Abstract
The precise and effective evaluation of surgical recovery presents significant challenges in the medical field, and current methods for evaluating recovery can be invasive, subjective, and expensive. Fluorescent silk, produced by a modified silkworm diet, has been proposed to have biomedical applications due to its desirable properties. In this study, silkworms were fed a diet supplemented with the fluorescent dyes Rhodamine B and Rhodamine 110, and a metal additive of Copper for approximately 5-14 days. The cocoons from these silkworms were then analyzed to determine whether silk fibers can be engineered to contain multiple fluorescent properties. The silk threads were then immersed in synthetic blood to determine how an environment like that inside of a human body may impact the fluorescent properties. Results show silkworms can produce multi-fluorescent silk that is similar in size and has up to 70% of the breaking load of commercially available surgical sutures (420-480g breaking force). Silk fibers containing green fluorophores showed around 50% reduction in fluorescence intensity after one week immersion in synthetic blood while fibers containing red fluorophores showed little change. Additionally, the concentration of chemicals the silkworms were fed affected their growth rate. The silk produced through the methods described in this study is comparable to that of synthetic sutures in size and function with the additional property of being multi-fluorescent. The differential degradation rates observed in a simulated physiological environment demonstrate the feasibility of using multi-fluorescent silk as a sensor for blood exposure. This suggests a potential clinical application where fluorescence loss may be correlated, following in vivo validation, with perfusion-dependent wound healing.
Keywords: Bombyx mori, Fluorescence, Surgical Sutures, Rhodamine B, Wound Monitoring
Background
A significant and ongoing challenge in the medical field is precisely and effectively evaluating recovery after patients undergo a surgical operation1 . For internal operations in particular, where sutures are applied on tissues that lie beneath the skin, current methods for evaluating recovery can be invasive, inaccurate, and expensive. Aside from asking a patient how they are feeling to gauge recovery, doctors rely on a visual inspection to determine the progression of healing, thus accurately monitoring wound healing of an internal site of operation is especially difficult. Additionally, there are currently no minimally invasive methods to track cumulative blood exposure of body tissue over time. This project aims to fill that gap by engineering a thread with fluorescent properties that degrade when immersed in synthetic blood, establishing a material that can potentially be applied as a perfusion-based recovery indicator.
5,000 years ago, the wild silkworm, Bombyx mandarina, was domesticated by Chinese farmers for the production of silk, and this gave rise to what is now known as the silkworm, Bombyx mori 2. In addition to their value as a raw material for textiles, the silk produced by Bombyx mori has been found to be biodegradable, have excellent mechanical properties, and be biocompatible with the human body3. In the field of biomaterials engineering, there has been a growing interest in advancing the various applications of fluorescent silk in a variety of fields, such as bioimaging, tissue engineering, and drug delivery4.
In the past couple of years, scientists have been developing a reliable way to create stable, fluorescent silk through the process of feeding silkworms a diet supplemented with fluorescent additives, such as Rhodamine B (RhB) and Rhodamine 110 (Rh110)4. When these additives are combined into the silkworms’ feed, these small fluorescent molecules are ingested, and absorbed into the silkworm’s body. They are then transported into the glands where silk is produced. The fluorescent molecules then become incorporated into the developing silk proteins either by being embedded within the fibroin matrix or binding to sericin proteins, giving the fibers stable fluorescence at the molecular level3. Methods to reliably produce silk that exhibits a single color of fluorescence have been established4. However, it remains unknown whether this process can be modified to have silkworms incorporate multiple fluorophores into their silk to create silk threads with fluorescence of more than one color. It is also unknown whether the process of feeding silkworms multiple fluorescent molecules may affect the incorporation of those molecules into the silk as well as the subsequent fluorescence emission intensity. Being that silkworm silk has already been used in biomedical applications and a process for generating single-fluorescence silkworm thread has been established, remaining gaps in knowledge highlight the importance to further investigate whether multi-fluorescent silk can be produced reliably and whether it could have biomedical use.
Fluorescence occurs when a molecule absorbs light at one wavelength and then emits light at a different wavelength of a visible color5. In biology research, fluorescence microscopy is commonly used to track the production and migration of molecules in cells as well as mark specific tissues to visualize physiological processes since fluorescent molecules provide high contrast colors and are localized to specific areas6. One such fluorophore, Rhodamine B (RhB), a chemical used in this study, absorbs green-yellow light and emits orange-red light. Rhodamine 110 (Rh110), another chemical used in this study, absorbs blue-green light and emits green light4. RhB and Rh110 are organic, fluorescent small molecule dyes that are typically used in fluorescence sensing and imaging due to them being cost-effective and having an outstanding fluorescence quantum yield4. Additionally, RhB and Rh110 have been shown to be utilized in applications such as environmental monitoring and in laser dyes. While elemental copper (Cu) is not typically used as a fluorophore like RhB, some types of copper on the other hand, has been shown to induce fluorescence in biomaterials through the formation of metal-protein complexes or nanoclusters, and these have been used as biocompatible probes for bioimaging and biosensing, particularly in early disease detection7.
One particular characteristic of fluorophores that this study aims to leverage is that the intensity of fluorophores has been shown to degrade at different rates: the emissions intensity of red-shifted fluorophores tend to be more photostable and degrade at a slower rate than green-shifted fluorophores8. In considering how the internal environment of a warm-blooded mammalian body can impact fluorescence through potential chemical and enzymatic degradation, the optical properties of blood itself may influence fluorescence visibility. In particular, the primary oxygen-carrying protein in blood, hemoglobin, may play a role. Due to hemoglobin having similar absorption peaks in the visible spectrum as green fluorophores (540nm–580nm), environments that have ample hemoglobin may actually be able to conceal green fluorescence as a result of the overlap. This spectral overlap means that the hemoglobin present in the body’s wounds may be able to absorb parts of the emitted green signal, reducing its observed fluorescence intensity from the surrounding background. This filtering effect further supports the hypothesis that multi-fluorescent silk exposed to a blood-like environment has the potential to exhibit differential signal decay depending on fluorophore wavelength stability and overlap with hemoglobin absorption within the spectrum. Therefore, it is hypothesized that silk threads containing several fluorophores of different colors can be used as a way to indicate the extent of perfusion, a proxy of healing. Additionally, past studies have shown that pH fluctuations and oxidative reactions, all things that can occur in blood may result in structural modifications or degradation of the emissions intensity of fluorescent molecules9. Together, these chemical and optical mechanisms support the idea that surgical sutures created from silk threads with multi-fluorescent properties will exhibit a predictable reduction in fluorescence intensity after being submerged in blood or a blood substitute.
In the context of post-operative recovery following an invasive surgical procedure that requires internal sutures, adequate blood flow to provide oxygen through the surgical site is one of the strongest indicators of tissue healing and recovery10. Oxygenation of a wound is crucial for the healing process, as impaired perfusion is associated with delayed wound healing and infection risk11. Interestingly, these same properties of blood that serve to benefit the body may also act to degrade the fluorescence intensity of silk engineered through the method previously described by destabilizing fluorophores, leading to diminished fluorescence8,9.This suggests that sutures made from fluorescent silk have the potential to be used, with in vivo validation, to monitor the presence of perfusion in an area over time. Moreover, the inclusion of different fluorophores in a single strand of silk may result in a temporally predictable pattern of fluorescence degradation, since different fluorophores may degrade at different rates as previously described.
This project aims to develop a novel material that exhibits a change in its fluorescent properties over time after being exposed to an environment that mimics the perfusion that occurs within the human body. A potential clinical application of the material aimed to be developed in this study is utilizing the multi-fluorescent silk as a material for sutures, allowing doctors to track the process of recovering surgeries through a change in fluorescence intensity over time. Measuring the change in fluorescence could thus provide a minimally invasive way to monitor healing. This study aims to take the first steps toward making post-operative recovery monitoring more accurate, decreasing the chances of patients reinjuring themselves and increasing the total number of patients that doctors can attend to. This study utilizes silkworms to produce multi-fluorescent silk in vivo, followed by experiments that investigate the behavior of the silk produced in a physiological environment that is simulated in vitro.
Methods
The independent variables in this study were the type of dietary additive (Rhodamine B, Rhodamine 110, Copper) and the concentration of these additives in the silkworm diet (Low, Medium, High). The dependent variables measured were the fluorescence intensity (quantified via ImageJ), the mechanical breaking load of the fibers (grams), and the degradation rate of fluorescence over time.
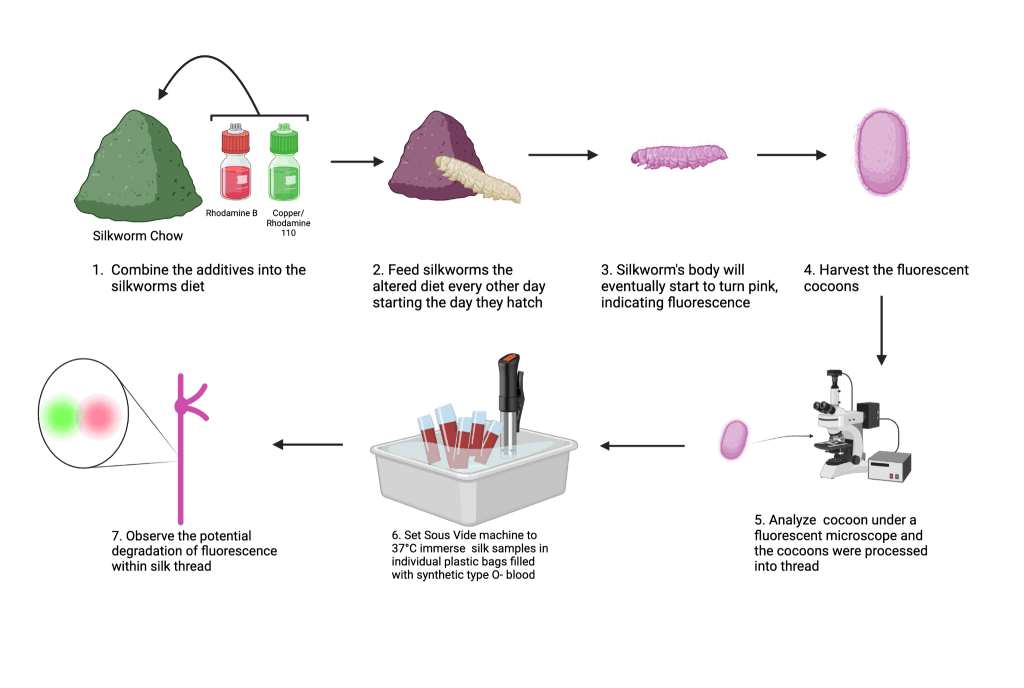
For the production of silk, juvenile (2-week old) silkworms were obtained from Coastal Silkworms and raised by feeding them with a pre-made chow (Coastal Silkworms SWF-P1). Worms were split into 7 groups (6 experimental and 1 control), each group representing either a high, medium or low concentration of a combination of either RhB and Rh110 or RhB and Cu. At each daily feeding, the appropriate combination and concentration of fluorescent chemicals were mixed into the chow before being placed into the container where the silkworms were housed. This modified diet was fed to the silkworms until all of the worms in each group cocooned (n=14). The cocoons were then collected, examined under a fluorescent microscope to observe color emission and fluorescence intensity (Figure 1), then degummed following established protocols to isolate single fibers12.
To replicate one aspect of the human body’s internal environment, a sous vide immersion circulator was used to create a water bath that was held at a constant temperature of 37°C, the same as the normal homeostatic temperature of humans. The cocoons collected from each of the seven groups were then degummed and placed into individual bags containing synthetic Type O- blood (Carolina 700173). These bags were then immersed in the 37°C water bath and the entire apparatus was left in a dark environment. The control and experimental silk threads were removed from the blood following one and three weeks of immersion, and the fluorescence emission of these threads were examined using a fluorescence microscope.
Images of the silk were taken using an AmScope FM200B upright epi-fluorescence microscope that was equipped with a 100W Mercury HBO arc-lamp illumination and a C-mount digital microscope camera. To be able to compare samples across the timepoints, the microscope settings such as the exposure time, gain, and excitation light intensity were kept constant across all imaging time points. Samples that included Rhodamine 110 or Copper were imaged using the Blue Excitation filter block (Excitation: ~450-490 nm; Emission: ~520 nm), and Rhodamine B and Copper samples were imaged using the Green Excitation filter block (Excitation: ~510-560 nm; Emission: ~590 nm).
ImageJ software was used to analyze images of the silk. To quantify the intensity of fluorescence, the channels of each image were split using ImageJ, and a Region of Interest (ROI) was manually defined to outline a single fiber of silk. Then, the Mean Gray Value (fluorescence intensity) was measured for the ROI of the fiber. To keep the measurements as accurate as possible, three random background regions near each fiber were measured and averaged. These two values, the fluorescence intensity of the outlined fiber, and background were subtracted from each other to obtain final fluorescence intensity (IFinal = IFiber – IBackground). Quantitative data (Table 3 and 4) are presented as mean ± standard deviation (SD). The sample size was n=14 individual silkworms or silk threads for each experimental condition. All data processing and descriptive statistical calculations were performed using Google Sheets. One-way ANOVA was used to compare fluorescence intensity and mechanical properties across experimental groups at a certain timepoint. Two-way ANOVA was then used in order to evaluate the combined effects of additive composition and immersion time on fluorescence degradation. A p-value of less than 0.05 (p<0.05) was considered statistically significant. Analysis was conducted using XLMiner Analysis ToolPak through Google Spreadsheets.
Finally, to assess strength of the silk threads and compare it against commercially available sutures to determine whether the multi-fluorescent silk can be a viable suture material, the amount of static weight that the multi-fluorescent silk thread from the control and experimental could hold before breaking was determined. For mechanical and fluorescence testing, a total of n=14 distinct silk threads were processed and averaged per condition.
All silkworms were housed in a temperature controlled environment with regular and adequate food supply according to the supplier’s instructions. Handling of chemicals, specifically RhB and Cu, was done according to standard laboratory safety protocols, including the use of PPE and proper hazardous waste disposal, to ensure researcher safety and environmental protection.
Results
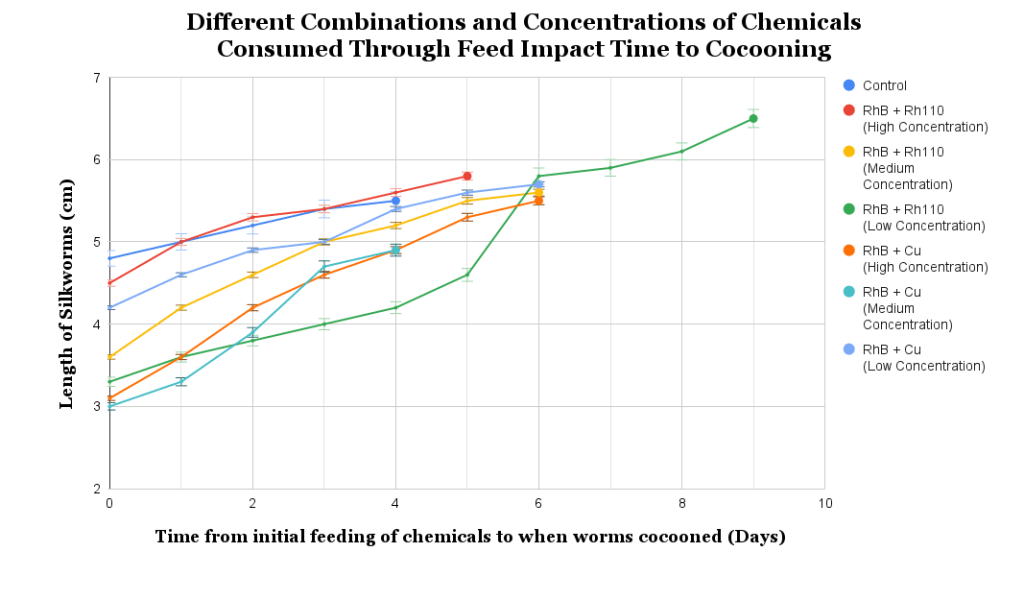
The silkworms were fed low, medium, and high concentrations of a combination of RhB and Rh110, and RhB and Cu, respectively (Table 1 and 2), and it was observed that the composition of the chow fed to the silkworms significantly affected their growth (F (6,91) = 6.936, p < 0.0001). As shown in Figure 2, the low concentration RhB and Rh110 group exhibited the greatest mean growth (6.49 ± 0.83), representing a notable increase compared to the control group (5.50 ± 0.44). On the other hand, the medium concentration of the RhB and Cu combination showed the least growth (4.81 ± 0.89). The F-value (6.936) significantly exceeded the critical F-value of 2.199, suggesting that observed variations were associated with the presence of chemical additives rather than random biological variance (n2 = 0.315).
| High | Medium | Low | |
| Rhodamine B from Kremer Pigmente (94900.12025.108) | 1.00g | 0.50g | 0.25g |
| Rhodamine 110 from Sigma Aldrich (83695) | 0.02g | 0.01g | 0.005g |
| High | Medium | Low | |
| Rhodamine B from Kremer Pigmente (94900.12025.108) | 1.00g | 0.50g | 0.25g |
| Copper Powder from Home Science Tools (7440-50-8) | 3.00g | 2.00g | 1.00g |
The combinations of chemicals, at all three concentrations, resulted in both red and green fluorescence being observed in the silk. All of the cocoons formed by worms that consumed RhB exhibited a consistent bright red fluorescence. The addition of Rh110 and Cu into the worms’ diet resulted in the emission of a dimmer but distinct green fluorescence (Figure 3). Within the varying chemical concentrations, the brightness of the red and green fluorophores were observed to be very similar across the different concentrations of the RhB and Rh110 combination. However, within the combination of RhB and Cu, the low concentration was the dimmest while the high concentration was the brightest, suggesting that the fluorescence intensity followed a linear pattern. Linear regression analysis of the fluorescence from the RhB and Cu groups showed a strong positive correlation between copper concentration and red fluorescence intensity (R² = 0.91, p < 0.01), supporting the pattern of high concentrations of chemicals resulting in higher fluorescence intensity in silk fibers in this particular group.
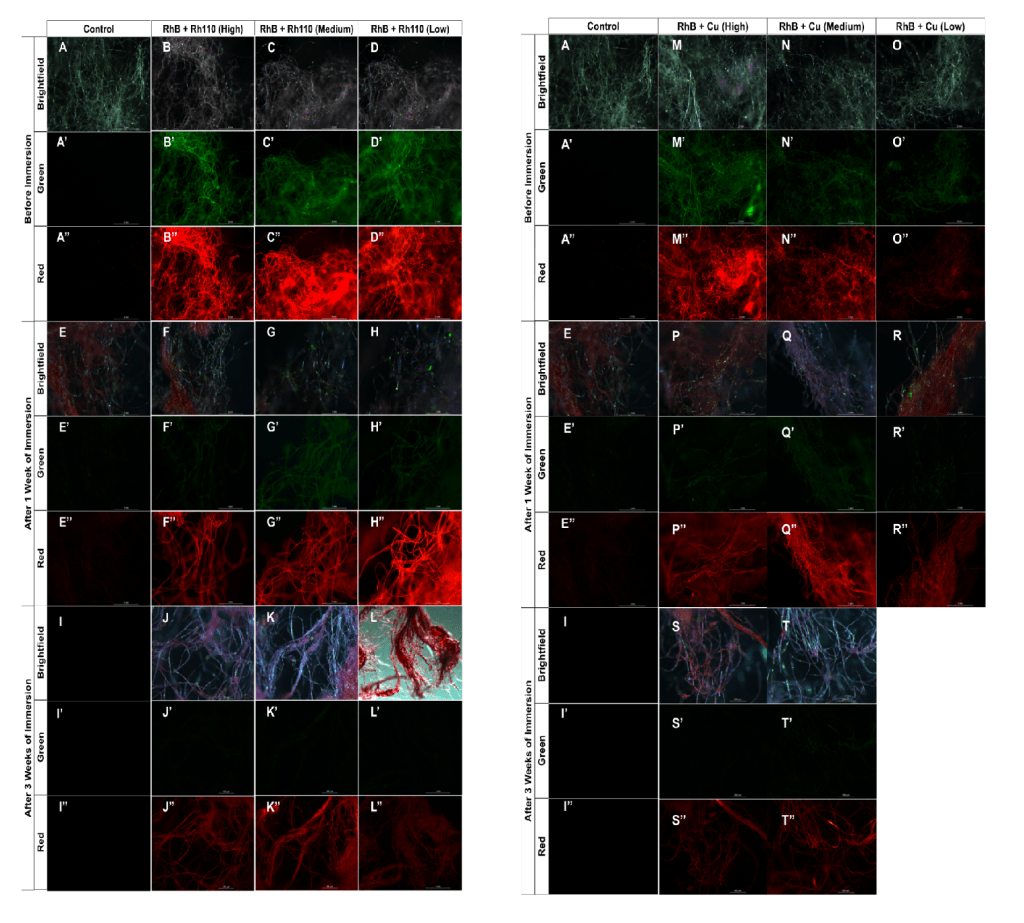
After the images of the silk from the fluorescent cocoons were taken prior to immersion initially, the fluorescent cocoons were degummed and then spun into a thread to prepare for immersion in synthetic Type O- blood. Three silk threads from the six experimental and one control group were removed from immersion after one week, imaged using an epifluorescence microscope, and processed via ImageJ to quantify the change in fluorescence. It was found that the majority of green fluorescence produced by both the Cu and Rh110 was lost after immersion, while the red produced by RhB remained consistently present in the silk (Figure 3).
Following one week of immersion, the red fluorescence of the RhB and Rh110 groups remained relatively stable, decreasing by 25%, 20%, and 6% at high, medium, and low concentrations, respectively. In contrast, green fluorescence declined substantially, decreasing by 82%, 40%, and 73% across the same concentration levels. Similarly, the RhB and Cu groups exhibited reductions in green fluorescence of 61%, 69%, and 51% at high, medium, and low concentrations, respectively. Red fluorescence in the RhB and Cu groups demonstrated greater variability, decreasing by 59%, 1%, and 52% across high, medium, and low concentrations (Tables 3 and 4).
After three weeks, a second batch of silk from the experimental and control groups was removed from synthetic blood. Using the same imaging and quantification workflow, green fluorescence intensity across all concentrations in both fluorophore combinations diminished almost entirely. In the RhB and Rh110 groups, red fluorescence decreased by 82%, 56%, and 88%, while green fluorescence decreased by 92%, 82%, and 85% at high, medium, and low concentrations, respectively. In the RhB and Cu groups, red fluorescence decreased by 69%, 33%, and dissolution occurred in the low concentration group prior to measurement, while green fluorescence decreased by 90%, 82%, and dissolution prevented quantification at the low concentration (Tables 3 and 4).
A two-factor ANOVA evaluating additive composition and immersion time indicated significant degradation trends to evaluate the effects of time and additive composition on fluorescence intensity. For all experimental groups, a highly significant degradation in both red and green fluorescence was observed over the 3-week immersion period (p < 0.001). Notably, green fluorescence exhibited a more rapid initial decline (averaging an 81% reduction by Week 1 for the RhB and Rh110 high concentration group) compared to red fluorescence, supporting the idea that there would be different degradation rates for multi-signal sensing.
| RhB + Rh110 (H) | RhB + Rh110 (M) | RhB + Rh110 (L) | RhB + Cu (H) | RhB + Cu (M) | RhB + Cu (L) | |
| Before | 76.203 ± 5.58 | 59.207 ± 1.92 | 68.246 ± 1.11 | 63.414 ± 2.22 | 51.913 ± 9.59 | 30.038 ± 2.49 |
| 1 week | 56.796 ±1.84 | 47.345 ± 6.27 | 63.964 ± 10.47 | 26.017 ± 6.20 | 51.539 ± 4.40 | 14.425 ± 3.25 |
| 3 week | 13.503 ± 4.58 | 26.106 ± 0.40 | 7.879 ± 0.51 | 19.466 ± 1.86 | 34.731 ± 6.75 | N/A |
| RhB + Rh110 (H) | RhB + Rh110 (M) | RhB + Rh110 (L) | RhB + Cu (H) | RhB + Cu (M) | RhB + Cu (L) | |
| Before | 37.024 ± 2.39 | 18.48 ± 1.48 | 18.050 ± 1.62 | 22.978 ± 5.04 | 20.830 ± 5.74 | 12.757 ± 1.34 |
| 1 week | 6.778 ± 0.32 | 11.173 ± 2.19 | 4.808 ± 4.42 | 9.055 ± 3.40 | 6.471 ± 2.98 | 6.290 ± 2.82 |
| 3 week | 2.794 ± 1.22 | 3.400 ± 0.81 | 2.634 ± 0.29 | 2.359 ± 0.93 | 3.848 ± 0.57 | N/A |
To quantify and evaluate differences in fluorescence intensity before and after immersion among additive formulations, statistical analyses were performed on the fluorescence measurements taken after each immersion timepoint. One-way ANOVA demonstrated significant differences in red fluorescence intensity among all additive combinations and concentrations prior to immersion (F(5,78) = 61.2, p < 0.001, n² = 0.80), after one week of immersion (F(5,78) = 54.6, p < 0.001, n² = 0.78), and after three weeks of immersion (F(4,65) = 47.3, p < 0.001, n² = 0.74), indicating substantial variation in fluorescence. Significant differences in green fluorescence intensity were also observed across additive groups prior to immersion (F(5,78) = 19.1, p < 0.001, n² = 0.55), after one week (F(5,78) = 8.4, p < 0.001, n² = 0.35), and after three weeks (F(4,65) = 3.9, p = 0.007, n² = 0.19), indicating that the degradation of green fluorescence between the weeks were significantly different.
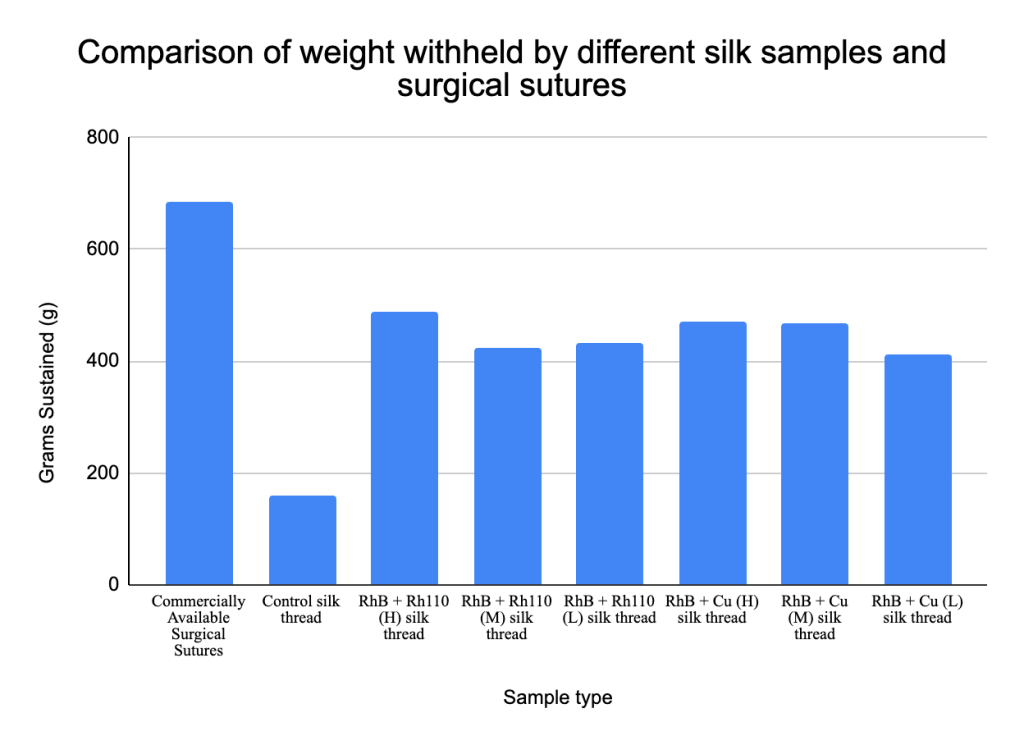
In order to test their potential application as a material to be made into sutures, the silk was spun into threads and then compared to commercially available sutures in terms of their strength. A test was conducted to determine the mechanical breaking load of the multi-fluorescent silk, and it was found that the experimental silk was approximately 70% the strength of commercially available surgical sutures. All of the silk from the experimental groups were able to sustain at least 420 grams of downward weight, with the maximum weight sustained being 480 grams by the RhB and Rh110 high concentration group (Figure 4).
The data collected suggest that incorporation of additives enhanced the mechanical strength of the silk fibers. The RhB and Rh110 (High) silk thread achieved a breaking load of 488g, representing a 206.9% increase (3.06-fold) over the Control silk thread (159g). The RhB and Cu (High) variant also showed a notable increase in reinforcement, reaching a load of 471 (196.2% increase). While these values are lower than the tested commercially available surgical sutures (686g), the data indicated that the chemical modifications may have bolstered the strength of the silkworm silk matrix. To ensure that the differences in breaking load among the silk variants and commercially available silk sutures were significant, a one-way ANOVA was conducted. It revealed that the additives impacted the breaking load of the sutures (F(7, 104) = 72.4, p < 0.001, n2 = 0.83), indicating that all additive containing silk fibers demonstrated substantially higher breaking loads as a result of the additives, not confounding factors.
Discussion
In this experiment, silkworms fed supplemented diets were able to produce silk that is both multi-fluorescent, and comparable in strength to modern day sutures. Throughout this study, it was also discovered that there were distinct differences in fluorescence intensity and mechanical strength depending on the combination and concentrations used. Specifically, the red fluorescence stemming from RhB was found to be more photostable than green fluorescence, whether RhB was combined with either Rh110 or Cu, the RhB containing silk threads retained a visible red signal even after three weeks of immersion in synthetic blood. This observation was actually consistent with previous research that showed red-shifted fluorophores degrading at slower rates than green fluorophores8. On the other hand, the green fluorescence was both dimmer initially and the fluorescence intensity decreased more rapidly following immersion in synthetic blood. One possible reason that the green fluorescence initially appeared dimmer could be caused by the green-emitting species of the Rh110 and Cu being inefficiently incorporated into the silk fibers during the feeding process. However, further analysis of the engineered silk threads would need to be conducted to confirm the amount of fluorescent material integrated into the fibers. The observation that the green fluorescence dimmed more rapidly following immersion supports the idea that some green-emitting species, such Rh110 and Cu, dim more quickly than red fluorophores (from RhB) in an environment like that of blood8. It should be noted that while RhB and Rh110 are intrinsic fluorophores, elemental copper is typically not. The green fluorescence observed in the experimental groups where copper was supplemented into the silkworm chow is likely not due to the element copper itself, but can be attributed to the formation of copper nanoclusters or copper-protein complexes within the fibroin matrix during the silk spinning process7.
It is important to acknowledge that the experimental design described in the methodology varied the concentration of fluorescent additives (RhB/Rh110 and RhB/Cu) simultaneously within the diet mixtures. Consequently, the observed effects on growth rate, fluorescence intensity, and mechanical strength represent a result caused by the presence of two additives together rather than the effect of a single additive. While this approach successfully identifies a baseline combination and concentration for generating multi-fluorescent silk, it precludes an important factor: the definitive attribution of an observed outcome to a specific chemical additives, whether it be the joining of the additives or one chemical itself. Future studies should test single-additive controls to first confirm that the color of fluorescence seen was the result of the respective chemical, and to discover what specific uptake efficiencies, toxicities, and impacts to physical integrity that each fluorophore has on the silk threads. Additionally, in preliminary experiments, higher concentrations of the same chemicals used in this study were applied, but resulted in worm lethality before reaching the cocooning stage. Thus, further work can be done to optimize fluorescence intensity while minimizing stress to the silkworms.
It should be noted that the observed decrease in fluorescence among the different concentrations may be attributed to other factors, one them being the absorption of hemoglobin into the silk threads. Hemoglobin exhibits strong absorption bands in the 540–580 nm wavelength range, and this also happens to overlap with the excitation and emission of green fluorophores13,14. This absorption can mask both the excitation light that reaches the fluorophore and also the observed fluorescence that is displayed on the microscope, making it seem as if chemical degradation occurred when it did not. Interestingly, threads containing RhB exhibited less red fluorescence degradation compared to the reduction in green fluorescence observed in the Rh110 containing groups. This difference most likely reflects the inherent photochemical stability of RhB rather than the interaction between the two fluorophores. The chemical structure of Rhodamine B includes alkyl substitutions that increase resistance to hydrolytic and photochemical degradation in comparison to the less substituted structure of Rhodamine 110, which may contribute to the differential signal persistence observed in this study15. Another possible explanation for the general decrease in fluorescence is the leakage of the fluorophores from the fiber matrix into the surrounding solution. While chemical degradation leading to fluorescence reduction is a desired mechanism for how this engineered silk functions as an indicator of healing, distinguishing between fluorescence degradation by blood versus leakage of fluorophores from the silk is critical for accurately interpreting the observed results.
It is important to note, however, that the degradation of fluorescence following immersion in synthetic blood may not recapitulate what may occur in an in vivo environment to clinically measure for recovery. Since adequate blood perfusion is a physiological prerequisite for wound healing, the predictable decay of the green signal against the stable red signal supports the hypothesis that this material could indicate the presence of blood flow in the wound. While this study establishes the behavior of this engineered silk in an in vitro environment, achieving clinical application would require correlating this optical fading with specific perfusion milestones in order to prove that multi-fluorescent silk could serve as an indicator of surgical recovery, utilizing the mechanism of different colored fluorophores fading at different rates to serve as a readout of cumulative biochemical changes in the surgical site.
An important result to consider is the complete dissolution of the low concentration RhB and Cu silk fibers following three weeks of immersion in synthetic blood; there may be some dynamic between this particular combination of chemicals and that of silkworm silk that interferes with the silk fibers’ integrity. While biodegradability is a desirable property for certain sutures, uncontrolled or premature dissolution poses a clinical risk of wounds re-opening. The complete dissolution of the low concentration RhB and Cu formulation suggests that insufficient additive incorporation has the potential to compromise fiber integrity under long periods of immersion. However, because the groups that did not contain copper (RhB/Rh110 groups) maintained their integrity, it can be inferred that copper incorporation is not inherently necessary for silk stability. Rather, the dissolution of the thread suggests that the overall additive amount and its interactions with the fibroin matrix likely influenced the structural durability. Future studies are needed to determine whether copper contributes to stabilization under specific concentration thresholds or whether observed effects are driven by broader changes in additive distribution within the fiber.
During mechanical tests of the experimental and control groups, the experimental silk exhibited a significantly higher breaking strength (420–480g) compared to the control silk (159g). This significant increase in strength may be attributed to the additives serving as a structural reinforcement within the silk fibroin matrix. This observed reinforcement may be attributed to the incorporation of Copper ions (Cu2+) with the amide bonds in the protein backbone, as previous studies have shown that metal ions such as Cu2+ and K+ can result in coils to form crystalline 𝛽-sheets, and in turn enhancing the mechanical properties and toughness of the fiber16. Furthermore, the incorporation of RhB may also contribute to the molecular strengthening of the silk. It may act as a molecular filler, where the RhB molecules occupy empty space within the fibers or form stable hydrogen bonds within the fibroin, which results in a stronger silk17. It is also possible that the presence of these additives protected the fibroin from hydrolytic degradation during the degumming process, resulting in a more intact and robust fiber compared to the control.
Mechanically, the silk produced from this study possessed approximately 70% of the breaking load of commercially available sutures. The difference in strength between the experimental silk and the commercially available sutures may be due to the removal of sericin during the degumming process, where individual silk threads are unwound from whole cocoons by boiling in an alkaline solution, which can weaken peptide bonds within the fibroin and in turn reduce the silk’s molecular weight and tensile strength18. Additionally, commercially available silk-based sutures undergo optimized twisting or braiding processes to enhance drug distribution and reduce friction, structural advantages that the engineered silk material lacked19. Although not yet equivalent in strength, initial measures of the breaking load shows that multi-fluorescent silk has the potential to be used as viable as an alternative suture material following further optimization of methods.
A significant limitation in the mechanical characterization is that only breaking load (grams) was measured, rather than tensile strength (MPa). Since the cross-sectional area of the silk fibers was not quantified due to technical limitations, a direct material-to-material comparison with commercial sutures that accounts for variations in thread diameter was not able to be made. Attempts were taken to measure the diameter of the threads using calipers, but the intrinsic thinness of the threads resulted in no significant difference in measurement to be observed. Future studies should incorporate microscopy to measure fiber diameter, allowing for the calculation of tensile stress (MPa) and Young’s modulus, a property of a material that measures stiffness. Additionally, clinically relevant metrics such as knot-pull strength and elasticity will need to be assessed to fully validate the material’s clinical applicability.
Other limitations still remain. The use of synthetic Type O- blood doesn’t accurately reflect the complexities of human blood and the impact of immersion was only tested at two time points. The possibility of background fluorescence observed in the silk from being immersed in the synthetic blood itself also needs to be considered. As this experiment was performed in vitro using a stable blood substitute, the data cannot yet correlate a specific percentage of fluorescence loss to a specific stage of tissue repair and the body’s ever changing internal environment. Furthermore, a primary limitation of this study is the absence of biological healing endpoints. Additionally, biological variability was assessed within the cohort (n=14); however, given the investigative nature of this material engineering study, results should be interpreted as descriptive trends rather than predictive clinical outcomes. In consideration of a clinical application, it is possible that there was some leakage of fluorescent chemicals from the engineered silk into the blood during the immersion, something which may impair signal reliability and biocompatibility for human use. Future directions should therefore prioritize understanding the mechanisms of signal loss. To assess the leakage of chemicals, the blood used for immersion can be analyzed fluorometrically, where the presence of an exogenous fluorescent signal in the blood would suggest that a change in fluorescence occurred due to leaching rather than chemical degradation. Testing in animal blood and following a longer time-course of immersion would also help quantify the extent of signal masking versus fluorophore loss. Exploring alternative fluorophores with higher brightness or stronger stability would also improve detection and quantification. Overall, the results in this study come together to show that multi-fluorescent silk can be produced and that its signal changes when exposed to a blood-like environment. While significant optimization and in vivo testing are needed, this work represents an initial step toward exploring fluorescent silk as an indicator for perfusion based healing in a clinical setting.
Supplementary Methods: Detailed Experimental Protocols
Animal Husbandry Methods
The silkworms used in this study were raised in controlled indoor conditions to ensure consistent development and silk production. Larvae were housed in ventilated containers lined with clean paper and a temperature of approximately 70℉ with relatively consistent humidity levels was maintained throughout all growth stages. A consistent light-dark cycle mimicking that of the natural day-night cycle was also maintained to simulate natural environmental conditions.
All of the silkworms were fed silkworm chow, which was refrigerated prior to feeding to maintain freshness. Waste material and uneaten food was removed regularly from the boxes in which the silkworms were housed to maintain sanitary living conditions. The larvae were monitored throughout development to ensure proper health and growth prior to cocoon formation and silk harvesting.
All organisms were handled carefully to minimize stress or physical damage during maintenance and experimental procedures.
Degumming and Silk Fiber Preparation
The degumming process to obtain individual fibers from intact cocoons was performed according to the method described by George Weil (“How to Degum Reeled Silk”)12. Cocoons from each experimental group were cut into approximately 5 cm segments. Each silk segment was then soaked in warm tap water (approximately 50–60 °C) for 10-15 min to begin loosening the sericin coating. Fibers were gently drawn apart using fine tipped forceps while still immersed in warm water, and rinsed repeatedly until sericin was visibly removed and fibers appeared smooth and glossy. Degummed fibers were then air-dried at room temperature in a dark room to minimize photobleaching prior to analysis.
The dried and degummed fibers were manually aligned and gently twisted to form single testable threads. Each thread consisted of fibers derived from a single cocoon to preserve biological independence between replicates. Threads were cut to standardized lengths of approximately 5 cm prior to mechanical testing or immersion experiments.
Fluorescence Imaging Acquisition Parameters
Fluorescence imaging was performed using an upright epifluorescence microscope equipped with a 100W mercury arc illumination source and a C-mount digital imaging camera. To ensure that quantitative comparability was maintained between timepoints and experimental groups, all imaging parameters were held constant across all samples. The microscope filter blocks used to detect fluorescence were selected based on fluorophore excitation characteristics of the fluorescent additives used in this study. A blue excitation filter block (450–490 nm excitation, ~520 nm emission) was used for detecting Rhodamine 110 and copper-associated fluorescence, while a green excitation filter block (510–560 nm excitation, ~590 nm emission) was used for detecting Rhodamine B fluorescence. Exposure time, camera gain, illumination intensity, and camera sensitivity settings were maintained consistent across imaging sessions. All imaging was conducted in the dark to minimize ambient light interference and photobleaching.
ImageJ Fluorescence Quantification Workflow
ImageJ software was used to quantify fluorescence intensity measurements following a standardized workflow. Images taken of the fluorescent silk were imported as uncompressed image files and separated into individual color channels using the Split Channels function. A region of interest (ROI) was manually drawn along the visible length of each silk fiber in each image, and the mean gray value of the ROI was recorded as the raw fluorescence intensity. Three background ROIs were then selected per image in non-fluorescent regions adjacent to the fiber, and the average of these values was calculated to determine background fluorescence intensity. The background-corrected fluorescence intensity was calculated by subtracting background intensity from fiber intensity. Fluorescence intensity values were subsequently normalized relative to the mean baseline fluorescence intensity of silk fibers from each experimental group prior to the immersion step.
Synthetic Blood Immersion Protocol
For immersion testing, degummed silk fibers were placed individually into sealed ziploc bags containing synthetic Type O- blood. The bags were submerged in a temperature-controlled water bath maintained at 37°C using a sous vide circulator. Samples were stored in darkness throughout immersion. Fibers were removed at predetermined timepoints of one week and three weeks, gently rinsed with distilled water, and immediately imaged to assess fluorescence retention.
Mechanical Breaking Load Testing
Mechanical strength testing was conducted using a custom static load testing apparatus constructed to measure breaking load. Silk threads were secured to the hook of an inverted wire hanger positioned above a digital kitchen scale placed between two stable chair supports. Threads were attached to the hanger hook using consistent knotting methods to minimize variability in attachment stress. Downward force was applied manually by gradually pulling the hanger assembly vertically while monitoring the force measurement displayed on the digital scale. Force was increased steadily until thread rupture occurred, and the maximum force recorded immediately prior to breakage was documented as the breaking load. To maintain consistency, all tests were conducted using the same apparatus configuration and loading procedure and the kitchen scale was verified for zero calibration prior to each trial.
References
- L. Xu, H. Zhang, J. Wang, A. Li, S. Song, H. Ren, L. Qi, J. Gu, M. Meng. Information loss challenges in surgical navigation systems: From information fusion to AI-based approaches. Information Fusion. 2022;92. https://doi.org/10.1016/j.inffus.2022.11.015 [↩]
- J. Nagaraju, M.R. Goldsmith. Silkworm genomics – progress and prospects. Current Science. 2002;83(4):415–425. http://www.jstor.org/stable/24106841 [↩]
- O.J. Lee, M.T. Sultan, H. Hong, Y.J. Lee, J.S. Lee, H. Lee, S.H. Kim, C.H. Park. Recent advances in fluorescent silk fibroin. Frontiers in Materials. 2020;7:50. https://doi.org/10.3389/fmats.2020.00050 [↩] [↩]
- Z.F. Wu, Z.N. Sun, H.M. Xiong. Fluorescent silk obtained by feeding silkworms with fluorescent materials. Chinese Journal of Chemistry. 2023;41:2035–2046. https://doi.org/10.1002/cjoc.202300043 [↩] [↩] [↩] [↩] [↩]
- M.J. Sanderson, I. Smith, I. Parker, M.D. Bootman. Fluorescence microscopy. Cold Spring Harbor Protocols. 2014;2014(10):pdb.top071795. https://doi.org/10.1101/pdb.top071795 [↩]
- N. Li, R. Zhao, Y. Sun, Z. Ye, K. He, X. Fang. Single-molecule imaging and tracking of molecular dynamics in living cells. National Science Review. 2017;4. https://doi.org/10.1093/nsr/nww055 [↩]
- K. Babu Busi, M. Palanivel, K. Kanta Ghosh, W. Basu Ball, B. Gulyás, P. Padmanabhan, S. Chakrabortty. The multifarious applications of copper nanoclusters in biosensing and bioimaging and their translational role in early disease detection. Nanomaterials (Basel, Switzerland). 2022;12(3):301. https://doi.org/10.3390/nano12030301 [↩] [↩]
- A. Khmelinskii, M. Meurer, C.T. Ho, B. Besenbeck, J. Füller, M.K. Lemberg, B. Bukau, A. Mogk, M. Knop. Incomplete proteasomal degradation of green fluorescent proteins in the context of tandem fluorescent protein timers. Molecular Biology of the Cell. 2016;27(2):360–370. https://doi.org/10.1091/mbc.E15-07-0525 [↩] [↩] [↩] [↩]
- Khalid Hamood Al-Behadili W., M. Jawad Y., Hasan H. J. Study the Effect of PH on Absorption and Fluorescence Spectra of Eosin Y Dye in Ethanol. Asian Journal of Green Chemistry, 2023; 7(3): 163-174. https://doi.org/10.22034/ajgc.2023.393691.1381 [↩] [↩]
- S. Guo, L.A. Dipietro. Factors affecting wound healing. Journal of Dental Research. 2010;89(3):219–229. https://doi.org/10.1177/0022034509359125 [↩]
- F. Gottrup. Oxygen in wound healing and infection. World Journal of Surgery. 2004;28:312–315. https://doi.org/10.1007/s00268-003-7398-5 [↩]
- G. Weil. How to degum reeled silk. 2015. https://www.georgeweil.com/blog/how-to-degum-reeled-silk/. [↩] [↩]
- O.O. Abugo, R. Nair, J.R. Lakowicz. Fluorescence properties of rhodamine 800 in whole blood and plasma. Analytical Biochemistry. 2000;279(2):142–150. https://doi.org/10.1006/abio.2000.4486 [↩]
- Ma Y. Fluorescence characteristics analysis of rhodamine B. Journal of Physics: Conference Series. 2023;2468:012022. https://doi.org/10.1088/1742-6596/2468/1/012022 [↩]
- M. Beija, C.A. Afonso, J.M. Martinho. Synthesis and applications of Rhodamine derivatives as fluorescent probes. Chemical Society Reviews. 2009;38(8):2410–2433. https://doi.org/10.1039/b901612k [↩]
- X. Wang, Y. Li, Q. Liu, Q. Chen, Q. Xia, P. Zhao. In vivo effects of metal ions on conformation and mechanical performance of silkworm silks. Biochimica et Biophysica Acta. General Subjects. 2017;1861(3):567–576. https://doi.org/10.1016/j.bbagen.2016.11.025 [↩]
- N.C. Tansil, Y. Li, L.D. Koh, T.C. Peng, K.Y. Win, X.Y. Liu, M.Y. Han. The use of molecular fluorescent markers to monitor absorption and distribution of xenobiotics in a silkworm model. Biomaterials. 2011;32(36):9576–9583. https://doi.org/10.1016/j.biomaterials.2011.08.081 [↩]
- P. Jiang, H. Liu, C. Wang, L. Wu, J. Huang, C. Guo. Tensile behavior and morphology of differently degummed silkworm (Bombyx mori) cocoon fibres. Materials Letters. 2006;60:919–925. https://doi.org/10.1016/j.matlet.2005.10.056 [↩]
- S. Kundu, R. Rajkhowa, B. Kundu, X. Wang. Silk fibroin biomaterials for tissue regenerations. Advanced Drug Delivery Reviews. 2013;65(4):457–470. https://doi.org/10.1016/j.addr.2012.09.043 [↩]