Abstract
Wound healing in cardiovascular disease (CVD) patients is a major problem leading to complex interactions between impaired perfusion, systemic inflammation, and other comorbidities. Conditions like coronary artery disease, peripheral artery disease and heart failure mitigate oxygen and nutrient supply to the wound sites and thus hinder the normal wound healing stages of hemostasis, inflammation, proliferation, and remodelling. Comorbidities that are typically seen with CVD, such as diabetes mellitus, obesity, and hypertension, also delay wound healing due to neuropathy, endothelial dysfunction and chronic inflammation. These comorbidities can later lead to more chronic wounds, such as diabetic foot ulcers and post-surgical wounds, which result in risks of inflammation, infection and amputation. This review integrates evidence of the last decade to explain the mechanisms involved with delayed wound healing in patients with CVD on the systemic, cellular, and molecular levels. Furthermore, this review goes into analyzing currently-used and emerging treatments, these involving biomaterial dressings, revascularization, and immunomodulatory drugs so that cardiovascular care along with wound care is targeted to enhance treatment outcomes in patients. Pointing out the limitations with some of these current treatment methods, this review emphasizes the importance of taking a CVD oriented wound healing treatment process to help improve wound healing in this growing population of CVD patients.
Introduction
Effective wound healing is important to avoid infection, amputation, and loss of function. Patients with cardiovascular disease (CVD), such as coronary artery disease (CAD), peripheral artery disease (PAD), and heart failure (HF), commonly have compromised healing. CVD restricts perfusion of tissues by atherosclerosis, decreased cardiac output, and venous congestion, all of which result in deprivation of oxygen and nutrients for wounds1,2,3. For instance, congestive HF was found to predict delayed healing in diabetic foot ulcers independently4. PAD’s chronic ischemia produces ulcers that tend to stagnate in the absence of revascularization5,6. People with chronic wounds face ongoing pain together with limited movement and work disability and mental health problems that include anxiety and depression7.
The healthcare expenses for chronic wound care in developed countries amount to 2–4% of national healthcare spending because patients require prolonged hospitalization and repeated dressing changes and outpatient visits8. The United States spends more than USD 25 billion annually on chronic wound care but developing nations achieve worse results because they lack access to modern medical treatments9. For PAD, between 2000 and 2015, there was an increase of 58% prevalence in low and middle income countries, with women in these having higher rates10. This compromised healing in CVD patients stems from restricted perfusion due to atherosclerosis, reduced cardiac output, and venous congestion. In fact, those with CVD tend to have lower oxygen supply to wounds, the consequences of which can burden their healing process. The effect was emphasized in a study done by Ichioka et al. which explored the effect of oxygen on wound healing in mice. An experiment was performed where different membranes were used to manipulate the amount of oxygen to a wound on a mouse. There were approximately 20 mice who had the percentage of the original wound area remaining measured after 7 days. In the permeable group, the oxygen tension was regular being 114.1±29.8 mmHg, while in the impermeable group, it was low with a pressure of 5.72±2.99 mmHg. They then observed the healing between the normoxia and hypoxia groups, where they found that in the normoxia group, there was a wound area (11.1±1.66% of the original wound size) which was significantly lower than the wound size of the hypoxia group (27.6±3.47%). This demonstrates how reducing oxygen exposure to wounds can result in delayed wound healing.11. Ultimately, CVD disrupts the typical stages of wound repair: hemostasis, inflammation, proliferation, and remodeling, resulting in the development of persistent, non-healing wounds.
Diabetes mellitus, a common comorbidity of CVD, impairs healing by causing neuropathy, microvascular impairment, and chronic inflammation. Chronic hyperglycemia also inhibits growth factor signaling, angiogenesis, and prolongs the inflammatory stage12. Obesity triggers a state of chronic inflammation that compromises immune function and disrupts the inflammatory phase to tissue regeneration phase transition13. Hypertension and metabolic syndrome further add to vascular stress and endothelial dysfunction, which further compromise microcirculatory support needed for wound repair. All combined, CVD and these comorbidities perpetuate a vicious cycle of compromised perfusion, dysfunctional angiogenesis, neuropathy, and increased infection risk that severely hinders wound healing. This review aims to examine the current understanding of wound healing in patients with cardiovascular disease, drawing on evidence from the past decade. It will explore the underlying pathophysiological mechanisms, analyze the clinical and economic implications, and identify key areas where further research and integrated care models are needed to improve outcomes in this vulnerable patient population. Furthermore, this review will look at both currently used and newer treatments and dressings for chronic wounds.
Methods
To collect data, a literature review strategy was used to find studies which discussed the relationship between cardiovascular disease and impaired wound healing. These studies were collected from the PubMed and Google Scholar databases.
To conduct these searches, keywords relating to the content discussed in this review were used: “cardiovascular disease” OR “peripheral artery disease” OR “heart failure” AND “wound healing” OR “chronic wounds” OR “ischemic ulcers” AND “angiogenesis” OR “hypoxia” OR “inflammation.”
Studies which were published between 2000 and 2025 were considered. To ensure that all studies were relevant to the topic being studied, each was analyzed to ensure that they studied either the mechanisms or the treatments of wound healing in patients with cardiovascular disease.
The titles and abstracts of the papers were screened first to determine if an article was relevant and then the full articles were analyzed after to ensure that the article is fully relevant. Articles were excluded if they were not peer-reviewed or relevant to the content being studied in this review.
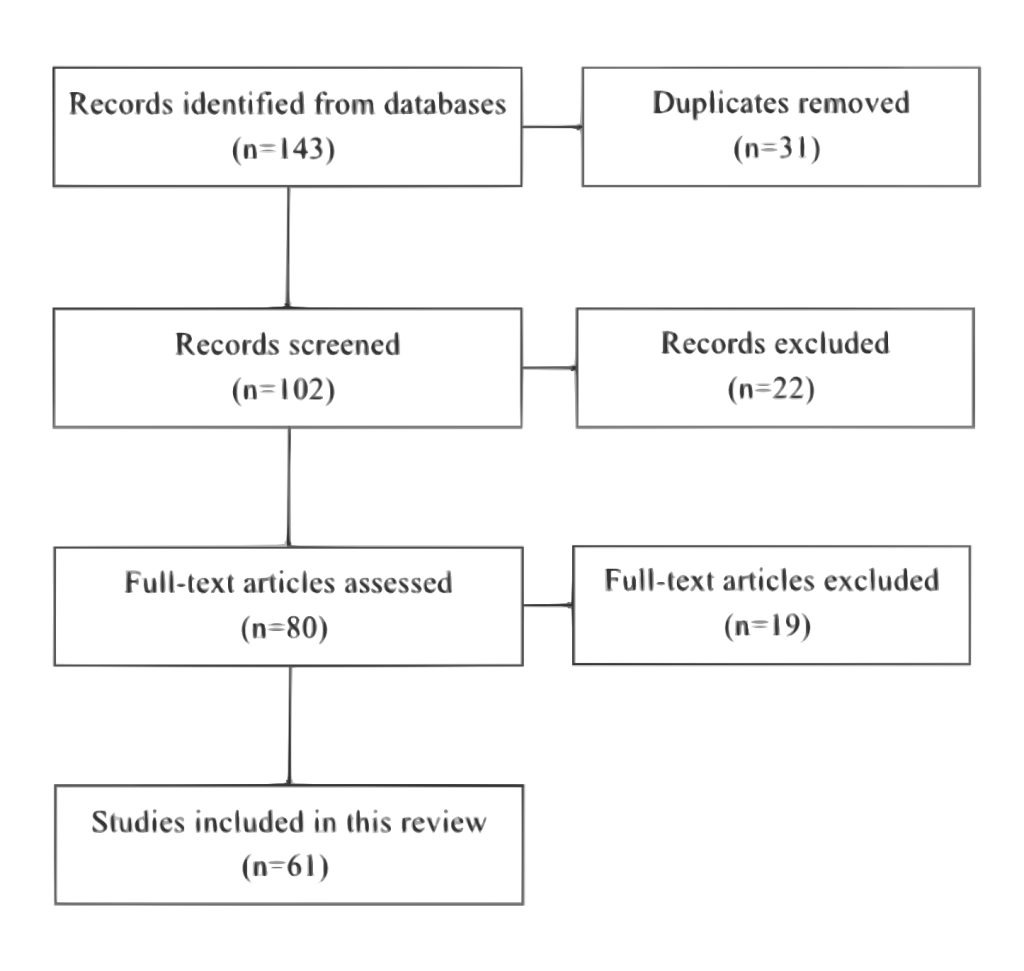
As depicted in figure 1, a total of 143 studies were collected searching through the Pubmed and Google Scholar databases. Among these, 31 studies were removed as they were duplicates of each other. After this, 102 studies were screened based on title and abstract, and a total of 22 studies were removed. Then, 80 full text articles were assessed for relevance to the topic and 19 were excluded. These exclusions resulted in a total of 61 articles being used for this review.
| Author | Design | Sample | Population | CVD | Wound | Findings | Limitations | Level of Evidence |
| Pathophysiology Studies | ||||||||
| Sano et al., 2012 | Animal | ~20 mice | Mouse wound model | Ischemia | Skin wounds | Reducing oxygen tension delayed wound healing | The model was an animal model so it cannot easily to translated to humans | Preclinical |
| Zhao et al., 2021 | Experimental | Animal model | Glucose exposure model | Diabetes effect on vascular function | Cutaneous wounds | Hyperglycemia lead to vascular dysfunction which leads to delayed wound healing | Mechanistic model | Preclinical |
| Malone et al., 2017 | Clinical study | 17 patients | Chronic diabetic foot ulcers | Diabetes and PAD | Chronic diabetic foot ulcers | Biofilms led to a greater microbial conditions resulting in wound healing being delayed | Small sample size | Moderate |
| Clinical Outcome Studies | ||||||||
| Nishio et al., 2022 | Observational study | 118 patients | Patients who suffer from heel pressure ulcers | PAD | Pressure ulcers | PAD lead to decreased rate of wound healing when compared to patients without PAD | Single site for study | Moderate |
| Hafner et al., 2000 | Clinical observations | 61 patients | Patients suffering from arterial leg ulcers | PAD | Arterial leg ulcers | Ischemic conditions lead to delayed wound healing | Older dataset | Moderate |
| Rhou et al., 2015 | Clinical cohort | 314 patients | Patients with diabetic foot ulcers | Congestive heart failure | Diabetic foot ulcers | Those with congestive heart failure took longer times for their wounds to heal | Observational design | Moderate |
| Ahmed et al., 2011 | Clinical cohort | 121 patients | Patients having a hip arthroplasty | Hypertension | Surgical wounds | Patients with hypertension exhibited longer healing times compared to those who were normotensive | Focused only on surgical wounds | Moderate |
| Treatment Studies | ||||||||
| Hendri et al., 2023 | Clinical comparative study | 46 patients | Patients suffering from diabetic foot ulcers and also PAD | PAD | Diabetic foot ulcers | Patients who underwent revascularization had faster healing rates compared to those who did not undergo revascularization | Small sample size | Moderate |
| Desai et al., 2021 | Meta-analysis | Multiple studies | Patients who were going through revascularization | PAD | Ischemic limb wounds | After revascularization, the use of cilostazol therapy improved the rates of limb salvation | Study heterogeneity | High |
| Li et al., 2022 | Animal study | Rodent model | MRSA infected wounds | Chronic wounds related to infection | Skin wounds | Using antioxidant hydrogel led to increased antibacterial activities and also improved wound healing rates | Preclinical model | Preclinical |
Wound healing
The skin is the body’s largest organ, covering about 1.85 m² and making up roughly 15% of total body weight. The body uses this system as a protective shield that detects and controls the internal environment. The skin structure contains three primary sections which include the epidermis, dermis, and hypodermis (subcutaneous tissue).
Epidermis
It is the layer of skin that is outermost and serves as the primary protection against pathogens and UV radiation and chemical and mechanical harm.
Stratum basale (basal layer) is the deepest skin tissue that supports continuous cell division for keratinocyte production and contains melanocytes which create skin pigmentation and Merkel cells which sense touch. The Stratum spinosum (spiny layer) consists of keratinocytes that bond through desmosomes to create a protective structure which provides strength and elasticity and guards against friction. Stratum granulosum (granular layer) contains cells which start to die while accumulating keratin and lipids to create a waterproof barrier that prevents fluid loss. Stratum corneum (horny layer) – the outermost layer of flattened, dead keratinized cells that are continuously shed and replaced, forming a tough, protective surface.
Dermis
Located beneath the epidermis, the dermis makes up about 90% of the skin’s weight. It contains collagen and elastin fibers that give skin its strength and flexibility. The dermis contains hair follicles as well as sweat and oil glands and blood vessels and lymphatics and sensory nerves which sense touch and temperature and pain. Its rich blood supply delivers nutrients and oxygen to both the dermis and the avascular epidermis.
Hypodermis
The deepest layer of skin tissue consists of fat cells and loose connective tissue which forms the hypodermis. The body fat functions as an energy reservoir while providing insulation and shock absorption to the body. Large blood vessels in this layer branch upward to feed the dermis. The extent of skin damage from injuries determines wound severity based on the depth of injury14.
Stages of wound healing
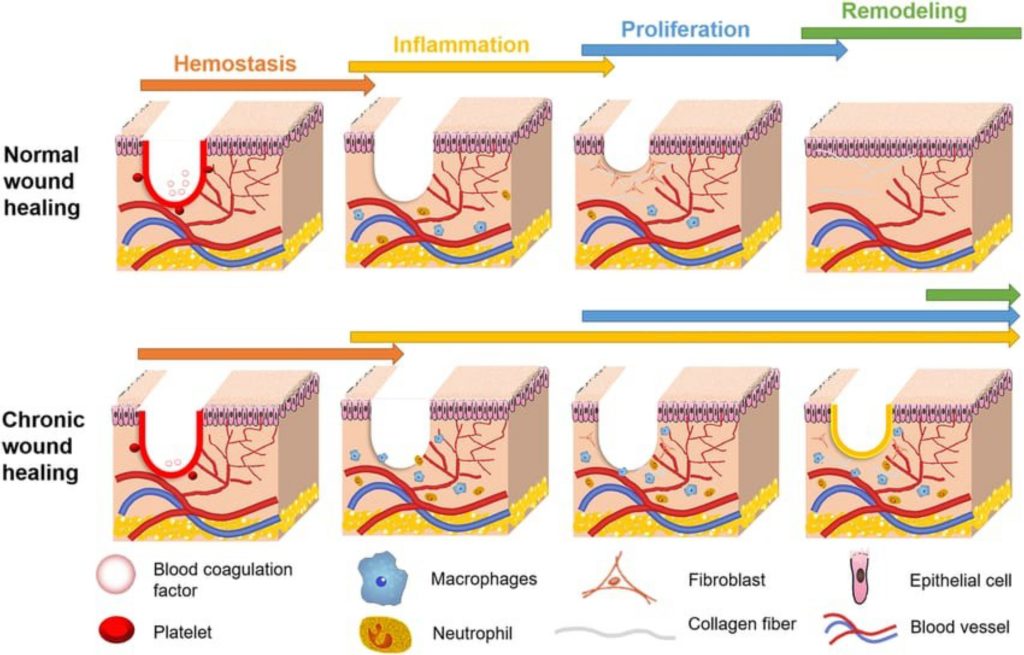
Once the integrity of the skin is compromised, a highly coordinated series of biological events is initiated to restore tissue homeostasis. The healing process consists of multiple stages which follow each other in a specific order of cellular and molecular events.
Hemostasis
Immediately after injury, the body works to stop bleeding. The damaged vessels become narrower while platelets start to activate and aggregate into a blood clot. This clot acts as both a barrier and a temporary scaffold, made mainly of fibrin and fibronectin. It also traps signalling molecules that attract immune cells to the wound site.
Inflammation
The wound attracts neutrophils which arrive within hours to perform phagocytosis and produce reactive oxygen species for clearing pathogens and dead cells and debris. As neutrophils decline, monocytes arrive and become macrophages, which continue cleaning and releasing growth factors like vascular endothelial growth factor (VEGF) and transforming growth factor beta (TGF-β) to promote healing.
Proliferation
The wound attracts fibroblasts which migrate to the site to produce collagen type III and multiple matrix proteins that create granulation tissue. Keratinocytes migrate to the edges of the epidermis to rebuild layers and angiogenesis creates new blood vessels which follow growth factor concentration gradients. The return of oxygen and nutrients results in higher cell metabolic activity and matrix production. Regulatory T cells serve as a system to control inflammation while supporting the natural healing of tissues.
Remodelling
The tissue becomes stronger after granulation and re-epithelialization finishes because collagen III transforms into collagen I. The wound edges contract through myofibroblast activity which decreases the wound size and results in less scarring. The normal blood flow leads to the disappearance of new capillaries which results in scar tissue that maintains 80% of normal skin strength but becomes less flexible15,16. The standard sequence of wound healing exists but different elements can cause interruptions or prolongations of these stages. Chronic wounds develop from these disturbances because they create wounds with special pathophysiological characteristics which need personalized clinical care. The following section outlines the key distinctions between these two types of wounds.
Chronic Wounds
Acute and chronic wounds heal at different rates because they need distinct treatment approaches and healing times. The healing process for acute wounds including surgical cuts and abrasions and burns and chemical injuries takes 8 to 12 weeks9. The healing process unfolds in a specific order which includes bleeding control followed by inflammation and then tissue formation and finally remodeling. Chronic wounds do not follow the typical healing stages because they remain stuck in one phase for extended periods of time. The tissues stay in an extended inflammatory state because leukocytes stay active and cytokines and ROS levels stay elevated and enzymes continue to degrade tissue at a rate that exceeds the body’s repair capabilities. The poor function of fibroblasts and keratinocytes occurs with insufficient angiogenesis and alkaline pH which creates an environment that supports bacterial growth including antibiotic-resistant biofilms. Biofilms appear frequently in chronic wounds, but they are not commonly found in acute wounds. The main causes of chronic wounds include diabetes and poor circulation as well as pressure and infection and radiation and autoimmune disorders. The treatment for these wounds focuses on these core problems by enhancing blood circulation and managing blood sugar levels and removing dead tissue and biofilm deposits. The management of chronic wounds results in major financial and social challenges because it requires ongoing dressing treatments and medical appointments and limits patient mobility at the same time as creating substantial healthcare expenses17.
Infection, Biofilms, and Microbiome Immune Interactions
The formation of bacterial biofilms along with microbe colonization can also severely impact chronic wounds. In chronic wounds, because of the conditions which allow for stable microbial populations to develop, extracellular matrices called biofilms are able to form over wounds. These biofilms further infection, as they can protect the microbial communities from immune responses. Furthermore, they can also impair the ability of antibiotic therapies.
With biofilms, the immune environment for that wound is also impaired, as the prolonged duration of the presence of bacteria can lead to the stimulation of neutrophils and macrophages. With this prolonged immune response, a pro-inflammatory environment is created in the wound, damaging tissue in the area, and preventing the wound from leaving the inflammatory stage of its healing. For diabetic foot ulcers, clinical studies have shown how biofilm formation in these wounds can limit wound healing processes.
These findings highlight the importance of implementing strategies to disrupt biofilms, in order to remove the prolonged inflammatory and infectious conditions they can set in wounds, and also improve the quality of wound healing in patients with CVD.
Pathogenesis of impaired wound healing in diabetes:
Diabetes stands as a leading cause of prolonged wound healing problems which primarily affect diabetic foot ulcers (DFUs) in patients. The body fails to heal diabetic wounds properly because blood sugar levels remain elevated which serves as the main reason for this problem18,19.
Chronic hyperglycemia can have various impacts: The body creates advanced glycation end-products (AGEs) through elevated blood glucose which causes collagen cross-linking that makes tissues stiff and prevents their natural remodeling process. The binding of AGEs to RAGE receptors on macrophages and endothelial cells triggers continuous generation of interleukin-6 (IL-6) and interleukin-1 beta (IL-1β) and tumor necrosis factor alpha (TNF-α) which are cytokines that extend the inflammatory response20. The development of diabetic neuropathy causes patients to lose their pain sensation which leads to unperceived repetitive injuries that produce pressure sores that eventually develop into ulcers21. The insufficient delivery of oxygen and nutrients because of peripheral arterial disease and microvascular dysfunction causes impaired leukocyte function and defective angiogenesis through reduced VEGF and HIF-1α signaling12. The immune system fails to control biofilm growth because neutrophil extracellular traps (NETs) form in excess but fail to function correctly and macrophages stay in a prolonged M1 inflammatory state instead of transitioning to the M2 anti-inflammatory state22.
The combination of these factors creates a self-sustaining inflammatory response in diabetic wounds which produces high levels of ROS and proteases and proinflammatory cytokines while making it impossible for the body to enter the productive phases of wound healing23.
Cardiovascular disease (CVD)
Cardiovascular disease (CVD) consists of a group of diseases which impact the functioning of the heart and blood vessels. There are different types of CVDs such as heart failures, stroke, coronary artery disease, peripheral artery disease (PAD), and arrhythmias. While atherosclerosis contributes to many cardiovascular diseases, there are other mechanisms which also contribute to cardiovascular diseases. Some of these include reduced cardiac output or irregularities in electrical conductivity. The progression of CVD induces systemic alterations, including chronic inflammation, oxidative stress and neurohormonal activation. Endothelial function is impaired by these modifications, which also impede the release of angiogenic factors such as platelet-derived growth factor (PDGF) and VEGF. In addition, patients with cardiovascular disease have hyperactivated inflammatory cells, which contributes to the extension of lesion inflammation24. Progressive CVD causes a decrease in perfusion pressure and an increase in circulating vasoconstrictors, such as angiotensin II, which further impedes microcirculation. Clinical research supports the notion that decreased perfusion impedes the proliferative healing phase, as it has been demonstrated that comorbid congestive heart failure (CHF) is associated with significantly delayed wound closure in diabetic foot ulcers. Inflammation has a key role in the steps involved with atherosclerosis and atherothrombosis, forms of cardiovascular disease25. ECD and the accumulation of LDLs in the arterial intima commence a subintimal inflammatory response. This causes adhesion molecules to aid monocytes and T helper cells to attach to early plaque initiation sites. The monocytes then become macrophages that form the Nod-like receptor protein 3 (NLRP3) inside them. The formation of this inflammasome results in the production of IL-1B from pro-IL-1B and interleukin-18 (IL-18) from pro-IL-18 which in turn activate multiple different inflammatory cells and also create IL-6 which helps produce CRP that increases the inflammatory cascade. Additionally, there are a variety of other inflammatory mechanisms that help further develop and increase progression of the atherosclerotic plaque. These include the increased amounts of interleukins, cytokines, and ROSs, and these also include inflammatory cells such as mast cells, and dendritic cells, and T-cells, which increase production and signaling of cytokines which regulate the progression and formation of plaque. Ultimately, foam cells lead to the creation of a necrotic lipid core.26
Endothelial dysfunction and atherosclerosis
Atherosclerosis, which is characterized by lipid-rich plaques in arteries, results in chronic endothelial dysfunction and compromised tissue perfusion. In a healthy state, the endothelium maintains blood fluidity and vasodilation; however, in atherosclerosis, it becomes prothrombotic and vasoconstrictive, depriving the healing tissues of oxygen and growth factors. Dysfunctional angiogenesis is a defining characteristic of both chronic wounds and atherosclerosis, and it is the primary cause of poor healing outcomes12.
Thrombosis that results from the rupture of atherosclerotic plaques may further compromise perfusion. Furthermore, endothelial injury induces an increase in oxidative stress and the release of inflammatory cytokines, which impedes the migration of keratinocytes and fibroblasts. In severe peripheral vascular disease, acute lesions develop into chronic, non-healing ulcers as a result of chronic ischemia. Consequently, advanced atherosclerosis impedes wound healing by disrupting the wound-healing cascade at multiple levels, such as angiogenesis and hemostasis12.
Tissue Repair, inflammation and ischemia in myocardial infarction
Myocardial infarction (MI) induces a wound-healing response in the heart. Chalise et al. (2023) assert that MI initiates a two-stage healing process: a reparative phase during which macrophages promote fibroblast proliferation and scar formation, and an initial inflammatory phase characterized by neutrophil and monocyte infiltration27.
Nevertheless, the quality of healing following MI is contingent upon the preservation of a balance between matrix synthesis and degradation. Inadequate fibrosis leads to dilatation, whereas excessive inflammation can result in wall thinning and perforation. The faster healing of skin wounds was significantly correlated with the enhanced post-MI survival of mice, indicating that all tissues possess a systemic healing competence27.
Reduced angiogenesis and peripheral arterial diseases
Peripheral arterial disease (PAD) results in chronic hypoxia, reduced limb perfusion, and delayed wound healing. In observational cohort studies, it has been observed that 50–65% of patients with critical limb ischemia (CLI) in populations of those undergoing vascular surgery die within five years. 20–30% have their extremities amputated within a year in cases when the wounds are non healable or where revascularization cannot occur, as per Iyer et al. (2017)28. These findings demonstrate that, despite the implementation of sophisticated wound care, it is impossible to reestablish adequate perfusion. However, ulcer healing and amputation-free survival have not been significantly improved in therapeutic angiogenesis trials that utilize VEGF, FGF, or stem cells28. Perfusion imaging has been enhanced in these trials. These results indicate that PAD suppresses the downstream response necessary for the formation of collateral vessels, even in the presence of elevated angiogenic signals. Matrix metalloproteinases (MMPs) are important molecules for the extracellular matrix (ECM) because of their power to degrade many of the parts of the ECM, a process important for wound healing. To regulate the activity of these MMPs, there are tissue inhibitors of MMPs (TIMPs) which inhibit the ability of MMPs to degrade proteins in the ECM. In patients with diabetes mellitus (DM), due to hyperglycemia, there is an increase in oxidative stress which in turn induces the activity of MMP-9 harming the ECM and vascular system. TIMPs trying to regulate these MMPs can try to ameliorate the situation29.
Systemic consequences of heart failure on wound healing
Heart failure (HF) induces a state of elevated inflammation and diminished flow. The delivery of oxygen to the peripheral tissues is restricted by a reduction in cardiac output, and the process of healing is impeded by the increased interstitial pressure that results from venous congestion. Increased cytokine levels (e.g., TNF-α, IL-6) in HF patients impede regeneration by promoting catabolism and inhibiting the growth of new blood vessels25,30. Clinical audits indicate that diabetic foot ulcers in patients with heart failure resolve at a slower pace. Tissue regeneration may be directly influenced by compromised perfusion and systemic inflammation, as HF was a predictor of non-healing on its own. In a study performed by Rhou et al., it was observed that among patients with Type 1 or 2 Diabetes who had diabetic foot ulcers (DFUs), 95 out of the 107 patients healed overall. However, it was also found that among the rest who had problems during the healing process such as failure of healing, delayed healing at 12 weeks, or reduced healing rates, these effects were independently predicted by comorbid congestive heart failure25.
Hypertension-induced vascular damage and repair limitations
Hypertension impairs microcirculation by increasing blood vessel stiffness and decreasing capillary density. Ahmed et al. (2011) found that patients with hypertension experienced significantly prolonged wound drainage (3.79 days compared to 2.03 days for normotensives) in their analysis of 120 hip replacement patients31. The impact of hypertension on the phases of hemostasis and proliferation is underscored by the nearly twofold delay in wound closure.
In hypertensive patients, vascular stiffening reduces endothelial responsiveness and nitric oxide availability, both of which are essential for angiogenesis and the formation of granulation tissue31.
Diabetic cardiomyopathy and delayed wound healing
Chronic inflammation, immunological dysfunction, and vascular insufficiency are all exacerbated by diabetes, which impedes the healing process of wounds. Approximately 15–25% of diabetic patients develop foot ulcers, and 14–24% of these ulcers necessitate amputation. Amputation results in the death of more than 50% of individuals within five years21. Hyperglycemia results in the glycation of vessel walls, impaired angiogenesis, and endothelial cell dysfunction. In diabetic wounds, macrophages fail to transition to a reparative phenotype, which leads to protracted inflammation and diminished capillary formation12,24. In a study, the effect of hyperglycemia and metabolic memory (MM) on the wound healing duration of mice, it was found that hyperglycemia and MM increased the healing time for wounds significantly. The experiment had three groups, a control group, a group which contained mice with induced diabetes, and a group of mice with metabolic memory. In the healing process, on the 3rd day, the wound closure rate for the control group was 30% while it was less than 15% for both the diabetic and MM group. On the 7th day, for the control group the closure rate was 60%, while it was 15% and 17% for the diabetic and MM groups, respectively. By the 14th day, the control group had completely reached closure, while the diabetic group and MM groups still had 41% and 37% unhealed wound areas, respectively32.
Wound healing necessitates a delicate equilibrium between fibrinolysis and thrombosis. TGF-β and PDGF are released by platelets during the formation of a thrombus, which initiates the healing process. The fibrin clot is disintegrated by plasmin-mediated fibrinolysis, which enables cell migration and angiogenesis. Impaired fibrinolysis can result in fibrin persistence, which can delay healing33. Inflammation and protracted clot retention are facilitated by elevated plasminogen activator inhibitor 1 (PAI-1) levels in CVD, which inhibit fibrinolysis. This is especially evident in diabetic wounds, where hypercoagulability results in microthrombi in the wound bed, which exacerbates chronic, non-healing ulcers33.
Cardiovascular diseases significantly impede wound healing. These disorders, which encompass a wide variety of conditions, including atherosclerosis, MI, PAD, and diabetic cardiomyopathy, disrupt the equilibrium between coagulation and fibrinolysis, delay the resolution of inflammation, and impair angiogenesis. The practical implications of these conditions are illustrated by clinical study data, such as the high amputation rates in CLI and the extended wound healing periods in hypertensive patients. The effective therapy of non-healing wounds necessitates an integrated strategy that addresses systemic vascular and metabolic dysfunctions.
Reasons for delayed wound-healing mechanisms in CVD patients
Endothelial dysfunction, nitric oxide and hypoxia
One of the first factors that was observed to lead to endothelial dysfunction was abnormalities in the production of vasoactive peptides (molecules that impact blood flow, blood vessel diameter, and blood pressure) such as nitric oxide (NO). NO is critical as it functions as a vasodilator. Inhibition of the process which metabolizes L-arginine into NO can potentially yield negative results: the decrease of NO causes atherogenesis which can lead to atherosclerosis. This process is exacerbated through endothelial dysfunction as it allows for low-density lipoproteins (LDL) to increase in the arterial intima34,35. Endothelial dysfunction can also bring in oxidative stress. Oxidative stress occurs when there are imbalances between reactive oxygen species (ROS) and antioxidants. For example, when endothelial dysfunction allows for the increase of LDL, they become oxidized by ROS which leads to the endothelial cells calling inflammatory leukocytes which increase pro-inflammatory cytokines and interleukins which increase matrix and lipid deposition. These all together eventually lead to the development of atherosclerosis as atherosclerotic plaque begins to develop36. NO is an important molecule for the cardiovascular system due to vasodilating properties it has. One of the main enzymes which helps synthesize it in the vascular endothelium is the endothelial nitric oxide synthase (eNOS). This enzyme has the role of catalyzing the reaction of L-arginine to L-citrulline. This reaction also produces NO with O2 and nicotinamide adenine dinucleotide phosphate (NADPH). The proper functioning of eNOS is very crucial as its product NO signals vasodilation. Its importance is even further emphasized by the other various benefits from NO. NO derived from platelets can have paracrine and autocrine inhibitory effects which decrease clot formation. NO can also limit the amount of leukocytes going to the endothelium, decreasing chances of endothelial dysfunction37. With all these important jobs that NO holds in the vascular endothelium, the proper functioning of eNOS is necessary to decrease the chances of cardiovascular disease, such as endothelial dysfunction.
The vascular endothelial growth factor (VEGF) and fibroblast growth factor (FGF) are essential molecules for the process of angiogenesis as they are the ones who influence endothelial cells in this process. They normally function by inducing the endothelial cells to release proteases and plasminogen activators. These in turn help degrade the surroundings to allow endothelial cells to migrate and differentiate and form new blood vessels. This process, however, is very delicate, and changes in the functions of VEGF and FGF can cause detrimental effects for angiogenesis. When there is disruption of the genes of either molecule, the functions of the molecules tend to change and this causes irregular behaviors in the molecules. These behaviors can then negatively impact the process of angiogenesis negatively as they would not be able to fully regulate angiogenesis. This flaw ends up failing angiogenesis as endothelial cells would not be able to differentiate, migrate, or even proliferate along with a variety of other effects38. The vascular endothelial growth factor (VEGF) is another important molecule involved with the cardiovascular system which helps primarily with the movement of endothelial cells during angiogenesis. There is also an unclear relationship between VEGF and atherogenesis, with lots of understanding that it is involved with atherogenesis yet little understanding on how it influences it. VEGF could also potentially have a connection with other cardiovascular diseases as well such as MI and CAD. It was found in these diseases and other acute coronary syndromes, the concentrations of VEGF varied suggesting that it is involved. Furthermore, genetic polymorphisms of VEGF can increase the susceptibility of cardiovascular disease39.
At the beginning of the inflammatory response process, the site of the wound is often hypoxic. This happens due to the disruption of the vasoculture near the wound and also the increased inflammatory cells from the inflammatory process using up lots of the oxygen, leaving less for the wound. When not enough oxygen is present, it can lead to a decreased production of the extracellular matrix (ECM) which is seen in a lot of chronic wounds as the process which is responsible for this, fibroblast production of collagen, depends on oxygen.
The hypoxia-inducible factor 1 (HIF-1) is another important molecule due to its ability to control and regulate oxygen homeostasis, and healing ischemic wounds. This molecule is typically induced in hypoxic conditions. In studies done on mice, it was found how with HIF-1 alpha, there was more blood flow following injury allowing the mice to heal better with an ischemic wound. However, using HIF-1 alpha was more successful in the younger mice implying that for the case of humans, humans that are older may not as benefitted from this as much as younger humans. However, this does not diminish the value of the positive effect that HIF-1 alpha has on reducing ischemic heart disease (IHD). This could imply that the dysregulation of this molecule could lead to worsening of IHD and eventual tissue loss40. Reactive oxygen species (ROS) are molecules that can cause some serious problems to the cardiovascular system. These problems include MI, atherosclerosis, diabetes, cell damage, and necrosis and cell apoptosis. ROS can also play a major role in cardiac hypertrophy, where they can either directly or indirectly activate signaling pathways such as mitogen activated kinases (MAP kinases), protein kinase C (PKC), nuclear factor kappa B (NFkB), calcineurin, and tyrosine kinases, ultimately leading to the possible result of cardiac hypertrophy.
ROS can also have an impact on cardiac fibrosis. During the increased expression of procollagen I and III, there is increased NADPH oxidase activation which produces more and more ROS. This can have a negative impact as these ROS can indirectly affect the cardiac contractility and it can also directly affect the contractile system of the heart itself41.
Extracellular matrix remodeling and MMP dysregulation
The extracellular matrix (ECM) is an intricate arrangement of proteins which all together form the environment outside the cells. The ECM maintains tissue homeostasis by remodeling over and over again. The ECM can also have major impacts on the cardiovascular system as many problems with this system can be associated with changes in the ECM. For instance, during myocardial infarction, fibronectin, a glycoprotein, has unregulated expression42. Matrix metalloproteinases (MMPs) are proteolytic enzymes that help degrade the myocardial extracellular proteins and play a big role in ECM remodeling. These enzymes can have negative impacts as well, however, as its dysregulation due to excessive exposure can lead to diseases such as plaque rupture, tumor angiogenesis and metastasis, and vascular neointimal hyperplasia, potentially causing other major cardiovascular diseases, such as myocardial infarction or a stroke. Furthermore, increased MMP exposure and particularly powerful MMPs that can break up lots of the ECM are found in those with end-stage CHF. For example, while interstitial collagenase MMP-13 is supposed to be very lightly expressed, in those with end-stage CHF, its expression is much larger than appropriate43.
TGF-beta and fibrosis in impaired regeneration
Fibrosis is a condition in which there is a large accumulation of fibrous tissue and is normally seen in those with chronic inflammation. TGF-beta is a profibrotic cytokine which helps wound healing and also decreases inflammation. However, increases in the levels of this cytokine is correlated with fibrosis-related diseases and treatment with this can induce fibrosis. Based on this relationship, by decreasing the concentration of TGF-beta, fibrosis can be reduced and the inflammation in the wound would not be exacerbated44. Fibrosis can also have the impact of impairing angiogenesis. Fibrosis causes tissue hypoxia in these areas which in turn leads to the stimulation of angiogenesis. However, due to the angiogenesis being inefficient as it cannot keep up, it causes more harm and exacerbates the fibrosis45. Linking this to TGF-beta could show that with increased TGF-beta, there can be induction of fibrosis which can lead to inefficient and impaired angiogenesis.
Macrophage polarization imbalance in wound resolution
Macrophages are important to the cardiovascular system as they play an important role in wound healing and the ending of inflammation. There are two main types of macrophages; M1 and M2 with proinflammatory and anti-inflammatory wound healing properties, respectively. Wound healing greatly benefits from the proper expression of type of macrophages. In the early stages of healing, M1 macrophages are utilized while later one the M2 macrophages are used when there is development of new tissue. In a study done on mice, it was found that wounds without macrophages exhibited slowed and reduced healing, further emphasizing the importance of macrophages. Specifically, the decrease in macrophages in these wounds also inhibited angiogenesis in these wounds46.
The platelet-derived growth factor (PDGF) is a molecule that is involved in a variety of things, such as tissue fibrosis, atherosclerosis, and tumor growth. PDGFs are molecules that are involved in a variety of processes such as cell growth, migration, and cell division. This factor can have an impact on cardiovascular disease as well, however. In a study conducted to see the impact of PDGF-D on the cardiovascular system, mouse hearts produced extra PDGF-D and then were observed. This led to the buildup of interstitial fibroblasts which led to the increase of collagen. This enlarged the heart and led to heart failure in the mice. This study had similar results as the effects of increased PDGF-D in humans which are cardiac hypertrophy and dilated cardiomyopathy47.
Pro-inflammatory cytokines: IL-1 beta, IL-6 and TNF-alpha
Cytokines are important molecules in our bodies that are used in an autocrine, paracrine, or endocrine factor. They can also act in either pro-inflammatory or anti-inflammatory manners. Pro-inflammatory cytokines can actually have an impact on cardiovascular disease in a person. When looking at atherosclerosis, we can see the impact of pro-inflammatory cytokines such as IL-6 as there are normally lots of mRNA transcripts and proteins in the atherosclerotic plaques. In fact, the presence of IL-6 could also increase the presence of other pro-inflammatory cytokines further impacting these pro-inflammatory cytokines effects.
Thrombosis can also occur due to the increased presence of these proinflammatory cytokines. When looking at a study involving baboons, it was shown how TNF-alpha started the coagulation and fibrinolysis, and TNF-alpha and IL-1 were found to have activated the endothelial cells which in turn shifted into a pro-thrombotic state. Additionally, there was also increased risk of stroke in those with higher IL-6 levels and increased plasma viscosity48.
Due to the aforementioned factors, wound care is delayed in CVD patients; if appropriate treatment and care are not given, chronic wounds may develop and may even be fatal.
Treatments
Humans have known since ancient times that wounds require coverings in order to be treated and healed. Wound dressing technology has evolved from using basic natural materials to creating advanced biomedical engineering solutions. Wound coverage before the 18th century relied on natural materials including animal skin, honey and plant fibers. The materials used for treating wounds frequently trapped dust particles and bacteria which caused inflammation that extended the healing process49. Despite medical advancements, wound healing remains a persistent clinical challenge, particularly in chronic wounds strongly associated with cardiovascular disease. The World Health Organization states that millions of people worldwide suffer from chronic wounds which generate major social and economic problems because of their prolonged treatment duration and limited mobility. The physical damage from chronic wounds leads to major social problems and economic burdens. Research shows that 2–6% of global population develops chronic wounds at any time, yet the numbers keep increasing because of aging populations and rising diabetes cases50. People with chronic wounds face ongoing pain together with limited movement and work disability and mental health problems that include anxiety and depression. Healthcare systems face major economic challenges when managing chronic wounds according to economic analysis. The healthcare expenses for chronic wound care in developed countries amount to 2–4% of national healthcare spending because patients require prolonged hospitalization and repeated dressing changes and outpatient visits8. The United States spends more than USD 25 billion annually on chronic wound care but developing nations achieve worse results because they lack access to modern medical treatment9. The worldwide diabetic population exceeds 550 million people while diabetic foot ulcers develop in 18.6 million patients annually. The treatment of diabetic foot ulcers leads to lower-limb amputation for 20% of patients who receive this procedure. Even after treatment, diabetic foot ulcers have a 42% recurrence rate during the first year after healing and patients face a 30.5% risk of death within five years of the amputation. The growing number of people with diabetes and obesity and aging population has resulted in a substantial increase of chronic wound cases which now represents a significant public health challenge. The primary contributing element is the recurrent infection of the local wound area, resulting in biofilms of bacteria that inhibit the development of granulation tissue and hinder wound healing17.
These socio-economic challenges emphasize the critical need for innovative and cost-effective therapeutic strategies that accelerate wound closure, prevent infection, and restore normal skin function. Achieving these outcomes requires a comprehensive understanding of skin anatomy and physiology, as the skin serves as the primary barrier protecting the body from external insults and plays a central role in tissue regeneration.
Although there have been various pathways which can be focused on to improve wound healing in patients struggling with CVD, there have been challenges with clinical implementation. When considering treatments such as therapeutic angiogenesis, promising results were demonstrated in preclinical models however there has been limited success in clinical implementations. These findings show that while understanding the mechanisms provides possibilities of treatments, clinical studies need to be performed to understand how these treatments affect real populations of people.
The treatments discussed in this section focus on attacking the mechanisms explained earlier in this review. These mechanisms include ischemia, inflammation, reduced angiogenesis, and also microbial environments. By combating these mechanisms, these treatments try to improve the quality of wound healing in patients with CVD.
Revascularization
Revascularization is a widely used method to help treat tissues experiencing ischemia. This process often involves recovering blood flow to these tissues to ensure a healthy source of oxygen to the wound. This healing procedure is especially helpful in the case of PAD, which can eventually lead to chronic limb-threatening ischemia, by preventing the possibility of removing a limb. There are a variety of approaches to revascularization including both endovascular and open surgical approaches. For endovascular, there are two approaches including balloon angioplasty, where a balloon opens up blocked arteries, and stenting, where stents are used to support the lumen of the artery enhancing blood flow. Open surgical methods involve bypass surgeries where blood flow is redirected using conduit which can either be a manmade prosthetic or autogenous51. Considering a study performed by Hendri et al. (2023) which analyzed a group of 43 patients who had diabetic foot ulcers and PAD. They were split into two groups with 23 members in each where one group was healed through revascularization and one was not. When comparing the wound healing rates, the group receiving revascularization had a rate of 78.3% patients compared to the 26.1% patients in the non-revascularization group52. The findings of this research signifies the importance that vascular restoration has in patients with PAD and ischemic wounds. Additionally, heart failure can also delay wound healing due to reduced perfusion of tissue. There are a variety of drugs which can be used to help treat cardiovascular diseases and improve wound healing. One of such drugs is cilostazol. This drug is a phosphodiesterase III inhibitor which creates a vasodilatory effect on the wounds and also reduces platelet activity. In patients with PAD, this drug often increases the walking distance that patients can walk before feeling pain. There is also an increased perfusion effect of this drug, where there is a greater blood flow throughout the body, leading to an increase in oxygenation. This was proven by a randomized study in which patients taking this drug resulted in increased oxygen supply to the skin. This ability to improve perfusion in the body and raise oxygen levels for tissues enhances the wound healing process allowing these wounds to heal much faster53. There are also other drugs which can focus on inflammation. These immunomodulatory drugs have potential to reduce the inflammatory risk which in PAD patients is typically caused to the inflammasome-interleukin axis. There are a variety of these drugs, including Canakinumab, which was able to protect patients from cardiovascular events by effectively inhibiting inflammatory molecules such as IL-1ß and IL-6. Colchicine is another medication, where pro-inflammatory cytokines such as IL-1ß and IL-18 were inhibited, creating a potential.
By restoring the blood flow, revascularization is able to increase the delivery of vital nutrients to wounds promoting wound healing. These improved conditions for the wound allow angiogenesis, immune function, and fibroblast activity to be promoted. These allow for improved wound healing.
Current wound treatment approaches:
| Wound treatments | Pros | Cons | Products |
| Gauzes | – Inexpensive and widely available. – Easy to apply and remove. – Good absorbency for exudates. | – Poor moisture retention. – May cause trauma upon removal. – Requires frequent changes. | – Kerlix® Steri-Pad™ |
| Films | – Thin, transparent, and flexible. – Semi-permeable to gases and water vapor. – Maintains a moist wound environment. | – Non-absorbent; may lead to maceration. – Leakage risk with heavy exudate. – Not suitable for infected wounds. | – Opsite® Films |
| Foams | – Excellent absorbency. – Provides cushioning and thermal insulation. – Maintains a moist environment. | – Weak adhesion; often needs secondary dressing. – May cause trauma upon removal. – Can dry out if exudate is minimal. | – Allevyn (Smith and Nephew) – Mepilex® and Mepilex Ag® |
| Hydrocolloids | – Self-adhesive and easy to handle. – Promotes autolytic debridement. – Maintains moisture balance. | – Opaque—limits wound observation. – May produce odor or mimic infection. – Not suitable for highly exuding wounds. | – DuoDERM® – Tegaderm™ hydrocolloid dressing |
Traditional wound dressings consisting of cotton wool and gauze and compression bandages function primarily as protective barriers. The dressing absorbs liquids while preventing bacterial entry but it can cause wound drying which hinders keratinocyte migration and slows down the healing process. The process of removing these dressings can result in damage to newly formed tissue because of adhesion. These materials work best for treating simple wounds that have not caused significant damage and will heal on their own without any issues. The advanced conventional products hydrocolloids, gels and foams work to preserve moisture which enables natural healing processes. Moist environments promote cell migration and the breakdown of dead tissue without the trauma of dry dressings. The development of wound care has shifted from basic protective materials to biomaterials which perform various functions and adapt to different environmental conditions. The modern systems protect the body structure while managing biological problems which include maintaining moisture balance and preventing infections and delivering oxygen and pH regulation and supporting tissue repair especially for long-term or complex wounds14.
Hydrogels
Hydrogels have emerged as a popular biomaterial for treating wounds because they provide a moist healing environment through their soft jelly-like structure and high water content and biocompatible nature. The performance of these hydrogels largely depends on the polymers used in their formulation. The following are some commonly used natural polymers for hydrogels:
Chitosan
Chitosan emerges when chitin undergoes a deacetylation; the resulting linear polysaccharide is built with N‑acetyl‑D‑glucosamine interspersed with β‑(1→4)‑linked D‑glucosamine. Its skeleton resembles the glycosaminoglycans that populate the matrix. Both the polymer’s molecular weight and the extent of deacetylation dictate its mechanical traits. Thanks, to its charge the polymer can knock out microbes by pulling on their charged surfaces, a property that makes it handy for coating nanoparticles and, for supporting wound‑healing applications. Chitosan hydrogels ward off infection, boost leukocyte activity and tend to limit scar formation. Their mechanical sturdiness however is often lacking, a drawback that can be remedied by incorporating cross‑linkers such, as glutaraldehyde or genipin via gelation. Injectable chitosan‑based hydrogels have shown 96.4 % inhibition of Pseudomonas aeruginosa and 95.0 % inhibition of Staphylococcus aureus and have driven wound closure, up to 99.8 % within two weeks. In work carboxymethyl chitosan (CMCS) hydrogels have been engineered to boost solubility and speed up wound healing in models. At 37 °C the gel swells to 132 % of its volume soaks up exudate with ease and drives wound closure to roughly 99 % within two weeks behaving much like the extracellular matrix, in orchestrating tissue repair16.
Gelatin
Gelatin can be thought of as a polymer derived from collagen. It is peppered with RGD motifs that act like microscopic Velcro encouraging cells to adhere and proliferate. Its propensity to degrade harmlessly and its low immunogenic profile make it a favourite, in tissue‑engineering applications. The downside is its strength, which necessitates cross‑linking. Typical cross‑linkers—glutaraldehyde, formaldehyde and the carbodiimide EDC enhance stability while remaining external, to the gelatin matrix. Gelatin’s porous matrices soak up wound exudate. Keep the site moist, which in turn supports healing. Because gelatin lacks activity it is usually paired with antimicrobial agents or blended into hybrid polymers. Wang et al. (2023) Engineered a bilayer gelatin methacryloyl (GelMA) hydrogel loaded with epidermal growth factor (EGF) demonstrating both biofilm eradication and enhanced proliferation54. After twelve days of treatment E. coli‑infected wounds shrank to 7.9 % of their area54.
Hyaluronic Acid (HA)
Hyaluronic acid (HA) is a glycosaminoglycan built from N‑acetyl‑D‑glucosamine and D-glucuronic naturally residing in connective tissues and also producible, by microbes. Its strong affinity for water enables it to soak up exudate while simultaneously encouraging cell adhesion. By grafting groups onto acid and stabilizing the network through thiol–disulfide exchange, with oxidized glutathione a robust hydrogel emerges that resists hydrolytic degradation. Across the wound‑healing timeline HA‑based hydrogels can temper inflammation, spur angiogenesis and nurture endothelial cell proliferation. In a 2022 report Li et al. Fashioned an HA hydrogel cross‑linked with benzaldehyde‑functionalized PEG–polyglycerol caprate (PEGSB). This formulation can be activated by near‑infrared light to unleash activity against both E. coli and MRSA and delivers complete wound closure within fourteen days55.
Alginate
The biopolymer alginate exists as a natural substance which scientists obtain from brown algae and certain bacterial species through the combination of D-mannuronate (M) and L-guluronate (G) monomers. High-G alginates create stiff hydrogels through Ca²⁺-mediated ionic cross-linking. The exchange of Ca²⁺ ions with Na⁺ ions from wound exudate creates a protective gel-like barrier which helps the healing process.. The application of alginate triggers macrophage activation which leads to cytokine production (IL-6 and TNF-α) that promotes faster recovery of chronic wounds. Alginate functions as the main ingredient in commercial hydrogel dressings because it absorbs liquids while keeping wounds moist and enables easy dressing removal.
The physical and biological properties of hydrogels depend on the selection of appropriate polymers yet the biochemical conditions of the wound site determine how well the healing process will progress. Oxygen availability stands as one of the essential factors among these elements.
Through creating environments full of moisture and supporting the formation of ECM, hydrogels are able to perform functions such as regaining the migration of fibroblasts and also the proliferation of keratinocytes. By performing these functions, they can fight mechanisms which lead to impaired wound healing responses.
Conclusion
Chronic wound burden in the population is immense. Sen (2023) indicates that chronic wounds impose a burden on about 2.5 percent of the American population, with the highest rates in the elderly with diabetes and CVD56. Such patients typically have a significant decrease in health-related quality of life, particularly in the areas of physical function and mobility57. The economic burden is also high. Rice et al. (2014) projected that diabetic foot ulcers alone cost an extra $11,710 a year in healthcare costs per patient and add up to a national expenditure of $9 to $13 billion a year in the United States58. Research and clinical care continue to be disorganized despite increasing awareness. The relationship between cardiovascular dysfunction and impaired wound healing is not well understood. Mahmoudi and Gould (2020) highlight the absence of interdisciplinary teams in wound care research and endorse teams integrating vascular medicine, dermatology, endocrinology, and infectious disease expertise59. Likewise, Bus et al. (2020) posit the requirement for multidisciplinary strategies to cater to the holistic needs of patients with diabetic foot ulcers and associated comorbidities60. The growing number of people with diabetes and CVD and aging population has resulted in a substantial increase of chronic wound cases which now represents a significant public health challenge. Better care for CVD patients with chronic wound management is possible with a thorough understanding of the primary signaling pathways and the scientific rationale behind them. Instead of treating CVD and chronic wounds independently, the approach should be to identify the underlying cause.
Through multiple systemic and local biological processes, patients with CVD struggle with impaired wound healing. For example, vascular issues can lead to reduced perfusion of tissue and issues such as ischemia. Furthermore, conditions such as inflammation and oxidative stress can limit the steps of wound healing. These various issues create environments with factors which promote inflammation and reduced wound healing. Although there has been significant research into these mechanisms, this research should be converted into more clinical treatments and therapies.
Limitations
While making this review, there were a few limitations which hindered the quality of the data reported. Firstly, while there was an attempt to focus on human related studies, a lot of the data collected in the paper was from animal models. While animal models may represent how certain mechanisms work, they cannot completely apply to human populations. Secondly, the design of the studies analyzed in this review varied from one another. These differences led to different outcome measurements and populations which cannot be easily compared to each other. Finally, since the field of wound healing is constantly growing, newer therapies, treatments, and technologies do not yet have enough evidence demonstrating long-term success, meaning that they may not necessarily work when implemented fully.
Future Directions
Future research in this field should focus on looking at large-scale clinical studies which look at the wound healing outcomes for patients who have cardiovascular diseases. Many studies focus on animal models so future research focusing on clinical studies can help these findings spread to clinical practice. Furthermore, researching further in areas such as immune modulation, angiogenesis, and also microbiome immune interactions can lead to new treatments being produced.
References
- R. J. White, S. Fumarola, J. Denyer. Role of oxygen in wound healing: a review of evidence. Journal of Wound Care. Vol. 20, pg. 192, 2011 https://doi.org/10.12968/JOWC.2011.20.4.192 [↩]
- W. W. Li, M. J. Carter, E. Mashiach, S. D. Guthrie. Vascular assessment of wound healing: a clinical review. International Wound Journal. Vol. 14, pg. 460, 2016 https://doi.org/10.1111/IWJ.12622 [↩]
- S. Schreml, R. M. Szeimies, L. Prantl, S. Karrer, M. Landthaler, P. Babilas. Oxygen in acute and chronic wound healing. The British Journal of Dermatology. Vol. 163, pg. 257–268, 2010 https://doi.org/10.1111/J.1365-2133.2010.09804.X [↩]
- D. J. Margolis, L. Allen-Taylor, O. Hoffstad, J. A. Berlin. Diabetic neuropathic foot ulcers. Diabetes Care. Vol. 25, pg. 1835–1839, 2002 https://doi.org/10.2337/DIACARE.25.10.1835 [↩]
- Y. Nishio, Y. Tsuji, I. Kitano, H. Terashi. Influence of peripheral arterial disease on wound healing in heel pressure ulcers. Kobe Journal of Medical Sciences. Vol. 67, pg. E146, 2022 [↩]
- J. Hafner, I. Schaad, E. Schneider, B. Seifert, G. Burg, P. C. Cassina. Leg ulcers in peripheral arterial disease (arterial leg ulcers): impaired wound healing above the threshold of chronic critical limb ischemia. Journal of the American Academy of Dermatology. Vol. 43, pg. 1001–1008, 2000 https://doi.org/10.1067/mjd.2000.108375 [↩]
- M. C. Redmond, G. Gethin, D. P. Finn. A review of chronic wounds and their impact on negative affect, cognition, and quality of life. International Wound Journal. Vol. 22, pg. e70748, 2025 https://doi.org/10.1111/IWJ.70748 [↩]
- Y. He, Q. Chang, F. Lu. Oxygen-releasing biomaterials for chronic wounds breathing: from theoretical mechanism to application prospect. Materials Today Bio. Vol. 20, pg. 100687, 2023 https://doi.org/10.1016/J.MTBIO.2023.100687 [↩] [↩]
- V. Gounden, M. Singh. Hydrogels and wound healing: current and future prospects. Gels 2024, Vol. 10, Page 43. Vol. 10, pg. 43, 2024 https://doi.org/10.3390/GELS10010043 [↩] [↩] [↩]
- A. T. Abraham, S. Mojaddedi, I. H. Loseke, C. Bray, A. T. Abraham, S. Mojaddedi, I. H. Loseke, C. Bray. Hypertension in patients with peripheral artery disease: an updated literature review. Cureus. Vol. 16, 2024 https://doi.org/10.7759/CUREUS.62246 [↩]
- H. Sano, S. Ichioka, N. Sekiya. Influence of oxygen on wound healing dynamics: assessment in a novel wound mouse model under a variable oxygen environment. PLOS ONE. Vol. 7, pg. e50212, 2012 https://doi.org/10.1371/JOURNAL.PONE.0050212 [↩]
- U. A. Okonkwo, L. A. Dipietro. Diabetes and wound angiogenesis. International Journal of Molecular Sciences. Vol. 18, 2017 https://doi.org/10.3390/IJMS18071419 [↩] [↩] [↩] [↩] [↩]
- Y. N. Pierpont, T. P. Dinh, R. E. Salas, E. L. Johnson, T. G. Wright, M. C. Robson, W. G. Payne. Obesity and surgical wound healing: a current review. ISRN Obesity. Vol. 2014, pg. 638936, 2014 https://doi.org/10.1155/2014/638936 [↩]
- I. J. Das, T. Bal. PH factors in chronic wound and ph-responsive polysaccharide-based hydrogel dressings. International Journal of Biological Macromolecules. Vol. 279, pg. 135118, 2024 https://doi.org/10.1016/J.IJBIOMAC.2024.135118 [↩] [↩]
- M. Zawani, M. B. Fauzi. Injectable hydrogels for chronic skin wound management: a concise review. Biomedicines 2021, Vol. 9, Page 527. Vol. 9, pg. 527, 2021 https://doi.org/10.3390/BIOMEDICINES9050527 [↩]
- S. R. B, D. S. R. Rajkumar, K. K, V. Vijayaragavan, S. R. B, D. S. R. Rajkumar, K. K, V. Vijayaragavan. Chitosan-based biomaterial in wound healing: a review. Cureus. Vol. 16, 2024 https://doi.org/10.7759/CUREUS.55193 [↩] [↩]
- Y. Hu, L. Yu, Q. Dai, X. Hu, Y. Shen, Y. Shen. Multifunctional antibacterial hydrogels for chronic wound management. Biomaterials Science. Vol. 12, pg. 2460–2479, 2024 https://doi.org/10.1039/D4BM00155A [↩] [↩]
- V. Falanga. Wound healing and its impairment in the diabetic foot. Lancet. Vol. 366, pg. 1736–1743, 2005 https://doi.org/10.1016/S0140-6736(05)67700-8 [↩]
- S. Patel, S. Srivastava, M. R. Singh, D. Singh. Mechanistic insight into diabetic wounds: pathogenesis, molecular targets and treatment strategies to pace wound healing. Biomedicine & Pharmacotherapy. Vol. 112, pg. 108615, 2019 https://doi.org/10.1016/J.BIOPHA.2019.108615 [↩]
- H. Brem, M. Tomic-Canic. Cellular and molecular basis of wound healing in diabetes. The Journal of Clinical Investigation. Vol. 117, pg. 1219–1222, 2007 https://doi.org/10.1172/JCI32169 [↩]
- D. G. Armstrong, T. W. Tan, A. J. M. Boulton, S. A. Bus. Diabetic foot ulcers: a review. JAMA. Vol. 330, pg. 62–75, 2023 https://doi.org/10.1001/JAMA.2023.10578 [↩] [↩]
- A. E. Louiselle, S. M. Niemiec, C. Zgheib, K. W. Liechty. Macrophage polarization and diabetic wound healing. Translational Research. Vol. 236, pg. 109–116, 2021 https://doi.org/10.1016/j.trsl.2021.05.006 [↩]
- M. Malone, K. Johani, S. O. Jensen, I. B. Gosbell, H. G. Dickson, S. McLennan, H. Hu, K. Vickery. Effect of cadexomer iodine on the microbial load and diversity of chronic non-healing diabetic foot ulcers complicated by biofilm in vivo. The Journal of Antimicrobial Chemotherapy. Vol. 72, pg. 2093–2101, 2017 https://doi.org/10.1093/JAC/DKX099 [↩]
- S. F. Spampinato, G. I. Caruso, R. De Pasquale, M. A. Sortino, S. Merlo. The treatment of impaired wound healing in diabetes: looking among old drugs. Pharmaceuticals 2020, Vol. 13, Page 60. Vol. 13, pg. 60, 2020 https://doi.org/10.3390/PH13040060 [↩] [↩]
- Y. J. J. Rhou, F. R. Henshaw, M. J. McGill, S. M. Twigg. Congestive heart failure presence predicts delayed healing of foot ulcers in diabetes: an audit from a multidisciplinary high-risk foot clinic. Journal of Diabetes and Its Complications. Vol. 29, pg. 556–562, 2015 https://doi.org/10.1016/J.JDIACOMP.2015.02.009 [↩] [↩] [↩]
- A. Alfaddagh, S. S. Martin, T. M. Leucker, E. D. Michos, M. J. Blaha, C. J. Lowenstein, S. R. Jones, P. P. Toth. Inflammation and cardiovascular disease: from mechanisms to therapeutics. American Journal of Preventive Cardiology. Vol. 4, pg. 100130, 2020 https://doi.org/10.1016/J.AJPC.2020.100130 [↩]
- U. Chalise, M. Becirovic-Agic, M. L. Lindsey. The cardiac wound healing response to myocardial infarction. WIREs Mechanisms of Disease. Vol. 15, 2023 https://doi.org/10.1002/WSBM.1584 [↩] [↩]
- S. R. Iyer, B. H. Annex. Therapeutic angiogenesis for peripheral artery disease: lessons learned in translational science. JACC: Basic to Translational Science. Vol. 2, pg. 503–512, 2017 https://doi.org/10.1016/J.JACBTS.2017.07.012 [↩] [↩]
- G. A. Cabral-Pacheco, I. Garza-Veloz, C. C. D. La Rosa, J. M. Ramirez-Acuña, B. A. Perez-Romero, J. F. Guerrero-Rodriguez, N. Martinez-Avila, M. L. Martinez-Fierro. The roles of matrix metalloproteinases and their inhibitors in human diseases. International Journal of Molecular Sciences 2020, Vol. 21, Page 9739. Vol. 21, pg. 9739, 2020 https://doi.org/10.3390/IJMS21249739 [↩]
- S. Van Linthout, C. Tschöpe. Inflammation – cause or consequence of heart failure or both? Current Heart Failure Reports. Vol. 14, pg. 251–265, 2017 https://doi.org/10.1007/S11897-017-0337-9 [↩]
- A. A. Ahmed, P. A. Mooar, M. Kleiner, J. S. Torg, C. T. Miyamoto. Hypertensive patients show delayed wound healing following total hip arthroplasty. PLoS ONE. Vol. 6, pg. e23224, 2011 https://doi.org/10.1371/JOURNAL.PONE.0023224 [↩] [↩]
- J. Zhao, S. Yang, B. Shu, L. Chen, R. Yang, Y. Xu, J. Xie, X. Liu, S. Qi. Transient high glucose causes persistent vascular dysfunction and delayed wound healing by the dnmt1-mediated ang-1/nf-κb pathway. Journal of Investigative Dermatology. Vol. 141, pg. 1573–1584, 2021 https://doi.org/10.1016/j.jid.2020.10.023 [↩]
- A. Opneja, S. Kapoor, E. X. Stavrou. Contribution of platelets, the coagulation and fibrinolytic systems to cutaneous wound healing. Thrombosis Research. Vol. 179, pg. 56–63, 2019 https://doi.org/10.1016/j.thromres.2019.05.001 [↩] [↩]
- R. J. Esper, R. A. Nordaby, J. O. Vilariño, A. Paragano, J. L. Cacharrón, R. A. Machado. Endothelial dysfunction: a comprehensive appraisal. Cardiovascular Diabetology 2006 5:1. Vol. 5, pg. 4-, 2006 https://doi.org/10.1186/1475-2840-5-4 [↩]
- M. Shimabukuro. L-arginine, nitric oxide, and endothelial dysfunction underlying atherosclerotic cardiovascular disease (ascvd). Journal of Atherosclerosis and Thrombosis. Vol. 30, pg. 1311, 2023 https://doi.org/10.5551/JAT.ED235 [↩]
- A. Shaito, K. Aramouni, R. Assaf, A. Parenti, A. Orekhov, A. El Yazbi, G. Pintus, A. H. Eid. Oxidative stress-induced endothelial dysfunction in cardiovascular diseases. Frontiers in Bioscience (Landmark Edition). Vol. 27, 2022 https://doi.org/10.31083/J.FBL2703105 [↩]
- A. R. Cyr, L. V. Huckaby, S. S. Shiva, B. S. Zuckerbraun. Nitric oxide and endothelial dysfunction. Critical Care Clinics. Vol. 36, pg. 307, 2020 https://doi.org/10.1016/J.CCC.2019.12.009 [↩]
- M. J. Cross, L. Claesson-Welsh. FGF and vegf function in angiogenesis: signalling pathways, biological responses and therapeutic inhibition. Trends in Pharmacological Sciences. Vol. 22, pg. 201–207, 2001 https://doi.org/10.1016/S0165-6147(00)01676-X [↩]
- K. Florek, D. Mendyka, K. Gomułka. Vascular endothelial growth factor (vegf) and its role in the cardiovascular system. Biomedicines 2024, Vol. 12, Page 1055. Vol. 12, pg. 1055, 2024 https://doi.org/10.3390/BIOMEDICINES12051055 [↩]
- G. L. Semenza. Hypoxia-inducible factor 1 and cardiovascular disease. Annual Review of Physiology. Vol. 76, pg. 39–56, 2014 https://doi.org/10.1146/ANNUREV-PHYSIOL-021113-170322/CITE/REFWORKS [↩]
- D. Moris, M. Spartalis, E. Spartalis, G. S. Karachaliou, G. I. Karaolanis, G. Tsourouflis, D. I. Tsilimigras, E. Tzatzaki, S. Theocharis. The role of reactive oxygen species in the pathophysiology of cardiovascular diseases and the clinical significance of myocardial redox. Annals of Translational Medicine. Vol. 5, pg. 326–326, 2017 https://doi.org/10.21037/ATM.2017.06.27 [↩]
- X. Wang, S. Yu, L. Xie, M. Xiang, H. Ma. The role of the extracellular matrix in cardiac regeneration. Heliyon. Vol. 11, 2025 https://doi.org/10.1016/j.heliyon.2024.e41157 [↩]
- F. G. Spinale. Matrix metalloproteinases: regulation and dysregulation in the failing heart. Circulation Research. Vol. 90, pg. 520–530, 2002 https://doi.org/10.1161/01.RES.0000013290.12884.A3/FORMAT/EPUB [↩]
- C. H. Ong, C. L. Tham, H. H. Harith, N. Firdaus, D. A. Israf. TGF-β-induced fibrosis: a review on the underlying mechanism and potential therapeutic strategies. European Journal of Pharmacology. Vol. 911, 2021 https://doi.org/10.1016/j.ejphar.2021.174510 [↩]
- C. Bocca, E. Novo, A. Miglietta, M. Parola. Angiogenesis and fibrogenesis in chronic liver diseases. CMGH. Vol. 1, pg. 477–488, 2015 https://doi.org/10.1016/j.jcmgh.2015.06.011 [↩]
- C. J. Ferrante, S. J. Leibovich. Regulation of macrophage polarization and wound healing. Https://Home.Liebertpub.Com/Wound. Vol. 1, pg. 10–16, 2012 https://doi.org/10.1089/WOUND.2011.0307 [↩]
- A. Pontén, E. B. Folestad, K. Pietras, U. Eriksson. Platelet-derived growth factor d induces cardiac fibrosis and proliferation of vascular smooth muscle cells in heart-specific transgenic mice. Circulation Research. Vol. 97, pg. 1036–1045, 2005 https://doi.org/10.1161/01.RES.0000190590.31545.D4;WGROUP:STRING:PUBLICATION [↩]
- H. Zhang, N. S. Dhalla. The role of pro-inflammatory cytokines in the pathogenesis of cardiovascular disease. International Journal of Molecular Sciences 2024, Vol. 25, Page 1082. Vol. 25, pg. 1082, 2024 https://doi.org/10.3390/IJMS25021082 [↩]
- M. Ansari, A. Darvishi. A review of the current state of natural biomaterials in wound healing applications. Frontiers in Bioengineering and Biotechnology. Vol. 12, pg. 1309541, 2024 https://doi.org/10.3389/FBIOE.2024.1309541/FULL [↩]
- A. Al Mamun, A. Ullah, M. E. H. Chowdhury, H. E. Marei, A. P. Madappura, M. Hassan, M. Rizwan, V. G. Gomes, A. Amirfazli, A. Hasan. Oxygen releasing patches based on carbohydrate polymer and protein hydrogels for diabetic wound healing: a review. International Journal of Biological Macromolecules. Vol. 250, pg. 126174, 2023 https://doi.org/10.1016/J.IJBIOMAC.2023.126174 [↩]
- J. A. Beckman, P. A. Schneider, M. S. Conte. Advances in revascularization for peripheral artery disease: revascularization in pad. Circulation Research. Vol. 128, pg. 1885–1912, 2021 https://doi.org/10.1161/CIRCRESAHA.121.318261 [↩]
- Hendri, R. Suhartono, A. Kekalih, J. R. Adriani, M. Faruk. Healing rate comparison of revascularized and non-revascularized diabetic foot ulcers with peripheral arterial disease. Surgery Open Science. Vol. 16, pg. 235–241, 2023 https://doi.org/10.1016/j.sopen.2023.11.008 [↩]
- K. Desai, B. Han, L. Kuziez, Y. Yan, M. A. Zayed. Literature review and meta-analysis of the efficacy of cilostazol on limb salvage rates after infrainguinal endovascular and open revascularization. Journal of Vascular Surgery. Vol. 73, pg. 711-721.e3, 2021 https://doi.org/10.1016/j.jvs.2020.08.125 [↩]
- Y. Chen, X. Wang, S. Tao, Q. Wang, P. Q. Ma, Z. B. Li, Y. L. Wu, D. W. Li. Research advances in smart responsive-hydrogel dressings with potential clinical diabetic wound healing properties. Military Medical Research 2023 10:1. Vol. 10, pg. 37-, 2023 https://doi.org/10.1186/S40779-023-00473-9 [↩] [↩]
- M. Li, Y. Liang, Y. Liang, G. Pan, B. Guo. Injectable stretchable self-healing dual dynamic network hydrogel as adhesive anti-oxidant wound dressing for photothermal clearance of bacteria and promoting wound healing of mrsa infected motion wounds. Chemical Engineering Journal. Vol. 427, pg. 132039, 2022 https://doi.org/10.1016/J.CEJ.2021.132039 [↩]
- C. K. Sen. Human wound and its burden: updated 2022 compendium of estimates. Advances in Wound Care. Vol. 12, pg. 657, 2023 https://doi.org/10.1089/WOUND.2023.0150 [↩]
- M. Olsson, K. Järbrink, U. Divakar, R. Bajpai, Z. Upton, A. Schmidtchen, J. Car. The humanistic and economic burden of chronic wounds: a systematic review. Wound Repair and Regeneration. Vol. 27, pg. 114–125, 2019 https://doi.org/10.1111/WRR.12683 [↩]
- J. B. Rice, U. Desai, A. K. G. Cummings, H. G. Birnbaum, M. Skornicki, N. B. Parsons. Burden of diabetic foot ulcers for medicare and private insurers. Diabetes Care. Vol. 37, pg. 651–658, 2014 https://doi.org/10.2337/DC13-2176 [↩]
- M. Mahmoudi, L. Gould. Opportunities and challenges of the management of chronic wounds: a multidisciplinary viewpoint. Chronic Wound Care Management and Research. Vol. Volume 7, pg. 27–36, 2020 https://doi.org/10.2147/CWCMR.S260136 [↩]
- S. A. Bus, L. A. Lavery, M. Monteiro-Soares, A. Rasmussen, A. Raspovic, I. C. Sacco, J. J. van Netten, Z. Twente. Guidelines on the prevention of foot ulcers in persons with diabetes (iwgdf 2019 update). 2020 https://doi.org/10.1002/dmrr.3269 [↩]