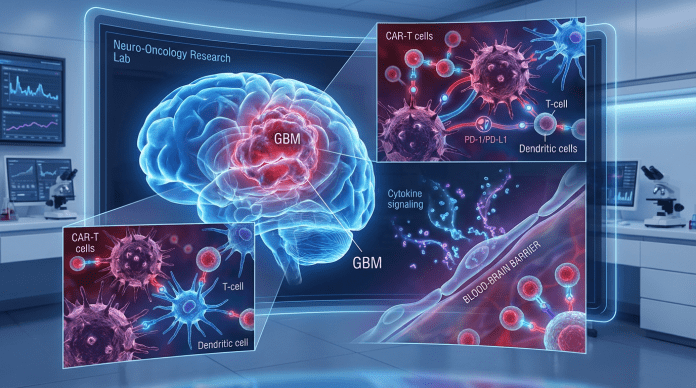
Abstract
Glioblastoma multiforme (GBM) is a malignant brain tumor affecting over 300,000 people each year worldwide. The current standard of care includes radiation, chemotherapy with temozolomide (TMZ), and surgical resection. Given the 5-year survival rate for patients suffering from GBM is 6.9%, improving the current standard of care is necessary. Recently, immunotherapeutics such as chimeric antigen receptor (CAR)-T cells, dendritic cell vaccines, and immune checkpoint blockade have entered clinical trials for the treatment of GBM. CAR-T cell therapy relies on engineered T-cells to eradicate tumor antigens and has been shown to increase tumor-specific killing and eliminate immune suppressive pathways, in the context of GBM. Dendritic cell vaccines, for GBM, boost the immune system, by releasing cytokines and stimulators of T-cells, to attack foreign antigens and have been shown to increase overall long-term survival rates by months, with minor side effects. Lastly, immune checkpoint blockade drugs target specific checkpoints such as cytotoxic T-lymphocyte-associated protein 4 (CTLA4) and Programmed Death-Ligand 1 (PDL1), that hinder the immune system and have improved survival rates and progression-free survival. Overall, the results of these clinical trials suggest that immunotherapy is a promising therapeutic option for GBM patients and these therapeutics should continue to be researched to further elucidate their safety profiles and efficacy.
Keywords: Glioblastoma multiforme, Immunotherapy, CAR-T Cell Therapy, Dendritic cell vaccine, Immune checkpoint inhibitor, Tumor microenvironment, IL-10, Temozolomide, Blood-brain barrier, Survival, Clinical trials, Tumor-associated antigen, Cytokines, T-cell exhaustion, Combination therapy, Adverse events, Patient outcomes, Preclinical models, Translational research
Introduction
Glioblastoma multiforme (GBM) is a highly aggressive and fast-growing malignant brain tumor that accounts for over 47.7% of cases of all central nervous system (CNS) tumors, with over 300,000 individuals diagnosed each year worldwide1. GBM is most commonly diagnosed in individuals aged 65 and older and has a poor prognosis, with an average survival of only 14 months2 and a 5-year survival rate of 6.9%3. GBM’s high propensity for relapse is due to the aggressive nature of the tumor. The average age of diagnosis for GBM is 64 years4. GBM patients often experience recurring headaches, vomiting, a loss of appetite, possible mood changes, shifts in the ability to think and learn, and speech difficulties, among other symptoms4. In general, the main causes of GBM are unknown, however, in rare cases, it can occur due to genetic syndromes. The main risk factor for developing GBM is prior ionizing radiation therapy, which uses high-energy waves to eradicate cancer cells and can damage, healthy cells in the process.
The current standard of care for GBM is surgical resection, followed by radiation and chemotherapy with the drug temozolomide (TMZ). TMZ works by causing mutations in replicating cells which results in cell cycle arrest and prevents further replication, eventually resulting in apoptosis or cell death5. TMZ alone has not been successful in over 55% of patients diagnosed with malignant gliomas due to the depth of the tumor6. When TMZ crosses the blood-brain barrier, only 20% of the drug is delivered to the brain tumor7. TMZ must be administered in high doses for a significant impact in eradicating the tumor, so TMZ can accumulate in a higher amount in the brain, but a negative side effect includes local toxicity. Hence, researching new novel treatments is critical. The reason is that GBM generally recurs, and most TMZ resistance comes from cells overexpressing methylguanine methyltransferase (MGMT) or the cells lacking a functional Deoxyribonucleic Acid (DNA) damage repair pathway resulting in resistance to TMZ, since TMZ does not only target cancer cells but also healthy cells, the toxicity is significant. TMZ resistance and the poor prognosis of GBM suggest that novel therapeutic options are necessary.
Immunotherapy boosts and strengthens the immune system to fight cancer itself. For GBM and other cancers, it has been shown that immunotherapies increase survival rates, life longevity, and overall improvement in quality of life for the patients. Immunotherapies rationale demonstrates how it is overall improving the quality of life and length of life for patients. This review focuses on investigating the efficacy of immunotherapeutics such as CAR-T cell therapy, dendritic cell vaccines, and immune checkpoint blockade in the context of GBM treatment.
However, despite the use of surgical resection, radiation, and chemotherapy, patient outcomes remain poor due to resistance mechanisms, and significant toxicity2,3,4,5. These limitations highlight the urgent need to explore alternative therapeutic approaches8,9. Therefore, the objective of this systematic review is to critically assess the effectiveness and safety of emerging immunotherapeutic strategies, including CAR-T cell therapy, dendritic cell vaccines, and immune checkpoint blockade in the treatment of GBM. The review aims to evaluate clinical outcomes, identify key limitations, and suggest future directions for research that may enhance the standard of care for GBM patients.
The Immune System
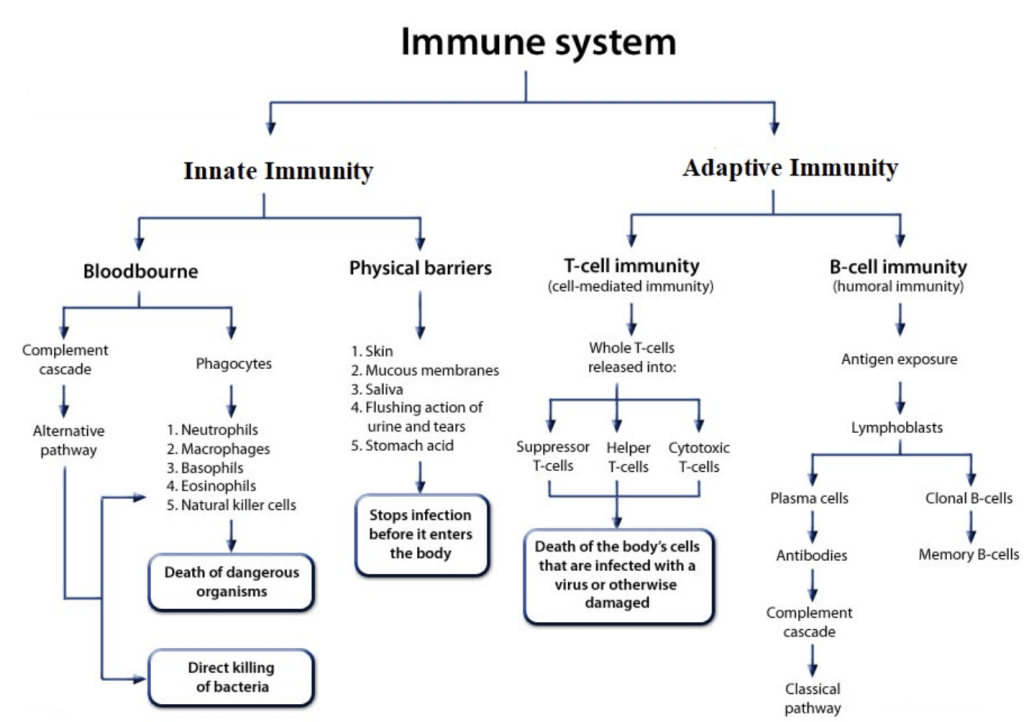
The immune system contains various cell types and processes helpful in protecting the body from foreign substances/particles, viruses, bacteria, diseases, etc. There are two main types of immunity: innate and adaptive immunity (Figure 1)10. Innate immunity refers to the immediate clearance of pathogens from the body by cell types such as macrophages, neutrophils, mast cells, etc10. Adaptive immunity refers to the longer-term response by B cells, T cells, and APCs that provide memory to specific pathogens following an exposure/vaccine10.
Innate immunity also encompasses physical barriers such as the skin, mucous membranes, saliva, flushing actions of urine/tears, and stomach acids, which all stop an infection from entering the body (Figure 1)10. Innate immunity consists of the bloodborne defense made up of phagocytes such as neutrophils, macrophages, basophils, and eosinophils10 which all consume cancerous cells affecting the body negatively (Figure 1)11. Through bloodborne pathways, foreign particles(bacteria, viruses, etc.) are destroyed and eradicated11,10.
Contrasting innate immunity, adaptive immunity has a longer response time, but results in long-term protection and memory, and can be split into T-cell (cell-mediated) immunity and B-cell (humoral) immunity (Figure 1)10. Cell-mediated immunity involves the activity of suppressor T-cells, helper T-cells, and cytotoxic T-cells, which all lead to the death of infected cells and cancerous cells10. Suppressor T-cells aid in controlling T-cell production and function, while helper T-cells connect to the humoral immunity of the B-cells to develop antibodies and killer cells12,13. The cytotoxic T-cells focus on destroying the cancerous cells12. Humoral immunity focuses on B-cells creating antibodies, which are Y-shaped proteins that have specificity to certain pathogens that mark invading cells as foreign to be destroyed14. An antibody then recognizes the antigen which triggers the immune response of T-cell elimination14.
GBM evades immune surveillance by creating a suppressive environment that weakens immune responses and blocks immune cells from attacking the tumor. This includes releasing molecules that inhibit immune activity and attracting cells that prevent effective immune attack. These evasion mechanisms make it challenging for immunotherapies to work, as treatments must overcome the tumor’s defenses and reach the cancer cells to be effective15.
Immunotherapies for GBM
As previously mentioned, immunotherapy boosts the immune system and can be used in the context of cancer8. Immunotherapy uses substances produced by the body to destroy the cancer cells. Immunotherapy may be used together with radiation/chemotherapy using substances such as Anti-Programmed Death-1 (Anti-PD-1)/PD-L1 antibody therapy, targeting monoclonal antibodies, and adoptive cell transfer by the body to destroy cancer cells having been tested on the breast to highly harmful melanomas, becoming Food and Drug Administration (FDA) approved16,17. The immunotherapies currently being investigated for GBM focus on overcoming GBM’s barriers of the immunosuppressive environment and the low immunogenicity (triggering adaptive immunity) of GBM and if overcome and reduce severe effects of GBM on the body9.
CAR-T Cell Therapy
An immunotherapy being explored for GBM is chimeric antigen receptor (CAR) T-cell therapy18. CAR-T cells function through engineered receptors that guide T-cells to target and destroy cells of a tumor-associated antigen (TAA), which is a substance in the body that can be destroyed with an immune response of antibodies, specifically for tumor cells in the body at high specificity by the CAR-T cells19. CAR-T cell therapy has resulted in 40% of patients experiencing long-term remission in B-cell lymphoma and is expected to show similar, if not, better results from GBM20. CAR-T cell therapy involves removing T-cells from the patient’s body and modifying them to secrete synthetic antigen receptors which allows them to bind to the target tumor antigens and this immunotherapy uses the modified T-cells to identify and target GBM cancer cells21. When the CAR-T cell attaches to the surface, this binding ensures that the CD3ζ module is triggered to complete its cytotoxic function of cell killing by releasing perforins, granzymes, and cytokines19. CARs ensure that the immune receptor is created specifically for the tumor-associated antigens and that the CAR-T cells connect with and destroy the tumor cells, like the “lock and key” concept22. For example, recent research has focused on the role of B7-H3 (an immune checkpoint molecule that is recognized and highly expressed in GBM among other cancers) in GBM23. Figure 2c is the full setup of the CAR-T cells conjoined together to recognize and attach to tumor-associated antigens on the tumor surface. The T-cell receptor (TCR) complex has specific variable chains that are attached to signaling transduction modules. At the TCR, the T-cells are triggered to bind to the antigens on the tumor cells and the signal transduction pathway ensures that the external TCR is attached to the cell surface24. During CAR-T cell therapy, the CAR-T cells expand, and the increase in cytokines triggers an immune response. The basic structure of the CAR includes the antigen binding region and the signaling domain and together the CAR will express a protein, for instance, cytokines or chemokines19.
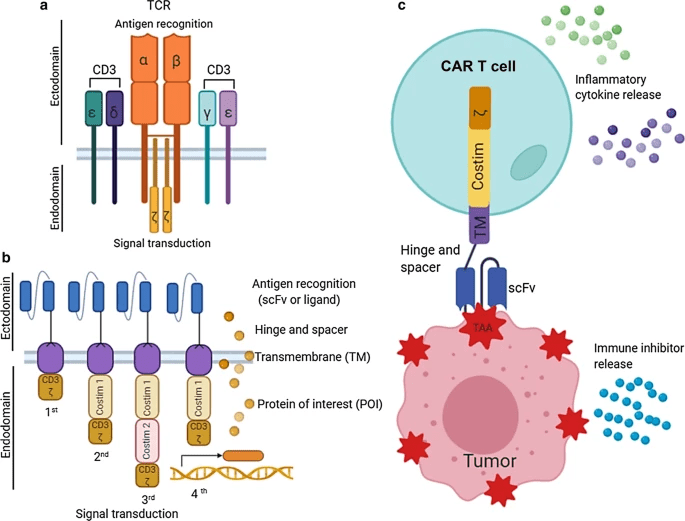
CAR-T Cell immunotherapy has shown encouraging results in pre-clinical studies, such as the expression of Interleukin‑13 receptor subunit alpha‑2 (IL-13 Ra2) (a primary tumor-associated antigen) CAR-T cells and the use of Epidermal Growth Factor Receptor variant III (EGFRvIII) CAR-T cells for GBM25. However, these findings are based on small sample sizes and limited follow-up, and clinical trials in GBM remain minimal. Therefore, these results should be interpreted as preliminary21. Common mild side effects include fever and chills, trouble breathing, severe nausea, vomiting, possible diarrhea, dizziness, light-headedness, headaches, and rapid heartbeats among others. The side effects of CAR-T cell therapy apply to any cancer22.
A Phase 1 clinical trial investigated the safety and feasibility of administering IL13Ra2-CAR T cells, intracranially, to patients with recurrent GBM18. IL13Ra2 is a primary tumor-associated antigen, present in over 75% of GBM, and has limited expression in the normal brain and other tissues of the body18. Of seven individuals in this study, one patient who was treated with IL13Ra2-CAR T cells demonstrated a complete response (with all indications of cancer disappearing with the treatment)18. While the sample size of the clinical trial was small, it demonstrated that IL13Ra2-CAR-T cells could safely be administered to this patient population and helped show that increasing cytotoxic T-lymphocyte tumor trafficking is a critical aspect of cancer immunotherapies18,26. The CAR-T cells induced endogenous immunity to help focus on controlling the increase in tumor reactivity and improved tumor-specific killing18. These results suggest IL13Ra2 CAR-T cells may have potential antitumor activity in GBM that could potentially expand to other solid tumors18. While a complete response was only seen in one patient, the potential for this immunotherapy must continue to be investigated in future clinical trials with larger sample sizes18.
Another phase 1 clinical trial investigated the efficacy of the antigen EGFRvIII, in CAR-T cell therapy in reducing toxicity27. EGFR contains EGF receptors that have regulated tumor progression through hindering cell proliferation and extending survival EGFR is most commonly found in newly diagnosed GBM27. EGFR is a mutated form of the antigen found in GBM and is the most common EGFR form of EGFR variant III, in GBM27. This study was conducted in 10 patients with recurrent GBM and 7 of these patients had a surgical resection following the CAR-T cell therapy27. This surgical resection allowed for specific analysis of the tumor tissue and assessment of specific endpoints, which looked at T cell trafficking to the tumor, examined the tumor microenvironment in situ, and the post-therapy target antigen expression for the CAR-T cells28. In this study, the treatment led to removal of the EGFRvIII-positive tumor and was associated with a whole-tumor immune response, suggesting improved anti-tumor immunity duration27. The study also observed engraftment of CAR-T cells in the bloodstream and demonstrated that CAR-T cells could be trafficked into the brain27. However, these findings are based on a small patient cohort in an early phase trial, and should be interpreted as preliminary.
Limitations of CAR-T Cells
A limitation for CAR-T cells is that they require the Target Antigen (TA) to be located on the cell membrane which can be an issue as T cell receptors recognize the TA intracellularly 29. Another limitation of the CAR-T cells is the selectivity and heterogeneity of the targeted TA, as the ideal TA is expected to be homogeneously spread out on all cancer-initiating cells (CICs)29. TA expression must also be reduced in normal tissues as the CAR-T cells eradicate any cell with any amount of the TA which increases toxicity resulting in some of the negative side effects of CAR-T cell therapy30. Another limitation of CAR-T cells is that CAR-T cell therapy ensures that the CAR-T cells can be trafficked to the tumor, which in GBM is in the brain and many individuals have struggled in designing CAR-T cells that can cross the blood-brain barrier (BBB). It becomes difficult to target the tumors using immunotherapy because the CAR-T cells are unable to reach the tumor, especially because it is unable to pass through the BBB. Moreover, CAR-T cell therapies are extremely costly, making them virtually inaccessible for most people. Furthermore, CAR-T cells can enter a state of exhaustion, rendering them ineffective in eliminating the tumor cells, in later stages of treatment proving that it may not have longevity in their response. It is also important to note that confounding factors—such as differences in patient characteristics, tumor biology, and prior treatments—can significantly influence individual responses and outcomes in these studies. This is especially relevant for small, non-randomized trials, where the potential for selection bias is high. Individual patient differences, tumor biology, and prior treatments may influence CAR-T cell therapy outcomes, especially in small, non-randomized studies, introducing potential selection bias15. These limitations should be considered when interpreting the reported results.
CAR-T cell therapy can cause mild to moderate side effects such as fever, nausea, and off-target toxicity, with additional risks from difficulty crossing the blood-brain barrier and T-cell exhaustion22,30.
Dendritic Cell (DC) Vaccine
Dendritic cells are a type of antigen-presenting cell (APC), that defend the body from foreign antigens and connect the innate and adaptive immune systems31. These cells protect and defend the body by searching for pathogen-associated molecular patterns (PAMPs) with pathogen recognition receptors (PRRs) which help activate the immune responses in the body31. When a pathogen is recognized, the dendritic cells immediately endocytose (engulf the external substance with the cell membrane of the cell) the protein and travel to the B-cell and T-cell-rich sites of the lymph nodes31, where the foreign peptide is presented. Once the dendritic cells are activated, they begin to create proinflammatory cytokines and potential stimulators of specific T-cells32. Dendritic cells are capable of passing through the blood-brain barrier, suggesting they might be an attractive therapeutic option for GBM. Additionally, the dendritic cells go through the BBB, which ensures that they reach the tumor and fight/destroy it.
Dendritic cells are commonly administered by being incubated with tumor cells and administered with an adjuvant to stimulate the immune response upon injection, for example, one DC vaccine contains PD1+ or PD1− cytotoxic T cells that are injected into the lymph nodes (Figure 3). This activates T cells that are transmitted through the peripheral blood to the brain. The GBM antigen-activated CD8*T dendritic cells then reach the tumor microenvironment and based on the quantity of PD-1+ cytotoxic T cells, it determines the quantifiable effectiveness of cytotoxic T cells and tumor killing resulting in tumor cell death33. DC vaccines for GBM have started to enter clinical trials, proving their usage as an immunotherapy.
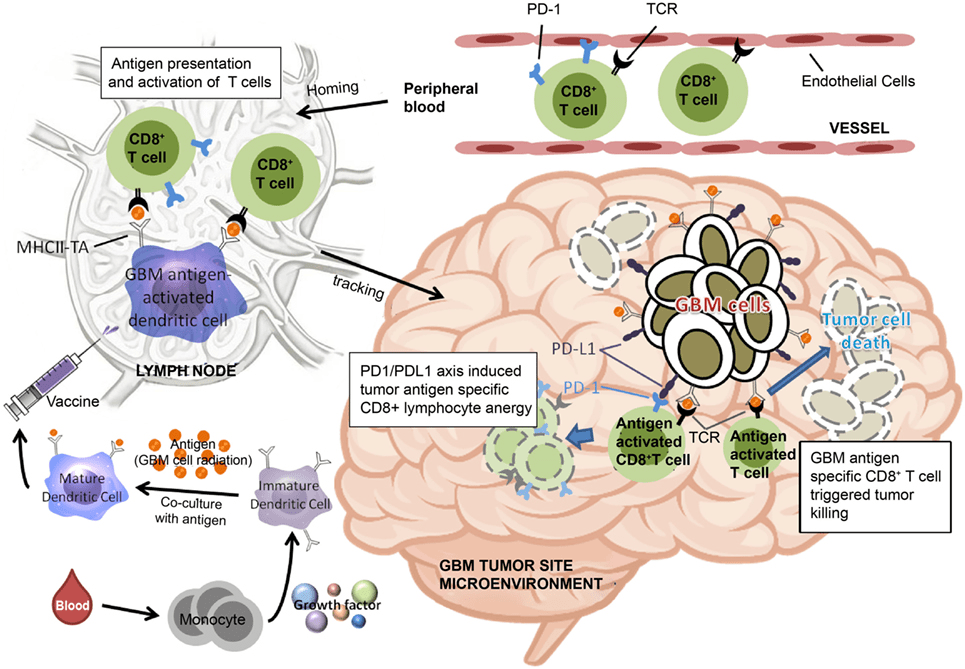
The VICTOR I study was conducted on 15 patients – 12 of whom received a dendritic cell vaccine of cells induced with anEGFRvIII peptide (3 patients did not receive the vaccine due to improvement from radiation therapy alone)32. After the patients underwent surgical resection of the tumor, 3 vaccine doses were administered within the first 2 weeks of the trial, and then 2 more doses were consistently administered over 2-week intervals32. The comparison was shown of the patients who did and didn’t receive Temozolomide (TMZ) and who only lived for 6.4 and 7.1 months, respectively, post-treatment32. In this small, early-phase trial, some patients did not show progression of symptoms or tumor, and experienced increased longevity32. Two patients had encouraging responses when monitored over Magnetic Resonance Imaging (MRI), including one patient who was not showing symptoms or progression of the tumor32. However, these findings are based on a limited patient group. Another patient succeeded significantly with the vaccine, as she remained alive for 6.2 years after diagnosis and treatment32. Two of the three patients with Grade III tumors were monitored and confirmed to not have any tumor progression for 66.2 to 123.7 months post-treatment32. However, the patients with only TMZ treatment showed that they only had 13.7 months to 14.5 months, essentially a smaller period of survival32. Minimal toxicity was seen with the administration of the dendritic cell vaccine and, it was concluded that the vaccine was safe for use in humans32. In addition, this study suggests a new potential survival benefit with a median time to progression of 18.7 and a median survival of 22.8 months32.
A randomized double-blind phase III trial was conducted on 331 patients with newly diagnosed or recurrent GBM34. All patients received the current standard of care however, in addition, one group received a placebo, and the other received DCVax-L (an autologous tumor lysate-loaded dendritic cell vaccination)34. As a reference, Autologous cell therapy uses the patient’s own cells or tumor material, while allogeneic cell therapy uses cells or material from a donor31. The overall survival of the DCVax-L group compared to the placebo group was assessed as the endpoint of the trial34. However, due to the changes during treatment, it was monitored for patients who had the DCVax-L along with the current standard of care to another group who only received the current standard of care and not the placebo, showing the limitation of using an outside group34. The DCVax-L treated group for newly diagnosed GBM had an overall survival of 19.3 months, which was longer than the control group’s overall survival of 16.5 months34. Recurrent GBM, similar to the newly diagnosed group, had an overall survival improvement of 13.2 months versus the control group which survived 7.8 months34. The survival advantage from the DCVax-L seemed to apply more to those with poor prognosis, elderly patients, patients who received a suboptimal surgical resection, and patients who had a deteriorating condition of the recurring disease34. Additionally, the vaccine appeared to be well tolerated and the patients did not have any severe side effects including grade 2/3 intracranial edema, grade 3 nausea, and grade 3 infection34. The evidence demonstrated a significant overall survival improvement for patients, whether that was the newly diagnosed GBM patients or the recurrent GBM patients34. Some limitations within this clinical trial included that the data was compared to an external control group, and an algorithm was used to generate the potential survival data, which did not provide the most accurate data34.
Limitations of Dendritic Cell Vaccines
The main limitation of dendritic cell vaccines has been tumor-mediated immunosuppression13. Tumor-mediated immunosuppression is essentially how cancer cells can weaken the body’s immune response by attracting immunosuppressive cells which hinders the immune responses through the use of apoptosis in tumor-infiltrating lymphocytes13. Tumors may also release immunosuppressive molecules such as cytokines, creating an immunosuppressive tumor microenvironment that can make it difficult for the immune system to act on the tumor cells13. Results from dendritic cell vaccine trials may be affected by heterogeneity in patient characteristics and prior therapies, and the risk of selection bias is heightened in small, non-randomized cohorts15.
Dendritic cell vaccines generally show minimal toxicity and are well tolerated, though tumor-mediated immunosuppression may limit their effectiveness15.
Immune Checkpoint Blockade
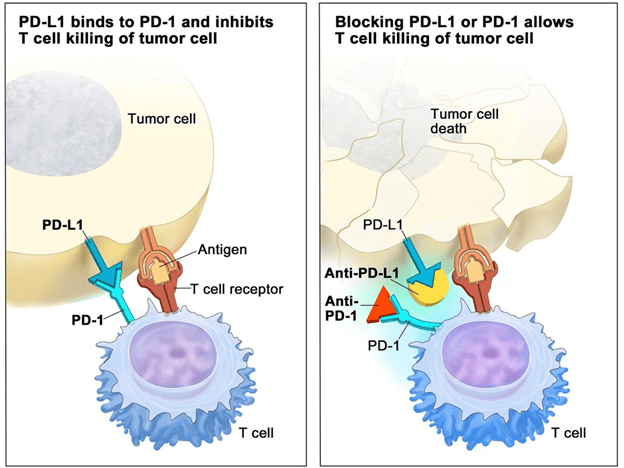
Immune checkpoints are considered natural brakes in the body for the immune system35. Immune checkpoint inhibitors block the immune checkpoints which helps enhance accessibility for the immune system to attack and destroy cancer cells35. This is because the drugs will ensure that the immune system can fight off the cancerous cells by targeting the immune checkpoints35. Specific drugs, such as ipilimumab, nivolumab, and bevacizumab targeting checkpoints such as CTLA-4, PD-1, and PD-L1 have been used to dampen the effects on the immune system35. The tumors use immune checkpoints to avoid and escape immune surveillance to protect against autoimmunity35. Immune checkpoint inhibitor drugs will go through the BBB under environmental, physical, or chemical disruptions to reach the tumor, so the immune system can attack the tumor with ease in the context of GBM36. An example of immune checkpoint blockade therapy in the context of various cancers is targeting checkpoint proteins, PD-L1 on tumor cells and PD-1 on T cells, which keep a watch over the immune system functions and responses37. When PD-L1 is bound to PD-1, this ensures that the T cells of the immune system cannot reach the tumor cells and destroy them (Figure 4)37. The attachment of PD-L1 to PD-1 results in the tumor cell staying the way it is, and the T cells are blocked from killing the tumor cell37. When the attachment of PD-L1 to the PD-1 is broken and the PD-1 is attached to the anti-PD-1, and the PD-L1 is bound to an anti-PD-L1, there is a block on the checkpoint proteins37. This allows for the T cells to eradicate the tumor cell37.
A phase 1 clinical trial investigated the efficacy of checkpoint inhibitors Ipilimumab and nivolumab in 40 patients with recurrent GBM35. Ipilimumab blocks the checkpoint, CTLA-4, and Nivolumab blocks the checkpoint PD-1. Researchers gave nivolumab alone to one group and in combination with ipilimumab to another group35. They found that 20% of the patients were able to resist progression of the tumor for a minimum of 12 weeks and 5 of the patients were able to live for 25 more months including the 12 weeks35. The combination of nivolumab and ipilimumab resulted in adverse effects/events, so nivolumab alone was better tolerated, and it was better suited for the phase III trial35. A phase III trial was then conducted to determine the efficiency and safety of nivolumab versus bevacizumab, an immune checkpoint inhibitor (vascular endothelial growth factor)35. This resulted in recurring improvements in efficiency and safety in recurrent GBM patients of resistance to the progression of the disease35. Results displayed that the nivolumab alone in comparison with bevacizumab did not have a significant advantage in survival35.
Another clinical trial was done on 369 GBM patients, 347 of whom were randomly assigned to either the nivolumab (184) or bevacizumab (185) treatment group32. The final analysis was complete when 301 patients passed away32. Both drugs were administered intravenously32. There was no significant difference in the overall survival between the treatment groups with 9.8 months for nivolumab and 10 months for bevacizumab32. As for progression-free survival, nivolumab had a shorter time frame of 1.5 months compared to 3.5 months with bevacizumab32. The findings in efficacy and side effect profile demonstrate that while there are minimal improvements and benefits to the immune checkpoint blockade, it does not have as large of a positive effect as other immunotherapies32.
Limitations of Immune Checkpoint Blockade
Immune checkpoint blockade has had minimal benefits and advantages when it comes to improving the conditions of patients with GBM and other diseases, however, there have been some significant limitations affecting patients as well. Specifically, there have been many side effects and symptoms resulting from the blockade therapy38. Possible side-effects that were studied of the immune checkpoint inhibitors include diarrhea, fatigue, cough, nausea, skin rash, poor appetite, and many more. Certain infusion and autoimmune reactions tend to be less common than the side effects mentioned. Immune-related adverse events have resulted from this treatment to cancer patients, resulting in hospitalizations and other treatments being necessary to help with the side effects of the immune checkpoint blockade therapy. Additionally, patients have experienced problems from the immunosuppressive tumor microenvironments (TME) which weakens the antitumor immune responses39 preventing patients from gaining the advantages of the therapy40. Interpretation of immune checkpoint blockade studies should account for confounding factors and possible selection bias, as outcomes may vary due to differences in patient populations and study design38,39,40.
Immune checkpoint inhibitors are associated with adverse events such as diarrhea, fatigue, and immune-related reactions, sometimes requiring hospitalization, and their efficacy may be reduced by the immunosuppressive tumor microenvironment38,39,40.
Future Directions
With the various immunotherapies and current standards of care that have been used, there have been significant improvements in survival, efficiency, and safety, in patients with GBM, with each immunotherapy having its benefits and limitations. Taking the limitations of the therapies such as how they are unable to cross the BBB, and creating a better immunotherapy can be even more beneficial for a GBM patient. Immune checkpoint inhibitors holding many limitations can be targeted to lessen the side effects as they block the tumor from being killed by T-cells and other immune system functions. Immune checkpoint inhibitors, such as anti-PD-1 monoclonal antibodies, work by blocking inhibitory signals that tumors use to suppress immune responses. This restores the immune system’s ability to attack cancer cells, rather than blocking immune function. However, this immune activation can result in immune-related adverse events, sometimes severe, as noted in the Limitations section. Future research aims to design checkpoint inhibitors that minimize these side effects while maintaining their effectiveness against tumor-induced immune suppression38. Enhancing the specificity of CAR-T cells can allow them to avoid recognizing antigens of healthy cells, and in treatment, the healthy cells’ antigens expression can be reduced to improve the efficacy of immunotherapy. The tumor microenvironment in GBM secretes immune-inhibitory cytokines such as IL-10, which can suppress immune cell function and lead to exhaustion or reduced efficacy of therapies like CAR-T cells and dendritic cell vaccines. Recent evidence suggests that combining immune checkpoint inhibitors (such as anti-PD-1 antibodies) with other immunotherapies may help overcome these barriers, restore T-cell activity, and improve anti-tumor responses and patient outcomes in GBM41. TMZ and immunotherapy combinations can be further explored to see how the current standard of care can be improved to result in better outcomes and reduce possibilities of relapse or post-treatment difficulties as the goal is to ensure the patient lives a long and healthy life without difficulty. Moreover, some other promising immunotherapies for GBM that can be experimented with and studied more are cytokine-based treatment, oncolytic virus, and other vaccine-based techniques excluding dendritic cell vaccines42. Overall, each immunotherapy has benefits to the patient in improving their condition, restricting the progression of the tumor, and increasing overall survival, but with future research, there could be opportunities to minimize their limitations and make them more feasible to use in the context of GBM (or get the immunotherapies to FDA approval).
Larger, well-controlled trials are needed to confirm the promise of immunotherapies for GBM. Future studies should directly compare these treatments to standard care, assess overall survival and quality of life, and evaluate safety in broader patient populations.
One significant limitation in the development of immune therapies for GBM is the lack of suitable GBM mouse models. This gap often leads to divergent results between preclinical animal studies and clinical applications, as current models may not accurately replicate the complexity of human GBM and its immune microenvironment. As a result, promising therapies in animal studies may not translate effectively to patient outcomes in clinical trials43.
Conclusion
Glioblastoma multiforme is a malignant tumor and rapidly spreading disease has had dire effects on patients suffering from it. The current standard of care of radiation, chemotherapy with temozolomide, and surgery has been overall successful but encounters issues with TMZ resistance. Immunotherapies are hoped to have more effective and positive results as they prove higher survival rates, safety, and life longevity. CAR-T cell therapy, dendritic cell vaccines, and immune checkpoint blockade have all been investigated in the context of GBM. CAR-T cell therapy is used with a receptor that brings T-cells to the targeted foreign antigens to be destroyed and has been proven successful in antitumor activity and increased duration of such activity. Dendritic cell vaccines release cytokines from the immune system to ensure the cytotoxic particles attack the foreign antigens/particles of the tumor ensuring increased survival and improvement of conditions. The immune checkpoint blockade uses checkpoint inhibitors to allow T-cells to attack the foreign particles on the tumors’ surface ensuring long-term survival and duration of response. Each immunotherapy has been successful in overall survival, duration of treatment response, and longevity, demonstrating that the malignant tumor’s spread can be stopped. The addition of immunotherapy to glioblastoma multiforme treatment has significant potential to enhance the current standard of care for patients.
References
- Cooling brain tumor cells could make headway in glioblastoma, UTSW researcher finds. (2023, January 12). https://www.utsouthwestern.edu/newsroom/articles/year-2023/january-cooling-brain-tumor-cells.html [↩]
- Mohammed S, Dinesan M, Ajayakumar T. Survival and quality of life analysis in glioblastoma multiforme with adjuvant chemoradiotherapy: a retrospective study. Rep Pract Oncol Radiother. 2022 Dec 29;27(6):1026-1036. doi: 10.5603/RPOR.a2022.0113. PMID [↩] [↩]
- About Glioblastoma – National Brain Tumor Society. (2023, July 20). National Brain Tumor Society. https://braintumor.org/events/glioblastoma-awareness-day/about-glioblastoma/ [↩] [↩]
- Glioblastoma multiforme – symptoms, diagnosis, and treatment options. (n.d.). https://www.aans.org/en/Patients/Neurosurgical-Conditions-and-Treatments/Glioblastoma-Multiforme [↩] [↩] [↩]
- Ortiz R, Perazzoli G, Cabeza L, Jiménez-Luna C, Luque R, Prados J, Melguizo C. Temozolomide: An Updated Overview of Resistance Mechanisms, Nanotechnology Advances and Clinical Applications. Curr Neuropharmacol. 2021;19(4):513-537. doi: 10.2174/1570159X18666200626204005. PMID: 32589560; PMCID: PMC8206461 [↩] [↩]
- Sengupta, S., Marrinan, J. E., Frishman, C., & Sampath, P. (2012). Impact of Temozolomide on Immune Response during Malignant Glioma Chemotherapy. Clinical & Developmental Immunology, 2012, 1–7. https://doi.org/10.1155/2012/831090 [↩]
- Park SH, Kim MJ, Jung HH, Chang WS, Choi HS, Rachmilevitch I, Zadicario E, Chang JW. One-Year Outcome of Multiple Blood-Brain Barrier Disruptions With Temozolomide for the Treatment of Glioblastoma. Front Oncol. 2020 Sep 10;10:1663. doi: 10.3389/fonc.2020.01663. PMID: 33014832; PMCID: PMC7511634 [↩]
- What is immunotherapy? | Immunotherapy for cancer. (n.d.). American Cancer Society. https://www.cancer.org/cancer/managing-cancer/treatment-types/immunotherapy.html [↩] [↩]
- Cancer immunotherapy. (2022, June 30). Yale Medicine. https://www.yalemedicine.org/conditions/cancer-immunotherapy [↩] [↩]
- Paul, S., Hmar, E. L., & Sharma, H. K. (2020). STRENGTHENING IMMUNITY WITH IMMUNOSTIMULANTS: A REVIEW. ResearchGate. https://www.researchgate.net/publication/342663166_STRENGTHENING_IMMUNITY_WITH_IMMUNOSTIMULANTS_A_REVIEW [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- The Editors of Encyclopaedia Britannica. (2009, February 6). Phagocyte | Definition, Function, Examples, & Facts. Encyclopedia Britannica. https://www.britannica.com/science/phagocyte [↩] [↩]
- B-cells vs. T-cells: What are Helper, Killer and Cytotoxic T cells. (n.d.). City of Hope. https://www.cancercenter.com/what-are-b-cells-vs-t-cells [↩] [↩]
- Professional, C. C. M. (n.d.). T-Cells. Cleveland Clinic. https://my.clevelandclinic.org/health/body/24630-t-cells [↩] [↩] [↩] [↩]
- News-Medical. (2022, November 18). What is an Antibody? https://www.news-medical.net/health/What-is-an-Antibody.aspx [↩] [↩]
- Schlößer HA, Theurich S, Shimabukuro-Vornhagen A, Holtick U, Stippel DL, von Bergwelt-Baildon M. Overcoming tumor-mediated immunosuppression. Immunotherapy. 2014;6(9):973-88. doi: 10.2217/imt.14.58. PMID: 25341119 [↩] [↩] [↩] [↩]
- What is immunotherapy? (2022, May 26). Cancer.Net. https://www.cancer.net/navigating-cancer-care/how-cancer-treated/immunotherapy-and-vaccines/what-immunotherapy [↩]
- Himes BT, Geiger PA, Ayasoufi K, Bhargav AG, Brown DA, Parney IF. Immunosuppression in Glioblastoma: Current Understanding and Therapeutic Implications. Front Oncol. 2021 Oct 28;11:770561. doi: 10.3389/fonc.2021.770561. PMID: 34778089; PMCID: PMC8581618 [↩]
- Alizadeh D, Wong RA, Gholamin S, Maker M, Aftabizadeh M, Yang X, Pecoraro JR, Jeppson JD, Wang D, Aguilar B, Starr R, Larmonier CB, Larmonier N, Chen MH, Wu X, Ribas A, Badie B, Forman SJ, Brown CE. IFNγ Is Critical for CAR T Cell-Mediated Myeloid Activation and Induction of Endogenous Immunity. Cancer Discov. 2021 Sep;11(9):2248-2265. doi: 10.1158/2159-8290.CD-20-1661. Epub 2021 Apr 9. PMID: 33837065; PMCID: PMC8561746 [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- Land, C. A., Musich, P. R., Haydar, D., Krenciute, G., & Xie, Q. (2020). Chimeric antigen receptor T-cell therapy in glioblastoma: charging the T cells to fight. Journal of Translational Medicine, 18(1). https://doi.org/10.1186/s12967-020-02598-0 [↩] [↩] [↩] [↩]
- Harris, J. (2023, March 29). New model predicts long-term response to CAR T-cell therapy in B-cell lymphoma. OncLive. https://www.onclive.com/view/new-model-predicts-long-term-response-to-car-t-cell-therapy-in-b-cell-lymphoma [↩]
- Luksik AS, Yazigi E, Shah P, Jackson CM. CAR T Cell Therapy in Glioblastoma: Overcoming Challenges Related to Antigen Expression. Cancers (Basel). 2023 Feb 23;15(5):1414. doi: 10.3390/cancers15051414. PMID: 36900205; PMCID: PMC10000604 [↩] [↩]
- CAR T-cell therapy and its side effects. (n.d.). American Cancer Society. https://www.cancer.org/cancer/managing-cancer/treatment-types/immunotherapy/car-t-cell1.html [↩] [↩] [↩]
- Nakagawa T, Kijima N, Hasegawa K, Ikeda S, Yaga M, Wibowo T, Tachi T, Kuroda H, Hirayama R, Okita Y, Kinoshita M, Kagawa N, Kanemura Y, Hosen N, Kishima H. Identification of glioblastoma-specific antigens expressed in patient-derived tumor cells as candidate targets for chimeric antigen receptor T cell therapy. Neurooncol Adv. 2022 Nov 15;5(1):vdac177. doi: 10.1093/noajnl/vdac177. PMID: 36601313; PMCID: PMC9798403 [↩]
- Lal, R., Ritchie, J., Richmond, L., & Keeshan, K. (2022). Detecting endogenous TRIB2 protein expression by flow cytometry and Western blotting. In Methods in Enzymology (pp. 59–77). https://doi.org/10.1016/bs.mie.2022.03.024 [↩]
- Bagley SJ, Desai AS, Linette GP, June CH, O’Rourke DM. CAR T-cell therapy for glioblastoma: recent clinical advances and future challenges. Neuro Oncol. 2018 Oct 9;20(11):1429-1438. doi: 10.1093/neuonc/noy032. PMID: 29509936; PMCID: PMC6176794 [↩]
- Slaney CY, Kershaw MH, Darcy PK. Trafficking of T cells into tumors. Cancer Res. 2014 Dec 15;74(24):7168-74. doi: 10.1158/0008-5472.CAN-14-2458. Epub 2014 Dec 4. PMID: 25477332 [↩]
- O’Rourke DM, Nasrallah MP, Desai A, Melenhorst JJ, Mansfield K, Morrissette JJD, Martinez-Lage M, Brem S, Maloney E, Shen A, Isaacs R, Mohan S, Plesa G, Lacey SF, Navenot JM, Zheng Z, Levine BL, Okada H, June CH, Brogdon JL, Maus MV. A single dose of peripherally infused EGFRvIII-directed CAR T cells mediates antigen loss and induces adaptive resistance in patients with recurrent glioblastoma. Sci Transl Med. 2017 Jul 19;9(399):eaaa0984. doi: 10.1126/scitranslmed.aaa0984. PMID: 28724573; PMCID: PMC5762203 [↩] [↩] [↩] [↩] [↩] [↩]
- Sener U, Ruff MW, Campian JL. Immunotherapy in Glioblastoma: Current Approaches and Future Perspectives. Int J Mol Sci. 2022 Jun 24;23(13):7046. doi: 10.3390/ijms23137046. PMID: 35806051; PMCID: PMC9266573 [↩]
- Maggs, L., Cattaneo, G., Dal, A. E., Moghaddam, A. S., & Ferrone, S. (2021). CAR T Cell-Based immunotherapy for the treatment of glioblastoma. Frontiers in Neuroscience, 15. https://doi.org/10.3389/fnins.2021.662064 [↩] [↩]
- Del Baldo G, Del Bufalo F, Pinacchio C, Carai A, Quintarelli C, De Angelis B, Merli P, Cacchione A, Locatelli F, Mastronuzzi A. The peculiar challenge of bringing CAR-T cells into the brain: Perspectives in the clinical application to the treatment of pediatric central nervous system tumors. Front Immunol. 2023 Mar 21;14:1142597. doi: 10.3389/fimmu.2023.1142597. PMID: 37025994; PMCID: PMC10072260 [↩] [↩]
- Constant, O., Maarifi, G., Blanchet, F. P., Van De Perre, P., Simonin, Y., & Salinas, S. (2022). Role of dendritic cells in viral brain infections. Frontiers in Immunology, 13. https://doi.org/10.3389/fimmu.2022.862053 [↩] [↩] [↩] [↩]
- Jackson C, Ruzevick J, Brem H, Lim M. Vaccine strategies for glioblastoma: progress and future directions. Immunotherapy. 2013 Feb;5(2):155-67. doi: 10.2217/imt.12.155. PMID: 23413907; PMCID: PMC4086484 [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- Jan, C. I., Tsai, W. C., Harn, H. J., Shyu, W. C., Liu, M. C., Lu, H. M., Chiu, S., & Cho, D. (2018). Predictors of response to autologous dendritic cell therapy in glioblastoma multiforme. Frontiers in Immunology, 9. https://doi.org/10.3389/fimmu.2018.00727 [↩] [↩]
- Gatto L, Di Nunno V, Tosoni A, Bartolini S, Ranieri L, Franceschi E. DCVax-L Vaccination in Patients with Glioblastoma: Real Promise or Negative Trial? The Debate Is Open. Cancers (Basel). 2023 Jun 20;15(12):3251. doi: 10.3390/cancers15123251. PMID: 37370860; PMCID: PMC10296384 [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- Rong L, Li N, Zhang Z. Emerging therapies for glioblastoma: current state and future directions. J Exp Clin Cancer Res. 2022 Apr 15;41(1):142. doi: 10.1186/s13046-022-02349-7. PMID: 35428347; PMCID: PMC9013078 [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- Sanders S, Debinski W. Challenges to Successful Implementation of the Immune Checkpoint Inhibitors for Treatment of Glioblastoma. Int J Mol Sci. 2020 Apr 16;21(8):2759. doi: 10.3390/ijms21082759. PMID: 32316096; PMCID: PMC7215941 [↩]
- Immune checkpoint inhibitors. (2022, April 7). National Cancer Institute. https://www.cancer.gov/about-cancer/treatment/types/immunotherapy/checkpoint-inhibitors [↩] [↩] [↩] [↩] [↩] [↩]
- Immune checkpoint inhibitors and their side effects. (n.d.). American Cancer Society. https://www.cancer.org/cancer/managing-cancer/treatment-types/immunotherapy/immune-checkpoint-inhibitors.html [↩] [↩] [↩] [↩]
- A look inside a tumor: mechanisms of tumor evasion and immunosuppression in the tumor microenvironment. (n.d.). www.rndsystems.com. https://www.rndsystems.com/resources/posters/look-inside-tumor-mechanism-tumor-evasion-and-immunosuppression-tumor [↩] [↩] [↩]
- Tormoen GW, Crittenden MR, Gough MJ. Role of the immunosuppressive microenvironment in immunotherapy. Adv Radiat Oncol. 2018 Oct 23;3(4):520-526. doi: 10.1016/j.adro.2018.08.018. PMID: 30370351; PMCID: PMC6200899 [↩] [↩] [↩]
- Zhu P, Li SY, Ding J, Fei Z, Sun SN, Zheng ZH, Wei D, Jiang J, Miao JL, Li SZ, Luo X, Zhang K, Wang B, Zhang K, Pu S, Wang QT, Zhang XY, Wen GL, Liu JO, August JT, Bian H, Chen ZN, He YW. Combination immunotherapy of glioblastoma with dendritic cell cancer vaccines, anti-PD-1 and poly I:C. J Pharm Anal. 2023 Jun;13(6):616-624. doi: 10.1016/j.jpha.2023.04.012. Epub 2023 Apr 21. PMID: 37440907; PMCID: PMC10334272 [↩]
- Rocha Pinheiro SL, Lemos FFB, Marques HS, Silva Luz M, de Oliveira Silva LG, Faria Souza Mendes Dos Santos C, da Costa Evangelista K, Calmon MS, Sande Loureiro M, Freire de Melo F. Immunotherapy in glioblastoma treatment: Current state and future prospects. World J Clin Oncol. 2023 Apr 24;14(4):138-159. doi: 10.5306/wjco.v14.i4.138. PMID: 37124134; PMCID: PMC10134201 [↩]
- Kringel R, Lamszus K, Mohme M. Chimeric Antigen Receptor T Cells in Glioblastoma-Current Concepts and Promising Future. Cells. 2023 Jul 3;12(13):1770. doi: 10.3390/cells12131770. PMID: 37443804; PMCID: PMC10340625 [↩]