Abstract
This systematic literature review evaluates evidence from 25 primary studies, including human experimental trials and controlled animal models to examine how acute (24–64 hours) and chronic (≤6 hours/night for ≥3 nights) sleep deprivation affects immune function and susceptibility to disease. The review used a systematic search of PubMed, Web of Science, and Scopus using already defined inclusion and exclusion criteria. In addition, a PRISMA flow process documented study selection where the search identified 342 records, 298 after duplicates, 68 full texts screened, and 25 studies included. The evidence analyzed shows that acute sleep loss is associated with rapid increases in pro-inflammatory cytokines, including interleukin‑6 (IL‑6), tumor necrosis factor‑α (TNF‑α), and temporary reductions in natural killer (NK) cell cytotoxicity, showing an immediate but short immune disruption. Chronic sleep restriction, in contrast, seems to induce continuous low-grade inflammation, sustained NK cell suppression, and circadian dysregulation of immune-related gene expression, which all increase the chances of getting infections and chronic diseases. Overall, the literature demonstrates that insufficient sleep simultaneously triggers inflammatory pathways and impairs protective immune defenses, which highlights the important role of adequate sleep in maintaining immune homeostasis and overall health, with acute sleep loss producing transient immune disruption and chronic sleep restriction contributing to sustained low-grade inflammation and increased susceptibility to infectious and inflammatory diseases. These findings indicate that insufficient sleep is a modifiable risk factor for immune dysregulation and increased disease susceptibility, which is why this review intentionally presents a broad mechanistic overview of inflammatory, innate, and circadian pathways.
Keywords: Sleep deprivation; immune function; disease susceptibility; cytokines; inflammation; circadian rhythm; autoimmune diseases; Natural Killer cells; C-reactive protein; insomnia.
Introduction
Previous research discovered shows that sleep influences cytokine production, leukocyte trafficking, and adaptive immune responses since it affects both innate and adaptive defenses1. Specifically, sleep is able to regulate cytokine production, leukocyte trafficking, and antigen-specific immune responses, which are all controlled temporarily by circadian rhythms2. Insufficient sleep can also disrupt cytokine balances, reduce natural killer (NK) cell cytotoxicity, and change circadian regulations of immune gene expression, which all affect the body’s ability to respond to infections and to maintain homeostasis3. However, the mechanisms linking sleep deprivation to immune dysfunction depends on the duration of sleep loss, study design, and population studied. These conditions will create distinct physiological responses and have to be evaluated separately.
Firstly, acute sleep deprivation, which is defined as 24–64 hours of continuous wakefulness, produces rapid immune changes. For example, controlled laboratory studies have shown that 40 hours of total sleep deprivation significantly increases circulating interleukin-6 (IL-6), indicating acute inflammatory activation, while 64 hours of wakefulness is linked with leukocytosis and altered immune cell distribution4. Concurrently, reductions in NK cell cytotoxicity were seen following prolonged wakefulness5.
Meanwhile, chronic sleep restriction, where one has ≤6 hours of sleep per night for multiple consecutive nights, is seen to produce more sustained immune alterations. This is recognized in population-based studies where habitual short sleep duration has been related with elevated C-reactive protein (CRP), a marker of systemic inflammation and cardiovascular risk, while experimental restriction studies demonstrate cumulative increases in inflammatory signaling across consecutive nights of insufficient sleep6,7.
Transcriptomic studies have shown that repeated nights of restricted sleep upregulate pro-inflammatory gene pathways and reduce circadian rhythmicity in immune-related genes8. The same is for chronic insufficient sleep, which also reduces NK cell activity over time1. This suggests that there is a persistent impairment of innate immunity and potential vulnerability to infections and chronic diseases.
Gaps still remain despite discoveries since many reviews blend acute and chronic sleep deprivation, or combine human and animal evidence without clearly distinguishing underlying mechanisms. Additionally, cytokine responses and NK cell activity show inconsistent results across studies, likely due to differences in sleep deprivation protocols, circadian timing, and measurement methods9,10. Most studies also focused on short-term outcomes in healthy adult populations, limiting generalizability to older or medically vulnerable groups.
This review addresses these limitations by systematically examining acute versus chronic sleep deprivation across inflammatory, innate immune, and circadian pathways in both human and animal models.
Methods
This systematic literature review synthesizes 25 primary studies, including human experimental trials, observational studies, and controlled animal experiments, to clarify how sleep deprivation affects inflammatory cytokines and NK cell activity. By explicitly distinguishing acute versus chronic sleep loss and integrating human and animal evidence, this review aims to provide a mechanistic understanding of how insufficient sleep decides immune function and how it identifies priorities for future research by examining peer-reviewed research, investigating the effects of sleep deprivation on immune function and disease susceptibility, and the human and animal studies that reported primary research data.
Search Strategy:
A search was conducted in PubMed, Web of Science, and Scopus for studies published up to January 2025. Keywords included “sleep deprivation,” “sleep restriction,” “immune function,” “cytokines,” “natural killer cells,” “inflammation,” “vaccination response,” and “disease susceptibility.” (“AND,” “OR”) were used to combine terms, which ensured the inclusion of studies which examined immune outcomes in response towards acute or chronic sleep disruption.
Inclusion Criteria:
Studies were included if they:
- Were published in peer-reviewed journals.
- Reported primary research data in human or animal models.
- Examined total sleep deprivation (≥24 hours) or partial sleep restriction (<6 hours/night for ≥3 nights).
- Measured immune outcomes such as cytokine production, natural killer cell activity, antibody responses, C-reactive protein, or infection susceptibility.
- Were written in English.
Exclusion Criteria:
Studies were excluded if they:
- Focused on sleep disorders unrelated to experimental or voluntary sleep deprivation.
- Were secondary analyses, literature reviews, or opinion pieces.
- Lacked quantifiable immune or inflammatory outcomes.
- Were non-peer-reviewed, conference abstracts, or unpublished data.
Study Selection:
Titles and abstracts of all available studies were analyzed for relevance. Full texts were then reviewed for use based on inclusion and exclusion criteria. The final review included 25 studies spanning human experimental trials, observational studies, and controlled animal experiments. A Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) flow showed the study selection process, including reasons for exclusion at each stage:
Records identified through database searching: 342
- PubMed: 125
- Web of Science: 110
- Scopus: 107
Records after duplicates removed: 298
Records screened (title/abstract): 298
- Records excluded: 230
Full-text articles assessed for eligibility: 68
● Full-text articles excluded (with reasons): 43
- Not primary research: 15
- Non-relevant outcomes: 12
- Non-peer-reviewed / conference abstracts: 8
- Insufficient sleep deprivation definition: 8
- Studies included in qualitative synthesis: 25
Data Extraction and Synthesis:
Data extracted included study population, sample size, type and duration of sleep deprivation, immune outcomes measured, key findings, and study design. Studies were organized into innate immunity, adaptive immunity, inflammatory markers, circadian mechanisms, and disease susceptibility. Trends and inconsistencies were summarized though, as most included studies were observational or experimental but not interventional.
Results
Effects of Sleep Deprivation on Inflammatory Cytokines and Systemic Inflammation
Controlled laboratory studies show that acute total sleep deprivation (24–64 hours) increases circulating inflammatory markers11. Specifically, IL-6 and TNF-α are the most consistently reported cytokines to increase under acute sleep deprivation conditions, although the magnitude and timing of these responses can vary with the circadian phase and sampling protocols. For instance, in a 40-hour sleep deprivation protocol, healthy adults exhibited significant elevations in inflammatory markers including interleukin-6 (IL-6) and tumor necrosis factor-related activity4. Similarly, exposure to 64 hours of continuous wakefulness resulted in leukocytosis and increased immune activation markers alongside physiological fatigue12.
Meanwhile, partial sleep restriction studies show comparable but more gradual effects. Restricting sleep over multiple nights significantly increased circulating inflammatory cytokines, including IL-6 and TNF-α, with moderate sleep loss producing measurable immune activation13. Sustained sleep restriction protocols also resulted in elevated inflammatory markers across repeated measures14.
At the molecular level, sleep deprivation has shown to activate pro-inflammatory gene expression pathways, as sleep loss induces increased regulation of inflammatory transcriptional pathways and increased expression of genes involved in innate immune activation8. Complementary transcriptomic evidence also indicates that insufficient sleep reduces the amplitude of circadian gene expression while also dysregulating immune-related pathways3.
Furthermore, population-based studies further support these findings. Short sleep duration has been associated with elevated biomarkers of systemic inflammation, including C-reactive protein (CRP)7. Experimental sleep restriction has also been shown to increase CRP levels in controlled conditions, with convergence between epidemiological and laboratory findings6.
However, cytokine responses are not the same across studies. For example, while several studies report significant elevations in IL-6 because of sleep deprivation, other studies found no change or time-dependent variation, suggesting that cytokine responses are really influenced by circadian timing, duration of deprivation, and individual variability15. In addition, combined sleep deprivation and circadian misalignment further expands inflammatory responses beyond sleep loss alone4.
Effects on Innate Immune Function: Natural Killer Cells and Cellular Immunity
Sleep deprivation has been shown to impair innate immune defenses, particularly natural killer (NK) cell activity and cellular immune responses, both short and long term. Short-term partial sleep deprivation significantly reduces NK cell cytotoxic activity in humans. These reductions in NK cell activity indicate impaired innate immune defense, as the NK cells play a critical role in early antiviral and antitumor responses. One controlled study demonstrated that even a single night of restricted sleep led to measurable reductions in NK cell function and cellular immune responsiveness1. Likewise, prolonged wakefulness over 64 hours was associated with altered NK cell activity alongside broader immune dysregulation16. Circadian disruption further contributes to these effects. Acute sleep deprivation alters the normal diurnal distribution of immune cells, including lymphocytes and leukocytes, with sleep loss disrupting immune cell trafficking and timing2.
Animal studies provide additional mechanistic support, as sustained sleep deprivation in rodent models results in impaired host defense, which includes increased susceptibility to infection and failure to maintain immune homeostasis17. Follow-up work confirmed that prolonged sleep loss leads to systemic immune failure and increased mortality risk in animal models18. In infected rat models, sleep deprivation worsens immune dysfunction, altering immune responses to pathogens and worsening disease outcomes19.
Effects on Adaptive Immunity and Vaccine Response
Sleep also plays a critical role in adaptive immune processes, particularly antigen-specific responses and immunological memory formation. Experimental studies demonstrate that sleep enhances antibody production following vaccination, as individuals who obtained normal sleep after vaccination exhibited stronger antibody responses compared to those dealing with sleep deprivation20. Moreover, sleep duration positively predicted antibody response to hepatitis B vaccination in healthy adults21.
However, findings are not entirely consistent, as some studies report no long-term impairment in antibody titers following acute sleep deprivation, suggesting that the timing and duration of sleep loss relative to antigen exposure are critical determinants of adaptive immune outcomes. Individuals subjected to restricted sleep schedules produced significantly lower antibody titers following immunization22. Supporting this, early findings demonstrated reduced immunological responses to vaccination under sleep deprivation conditions23.
The same pattern is visible for other antigens as well. For instance, at the cellular level, sleep also enhances antigen-specific T cell responses, suggesting that sleep contributes directly to adaptive immune activation24. Again though, not all findings are consistent. One study examining H1N1 vaccination found that acute sleep deprivation did not produce long-term reductions in antibody titers and that short-term sleep loss may not uniformly impair adaptive immune outcomes25. The study used timing, duration, and type of immune challenge to influence observed effects.
Circadian Disruption, Hormonal Regulation, and Immune Timing
Sleep deprivation disrupts circadian rhythms, which in turn affects immune regulation and hormonal signaling26. Experimental evidence shows that sleep loss alters melatonin secretion and circadian clock gene expression through disruption of central biological timing systems2. These disruptions extend to immune-relevant transcriptional processes, including reduced rhythmicity in genes associated with immune functions3. Sleep deprivation also alters circulating immune signaling molecules linked to circadian regulation. Increased levels of IL-6 and soluble TNF-α receptors have been recorded under sleep deprivation conditions, as both inflammatory activation and altered immune signaling dynamics were affected27.
These circadian disruptions are further associated with broader physiological consequences, with a major example being how short sleep duration has been linked to metabolic dysregulation, which includes alterations in endocrine signaling pathways that interact with immune function28.
Human Pattern Intercrossing With Animal Studies
Human laboratory studies demonstrate that acute sleep loss induces rapid inflammatory activation and suppresses innate immune function, particularly NK cell activity and cytokine balance4. Multi-night sleep restriction studies also show cumulative increases in inflammatory markers and sustained immune dysregulation14.
Animal studies extend these findings by demonstrating that prolonged sleep deprivation leads to severe impairment in host defense and increased mortality, giving mechanistic evidence of immune system failure under chronic sleep loss17.
At the population level, shorter habitual sleep duration is associated with elevated systemic inflammation, suggesting that these experimental findings translate into real-world physiological patterns although these associations are largely correlational and do not fully conclude a causality between sleep duration and disease outcomes7.
Finally, across adaptive immune studies, sleep consistently supports antigen-specific immune responses, although variability again exists depending on experimental design and timing of sleep deprivation relative to immune challenge21.
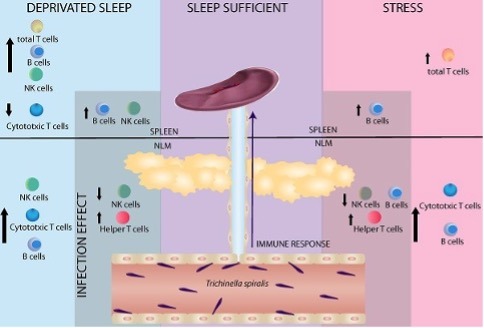
Description: Immune cell counts in the spleen and non-lymphoid mononuclear (NLM) compartment of infected rats under three conditions: sleep deprivation, adequate sleep, and psychological stress. Sleep deprivation elevated total T cells, B cells, NK cells, and cytotoxic T cells, suggesting heightened but potentially dysregulated immune activation. Adequate sleep supported balanced immune enhancement across cell types. Stress increased total T cells and B cells but reduced NK cells, cytotoxic T cells, and helper T cells, resulting in impaired cellular immunity. The figure illustrates how sleep and stress modulate host immune responses during parasitic infection19.
Discussion
Mechanistic Integration: Inflammation, Immune Suppression, and Circadian Disruption
The findings synthesized in this review show that sleep deprivation can produce numerous effects on immune function through three primary mechanisms: pro-inflammatory activation, suppression of innate immune defenses, and disruption of circadian immune regulation. These changes are further supported by transcriptional evidence demonstrating increased regulation of pro-inflammatory gene expression pathways under conditions of sleep loss8. Together, these findings indicate that sleep deprivation shifts the immune system toward a pro-inflammatory phenotype characterized by increased production of IL-6, TNF-α, and CRP alongside transcriptional activation of inflammatory signaling pathways, which may contribute to disease vulnerability.
Concurrently, sleep deprivation impairs innate immune function, particularly through reductions in natural killer (NK) cell activity and alterations in immune cell trafficking1,2. This suppression of cellular immune defenses suggests that sleep loss not only activates inflammatory pathways but also compromises the body’s ability to respond effectively to pathogens. This is particularly evident in reduced NK cell cytotoxicity and altered immune cell trafficking, which together weaken early immune defense mechanisms.
These immune alterations are further modulated by disruption of circadian rhythms. Sleep deprivation alters melatonin secretion, clock gene expression, and the rhythmic regulation of immune-related genes2,3. Because immune responses, which include cytokine release and immune cell distribution, are tightly regulated by circadian timing, disruption of these rhythms likely amplifies both inflammatory dysregulation and immune suppression.
In summary, the evidence shows that sleep deprivation produces simultaneous immune activation and dysfunction, rather than a simple unidirectional effect. This dual response is characterized by increased inflammation alongside impaired immune defense capacity.
Acute Versus Chronic Sleep Deprivation
A distinction shown from the literature is the difference between acute total sleep deprivation and chronic partial sleep restriction, which produce overlapping but not identical immune outcomes.
Acute sleep deprivation studies, which usually involves 24 to 64 hours of continuous wakefulness, demonstrate rapid increases in inflammatory markers and immediate reductions in NK cell activity16,4. These effects appear to be transient but profouned, reflecting short-term physiological stress responses. In contrast, chronic sleep restriction studies show more gradual but sustained immune alterations. Repeated nights of insufficient sleep result in persistent elevations in inflammatory cytokines and cumulative dysregulation of immune signaling pathways14,7. Importantly, chronic sleep loss is also associated with broader systemic effects, including metabolic dysregulation, which may further interact with immune pathways28.
Animal studies extend these findings by demonstrating that prolonged sleep deprivation leads to severe impairment of host defense and increased mortality17,18. These results suggest that while acute sleep loss produces immediate immune disruption, chronic deprivation may have more profound and potentially irreversible physiological consequences. Importantly, while acute sleep deprivation effects are generally reversible following recovery sleep, chronic sleep restriction may lead to potentially cause long-lasting alterations in immune regulation, as acute sleep loss helps initiates a state of simultaneous immune activation and suppression, with rapid but brief effects on cytokines and NK cell activity.
Adaptive Immunity and Variability in Vaccine Response
The effects of sleep on adaptive immunity are generally consistent but exhibit important variability across studies. Multiple studies demonstrate that sleep enhances antibody production and antigen-specific immune responses following vaccination20,21. Sleep also promotes T cell activation and immunological memory formation, indicating a critical role in adaptive immune function24.
However, not all findings are the same, as the studies analyzed report that there was a reduced vaccine response under sleep restriction conditions22. Meanwhile, other studies also find that there was no lasting impact of acute sleep deprivation on antibody titers25. These inconsistencies may reflect differences in experimental design, including the timing of sleep deprivation relative to vaccination, duration of sleep loss, and type of immune challenge.
This variability highlights that the relationship between sleep and adaptive immunity is context-dependent rather than absolute, reinforcing the importance of considering study conditions when interpreting findings.
Contradictions and Inconsistencies Across Studies
A key strength of the literature is also a source of complexity: findings are not entirely consistent across all studies.
For example, while many studies report increases in IL-6 following sleep deprivation, others demonstrate variable or condition-dependent responses15. Differences in sampling time, circadian phase, and duration of sleep restriction may account for these discrepancies. Similarly, while inflammatory activation is widely observed, the possibility of change varies across populations and experimental conditions.
In adaptive immunity, conflicting findings regarding vaccine response further illustrate these inconsistencies. While several studies demonstrate enhanced immune responses with sufficient sleep, others show minimal or no long-term impairment following acute deprivation25.
Differences between human and animal studies should be considered. Animal models often demonstrate more severe outcomes, including immune system failure and mortality under prolonged sleep deprivation17. In contrast, human studies typically observe more moderate physiological changes, reflecting differences for deciding experimental feasibility and ethical constraints. These inconsistencies overall highlight that immune responses to sleep deprivation are not uniform and depend heavily on experimental design, including duration of sleep loss, circadian timing, and the specific immune markers measured.
Limitations and Gaps
Several methodological limitations across the reviewed studies constrain the interpretation and generalizability of findings.
First, many experimental studies rely on small sample sizes and homogeneous populations, often consisting of healthy young adults. Evidence from adolescent populations indicates that sleep deprivation is also associated with increased inflammatory activity, suggesting that age and developmental stage may influence immune responses to sleep loss29. This limits the ability to generalize findings to broader populations, including older adults or individuals with underlying health conditions.
Second, there is substantial variability in study design, including differences in sleep deprivation protocols (total vs. partial), duration (acute vs. chronic), and outcome measures. This heterogeneity complicates direct comparisons across studies and could cause inconsistent findings.
Third, measurement of immune function varies widely, ranging from circulating cytokine levels to gene expression and functional immune assays. These differences in measurement approaches can produce contrasting results even when underlying physiological processes are similar.
Fourth, many studies assess short-term outcomes, limiting insight into long-term clinical consequences of sleep deprivation. While epidemiological studies suggest associations between short sleep duration and disease risk, causal relationships cannot be established based on the available evidence7.
Finally, the interaction between circadian disruption and sleep loss is not consistently controlled across studies. Because circadian misalignment independently affects immune function, failure to isolate these variables introduces additional complexity4. Additionally, variability in cytokine measurement techniques and differences in defining sleep deprivation across studies introduce methodological heterogeneity that complicates direct comparison of findings.
Implications for Intervention and Future Research
The findings reviewed here suggest that interventions targeting sleep may influence immune function, but the evidence remains indirect and context-dependent.
Improving sleep duration and quality may enhance adaptive immune responses, as demonstrated in vaccination studies21. However, variability across studies indicates that the effectiveness of such interventions depends on timing, duration, and individual physiological factors.
Future research should prioritize:
- Standardized definitions of sleep deprivation and measurement protocols
- Longitudinal studies examining long-term immune and disease outcomes
- Greater inclusion of diverse populations
- Integration of circadian biology into experimental design
By addressing these gaps, future work can more precisely define the relationship between sleep and immune health.
Conclusion
This literature review demonstrates that sleep deprivation alters immune function through coordinated effects on inflammatory signaling and innate immune defenses. Acute sleep loss rapidly elevates pro-inflammatory cytokines (IL‑6, TNF‑α) and reduces NK cell activity, causing immediate but temporary immune disruption, while chronic sleep restriction induces persistent low-grade inflammation, sustained NK cell suppression, and circadian dysregulation of immune gene expression, collectively heightening vulnerability to infections and chronic diseases. Although some findings vary depending on study design, timing, and population, the overall pattern highlights a dual effect: simultaneous immune activation and functional suppression.
Human and animal studies converge to show that both acute and chronic insufficient sleep compromise host defense, though human outcomes are generally less severe than in animal models. Key limitations include small sample sizes, heterogeneous sleep protocols, short-term follow-up, and underrepresentation of diverse populations. Future research should standardize sleep deprivation definitions, incorporate longitudinal immune assessments, and integrate circadian biology to clarify mechanistic pathways and guide intervention strategies.
From a public health perspective, these findings show that maintaining adequate sleep is a modifiable factor crucial for immune health, with implications for infection prevention, vaccine efficacy, and chronic disease risk reduction. Together, these findings highlight sleep as a modifiable determinant of immune health and a priority for future mechanistic and clinical research.
Acknowledgments
I would like to express gratitude to my science teachers for their guidance and support throughout the development of this research paper. Their feedback and instruction were invaluable in helping me learn how to structure, refine, and present my work effectively.
References
- Irwin, M., McClintick, J., Costlow, C., Fortner, M., White, J., & Gillin, J. C. (1996). Partial night sleep deprivation reduces natural killer and celhdar immune responses in humans. FASEB Journal, 10(5), 643–653. https://doi.org/10.1096/fasebj.10.5.8621064 [↩] [↩] [↩] [↩]
- Ackermann, K., Plomp, R., Lao, O., Middleton, B., Revell, V. L., Skene, D. J., & Kayser, M. (2013). Effect of sleep deprivation on rhythms of clock gene expression and melatonin in humans. Chronobiology International, 30(7), 901–909. https://doi.org/10.3109/07420528.2013.784773 [↩] [↩] [↩] [↩] [↩]
- Möller-Levet, C. S., Archer, S. N., Bucca, G., Laing, E. E., Slak, A., Kabiljo, R., Lo, J. C., Santhi, N., von Schantz, M., Smith, C. P., & Dijk, D. J. (2013). Effects of insufficient sleep on circadian rhythmicity and expression amplitude of the human blood transcriptome. Proceedings of the National Academy of Sciences, 110(12), E1132–E1141. https://doi.org/10.1073/pnas.1217154110. [↩] [↩] [↩] [↩]
- Wright, K. P., Jr, Drake, A. L., Frey, D. J., Fleshner, M., Desouza, C. A., Gronfier, C., & Czeisler, C. A. (2015). Influence of sleep deprivation and circadian misalignment on cortisol, inflammatory markers, and cytokine balance. Brain, Behavior, and Immunity, 47, 24–34. https://doi.org/10.1016/j.bbi.2015.01.004 [↩] [↩] [↩] [↩] [↩] [↩]
- Dinges, D. F., Douglas, S. D., Zaugg, L., Campbell, D. E., McMann, J. M., Whitehouse, W. G., Orne, E. C., Kapoor, S. C., Icaza, E., & Orne, M. T. (1994). Leukocytosis and natural killer cell function parallel neurobehavioral fatigue induced by 64 hours of sleep deprivation. The Journal of Clinical Investigation, 93(5), 1930–1939. https://doi.org/10.1172/JCI117184. [↩]
- Meier-Ewert, H. K., Ridker, P. M., Rifai, N., Regan, M. M., Price, N. J., Dinges, D. F., & Mullington, J. M. (2004). Effect of sleep loss on C-Reactive protein, an inflammatory marker of cardiovascular risk. Journal of the American College of Cardiology, 43(4), 678–683. https://doi.org/10.1016/j.jacc.2003.07.050. [↩] [↩]
- Patel, S. R., Zhu, X., Storfer-Isser, A., Mehra, R., Jenny, N. S., Tracy, R., & Redline, S. (2009). Sleep duration and biomarkers of inflammation. Sleep, 32(2), 200–204. https://doi.org/10.1093/sleep/32.2.200. [↩] [↩] [↩] [↩] [↩]
- Irwin, M. R., Wang, M., Campomayor, C. O., Collado-Hidalgo, A., & Cole, S. (2006). Sleep Deprivation and Activation of Morning Levels of Cellular and Genomic Markers of Inflammation. Archives of Internal Medicine, 166(16), 1756–1762. https://doi.org/10.1001/archinte.166.16.1756 [↩] [↩] [↩]
- Dinges, D. F., Douglas, S. D., Zaugg, L., Campbell, D. E., McMann, J. M., Whitehouse, W. G., Orne, E. C., Kapoor, S. C., Icaza, E., & Orne, M. T. (1994). Leukocytosis and natural killer cell function parallel neurobehavioral fatigue induced by 64 hours of sleep deprivation. The Journal of Clinical Investigation, 93(5), 1930–1939. https://doi.org/10.1172/JCI117184. [↩]
- Haack, M., Sanchez, E., Mullington, J. M. (2007). Elevated Inflammatory Markers in Response to Prolonged Sleep Restriction Are Associated With Increased Pain Experience in Healthy Volunteers. Sleep, 30(9), 1145–1152. https://doi.org/10.1093/sleep/30.9.1145. [↩]
- Wright, K. P., Jr, Drake, A. L., Frey, D. J., Fleshner, M., Desouza, C. A., Gronfier, C., & Czeisler, C. A. (2015). Influence of sleep deprivation and circadian misalignment on cortisol, inflammatory markers, and cytokine balance. Brain, Behavior, and Immunity, 47, 24–34. https://doi.org/10.1016/j.bbi.2015.01.004 [↩]
- Dinges, D. F., Douglas, S. D., Zaugg, L., Campbell, D. E., McMann, J. M., Whitehouse, W. G., Orne, E. C., Kapoor, S. C., Icaza, E., & Orne, M. T. (1994). Leukocytosis and natural killer cell function parallel neurobehavioral fatigue induced by 64 hours of sleep deprivation. The Journal of Clinical Investigation, 93(5), 1930–1939. https://doi.org/10.1172/JCI117184. [↩]
- Vgontzas, A. N., Zoumakis, E., Bixler, E. O., Lin, H. M., Prolo, P., Vela-Bueno, A., & Chrousos, G. P. (2004). Adverse Effects of Modest Sleep Restriction on Sleepiness, Performance, and Inflammatory Cytokines. Journal of Clinical Endocrinology & Metabolism, 89(5), 2119–2126. https://doi.org/10.1210/jc.2003-031562. [↩]
- Haack, M., Sanchez, E., Mullington, J. M. (2007). Elevated Inflammatory Markers in Response to Prolonged Sleep Restriction Are Associated With Increased Pain Experience in Healthy Volunteers. Sleep, 30(9), 1145–1152. https://doi.org/10.1093/sleep/30.9.1145. [↩] [↩] [↩]
- Redwine, L., Hauger, R. L., Gillin, J. C., & Irwin, M. (2000). Effects of sleep and sleep deprivation on interleukin-6, growth hormone, cortisol, and melatonin levels in humans. The Journal of Clinical Endocrinology and Metabolism, 85(10), 3597–3603. https://doi.org/10.1210/jcem.85.10.6871. [↩] [↩]
- Dinges, D. F., Douglas, S. D., Zaugg, L., Campbell, D. E., McMann, J. M., Whitehouse, W. G., Orne, E. C., Kapoor, S. C., Icaza, E., & Orne, M. T. (1994). Leukocytosis and natural killer cell function parallel neurobehavioral fatigue induced by 64 hours of sleep deprivation. The Journal of Clinical Investigation, 93(5), 1930–1939. https://doi.org/10.1172/JCI117184. [↩] [↩]
- Everson C. A. (1993). Sustained sleep deprivation impairs host defense. The American journal of physiology, 265(5 Pt 2), R1148–R1154. https://doi.org/10.1152/ajpregu.1993.265.5.R1148. [↩] [↩] [↩] [↩]
- Rechtschaffen, A., & Bergmann, B. M. (2002). Sleep Deprivation in the Rat: An Update of the 1989 Paper. Sleep, 25(1), 18–24. https://doi.org/10.1093/sleep/25.1.18. [↩] [↩]
- Ibarra-Coronado, E. G., Pantaleón-Martínez, A. M., Velazquéz-Moctezuma, J., Prospéro-García, O., Méndez-Díaz, M., Pérez-Tapia, M., Pavón, L., & Morales-Montor, J. (2015). Sleep Deprivation Induces Changes in Immunity in Trichinella spiralis Infected Rats. International Journal of Biological Sciences, 11(8), 901–912. https://doi.org/10.7150/ijbs.11907 [↩] [↩]
- Lange, T., Perras, B., Fehm, H. L., & Born, J. (2003). Sleep enhances the human antibody response to hepatitis A vaccination. Psychosomatic Medicine, 65(5), 831–835. https://doi.org/10.1097/01.PSY.0000091382.61178.F1 [↩] [↩]
- Prather, A. A., Hall, M., Fury, J. M., Ross, D. C., Muldoon, M. F., Cohen, S., & Marsland, A. L. (2012). Sleep and antibody response to hepatitis B vaccination. Sleep, 35(8), 1063–1069. https://doi.org/10.5665/sleep.1990. [↩] [↩] [↩] [↩]
- Van Leeuwen, W. M., Lehto, M., Karisola, P., Lindholm, H., Luukkonen, R., Sallinen, M., Härmä, M., Porkka-Heiskanen, T., & Alenius, H. (2009). Sleep restriction increases the risk of developing cardiovascular diseases by augmenting proinflammatory responses through IL-17 and CRP. PLoS One, 4(2), e4589. https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0004589 [↩] [↩]
- Spiegel, K., Sheridan, J. F., & Van Cauter, E. (2002). Effect of sleep deprivation on response to immunization. JAMA, 288(12), 1471–1472. https://doi.org/10.1001/jama.288.12.1471-a. [↩]
- Dimitrov, S., Lange, T., Benedict, C., Nowell, M. A., Jones, S. A., Scheller, J., Rose-John, S., & Born, J. (2006). Sleep enhances IL-6 trans-signaling in humans. FASEB Journal, 20(12), 2174–2176. https://doi.org/10.1096/fj.06-5754fje. [↩] [↩]
- Benedict, C., Brytting, M., Markström, A., Broman, J. E., & Schiöth, H. B. (2012). Acute sleep deprivation has no lasting effects on the human antibody titer response following a novel influenza A H1N1 virus vaccination. BMC Immunology, 13, 1. https://doi.org/10.1186/1471-2172-13-1. [↩] [↩] [↩]
- Meier-Ewert, H. K., Ridker, P. M., Rifai, N., Regan, M. M., Price, N. J., Dinges, D. F., & Mullington, J. M. (2004). Effect of sleep loss on C-Reactive protein, an inflammatory marker of cardiovascular risk. Journal of the American College of Cardiology, 43(4), 678–683. https://doi.org/10.1016/j.jacc.2003.07.050. [↩]
- Shearer, W. T., Reuben, J. M., Mullington, J. M., Price, N. J., Lee, B. N., Smith, E. O., Szuba, M. P., Van Dongen, H. P. A., & Dinges, D. F. (2001). Soluble TNF-α receptor 1 and IL-6 plasma levels in humans subjected to the sleep deprivation model of spaceflight. Journal of Allergy and Clinical Immunology, 107(1), 165–170. https://doi.org/10.1067/mai.2001.112270. [↩]
- Taheri, S., Lin, L., Austin, D., Young, T., & Mignot, E. (2004). Short Sleep Duration Is Associated with Reduced Leptin, Elevated Ghrelin, and Increased Body Mass Index. PLoS Medicine, 1(3), e62. https://doi.org/10.1371/journal.pmed.0010062. [↩] [↩]
- Irwin, M. R., Olmstead, R., & Carroll, J. E. (2016). Sleep and Inflammation During Adolescence. Psychosomatic Medicine, 78(5), 545–552. https://doi.org/10.1097/PSY.0000000000000340. [↩]






