combination with Ipilimumab (an immunotherapy that inhibits CTLA-4) to treat mCRC patients with dMMR-MSI-H.
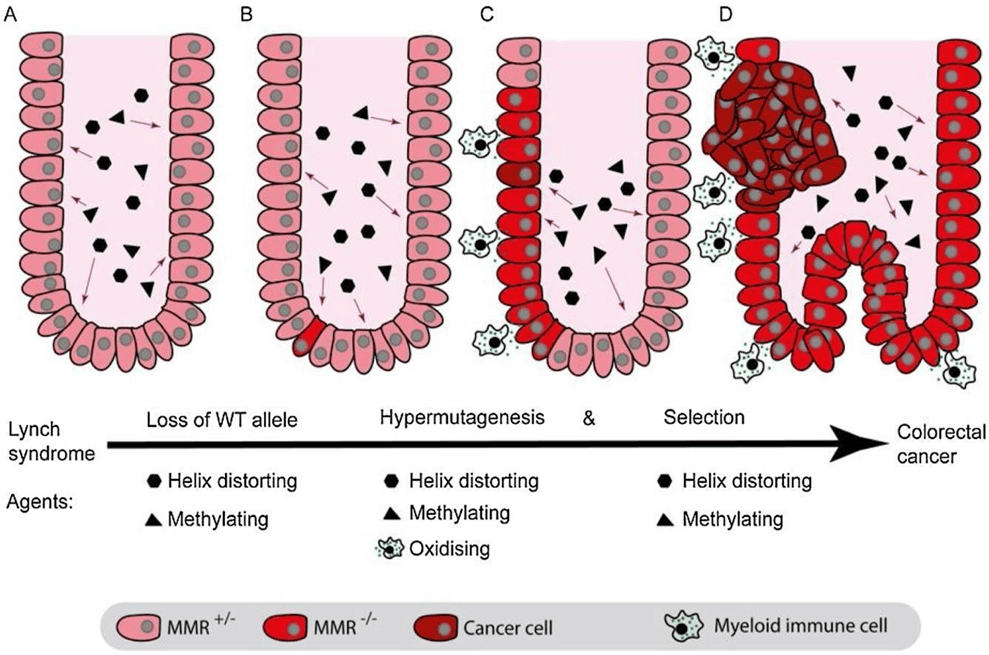
Colorectal cancer treatment modalities have evolved over the last six decades, as illustrated in Figure 5. These modalities include classical chemotherapies, targeted monoclonal antibody therapies, targeted small molecule inhibitor therapies, and more recently immunotherapies, which have only been available since 2015. Each modality has its advantages and disadvantages, as summarized in Figure 5. Many of the disadvantages of earlier therapies include the resistance of cancer cells developing over time, and lower specificity of treatments. The impact of these disadvantages is reduced with immunotherapies.
Figure 5 shows the evolution of colorectal cancer treatments from 1962 to 2020 up to Pembrolizumab and Nivolumab. This includes classical chemotherapeutic agents (red), targeted therapies with the use of monoclonal antibiotics (light blue), small molecule inhibitor (dark blue) and immunotherapeutic agents (light green). The boxes below give a brief overview of their advantages and disadvantages. This is adapted from Kciuk 202516.
Pembrolizumab
Pembrolizumab is a humanized monoclonal anti PD-1 antibody. It blocks the interaction between PD-1 and its ligands. Large scale studies and long term follow ups show high, durable disease control for patients with MSI-H colorectal cancer with Pembrolizumab. The hallmark of MSI-H colorectal cancer is high tumor burden leading to neoantigens, for which immune checkpoint inhibitors have high efficacy20.
Studies have shown that Pembrolizumab significantly prolonged progression free survival (PFS) and overall survival (OS) compared to chemotherapy when used as initial treatment for mCRC with MSI-H and dMMR. KEYNOTE-177 is a phase III study, a trial that tests a treatment efficacy in a larger population relative to a phase II study, that assessed Pembrolizumab monotherapy with standard of care treatment for previously untreated stage four dMMR-MSI-H colorectal cancer patients. The median PFS was 16.5 months in the Pembrolizumab group compared to 8.2 months in the chemotherapy group (hazard ratio 0.59 with 95% CI 0.45-0.79). The median OS rates was 77.5 months with Pembrolizumab versus 36.7 months with chemotherapy (hazard ratio, 0.73; 95% confidence interval 0.53-0.99) with 5-year OS rates being 54.8% versus 44.2%19’21’22.
However, there are also various negative sides to this treatment. For example, having a targeted therapy has limited efficacy. The efficacy of Pembrolizumab is particular to with those who have dMMR-MSI-H tumors. These tumor burdens can lead to a high production of neoantigens, which can make the tumor more prominent to Pembrolizumab, an immune checkpoint inhibitor. Pembrolizumab’s benefit in microsatellite stable (MSS) colorectal cancer, which makes up the majority of colorectal cancer, is still limited due to the lower immunogenicity and less inflamed tumor environment. MSS colorectal cancer tends to have lower tumor mutational burden and lowered immune infiltration which makes it respond poorly to checkpoint blockage16.
Pembrolizumab, like all treatments, does have side effects. These can affect different organs in the body including the skin, the gastrointestinal system, the endocrinological system, the neurological system, the respiratory system, and more23. Additionally, infusion reactions can also occur. Pembrolizumab’s optimal duration of treatment is undefined and may be given up to two years. Therefore, immune related adverse events can occur later and even after cessation of treatment. There have been rare life-threatening or fatal adverse events that have been reported with Pembrolizumab including skin reaction and type 1 diabetes mellitus. The frequency of these adverse reactions ranges from 1.1%-27.3% as illustrated in Table 124. Although many of the side effects are generalized, such as fatigue and pruritus, organ specific side effects like pneumonitis or enterocolitis also occur as illustrated in Table 124. Some of these were fatal.
| Adverse Event | All grade, n(%) | Grade ≥ 3, n(%) |
| Dermatologic | 49 (55.7) | 3 (3.4) |
| Pruritus | 24 (27.3) | — |
| Rash | 23 (26.1) | 3 (3.4) |
| Vitiligo | 2 (2.3) | — |
| Musculoskeletal | 12 (13.6) | 2 (2.3) |
| Myalgia | 5 (5.7) | — |
| Myositis | 1 (1.1) | 1 (1.1) |
| Myasthenia gravis | 1 (1.1) | 1 (1.1) |
| Arthralgia | 3 (3.4) | — |
| Arthritis | 2 (2.3) | — |
| Pulmonary | 11 (12.5) | 9 (10.2) |
| Endocrinopathy | 7 (8.0) | — |
| Gastrointestinal | 5 (5.7) | 1 (1.1) |
| Diarrhea | 4 (4.6) | — |
| Enterocolitis | 1 (1.1) | 1 (1.1) |
| Hepatic | 3 (3.4) | 1 (1.1) |
| Dry mouth | 1 (1.1) | — |
| Total | 88 (100.0) | 16 (18.2) |
Endocrine side effects like hypothyroidism and diabetes are treated without disrupting the Pembrolizumab treatment. For more serious adverse events, for example pneumonitis, the treatment differs. For grade 1 pneumonitis (asymptomatic radiographic changes only), close clinical and radiographic monitoring are usually needed. For grade 2 pneumonitis (moderate symptoms), treatment with corticosteroids is warranted. For grade 3 (severe symptoms) and 4 (life threatening symptoms) pneumonitis, permanent discontinuation of treatment and high dosage corticosteroid are required. In general, cessation of therapy is recommended for any life threatening (grade 4) toxicities, severe (grade 3) toxicities that are recurring, or moderate (grade 2) toxicities that do not resolve with appropriate treatment in 3 months23.
Nivolumab
Nivolumab is a PD-1 inhibitor used to treat colorectal cancer. Nivolumab is usually used in combination therapy with Ipilimumab as it enhances the effectiveness of treatment compared to monotherapy in colorectal cancer16. It has been FDA approved to be used in mCRC with dMMR and MSI-H as monotherapy or in combination with Ipilimumab.
Nivolumab seems to work better for certain atypical malignancies like squamous cell carcinoma of the anal canal (SCCA) compared to conventional chemotherapy as well. SCCA is associated with Human Papillomavirus (HPV), which shows intramural HPV oncoproteins that have been observed to increase the expression of PD-1, which could explain why treatment with Nivolumab is more effective16.
CheckMate 142 is a trial that evaluated efficacy and safety of first line treatment with combined Nivolumab and Ipilimumab in patients with dMMR-MSI-H tumors. When followed up at 13.4 months, the overall response rate (ORR) and disease control rate (DCR) for ≥ 12 weeks was 55% (95% CI, 45.2 to 63.8), and 80%. Additionally, a follow-up at 29 months, DCR was around 84% (95% CI, 70.5 to 93.5) and ORR was 69% (95% CI, 53 to 82)25’26.
Unfortunately, adverse events were also noted with this treatment. Severe infusion reactions are uncommon but have been recorded. However, as with other immunotherapies, there are some immune related adverse events that occur as a consequence of aberrant stimulation of the immune system against normal tissues. The Checkmate-142 trial itself showed that 70·3% had drug-related adverse events, 48·6% had grade 1 or 2 events, 17·6% had grade 3 events and 2·7% had grade 4 events. 1 patient (1.4%) had a grade 5 event of sudden death. The most common side effects were fatigue, diarrhea, pruritus and rash. Other symptoms include hepatitis, hypothyroidism, kidney injury and adrenal insufficiency26.
Discussion
DNA mismatch repair is a complex and significant mechanism utilized by the cells in our body to ensure genomic stability. The MMR machinery plays an integral role in correcting errors in DNA, both during DNA replication and when there are DNA damaged-induced mutagenic insults. Defects in MMR causes multiple negative outcomes including microsatellite instability and carcinogenesis. This article highlights the ways these defects can lead to colorectal cancer. Studies have shown that these tumor cells can manipulate checkpoint inhibitors to evade detection and destruction. Several immunotherapies have utilized these characteristics of the tumor cells to provide checkpoint inhibitor targeted therapies for mCRC, including Pembrolizumab and Nivolumab. These therapies are specifically for dMMR and MSI-H patients. Microsatellite stable (MSS) colorectal cancer, which is characterized by less tumor mutational burden and lower immune infiltration, responds poorly to checkpoint blockage therapies16.
Understanding both the basic mechanism of DNA mismatch repair at each step and how it can go wrong can help guide future studies as well as treatments for a variety of diseases. This paper focuses on one type of cancer (colorectal cancer), but there are numerous diseases that require further research which can lead to better treatments.
While I have explained defects in the MMR’s contribution to the development of colon cancer, there are multiple other DNA defects that may lead to colorectal cancer that is unrelated to mismatch repair. These defects and their mechanisms to the development of colorectal cancer may need to be further studied to fully combat colorectal cancer.
Conclusion
In conclusion, understanding DNA mismatch repair is critical to diagnose and treat certain types of colorectal cancer. As research shows, defects in MMR can lead to mutagenesis and genomic instability, but immunotherapies such as Pembrolizumab and Nivolumab provide effective treatments as an alternative to conventional chemotherapy. Further research to gain a wider understanding of MMR and colorectal cancer can lead to the development of new treatments including other immunotherapies that are effective and safe for a wider variety of patients, with pathologies outside of dMMR and MSI-H.
References
- R. Ijsselsteijn, J.G. Jansen, N.D. Wind. DNA mismatch repair-dependent DNA damage responses and cancer. DNA Repair. Vol. 93, pg. 102923, 2020, https://doi.org/10.1016/j.dnarep.2020.102923 [↩] [↩] [↩] [↩] [↩]
- T. Pal, J. Permuth-Wey, T.A. Sellers. A review of the clinical relevance of mismatch-repair deficiency in ovarian cancer. Cancer, Vol 113(4), pg. 733–742, 2008 https://doi.org/10.1002/cncr.23601 [↩]
- A.C. Bateman. DNA mismatch repair proteins: scientific update and practical guide. Journal of Clinical Pathology. Vol. 74, pg. 264-268, 2021, https://doi.org/10.1111/his.14367 [↩] [↩] [↩]
- R.R. Iyer, A. Pluciennik (2021). DNA Mismatch Repair and its Role in Huntington’s Disease. Journal of Huntington’s disease. Vol. 10(1), pg. 75–94, 2021, https://doi.org/10.3233/JHD-200438A [↩] [↩]
- Klinakis, D. Karagiannis, T.Rampias. Targeting DNA repair in cancer: current state and novel approaches. Cellular and Molecular Life Sciences. Vol. 77, pg. 677-703, 2020, https://doi.org/10.1007/s00018-019-03299-8 [↩] [↩] [↩] [↩] [↩] [↩]
- N.C. Ramchander, N.A. Ryan, E.J. Crosbie, D.G. Evans. Homozygous germ-line mutation of the PMS2 mismatch repair gene: a unique case report of constitutional mismatch repair deficiency (CMMRD). BMC medical genetics, Vol. 18(1), pg. 40, 2017 https://doi.org/10.1186/s12881-017-0391-x [↩]
- A.S Mastrocola, C.D Heinen. Lynch syndrome-associated mutations in MSH2 alter DNA repair and checkpoint response functions in vivo. Human mutation. Vol. 31, pg. 10, 2010, https://doi.org/10.1002/humu.21333 [↩] [↩] [↩]
- P. Peltomäki, M. Nyström, J. Mecklin, T.T. Seppälä. Lynch Syndrome Genetics and Clinical Implications. Gastroenterology. Vol. 164 (5), pg. 783 – 799, 2023, https://doi.org/10.1053/j.gastro.2022.08.058 [↩] [↩]
- M. Elez. Mismatch Repair: From preserving genome stability to enabling mutation studies in real-time single cells. Cells. Vol. 10, pg. 1535, 2021, https://doi.org/10.3390/cells10061535 [↩]
- K. Fukui. DNA mismatch repair in eukaryotes and bacteria. Journal of nucleic acids. 260512, 2010, https://doi.org/10.4061/2010/260512 [↩]
- K.Curtius, S. Gupta, C.R.Boland. Review article: Lynch Syndrome-a mechanistic and clinical management update. Alimentary pharmacology & therapeutics. Vol. 55(8), pg.960–977, 2022, https://doi.org/10.1111/apt.16826 [↩]
- E.J. Kuipers, W.M. Grady, D. Lieberman, T. Seufferlein, J.J. Sung, P.G. Boelens, C.J. Van de Velde, T. Watanabe. Colorectal cancer. Nature reviews. Disease primers. Vol. 1, 15065, 2015, https://doi.org/10.1038/nrdp.2015.65 [↩]
- A. Naimi, R.N. Mohammed, A. Raji, S. Chupradit, A.V. Yumashev, W. Suksatan, M.N. Shalaby, L. Thangavelu, S. Kamrava, N. Shomali, A.D. Sohrabi, A. Adili, A. Noroozi-Aghideh, E. Razeghian. Tumor immunotherapies by immune checkpoint inhibitors (ICIs); the pros and cons. Cell communication and signaling: CCS. Vol. 20(1), pg. 44, 2022, https://doi.org/10.1186/s12964-022-00854-y [↩] [↩] [↩] [↩] [↩]
- D.J. Byun, J.D. Wolchok, L.M. Rosenberg, M. Girotra. Cancer immunotherapy – immune checkpoint blockade and associated endocrinopathies. Nature reviews. Endocrinology, Vol. 13(4), pg. 195–207, 2017. https://doi.org/10.1038/nrendo.2016.205 [↩]
- E.I. Buchbinder, A. Desai. CTLA-4 and PD-1 Pathways: Similarities, Differences, and Implications of Their Inhibition. American journal of clinical oncology. Vol. 39(1), pg. 98–106, 2016, https://doi.org/10.1097/COC.0000000000000239 [↩]
- M. Kciuk, K.Wanke, W.Cruczkowska, B.Marciniak, R.Kontek. Focus on PD-1/PD-L1 – Targeting antibodies in colorectal cancer: are there options beyond Dostarlimab, Nivolumab, and Pembrolizumab? A comprehensive review. Molecules. Vol. 30(13), 2686, 2025, https://doi.org/10.3390/molecules30132686 [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- C.M. Tilsed, S.A. Fisher, A.K. Nowak, R.A. Lake, W.J. Lesterhuis. Cancer chemotherapy: insights into cellular and tumor microenvironmental mechanisms of action. Frontiers in oncology, Vol. 12, pg. 960317, 2022. https://doi.org/10.3389/fonc.2022.960317 [↩]
- M.V. Blagosklonny. (2023). Selective protection of normal cells from chemotherapy, while killing drug-resistant cancer cells. Oncotarget, Vol. 14, pg. 193–206, 2023 https://doi.org/10.18632/oncotarget.28382 [↩]
- A. Fan, B. Wang, X. Wang, Y. Nie, D. Fan, X. Zhao, Y, Lu. Immunotherapy in colorectal cancer: current achievements and future perspective. International Journal Of Biological Sciences. Vol. 17, pg. 3837-3849, 2021, https://doi.org/10.7150/ijbs.64077 [↩] [↩]
- D.C. Guven, G. Kaygaci, E.Erul, M.P.Syed, T. Magge, A. Saeed, S. Yalcin, I.H. Sahin. The efficacy of immune checkpoint inhibitors in microsatellite stable colorectal cancer: a systemic review. The Oncologist. Vol. 29(5), pg. 5, 2024, https://doi.org/10.1093/oncolo/oyae013 [↩]
- L.A Diaz Jr., K.K Shiu, T.W. Kim, B.V. Jensen, L.H. Jensen, C. Punt, D.Smith, R. Garcia-Carbonero, M. Benavides, P. Gibbs, C. de la Fourchardiere, F. Rivera, E. Elez, D.T. Le, T. Yoshino, W.Y. Zhong, D. Fogelman, P. Marinello, T. Andre, KEYNOTE-177 Investigators. Pembrolizumab versus chemotherapy for microsatellite instability-high or mismatch repair-deficient metastatic colorectal cancer (KEYNOTE-177): final analysis of a randomized, open-label, phase 3 study. The Lancet. Oncology, Vol. 23(5), pg. 659–670, 2022, https://doi.org/10.1016/S1470-2045(22)00197-8 [↩]
- T. André, K.K. Shiu, T.W. Kim, V. Jensen, B. L.H. Jensen, C.J.A. Punt, D. Smith, R. Garcia-Carbonero, J. Alcaide-Garcia, P. Gibbs, C. de la Fouchardiere, F. Rivera, E. Elez, D. T. Le, T. Yoshino, Y. Zuo, D. Fogelman, D. Adelberg, L.A. Diaz. Pembrolizumab versus chemotherapy in microsatellite instability-high or mismatch repair-deficient metastatic colorectal cancer: 5-year follow-up from the randomized phase III KEYNOTE-177 study. Annals of oncology : official journal of the European Society for Medical Oncology, Vol. 36(3), pg. 277–284, 2024. https://doi.org/10.1016/j.annonc.2024.11.012 [↩]
- G. Kwok, T.C.C. Yau, J.W. Chiu, E. Tse, Y.L. Kwong. Pembrolizumab (Keytruda). Human Vaccines & Immunotherapies. Vol. 12, pg. 2777-2789, 2016, https://doi.org/10.1080/21645515.2016.1199310 [↩] [↩]
- T. Eun, I.Y. Kim, J.M. Sun, J. Lee, H.S. Cha, E.M. Koh, H. Kim, J. Lee. Risk factors for immune-related adverse events associated with anti-PD-1 pembrolizumab. Scientific reports, Vol. 9(1), pg. 14039, 2019. https://doi.org/10.1038/s41598-019-50574-6 [↩] [↩] [↩]
- H.J. Lenz, E. Van Cutsem, M. Luisa Limon, K.Y.M. Wong, A. Hendlisz, M.Aglietta, P. García-Alfonso, B. Neyns, G. Luppi, D.B. Cardin, T. Dragovich, U. Shah, S. Abdullaev, J. Gricar, J.M. Ledeine, M.J. Overman, S. Lonardi. First-Line Nivolumab Plus Low-Dose Ipilimumab for Microsatellite Instability-High/Mismatch Repair-Deficient Metastatic Colorectal Cancer: The Phase II CheckMate 142 Study. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. Vol. 40(2), pg. 161–170, 2022, https://doi.org/10.1200/JCO.21.01015 [↩]
- M.J. Overman, S. Lonardi, K.Y.M. Wong, H.J. Lenz, F. Gelsomino, M. Aglietta, M.A. Morse, E. Van Cutsem, R.McDermott, A.Hill, M.B. Sawyer, A. Hendlisz, B. Neyns, M. Svrcek, R.A. Moss, J.M. Ledeine, Z.A. Cao, S. Kamble, S. Kopetz, T. André. Durable Clinical Benefit With Nivolumab Plus Ipilimumab in DNA Mismatch Repair-Deficient/Microsatellite Instability-High Metastatic Colorectal Cancer. Journal of clinical oncology: official journal of the American Society of Clinical Oncology. Vol. 36(8), pg. 773–779, 2018. https://doi.org/10.1200/JCO.2017.76.9901 [↩] [↩]