Abstract
The global prevalence of Type 2 Diabetes Mellitus (T2DM) has increased substantially in recent decades. Although pharmacotherapy remains a cornerstone of clinical management for T2DM, there is growing interest in structured nonpharmacological strategies that target the underlying metabolic dysfunction. This observational case study followed a 50-year-old South Asian male in the United States for 44 weeks following his diagnosis of severe T2DM (with a baseline HbA1c of 12.0%). It describes the metabolic changes observed during his adoption of a structured, evidence-based lifestyle approach (hereafter referred to as the Insulight Protocol). Following discussion with his physician, the subject elected to pursue intensive lifestyle modification, and only initiate pharmacotherapy if metabolic markers failed to improve in three months. He eliminated dietary sugars, refined carbohydrates, and gluten-containing foods; prioritized fiber-rich foods, proteins, and unsaturated fats; adopted consistent hydration practices; incorporated structured physical activity; maintained a daily 16-hour fast, and consumed selected phytochemical-rich foods during the fasted state. Adherence support included education on the scientific rationale underlying the protocol, and use of a non-clinical AI-assisted informational tool intended to guide real-time dietary decisions. At 16 weeks, the subject’s HbA1c, body weight, and triglycerides measured 5.3% (down from 12.0%), 196 lbs. (down 14% from 229 lbs.), and 135 mg/dL (down from a baseline 619 mg/dL) respectively. At 44 weeks, HbA1c, body weight, and triglycerides measured 5.3%, 187 lbs, and 104 mg/dL respectively. By the 2021 ADA consensus definition – HbA1c below 6.5% sustained for at least three months without glucose-lowering pharmacotherapy – the observed outcomes at 44 weeks meet criteria for remission. These improvements were observed without the initiation of glucose-lowering pharmacotherapy. While causality cannot be inferred from a single case, the magnitude, velocity, and durability of change support further investigation in larger and more diverse populations.
Keywords: Type 2 Diabetes Mellitus, T2DM, Diabetes Remission, Hyperglycemia, Hypertriglyceridemia, Metabolic Reversal, HbA1c, South Asian Phenotype, Intermittent Fasting, Time-Restricted Eating, Nonpharmacological Intervention, Phytochemicals, Lifestyle Medicine, Structured Adherence.
Introduction
Type 2 Diabetes Mellitus (T2DM) represents a major and growing global public health challenge with an estimated 589 million adults living with diabetes and an additional 1.1 billion individuals with impaired glucose tolerance (IGT) or impaired fasting glucose (IFG), placing them at an elevated risk of T2DM1,2. While pharmacotherapy remains a cornerstone of T2DM management, it primarily addresses downstream glycemic control rather than upstream metabolic drivers such as ectopic lipid accumulation in the liver and pancreas, which in turn contribute to insulin resistance and the impairment of insulin-producing pancreatic β-cells3. This distinction has fueled interest in structured nonpharmacological interventions that attempt to modify the underlying pathophysiology. Prior research3 has also demonstrated the potential reversibility of T2DM in selected individuals, with the DiRECT (UK) trial4 showing that a structured, nonpharmacological lifestyle approach emphasizing weight loss and behavioral support enabled approximately one-third of participants to achieve and maintain diabetes remission at two years. Despite these findings, rapid normalization of severe hyperglycemia through nonpharmacological approaches alone remains uncommon in routine clinical practice, particularly in individuals presenting with markedly elevated HbA1c levels5. Additionally, standard pharmacological therapy is generally associated with HbA1c reductions of approximately 1–2% over a six-month period5. Individual cases demonstrating larger improvements in glycemic markers over shorter timeframes may offer useful insights into the feasibility of intensive lifestyle strategies.
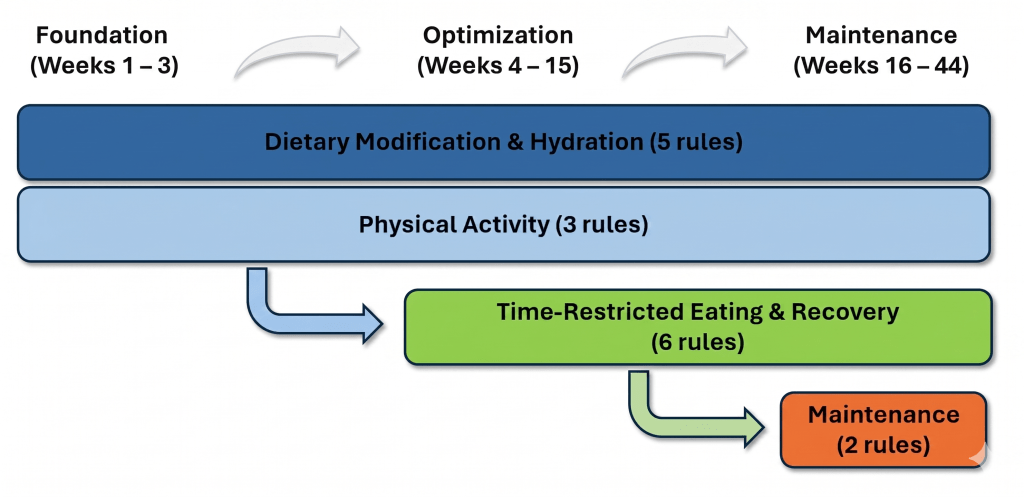
This case report describes a 50-year-old South Asian male with newly diagnosed severe T2DM (HbA1c 12.0%), in whom rapid improvement in glycemic markers was observed following the adoption of a structured, evidence-based lifestyle approach. The full set of 16 rules was compiled within the first few days following diagnosis, through rapid consultation between the subject, his family and the subject’s physician. The rules were implemented in deliberate phases: eight foundation rules were introduced in Week 1; six additional rules covering time-restricted eating, phytochemical adjuncts, and recovery practices were introduced in Week 4, deferred from Week 1 to align with the completion of a scheduled travel commitment; and two maintenance rules, while identified at the outset, were held pending laboratory confirmation of glycemic normalization, which occurred at Week 16. This phased implementation was prospectively planned and not a post-hoc adaptation. The approach was undertaken as a voluntary lifestyle change with agreement that pharmacotherapy would be initiated within three months if the metabolic markers did not adequately improve. The framework was later formalized and named the Insulight Protocol by the investigator for descriptive and analytical purposes, and emphasized strategies associated in prior literature with lower insulin demand and increased lipid oxidation3,4,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28.
During the observational period, HbA1c decreased by 6.7 percentage points (from 12.0% to 5.3%) over approximately 16 weeks, and remained stable at 44 weeks in the absence of glucose-lowering pharmacotherapy. This report serves as a hypothesis-generating case observation, intended to document feasibility and prompt further investigation, and should not be interpreted as evidence of a generalizable treatment approach. The findings provide a foundation for future research in larger and more diverse cohorts.
To contextualize the observed metabolic changes, daily adherence logs were aggregated weekly and compared against longitudinal biomarker trends; all laboratory results were cross-referenced with physician records to confirm accuracy.
Scientific Foundation For The Lifestyle Approach
The scientific rationale for the various strategies adopted by the subject in this structured lifestyle approach is summarized below, organized into evidence-supported core components, and adjunctive, hypothesis-generating components.
Evidence-Supported Core Components
The Twin Cycle Hypothesis proposed by Taylor3 describes T2DM as a condition potentially driven by excess fat accumulation in the liver and pancreas, resulting in hepatic insulin resistance and progressive pancreatic β-cell dysfunction. Sustained weight loss may reduce ectopic fat in these organs, improving insulin response and supporting β-cell recovery. The DiRECT (UK) trial provided clinical support, showing that a structured weight-focused lifestyle program enabled approximately one-third of participants to achieve T2DM remission4. The Carbohydrate-Insulin Model, described by Ludwig et al.7, posits that meals with a high glycemic load can drive chronic hyperinsulinemia and increase anabolic signaling, promoting fat storage and limiting access to endogenous energy reserves. Reducing dietary glycemic load may lower insulin demand and shift energy utilization toward increased lipolysis and lipid oxidation8,27, potentially reducing ectopic fat burden and improving insulin sensitivity over time.
Skeletal muscle serves as a primary site of glucose disposal. Low-intensity physical activity, such as walking, stimulates the insulin-independent translocation of GLUT4 transporters to the muscle cell membrane, increasing glucose uptake and attenuating postprandial glucose excursions even in insulin-resistant states12,13.Nutrient sequencing (consuming fiber-rich foods before proteins and carbohydrates) slows gastric emptying and reduces both glucose absorption rates and postprandial demand on pancreatic β-cells to produce insulin6.Resistance training further expands glucose-handling capacity by increasing muscle mass, glycogen storage, and GLUT4 expression14. Time-restricted eating can reduce cumulative daily insulin exposure by extending the post-absorptive period during which insulin levels remain low9. Intermittent-fasting and time-restricted eating have been associated with improvements in metabolic flexibility, mitochondrial efficiency, and insulin sensitivity9. By reducing cumulative insulin exposure, time-restricted eating may support insulin sensitivity in the liver, skeletal muscle, and adipose tissue while reducing demand on pancreatic β-cells to produce insulin9,25,26.
Psychological stress and sleep deprivation activate the hypothalamic-pituitary-adrenal (HPA) axis, increasing cortisol secretion and stimulating hepatic gluconeogenesis, raising glucose levels even in the fasted state21. Disrupted sleep impairs appetite regulation by suppressing leptin and increasing ghrelin, promoting increased caloric intake and preference for high-glycemic foods22. Inflammation further compounds insulin resistance by interfering with insulin signaling in skeletal muscle and the liver8,18.To address these factors, the approach emphasized sleep hygiene and nature-based physical activity for stress regulation29,30.
Adjunctive, Hypothesis-Generating Components
Several additional components were incorporated based on mechanistic plausibility rather than established clinical efficacy at the doses and durations used in this case. Consistent hydration practices, including limiting fluid intake during meals, and avoiding very cold beverages during meals, were adopted to potentially modulate gastric emptying and nutrient delivery19,20,31. Prolonged fasting periods may also support autophagy, a cellular recycling process associated with sustained low-insulin states9, though this was not directly measured in this case. Selected dietary phytochemicals in whole-food form, including gingerol (from ginger), curcumin (from turmeric, with black pepper to enhance bioavailability), allicin (from garlic), and cinnamaldehyde (from Ceylon cinnamon) included as hypothesis-generating adjuncts based on prior literature reporting associations with anti-inflammatory and insulin-sensitizing effects10,11,16; clinical efficacy at the doses and durations used in this case has not been established. Organic acids, such as citric or acetic acid (e.g., lemon or vinegar) have been associated in some studies with attenuated postprandial glycemic responses23,24 and are similarly considered adjunctive and hypothesis-generating.
Together, these mechanisms provide a scientific rationale for the lifestyle approach adopted by the subject.
Methods
Participant
The subject was a 50-year-old male of South Asian descent residing in the United States. Ethnicity is a relevant clinical factor in this case, as South Asian populations have been shown to exhibit increased cardiometabolic risk, including insulin resistance and visceral adiposity, at lower body mass index (BMI) thresholds, a pattern often referred to as the South Asian phenotype32. The subject had no prior history of pharmacological treatment for diabetes but had been receiving treatment for hypertension for more than 20 years. Prior to adopting this lifestyle approach, the subject’s routine was predominantly sedentary and characterized by a dietary pattern with high glycemic load (e.g., refined carbohydrates, processed snack foods, sugar-sweetened beverages, and desserts), which has been associated with chronic hyperinsulinemia, a metabolic state that promotes adipogenesis (fat storage) while inhibiting lipid oxidation7,8. With multiple family members being diagnosed with diabetes, the subject’s family history suggests increased genetic susceptibility to the disease.
The subject’s clinical presentation began in late October 2024 with the sudden onset of blurred vision while initiating computer-based work. Subsequent laboratory testing revealed a markedly elevated HbA1c level of 12.0%, a fasting plasma glucose of 342 mg/dL, and a markedly abnormal lipid profile, including serum triglycerides at 619 mg/dL, more than four times the upper limit of the reference range. Based on these findings, the subject was diagnosed with T2DM accompanied by severe hyperglycemia and severe hypertriglyceridemia. The subject’s complete medication list at baseline consisted of Losartan 50 mg daily for hypertension, which had been ongoing for more than 5 years and remained unchanged throughout the 44-week observation period. No lipid-lowering agents, glucose-lowering medications, or anticoagulants were prescribed at any point during the observation period. Additional clinical markers including renal function, fasting insulin, C-peptide, ketones, and autoimmune markers were not assessed; waist circumference was self-measured at 42 inches at baseline, 38 inches at 16 weeks, and 35 inches at 44 weeks. At the start of the observation period in November 2024 (Week 1), the subject weighed 229 lbs. with a height of 5’8″, corresponding to a BMI of 35.3. While this meets criteria for Class II obesity under CDC standards33, it may represent elevated metabolic risk when evaluated against BMI thresholds adjusted for Asian populations34. The diagnosis also provided context for several symptoms that the subject had experienced in the preceding months, including unintentional weight loss (from 246 lbs. in August 2024 to 229 lbs. in November 2024), chronic polydipsia (excessive thirst), palmar pruritus (itchy palms), and pronounced cognitive fatigue (brain fog).
Following the diagnosis, the physician recommended prompt initiation of pharmacotherapy (metformin) in conjunction with lifestyle modification; however, the subject elected to pursue an intensive nonpharmacological approach, with the understanding that pharmacotherapy would be initiated if the metabolic markers (HbA1c, fasting plasma glucose, and serum triglycerides) did not improve within three months, thereby ensuring a clear contingency for standard medical care.
Research Design
The subject was observed longitudinally for a total duration of 44 weeks, beginning three days after the diagnosis of severe T2DM.
Layer 1: 16 Core Physiological Rules
The subject, in consultation with his family and his physician, adopted 16 evidence-based rules that comprise the core physiological layer, or Layer 1, of the structured lifestyle approach. Because these rules addressed diet, hydration, physical activity, meal timing, sleep, and adjunctive phytochemical intake simultaneously, this is best characterized as a multi-component lifestyle intervention. The contribution of any individual rule to the observed outcomes cannot be determined from this single-subject design. These rules were adopted in three sequential phases. Eight foundation rules were introduced in Week 1, and included five rules related to dietary modification and hydration, and three related to physical activity. In Week 4, at the start of the optimization phase (Weeks 4-15), six additional rules relating to time-restricted eating, intake of phytochemical-rich dietary adjuncts and recovery practices were introduced. As the subject’s HbA1c reached the non-diabetic range in Week 15, the approach transitioned to the maintenance phase (Weeks 16-44), during which two additional rules were introduced. No formal caloric restriction target was implemented during the observation period, and alcohol consumption was not a component of the subject’s baseline dietary pattern and therefore was not explicitly addressed within the structural framework.
Weeks 1–44: Dietary Modification, Hydration and Physical Activity
Beginning Week 1, the subject focused on dietary modification, hydration practices, and physical activity that were sustained throughout the observation period, until Week 44. Given the subject’s markedly elevated baseline HbA1c (12.0%), the primary focus of these eight rules was reduction of insulin demand and postprandial glucose excursions. Rules 1 to 5 described here account for the Dietary Modification and Hydration rules, while Rules 6 to 8 account for Physical Activity rules.
Rule 01: Eliminate Sugars, Refined Carbohydrates, and Gluten from the Diet. The subject eliminated added sugars and refined carbohydrates from his diet for the entire duration of the observation period. Gluten-containing foods were explicitly excluded as a practical proxy for removing ultra-processed, calorie-dense convenience foods often associated with rapid glucose excursions. Adequate protein and healthy fats were prioritized with each meal. When additional satiety was required, low-glycemic whole foods such as nuts (e.g., walnuts and almonds), and berries (e.g., blueberries and strawberries), and full-fat plain yogurt were consumed selectively.
Rule 02: Strict Restriction of Glycemic Load. Given the severity of baseline hyperglycemia, the subject temporarily excluded foods with moderate to high natural sugar content including refined grains (e.g., white rice), starch-dense tubers (e.g., potatoes), and traditional starch-dense preparations (e.g., idly), in addition to fruits and other naturally sugar-containing foods (e.g., carrots, corn, beetroot, and coconut) during this phase. This restriction was intended to minimize postprandial glucose exposure during the early stage of the protocol. These foods were reintroduced in moderation during the maintenance phase in Week 16.
Rule 03: Hydration. The subject followed consistent hydration practices intended to support digestive efficiency and glycemic control. He consumed approximately 64 ounces of water daily, distributed evenly throughout the day. Fluid intake during meals was limited to 2-4 oz of room-temperature water, with larger volumes delayed for approximately 30 minutes post-meal, and ice-cold beverages were avoided. These practices were intended to modulate gastric emptying and potentially reduce rapid nutrient delivery following meals.
Rule 04: Fiber-First Food Intake During Meals. Food ingestion at meals followed a consistent sequence, with fiber-rich foods (e.g., vegetables) ingested before proteins and fats, intending to reduce the postprandial demand on pancreatic β-cells to produce insulin6.
Rule 05: Eliminate Snacking. The subject avoided snacking between meals to minimize repeated insulin stimulation and cumulative postprandial glucose exposure. In rare situations requiring additional energy expenditure (e.g., a long hike), low-glycemic whole foods such as nuts and berries were used selectively.
Rule 06: Walk 10,000 Steps Daily. The subject aimed to walk 10,000 steps per day, five days per week, intending to increase glucose uptake by skeletal muscle12 and may have helped reduce stress-related cortisol activity, which is known to increase hepatic glucose production21,29.
Rule 07: Postprandial Movement and Activation of the Soleus Muscle. The subject engaged in a 15-minute walk within 15–30 minutes after meals to blunt postprandial glucose excursions. Activation of oxidative muscle fibers, particularly the soleus muscle, was prioritized because of its capacity for sustained glucose utilization during low-intensity activity, including calf raises performed while seated at a desk12,13.
Rule 08: Low-to-Moderate Resistance Training. The subject incorporated low-intensity resistance training three times per week, for approximately 20 minutes per session, to preserve lean muscle mass and maintain skeletal muscle glycogen storage capacity14.
Weeks 4–44: Time-Restricted Eating and Metabolic Recovery
In Week 4, the subject incorporated six additional temporal strategies, Rules 9 to 14, while continuing prior dietary, hydration and physical activity practices.
Rule 09: Time-Restricted Eating (TRE, or 16-hour fast). The subject utilized an 8-hour daily feeding window (from 11:30 AM – 7:30 PM) intending to reduce cumulative insulin exposure and support metabolic flexibility, defined as the body’s ability to efficiently transition between glucose utilization in the fed state and lipid oxidation in the fasted state.
Rule 10: Weekly Metabolic Challenge (One-Meal-A-Day). Once per week, the subject consumed just one-meal-a-day (OMAD) intending to enhance metabolic flexibility and support autophagy, a cellular recycling process that can be altered in states of chronic hyperinsulinemia9,27.
Rule 11: Garlic Intake in the Fasted State. Each morning, the subject consumed one clove of raw garlic that had been crushed and allowed to rest for approximately 10 minutes prior to ingestion. This resting period allowed enzymatic conversion of alliin to allicin, a bioactive compound that has been associated with improvements in insulin sensitivity in prior studies10; clinical efficacy at the dose and duration used here has not been established, and garlic intake is considered an adjunctive, hypothesis-generating component of this approach.
Rule 12: Non-Caloric Herbal Preparations in the Fasted State. A home-made herbal beverage consisting of warm water, ginger, turmeric, lemon, and black pepper was consumed as an adjunctive zero-calorie beverage approximately 30 minutes after garlic ingestion. An alternative preparation using warm water, lemon, Ceylon cinnamon, and salt was also used intermittently.
Rule 13: Sleep Regularity. The subject maintained a consistent sleep schedule, targeting 7-8 hours of nightly sleep with a regular bedtime.
Rule 14: Temporary Suspension of Caffeine (Weeks 4-15). The subject discontinued caffeine until Week 15. In individuals with T2DM, caffeine intake has been shown, in some studies, to impair glucose tolerance and worsen glycemic responses15.
Week 16–44: Maintenance
By Week 15, laboratory testing showed HbA1c had decreased to 5.3%, within the non-diabetic range. From Week 16 onward, the lifestyle approach transitioned to a maintenance phase focused on sustaining metabolic stability.
Rule 15: Structured Caffeine Reintroduction (Week 16+). Caffeine was reintroduced in a structured manner, with the subject consuming one cup of black coffee during the fasted state and a second cup with unsweetened almond milk after the first meal. This staged approach allowed the subject to qualitatively note caffeine’s perceived effects on fasting and postprandial energy patterns.
Rule 16: Planned Dietary Deviations During Maintenance. After Week 15, the subject allowed himself one high-glycemic meal per month. Absence of prolonged postprandial fatigue and timely return to baseline energy levels were noted as qualitative, non-diagnostic observations of perceived metabolic resilience rather than objective measures of insulin sensitivity.
| Component | Rule(s) | Details | Phase |
| Example Breakfast | 1,2,4,5 | Sauteed Spinach & Mushrooms in Avocado Oil, 2 eggs (boiled, fried or omelette), ½ avocado, seasoned with Salt, Pepper, Chilli flakes and Lemon | Weeks 1-3 only; no breakfast Weeks 4+ (fasting window) |
| Example Lunch | 1,2,4,5 | Salad Bar: Mixed greens, Oven-roasted vegetables (broccoli, Brussels sprouts, green beans), 2 oz Grilled chicken and 4 oz baked salmon with Tabasco; 3 oz walnuts, 8–10 blueberries. Vegetables consumed before protein per Rule 4. | Weeks 1–44; first meal of day from Week 4 onward (~12 PM) |
| Example Dinner | 1,2,4,5 | 1 Small Cucumber with 1 tbsp hummus; two pan-roasted vegetables (e.g., cabbage, zucchini, eggplant, onions, tomatoes); 4 oz chicken or 6 oz steelhead or 1 cup lentils/chickpeas; 1-2 oz walnuts; ¼ cup berries with 2 oz plain yogurt. Vegetables consumed first. | Weeks 1–44; ~7 PM from Week 4 onward |
| Moderation – reintroduced foods | 2,16 | Once weekly at lunch (after vegetables): 2 idlies, or 1 dosa, or 1 chickpea/besan-batter crepe. 1 tsp sriracha with eggs 2–3×/week. One high-glycemic meal per month (e.g., gluten-free pizza). | Week 16–44 only |
| Fasting Pattern | 9,10 | 16-hour daily fast; feeding window 12 PM – 7 PM. One OMAD (one meal a day) day per week at 6 PM. | Weeks 4–44 |
| Morning Routine (Weeks 1-3) | 6,7,8 | 5:00–6:00 AM: treadmill walk (3.5–4 mph). 15-minute postprandial walks after lunch and dinner. Resistance training 3×/week (~15-20 min). | Weeks 1–3 |
| Morning Routine (Weeks 4-44) | 6,7,8,11,12 | 5:15 AM: crush garlic (rest 10 min). 5:15–5:25 AM: resistance bands (set 1, ~10 min, while garlic rests). 5:25 AM: consume garlic with 2 oz water. 5:25–6:10 AM: resistance bands (set 2) + brisk treadmill walk (3.5–4 mph) for ~30 min. 6:10 AM: warm water with lemon, ginger, turmeric, black pepper. 8:00 AM: warm water with cinnamon and salt (fasted). 15-minute postprandial walks after lunch and dinner; additional resistance/treadmill session at 6:15 PM 3×/week. | Weeks 4–44; resistance training suspended Weeks 23–33 due to injury |
| Caffeine | 14,15 | Weeks 1–3: 12 oz latte (sugar-free, due to Rule 1). Weeks 4–15: suspended. Week 16+: 12 oz black Americano (double shot) at 7:30 AM in fasted state; 8–12 oz latte with unsweetened almond milk and non-glycemic sweetener at ~1 PM. | Weeks 1–3: (sugar-free); Weeks 4–15: no caffeine; Week 16+: structured reintroduction |
Layer 2: Behavioral & Technological Support
Layer 2 incorporated behavioral and technological components for sustained adherence35. Three psychosocial factors supported adherence: perceived health risk (acute blurred vision and strong family history of cardiometabolic disease), self-efficacy reinforced by understanding of the scientific rationale36, and family accountability through shared meal planning, and daily routines, which has been linked to improved outcomes in chronic disease management37.
To further support adherence, the subject utilized a custom chatbot configured within a commercially available AI platform (ChatGPT, OpenAI), with the Insulight protocol rules as its knowledge base. This non-clinical tool provided real-time dietary compatibility feedback through text queries or photographs of ingredient labels, for example, verifying whether items like hummus, or corn were protocol-compatible. The application did not collect physiological data, offer medical advice, or replace clinical judgment, and was used solely to reduce decision uncertainty in complex food environments without replacing independent judgment or clinical oversight38.
The tool was developed by the author – a high-school student at the time of this study – for personal adherence support and to assist the subject; no commercial interest, trademark, copyright or independent application exists in connection with it at the time of submission. The author owns the underlying protocol rules but has no ownership of the AI platform itself. The author intends to make a version of this tool available to nonprofit organizations and individuals in the future, with a free-tier for basic use and an optional cost-recovery tier to offset translation and API expenses for more intensive usage; no such arrangement currently exists. Usage was informal and conversational; structured logs were not retained, though interaction records are available within the platform.
The behavioral and technological elements described here were supportive in nature and did not constitute therapeutic or clinical management.
Data Collection, Variables, Measurements
Primary measures included Hemoglobin A1c (HbA1c), fasting plasma glucose, body weight, serum triglycerides, high-density lipoprotein (HDL) cholesterol and low-density lipoprotein (LDL). Secondary outcome measures included hepatic enzymes (ALT and AST). Data were collected from clinical bloodwork and office visits at three time points: baseline (November 7, 2024), 16 weeks (February 24, 2025), and 44 weeks (September 8, 2025). For the purposes of this report, remission is defined per the 2021 ADA consensus criteria as an HbA1c below 6.5% measured at least three months after cessation of glucose-lowering pharmacotherapy39. All laboratory values used standard clinical units (mg/dL, U/L, and %), and were cross-referenced with physician records to ensure data integrity (Table 1).
Adherence was tracked daily, for each rule, via participant self-report in Excel. Weekly adherence percentages were computed by normalizing frequency counts against rule-specific weekly targets such as 7-day target for diet and hydration rules (Rules 1–5), a 5-day target for walking (Rule 6), 7-day target for postprandial activity (Rule 7), and 3-day target for resistance training (Rule 8), 7-day target for time-restricted eating & recovery rules (Rules 9, and 11–14), and a one-day target for Rule 10. Any adherence exceeding weekly targets (e.g., walking seven instead of five days per week) was capped at 100%. As with all self-reported adherence data, these records are subject to social desirability bias and cannot be independently verified. The weekly percentages derived from these logs should be interpreted as descriptive and exploratory estimates rather than validated measures of adherence.
Daily and weekly adherence data were compared across two time-intervals: Weeks 1–15 (the period between the diagnosis of T2DM with HbA1c of 12.0% to the first clinical follow-up in Week 15 where HbA1c decreased to 5.3% entering the non-diabetic range), and Weeks 16–44 (representing the maintenance phase). The subject incurred a musculoskeletal injury in Week 23, and therefore, the impact of this event on adherence and biological markers was also analyzed.
Ethical Consideration And Participant Consent
The subject provided written informed consent to participate in this single-patient observational case study and to the use of anonymized data for research purposes. Medical care was not withheld, and standard pharmacological treatment remained available throughout the observation period. The investigator maintained a non-interventional, observational role throughout; no clinical decisions were directed or influenced by the investigator, and all medical care was provided solely by the subject’s primary care physician. Formal IRB review was not required for this case report. Under federal research regulations (45 CFR 46), case reports involving a single patient with informed consent are generally exempt from IRB oversight when the primary intent is clinical documentation and hypothesis generation rather than the systematic collection of data intended to produce generalizable knowledge40. This report was conducted in that spirit. The subject reviewed and approved the final manuscript prior to submission.
The investigator is a family member of the subject. This relationship may introduce bias in observation, data interpretation, and reporting, as the investigator has a personal interest in the subject’s health outcomes. Mitigating factors include: all laboratory values were obtained from independent clinical records and cross-referenced with the subject’s physician to confirm accuracy; no clinical decisions were directed or influenced by the investigator; and the subject independently pursued the lifestyle approach under physician supervision. These mitigations reduce but do not eliminate the risk of observer bias, and readers should interpret the findings accordingly.
The Insulight Protocol name was coined by the investigator. The AI-based informational tool described in this report was developed by the investigator as a custom chatbot configuration within a commercially available platform (ChatGPT, OpenAI). At the time of submission, no trademark, copyright, commercial arrangement, or independent application exists in connection with the protocol or tool. The investigator intends to make a version of the tool available to nonprofit organizations and individuals in the future; no such arrangement currently exists. As the author of the Insulight Protocol, the investigator (a high school student at the time of this study) has a reputational interest in its scientific reception; readers should weigh this accordingly. No external funding was received for this work, and no other conflicts of interest are declared.
Given the severity of baseline hyperglycemia and the decision to defer pharmacotherapy, a safety monitoring framework was established at the outset. The subject and his physician agreed that pharmacotherapy would be initiated immediately if any of the three presenting symptoms — blurred vision, chronic polydipsia (thirst), or palmar pruritus (itchy palms) — recurred, or if metabolic markers failed to improve within three months. Clinical follow-up was scheduled at three months but conducted at Week 16 due to a travel commitment, with no interim physician visits or home glucose monitoring performed between diagnosis and that Week 16 laboratory assessment. The subject did not experience recurrence of any presenting symptoms during the observation period.
Results
Metabolic And Glycemic Trends
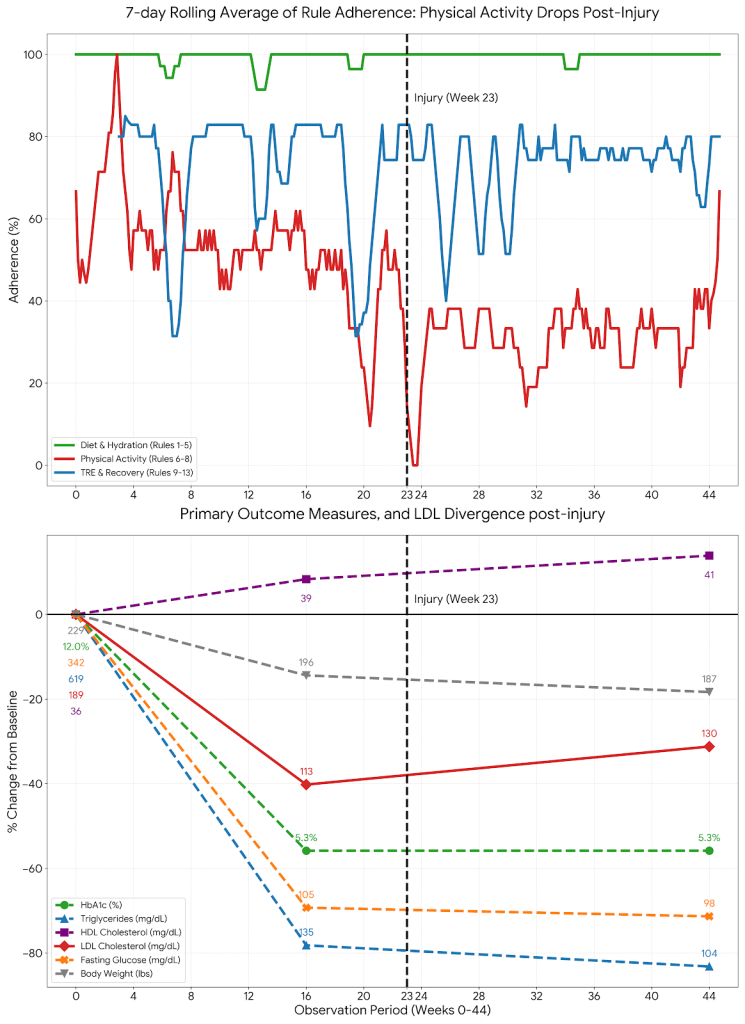
After approximately 16 weeks, changes in glycemic control and cardiometabolic markers were observed. HbA1c decreased from 12.0% at baseline to 5.3% at the first follow-up (16 weeks). During the same period, body weight decreased from 229 lbs to 196 lbs representing a 14% drop against baseline. At the 44-week follow-up, HbA1c remained 5.3% with body weight decreasing to 187 lbs, representing an overall reduction of 42 lbs in 44 weeks.
Serum triglyceride levels declined from 619 mg/dL at baseline to 135 mg/dL at 16 weeks and to 104 mg/dL at 44 weeks (reference <150 mg/dL). Fasting plasma glucose decreased from 342 mg/dL at baseline to 105 mg/dL at 16 weeks and to 98 mg/dL at 44 weeks. HDL cholesterol levels increased from 36 mg/dL (baseline) to 41 mg/dL over the 44-week period. LDL cholesterol decreased from 189 mg/dL (Direct LDL) at baseline to 113 mg/dL at 16 weeks, followed by an increase to 130 mg/dL at 44 weeks, approaching the upper limit of the optimal range. This elevation coincided with a self-reported musculoskeletal injury and subsequent reduction in volume of physical activity, specifically Rule 8 (resistance training), between Weeks 23 and 44.
Secondary laboratory measures also showed changes in hepatic markers. Serum alanine aminotransferase (ALT) decreased from 39 U/L at baseline to 12 U/L at 16 weeks and to 11 U/L at 44 weeks. Aspartate aminotransferase (AST) declined from 27 U/L at baseline to 15 U/L at 16 weeks and to 13 U/L at 44 weeks. Both values declined over the observation period and remained within laboratory-reference ranges throughout.
Longitudinal Laboratory Findings
The following table summarizes the longitudinal clinical markers for the subject over the observation period. Primary metabolic markers improved substantially within 16 weeks, and remained stable or continued to improve at the 44-week follow-up, with the exception of LDL cholesterol, which is discussed below.
| Component | Baseline (Nov 2024) | First Follow-up (Feb 2025) | Maintenance (Sept 2025) | Reference Range |
| Hemoglobin A1c | 12.0% | 5.3% | 5.3% | <5.7% |
| Fasting Glucose | 342 mg/dL | 105 mg/dL | 98 mg/dL | 60–99 mg/dL |
| Triglycerides | 619 mg/dL | 135 mg/dL | 104 mg/dL | 0–150 mg/dL |
| HDL Cholesterol | 36 mg/dL | 39 mg/dL | 41 mg/dL | >40 mg/dL |
| LDL Cholesterol | Direct LDL: 189 mg/dL | 113 mg/dL | 130 mg/dL | 0-129 mg/dL |
| ALT | 39 U/L | 12 U/L | 11 U/L | 0–55 U/L |
| AST | 27 U/L | 15 U/L | 13 U/L | 5–34 U/L |
| Body Weight | 229 lbs | 196 lbs | 187 lbs | N/A |
Adherence To The Structured Lifestyle Approach
Mean adherence across rules was approximately 90% during Weeks 1–15 (foundation and optimization phases), 76% during Weeks 16–44 (maintenance phase), and 82% across the entire observation period, Weeks 1–44. Adherence was consistently high for core dietary modification and hydration rules (rules 1-5), and time-restricted eating and recovery rules (rules 9-14), while adherence was lower for physical-activity rules (rules 6-8), especially postprandial movement (rule 7) and resistance training (rule 8), during the maintenance phase. A musculoskeletal injury in Week 23 coincided with low adherence for physical activity rules, particularly resistance training.
| Category | Rule | Adherence% | ||||
| Wks. 1-15 | Wks. 16-44 | Wks. 1-44 | ||||
| Dietary Modification and Hydration (Wks. 1-44) | Rule 01: Elimination of Sugars, Refined Carbohydrates and Gluten | 100% | 99.5% | 99.7% | ||
| Rule 02: Strict Restriction of Glycemic Load (e.g., potatoes, carrots, corn) | 100% | 100% | 100% | |||
| Rule 03: Hydration | 97% | 99.5% | 99% | |||
| Rule 04: Fiber-first Food Intake During Meals | 98% | 100% | 99% | |||
| Rule 05: Eliminate Snacking | 100% | 100% | 100% | |||
| Physical Activity (Wks. 1-44) | Rule 06: Walk 10K Steps per Day (five days per week) | 100% | 48% | 66% | ||
| Rule 07: Post Prandial Movement and Activation of the Soleus Muscle | 36% | 17% | 24% | |||
| Rule 08: Low-to-Moderate Resistance Training | 87% | 21% | 43% | |||
| Time-Restricted Eating and Metabolic Recovery (Wks. 4-44) | Rule 09: Time-Restricted Eating (16-hour fast) | 100% | 100% | 100% | ||
| Rule 10: Weekly Metabolic Challenge (One-Meal-A-Day) | 83% | 55% | 63% | |||
| Rule 11: Garlic Intake in Fasted State | 76% | 79% | 78% | |||
| Rule 12: Non-Caloric Herbal Preparations in the Fasted State | 81% | 74% | 76% | |||
| Rule 13: Sleep Regularity | 95% | 99% | 98% | |||
| Rule 14: Temporary Suspension of Caffeine (Weeks 4-15) | 100% | n/a | 100% | |||
| Maintenance (Wks. 16-44) | Rule 15: Structured Caffeine Reintroduction (Week 16+) | n/a | 100% | 100% | ||
| Rule 16: Planned Dietary Deviations During Maintenance | n/a | n/a | n/a | |||
| Overall Adherence% | 90% | 76% | 82% | |||
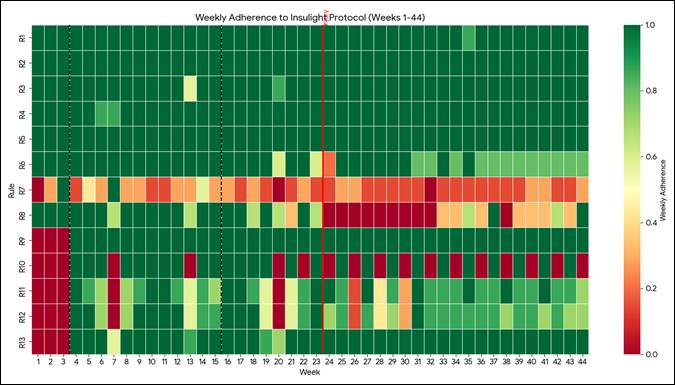
Figure 2 helps visualize the impact of a musculoskeletal injury at Week 23 through a temporal adherence heatmap. Rules 9–13 (Time-Restricted Eating and Recovery) were visually suppressed (set to null) during the foundation phase (Weeks 1–3) to accurately reflect that these components were not yet active in the subject’s adoption of the lifestyle approach. Rule 14 (caffeine suppression rule) was excluded from this analysis as it was a temporary rule, and Rules 15-16 were excluded because they applied only to the maintenance phase.
Figure 3 visualizes the primary outcome measures (HbA1c, fasting glucose, HDL, LDL Triglycerides and Body Weight) against the 7-day rolling average of daily adherence scores across three categories of rules: Dietary Modification and Hydration rules, Physical Activity and Time-Restricted Eating and Recovery. It also delineates the high-fidelity adherence during the foundation and optimization phases from the divergence observed following the musculoskeletal injury at Week 23.
Discussion
This observational case study describes substantial improvement in severe hyperglycemia in a subject who undertook an intensive lifestyle approach without pharmacological therapy. Over approximately 16 weeks, the subject’s HbA1c decreased from 12.0% to 5.3%, with sustained changes in glycemic and selected cardiometabolic markers observed at 44 weeks. These outcomes were accompanied by reductions in body weight, serum triglycerides, and hepatic enzyme levels, alongside an increase in HDL cholesterol, reflecting changes in metabolic markers beyond glycemic control. It is not possible to rule out the contribution of other factors, including natural disease course, regression to the mean, or unmeasured behavioral or physiological changes.
During the maintenance phase, LDL cholesterol declined substantially by 16 weeks but increased to 130 mg/dL by 44 weeks toward the upper limit of the near optimal range. This change coincided with a musculoskeletal injury at approximately Week 23 that resulted in a 10-week pause in resistance training, as well as a marked reduction in adherence to the walking target of 10,000 steps five times per week. Notably, dietary adherence remained consistently high throughout this period, while resistance training volume dropped substantially, and the increase in LDL coincided with this shift. Causal attribution cannot be made from a single observation with only two post-baseline LDL measurements and no detailed dietary data for that specific period; however, this temporal pattern is consistent with prior literature suggesting that lipid profiles may be sensitive to resistance training volume and sustained physical activity14.
Although limited by its single-subject design, this case is notable due to both the severity of baseline hyperglycemia and the subject’s demographic profile. South Asian populations face disproportionate metabolic risk, often at lower BMI thresholds than Western populations41. These findings raise the possibility that structured lifestyle approaches may be particularly relevant for such populations, though confirmation in larger studies is needed.
The metabolic improvements observed in this case coincided with coordinated dietary, activity and meal-timing modifications, along with adjunctive phytochemical intake. The combined pattern is consistent with reduced insulin demand and altered glucose disposal across multiple pathways. The early adoption of dietary modification and physical activity (Weeks 1–3) preceded the introduction of time-restricted eating and extended fasting. This sequencing is consistent with prior literature suggesting that cumulative insulin exposure may decrease over time and that lipolysis and cellular recycling processes such as autophagy may be supported27, though neither was directly measured in this case. Several components may have acted complementarily to one another. For example, postprandial walking and consuming fiber before proteins and carbohydrates may have worked by blunting sugar spikes after meals, though through different mechanisms6,12,13. Resistance training may have helped preserve skeletal muscle mass during weight loss, which in turn may have supported the body’s ability to clear glucose from the blood stream28. Collectively, these interactions are consistent with the hypothesis that glycemic trajectories may depend on both the intensity of lifestyle modification and integration across metabolic pathways. It should be noted that insulin resistance, β-cell function, ectopic fat distribution, and autophagy were not directly measured in this case and remain speculative as explanatory mechanisms.
The lifestyle approach described in this case was intensive, reflecting the subject’s markedly elevated baseline HbA1c and the decision to defer pharmacotherapy pending metabolic improvement. Given the severity of hyperglycemia, the subject elected to pursue an intensive lifestyle strategy as an initial approach to address short-term metabolic risk, with pharmacotherapy remaining available if needed42. However, this level of rigor may not be required for individuals with milder dysglycemia or prediabetes. Several components described in this case, such as phytochemical intake, OMAD, or structured postprandial movement, may be more appropriately applied in a modular or graduated manner depending on baseline metabolic status. Future research could explore whether tiered implementations of similar lifestyle approaches, emphasizing core dietary and activity principles while introducing more demanding practices selectively, are feasible and effective. Such flexibility may enhance scalability while preserving a focus on insulin dynamics, should similar approaches be validated in broader populations.
Real-world food environments present practical challenges. Restaurant and café settings often lack transparency regarding added sugars, refined starches, and hidden glycemic agents, complicating adherence even when “healthy” options are perceived to be available. Coffee shops frequently market sugar-containing milk substitutes and sweeteners as health-oriented products, increasing the risk of unintended glycemic exposure. A custom chatbot configured with the Insulight Protocol rules may have helped translate complex nutritional information into day-to-day choices, potentially reducing decision uncertainty in these environments38. These challenges illustrate that modern food environments may pose significant cognitive demands for individuals following low-insulin dietary patterns, highlighting the potential value of simple heuristics (e.g., “no sugar, no starch, no gluten”) and real-time informational tools43.
The nonpharmacological approach described in this case may carry clinical risk if undertaken without appropriate medical supervision. Accordingly, the findings reported here should not be interpreted as treatment recommendations. Any similar lifestyle approaches should be pursued with physician oversight and a clearly defined contingency plan for pharmacologic therapy. Notably, the subject had not yet initiated pharmacotherapy at the time he adopted this approach which simplified the initial adoption of the rules. For individuals already undergoing pharmacotherapy, the dietary and lifestyle changes described here – including elimination of sugars and refined carbohydrates, low-glycemic eating, postprandial movement, resistance training and time-restricted eating – may lower blood glucose substantially, potentially triggering hypoglycemia if medication doses are not adjusted accordingly5,44. Adjustments to medication in this context require close physician oversight, and any similar lifestyle modification should not be undertaken by individuals on glucose-lowering therapy without prior discussion with their healthcare provider.
Together, these observations suggest that a structured lifestyle approach to T2DM management may benefit from both evidence-based physiological rules and intentional behavioral support systems designed to navigate real-world adherence challenges.
Limitations And Unmeasured Outcomes
As a single-subject observational study, the findings in this report cannot be generalized without further validation. Direct measures of insulin resistance, such as HOMA-IR45, or insulin-assisted glucose tolerance testing were not performed; instead, improvements were inferred through glycemic and lipid markers. Additionally, qualitative tracking of diet and activity introduces potential reporting bias, though the 44-week laboratory trends suggest metabolic stability.
Implications, Future Gaps And Future Directions
This case highlights several areas for future investigation, including the potential applicability of structured nonpharmacological approaches in resource-limited settings where continuous glucose monitoring remains cost-prohibitive4.
Current clinical practice often treats dysglycemia as a single continuum; however, impaired fasting glucose (IFG) and impaired glucose tolerance (IGT) arise from distinct pathophysiological mechanisms involving hepatic versus peripheral insulin resistance2,32. How different components of this approach may affect each phenotype differently remains unclear. For example, fasted-state and circadian strategies may preferentially influence hepatic glucose output, whereas postprandial movement may play a larger role in peripheral glucose clearance9,12,13. Studies stratifying participants by IFG and IGT status could clarify whether certain components should be emphasized or sequenced differently.
Given the prevalence of the Thin Outside Fat Inside (TOFI) phenotype in South Asian populations – characterized by disproportionate visceral and ectopic fat accumulation despite relatively modest BMI values32,46 – future studies should examine how this fat responds to intensive lifestyle approaches, ideally incorporating imaging modalities such as MRI or DXA41,47,48. Whether a post-intervention HbA1c of 5.3% confers equivalent long-term risk reduction in South Asian populations also remains uncertain, as diagnostic and remission thresholds are largely derived from Western cohorts1,49.
Finally, the magnitude and pace of improvement observed in this case raise questions about inter-individual variability in response. Identifying biological or metabolic factors associated with faster response, such as insulin secretory reserve or inflammatory status, could support more personalized intervention strategies for high-risk individuals. Together, these gaps highlight the need for controlled, mechanistic, and population-specific studies to evaluate intensive lifestyle approaches more broadly. Future research could also attempt to isolate the contribution of core components, such as dietary modification and physical activity, from adjunctive components such as phytochemical intake, specific hydration practices, and circadian strategies, to identify which elements are essential and which may be supplementary.
Conclusion
This case report describes substantial improvement in severe hyperglycemia in a subject with T2DM who undertook an intensive lifestyle approach without pharmacological therapy. Over approximately 16 weeks, HbA1c decreased from 12.0% to 5.3%, alongside reductions in body weight, serum triglycerides, and hepatic enzyme levels, with these changes sustained at 44 weeks. The findings are consistent with the hypothesis that coordinated lifestyle components addressing dietary glycemic load, skeletal muscle glucose uptake, meal timing, and recovery behaviors may help reduce chronic hyperinsulinemia and support metabolic homeostasis in this case. The longitudinal adherence analysis further suggests that dietary consistency may be more readily sustained than physical activity targets, and that reductions in activity volume coincided with changes in lipid profiles even when dietary adherence remains high, though causal attribution cannot be made from a single observation.
The lifestyle changes described here relied on accessible, non-proprietary strategies rather than continuous pharmacological escalation. Further investigation is needed to assess generalizability, directly quantify changes in insulin resistance, and define optimal approach intensity across different risk profiles. That said, this case is consistent with the possibility that, in selected individuals, T2DM-associated hyperglycemia may represent a metabolically modifiable state when upstream insulin dynamics are systematically addressed.
Acknowledgements
I am grateful to Dr. Brydie R. Huckestein (PhD, Microbiology and Immunology), St. Jude Children’s Research Hospital for her support and feedback during the preparation of this manuscript.
Supplementary Information
References
- International Diabetes Federation. IDF Diabetes Atlas, 11th edn. https://diabetesatlas.org/ 2025 [↩] [↩]
- Global Estimates of Impaired Fasting Glucose and Impaired Glucose Tolerance. https://diabetesatlas.org/resources/idf-diabetes-atlas-2025/ 2024 [↩] [↩]
- R. Taylor. Banting memorial lecture 2012 reversing the twin cycles of type 2 diabetes. Diabetic Medicine. Vol. 30, pg. 267–275, 2013 https://doi.org/10.1111/dme.12039 [↩] [↩] [↩] [↩]
- M. E. J. Lean, W. S. Leslie, A. C. Barnes, N. Brosnahan, G. Thom, L. McCombie, C. Peters, S. Zhyzhneuskaya, A. Al-Mrabeh, K. G. Hollingsworth, A. M. Rodrigues, L. Rehackova, A. J. Adamson, F. F. Sniehotta, J. C. Mathers, H. M. Ross, Y. McIlvenna, P. Welsh, S. Kean, I. Ford, A. McConnachie, C.-M. Messow, N. Sattar, R. Taylor. Durability of a primary care-led weight-management intervention for remission of type 2 diabetes: 2-year results of the direct open-label, cluster-randomised trial. The Lancet Diabetes & Endocrinology. Vol. 7, pg. 344–355, 2019 https://doi.org/10.1016/S2213-8587(19)30068-3 [↩] [↩] [↩] [↩]
- American Diabetes Association Professional Practice Committee, N. A. ElSayed, G. Aleppo, R. R. Bannuru, D. Bruemmer, B. S. Collins, L. Ekhlaspour, J. L. Gaglia, M. E. Hilliard, E. L. Johnson, K. Khunti, I. Lingvay, G. Matfin, R. G. McCoy, M. L. Perry, S. J. Pilla, S. Polsky, P. Prahalad, R. E. Pratley, A. R. Segal, J. J. Seley, R. C. Stanton, R. A. Gabbay. 9. pharmacologic approaches to glycemic treatment: standards of care in diabetes—2024. Diabetes Care. Vol. 47, pg. S158–S178, 2024 https://doi.org/10.2337/dc24-S009 [↩] [↩] [↩]
- A. P. Shukla, R. G. Iliescu, C. E. Thomas, L. J. Aronne. Food order has a significant impact on postprandial glucose and insulin levels. Diabetes Care. Vol. 38, pg. e98–e99, 2015 https://doi.org/10.2337/dc15-0429 [↩] [↩] [↩] [↩]
- D. S. Ludwig, C. B. Ebbeling. The carbohydrate-insulin model of obesity: beyond “calories in, calories out”. JAMA Internal Medicine. Vol. 178, pg. 1098, 2018 https://doi.org/10.1001/jamainternmed.2018.2933 [↩] [↩] [↩]
- K. Kempf, B. Rose, C. Herder, U. Kleophas, S. Martin, H. Kolb. Inflammation in metabolic syndrome and type 2 diabetes: impact of dietary glucose. Annals of the New York Academy of Sciences. Vol. 1084, pg. 30–48, 2006 https://doi.org/10.1196/annals.1372.012 [↩] [↩] [↩] [↩]
- R. De Cabo, M. P. Mattson. Effects of intermittent fasting on health, aging, and disease. New England Journal of Medicine. Vol. 381, pg. 2541–2551, 2019 https://doi.org/10.1056/NEJMra1905136 [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- J. Wang, X. Zhang, H. Lan, W. Wang. Effect of garlic supplement in the management of type 2 diabetes mellitus (t2dm): a meta-analysis of randomized controlled trials. Food & Nutrition Research. Vol. 61, pg. 1377571, 2017 https://doi.org/10.1080/16546628.2017.1377571 [↩] [↩] [↩]
- T. Arablou, N. Aryaeian, M. Valizadeh, F. Sharifi, A. Hosseini, M. Djalali. The effect of ginger consumption on glycemic status, lipid profile and some inflammatory markers in patients with type 2 diabetes mellitus. International Journal of Food Sciences and Nutrition. Vol. 65, pg. 515–520, 2014 https://doi.org/10.3109/09637486.2014.880671 [↩] [↩]
- E. A. Richter, M. Hargreaves. Exercise, glut4, and skeletal muscle glucose uptake. Physiological Reviews. Vol. 93, pg. 993–1017, 2013 https://doi.org/10.1152/physrev.00038.2012 [↩] [↩] [↩] [↩] [↩] [↩]
- L. DiPietro, A. Gribok, M. S. Stevens, L. F. Hamm, W. Rumpler. Three 15-min bouts of moderate postmeal walking significantly improves 24-h glycemic control in older people at risk for impaired glucose tolerance. Diabetes Care. Vol. 36, pg. 3262–3268, 2013 https://doi.org/10.2337/dc13-0084 [↩] [↩] [↩] [↩] [↩]
- W. L. Westcott. Resistance training is medicine: effects of strength training on health. Current Sports Medicine Reports. Vol. 11, pg. 209–216, 2012 https://doi.org/10.1249/JSR.0b013e31825dabb8 [↩] [↩] [↩] [↩]
- L. E. Robinson, S. Savani, D. S. Battram, D. H. McLaren, P. Sathasivam, T. E. Graham. Caffeine ingestion before an oral glucose tolerance test impairs blood glucose management in men with type 2 diabetes. The Journal of Nutrition. Vol. 134, pg. 2528–2533, 2004 https://doi.org/10.1093/jn/134.10.2528 [↩] [↩]
- S. Prasad, B. B. Aggarwal. Turmeric, the golden spice: from traditional medicine to modern medicine. in Herbal Medicine: Biomolecular and Clinical Aspects (eds I. F. F. Benzie & S. Wachtel-Galor) CRC Press/Taylor & Francis, Boca Raton (FL), 2011 [↩] [↩]
- P. Ranasinghe, P. Galappaththy, G. R. Constantine, R. Jayawardena, H. D. Weeratunga, S. Premakumara, P. Katulanda. Cinnamomum zeylanicum (ceylon cinnamon) as a potential pharmaceutical agent for type-2 diabetes mellitus: study protocol for a randomized controlled trial. Trials. Vol. 18, pg. 446, 2017 https://doi.org/10.1186/s13063-017-2192-0 [↩]
- S. Tangvarasittichai. Oxidative stress, insulin resistance, dyslipidemia and type 2 diabetes mellitus. World Journal of Diabetes. Vol. 6, pg. 456, 2015 https://doi.org/10.4239/wjd.v6.i3.456 [↩] [↩]
- K. Fujihira, Y. Hamada, T. Yanaoka, R. Yamamoto, K. Suzuki, M. Miyashita. The effects of water temperature on gastric motility and energy intake in healthy young men. European Journal of Nutrition. Vol. 59, pg. 103–109, 2020 https://doi.org/10.1007/s00394-018-1888-6 [↩] [↩]
- J. L. Sievenpiper, V. Vuksan, E. Y. Wong, R. A. Mendelson, C. Bruce-Thompson. Effect of meal dilution on the postprandial glycemic response: implications for glycemic testing. Diabetes Care. Vol. 21, pg. 711–716, 1998 https://doi.org/10.2337/diacare.21.5.711 [↩] [↩]
- J. J. Joseph, S. H. Golden. Cortisol dysregulation: the bidirectional link between stress, depression, and type 2 diabetes mellitus. Annals of the New York Academy of Sciences. Vol. 1391, pg. 20–34, 2017 https://doi.org/10.1111/nyas.13217 [↩] [↩] [↩]
- K. Spiegel, E. Tasali, P. Penev, E. V. Cauter. Brief communication: sleep curtailment in healthy young men is associated with decreased leptin levels, elevated ghrelin levels, and increased hunger and appetite. Annals of Internal Medicine. Vol. 141, pg. 846–850, 2004 https://doi.org/10.7326/0003-4819-141-11-200412070-00008 [↩] [↩]
- C. S. Johnston, C. A. Gaas. Vinegar: medicinal uses and antiglycemic effect. MedGenMed: Medscape General Medicine. Vol. 8, pg. 61, 2006 [↩] [↩]
- D. Freitas, F. Boué, M. Benallaoua, G. Airinei, R. Benamouzig, S. Le Feunteun. Lemon juice, but not tea, reduces the glycemic response to bread in healthy volunteers: a randomized crossover trial. European Journal of Nutrition. Vol. 60, pg. 113–122, 2021 https://doi.org/10.1007/s00394-020-02228-x [↩] [↩]
- R. E. Patterson, G. A. Laughlin, A. Z. LaCroix, S. J. Hartman, L. Natarajan, C. M. Senger, M. E. Martínez, A. Villaseñor, D. D. Sears, C. R. Marinac, L. C. Gallo. Intermittent fasting and human metabolic health. Journal of the Academy of Nutrition and Dietetics. Vol. 115, pg. 1203–1212, 2015 https://doi.org/10.1016/j.jand.2015.02.018 [↩] [↩]
- E. F. Sutton, R. Beyl, K. S. Early, W. T. Cefalu, E. Ravussin, C. M. Peterson. Early time-restricted feeding improves insulin sensitivity, blood pressure, and oxidative stress even without weight loss in men with prediabetes. Cell Metabolism. Vol. 27, pg. 1212-1221.e3, 2018 https://doi.org/10.1016/j.cmet.2018.04.010 [↩] [↩]
- S. D. Anton, K. Moehl, W. T. Donahoo, K. Marosi, S. A. Lee, A. G. Mainous, C. Leeuwenburgh, M. P. Mattson. Flipping the metabolic switch: understanding and applying the health benefits of fasting. Obesity. Vol. 26, pg. 254–268, 2018 https://doi.org/10.1002/oby.22065 [↩] [↩] [↩] [↩]
- S. M. Phillips, R. A. Winett. Uncomplicated resistance training and health-related outcomes: evidence for a public health mandate. Current Sports Medicine Reports. Vol. 9, pg. 208–213, 2010 https://doi.org/10.1249/JSR.0b013e3181e7da73 [↩] [↩]
- M. R. Hunter, B. W. Gillespie, S. Y.-P. Chen. Urban nature experiences reduce stress in the context of daily life based on salivary biomarkers. Frontiers in Psychology. Vol. 10, pg. 722, 2019 https://doi.org/10.3389/fpsyg.2019.00722 [↩] [↩]
- L. Besedovsky, T. Lange, M. Haack. The sleep-immune crosstalk in health and disease. Physiological Reviews. Vol. 99, pg. 1325–1380, 2019 https://doi.org/10.1152/physrev.00010.2018 [↩]
- I. Torsdottir, H. Andersson. Effect on the postprandial glycaemic level of the addition of water to a meal ingested by healthy subjects and type 2 (non-insulin-dependent) diabetic patients. Diabetologia. Vol. 32, pg. 231–235, 1989 https://doi.org/10.1007/BF00285289 [↩]
- R. Unnikrishnan, R. M. Anjana, V. Mohan. Diabetes in south asians: is the phenotype different? Diabetes. Vol. 63, pg. 53–55, 2014 https://doi.org/10.2337/db13-1592 [↩] [↩] [↩]
- Centers for Disease Control and Prevention. Adult BMI Categories. https://www.cdc.gov/bmi/adult-calculator/bmi-categories.html 2024 [↩]
- WHO Expert Consultation. Appropriate body-mass index for asian populations and its implications for policy and intervention strategies. The Lancet. Vol. 363, pg. 157–163, 2004 https://doi.org/10.1016/S0140-6736(03)15268-3 [↩]
- Adherence to long-term therapies: evidence for action. World Health Organization, Geneva, 2003 [↩]
- A. Bandura. Health functioning. in Self-efficacy: the exercise of control pg. 259–318, W.H. Freeman, New York, 1997 [↩]
- M. Reblin, B. N. Uchino. Social and emotional support and its implication for health: Current Opinion in Psychiatry. Vol. 21, pg. 201–205, 2008 https://doi.org/10.1097/YCO.0b013e3282f3ad89 [↩]
- B. Singh, T. Olds, J. Brinsley, D. Dumuid, R. Virgara, L. Matricciani, A. Watson, K. Szeto, E. Eglitis, A. Miatke, C. E. M. Simpson, C. Vandelanotte, C. Maher. Systematic review and meta-analysis of the effectiveness of chatbots on lifestyle behaviours. Npj Digital Medicine. Vol. 6, pg. 118, 2023 https://doi.org/10.1038/s41746-023-00856-1 [↩] [↩]
- M. C. Riddle, W. T. Cefalu, P. H. Evans, H. C. Gerstein, M. A. Nauck, W. K. Oh, A. E. Rothberg, C. W. Le Roux, F. Rubino, P. Schauer, R. Taylor, D. Twenefour. Consensus report: definition and interpretation of remission in type 2 diabetes. Diabetes Care. Vol. 44, pg. 2438–2444, 2021 https://doi.org/10.2337/dci21-0034 [↩]
- U.S. Department of Health and Human Services. Protection of Human Subjects. 45 C.F.R. § 46. https://www.hhs.gov/ohrp/regulations-and-policy/regulations/45-cfr-46/index.html 1991 [↩]
- A. Misra, U. Shrivastava. Obesity and dyslipidemia in south asians. Nutrients. Vol. 5, pg. 2708–2733, 2013 https://doi.org/10.3390/nu5072708 [↩] [↩]
- American Diabetes Association Professional Practice Committee, N. A. ElSayed, G. Aleppo, R. R. Bannuru, E. A. Beverly, D. Bruemmer, B. S. Collins, A. Darville, L. Ekhlaspour, M. Hassanein, M. E. Hilliard, E. L. Johnson, K. Khunti, I. Lingvay, G. Matfin, R. G. McCoy, M. L. Perry, S. J. Pilla, S. Polsky, P. Prahalad, R. E. Pratley, A. R. Segal, J. J. Seley, R. C. Stanton, R. A. Gabbay. 5. facilitating positive health behaviors and well-being to improve health outcomes: standards of care in diabetes—2024. Diabetes Care. Vol. 47, pg. S77–S110, 2024 https://doi.org/10.2337/dc24-S005 [↩]
- C. A. Monteiro, J. ‐C. Moubarac, G. Cannon, S. W. Ng, B. Popkin. Ultra‐processed products are becoming dominant in the global food system. Obesity Reviews. Vol. 14, pg. 21–28, 2013 https://doi.org/10.1111/obr.12107 [↩]
- E. R. Seaquist, J. Anderson, B. Childs, P. Cryer, S. Dagogo-Jack, L. Fish, S. R. Heller, H. Rodriguez, J. Rosenzweig, R. Vigersky. Hypoglycemia and diabetes: a report of a workgroup of the american diabetes association and the endocrine society. Diabetes Care. Vol. 36, pg. 1384–1395, 2013 https://doi.org/10.2337/dc12-2480 [↩]
- D. R. Matthews, J. P. Hosker, A. S. Rudenski, B. A. Naylor, D. F. Treacher, R. C. Turner. Homeostasis model assessment: insulin resistance and ?-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. Vol. 28, pg. 412–419, 1985 https://doi.org/10.1007/BF00280883 [↩]
- R. M. Anjana, R. Unnikrishnan, M. Deepa, R. Pradeepa, N. Tandon, A. K. Das, S. Joshi, S. Bajaj, P. K. Jabbar, H. K. Das, A. Kumar, V. K. Dhandhania, A. Bhansali, P. V. Rao, A. Desai, S. Kalra, A. Gupta, R. Lakshmy, S. V. Madhu, N. Elangovan, S. Chowdhury, U. Venkatesan, R. Subashini, T. Kaur, R. S. Dhaliwal, V. Mohan. Metabolic non-communicable disease health report of india: the icmr-indiab national cross-sectional study (icmr-indiab-17). The Lancet Diabetes & Endocrinology. Vol. 11, pg. 474–489, 2023 https://doi.org/10.1016/S2213-8587(23)00119-5 [↩]
- E. L. Thomas, J. A. Fitzpatrick, S. J. Malik, S. D. Taylor-Robinson, J. D. Bell. Whole body fat: content and distribution. Progress in Nuclear Magnetic Resonance Spectroscopy. Vol. 73, pg. 56–80, 2013 https://doi.org/10.1016/j.pnmrs.2013.04.001 [↩]
- E. L. Lim, K. G. Hollingsworth, B. S. Aribisala, M. J. Chen, J. C. Mathers, R. Taylor. Reversal of type 2 diabetes: normalisation of beta cell function in association with decreased pancreas and liver triacylglycerol. Diabetologia. Vol. 54, pg. 2506–2514, 2011 https://doi.org/10.1007/s00125-011-2204-7 [↩]
- Centers for Disease Control and Prevention. National Diabetes Statistics Report. https://www.cdc.gov/diabetes/php/data-research/index.html 2024 [↩]