Abstract
The rise of antibiotic-resistant bacteria has created a necessity for alternative methods for treating wound infections. The effectiveness of modern antibiotics is drastically decreasing which leads to prolonged healing, greater health risks, and more. This study evaluates the use of bacteriophages – viruses that selectively target bacteria – as a potential restorative solution to bacterial wound infections. 60 Mueller Hinton Agar plates were prepared and spread with a full lawn of Escherichia coli K-12 (E. coli K-12) using an L-shaped spreader. The hydrogel was synthesized using a hybrid mixture of sodium alginate & chitosan which was then crosslinked with calcium chloride to provide more structure, stability, controlled release, and effectiveness of the bacteriophages. T4R+ bacteriophages were infused into the hydrogel and then applied to 1cm² sections of sterile gauze. The hydrogel-coated bandages were then applied onto 30 agar plates. 30 control plates were applied with sterile discs soaked with distilled water, then all plates were incubated at 37°C for 24 hours to allow bacterial growth. After incubation, the zones of inhibition were measured using rulers. Average inhibition zones were calculated for both groups of plates to determine bacteriophage-hydrogel efficacy. The results proved the bacteriophage-hydrogel bandage was highly effective in inhibiting bacterial growth, with the mean zone of inhibition being 36.5mm. The Mann-Whitney U test yielded a Z value of 7.1058, indicating a significant difference between the bacteriophage hydrogel and distilled water. These findings open the potential for bacteriophage-infused treatments to become viable alternatives to traditional antibiotics for combating bacterial wound infections.
Keywords: Bacteriophage, hydrogel, antibiotic resistance, Escherichia coli, phage therapy, antimicrobial biomaterials, wound healing, T4R+ phage, biopolymer delivery systems, chitosan, alginate
Introduction
Bacteriophages are viruses that only infect and replicate in bacterial cells. They are extremely diverse in size and morphology and are recognized as the earth’s most abundant biological agent. There are many different phage types; some of these types appear to have “heads,” “legs,” and “tails.” Bacteriophages are non-motile and they need Brownian motion to help them reach their targets1. Bacteriophages are species specific about their hosts so they usually only infect a single bacterial species or even specific strains within a species.
Once a bacteriophage attaches to a susceptible host, it pursues either lytic replication or lysogenic replication. In a lytic replication cycle, a phage attaches to a susceptible host, introduces its genome into the host cell’s cytoplasm, and uses the host’s ribosomes to create its proteins. The host cell’s resources are quickly converted to viral genomes and capsid proteins, and these assemble into multiple copies of the original phage. The host cell dies while being either actively or passively lysed, releasing the new bacteriophage to infect another host cell.
In the lysogenic replication cycle, the bacteriophage attaches to a susceptible host bacterium and it introduces its genome into the host cell’s cytoplasm. The bacteriophage genome is integrated into the bacterial cell chromosome or it is maintained as an episomal element where it is replicated and passed on to daughter bacterial cells without killing them. The integrated phage genomes can convert to a lytic replication cycle and kill their host, often in response to changing environmental conditions1.
Escherichia coli K-12 was selected for this study because it is a non-pathogenic lab strain that poses minimal safety risk to humans and is widely used in antimicrobial research.
T4 bacteriophages (a specific class of bacteriophages that target Eschrichea coli) use their tail fibers to attach to a bacterium. Then, using enzymes on its tail, it will make a hole in the wall or membrane. Its tail sheath will contract which causes the DNA to leave the protein coat and go into the host cell. The DNA then goes to the nucleus and is used as a template by the cell to make viral mRNA. This mRNA travels to the host cell’s ribosomes and is then used to create viral proteins. Once viral protein subunits (also called capsomeres) are formed inside the cell, they spontaneously self-assemble into a complete protein coat. Then, viral DNA is also replicated by the cell, and it combines with the protein coat to make a new intact virus. Finally, lysosome-like viral enzymes break down the cell wall and the host cell lyses as the newly created viruses exit the cell.
There are different strains of bacteriophage T4 including T4r and T4r+. The “r” means that there is a mutation that leads to the rapid lysis of the bacterial host. T4r differs from the wild type T4r+ strain in its size. T4r is physically larger than T4r+ which would mean that T4r would diffuse through a bacterial culture more slowly. This slow diffusion rate of T4 would mean that T4r+ may be the most effective at infecting E. coli K-122.
A wound is a disturbance in the typical structure and function of the epidermis which is the outer layer of skin. Wound healing is a complex process that contains many interdependent immunological and pathophysiological mediators to restore the tissue3. Wound healing has many potential factors that can delay healing. One of these factors are bacterial infections. Bacterial infections of wounds are detrimental to the wound healing process and there is increasing evidence that bacteria within chronic wounds live within biofilm communities in which the bacteria are resistant to host defenses and could develop resistance to antibiotic treatment4.
Bacteriophages are used in the hospital setting and can be administered in various methods. The most efficient method of phage delivery in treating systemic infections is by injection. Oral delivery is possible, but it isn’t helpful in treating these infections because of their quick clearance from the gut5. Phage cocktail formulations usually guarantee a broader spectrum of activity because of phages’ high specificity for their bacterial host6. Once phages enter the body, they reach most organs which includes the central nervous system. Some data suggests that the immune system removes phages from the body even when no specific immune response to phages has developed. With regards to phage movement within the human body, when delivered systemically, phages are not detected in other tissues in large quantities5.
Phages have been proven to work in animals such as mice in treating different bacteria such as Pseudomonas aeruginosa, and human trials in phage therapy are also showing promising results. In a six patient case series of antibiotic-unresponsive diabetic foot ulcers, the topical application of a phage that targets Staphylococcus aureus (S. aureus) was sufficient for the recovery of each of the six individuals7. In a clinical trial involving nine diabetic patients with toe ulcers caused by S. aureus, phage therapy showed promising results. All nine patients were unresponsive to conventional therapy between 10 days to seven weeks before the phage treatment. When the Staphylococcal phage Sb-1 was applied topically to the ulcerations once a week paired with standard wound care, the ulcers healed in around seven weeks and severe ulcers healed in around 18 weeks. Phages were also tested on 20 patients with chronic nonhealing wounds that didn’t respond to conventional local debridement and antibiotic treatment. These wounds contained E. coli, S. aureus, and P. aeruginosa. Three to five doses of phage therapy led to complete healing or healthy margins and healthy granulation tissue in the wounds (a positive sign of wound healing)6.
Escherichia coli (E. Coli) are rod shaped, gram negative, and facultative anaerobic bacteria8. A single E. Coli can divide every 20 minutes9. However, some strains have evolved into pathogenic E. Coli, acquiring their virulence factors through different things like plasmids and bacteriophages8. When skin is damaged, E. Coli can cause skin infections such as cellulitis. Incidents of cellulitis being caused by E. Coli however, are rare and mainly occur in immunodeficient patients where the E. Coli infects their blood before infecting their skin. E. Coli isolates from skin infections exhibit a remarkable virulence potential which is comparable to E. Coli isolates found in urinary tract infections. E. Coli skin infections can be treated by antibiotics9.
Hydrogel drug delivery systems have attracted huge interest in wound healing because of their biocompatibility, versatility, and moisture retention capacity at the trauma site. Hydrogels offer a protective dressing, which provides an optimal environment for healing and a controlled release of therapeutic agents, including bacteriophages7. The application of bacteriophages in hydrogels allows for a prolonged and localized release of the agents to the wound environment, augmenting bacterial eradication while reducing the side effects10.
The embedding process of the bacteriophages into the hydrogel must be done carefully in order to preserve phage viability and activity. Physical entrapment, ionic gelation, or covalent conjugation techniques can be used to embed phages with retention of activity. Physical entrapment preserves the integrity of phages in the hydrogel, whereas ionic gelation provides for their inclusion by ionic interactions with the polymer11. Covalent conjugation is a chemical attachment process of the phage to the polymer, allowing for a controlled release. The incorporation strategy is dependent on the release profile and phage stability in the hydrogel12. Hydrogels create an environment of high humidity which is critical to wound healing. While bacteriophages have a high therapeutic modality towards infection reduction without disrupting healthy tissues. This combination has a double faced approach where the hydrogel helps facilitate the wound healing process while at the same time releasing the bacteriophages that act specifically to combat bacterial infection. The difficulty of accomplishing this experiment successfully relies on maintaining the stability, viability, and activity of the bacteriophages in the hydrogel itself13. Apart from their use in infection control, hydrogel-based phage delivery systems can also reduce the number of dressing changes, therefore, promoting patient comfort and accelerating healing processes. This method is especially useful in wound treatment because of high bacteria loads or risk of infection, as it provides a more durable, localized, and controlled means of administering bacteriophage therapy to the wound6.
While deciding how to form the hydrogels for wound healing, there are multiple key aspects to consider to ensure that they are both effective and safe for use. Biodegradability and biocompatibility are some of the critical properties, since the hydrogel must not cause any adverse reactions from the body and must be able to naturally degrade over time without toxic residues14. Mechanical strength is a key consideration, as the hydrogel must maintain its structural integrity while conforming to the shape of the wound bed and allowing motion. In addition, the hydrogel must release the bacteriophages properly. Moisture retention is important, as it creates a conducive environment for both cellular migration and tissue regeneration. Hydrogels that provide a moist wound environment facilitate faster healing by inhibiting the formation of scabs and encouraging the development of new tissue15. Another critical thought is that hydrogels also need to be developed in such a way that it’s easy to incorporate the bacteriophages without compromising their stability and antimicrobial activity during treatment.
The difference in materials used for forming the hydrogels significantly influences their properties, function, and applicability in wound healing16. The natural polymers, alginate and chitosan are biocompatible and naturally antimicrobial. Alginate is derived from seaweed and forms hydrogels that are highly absorbent of exudates and maintain a moist environment at the wound site. Chitosan, derived from shellfish, has significant antimicrobial properties, thus making it particularly useful in the healing of infected wounds. These materials effectively mimic the extracellular matrix, meaning they promote tissue regeneration17. Synthetic polymers, such as polyethylene glycol (PEG), provide greater control over mechanical properties and degradation rates but may lack the bioactivity of natural polymers18. However, PEG use is preferable for specific applications where gelation must be controlled. Hybrid materials however, blend natural and synthetic constituents, providing a balance between these advantages, making them suitable for applications in wound healing19.
The physical properties of hydrogels are directly influenced by their composition, which dictates their swelling, moisture retention, and overall stability. Natural polymers, like alginate, chitosan, and gelatin, can provide a more favorable swelling behavior and moisture retention due to their biocompatibility and inherent hydrophilicity20. However, their mechanical properties can vary significantly depending on the degree of crosslinking and the presence of any additional chemical modifications. Synthetic polymers have more control over physical properties, particularly in regards to swelling, mechanical strength, and stability.
It is important to preserve the viability of the bacteriophages when they are embedded in the hydrogel in order to achieve their full antimicrobial activity. There are various strategies used to reduce phage damage or denaturation during the embedding process. Encapsulation is a widely used method in which the phages are entrapped in the hydrogel matrix through the use of physical forces like ionic or hydrogen bonding. It is used to shield the phages from the external environment, e.g., UV radiation and temperature fluctuations.
The ability to release the bacteriophages from the hydrogel is dependent on several factors that need to be controlled in order to have optimal effects. Of these, hydrogel degradation is the most significant trigger for phage release, as degradation of the matrix facilitates the gradual diffusion of phages into the wound. The degradation rate of hydrogel can be affected by its formulation, e.g., the type of polymer used and crosslink density. The lower the degradation rate, the longer phage release is, which is desirable when treating long-term infections. In addition, the diffusion rates of phages in the hydrogel matrix is another critical factor. The diffusion is a function of the molecular size of the phages, hydrogel porosity, and phage-polymer chain interaction. A slow diffusion rate is usually desired in a sustained release, which can be regulated by altering the structure of the gel network. External stimuli such as temperature or pH play a significant role in influencing the rate at which the bacteriophages are released. For example, acidic environments which are commonly found in infected wounds cause the breakdown of pH-sensitive hydrogels, causing the phages to be released at a faster rate and therefore decreasing effectiveness. Similarly, enzyme-degradable hydrogels can be made to respond to the presence of specific enzymes, e.g., those produced by bacteria, releasing phages in a targeted fashion.
The use of hydrogels combined with bacteriophages has numerous benefits compared to conventional wound treatment methods, which commonly involve the application of broad-spectrum antibiotics and antiseptics21. The efficacy of antibiotic-based treatments is gaining less traction due to new antibiotic-resistant bacterial strains emerging, therefore reducing their effectiveness in controlling infections22. Bacteriophages, on the other hand, are highly specific, targeting only pathogenic bacteria and leaving beneficial microbiota unharmed. Furthermore, as stated before, hydrogels provide a sustained and controlled release of bacteriophages, thus maintaining the antimicrobial properties at the site of infection for longer durations. This aspect eliminates the need for frequent dressing changes and enables uninterrupted treatment. Making this matrix of hydrogel and bacteriophages to be more effective and convenient for the patient. Furthermore, the possibility to customize the hydrogels based on the specific nature of the wound environment significantly increases their healing capabilities, therefore, providing a more effective approach than conventional techniques. While previous studies have explored phage-infused hydrogels, our approach uses a hybrid chitosan-alginate matrix paired specifically with T4r+ bacteriophages. This combination, applied directly to the gauze, more closely mimics real-world wound care scenarios than standard in-gel models.
The purpose of our project was to evaluate whether a bacteriophage-infused hydrogel could act as an innovative and effective alternative to traditional antibiotic treatments for bacterial infections. With the increasing threat of antibiotic resistance, there is an urgent need for new approaches for infection control, particularly in wound care. By integrating T4r+ bacteriophages into a chitosan-alginate hybrid hydrogel, we aimed to create a bandage capable of actively targeting and inhibiting the growth of Escherichia. coli K-12. We hypothesized that the bacteriophage-infused hydrogel bandage would effectively inhibit the growth of Escherichia coli K-12, resulting in a measurable zone of bacterial inhibition of at least 20 mm. Given the well known ability of T4r+ bacteriophages to infect and lyse Escherichia. coli, we expected that our engineered hydrogel would demonstrate significant antimicrobial activity while maintaining the structural and functional properties necessary for practical wound application.
Results
Discussion
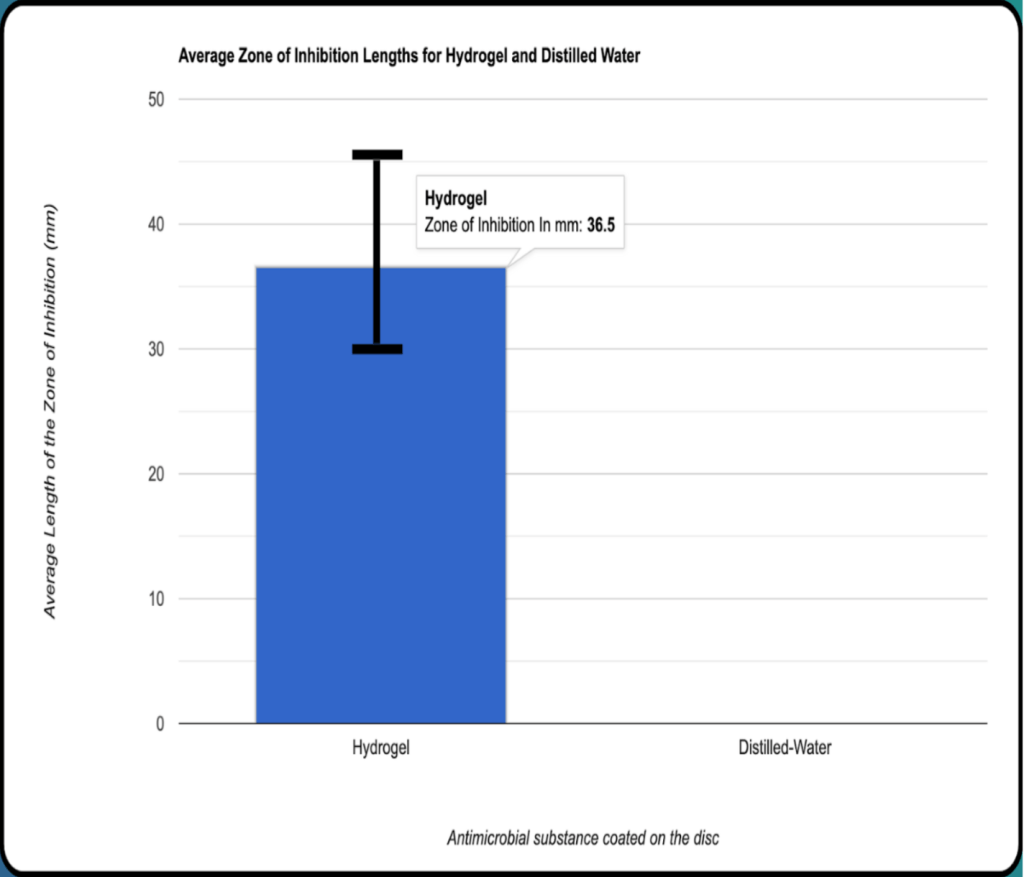
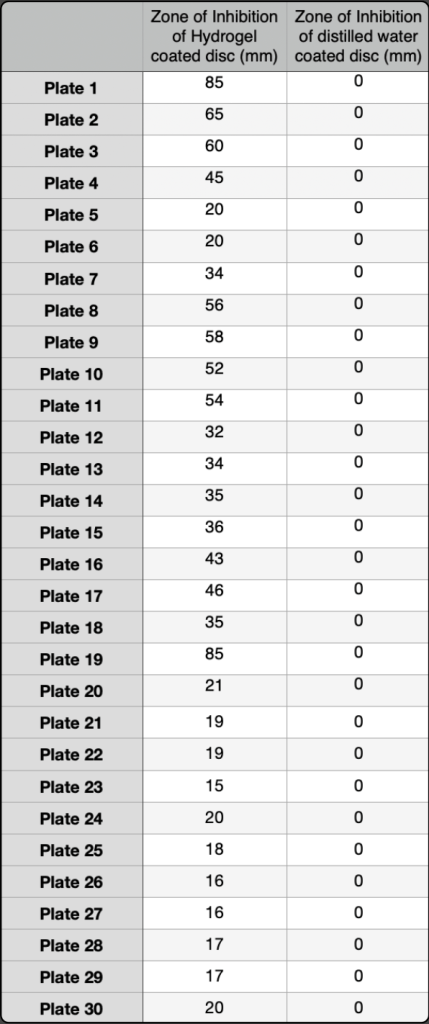
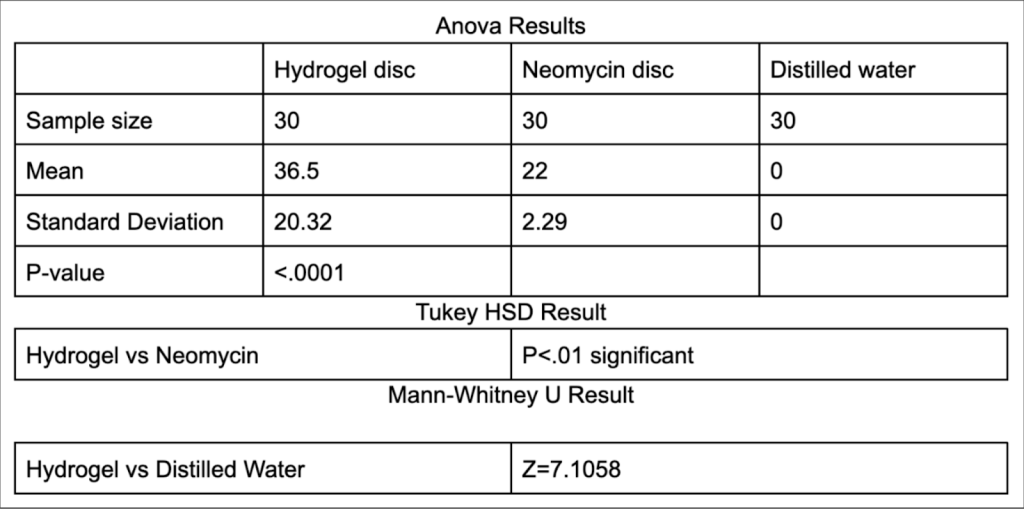
When comparing the averages of the sample sets for zones of inhibition size for distilled water and the hydrogel, the hydrogel’s average stands out as greater than that of the distilled water with the hydrogel’s mean/average being 36.5 mm and the distilled water’s mean/average being 0mm. From the ANOVA results for three data samples of zones of inhibition of substances coated onto sterile discs (hydrogel, neomycin from a previous controlled trial using identical
methodology, and distilled water), a P-value of less than .0001 is obtained. When comparing the data sets of the zones of inhibition of the hydrogel and neomycin in the Tukey HSD Test, a P-value of less than 0.01 is obtained.
Based on the results, the bacteriophage-infused hydrogel was effective in inhibiting bacterial growth. The mean zone of inhibition for the hydrogel was significantly larger than that of distilled water which was expected as distilled water has no antimicrobial properties itself. The Mann-Whitney U test yielded a high Z value of 7.1, confirming that the difference between the hydrogel and distilled water was statistically significant. Additionally, when compared to antibiotics, the Tukey HSD test showed a significant difference between the hydrogel and Neomycin, with a P value less than 0.01 This shows that there is a significant difference between the bacteriophage hydrogel and the neomycin . The ANOVA results also demonstrated a statistically significant difference among the groups (hydrogel, Neomycin, and distilled water), with a P-value less than 0.0001. All the results and conclusions support bacteriophage-hydrogel based treatments being used as viable alternatives for traditional antibiotics. The ability of the hydrogel to consistently inhibit bacterial growth shows its potential for tests involving more in vitro models which could potentially pave the way for tests involving humans.
Several uncontrollable factors may have influenced the results. Variations in bacteriophage concentration within the hydrogel could have affected its effectiveness. This could be prevented by leaving the hydrogel mixture to stir for an additional 20 minutes which would help spread out the bacteriophages uniformly within the hydrogel. Additionally, the bacteriophage’s ability to target and kill E. coli K-12 may have impacted the outcome. This could have been prevented if we tested the bacteriophage’s ability to kill the E. coli without the hydrogel mixture first. Also, environmental factors like temperature fluctuations could also have affected both the bacteriophages and bacteria. This could have been prevented by making sure the room that the experiment was performed in stayed the same temperature throughout the whole procedure. Other possible errors may have included not adding equal amounts of hydrogel to each disc before plating. If equal amounts of hydrogel were used, then the standard deviation of the zones of inhibition lengths could possibly be significantly less. This could be avoided in the future by using a pipette to get exact amounts of the hydrogel on each disc instead of dipping each disc into the whole hydrogel mixture.
Further research could possibly explore optimizing the hydrogel formula and testing its efficacy against a broader range of bacterial strains as well as comparing it to a broader range of antibiotics and antiseptics.
Materials/Methods
Materials:
Microbiology Equipment & Safety Gear
- BSL-1 Certified School Laboratory
- Latex Gloves
- Lab Coats
- Safety Goggles
- Bunsen Burner
- Ethanol (70% solution, 500 mL bottles)
- Clorox Disinfecting Wipes
- Bleach (10% & 20% solution)
Bacterial & Bacteriophage Culturing Materials
- Escherichia coli K-12 Strain (Lyophilized Culture)
- T4R+ Bacteriophage Stock (2×10⁹ PFU(plaque-forming units)/mL concentration)
- Mueller-Hinton Agar
- Sterile L-Shaped Spreaders (Polypropylene, individually wrapped)
Hydrogel Preparation Materials
- Sodium Alginate Powder (High Purity, 500 g, Sigma-Aldrich)
- Chitosan Powder (Food grade)
- Calcium Chloride (CaCl₂) Solution (0.5% w/v, 500 mL, anhydrous)
- Acetic Acid (Glacial)
- Distilled Water – Used for preparing all solutions and hydrogel formulations.
- Magnetic Stir Bar
- Hot Plate with Stirrer
- pH paper
- Beakers (100 mL, 500 mL, 1,000 mL)
- Graduated Cylinders (10 mL, 50 mL, 100 mL, Glass)
- Sterile Pipettes (1 mL, 5 mL, 10 mL, individually wrapped, sterile)
- Pipette Tips (Sterile, disposable)
Application & Data Collection Materials
- 4×4 8-Ply Non-Woven Sterile Gauze Pads (1 cm² Sections, Non-Adherent)
- Sterile Discs (1 cm diameter)
- Sterile Forceps (Stainless Steel, Fine-Tip)
- Digital Caliper (0.1mm Precision, stainless steel/plastic, LCD Display)
- Incubator (37°C, Digital Temperature Control, Forced-Air Circulation)
- Sterile Petri Dishes (100 mm × 15 mm)
Method/Procedure:
Step 1: Laboratory Preparation and Safety Measures
- Wash hands thoroughly with antibacterial soap and warm water.
- Apply PPE.
- Disinfect the work area using 70% ethanol and Clorox wipes.
- Prepare a 10% bleach solution for decontaminating used materials.
- Set up a Bunsen burner for aseptic techniques.
- Arrange all materials in an organized, sterile manner before proceeding.
Step 2: Preparation of Bacterial Lawn on Mueller-Hinton Agar Plates
- Make sure the Mueller-Hinton Agar (MHA) plates are at room temperature.
- Label all agar plates with names and dates for identification.
- Inoculate the agar plates: Pipette 150 µL of bacterial suspension onto the agar.
- Use a sterile L-shaped spreader to evenly distribute bacteria.
- Allow plates to sit and dry for 10 minutes for proper adhesion.
Step 3: Hydrogel Synthesis and Bacteriophage Infusion
- Prepare sodium alginate-chitosan hydrogel: In a 250 mL beaker, dissolve 2% (w/v) sodium alginate in 100 mL of distilled water.
- In a separate beaker, dissolve 1% (w/v) chitosan in distilled water, adjusting the pH to 6 with acetic acid.
- Stir both solutions at 50°C for 1 hour until fully dissolved.
- Combine hydrogel components: Slowly add the chitosan solution into the sodium alginate solution while stirring continuously, not the other way around.
- Crosslinking the hydrogel: Add 0.5% (w/v) calcium chloride solution in drop by drop while stirring.
- Stir for an additional 30 minutes to ensure uniform consistency.
- Slowly mix the bacteriophage solution into the hydrogel for 10 minutes while stirring gently to ensure even distribution/concentration of the bacteriophage solution.
Step 4: Hydrogel-Coated Bandage Preparation
- Cut sterile gauze into 1 cm² sections using sterile scissors.
- Using sterile forceps, dip each gauze piece into the hydrogel solution for 5 seconds.
Step 5: Application of Hydrogel-Coated Bandages to Bacterial Plates
- Using sterile forceps, place one hydrogel-coated bandage onto each agar plate.
- Prepare control samples: Soak sterile discs in distilled water.
- Using sterile forceps, place them onto separate agar plates.
- Press each sample down gently to ensure adhesion to the agar.
Step 6: Incubation and Measurement of Bacterial Inhibition
- Place all inoculated plates into an incubator set to 37°C.
- Incubate for 24 hours to allow full bacterial growth and phage activity.
- After incubation, take the plates out and measure the zones of inhibition using a digital caliper. Measure the diameter of the clear zones (in mm) around each bandage or sterile disc.
- Record all data in a spreadsheet for later analysis.
Step 7: Data Analysis and Statistical Testing
- Calculate the average inhibition zone size for hydrogel-coated bandages and control discs.
- Perform statistical tests to analyze results: One way is the ANOVA test that compares the hydrogel, neomycin, and distilled water groups. The Tukey HSD test compares the hydrogel vs. neomycin. While the Mann-Whitney test compares the hydrogel vs. distilled water. (Neomycin results are based on previous year’s project)
- Create bar graphs with error bars to visualize the differences.
Step 8: Cleanup and Safety Protocols
- Decontaminate all used materials by soaking them in 10% for bacteria only materials and 20% bleach for bacteriophage materials at least 30 minutes.
- Sterilize all of the workspace by wiping down surfaces using 70% alcohol and Clorox wipes.
- Dispose of all biohazardous waste according to laboratory protocols.
- Remove PPE and wash hands thoroughly.
Extra Notes: Maintain aseptic technique at all times to prevent contamination. Use a fume hood when handling chemicals like acetic acid. Use sterile forceps when handling bandages or discs to prevent cross-contamination.
Acknowledgements
We would like to extend our gratitude to Mrs. Jessica Loos for her support and guidance throughout our research project. We would also like to give our sincerest thanks to the Tracy Family Foundation and Science Coach for their generous funding of the materials we used for our project. Their support allowed us to buy the materials necessary to construct and test our bacteriophage-infused hydrogel bandage, thereby allowing us to complete this research. If it were not for their contribution, this project would not have been feasible. Last but not least, we would like to thank all the people who offered advice, support, and resources along the way of this research process. The information and support that we received added greatly to the success of our study, and we greatly appreciate the efforts of all involved.
References
- L. M. Kasman, L. D. Porter. Bacteriophages. StatPearls. (2022). [↩] [↩]
- L. Hudanich, C. Hudanich, J. Carey. Comparing Virulence of Three T4 Bacteriophage Strains on Ampicillin-Resistant and Sensitive E. coli Bacteria. Journal of Emerging Investigators. 3 (2020). [↩]
- H. Tashkandi. Honey in wound healing: An updated review. Open Life Sciences. 16, 1091-1100 (2021). [↩]
- R. Edwards, K. G. Harding. Bacteria and Wound Healing. Current Opinion in Infectious Diseases. 17, 91-96 (2004). [↩]
- H. A. Sohail, A. Coffey, K. Debrowska, I. M. Meyer, M. Middelboe, M. Sohail, M. RJ. Clokie. Bacteriophages: Emerging Applications in Medicine, Food, and Biotechnology. Phage Therapy, Applications, and Research. 1, 75-82 (2020). [↩] [↩]
- A. M. Pinto, M. A. Cerqueira, M. Bañobre-Lópes, L. M. Pastrana, S. Sillankorva. Bacteriophages for Chronic Wound Treatment: From Traditional to Novel Delivery Systems. Viruses. 12, 235 (2020). [↩] [↩] [↩]
- D. M. Lin, B. Koskella, H. C. Lin. Phage therapy: An alternative to antibiotics in the age of multi-drug resistance. World Journal of Gastrointestinal Pharmacology and Therapeutics. 8, 162-173 (2017). [↩] [↩]
- J. Y. Lim, J. W. Yoon, C. J. Hovde. A Brief Overview of Escherichia coli O157:H7 and Its Plasmid O157. Journal of Microbiology and Biotechnology. 20, 5-14 (2010). [↩] [↩]
- B. Brockway. Introducing Escherichia coli (E. coli) part 1. https://thesecretlifeofskin.com/2020/11/10/introducing-e-coli/# (2020). [↩] [↩]
- N. M. Hitchcock, D. D. G. Nunes, J. Shiach, K. V. S. Hodel, J. D. V. Barbosa, L. A. P. Rodrigues, B. S. Coler, M. B. P. Soares, R. Badaró. Current Clinical Landscape and Global Potential of Bacteriophage Therapy. Viruses. 15, 1020 (2023). [↩]
- V. H. Pérez-Luna, O. González-Reynoso. Encapsulation of Biological Agents in Hydrogels for Therapeutic Applications. Gels. 4, 61 (2018). [↩]
- M. Li, D. Shi, Y. Li, Y. Xiao, M. Chen, L. Chen, H. Du, W. Zhang. Recombination of T4-like Phages and Its Activity against Pathogenic Escherichia coli in Planktonic and Biofilm Forms. Virologica Sinica. 35, 651-661 (2020). [↩]
- V. Beschastnov, M. Ryabkov, A. Leontiev, A. Tulupov, T. Yudanova, I. Shirokova, N. Belyanina, O. Kovalishena. Viability of Bacteriophages in the Complex Hydrogel Wound Dressings in vitro. Sovremennye Tehnologii v Medicine. 13, 32-38 (2021). [↩]
- X. Ma, X. Sun, D. Hargrove, J. Chen, D. Song, Q. Dong, X. Lu, T. Fan, Y. Fu, Y. Lei. A Biocompatible and Biodegradable Protein Hydrogel with Green and Red Autofluorescence: Preparation, Characterization and In Vivo Biodegradation Tracking and Modeling. Scientific Reports. 6, 19370 (2016). [↩]
- G. Olteanu, S. M. Neacşu, F. A. Joița, A. M. Musuc, E. C. Lupu, C. Ioniță-Mîndrican, D. Lupuliasa, M. Mititelu. Advancements in Regenerative Hydrogels in Skin Wound Treatment: A Comprehensive Review. International Journal of Molecular Sciences. 25, 3849 (2024). [↩]
- T. Ho, C. Chang, H. Chan, T. Chung, C. Shu, K. Chuang, T. Duh, M. Yang, Y. Tyan. Hydrogels: Properties and Applications in Biomedicine. Molecules. 27, 2902 (2022). [↩]
- N. Wathoni, Y. Herdiana, C. Suhandi, A. F. A. Mohammed, A. El-Rayyes, A. C. Narsa. Chitosan/Alginate-Based Nanoparticles for Antibacterial Agents Delivery. International Journal of Nanomedicine. 19, 5021-5044 (2024). [↩]
- S. P. Zustiak, J. B. Leach. Hydrolytically degradable poly(ethylene glycol) hydrogel scaffolds with tunable degradation and mechanical properties. Biomacromolecules. 11, 1348-1357 (2010). [↩]
- C. Vasile, D. Pamfil, E. Stoleru, M. Baican. New Developments in Medical Applications of Hybrid Hydrogels Containing Natural Polymers. Molecules. 25, 1539 (2020). [↩]
- X. Che, T. Zhao, J. Hu, K. Yang, N. Ma, A. Li, Q. Sun, C. Ding, Q. Ding. Application of Chitosan-Based Hydrogel in Promoting Wound Healing: A Review. Polymers. 16, 344 (2024). [↩]
- S. Abed, M. Beig, S. M. Barzi, M. Shafiei, A. H. Shahraki, S. Sadeghi, A. Sohrabi. Development of phage-containing hydrogel for treating Enterococcus faecalis-infected wounds. PLoS ONE. 19, e0312469 (2024). [↩]
- C. L. Ventola. The Antibiotic Resistance Crisis. Pharmacy and Therapeutics. 40, 277-283 (2015). [↩]