Abstract
Adolescent mental health problems are major factors that impair academic achievement and quality of life. Emerging evidence suggests that the gut–brain axis plays a crucial role in regulating emotion and cognition. This study aimed to evaluate the multi-layered effects of probiotic intake on mental health through the gut–brain axis by integrating human surveys and cellular models. In a two-week randomized controlled trial, healthy adolescents (n=20) were assigned to probiotic or control groups, and changes in well-being (WHO-5), depression (PHQ-2), stress, and cognitive performance (Stroop, N-back) were assessed. In parallel, filtered supernatants from the same probiotic drink were applied to a Caco-2 Transwell intestinal epithelial model to measure transepithelial electrical resistance (TEER), FITC-dextran permeability, and tight junction protein expression (ZO-1, Claudin-1). Probiotic-conditioned medium collected from the basolateral chamber was subsequently applied to SH-SY5Y neuroblastoma cells, followed by RNA-seq-based gene ontology (GO) pathway analysis, BDNF expression by RT-qPCR, and cell viability by MTT assay. Probiotic intake was associated with increased well-being, reduced depressive and stress scores, and improved cognitive performance, alongside enhanced intestinal barrier function and upregulation of neuroplasticity-related pathways in neuronal cells. This integrated approach provides supportive in vitro mechanistic insights linking probiotic-derived signals to intestinal barrier function and neuronal responses.
Keyword: gut-brain axis, probiotics, Caco-2, SH-SY5Y, BDNF, RNA-seq, WHO-5, TEER, FITC-dextran
Introduction
Adolescence is a period of rapid physical and psychological development, making adolescents particularly vulnerable to mental health issues like depression and anxiety due to academic and social pressures1,2. These challenges not only compromise their quality of life but also negatively impact their academic achievement and social functioning3,4. Over the past few decades, the concept of the gut-brain axis, which suggests a direct influence of the gut microbiome on brain function, has gained prominence, leading to an increased interest in microbiome-based mental health intervention strategies5,6,7. The gut microbiome synthesizes precursors for neurotransmitters7 and produces metabolites such as short-chain fatty acids (SCFAs)8, which are vital for maintaining the integrity of the intestinal epithelial barrier9 and modulating systemic inflammatory responses10,11. In particular, probiotics are reported to increase the expression of key signaling molecules that regulate neuroplasticity, such as brain-derived neurotrophic factor (BDNF)12,13, through these mechanisms. However, most existing studies have focused on animal models or specific molecular pathways14,15, and research that simultaneously validates both clinical human effects and molecular mechanistic effects within a single, integrated framework is limited16,17.
Although accumulating evidence from animal models suggests that probiotics can influence stress-related behaviors and neuroplasticity18, many of these studies are conducted in disease or stress-induced models19, limiting their direct translation to healthy human populations. Human studies, particularly in adolescents, have largely relied on questionnaire-based outcomes without accompanying mechanistic validation20. Conversely, mechanistic studies often lack direct relevance to human behavioral data21. As a result, there remains a critical gap in research that simultaneously evaluates behavioral outcomes in healthy adolescents while providing supportive mechanistic insights using reproducible in vitro models. The present pilot study was designed to address this gap by integrating a randomized controlled human intervention with intestinal and neuronal cell-based assays.
This study was designed to bridge this gap by proposing a multi-layered research design. We aimed to (1) confirm the effects of a two-week probiotic beverage consumption on adolescent emotional and cognitive function through a human-based study, while also (2) mechanistically validating the effects of the same sample at the molecular level using a reproducible cell model. We hypothesized that a two-week probiotic beverage consumption would (1) improve emotional and cognitive function in humans, (2) strengthen the intestinal epithelial cell barrier, and (3) activate neuroplasticity-related signaling pathways in neuronal cells. The originality of this research lies in its ability to connect the phenomenon observed in human surveys with the mechanism demonstrated in cell models in a continuous research flow. This approach provides a new framework for comprehensively elucidating the effects of probiotic-based nutritional interventions on mental health and is expected to provide a crucial scientific foundation for future mental health promotion programs.
Methods
Human Study: Two-week Randomized Controlled Trial
Participants
Healthy adolescents aged 15–18 years were recruited with informed consent obtained from both participants and their guardians. Exclusion criteria included antibiotic use within the past month, chronic gastrointestinal or psychiatric disorders, and current use of psychotropic medications.
A total of 20 adolescents participated in the study (probiotic group: n = 10; control group: n = 10). The mean age of participants was 16.4 ± 0.8 years in the probiotic group and 16.6 ± 0.7 years in the control group, with no significant difference between groups (p = 0.58). Gender distribution was comparable between groups (probiotic: 5 males / 5 females; control: 4 males / 6 females; p = 0.67).
Study Design
Participants were randomly assigned to either a probiotic or control group. The probiotic group consumed one commercially available probiotic beverage daily for 14 days, whereas the control group received a placebo beverage matched for taste and caloric content but lacking live bacteria.
Randomization and Blinding
Participants were randomly assigned to either the probiotic or control group using a simple randomization procedure. Due to logistical constraints, the study employed a single-blind design, in which participants were blinded to group allocation, while investigators were not.
Intervention product detail
The intervention group consumed one commercially available probiotic beverage per day for 14 consecutive days. The control group received a placebo beverage matched for taste and caloric content but lacking live probiotic cultures.
Compliance tracking
Compliance with beverage consumption was monitored using daily self-reported intake logs completed by participants throughout the intervention period.
Survey and Cognitive Measures
Psychological and cognitive outcomes were evaluated at baseline and after the two-week intervention.
- Well-being: WHO-5 Well-Being Index
- Depression: Patient Health Questionnaire-2 (PHQ-2)
- Stress: Visual Analogue Scale (VAS, 0–100)
- Gastrointestinal symptoms: Self-reported bloating, abdominal pain, and stool form
- Sleep: Average daily sleep duration and subjective sleep quality
- Cognition: Stroop task (reaction time and error rate) and 2-back task (accuracy)
Statistical Analysis
Pre–post changes (Δ) were calculated within each group, and between-group comparisons were analyzed using independent t-tests or ANCOVA, with sleep, caffeine intake, and diet included as covariates. A significance level of p < 0.05 was applied.
Preparation of Probiotic Supernatants
The probiotic beverage was centrifuged at 4,000 × g for 15 min at 4 °C. The resulting supernatant was collected and sterilized through a 0.22 μm filter. Filtrates were diluted in serum-free DMEM to a final concentration of 5% (v/v) before application to Caco-2 monolayers. The filtered supernatant used in this study contains soluble components derived from the commercial probiotic beverage but does not contain live bacteria.
Caco-2 Intestinal Barrier Model
-Cell Culture and Differentiation
Caco-2 cells (human colorectal adenocarcinoma line, Korea Cell Line Bank) were seeded onto Transwell polyester membrane inserts (pore size 0.4 μm, growth area 1.12 cm², 12-well format) at a density of 3.0 × 10⁴ cells per insert. Cells were cultured for 14 days in DMEM supplemented with 10% fetal bovine serum (FBS), 1% non-essential amino acids, and 1% penicillin–streptomycin. Medium in both apical and basolateral chambers was replaced every 2–3 days until monolayer differentiation, confirmed by stable transepithelial electrical resistance (TEER) values exceeding 300 Ω·cm².
–Probiotic-treated Caco-2 cells and Collection of Conditioned Media
Following differentiation, sterilized probiotic supernatant diluted in serum-free DMEM was applied to the apical chamber. Fresh supernatant was replaced every 24 h, while the basolateral medium was exchanged every 48 h. After 48 h of treatment, basolateral conditioned media (CM) were collected, centrifuged at 1,000 × g for 10 min, and filtered (0.22 μm) to remove debris prior to neuronal assays.
-Barrier Integrity Assays
- TEER Measurement: TEER was measured using an EVOM2 voltohmmeter with STX2 electrodes. Resistance of blank inserts was subtracted, and TEER values were calculated as (R_sample – R_blank) × surface area (Ω·cm²).
- FITC-dextran Permeability Assay: FITC-dextran (4 kDa, 1 mg/mL) was added to the apical chamber, and basolateral samples were collected after 60 min. Fluorescence was measured at excitation 485 nm/emission 530 nm and expressed as relative flux compared to control.
- Immunofluorescence Staining: Cells were fixed with 4% paraformaldehyde, permeabilized with 0.1% Triton X-100, and blocked with 2% BSA. Primary antibodies against ZO-1 (1:500) and Claudin-1 (1:500) were applied, followed by Alexa Fluor 488/594–conjugated secondary antibodies. Nuclei were counterstained with DAPI, and images were acquired by confocal microscopy.
SH-SY5Y Neuronal Cell Assays
-Cell Culture and Treatment
SH-SY5Y human neuroblastoma cells were maintained in DMEM/F12 supplemented with 10% FBS and 1% penicillin–streptomycin. For experiments, cells were treated with 25% (v/v) conditioned media (control-CM or probiotic-CM) diluted in serum-free medium. Treatment durations were 6, 12, 24, and 48 h depending on the specific assay. All in vitro experiments were independently repeated at least three times using separate cell cultures.
-RNA-seq and Gene Ontology Analysis
Total RNA was extracted using TRIzol reagent, and integrity was confirmed (RIN > 8.0). Libraries were prepared and sequenced on an Illumina platform. Differential gene expression analysis was performed using the DESeq2 package in R. Raw count data were normalized using DESeq2’s default median-of-ratios method. Genes with low counts were filtered prior to analysis.
Differentially expressed genes (DEGs) were defined as those with an absolute log2 fold change ≥ 1 and an adjusted p-value (FDR q) < 0.05, corrected using the Benjamini–Hochberg method. Gene ontology (GO) enrichment analysis was conducted using the clusterProfiler package, with significance determined at FDR q < 0.05.
-RT-qPCR Validation
For validation, BDNF expression was quantified by RT-qPCR. cDNA was synthesized from 1 μg of RNA, and qPCR was performed with SYBR Green chemistry using GAPDH as the housekeeping gene. Relative expression levels were calculated using the ΔΔCt method. (BDNF_Forward : GAGCCCTGTATCAACCCAGA, BDNF_Reverse: TCAAATACCATGCCCCACCT)
-MTT Cell Viability Assay
SH-SY5Y cells were seeded in a 96-well plate and treated with conditioned culture medium for 24 hours. Time points of 6 hours, 12 hours, 24 hours and 48 hours post-treatment were established. MTT solution was added at a final concentration of 0.5 mg/mL and incubated for 4 h at 37 °C. Formazan crystals were dissolved in DMSO, and absorbance was measured at 570 nm. Cell viability was expressed as a percentage relative to cells treated with control-CM.
-Statistical analysis
Statistical analyses were performed using R and GraphPad Prism. Data distributions were assessed for normality using the Shapiro–Wilk test. For normally distributed data, parametric tests, including independent or paired t-tests and ANCOVA where appropriate, were applied. When normality assumptions were not met, non-parametric alternatives were considered.
To account for multiple hypothesis testing, correction procedures were applied where appropriate. For RNA-seq differential expression analysis, p-values were adjusted using the Benjamini–Hochberg false discovery rate (FDR) method, with genes considered significant at an adjusted p (q) < 0.05. For behavioral and cellular assays involving multiple outcome measures, results were interpreted with caution and are reported alongside exact p-values.
Data are presented as mean ± standard error of the mean (SEM), unless otherwise stated. Statistical significance was set at p < 0.05.
Results
1. Effects of a two-week probiotic intervention on emotional and cognitive outcomes in adolescents.
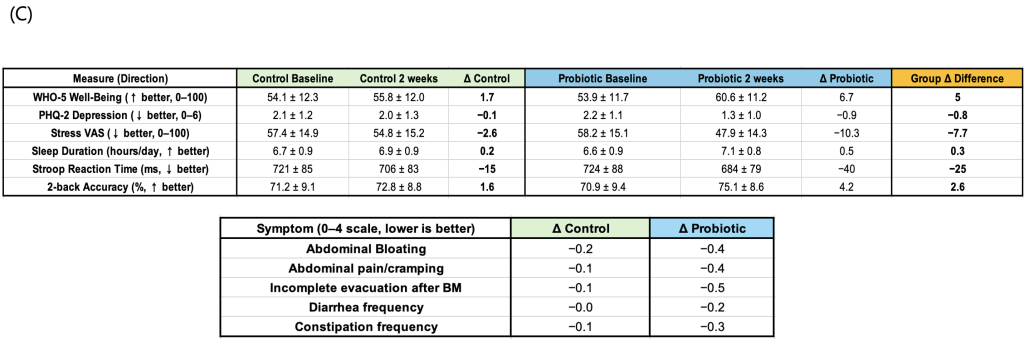
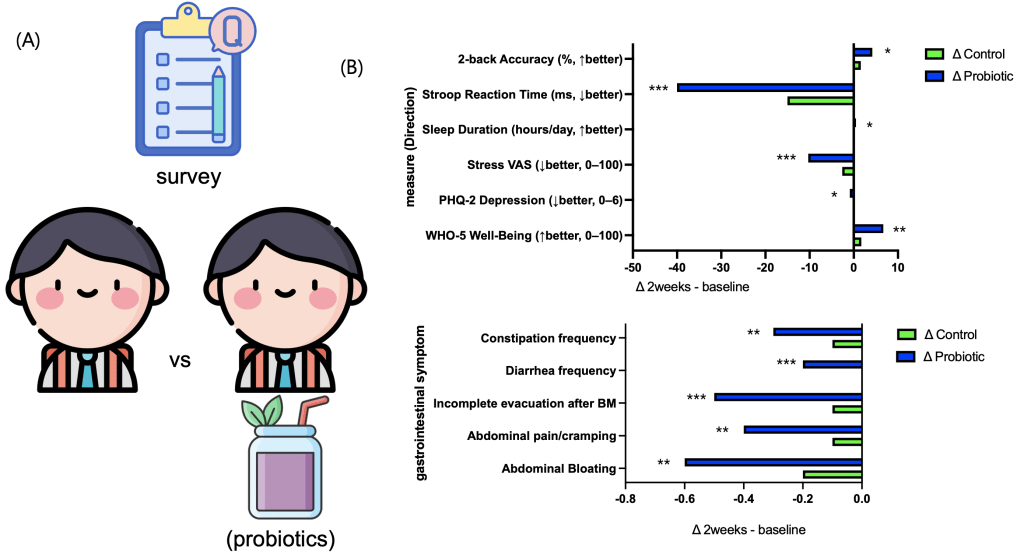
A randomized controlled trial was conducted to assess whether short-term probiotic consumption could improve well-being, reduce depressive symptoms and stress, and enhance cognitive performance. After the two-week intervention, the probiotic group showed positive changes across all key indicators compared to the control group. The Well-Being Index (The probiotic group showed a significant increase in WHO-5 scores compared to controls (mean difference = 4.8, 95% CI [1.2, 8.4], p = 0.01, Cohen’s d = 0.82).-5) significantly increased (Δ=+5.0, p<0.05), while the Depression Index (PHQ-2) and stress levels (VAS) significantly decreased (ΔPHQ-2=−0.7, ΔVAS=−5.1, p<0.05). The probiotic group showed a significant increase in WHO-5 scores compared to controls (mean difference = 4.8, 95% CI [1.2, 8.4], p = 0.01, Cohen’s d = 0.82). Improvements were also observed in cognitive tasks: average reaction time on the Stroop task was significantly reduced (Δ=−10 ms, p<0.01), and accuracy on the N-back task significantly increased (Δ=+1.0%, p<0.05). Gastrointestinal discomfort, including bloating and abdominal pain, was modestly alleviated, and both sleep duration and sleep quality showed slight improvements (fig. 1a-c). Collectively, these findings indicate that even short-term probiotic intake can meaningfully enhance emotional stability and cognitive function in adolescents.
2. Experimental design linking survey outcomes with the gut–brain axis.

To bridge the improvements in emotional and cognitive indices observed in the human survey with the biological pathway of the gut–brain axis, in vitro models representing the intestine (Caco-2 epithelial cells) and the brain (SH-SY5Y neuronal cells) were employed (fig. 2A). Probiotic beverages were centrifuged and filtered to remove viable bacteria, and the resulting diluted supernatant was applied to the apical compartment of Caco-2 cells cultured on Transwell inserts. After a two-week differentiation period, media collected from the basolateral compartment, referred to as conditioned media, were subsequently applied to SH-SY5Y cells (fig. 2B). This approach was designed to recapitulate the sequential pathway of probiotic intake → intestinal barrier modulation and signal generation → neuronal functional response within an in vitro gut–brain axis model.
3. Assessment of intestinal barrier integrity in Caco-2 cells.
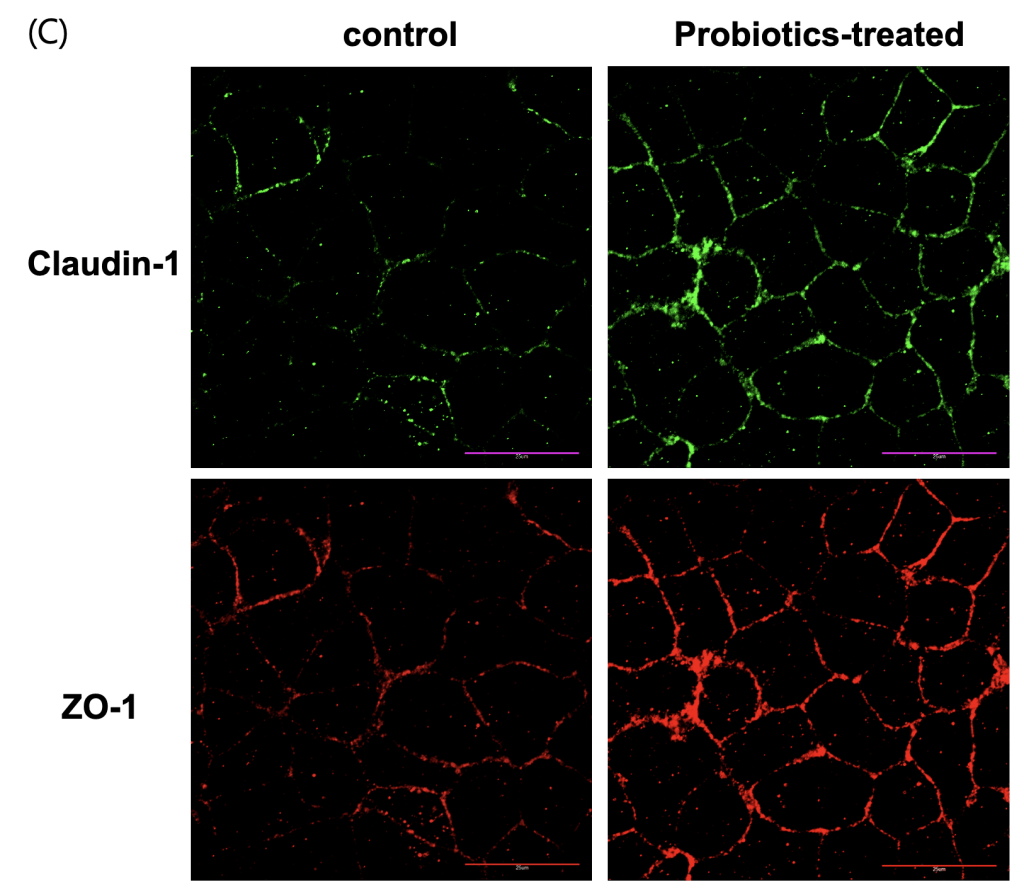
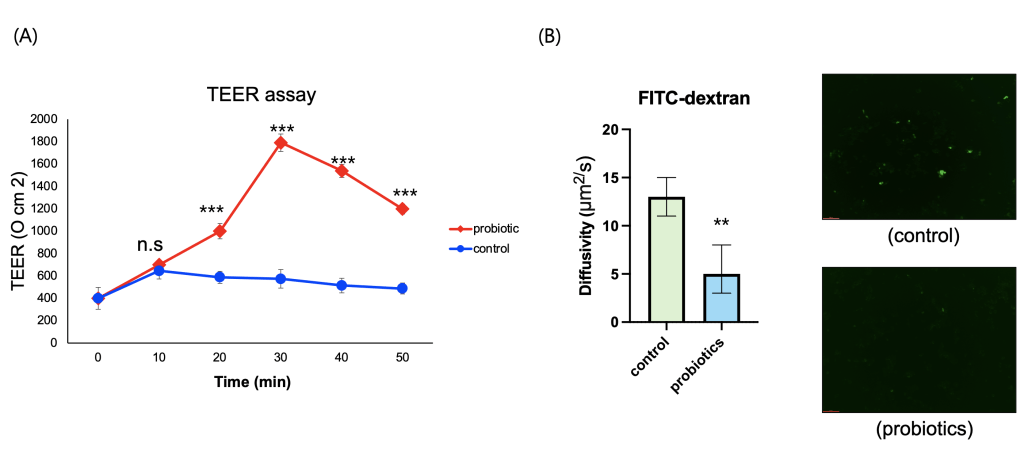
To determine whether probiotic-derived supernatant could enhance intestinal epithelial barrier function, differentiated Caco-2 monolayer cultures were treated with sterilized probiotic supernatant. Barrier integrity was assessed through multiple complementary approaches: trans-epithelial electrical resistance (TEER) to quantify ion tightness, FITC-dextran permeability to evaluate macromolecular leakage, and immunofluorescence staining for ZO-1 and claudin-1 to visualize the structural organization of tight junctions. Probiotic-treated cells showed significant improvements in all analyses. TEER values increased progressively over 30 mins, showing an approximately 3-fold increase (p<0.001) compared to the control group (Fig. 3A). FITC-dextran flux in the probiotic group was approximately 65% lower than the control group (p<0.01) (Fig. 3B). ZO-1 and claudin-1 exhibited clearer and more continuous localization along the cell junctions (fig. 3C), suggesting enhanced structural organization of the tight junction complex in the probiotic-treated group compared to the untreated group.
4. Transcriptomic and functional neuronal responses to probiotic-conditioned media
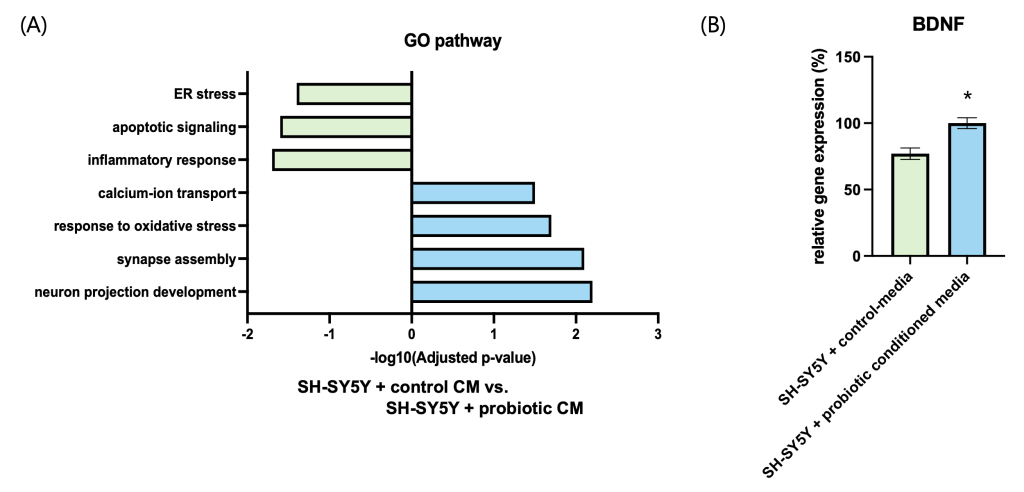
To evaluate whether probiotic-derived signals transmitted across the intestinal barrier could modulate neuronal function, SH-SY5Y cells were treated with basolateral conditioned media (CM) collected after two weeks from control or probiotic-treated Caco-2 monolayers. Transcriptomic profiling by RNA-seq revealed marked regulation of neuroplasticity-related pathways. Gene ontology analysis showed significant upregulation of neuron projection development, synapse assembly, and axon guidance in the probiotic-CM group, while pathways associated with inflammatory response and apoptotic process were downregulated (Fig. 4A). These findings suggest that gut-derived signals suppress pro-inflammatory and pro-apoptotic programs while promoting neurite growth and synaptic function.
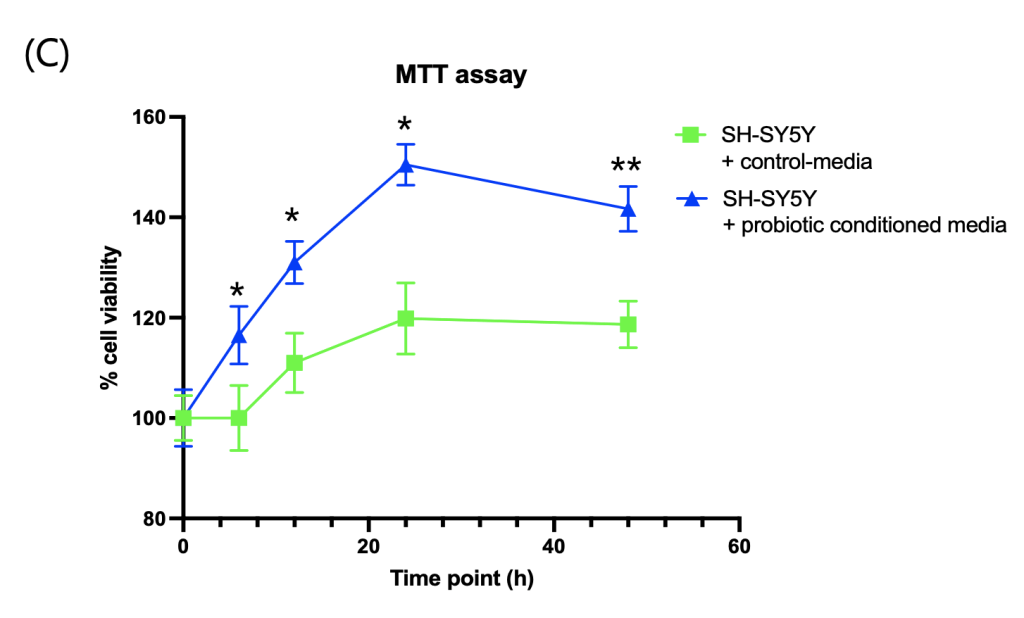
Consistent with these transcriptomic changes, RT-qPCR analysis demonstrated a ~25% increase in BDNF mRNA expression in the probiotic-CM group relative to control (Fig. 4B), highlighting activation of a key mediator of neuronal plasticity. Finally, MTT assays performed to assess metabolic activity and viability showed that probiotic-CM treatment enhanced SH-SY5Y cell viability by ~12.6% after 24 hours compared with control-CM (Fig. 4C). Together, these results indicate that probiotic-conditioned media not only reshape neuronal transcriptional programs toward plasticity but also confer measurable improvements in neuronal survival and function.
Discussion
Emerging evidence suggests that probiotics may influence stress and mental well-being through multiple, interrelated pathways within the gut–brain axis. One proposed mechanism involves modulation of the intestinal epithelial barrier, which plays a critical role in maintaining immune homeostasis and preventing systemic inflammatory signaling associated with psychological stress.
Strengthening of the intestinal barrier may reduce the translocation of pro-inflammatory molecules, thereby indirectly influencing stress-related neuroendocrine pathways. In parallel, probiotic-derived metabolites, such as short-chain fatty acids, have been shown to interact with neural and immune signaling cascades involved in emotional regulation.
In the present study, adolescents who consumed a probiotic beverage demonstrated improvements in self-reported well-being and perceived stress. Although these outcomes were assessed using questionnaires and do not provide direct biological measurements, they are consistent with prior human studies reporting associations between probiotic intake and reduced stress-related symptoms.
In the two-week human study, the probiotic group showed an increase in well-being indices, along with reductions in depression and stress scores, as well as improvements in cognitive performance compared with the control group (Fig. 1). These changes suggest a potential positive influence on adolescents’ daily quality of life and academic performance. In parallel, the Caco-2 intestinal epithelial model demonstrated increased TEER, decreased FITC-dextran permeability, and enhanced localization of ZO-1 and Claudin-1 (Fig. 3), indicating that probiotic-derived factors act to improve barrier integrity. Strengthening of the intestinal barrier may in turn contribute to the attenuation of systemic inflammatory signaling and stabilization of the gut–brain axis. The relatively small sample size limits statistical power and generalizability; however, this pilot study provides valuable preliminary insights.
Furthermore, to investigate the impact of signals transmitted across the intestinal epithelium on brain function, conditioned media were applied to SH-SY5Y neuronal cells. RNA-seq–based GO analysis revealed that probiotic-conditioned media upregulated genes associated with neurite outgrowth, synapse formation, and neuroplasticity, while downregulating pathways related to inflammation and apoptosis. Notably, the significant increase in BDNF expression provides compelling evidence that probiotic-treatment acts may promote the production of key mediators of neuroplasticity. Finally, MTT assays confirmed enhanced cellular viability in the conditioned media group, demonstrating that transcriptional changes were accompanied by functional improvements at the cellular level (Fig.4).
The central focus of this study lies in the signaling molecules contained within the conditioned media that passed through Caco-2 cells. Although RNA-seq allowed exploration of neuronal response pathways, the identity of these molecules was not directly determined. Based on previous studies, likely candidates include short-chain fatty acids (SCFAs), particularly butyrate and propionate, as well as exosomes and precursors of neurotransmitters. These molecules are thought to regulate epithelial metabolism, suppress inflammatory responses, and potentially reach the brain via the circulation to directly influence neuronal function.
Despite its multilayered design, this study has limitations. The human intervention was conducted over a relatively short period with a limited sample size, and the cell-based models do not fully replicate the complexity of the in vivo gut–brain axis. Moreover, an important limitation of the present study is the absence of a beverage-matched supernatant control in the in vitro experiments. Because the filtered supernatant contains not only probiotic-derived metabolites but also other soluble components of the beverage, such as sugars and organic acids, the observed effects on intestinal barrier integrity and neuronal gene expression cannot be attributed exclusively to probiotics. Furthermore, the filtration process removes live bacteria, indicating that the observed cellular effects are mediated by bacteria-free soluble factors rather than by intact probiotic organisms. Accordingly, the in vitro findings should be interpreted as supportive mechanistic observations, rather than definitive evidence of probiotic-specific effects. Future studies incorporating beverage-matched controls and live-probiotic conditions will be required to disentangle these effects. As biological samples such as blood were not collected from participants, the present study cannot establish a direct causal link between probiotic intake and molecular or biochemical changes in vivo.
Therefore, the cellular findings should be interpreted as supportive mechanistic observations, rather than as direct explanations of the human outcomes observed. Nevertheless, the consistent improvements observed in both human and cellular indices strongly suggest that probiotic treatments may exert beneficial effects on mental health through modulation of the gut–brain axis.
The findings of this study also provide important social insights. First, they point to a low-cost, high-efficiency strategy for mental health promotion. Compared with pharmacological or counseling approaches, probiotic or fermented food consumption is affordable and accessible, making it a practical alternative for populations with limited access to medical resources. Second, the results offer a scientific basis for school-based mental health promotion programs. Incorporating probiotic-enriched foods or dietary fiber into school meals, in combination with education on sleep hygiene and stress management, may contribute to the overall well-being of adolescents.
References
- Flisher, A.J., et al. Child and adolescent mental health in South Africa. Journal of Child & Adolescent Mental Health 24, 149–161 (2012). [↩]
- Lok, N. Factors affecting adolescent mental health. (2017). [↩]
- McLeod, J.D., Uemura, R. & Rohrman, S. Adolescent Mental Health, Behavior Problems, and Academic Achievement. Journal of Health and Social Behavior 53, 482–497 (2012). [↩]
- Joe, S., Joe, E. & Rowley, L.L. Consequences of Physical Health and Mental Illness Risks for Academic Achievement in Grades K–12. Review of Research in Education 33, 283–309 (2009). [↩]
- Sudo, N., et al. Postnatal microbial colonization programs the hypothalamic–pituitary–adrenal system for stress response in mice. The Journal of Physiology 558, 263–275 (2004). [↩]
- Verma, H., et al. Human Gut Microbiota and Mental Health: Advancements and Challenges in Microbe-Based Therapeutic Interventions. Indian Journal of Microbiology 60, 405–419 (2020). [↩]
- Maqsood, Q., Aman, M., Hussain, N., Saleem, M.Z. & Mahnoor, M. Microbiome-Based Therapies for Mental Health Disorders. in Human Microbiome: Techniques, Strategies, and Therapeutic Potential (eds. Khurshid, M. & Akash, M.S.H.) 463–493 (Springer Nature Singapore, Singapore, 2024). [↩] [↩]
- Topping, D.L. & Clifton, P.M. Short-chain fatty acids and human colonic function: roles of resistant starch and nonstarch polysaccharides. Physiol Rev 81, 1031–1064 (2001). [↩]
- Park, J.-h., et al. Promotion of Intestinal Epithelial Cell Turnover by Commensal Bacteria: Role of Short-Chain Fatty Acids. PLOS ONE 11, e0156334 (2016). [↩]
- Yao, Y., et al. The role of short-chain fatty acids in immunity, inflammation and metabolism. Critical Reviews in Food Science and Nutrition 62, 1–12 (2022). [↩]
- Liu, X.-f., et al. Regulation of short-chain fatty acids in the immune system. Frontiers in Immunology Volume 14 – 2023(2023). [↩]
- Beilharz, J.E., Kaakoush, N.O., Maniam, J. & Morris, M.J. Cafeteria diet and probiotic therapy: cross talk among memory, neuroplasticity, serotonin receptors and gut microbiota in the rat. Molecular Psychiatry 23, 351–361 (2018). [↩]
- Koutromanos, I., et al. Integrating gut microbiome and neuroplasticity genomics in alcohol use disorder therapy. Human Genomics 19, 78 (2025). [↩]
- Carabotti, M., Scirocco, A., Maselli, M.A. & Severi, C. The gut-brain axis: interactions between enteric microbiota, central and enteric nervous systems. Annals of Gastroenterology : Quarterly Publication of the Hellenic Society of Gastroenterology 28, 203 – 209 (2015). [↩]
- Bercik, P., Collins, S.M. & Verdu, E.F. Microbes and the gut-brain axis. Neurogastroenterology & Motility 24, 405–413 (2012). [↩]
- Simpson, J.B. & Redinbo, M.R. Multi-omic analysis of host-microbial interactions central to the gut-brain axis. Molecular Omics 18, 896–907 (2022). [↩]
- Lawrence, K. The microbiome and mental health. (2018). [↩]
- Li, N., et al. Oral Probiotics Ameliorate the Behavioral Deficits Induced by Chronic Mild Stress in Mice via the Gut Microbiota-Inflammation Axis. Frontiers in Behavioral Neuroscience Volume 12 – 2018(2018). [↩]
- Li, Q., et al. Probiotics alleviate depressive behavior in chronic unpredictable mild stress rat models by remodeling intestinal flora. NeuroReport 32, 686–693 (2021). [↩]
- Rianda, D., Agustina, R., Setiawan, E.A. & Manikam, N.R.M. Effect of probiotic supplementation on cognitive function in children and adolescents: a systematic review of randomised trials. Beneficial Microbes 10, 873–882 (2019). [↩]
- Salcedo Gómez, M. & García Aguilar, C. Limitations of Mechanistic Model of Explanation in Biological Psychiatry. European Psychiatry 65, S670–S670 (2022). [↩]