Abstract
Approximately twenty million people a year are diagnosed with cancer, and about ten million die from the disease annually. Cancer is traditionally treated by a combination of approaches geared to removing, killing, and/or controlling cancer cells, including surgery, chemotherapy, and radiation. One out of three cancer cases is refractory, meaning they do not respond to any of these traditional treatments, while one out of two of the ones that did respond will relapse, and the cancer will come back even after the initial success. Though the 5-year survival rate has been improving through these traditional treatments, especially because of early detection, the global mortality burden remains significant, and numerous novel treatments are in development in the hopes of turning more cancer victims into cancer survivors. This review focuses on one such promising new therapy, CAR-T cell therapy (chimeric antigen receptor T-cell therapy), which is a revolutionary immunotherapy that utilizes a patient’s own genetically modified T-cells to attack cancer cells with high specificity. The treatment has been demonstrated in clinical trials to be highly effective at treating an array of blood cancers, specifically ones that were previously considered untreatable by traditional means (relapsed/refractory), and there are now 7 FDA-approved CAR-T therapies for several different blood cancers. Further, there is promise in ongoing research and trials for this powerful technology to be an effective treatment against solid tumor cancers as well. This paper will review the application, results, and success rates of CAR-T cell therapy and discuss ongoing efforts to apply this therapy to additional blood cancers, as well as solid tumor cancers and even autoimmune diseases. Though the treatment is in the early stages of application and ongoing development and has some hurdles to overcome, CAR-T cell therapy offers hope for a possible one-and-done cure for cases where conventional treatments have failed. CAR-T cell therapy has the potential to become a leading cancer treatment for patients worldwide.
Introduction
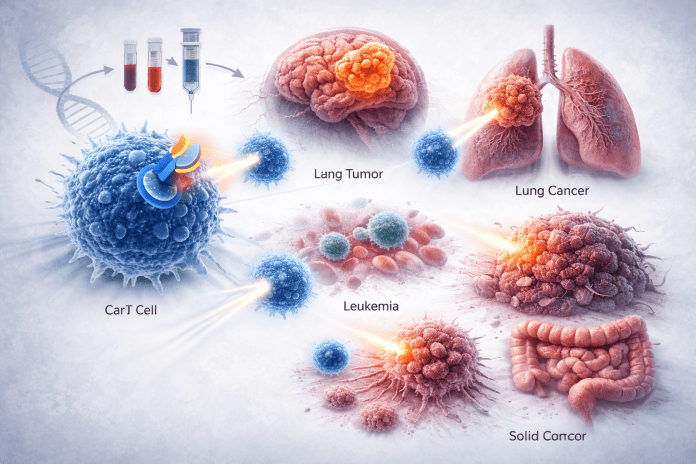
CAR-T cell therapy is an advanced immunotherapy that utilizes a patient’s own T cells, re-engineered as weapons to hunt and kill cancer cells in a highly targeted approach. Originally invented by Zelig Eshhar of the Weizmann Institute of Science in the 1980s, the technology was later refined and developed from the lab into the first CAR-T treatment for patients by Carl June of the University of Pennsylvania and Michel Sadelain of Memorial Sloan Kettering Cancer Center1. They called it Tisagenlecleucel, derived from T cell therapy specific antigen (Tisa), genetically engineered (gen), leucocytes (leu), cell therapy (cel). In 2010-2012, the first three adult patients with chronic lymphocytic leukemia were treated and achieved complete remission, demonstrating for the first time that the approach could produce durable responses in otherwise untreatable disease2. Then, in 2012, they tried the treatment on a 6-year-old girl named Emily Whitehead, who had acute lymphoblastic leukemia (ALL) and was ineligible for a bone marrow transplant. She received the treatment, achieved complete remission, and remains cancer-free to this day3. Branded by Novartis as Kymriah, this first CAR-T therapy gained FDA approval in 2017. The therapy has demonstrated remarkable success in clinical trials in cancer cases that were otherwise untreatable, and much follow-up research and development has since been completed4. Currently, there are seven FDA-approved CAR-T treatments for several relapsed or refractory B-cell malignancies.
Approximately 20 million people are diagnosed with cancer each year, and about 10 million die from the disease annually5‘6. The majority of these cancers are solid tumors, while a smaller proportion, approximately 8-10%, are hematologic cancers such as leukemias, lymphomas, and myeloma5‘6. All cancers are treated initially with surgery (when possible), chemotherapy, and radiation, and, for some blood cancers, hematopoietic stem cell transplant (HSCT). However, many patients do not respond fully to these treatments, and even among those who initially respond, relapse remains common6. The current overall 5-year survival rate of solid-tumor and blood cancers combined is approximately 70% in the United States and considerably lower in many parts of the world due to disparities in access to care6. These statistics drive massive efforts to develop novel, targeted cancer treatments like CAR-T cell therapy that can provide better options, especially where none exist, and achieve higher, lasting cure rates.
When a patient begins CAR-T cell treatment, their T cells are collected through leukapheresis, then genetically modified in a lab to produce a chimeric antigen receptor (CAR) that binds a specific protein on cancer cells (for example, CD19 in the case of Kymriah), expanded into millions, and reinfused back into the patient as a highly specific, single-dose therapy1. The whole process takes from 2 to 6 weeks, but differs from traditional treatments like chemotherapy and radiation because it involves only one infusion, rather than numerous sessions over an extended period1.
The goal of this paper is to review and understand the current state of CAR-T cell therapy and the hurdles that must be overcome to make this potential miracle cure more broadly accessible and cost-effective7. This paper will review the landmark clinical trials that paved the way for the treatment, the seven FDA-approved CAR-T therapies, and their real-world effectiveness in terms of complete remission (CR) rates and overall survival (OS) rates, major adverse effects (cytokine release syndrome and neurotoxicity)8, high manufacturing costs9, relapse due to “antigen escape,”7 and strategies for next-generation CAR-T therapies that could extend the treatment to solid-tumors and autoimmune diseases.
Methods
Literature Search
A literature review was conducted in PubMed on December 8, 2025. Articles published from 2020 to 2025 were searched using the terms “CAR-T,” “CAR T,” or “Chimeric Antigen Receptor” combined with keywords for approved therapies, pivotal trials (ELIANA, ZUMA-1, JULIET, KarMMa, CARTITUDE, etc.), real-world evidence, toxicities (CRS, ICANS), manufacturing and cost issues, solid-tumor applications, autoimmune uses, and antigen escape. Searches were limited to English-language, human subject studies and typically yielded several hundred to approximately 1,400 results per query. After removing duplicates and screening titles and abstracts for relevance, full texts were reviewed. Eighteen high-quality review articles and key clinical studies published between 2020 and 2025 were selected for inclusion. Reference lists of all included papers were also examined for additional sources. Data on active clinical trials were verified through ClinicalTrials.gov10.
Results
Early Academic Trials at the University of Pennsylvania
In August 2010, the first adult patient with chronic lymphocytic leukemia (CLL) was treated with a CD19 CAR-T (tisagenlecleucel) and achieved long-lasting, complete remission (CR)2. This initial case provided proof of concept that the technology could work and generated significant excitement in the oncology community. From 2011 to 2012, two additional adults with CLL were treated, and both also achieved CR2. Between 2012 and 2013, the first two children, including Emily Whitehead, were treated with tisagenlecleucel3. Both had relapsed, refractory ALL with little hope for survival, and incredibly, both achieved long-lasting CR3. Given the circumstances, especially that they were children with extremely poor prognoses and no remaining treatment options, these early successes were extraordinary.
Between 2013 and 2014, 30 additional young patients with ALL were treated with tisagenlecleucel, and 91% of them achieved CR3. Based on these unprecedented results, the FDA granted “Breakthrough Therapy Designation” to tisagenlecleucel, accelerating clinical testing and paving the way for broader development of CAR-T therapies1.
Landmark Clinical Trials for CAR-T Cell Therapies
Four trials directly supported the initial FDA (and later global) approvals of the first commercial CAR-T cell therapies: ELIANA, ZUMA-1, JULIET, and ELARA. Each served as a pivotal registrational study. All were prospective, multicenter, and single-arm trials (all patients received the treatment)1, appropriate given the patients enrolled had such poor prognoses that denying them the potentially curative treatment would be unethical1.
ELIANA
The ELIANA trial was a global phase 2 study that led to the approval of tisagenlecleucel11. Because dosing for tisagenlecleucel and manufacturing procedures had been established in the University of Pennsylvania academic studies, the FDA allowed the trial to begin at phase 211. A total of 79 children and young adults between the ages of 3 and 21 with relapsed B-cell ALL who had failed multiple prior treatments were infused11. After a single treatment, 81% achieved CR, a groundbreaking result consistent with earlier academic findings11.
ZUMA-1
Zuma-1 was a combined phase 1/2 trial evaluating axicabtagene ciloleucel for diffuse large B-cell lymphoma (DLBCL)12. 101 adults who had failed two prior lines of therapy were treated12. 58% of them achieved CR, leading to “Breakthrough Therapy Designation” and an accelerated path to approval12. Axicabtagene ciloleucel, branded as Yescarta, became the first approved CAR-T for lymphoma12.
JULIET
The JULIET trial was a global phase 2 study of tisagenlecleucel in 115 adults with relapsed or refractory DLBCL13. As with ELIANA, no phase 1 study was required because prior work had already established dosing13. A single infusion produced a 40% CR rate13. With Yescarta already approved, Kymriah became an alternative CAR-T option for adult lymphoma13.
ELARA
The ELARA trial evaluated tisagenlecleucel in 97 adults with relapsed or refractory follicular lymphoma (FL), a slow-growing but difficult-to-cure cancer14. 69% achieved lasting CR, leading to FDA approval for this indication14.
FDA-Approved CAR-T Cell Therapies (as of December 2025)
As a result of the pivotal trial breakthroughs, there are now seven FDA-approved CAR-T cell therapies, all indicated for relapsed or refractory B-cell malignancies. Five target CD19, a protein expressed on B-cells leukemias and lymphomas, and two target BCMA, a protein expressed on multiple myeloma cells. They are each achieving promising complete remission (CR) rates and overall survival (OS) rates, not only in the trial patients but also now in a growing population of patients being treated in the real world15‘16. They are summarized as follows:
1. Kymriah (tisagenlecleucel)
Approved in 2017, targeting CD1917.
Indications based on the ELIANA, JULIET, and ELARA trials include:
- Pediatric/young adult relapsed or refractory B-cell ALL11.
- Adult DLBCL13.
- Adult follicular lymphoma14.
Trial CR rates:
- ELIANA: 81%
- JULIET: 40%
- ELARA: 69–70%
Long-term outcomes (per trials):
- ELIANA:
 58% OS at 5 years
58% OS at 5 years - JULIET:
 43% OS at 5 years
43% OS at 5 years - ELARA: 82% OS at 4 years
2. Yescarta (axicabtagene ciloleucel)
Approved weeks after Kymriah, also targeting CD1912.
Indications:
- Adult relapsed or refractory DLBCL12.
- Adult relapsed or refractory FL
- ZUMA-1 trial CR rate: 58%
- ZUMA-1 OS:
 43% at 5 years
43% at 5 years
3. Tecartus (brexucabtagene autoleucel)
Targets CD19. Made by Kite/Gilead Sciences18.
Indications:
- Adult mantle cell lymphoma (MCL) based on ZUMA-2
- Adult B-cell ALL based on ZUMA-3
Trial outcomes:
- ZUMA-2: 62% CR, 39% OS at 5 years
- ZUMA-3: 63% CR,
 50% OS at latest follow-up
50% OS at latest follow-up
4. Breyanzi (lisocabtagene maraleucel)
Targets CD19 and is approved for several relapsed or refractory B-cell malignancies19.
Indications include LBCL, FL, MCL, and CLL/SLL. Trial CR rates (TRANSCEND NHL, TRANSFORM): ![]() 54% 2-year OS:
54% 2-year OS: ![]() 50–55%.
50–55%.
5. Abecma (idecabtagene vicleucel)
Targets BCMA for relapsed or refractory multiple myeloma (RRMM)7.
KarMMa trial:
- 33% CR
 50% OS
50% OS
6. Carvykti (ciltacabtagene autoleucel)
Also targets BCMA for RRMM20.
CARTITUDE-1 trial:
- 83% CR
- Estimated 62% OS at 5 years
(This 5-year OS is supported by published long-term follow-up in 2023–2024 updates.)
7. Aucatzyl (obecabtagene autoleucel)
Targets CD19 for adult relapsed or refractory B-cell ALL, approved in late 202415.
FELIX trial:
- 77% CR
 70% 1-year OS
70% 1-year OS
Real-world data are not yet available.
Challenges of CAR-T Cell Therapy
Negative side effects and high cost are among the two biggest challenges facing CAR-T therapy. Additionally, relapse can occur when the cancer cells stop expressing the target antigen and “hide” from the therapy7‘9, a problem known as antigen escape7. Perhaps the most difficult challenge, though, is expanding the technology so that it can effectively treat solid tumor cancers as well21‘22.
Adverse Effects
An estimated majority of patients receiving CAR-T infusions experience some degree of treatment-related toxicity8. The most common reaction is Cytokine release syndrome (CRS), where the CAR-T cells activate so strongly that they trigger large, rapid cytokine release. CRS can cause fever, fatigue, low blood pressure, and, in more severe cases, oxygen needs or difficulty breathing8. Another major toxicity is immune effector cell-associated neurotoxicity syndrome (ICANS), which can lead to confusion, tremors, or seizures8.
Most CRS and ICANS cases are manageable with standard interventions such as tocilizumab and corticosteroids, but in a subset of patients, the reaction is more serious. Severe toxicity occurs in roughly 10–30% of treated individuals, and early mortality related to toxicity occurs in approximately 3–7% of cases across studies and products8. Other complications—such as prolonged low blood counts or infections—occur less frequently but remain important considerations in the management of CAR-T recipients8.
Antigen Escape
Relapse due to tumor antigen escape is a major reason CAR-T therapy can ultimately fail7. After CAR-T cells initially kill the vast majority of cancer cells that display the target antigen (like CD19 or BCMA), a small subpopulation of tumor cells may survive because they already expressed low levels of the antigen or because they down-regulate its expression following therapy7‘21. These surviving ‘antigen-negative’ clones expand, leading to relapse.
In B-ALL, studies report that 30–60 % of relapses after CAR-T therapy lack CD19 on the cell surface7. In multiple myeloma treated with BCMA-targeted CAR-T, 20–50 % of relapses involve tumor clones with minimal or absent BCMA on the surface7. This occurs because tumors are not uniform; they contain many different clones, and the ones that lose the target gain the advantage for survival once the CAR-T cells are active.
To address this, researchers are developing dual target CAR-T cells (for example, CD19 + CD22 or BCMA + GPRC5D) that attack two antigens simultaneously to prevent antigen escape. Early studies of these next-generation, multi-antigen CAR-T cells report dramatically reduced relapse rates and very high remission rates, sometimes exceeding 90% in early phase trials22.
Manufacturing Costs
A major limitation of current CAR-T therapy is its high cost. Because each CAR-T treatment is custom-made from the patient’s own cells, production takes 2–6 weeks and typically costs ![]() 475,000 per treatment, not including hospitalization and supportive care9. Administration is highly specialized, and only a limited number of certified centers worldwide are equipped to offer it, restricting accessibility to treatment, particularly in lower-income regions23‘9. Additionally, manufacturing failures occur in some 5–20 % of cases, further contributing to the cost burden9. To address these challenges, researchers are developing allogeneic (“off-the-shelf”) CAR-T products that are made from healthy donor T cells rather than the patient’s own22. Point-of-care manufacturing technologies are also being tested to produce CAR-T products directly at hospitals, which could potentially shorten production time and reduce cost. These advances may make CAR-T therapy more widely available in the future22.
475,000 per treatment, not including hospitalization and supportive care9. Administration is highly specialized, and only a limited number of certified centers worldwide are equipped to offer it, restricting accessibility to treatment, particularly in lower-income regions23‘9. Additionally, manufacturing failures occur in some 5–20 % of cases, further contributing to the cost burden9. To address these challenges, researchers are developing allogeneic (“off-the-shelf”) CAR-T products that are made from healthy donor T cells rather than the patient’s own22. Point-of-care manufacturing technologies are also being tested to produce CAR-T products directly at hospitals, which could potentially shorten production time and reduce cost. These advances may make CAR-T therapy more widely available in the future22.
Next Generation CAR-Ts
xWhile blood cancers represent only a small percentage of overall cancer worldwide, they are uniquely suited to CAR-T therapy because malignant cells circulate in blood, bone marrow, and lymphoid tissues, where T cells can easily reach them21. By contrast, solid tumor cancers build fibrinogen and collagen walls and immunosuppressive microenvironments that block T-cell infiltration21‘22.
Efforts to expand CAR-T technology to solid tumors are focused on strategies that help CAR-T cells infiltrate the tumor and weaken the surrounding micro-environment so that the CAR-T cells can penetrate, survive, and ultimately kill the cancer cells22. These include:
- “Armoured CAR-T cells”, which are engineered to secrete cytokines or enzymes that can damage the tumor wall
- Multi-antigen CAR-T cells, engineered to recognize multiple tumor antigens simultaneously to counteract antigen heterogeneity and escape.
- CAR-T cells with checkpoint-resistant designs, such as PD-1 knockout constructs.
- These next-generation designs aim to overcome the biological barriers that have historically limited the success of CAR-T therapies in solid tumors22.
Autoimmune Diseases
CAR-T therapy is now being investigated as a treatment for autoimmune diseases, where the immune system mistakenly attacks the body’s own tissues and causes conditions like lupus and multiple sclerosis16. Early results (2024–2025) show dramatic B-cell depletion and long-lasting remission with far milder side effects than in cancer patients16. In one multicenter German study, fifteen patients with severe autoimmune conditions (including systemic lupus erythematosus, idiopathic inflammatory myositis, and systemic sclerosis) received low-dose CD19-targeted CAR-T therapy. Patients with lupus in this cohort achieved complete remission and were able to discontinue immunosuppressive medications, with remission sustained for more than a year in published follow-up16.
Similar early signals of deep, durable remission have been reported in small exploratory studies of multiple sclerosis, pemphigus, and inflammatory myopathies16‘24. Notably, autoimmune CAR-T patients experience far milder side effects than cancer patients because the required CAR-T dose is lower and inflammatory tumor burden is absent. These compelling early results have prompted the rapid expansion of clinical trials investigating CAR-T therapy across numerous autoimmune conditions16.
Ongoing Trials of Interest
Solid tumor cancers and autoimmune diseases currently have no FDA-approved CAR-T indications, but early phase 1/2 studies show initial promise. Diffuse Intrinsic Pontine Glioma (DIPG) is a rare, aggressive pediatric brain tumor that affects children aged 5-9, and is one of the earliest solid-tumor applications. In an ongoing trial (NCT04196413), children with H3K27M-mutant diffuse midline gliomas receive GD2-targeted CAR-T therapy delivered into the cerebrospinal fluid. Early results show meaningful tumor shrinkage in a majority of patients along with evidence of CAR-T trafficking into the central nervous system10.
Autoimmune applications are also advancing rapidly. A CD19-directed CAR-T trial for systemic lupus erythematosus (NCT03030976) reported complete remission in all treated lupus patients in early cohorts, with the ability to discontinue all immunosuppressive therapy and remain in remission for over a year16. These findings were later expanded in a 15-patient multicenter study that included lupus, myositis, and systemic sclerosis patients16.
Allogeneic “off-the-shelf” CAR-T therapies are also making progress. The ALPHA2 trial (NCT04416984), which tests the allogenic CD19 CAR-T product ALLO-501A for relapsed large B-cell lymphoma, has shown early overall response rates around 50%-70%, helping to address manufacturing delays and cost10.
Solid-tumor trials continue to broaden. Multiple phase 1 studies are investigating HER2-targeted, MUC1-targeted, and mesothelin-targeted CAR-Ts for breast cancer, pancreatic cancer, and epithelial tumors. Armored and multi-antigen CAR-T cells appear promising in early study readouts21‘22.
As of 2025, more than 1,400 CAR-T trials are active globally, with major areas of emphasis including dual-targeting, armored CAR-T designs, allogenic platforms, and combination therapies (such as CAR-T with PD-1 inhibitors)10. These developments illustrate the rapidly evolving efforts to expand CAR-T therapy into the diseases that currently lack curative treatments.
| Trial Name | Phase | Targets | Cancer/Disease | Design/Goal | Sponsor/Status | Est. Completion |
| NCT04196413 (GD2-CAR for DIPG/DMG) | 1/2 | GD2 | Pediatric diffuse midline glioma/DIPG | GD2 CAR-T delivered intracranially to overcome CNS barriers | Baylor College of Medicine / Recruiting | 2026 |
| NCT03030976 (CD19-CAR for SLE) | 1/2 | CD19 | Autoimmune (systemic lupus erythematosus) | CD19 CAR-T for B-cell depletion; remission in 100% lupus cohort | Charité–Universitätsmedizin Berlin/ Recruiting | 2027 |
| NCT04416984 (ALLO-501A for LBCL – ALPHA2) | 1 | CD19 (allogeneic) | Relapsed/refractory large B-cell lymphoma | Off-the-shelf CD19 CAR-T to address manufacturing delays and improve access | Allogene Therapeutics / Recruiting | 2026 |
| NCT05239143 (P-MUC1C-ALLO1 CAR-T) | 1 | MUC1-C | Solid tumors (breast, lung, pancreatic, ovarian, etc.) | Allogeneic CAR-T targeting MUC1-C; armored design | WindMIL/ Poseida/ Recruiting | 2028 |
| NCT05567741 (CD22 CAR-T for B-ALL after CD19 loss) | 2 | CD22 | B-ALL following CD19-negative relapse | CD22 CAR-T to overcome antigen escape after prior CD19 therapy | National Cancer Institute/ Recruiting | 2026 |
| NCT05041257 (CD70 CAR-T for RCC) | 1/2 | CD70 | Renal cell carcinoma | CD70-targeted CAR-T for RCC | Memorial Sloan Kettering/ Recruiting | 2027 |
| NCT04916800 (IL-1RA Armored CD19 CAR-T) | 1 | CD19 + IL-1RA | Autoimmune diseases (incl. lupus nephritis) | Armored CAR-T secreting IL-1 receptor antagonist to reduce inflammatory toxicity | Horizon Therapeutics/ Recruiting | 2028 |
| NCT05399480 (HER2 CAR-T for solid tumors) | 1 | HER2 | HER2-positive advanced solid tumors | HER2 CAR-T with cytokine-secreting enhancements | City of Hope / Recruiting | 2027 |
| NCT05274451 (Multi-antigen CAR-T for pancreatic cancer) | 1 | MUC1 + HER2 | Pancreatic adenocarcinoma | Dual MUC1/HER2 CAR with PD-1-modified signaling | Fred Hutchinson Cancer Center / Recruiting | 2027 |
Discussion
Both chemotherapy and hematopoietic stem cell transplant (HSCT) have been successful in treating some oncology patients; however, in CAR-T cell therapy, the chance of healthy cells being attacked by drugs (as can happen in chemotherapy) is greatly reduced, and rejection is not a possibility because CAR-T uses the patient’s own T cells, not donor cells3. As with any treatment, there are risks. CAR-T therapy can cause cytokine release syndrome (CRS), neurotoxicity, infection, fatigue, and other adverse effects8.
As shown in the results, CAR-T cell therapy is still relatively new, with most approved products based on phase I/2 or single-arm phase 2 studies1. Yet these studies have produced remarkable remission rates—often 40% to over 80% in patients who had no other options. These outcomes are particularly notable because the patients enrolled in these trials had relapsed or refractory disease with very poor prognoses. Unfortunately, as with all powerful immunotherapies, serious adverse effects can occur. CRS and ICANS remain the most common toxicities. Although most cases are manageable, approximately 10-30% are severe, and early mortality related to toxicity occurs in a small subset of patients (3–7%)8.
One benefit of reviewing the CAR-T literature is the opportunity to understand just how dramatically effective this therapy can be across various blood cancers, especially in cases where no viable options remain7. However, an important limitation in comparing CAR-T studies is the heterogeneity across clinical trial designs —different follow-up times, dosing strategies, conditioning regimens, lymphodepletion protocols, patient populations, and response definitions22. Because of this variability, direct comparisons between CAR-T products are difficult, as are cross-trial conclusions22.
Another major limitation is cost. Because CAR-T therapy is highly personalized and custom-manufactured for each patient, a single treatment costs ![]() 475,000 (plus hospitalization), putting it out of reach for many patients and healthcare systems9. This high price, combined with the need for specialized centers, severely limits global access23‘9.
475,000 (plus hospitalization), putting it out of reach for many patients and healthcare systems9. This high price, combined with the need for specialized centers, severely limits global access23‘9.
Despite these challenges, CAR-T has already transformed outcomes for thousands of patients with relapsed or refractory blood cancers and is now expanding into autoimmune diseases and, more slowly, solid tumors16‘24. Early studies in systemic lupus erythematosus and other autoimmune conditions have demonstrated high remission rates with relatively mild toxicity, suggesting a potentially broader role for CAR-T outside oncology16.
Ongoing research is focused on next-generation CAR-T designs that reduce relapse, limit toxicity, and expand applicability to difficult-to-treat diseases. These strategies include dual-antigen targeting to prevent antigen escape, armored CAR-T cells engineered to overcome the immunosuppressive tumor microenvironment, off-the-shelf allogenic CAR-T products to reduce cost and manufacturing time, and combination therapies with immune checkpoint inhibitors. With more than 1,400 clinical trials in progress worldwide, CAR-T cell therapy is rapidly evolving from a last-resort option into a potentially standard curative option for many currently incurable diseases10.
Conclusion
Although CAR-T cell therapy is still in relatively early stages of clinical application, it has already demonstrated the potential to benefit countless patients1. As of 2025, seven FDA-approved CAR-T treatments are commercially available for a variety of relapsed and refractory B-cell malignancies that were previously considered incurable. The dramatic success achieved in these diseases has driven major research and development efforts aimed at expanding the use of CAR-T technology7.
More than 1,400 ongoing clinical trials are now evaluating next-generation CAR-T products that use strategies such as dual-antigen targeting, armored CAR designs, and allogeneic, off-the-shelf products to improve specificity, reduce adverse effects, lower cost, and extend efficacy to solid-tumor cancers and autoimmune diseases.
This review has outlined what CAR-T cell therapy is, how it works, the landmark trials that led to its approval, current real-world outcomes, major challenges, and future directions. Cancer affects millions of individuals, families, and communities worldwide every year, regardless of background5‘6. CAR-T cell therapy offers real hope that many of these patients, especially those who have exhausted all other treatment options, may one day achieve long-lasting remission or even be cured with a single, personalized treatment1.
Acknowledgments
Thank you for the guidance of my mentor, Valentyna Kostiuk, from Yale University in the development of this research paper.
References
- C. H. June, M. Sadelain. Chimeric antigen receptor therapy. The New England Journal of Medicine. Vol. 379, pp. 64–73, 2018, https://doi.org/10.1056/NEJMra1706169. [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- D. L. Porter, W. T. Hwang, N. V. Frey, S. F. Lacey, P. A. Shaw, A. W. Loren, A. Bagg, K. T. Marcucci, A. Shen, V. Gonzalez, D. Ambrose, S. A. Grupp, A. Chew, Z. Zheng, M. C. Milone, B. L. Levine, J. J. Melenhorst, C. H. June.Chimeric antigen receptor T cells persist and induce sustained remissions in relapsed, refractory chronic lymphocytic leukemia. Science Translational Medicine. Vol. 7, pg. 303ra139, 2015, https://doi.org/10.1126/scitranslmed.aac5415. [↩] [↩] [↩]
- S. A. Grupp, M. Kalos, D. Barrett, R. Aplenc, D. L. Porter, S. R. Rheingold, D. T. Teachey, A. Chew, B. Hauck, J. F. Wright, M. C. Milone, B. L. Levine, C. H. June. Chimeric antigen receptor–modified T cells for acute lymphoid leukemia. The New England Journal of Medicine. Vol. 368, pp. 1509–1518, 2013, https://doi.org/10.1056/NEJMoa1215134. [↩] [↩] [↩] [↩] [↩]
- S. L. Maude, T. W. Laetsch, J. Buechner, S. Rives, M. Boyer, H. Bittencourt, P. Bader, M. R. Verneris, H. E. Stefanski, G. D. Myers, M. Qayed, B. De Moerloose, H. Hiramatsu, K. Schlis, K. L. Davis, P. L. Martin, E. R. Nemecek, G. A. Yanik, C. Peters, A. Baruchel, N. Boissel, F. Mechinaud, A. Balduzzi, J. Krueger, C. H. June, B. L. Levine, P. Wood, T. Taran, M. Leung, K. T. Mueller, Y. Zhang, K. Sen, D. Lebwohl, M. A. Pulsipher, S. A. Grupp.Tisagenlecleucel in children and young adults with B-cell lymphoblastic leukemia. The New England Journal of Medicine. Vol. 378, pp. 439–448, 2018, https://doi.org/10.1056/NEJMoa1709866. [↩]
- F. Bray, M. Laversanne, H. Sung, J. Ferlay, R. L. Siegel, I. Soerjomataram, A. Jemal. Global cancer statistics 2024: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer Journal for Clinicians. Vol. 74, pg. 229–263, 2024, https://doi.org/10.3322/caac.21834. [↩] [↩] [↩]
- R. L. Siegel, T. B. Kratzer, A. N. Giaquinto, H. Sung, A. Jemal. Cancer statistics, 2025. CA: A Cancer Journal for Clinicians. Vol. 75, pp. 10–45, 2025, https://doi.org/10.3322/caac.21871. [↩] [↩] [↩] [↩] [↩]
- Janssen Biotech, Inc. Carvykti (ciltacabtagene autoleucel) prescribing information. Horsham, PA: Janssen Biotech; 2025. [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- Y. Kong, J. Li, X. Zhao, Y. Wu, L. Chen. CAR-T cell therapy: developments, challenges and expanded applications from cancer to autoimmunity. Frontiers in Immunology. Vol. 15, Article 1519671, 2025, https://doi.org/10.3389/fimmu.2024.1519671. [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- S. H. Khan, Y. Choi, M. Veena, J. K. Lee, D. S. Shin. Advances in CAR T cell therapy: antigen selection, modifications, and current trials for solid tumors. Frontiers in Immunology. Vol. 15, Article 1489827, 2025, https://doi.org/10.3389/fimmu.2024.1489827. [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- ClinicalTrials.gov. Search terms: “CAR T” OR “CAR-T” AND “solid tumor” OR “autoimmune.” Accessed December 8, 2025. https://clinicaltrials.gov. [↩] [↩] [↩] [↩] [↩]
- S. S. Neelapu, F. L. Locke, N. L. Bartlett, L. J. Lekakis, D. B. Miklos, C. A. Jacobson, I. Braunschweig, O. O. Oluwole, T. Siddiqi, Y. Lin, J. M. Timmerman, P. J. Stiff, J. W. Friedberg, I. W. Flinn, A. Goy, B. T. Hill, M. R. Smith, A. Deol, U. Farooq, P. McSweeney, J. Munoz, I. Avivi, J. E. Castro, J. R. Westin, J. C. Chavez, A. Ghobadi, K. V. Komanduri, R. Levy, E. D. Jacobsen, T. E. Witzig, P. Reagan, A. Bot, J. Rossi, L. Navale, Y. Jiang, J. Aycock, M. Elias, D. Chang, J. Wiezorek, W. Y. Go. Axicabtagene ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. The New England Journal of Medicine. Vol. 377, pp. 2531–2544, 2017, https://doi.org/10.1056/NEJMoa1707447. [↩] [↩] [↩] [↩] [↩]
- S. J. Schuster, M. R. Bishop, C. S. Tam, E. K. Waller, P. Borchmann, J. P. McGuirk, U. Jäger, S. Jaglowski, C. Andreadis, J. R. Westin, I. Fleury, V. Bachanova, S. R. Foley, P. J. Ho, S. Mielke, J. M. Magenau, H. Holte, S. Pantano, L. B. Pacaud, R. Awasthi, J. Chu, Ö. Anak, G. Salles, R. T. Maziarz; JULIET Investigators.Tisagenlecleucel in adult relapsed or refractory diffuse large B-cell lymphoma. The New England Journal of Medicine. Vol. 380, pp. 45–56, 2019, https://doi.org/10.1056/NEJMoa1804980. [↩] [↩] [↩] [↩] [↩] [↩]
- N. H. Fowler, M. Dickinson, M. Dreyling, J. Martinez-Lopez, A. Kolstad, J. Butler, M. Ghosh, L. Popplewell, J. C. Chavez, E. Bachy, K. Kato, H. Harigae, M. J. Kersten, C. Andreadis, P. A. Riedell, J. P. McGuirk, A. L. Petzer, F. Offner, A. Viardot, P. L. Zinzani, R. Malladi, A. Zia, R. Awasthi, O. Anak, S. J. Schuster, C. Thieblemont.Tisagenlecleucel in adult relapsed or refractory follicular lymphoma (ELARA trial). Nature Medicine. Vol. 28, pp. 325–332, 2022, https://doi.org/10.1038/s41591-021-01622-0. [↩] [↩] [↩] [↩] [↩]
- Novartis Pharmaceuticals Corporation. Kymriah (tisagenlecleucel) prescribing information. East Hanover, NJ: Novartis; 2025. [↩] [↩] [↩]
- X. Deschênes-Simard, M. Bromberg, S. M. Devlin, M. Gonen, O. Beyar-Katz, A. Ip, R. Marcus, A. Avigdor, A. Ballweg, E. Rabinovich, M. Alhomoud, A. Rivas-Delgado, M. Corona De Lapuerta, A. Luna De Abia, M. L. Palomba, G. L. Shah, R. J. Lin, A. P. Boardman, L. Falchi, J. K. Lue, G. A. Salles, M.-A. Perales, R. Shouval, P. B. Dahi, M. Scordo. Comparative real-world outcomes of CD19-directed CAR T-cell therapies in large B-cell lymphoma. Blood Advances. Vol. 9, pp. 5571–5584, 2025, https://doi.org/10.1182/bloodadvances.2025016778. [↩] [↩]
- J. C. Chavez. Anti-CD19 chimeric antigen receptor T-cell therapy in B-cell lymphomas: current status and future directions. International Journal of Hematologic Oncology. Vol. 10, Article IJH33, 2021, https://doi.org/10.2217/ijh-2020-0021. [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- Kite Pharma, Inc. Yescarta (axicabtagene ciloleucel) prescribing information. Santa Monica, CA: Kite Pharma; 2025. [↩]
- Kite Pharma, Inc. Tecartus (brexucabtagene autoleucel) prescribing information. Santa Monica, CA: Kite Pharma; 2025. [↩]
- Bristol Myers Squibb. Breyanzi (lisocabtagene maraleucel) prescribing information. Summit, NJ: Bristol Myers Squibb; 2025. [↩]
- Autolus Therapeutics. Aucatzyl (obecabtagene autoleucel) prescribing information. London, UK: Autolus Therapeutics; 2025. [↩]
- S. K. Pal, B. Tran, J. B. A. G. Haanen, M. E. Hurwitz, A. Sacher, N. M. Tannir, L. E. Budde, S. J. Harrison, S. Klobuch, S. S. Patel, L. Meza, M.-L. Dequeant, A. Ma, Q. A. He, L. M. Williams, A. Keegan, E. B. Gurary, H. Dar, S. Karnik, C. Guo, H. Heath, R. R. Yuen, P. K. Morrow, N. Agarwal, S. A. Srour. CD70-targeted allogeneic CAR T-cell therapy for advanced clear cell renal cell carcinoma. Cancer Discovery. 2024;14(7):1176–1189. https://doi.org/10.1158/2159-8290.CD-24-0102. [↩] [↩] [↩] [↩] [↩]
- K. K. Patel, M. Tariveranmoshabad, S. Kadu, N. Shobaki, C. H. June. From concept to cure: the evolution of CAR-T cell therapy. Molecular Therapy. Vol. 33, pp. 2123–2140, 2025, https://doi.org/10.1016/j.ymthe.2025.03.005. [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- Bristol Myers Squibb. Abecma (idecabtagene vicleucel) prescribing information. Summit, NJ: Bristol Myers Squibb; 2025. [↩] [↩]
- L.-Y. Cao, Y. Zhao, Y. Chen, P. Ma, J.-C. Xie, X.-M. Pan, X. Zhang, Y.-C. Chen, Q. Wang, L.-L. Xie. CAR-T cell therapy clinical trials: global progress, challenges and future directions from ClinicalTrials.gov insights. Frontiers in Immunology. Vol. 16, Article 1583116, 2025, https://doi.org/10.3389/fimmu.2025.1583116. [↩] [↩]