Abstract
Background/Objective: Alzheimer’s disease is a rapidly growing degenerative neurological condition that is a major cause of dementia. Genetics as well as lifestyle factors play a role in its development. Histopathologically, the formation of amyloid beta plaques and tau neurofibrillary tangles leads to degeneration of neurons and loss of neurotransmitters. Recent advances in imaging techniques and molecular biomarkers have enabled early detection of this condition. Despite this, treatment of this condition remains challenging. This review presents a general overview of the causes, clinical presentation and traditional treatment options, followed by a focus on recent advances in diagnostic and treatment modalities.
Methods: A literature search of evidence-based medical databases and peer reviewed research articles is used to describe established causes, traditional diagnosis and medical therapy, recent laboratory and radiological diagnostic tools, newer disease modifying agents available and future research.
Results: Compared to older clinical tools that diagnosed Alzheimer’s disease later in symptomatic individuals, newer diagnostic modalities attempt to identify the disease in earlier stages. For treatment of Alzheimer’s disease, traditional treatment options have limited efficacy. In recent years, immunotherapy has emerged as a new treatment option. Monoclonal antibodies that target amyloid are being considered as disease modifying agents due to radiological reduction in plaque burden on PET scan, when used in early stages of the disease. However, their use is still limited due to availability and adverse effects. Several attempts have been made to develop a vaccine that would prevent or delay the onset of neuronal degeneration in high-risk individuals, with poor or limited results. Further research is needed into targeting the additional pathologies of Alzheimer’s, including the neurofibrillary tangles, and the development of an effective vaccine.
Keywords: Alzheimer’s disease, amyloid plaque, tau protein, Alzheimer’s biomarkers, amyloid targeted therapy, Alzheimer’s vaccine
Introduction
Dementia, a medical condition that impairs a person’s ability to think, reason, and remember to an extent that interferes with daily life, is a rapidly growing problem. It is estimated that the number of people with dementia would increase from 57 million globally in 2019 to 152 million in 20501. Alzheimer’s Disease (AD), named after German psychiatrist Alois Alzheimer, is the most common cause of dementia, affecting an estimated 7.2 million Americans above the age of 652. Other less common etiologies of dementia include cerebrovascular disease (strokes), frontotemporal degeneration, and Lewy body dementia, but AD is the most common and most rapidly growing category, as well as the seventh leading cause of death in the United States, as of 20222.
As opposed to other forms of dementia, individuals with AD demonstrate very characteristic changes in their brains, namely deposits of amyloid beta in extracellular tissue and accumulation of tau protein intracellularly3. These changes are the foundation of recent advances that have been made in the diagnosis and treatment of this disease.
This report highlights the following aspects of AD: 1) Genetics and Pathophysiology, 2) Traditional diagnostic tools and treatments, 3) Recent diagnostic techniques that enable early detection, 4) Novel immunotherapeutic treatment options, and 5) Areas of current and future research.
Methods
A literature search was done using the following keywords: Alzheimer’s disease, amyloid plaque, tau protein, Alzheimer’s biomarkers, amyloid targeted therapy, Alzheimer’s vaccine. Databases searched were Pubmed, UpToDate, Google Scholar, ClinicalTrials.gov, and FDA.gov.
The date range for the search was 1990 to current. A total of 74 articles were reviewed. Exclusion criteria were articles related to non-Alzheimer’s dementia, animal-only studies, and abstract-only publications
For established pathophysiology, clinical features and traditional management of AD, information from articles presenting a comprehensive review of AD, primary research articles, and peer reviewed articles pertaining to the diagnosis and treatment of AD, were selected. For recent advances review, articles published after 2000 describing Alzheimer’s laboratory assays, imaging techniques, amyloid-targeted therapy, immunotherapy, treatment trials and vaccine trials were included. 41 articles were listed as references.
Discussion
Genetics of Alzheimer’s disease
AD affects women more commonly than men, with a lifetime risk of nearly 1 in 5 for women and 1 in 10 for men2. Alzheimer’s can be either Early-Onset or Late-Onset.
Early-Onset AD is autosomal dominant and shows the strongest genetic link. The 3 genes with the strongest association to Early-Onset AD are amyloid precursor protein (APP), presenilin 1 (PSEN1), and presenilin 2 (PSEN2). APP is located on chromosome 21q, PSEN1 on chromosome 14q, and PSEN2 on chromosome 1q. Families with these genetic variants have individuals who develop symptoms between the ages of 30 and 60 years. The criteria for autosomal dominant AD are at least 3 affected individuals in 2 or more generations, with 2 of the individuals being first-degree relatives of the third. Individuals with Down syndrome (Trisomy 21) commonly develop AD by their fifth decade of life due to the presence of an extra copy of the APP gene on the additional chromosome 21 that they carry4.
Late-Onset AD has a more complex interplay of genetic and environmental factors, but the gene that has the most established connection is apolipoprotein E (APOE). APOE is located on chromosome 19 and has 3 alleles: epsilon 2, 3, and 4. The APOE epsilon 4 (APOE ϵ4) has the highest correlation with Late-Onset AD. However, unlike the genes in Early-Onset AD, the presence of APOE ϵ4 does not directly lead to the development of AD. Rather, APOE ϵ4 increases the susceptibility of the individual to AD depending on various factors like sex (women with APOE ϵ4 are more susceptible than men), smoking, diabetes, hypertension, hyperlipidemia, etc5 .
The proposed mechanism by which APOE ϵ4 leads to the development of AD is its effect on amyloid. Apolipoprotein is a prevalent lipoprotein in the brain, playing a role in neuronal protection and repair by facilitating the removal of amyloid-beta from the brain into the bloodstream. As opposed to the other alleles APOE ϵ2 (cysteine/cysteine) and APOE ϵ3 (cysteine/arginine), APOE ϵ4 (arginine/arginine) is not as efficient at transferring amyloid beta to the bloodstream, resulting in amyloid plaque buildup in the brain6.
AD is more commonly seen in women than men, especially related to the loss of estrogen after menopause. The Estrogen Receptor Gene (ESR) codes for estrogen receptors ER𝛂 and ERβ. These receptors are found in the hypothalamus, amygdala, hippocampus, and cortex and play a role in the higher prevalence of AD in women7.
Pathology of Alzheimer’s Disease:
The hallmark of AD is the deposition of amyloid beta protein in the extracellular brain tissue. There is overproduction and reduced clearance of amyloid beta peptides, resulting in the formation of amyloid plaques3,8,9. The other protein involved is tau, a microtubule-associated protein that aids in microtubule assembly and stabilization. In AD, tau is hyperphosphorylated, leading to the formation of intracellular neurofibrillary tangles. These changes lead to neuronal death and gradual brain atrophy10,11.
The above structural changes contribute to the loss of neurotransmitters, especially Acetylcholine (ACh). Acetylcholine plays a crucial role in several physiological processes, including memory, attention, sensory information, and learning. Lack of sufficient Ach leads to defects in memory and cognition7.
Another neurotransmitter that plays a role is Glutamate, which binds and excites N-methyl-D-aspartate (NMDA) receptors. Overstimulation of NMDA leads to excitotoxic neuronal damage12.
Understanding these pathological changes is critical in the development of drugs and therapies that specifically target these deficits.
Risk Factors for Alzheimer’s Disease
Age and family history have the highest correlation in developing AD. Although Early-Onset AD can be seen in people less than 65 years of age, it forms a small percentage of total Alzheimer’s cases. Being over 65 years of age or having a family history of AD in first-degree relatives has a strong association with the development of AD2.
Vascular risk factors like hypertension, dyslipidemia, cerebrovascular and cardiovascular disease, diabetes, and obesity are associated with AD. Aggressive management of these conditions can lead to reducing the risk and progression of the disease 13.
Lifestyle factors like reduced physical activity, poor diet, smoking, including exposure to secondhand smoke, excessive alcohol consumption, as well as exposure to pesticides play a role. Poor sleep quality and chronic stress also show a correlation. These are modifiable factors, and several studies have shown reduced cognitive decline in physically active individuals13.
Social isolation in the elderly is being recognized as a factor in cognitive decline and a contributor to AD14.
Clinical Features and Symptoms of Alzheimer’s Disease
Early symptoms
Impaired memory is the initial and most common symptom. Memory of recent events is affected in AD in the earliest stages, with relative sparing of memory of more distant events. Sleep disturbance is commonly seen and may occur early on. Other cognitive deficits include impaired language and word-finding difficulties. Psychosocial changes like apathy and social withdrawal may lead to a mistaken diagnosis of depression15.
Impairment of executive function and problem-solving leads to the affected individual appearing less organized and with particular difficulty in multitasking16. These concerns are usually reported by family members, as the patients themselves commonly have anosognosia (reduced insight into their deficits), causing them to offer explanations and excuses17.
Late symptoms
Loss of remote memory is seen in advanced disease and so is loss of procedural memory and motor learning skills (subcortical functions) Other behavioral changes like agitation, aggression, and wandering can be seen later in the course of the disease15.
Apraxia or dyspraxia, which is the inability or difficulty performing learned motor tasks, occurs later and leads to the individual being unable to perform activities of daily living like self–care18. Abnormal motor movements, including seizures, loss of reflexes, and incontinence, are also late findings19.
Diagnosis of Alzheimer’s Disease
Traditionally, the diagnosis of Alzheimer’s disease was dependent on a clinical evaluation using various cognitive assessment tests, while imaging studies like CT and MRI showed cerebral changes in advanced disease. In recent years, additional tools have been developed which include measurement of molecular markers in CSF and blood, and PET scans. Amyloid PET scans are being used more frequently, but Tau PET scans have limited availability.
Clinical Assessments
The initial assessment involves a clinical evaluation. Various tools are available; the commonest one used by most clinicians is the Mini-Mental State Examination (MMSE) (Figure 1), which is a series of questions answered by the patient that test their memory, ability to recall, and cognitive functions. Other standard mental status scales are the Montreal Cognitive Assessment (MoCA), National Institute on Aging and the Alzheimer’s Association (NIA-AA) scale, Diagnostic and Statistical Manual of Mental Disorders (DSM) criteria, and Clinical Dementia Rating (CDR).
| Max Score | Patient Score | Questions |
| 5 | What is the Year? Season? Date? Day? Month? (1 point each) | |
| 5 | Where are we: State? Country? Town? Hospital? Floor? (1 point each) | |
| 3 | Examiner names 3 objects and asks subject to repeat them (1 point each) | |
| 5 | 100-7 test (count backwards from 100 by sevens) OR spell WORLD backwards (D-L-R-O-W) | |
| 3 | Ask subject to recall the 3 objects above (1 point each) | |
| 2 | Show subject simple objects like watch and pencil and ask them to name them | |
| 1 | Have subject repeat the phrase “No ifs, ands or buts” | |
| 3 | Give subject a blank piece of paper and have them follow the instructions “ Take the paper in your right hand, fold in half, and put it on the floor” | |
| 1 | Read and follow the command “CLOSE YOUR EYES” | |
| 1 | Write a sentence (sentence should contain a noun and verb) | |
| 1 | Copy this picture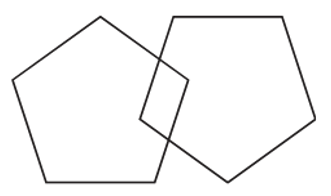 | |
| 30 | Total |
Neuroimaging:
MRI scans in AD show brain atrophy, particularly in the hippocampus and medial temporal lobe.
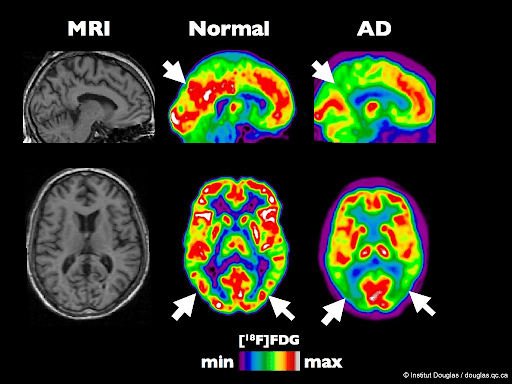
PET scan (Figure 2) shows areas of hypometabolism in brains of AD patients
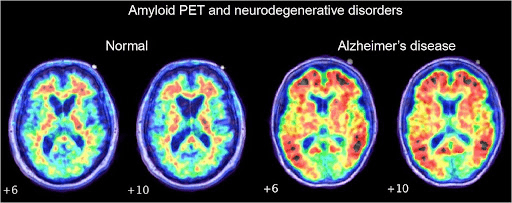
Amyloid PET (Figure 3) is a specialized PET scan that uses amyloid tracers (florbetapir F-18, florbetaben F-18) that highlight the amyloid deposits in the brain. This helps differentiate AD from other forms of dementia, and is a useful tool to use when amyloid-targeting medications like Lecanemab are being considered as treatment.
An alternate method of detection is Tau PET imaging, where PET scans using a tau tracer (flortaucipir F-18) can measure the tau burden in an AD brain.
Biomarkers:
Molecular markers can be measured either in the cerebrospinal fluid (CSF) or blood. In AD, as the amyloid accumulates in the brain, the CSF shows low levels of beta amyloid 42 (Aβ42) as it is secreted less into the cerebrospinal fluid. Low Aβ42 or low Aβ42/Aβ40 ratio in CSF is indicative of AD. On the other hand, CSF levels of Total tau and phospho-tau increase23.
Since obtaining CSF requires the patient to undergo a relatively invasive procedure, namely lumbar puncture, blood tests have been developed that can measure plasma levels of these biomarkers and are a significant landmark in the early detection and diagnosis of AD.
Various laboratory assays are being developed to measure different biomarker levels. The Simoa pTau assays for different pTau isoforms have shown elevated levels of pTau in AD. For example, the Simoa pTau181 assay was seen to have 100% sensitivity and 89% specificity , with AUC of 0.938 (95% confidence interval of 0.872 – 1.000). The SimoapTau217 had a sensitivity of 92% and specificity of 100% , with AUC 0.995 (95% CI 0.987-1.000)23.
The Lumipulse pTau217 assay showed a positive predictive value of 92% and negative predictive value of 85%, with AUC of 0.95 (95% CI 0.94 – 0.96) 24. In May 2025, the FDA granted a Breakthrough Device designation to the Lumipulse G pTau217/beta amyloid 1-42 ratio test, based on its PPV of 91.7% and NPV of 97%25. Given the invasiveness of CSF studies, and the high cost as well as limited availability of amyloid PET scans, these blood tests are emerging as convenient, easily accessible, and more cost-effective diagnostic tests for AD26.
Additional blood tests for other markers associated with AD are currently being studied, including microRNAs (miRNAs) and neurofilament light chain levels15.
Given the development and availability of these recent diagnostic tools, the National Institute on Aging and Alzheimer’s Association has proposed a scheme for a standardized biological definition of AD called the ATN classification based on the Amyloid, Tau and Neurodegeneration markers as follows:
A: Aggregated Aβ = CSF Aβ42 or Aβ42/Aβ40 ratio, Amyloid PET
T: Aggregated Tau = CSF pTau, Tau PET
N: Neurodegeneration = Neuronal injury seen on MRI, FDG PET or CSF total tau.
Presence of both A and T markers is required to diagnose “Alzheimer’s Disease”, while presence of A without the T markers is designated “Alzheimer’s pathologic change”. The N markers, although not strictly required to diagnose AD, provide a strong correlation to pathologic staging and cognitive decline27.
Clinical Classification of Alzheimer’s Disease
Using all the diagnostic tools mentioned above, AD can be classified into: Preclinical Alzheimer’s Disease, Mild Cognitive Impairment (MCI), and clinical Alzheimer’s Dementia28,29.
Preclinical Alzheimer’s disease:
- Stage 1: Asymptomatic amyloidosis – High PET amyloid tracer retention, low CSF Aβ42
- Stage 2: Amyloidosis + Neurodegeneration – Neuronal dysfunction on FDG PET, high CSF tau/phospho-tau, atrophy on MRI
- Stage 3: Amyloidosis + Neurodegeneration + Subtle Cognitive Decline – demonstrate subtle change from baseline in cognitive function, poor performance on more challenging cognitive tests, but do not meet criteria for MCI.
Mild Cognitive Impairment (MCI):
There is evidence of memory loss, but general cognitive and social functioning is preserved. The person can still function independently.
Clinical Alzheimer’s Dementia:
The cognitive decline is significant enough to affect daily functioning. This is further classified into
- Mild dementia – MMSE score 19 to 26
- Moderate dementia – MMSE score 10 to 18
- Severe dementia – MMSE score <10.
Treatment of Alzheimer’s Disease
Although AD has been a progressive condition leading to inevitable neurological decline, some therapies are available that improve cognitive functions and slow down the progression of the disease.
Cholinesterase inhibitors:
People affected by AD have reduced levels of acetylcholine, a key neurotransmitter required for cortical function. The enzyme cholinesterase plays a role in breaking down acetylcholine. Cholinesterase inhibitors like donepezil, rivastigmine, and galantamine reduce the activity of this enzyme, thereby increasing the levels of acetylcholine. Although these drugs do not change the underlying progression of the disease, they do improve symptoms by having a positive impact on cognition and daily functioning. Their use is limited by their adverse effects, such as nausea, dizziness, fatigue, headaches, increased urination, and incontinence, as well as by the worsening of the underlying disease12,30.
Memantine:
The cortical and hippocampal neurons in AD show an excessive activity of receptors called N-methyl-D-aspartate (NMDA). Memantine blocks these NMDA receptors and improves memory function. However, it too cannot modify the course of the underlying disease12,30.
Amyloid Targeted Therapies:
Immunotherapy has emerged as the latest frontier in the treatment of AD. Recent development of recombinant monoclonal antibodies like lecanemab and donanemab has offered new strategies in the treatment of AD. These antibodies bind to and remove amyloid, resulting in a reduction in amyloid plaque burden on PET scans. Thus, they are considered to be disease-modifying and can slow the progression of AD12,30.
The pivotal clinical trials for these drugs are described below (Table 1, Table 2), and showed reduction in amyloid on imaging studies, with modest improvements in clinical scores and disease progression31,32,33,34.
Similarly, antibodies that target tau are in the process of being developed and studied, although none are currently approved by the FDA7.
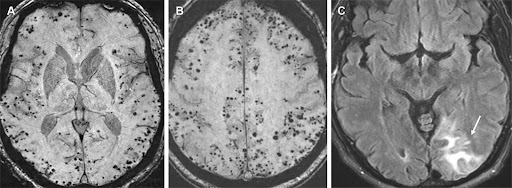
Although these antibodies have offered a novel treatment approach for AD patients, they have also been linked to certain complications, notably Amyloid-related imaging abnormalities or ARIA (Figure 4). ARIA is being increasingly recognized in people treated with Lecanemab and Donanemab, in the form of ARIA-E (edema or effusion) and ARIA-H (hemosiderosis or hemorrhage). ARIA-E is characterized by swelling or fluid under the meninges. ARIA-H presents as bleeding, usually microhemorrhages, or iron deposition (hemosiderosis) from the breakdown of these areas of hemorrhage. Although most patients are asymptomatic, some may develop headaches, confusion, nausea, vomiting, and vision or gait disturbances35,36.
| Study (Ref) | Design | Phase | Severity of AD | Drug | Duration |
| Swanson et al 2021 n = 854 (28) | Multicenter, randomized, double blind, placebo controlled | 2 | Mild | Lecanemab | 18 months |
| Van Dyck et al 2023 n =1795 (29) | Multicenter, randomized, double blind, placebo controlled | 3 | Mild | Lecanemab | 18 months |
| Mintun et al 2021 n = 257 (30) | Multicenter, randomized, double blind, placebo controlled | 2 | Early symptomatic AD | Donanemab | 76 weeks |
| Sims et al 2023 n = 1736 (31) | Multicenter, randomized, double blind, placebo controlled | 3 | MCI and Mild AD | Donanemab | 76 weeks |
| Study | Primary outcome: Change in clinical score | Secondary outcome: Change in amyloid on PET scan | ARIA – E | ARIA – H | Discontinuation rate |
| Swanson et al 2021 n = 854 (28) | Goal (80% probability to be better than placebo) not met | Goal (reduced brain amyloid) met | Drug 9.9% Placebo 6% | Drug 10.7% Placebo 5% | Drug 14.9% Placebo 6% |
| Van Dyck et al 2023 n =1795 (29) | Drug 1.21 Placebo 1.66 (in favor of drug) | Drug -55 Placebo +3.6 | Drug 12.6% Placebo 1.7% | Drug 17.3% Placebo 9% | Drug 6.9% Placebo 2.9% |
| Mintun et al 2021 n = 257 (30) | 25% slower disease progression | 67% achieved negative PET status | Drug 27% Placebo 0.8% | Drug 30.5% Placebo 7.2% | Drug 30.5% Placebo 7.2% |
| Sims et al 2023 n = 1736 (31) | 35% slowing of disease progression | 76% achieved amyloid clearance | Drug 24% Placebo 2% | Drug 19.7% Placebo 7.4% | Drug 13% Placebo 4.3% |
Management of Risk Factors
Treatment of underlying conditions like hypertension, hyperlipidemia, diabetes, obesity, vascular disease, and lifestyle interventions like exercise can reduce cognitive decline.
Non-pharmacological Management Strategies
Cognitive stimulation through mental games, reality orientation by providing environmental cues, and occupational therapy to reinforce fading skills can keep a person with AD more functional for longer periods of time. There is some evidence to show the beneficial effects of the Mediterranean diet. Since people suffering from AD have concomitant mood disorders like depression and anxiety, as well as disrupted sleep cycles, treatment of these conditions goes a long way in improving their quality of life. Good sleep hygiene and socializing also show some improvement in cognitive function15. Music therapy through MEAMs (music evoked autobiographical memories) can also be helpful38.
The benefits of regular physical activity in dementia are now clearly documented in several studies39. Aerobic exercise of 30 min per session, up to 150 min per week, and up to 3 times per week showed measurable improvement in cognition as seen by an increase in MMSE scores40.
These measures improve the functioning and quality of life of the person suffering from AD and also reduce caregiver burden.
Future Developments and Quest for a vaccine
Although amyloid targeted therapies have emerged as effective recent tools, further research is ongoing to address the other pathologies in AD, particularly tau neurofibrillary tangles7. Clinical trials studying agents such as tideglusib, saracatinib, and nilotinib that target tau protein have shown some promise41,42,43. Efforts are underway to develop a potential vaccine against beta amyloid. Additionally, stem cell therapy using neuronal stem cells is the target of future research15.
While amyloid targeted therapies like lecanemab and donanemab are examples of passive immunotherapy, research is being conducted to develop an effective vaccine, which would be a form of active immunotherapy, to stimulate a person’s immune system to produce antibodies that target and remove the amyloid plaques and tau tangles44.
Some initial vaccine trials (AN1792, CAD106) were terminated due to lack of safety and efficacy. Currently, two vaccine trials are underway. A phase 2A trial showed the UB-311 vaccine was safe, well-tolerated, and generated a robust immune response30,45,46. Another vaccine, ABvac40, also showed sustained immune response and no serious adverse effects in a phase 2 study30,47,46.
Conclusion
Alzheimer’s Disease is a chronic progressive neurodegenerative condition that leads to both mental and physical decline and significantly affects the quality of life of the people suffering from it as well as their caregivers. Its prevalence is projected to increase significantly and have a large impact on healthcare. Early diagnosis and intervention are crucial to maintain the mental faculties and functional status of the affected individual.
Recent advances in diagnostic tools enable early identification of affected individuals before their disease burden becomes severe. Simultaneously, the development of drugs and treatment strategies that can slow the progression of the disease and improve quality of life offers a glimmer of hope to persons affected by this terminal disease. Effective strides have been made in recent years, particularly in the area of immunotherapy, to develop novel treatment options. Ongoing research that explores new treatment targets and efforts to develop an effective vaccine are the cornerstone of future developments in the fight against this debilitating disease.
Acknowledgement
I am grateful to Dr Aman Deep, Neurologist at Mary Washington Healthcare, Fredericksburg, VA for his valuable time and input, as well as for the images he very kindly provided for use in this article.
Summary of All References
| Author | Journal | Type | Topic |
| GBD Dementia Collaborators, 20221. | Lancet Public Health | Statistics | Epidemiology |
| Alzheimer’s Association, 20252. | Alzheimer’s & Dementia | Statistics | Epidemiology |
| DeTure et al, 20193. | Molecular Neurodegeneration | Review | Pathology |
| Breijyeh et al, 20207. | Molecules | Review | Causes and Treatment |
| Cacace et al, 20164. | Alzheimer’s & Dementia | Review | Genetics |
| Bertram et al, 20075. | Nature Genetics | Systematic Meta-Analysis | Genetics |
| Slooter et al, 20046. | Neurology | Prospective cohort | Genetics |
| Han et al, 20178. | Scientific Reports | Basic research | Pathology |
| Vidoni et al, 20169. | Neurodegenerative Diseases | Retrospective cohort | Pathology |
| Medina et al, 201410. | Frontiers in Cellular Neuroscience | Review | Pathology |
| Braak et al, 200611. | Acta Neuropathologica | Basic research | Pathology |
| Zhang et al, 202412. | Signal Transduction and Targeted Therapy | Review | Pathology and Treatment |
| Silva et al, 201913. | Journal of Biomedical Science | Review | Risk factors |
| Drinkwater et al, 202214. | European Journal of Neuroscience | Review | Risk factors |
| Safiri et al, 202415. | Frontiers in Medicine | Review | Comprehensive AD review |
| Stokholm et al, 200616. | Dementia and Geriatric Cognitive Disorders | Case Control Study | Clinical symptoms |
| Harwood et al, 200517 | American Journal of Geriatric Psychology | Observational study | Clinical symptoms |
| Vakkila et al, 202318. | Journal of Clinical and Experimental Neuropsychology | Systematic Review | Clinical symptoms |
| Scarmeas et al, 200919. | Archives of Neurology | Prospective cohort study | Clinical symptoms |
| Bayoumy et al, 202123. | Alzheimer’s Research & Therapy | Case Control study | Diagnosis/ Biomarkers |
| Palqvist et al, 202524. | Nature Medicine | Cohort study | Diagnosis/Biomarkers |
| Figdore et al, 202426. | Alzheimer’s &Dementia | Retrospective Case Control study | Diagnosis/Biomarkers |
| Jack et al, 201827. | Alzheimer’s &Dementia | Guidelines | Diagnosis |
| McKhann et al, 201128 | Alzheimer’s &Dementia | Guidelines | Diagnosis |
| Sperling et al, 201129. | Alzheimer’s &Dementia | Guidelines | Classification |
| Mei et al, 2024,30. | Brain-X | Review | Treatment |
| Swanson et al, 202131. | Alzheimer’s Research & Therapy | Phase 2b Randomized Controlled Trial | Treatment/ Monoclonal antibodies |
| Van Dyck et al, 202332. | The New England Journal of Medicine | Phase 3 Randomized Controlled Trial | Treatment/ Monoclonal antibodies |
| Mintun et al, 202133. | The New England Journal of Medicine | Phase 2 Randomized Controlled Trial | Treatment/ Monoclonal antibodies |
| Sims et al, 202334. | Journal of American Medical Association | Phase 3 Randomized Controlled Trial | Treatment/ Monoclonal antibodies |
| Hampel et al, 202335. | Brain | Review | Treatment adverse effects |
| Cogswell et al, 202536. | American Journal of Neuroradiology | Guidelines | Treatment monitoring |
| Garcia-Navarro et al, 202238. | Nursing Reports | Systematic Review | Treatment |
| Zeng et al, 202339. | Geriatric Nursing | Meta-Analysis | Treatment |
| Zhang et al, 202240. | International Journal of Environmental Research and Public Health | Meta-Analysis | Treatment |
| Pluta et al, 202041. | Alzheimer’s Disease: Drug Discovery | Review | Experimental therapies |
| Lovestone et al, 201542. | Journal of Alzheimer’s Disease | Phase 2 Randomized Controlled Trial | Experimental therapies |
| Nygaard et al, 201543. | Alzheimer’s research & Therapy | Phase 1b Randomized Controlled Trial | Experimental therapies |
| Bhadane et al, 202444. | Brain Research | Review | Vaccine |
| Yu et al, 202345. | eBioMedicine | Phase 2a Randomized Controlled Trial | Vaccine |
| Pascual-Lucas et al, 202547. | Alzheimer’s &Dementia | Phase 2 Randomized Controlled Trial | Vaccine |
References
- GBD 2019 Dementia Forecasting Collaborators: Estimation of the global prevalence of dementia in 2019 and forecasted prevalence in 2050: an analysis for the Global Burden of Disease Study 2019. Lancet Public Health. Vol 7, pg. E105-e125, 2022, https://doi.org/10.1016/s2468-2667(21)00249-8. [↩] [↩]
- 2025 Alzheimer’s disease facts and figures. Alzheimer’s & Dementia. Vol. 21,pg.e70235, 2025, https://doi.org/10.1002/alz.70235. [↩] [↩] [↩] [↩] [↩]
- M.A. DeTure, D.W. Dickson. The neuropathological diagnosis of Alzheimer’s disease. Molecular Neurodegeneration. Vol. 14, pg. 32, 2019, https://doi.org/10.1186/s13024-019-0333-5 . [↩] [↩] [↩]
- R.Cacace, K. Sleegers, C. Van Broeckhoven. Molecular genetics of early-onset Alzheimer’s disease revisited. Alzheimer’s & Dementia. Vol 12, pg. 733-748, 2016, https://doi.org/10.1016/j.jalz.2016.01.012. [↩] [↩]
- L. Bertram, M.B. McQueen, K. Mullin, D. Blacker, R.E. Tanzi. Systematic meta-analysis of Alzheimer disease genetic association studies: the AlzGene database. Nature Genetics. Vol 39, pg. 17-23, 2007, https://doi.org/10.1038/ng1934. [↩] [↩]
- A.J. Slooter, M. Cruts, A. Hofman, P.J. Koudstaal, D. van der Kuip, M.A. Ridder, J.C. Witteman, M.M. Breteler, C. Van Broeckhoven, C.M. van Duijn. The impact of APOE on myocardial infarction, stroke, and dementia: the Rotterdam Study. Neurology. Vol 62, pg. 1196-1198, 2004, https://doi.org/10.1212/01.wnl.0000118302.66674.e1. [↩] [↩]
- Z. Breijyeh, R. Karaman. Comprehensive Review on Alzheimer’s Disease: Causes and Treatment. Molecules. Vol. 25, pg. 5789, 2020, https://doi.org/10.3390/molecules25245789. [↩] [↩] [↩] [↩] [↩]
- S. Han, M. Kollmer, D. Markx, S. Claus, P. Walther, M. Fandrich. Amyloid plaque structure and cell surface interactions of beta-amyloid fibrils revealed by electron tomography. Scientific Reports. Vol 7, 43577, 2017, https://doi.org/10.1038/srep43577. [↩] [↩]
- E.D. Vidoni, H.W. Yeh, J.K. Morris, K.L. Newell, A. Alqahtani, N.C. Burns, J.M. Burns, S.A. Billinger. Cerebral beta -amyloid angiopathy is associated with earlier dementia onset in Alzheimer’s Disease. Neurodegenerative Diseases. Vol 16, pg. 218-224, 2016, https://doi.org/10.1159/000441919. [↩] [↩]
- M. Medina, J. Avila. The role of extracellular Tau in the spreading of neurofibrillary pathology. Frontiers in Cellular Neuroscience. Vol 8, pg. 113, 2014, https://doi.org/10.3389/fncel.2014.00113. [↩] [↩]
- H. Braak, I. Alafuzoff, T. Arzberger, H. Kretzschmar, K. Del Tredici. Staging of Alzheimer disease associated neurofibrillary pathology using paraffin sections and immunohistochemistry. Acta Neuropathologica. Vol 112, pg. 389-404, 2006, https://doi.org/10.1007/s00401-006-0127-z. [↩] [↩]
- J. Zhang, Y. Zhang, J. Wang, Y. Xia, J. Zhang, L. Chen. Recent advances in Alzheimer’s disease: Mechanisms, clinical trials and new drug development strategies. Signal Transduction and Targeted Therapy. Vol. 9, pg.211, 2024, https://doi.org/10.1038/s41392-024-01911-3. [↩] [↩] [↩] [↩] [↩]
- M.V.F. Silva, C.M.G. Loures, L.C.V. Alves, L.C. de Souza, K.B.G. Borges, M.D.G. Carvalho. Alzheimer’s disease: risk factors and potentially protective measures. Journal of Biomedical Science. Vol 26, pg. 33, 2019, https://doi.org/10.1186/s12929-019-0524-y. [↩] [↩] [↩]
- E. Drinkwater, C. Davies, T.L. Spires-Jones. Potential neurobiological links between social isolation and Alzheimer’s disease risk. European Journal of Neuroscience. Vol. 56, pg 5397-5412, 2022, https://doi.org/10.1111/ejn.15373. [↩] [↩]
- S. Safiri , A. Ghaffari Jolfayi, A. Faziollahi, S. Morsali , A. Sarkesh, A. Daei Sorkhabi, B. Golabi, R. Aletaha, K. Motlagh Asghari , S. Hamidi, S.E. Mousavi, S. Jamalkhani, N. Karanzad, A. Shamekh, R. Mohammadinasab, MJM Sullman, F. Sahin, A.A. Kolahi. Alzheimer’s disease: a comprehensive review of epidemiology, risk factors, symptoms, diagnosis, management, caregiving, advanced treatments and associated challenges. Frontiers in Medicine. Vol. 11, pg. 1474043, 2024, https://doi.org/10.3389/fmed.2024.1474043. [↩] [↩] [↩] [↩] [↩] [↩]
- J. Stokholm, A. Vogel, A. Gade, G. Waldemar. Heterogeneity in executive impairment in patients with very mild Alzheimer’s disease. Dementia and Geriatric Cognitive Disorders. Vol 22, pg. 54-59, 2006, https://doi.org/10.1159/000093262. [↩] [↩]
- D.G. Harwood, D.L. Sultzer, D. Feil, L. Monserratt, E. Freedman, M.A. Mandelkern. Frontal lobe hypometabolism and impaired insight in Alzheimer disease. The American Journal of Geriatric Psychiatry. Vol 13, pg. 934-941, 2005, https://doi.org/10.1176/appi.ajgp.13.11.934. [↩] [↩]
- E. Vakkila, M. Jehkonen. Apraxia and dementia severity in Alzheimer’s disease: a systematic review. Journal of Clinical and Experimental Neuropsychology. Vol 45, pg. 84-103, 2023, https://doi.org/10.1080/13803395.2023.2199971. [↩] [↩]
- N. Scarmeas, L.S. Honig, H. Choi, J. Cantero, J. Brandt, D. Blacker, M. Albert, J.C. Amatniek, K. Marder, K. Beil, W.A. Hauser, Y. Stern. Seizures in Alzheimer disease:who, when, and how common? Archives of Neurology, Vol 66, pg. 992-997, 2009, https://doi.org/10.1001/archneurol.2009.130 [↩] [↩]
- https://muhc.ca/sites/default/files/micro/m-PT-OT/OT/Mini-Mental-State-Exam-(MMSE).pdf. [↩]
- https://www.flickr.com/photos/institut-douglas/2677257668. [↩]
- Amyloid PET scan.jpeg. Courtesy Dr Aman Deep, Neurologist, Mary Washington Healthcare. [↩]
- S. Bayoumy, I.M.W. Verberk, B. den Dulk, Z. Hussainali, M. Zwan, W.M. van der Flier, N.J. Ashton, H. Zetterberg, K. Blennow, J. Vanbrabant, E. Stoops, E. Vanmechelen, J.L. Dage, C.E. Teunissen. Clinical and analytical comparison of six Simoa assays for plasma P-tau isoforms P-tau 181, P-tau217, and P-tau231. Alzheimer’s Research & Therapy. Vol 13, pg. 198 2021, https://doi.org/10.1186/s13195-021-00939-9. [↩] [↩] [↩]
- S. Palqvist, N. Warmenhoven, F. Anastasi, A. Pilotto, S. Janelidze, P. Tideman, E. Stomrud, N. Mattsson-Carlgren, R. Smith, R. Ossenkoppele, K. Tan, A. Dittrich, I. Skoog, H. Zetterberg, V. Quaresima, C. Tolassi, K. Hoglund, D. Brugnoni, A. Puig-Pijoan, A. Fernandez-Lebrero, J. Contador, A. Padovani, M. Monane, P.B. Verghese, J.B. Braunstein, S. Kern, K. Blennow, N.J. Ashton, M. Suarez-Calvet, O. Hansson. Plasma phospho-tau217 for Alzheimer’s disease diagnosis in primary and secondary care using a fully automated platform. Nature Medicine. Vol 31, pg. 2036-2043, https://doi.org/10.1038/s41591-025-03622-w. [↩] [↩]
- https://www.fda.gov/news-events/press-announcements/fda-clears-first-blood-test-used-diagnosing-alzheimers-disease. [↩]
- D.J. Figdore, H.J. Wiste, J.A. Bornhorst, R.J. Bateman, Y. Li, J. Graff-Radford, D.S Knopman, P. Vemuri, V.J. Lowe, C.R. Jack Jr, R.C. Petersen, A. Algeciras-Schimnich. Performance of the Lumipulse plasma Aβ42/40 and pTau181 immunoassays in the detection of amyloid pathology. Alzheimer’s & Dementia. Vol. 16, pg. E12545, 2024, https://doi.org/10.1002/dad2.12545. [↩] [↩]
- C.R. Jack Jr, D.A. Bennett, K. Blennow, M.C. Carrillo, B. Dunn, S.B. Haeberlein, D.M. Holtzman, W. Jagust, F. Jessen, J. Karlawish, E. Liu, J.L. Molinuevo, T. Montine, C. Phelps, K.P. Rankin, C.C. Rowe, P. Scheltens, E. Siemers, H.M. Snyder, R. Sperling; Contributors. NIA-AA Research Framework: Toward a biological definition of Alzheimer’s disease. Alzheimer’s & Dementia. Vol 14, pg. 535-562, https://doi.org/10.1016/j.jalz.2018.02.018. [↩] [↩]
- G.M. McKhann, D.S. Knopman, H. Chertkow, B.T. Hyman, C.R. Jack Jr, C.H. Kawas, W.E. Klunk, W.J. Koroshetz, J.J. Manly, R. Mayeux, R.C Mohs, J.C. Morris, M.N. Rossor, P. Scheltens, M.C. Carrillo, B. Thies, S. Weintraub, C.H. Phelps. The diagnosis of dementia due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimer’s & Dementia. Vol 7, pg. 263-269, 2011, https://doi.org/10.1016/j.jalz.2011.03.005. [↩] [↩]
- R.A. Sperling, P.S. Aisen, L.A. Beckett, D.A. Bennett, S. Craft, A.M. Fagan, T. Iwatsubo, C.R. Jack Jr, J. Kaye, T.J. Montine, D.C. Park, E.M. Reiman, C.C. Rowe, E. Siemers, Y. Stern, K. Yaffe, M.C. Carrillo, B. Thies, M. Morrison-Bogorod, M.V. Wagster, C.H. Phelps. Toward defining the preclinical stages of Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimer’s &Dementia, Vol 7, pg. 280-92, 2011, https://doi.org/10.1016/j.jalz.2011.03.003. [↩] [↩]
- C. Mei, J. Zhan, S. Zhu, Y. Zhang, C. Xiong, J. Wang, Y.J. Xu, H. Zhong, J. Cheng. Advances of therapy for Alzheimer’s disease: An updated review. Brain-X. Vol. 2, pg. e68, 2024, https://doi.org/10.1002/brx2.68. [↩] [↩] [↩] [↩] [↩] [↩]
- C.J. Swanson, Y. Zhang, S. Dhadda, J. Wang, J. Kaplow, R.Y.K. Lai, L. Lannfelt, H. Bradley, M. Rabe, A. Koyama, L. Reyderman, D.A. Berry, S. Berry, R. Gordon, L.D. Kramer, J.L. Cummings. A randomized, double blind, phase 2b proof-of-concept clinical trial in early Alzheimer’s disease with lecanemab, an anti-Aβ protofibril antibody. Alzheimer’s Research and Therapy. Vol 13, pg. 80, 2021, https://doi.org/10.1186/s13195-021-00813-8. [↩] [↩]
- C.H. van Dyck, C.J. Swanson, P. Aisen, R.J. Bateman, C. Chen, M. Gee, M. Kanekiyo, D. Li, L. Reyderman, S. Cohen, L. Froelich, S. Katayama, M. Sabbagh, B. Vellas, D. Watson, S. Dhadda, M. Irizarry, L.D. Kramer, T. Iwatsubo. Lecanemab in Early Alzheimer’s Disease. The New England Journal of Medicine. Vol 388, pg. 9-21, 2023, https://doi.org/10.1056/nejmoa2212948. [↩] [↩]
- M.A. Mintun, A.C. Lo, C. Duggan Evans, A.M. Wessels, P.A. Ardayfio, S.W. Andersen, S. Shcherbinin, J. Sparks, J.R. Sims, M. Brys, L.G. Apostolova, S.P. Salloway, D.M. Skovronsky. Donanemab in Early Alzheimer’s Disease. The New England Journal of Medicine. Vol 384, pg.1691-1704, 2021, https://doi.org/10.1056/nejmoa2100708. [↩] [↩]
- J.R. Sims, J.A. Zimmer, C.D. Evans, M. Lu, P. Ardayfio, J. Sparks, A.M. Wessels, S. Shcherbinin, H. Wang, E.S. Monkul Nery, E.C. Collins, P. Solomon, S. Salloway, L.G. Apostolova, O. Hansson, C. Ritchie, D.A. Brooks, M. Mintun, D.M. Skovronsky; TRAILBLAZER-ALZ 2 Investigators. Donanemab in Early Symptomatic Alzheimer Disease: The TRAILBLAZER-ALZ 2 Randomized Clinical Trial. Journal of American Medical Association. Vol 330, pg. 512-527, 2023, https://doi.org/10.1001/jama.2023.13239. [↩] [↩]
- H. Hampel, A. Elhage, M. Cho, L.G. Apostolova, J.A.R. Nicoll, A. Atri. Amyloid-related imaging abnormalities (ARIA): radiological, biological and clinical characteristics. Brain. Vol. 146, pg. 4414-4424, 2023, https://doi.org/10.1093/brain/awad188. [↩] [↩]
- P.M. Cogswell, T.J. Andrews, J.A. Barakos, F. Barkhof, S. Bash, M.D. Benayoun, G.C. Chiang, A.M. Franceschi, C.R. Jack Jr, J.J. Pillai, T.Y. Poussaint, C.A. Raji, V.K.Ramanan, J. Tanabe, L. Tenenbaum, C.T. Whitlow, F.F. Yu, G. Zaharchuk, M. Zeinah, T.S. Benzinger, ASNR Alzheimer, ARIA, and Dementia Study Group. Alzheimer Disease Anti-Amyloid Immunotherapies: Imaging Recommendations and Practice Considerations for Monitoring of Amyloid-Related Imaging Abnormalities. American Journal of Neuroradiology. Vol. 46, pg. 24-32, 2025, https://doi.org/10.3174/ajnr.a8469. [↩] [↩]
- ARIA.jpeg. Courtesy Dr Aman Deep, Neurologist, Mary Washington Healthcare. [↩]
- E.B. Garcia-Navarro, A. Buzon-Perez, M. Cabillas-Romero. Effect of music therapy as a non-pharmacological measure applied to Alzheimer’s disease patients: a systematic review. Nursing Reports. Vol. 12, pg. 775-790, 2022, https://doi.org/10.3390/nursrep12040076. [↩] [↩]
- Y. Zeng, J. Wang, X. Cai, X. Zhang, J. Zhang, M. Peng, D. Xiao, H. Ouyang, F. Yan. Effects of physical activity interventions on executive function in older adults with dementia: A meta-analysis of randomized controlled trials. Geriatric Nursing. Vol. 51, pg. 369-377, 2023, https://doi.org/10.1016/j.gerinurse.2023.04.012. [↩] [↩]
- S. Zhang, K. Zhen, Q. Su, Y. Chen, Y. Lv, L. Yu. The effect of aerobic exercise on cognitive function in people with Alzheimer’s Disease: A systematic review and meta-analysis of randomized controlled trials. International Journal of Environmental Research and Public Health. Vol. 19, pg. 15700, 2022, https://doi.org/10.3390/ijerph192315700. [↩] [↩]
- R. Pluta, M. Ulamek-Koziol. Tau Protein-Targeted Therapies in Alzheimer’s Disease: Current state and future perspectives. In:Huang X , editor. Alzheimer’s Disease: Drug Discovery (Internet). Brisbane (AU): Exon Publications; 2020. Chapter 4. [↩] [↩]
- S. Lovestone, M. Boada, B. Dubois, M. Hull, J.O. Rinne, H.J. Huppertz, M. Calero, M.V. Andres, B. Gomez-Carrillo, T. Leon, T. del Ser; ARGO investigators.. A phase II trial of tideglusib in Alzheimer’s disease. Journal of Alzheimer’s Disease. Vol 45, pg. 75-88, 2015, https://doi.org/10.3233/jad-141959. [↩] [↩]
- H.B Nygaard, A.F. Wagner, G.S. Bowen, S.P. Good, M.G. MacAvoy, K.A. Strittmatter, A.C. Kaufman, B.J. Rosenberg, T. Sekine-Konno, P. Varma, K. Chen, A.J. Koleske, E.M. Reiman, S.M. Strittmatter, C.H. van Dyck. A phase Ib multiple ascending dose study of the safety, tolerability, and central nervous system availability of AZD0530 (saracatinib) in Alzheimer’s disease. Alzheimers Research & Therapy. Vol. 7, pg. 35, 2015, https://doi.org/10.1186/s13195-015-0119-0. [↩] [↩]
- P. Bhadane, K. Roul, S. Belemkar, D. Kumar. Immunotherapeutic approaches for Alzheimer’s disease: Exploring active and passive vaccine progress. Brain Research. Vol. 1840, pg.149018, 2024, https://doi.org/10.1016/j.brainres.2024.149018. [↩] [↩]
- H.J. Yu, S.P. Dickson, P.N. Wang, M.J. Chiu, C.C. Huang, C.C. Chang, H. Liu, S.B. Hendrix, J.C. Dodart, A. Verma, C.Y. Wang, J. Cummings. Safety, tolerability, immunogenicity, and efficacy of UB-311 in participants with mild Alzheimer’s disease: a randomised, double-blind, placebo-controlled , phase 2a study. eBioMedicine. Vol. 94, pg. 104665, 2023, https://doi.org/10.1016/j.ebiom.2023.104665. [↩] [↩]
- ClinicalTrials.gov. [↩] [↩]
- M. Pascual-Lucas, A.M. Lacosta, M. Montanes, J. Canudas, J. Loscos, I. Monleon, J.A. Allue, L. Sarasa, N. Fandos, J. Romero, M. Sarasa, M. Torres, D. Whyms D, J. Terencio, G. Pinol-Ripoll, M. Boada. Safety, tolerability, immunogenicity, and efficacy of ABvac40 active immunotherapy against A40 in patients with mild cognitive impairment or very mild Alzheimer’s disease: A randomized, double-blind, placebo-controlled phase 2 study. Alzheimer’s & Dementia. Vol. 21, pg. e70776, 2025, https://doi.org/10.1002/alz.70776. [↩] [↩]