Ria Jain, Solange Massa
Abstract
3D bioprinting is the process of 3D printing a biological structure using bioinks consisting of cells and other biological materials. This innovation can be used in the medical field for different procedures, especially for organ transplants. As of now, transplant patients run the risk of organ rejection, an instance where the body recognizes a donor organ as foreign body and enlists the immune system to treat it as such. Since the donor organ is necessary to the patient, other measures need to be taken to try to stop the immune system from fighting against it. The use of bioinks and specialization of 3D bioprinters can result in less rigorous medical therapies for these patients. 3D bioprinted organs were hypothesized to induce a better response from the immune system than current donor organs do. The conclusion that 3D bioprinting can lead to better lifestyles for organ transplant patients and less rejection by the human immune system was reached after research showing the use of autologous cells and specialized cells in 3D bioprinting. However, further research around 3D bioprinted organs must be conducted in order to make them more available for public medical procedures.
Introduction
Millions of humans need new organs, and even if they are able to receive one, there is always a chance of rejection and further medical treatments for life. A current solution that doctors have been able to derive is through organ transplants. Through surgical processes, physicians can take organs from recently deceased or living patients and transplant it in place of the current, medically harmful one. However, because this new organ is a foreign object to the patient, the patient’s body begins to reject it. The degree of rejection differs with every case, but it occurs nonetheless.13 To combat this issue, 3D bioprinting was developed; 3D bioprinting is a process in which biomaterials including cells are used to 3D print a human biological structure that can function in one’s body system.2 This solution can potentially save millions of lives, but more research to be completed before it can be regularly used in hospitals. As of now, surgical transplants from donors are still the most widely used procedure for organ transplants. Currently, 3D bioprinting has only been used in creating prototypes and simulations of human systems for drug and pharmaceutical testing and other research purposes. This new, potential solution can help to significantly decrease the deficit of organs available for transplants and also help decrease the risk of organ rejection by the body. However, where we are with printing skin and other biological components now is much different than for example printing an entire liver, as that is a much more intricate piece. The idea of achieving the printing of organs is still quite a ways away from where we are with research now.
Immune Response to Transplanted Donor Organs
To explore how 3D printed organs affect the immune response of the human body, it is critical to first delve into how the immune system responds to transplanted organs. Compatibility of a donor organ depends on blood type, height, weight, and other medical factors, which makes it difficult to find a match for patients.9 The immune system recognizes the organ as foreign and starts to fight it. The immune system distinguishes between its own antigens, referred to as “self” antigens, and foreign antigens, referred to as “nonself” antigens. The immune system is wired to take action when it recognizes nonself antigens, which is why when a donor organ, which is made up of nonself antigens, is transplanted, the immune system begins to fight it, referred to as rejection. Doctors try to ensure that the antigens match up as much as they can, but this rejection happens at every transplant.10 The degree to which it happens depends. When this occurs, doctors use immunosuppression therapy to suppress the immune system and try to lessen the degree of rejection.10 Immediate rejection is called acute rejection, and it can lead to chronic rejection, gradual loss of organ function. Immunosuppression stages include induction, maintenance, anti-rejection. This therapy is directed at stopping acute rejection and maximizing long term life. It includes inhibiting the two stages of T-cell activation, the sensitization stage, which includes antigen presenting cells and host T-cells, and the effector stage, the stimulation of costimulatory receptors. The induction stage includes initial suppression, which happens before the transplant occurs. The maintenance stage includes paralysis of the immune system and is aimed at stopping side effects and inducing long term acceptance of the donor organ. Anti-rejection happens last, and it is not included in treatments for all patients. It consists of giving the body heavy steroids.11 There are several different medicines to take, but many are only helpful in stopping acute rejection. Immunosuppression therapy can take a heavy toll on the patient because of the rigorous treatments and medicines that are included. There also lies the fact that the immune system, the body’s way of fighting infections and harmful bacteria, is compromised. 3D bioprinted organs can prove very helpful in lessening the aggressiveness of this type of therapy. However, we must first explore what 3D bioprinting is and the processes behind it.
3D Bioprinting Printers
3D bioprinters need to take into account the arrangement of cells, the extracellular matrix component, and the spacing of the cells in the tissue before actually printing the needed structure. Pre-processing must take place before the structure can be printed. This includes different forms of 3D imaging that would be used to create an image of the tissue, including computed tomography and magnetic resonance imaging.1 The image is divided into two layers and then sent to the printer, which helps create the right environment and ensures that the printer uses the correct materials. Many different types of printers can be used to synthesize a 3D bioprinted organ. Printers can build from the ground up or as separate layers that come together. One type of printer is an inkjet or droplet printer with cells functioning as the bioink. With an inkjet printer, droplets of bioinks are put on a substrate continuously until shape has been formed into tissue.1 It can print directly on wounds such as skin wounds, which can help in medical processes such as skin grafts.1 It is a fast way to produce a 3D bioprinted structure and is more available than others, with 85% cells staying viable. However, it is not as precise as others and the production requires low viscosity bioink.1 A micro-intrusion printer consists of a continuous stream of bioink through a nozzle, which is directed by CAD software in the printer. It can use high viscosity inks and can produce high cell density for tissue. However, it results in low cell viability because of the pressure from the printer, and the structure of tissue could be distorted. A laser-assisted printer consists of a laser pointed through a ribbon with bioink. When parts of the ribbon are vaporized, a high pressure bubble is created which moves specific bubbles to a substrate. This type of printing results in high cell viability and is very precise, but the cost remains very high. Stereolithography printing builds structures through layers by curing the bioink through digital light. This is also a very accurate 3D bioprinting process resulting in very high cell viability, but it includes high intensity UV light and does not use very compatible materials.1

3D Bioprinting Inks
As with regular printers, an ink is necessary to build the structure with. The main component for 3D bioprinters, as stated before, is bioink. It can maintain its shape after being used and is based on polymers such as collagen, gelatin, hyaluronan, silk, alginate, and nanocellulose. They are very frequently used because they can be used for diverse purposes and mimic extracellular matrix environments. The five main types of bioink include structural bioinks, which make up the basic components of the structure, sacrificial bioinks, which make up regions where flow of materials can be present such as voids and tunnels, functional bioinks, which lead the cell behavior towards specific phenotype and can release growth factors to aid in differentiation and other functions of cell, supportive bioinks, which help in creating rigid and supportive structures, and 4D bioinks, which can be changed when stimulated and used in muscle printing in the future.7 The three main bioinks used in 3D bioprinting are hydrogels, natural bioinks, and synthetic bioinks. Hydrogels are composed of hydrophilic polymers held together by covalent bonds or intramolecular interactions. Synthetic bioinks are composed of both synthetic and natural materials and can be differentiated based on what the user wants to create.1 Natural bioinks are made up of all natural resources, the effect of which being that these bioinks mimic the structure of the human body better than synthetic bioinks, but they leave less room for differentiation by the user. This differentiation is key, because it is necessary for the organ to be able to function and adapt to the environment it has been transplanted into. This requires both specialized cells and the ability of the cells in the organ to become specialized, which is why many 3D bioprinters use stem cells when printing structures.3

Comparison Between Immune Response to Transplanted Donor Organs and 3D Bioprinted Organs
Human immune systems do not attack their own cells because they are able to distinguish one’s own cells from foreign bodies, such as antigens. The presence of antigens or foreign bodies is what triggers the immune response from the body. The HLA proteins of a donor organ that are present in the organ will be different from that of the patient, unless they are genetically identical. This difference is what stimulates the immune response in the patient, resulting in rejection.4 The response to 3D bioprinted organs is a little bit different. The immune system willstill recognize the organ as a foreign object and begins to treat it as such. However, when tissue is being created, autologous cells, cells that are derived from the patient’s own body, can be inserted into tissue, which makes the immune system not able to recognize this body as completely foreign. Autologous cells can be procured using biopsies, autologous stem cells, and induced stem cells. As the cells in 3D bioprinted organs have to be able to multiply, 3D bioprinting uses stem cells and induced pluripotent stem cells, which will enable cells in bioinks to continue to multiply.3
Potential of 3D Bioprinted Organs
The main reason that the immune response is triggered by donor organs is because the immune system recognizes the organ as a foreign object. 3D bioprinted organs can be created with stem cells and autologous cells could lead to the patient not experiencing a rejection because the immune system will not recognize the 3D bioprinted organs as different from its own. The potential of this technology includes creating organs out of stem cells and guiding them into shape you want them to be. It also includes being able to differentiate the cells in the 3D bioprinted organs and creating them in a specific orientation that can lead to a replica of the organ one wants to create.6 This can hopefully lead to the avoidance of the issue of rejection that many patients face today.
Practical Application of 3D Bioprinting and 3D Bioprinting of Organs
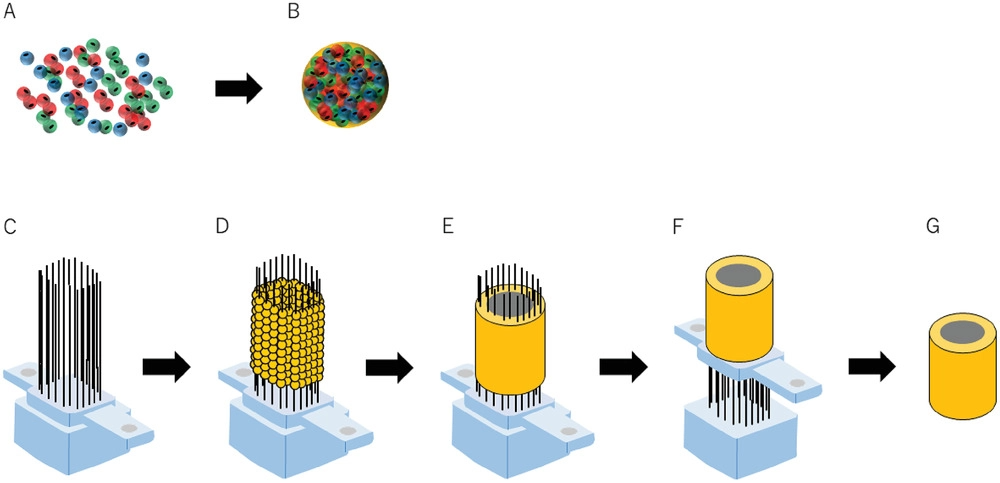
One of the main applications for 3D bioprinting itself is in the pharmaceutical industry. Dr. Solange Massa worked to create a chip that mimicked a cell environment to test certain drugs, using the 3D bioprinter, Organovo.5 Uses of 3D bioprinting itself also include dental and plastic surgery applications, due to the fact that 3D bioprinting is occasionally being used now for printing straight onto a wound or a patient.6 As discussed before, one of the largest practical applications for 3D bioprinted organs is for organ transplants. Medical innovations are helping to prolong and better the lives of millions of patients afflicted with disease, and this technology could help in decreasing the medical need for the hundreds of thousands of patients waiting for an organ. It aids in producing the much needed structures as well as decreasing the amount of therapy necessary after the procedure. Pauline Fenton was a patient at the Belfast City Hospital in Northern Ireland who required a kidney transplant due to complications such as late stages of kidney disease and renal cysts. It was extremely difficult to find her a viable donor organ in time, which resulted in surgeons eventually transplanting a 3D printed model of a viable organ for her body.8 3D bioprinting of organs can help patients like Pauline all over the world through the same processes with more research and testing in the future. While 3D bioprinting of organs is still very much in the research phase, 3D bioprinting has also been used in other ways in the medical field that can be potentially very beneficial to patients, especially in 2020. The Kenzan method has arisen and is the only 3D bioprinting method without the need for additional artificial material. The method includes adding spheroids on an array of microneedles labeled the Kenzan and then allowing them to coalesce; these needles are later removed and the spheroids become an applicable piece of tissue. In the field of heart tissue reconstruction, researchers used the Kenzan method to construct cardiac tissue using human pluripotent stem cells and ventricular cardiac fibroblasts to acquire cardiomyocytes as the spheroids. The graft was observed to contribute to the processes of the heart. Advancements have also been made in the area of peripheral nerve injuries. Researchers used human dermal fibroblasts as spheroids and printed the tubular cell constructs. These structures were placed on the sciatic nerve of a rat and proved to promote nerve reconstruction at the implant site. Lastly, the Kenzan method has been applied to esophageal reconstruction as well. Spheroids derived from human esophageal smooth muscle cells and human dermal fibroblasts were used to create structures implanted into rats, and the subjects were observed to pass food through the new structures without difficulties. While these are all research projects and much remains to be tested, the positive results give hope to these 3D bioprinting procedures as well as organ transplants being used in human patients in the future.14
Potential Societal Effect of 3D Bioprinting and 3D Bioprinted Organs
There remains research to be done before 3D bioprinters can be frequently used. The effects of 3D bioprinters on the environment are very harmful, and the cost to produce them is very high. This technology also raises ethical concerns around what effect this technology can have on society. If 3D bioprinters are able to print viable organs for transplant, it is not short of printing an entire human being. This can lead to a very valuable asset in drug, pharmaceutical, and other research, but it can also lead to disastrous implications for society. 3D bioprinting opens the potential of building enhanced body parts and more cosmetic uses. This could create a new division in social classes based on physical enhancement and how your body functions. As this technology evolves, these potential effects need to be taken into account before moving too far forward.
Conclusion
3D bioprinting of organs can lead to a change in the status quo of how organ transplants and further treatment are conducted. The reproducibility of 3D printed organs and the potential to almost exactly mimic the necessary organ, can help patients all over the world in need of an organ better than donor organs currently provide. However, several years of research still need to occur before this technology can be widespread. Currently, there still remains the thought of whether we will be able to reach printing these complex components of the human body. The size, 200-500 microns, and complexity of the organs make it very difficult to bioprint a viable one as of now.6 We are quite far from reaching this goal and whether we can achieve it also remains a question of “if”. Nevertheless, finding ways to overcome these issues can result in aiding millions of patients in the future.
Conflict of Interest
The author declares no conflict of interest.






