Abstract
Alzheimer’s Disease (AD) is a neurodegenerative disease that results in loss of cognitive functioning in numerous neurological areas. Imperative functions that are impacted by AD include basic motor and locomotor functions, thinking, memory, problem-solving skills, and more. The risk of AD increases with age, as seniors aged 60 and older are at the highest risk. Using Drosophila melanogaster as a model for AD, the neuroprotective peptide ACTH(6-9) PGP was tested in four different concentrations, 5, 10, 20, and 25 mg/mL, to examine its effect on the locomotor capability of diseased-AD flies. Using a climbing assay, the locomotor function of flies with AD was assessed to detect locomotor capability in the flies. The average climbing assay pass rate of disease control flies was 50%, with most concentrations of ACTH(6-9) PGP unable to surpass this in the Drosophila model, reflecting that the peptide could not significantly improve the locomotor capability of AD flies. However, flies exposed to 20 mg/mL of ACTH6-9 PGP were shown to have an average pass rate of 61%, exceeding that of the disease control’s 49% pass rate, highlighting the potential for more studies to be done with ACTH6-9 PGP in Alzheimer’s research. While ACTH(6-9) PGP may not have been a statistically significant treatment in lower dosages, higher dosages show slight potential for locomotor improvement in diseased fly models, but further studies are warranted to confirm the neuroprotective peptide’s viability. With further research, effective treatments can be developed to combat the effects of AD.
Keywords: alzheimer’s disease (AD), locomotor functions, neuroprotective, ACTH(6-9) PGP, Drosophila melanogaster
Introduction
Alzheimer’s Disease (AD) is a neurodegenerative disease characterized by loss of cognitive functioning in areas such as problem-solving, thinking, memory, and other neurological aspects important for day-to-day function in human beings. AD is a multifactorial disease with many biological pathways paving the way for the disease’s pathogenesis. These include, but are not limited to: amyloid plaque accumulation (via the aggregation of amyloid-beta (Aβ) peptides), Tau protein aggregation, oxidative stress, neuroinflammation, and more.
Regarding the amyloid plaque pathway, the breakdown of the amyloid precursor protein (APP) results in the release and aggregation of Aβ peptides in the brain, amongst other components from different hallmarks and pathways of Alzheimer’s development, such as Tau proteins and more1. These Aβ peptides come in two forms: Aβ40 and Aβ42. While both are important in the amyloid pathway of AD pathogenesis, Aβ42 is a more prominent factor. On a molecular level, Aβ42 differs from the former in amino acid structure as Aβ42 has two extra hydrophobic residues on the C-terminus. This makes Aβ42 more insoluble and hydrophobic than Aβ40, while also supporting that Aβ42 aggregates and forms amyloid plaques more readily than Aβ40. Upon aggregation, these Aβ peptides accumulate to form the amyloid plaques, which directly cause damage to the brain. This is done through acceleration of apoptosis in neurons, disruption of synapses and critical cognitive functions in the brain, blockages or ruptures of blood vessels, and more2. Typically, Aβ42 proteotoxicity is studied more frequently in Drosophila models than Aβ40, as the expression of secreted Aβ42 directly contributes to neuronal death more so than Aβ40, as previously mentioned, motivating researchers focusing on this pathway of Alzheimer’s pathogenesis to focus on mediating the intrinsic and external factors of Aβ42 neurotoxicity3.
Oxidative stress, an imbalance of antioxidants and oxidants, is another prominent pathway of Alzheimer’s pathogenesis. Resulting from a sudden increase or decrease in the amount of antioxidants within the brain, oxidative stress reduces hydrogen peroxide and the oxygen molecules of water, leading to the formation of reactive oxygen species (ROS) that can react with other substances in the brain, impairing crucial cognitive functions, as is seen in Alzheimer’s disease4.
Inflammation (also known and referred to as neuroinflammation) is another major pathway in the development of Alzheimer’s, serving as a core pathology of the disease. In previous studies, neuroinflammation has been observed as the activation of macrophages known as microglia and other cells native to the brain that exacerbate the progression of both amyloid-beta and Tau proteins, further contributing to the pathogenesis of Alzheimer’s5.
While it can occur in numerous different age groups, AD is most common in the senior population of those aged 65 and older, as the risk of acquiring the disease increases with age. The disease is also one of the top ten leading causes of death in the United States, according to the Centers for Disease Control. In the U.S. alone, 6.9 million people live with the disease, 45% of whom are seniors aged 75-84 years and 36% of whom are seniors aged 84+ years6. As of a 2025 study, Alzheimer’s is currently the seventh leading cause of death (in terms of disease), with 2023 data suggesting it could rise to the sixth7. Furthermore, the population of those with Alzheimer’s in the U.S. is expected to drastically increase in the coming decades, reaching as high as 14 million by the year 20508.
The purpose of this research experiment is to determine whether or not the neuroprotective peptide ACTH(6-9) PGP can improve locomotor capability in a model of Drosophila melanogaster (common fruit flies) with the neurodegenerative disorder Alzheimer’s Disease. It is hypothesized that ACTH(6-9) PGP can mitigate locomotor damage in Drosophila melanogaster models of Alzheimer’s disease and even improve the locomotor/flying capability of these fruit flies. Proponents of this hypothesis include the peptide’s neuroprotective properties and its ability to improve memory consolidation in a model of rats in previous research9. Furthermore, ACTH(6-9) PGP protects neurons from cell death as well as oxidative stress and has proliferative activity, allowing it to rapidly increase and grow in number10,11. Additionally, in this research, Drosophila melanogaster is used as the main organism model as its nervous system is widely similar to that of humans, and it can express many genes and diseases also expressed by human beings, making it an ideal in vivo model to study neurodegenerative diseases such as Alzheimer’s12. From improvement of memory and neurprotective properties to mitigating cell death and oxidative stress, ACTH(6-9) PGP would be an ideal treatment to combat the locomotor damage caused by Alzheimer’s disease. These past findings in research suggest ACTH(6-9) PGP may be an ideal candidate for developing a treatment that can help combat Alzheimer’s.
To best address the established hypothesis, two strains of flies were acquired: one healthy, normal strain of flies from Carolina Biological, and one strain of flies with Alzheimer’s Disease, specifically expressing the Aβ1-42 gene, from Bloomington Drosophila Stock Center. Before organizing them in their variable/control groups, the flies were age-matched and gender sorted to ensure that only female flies were used for the research. These flies were divided amongst three different fly groups: a wildtype (+) control of healthy flies that would not receive any treatment, a disease (-) control of Alzheimer’s flies that would not receive any treatment, and an experimental group of Alzheimer’s flies that would receive treatment. The experimental group was divided into four subsets of groups that would receive four different concentrations of the ACTH(6-9) PGP treatment, one group per concentration, with the different concentrations: 5 mg/mL, 10 mg/mL, 20 mg/mL, and 25 mg/mL. Concentrations of the peptide were mixed in their respective batch of fly food, which the different groups of experimental flies would consume. Climbing assays would be conducted on the groups of flies to record the flies’ pass rates, therefore testing their motor capability. The varying concentrations of the ACTH(6-9) PGP served as the independent variable (IV) while the climbing assay pass rates were the dependent variable (DV).
Methodology & Materials
Different Drosophila Strains
To best address the established hypothesis, two strains of Drosophila melanogaster were acquired: one healthy, normal strain from Carolina Biological and one strain with AD, specifically expressing aggregated levels of the Aβ42 peptides, from Bloomington Drosophila Stock Center. Stock #33769 from the Aβ42 model of Drosophila was acquired from Bloomington, while the wildtype (healthy and normal) model of Drosophila acquired from Carolina Biological was from item #172100. Pertinent information regarding the acquisition and details of the acquired fly strains may be found in the table below.
| Fly Strain Type | Source | Catalog/Identifier No. | Genotype | URLs (Necessary for Sourcing) |
| Wildtype (WT) | Carolina Biological | 172100 (Item No.) Identifier: N/A | Female: X+/X+, 2+/2+, 3+/3+, 4+/4+ Male: X+/Y, 2+/2+, 3+/3+, 4+/4+ | https://www.carolina.com/flies/drosophila–living–wild-type–vial-of-2515030/172100.pr |
| Aβ42 | Bloomington Drosophila Stock Center | 33769 (Stock No.) Identifier: RRID:BDSC_33769 | w[1118]; P{w[+mc]=UAS-APP.Abeta42.B}m26a | https://bdsc.indiana.edu/Home/Search |
Regarding important driver information for the Aβ42 Drosophila line from Bloomington, Stock No. 33769 was a UAS responder line designed to be used and crossed with GAL4 driver stock for the expression of the amyloid beta gene. Before it arrived in the experimental lab setting, the stock was crossed with an elav-GAL4 stock for pan-neuronal expression of the amyloid beta gene in all (or nearly all) of the neurons in the Drosophila nervous system. This was done to prepare an accurate Drosophila model of Alzheimer’s disease and its progression. Furthermore, this would ensure that any effects of the tested independent variable (IV) drugs are identified.
Expanding Stocks
The two stocks of flies, healthy/normal flies and the flies with Alzheimer’s disease (acquired from Carolina Biological and Bloomington Drosophila Stock Center, respectively), were expanded for 2-3 months to provide a sufficient amount for the research. This involved tapping the flies from different vials every 4-6 days, allowing the flies to reproduce quickly and repeatedly. As Drosophila are known to quickly reproduce, this allowed the two stocks of flies to be expanded fairly quickly, supplying the experiment with a sufficient amount for data collection. The flies were held in vials sealed with cotton flugs at the top and with 0.5 – 1 in of fly food at the bottom of the vial.
Regarding rearing conditions, both strains of flies were reared at room temperature (~ 25 degrees Celsius) under a 12-hour light/12-hour dark (LD 12:12) cycle at 40-50% relative humidity (RH). To ensure the consistency of fly growth and reproduction, these settings were kept consistent throughout the entirety of the experiment. Both female and male flies were initially provided in the vials and kept together to reproduce, providing more female and male flies. Upon eclosion, flies would typically age up to one week (seven days) before being disposed of via freezing in a -80 °C refrigerator, ensuring all flies were aged seven days or less (< 7 days).
Age Matching & Gender Sorting
Before organizing them in their variable/control groups, the Drosophila melanogaster were age-matched and gender sorted to ensure that only female flies were used for the research. Age matching ensured that the collected data would be consistent across all flies, as AD is a neurodegenerative disease with risk and effects that worsen with age. In other words, different age groups will reflect different symptoms of the disease, asserting the need for the Drosophila to be age-matched. Gender sorting was done to ensure a viable collection of data from the flies, as vials with both male and female flies would likely result in the reproduction of offspring, interfering with the reliability and consistency of any data collected. A vial of only male Drosophila could potentially result in aggression and violence with each other in a male-only environment. A vial consisting only of female Drosophila promotes the most viable and appropriate environment for data collection. To ensure randomization and avoid bias, multiple vials of age-matched flies mixed with males and females were used when sorting female flies into their respective vials. A total of ten flies were assigned per vial, with multiple vials utilized per variable group. Flies were gender sorted using anesthetization via freezing by tapping them into empty vials, pausing for two minutes (120 seconds) to allow them to adjust to their environment, and placing the vial in an ice bucket for five minutes. After freezing-induced anesthetization, flies were placed on a glass plate chilled on ice and were sorted appropriately. This methodology was unofficially nicknamed cold sorting in the research setting. While CO2 sorting, a sorting method using CO2 from CO2 tanks for anesthesia of flies, was available, this was not used as repeated CO2 exposure can cause changes in fruit fly neurology13. Anesthetization via freezing avoids any such changes in neurology and is more compatible with behavioral studies with fruit flies, such as this one, than CO2 sorting.
Variable Grouping
After expanding, age matching, and gender sorting, the Drosophila melanogaster were divided amongst three different fly groups: a wildtype (+) control of healthy flies that would not receive any treatment, a disease (-) control of Alzheimer’s flies that would not receive any treatment, and an experimental group of Alzheimer’s flies that would receive treatment. The experimental group was divided into four subsets of groups that would receive four different concentrations of the ACTH(6-9) PGP treatment, one group per concentration, with the different concentrations being 5 mg/mL, 10 mg/mL, 20 mg/mL, and 25 mg/mL. To follow and be consistent with appropriate binding procedures, appropriate labels (e.g, “A1”, “A2”, “B1”, “B2”, etc.) were assigned to the necessary vials after variable grouping to ensure that the different fly groups would not be mixed when performing the necessary assays.
Making Fly Food with and without ACTH(6-9) PGP
Two different types of fly food were made for the research. The first was regular fly food, which was fed to the wildtype (+) and disease (-) controls with no altered change of any kind to their normal diet. The second was fly food containing the peptide ACTH(6-9) PGP in the four different concentrations: 5 mg/mL, 10 mg/mL, 20 mg/mL, and 25 mg/mL. ACTH(6-9) PGP was acquired as a lyophilized (freeze-dried) powder at >97.0% purity from the vendor Prospec Protein Specialists (catalogue number: HOR-033). As a lyophilized powder, it was stored at -18 oC until necessary use, as, according to the acquired safety data sheet, it can remain stable at room temperature for only three weeks or less. To appropriately dissolve the ACTH(6-9) PGP in fly food, it would have to be dissolved into liquid form via reconstitution. The reconstitution process was as follows. As an appropriate solvent, distilled water was used to dissolve the ACTH(6-9) PGP powder in its vial. Distilled water was micropipetted to the sides of the vial containing the lyophilized powder; this was done to avoid dropping the distilled water directly onto the powder, because this prevents the powder from properly diluting. After the distilled water was added, the vial was closed and swirled for 1-2 minutes to ensure the lyophilized powder would fully dilute with the distilled water. Following reconstitution, the liquid ACTH(6-9) PGP was stored in a refrigerator at 4 oC until further use.
A separate batch of fly food was made for each concentration of the peptide. The experimental group of flies, as mentioned previously, was split into four groups, with each respective group receiving one of the four concentrations of ACTH(6-9) PGP in their fly food as part of their diet. In each case, regarding the batches of fly food that received the treatment, ACTH(6-9) PGP, in liquid form, was added to the freshly made fly food after preparation. The final volume of each batch of fly food was 500 mL, which was appropriately measured with a negligible margin of error within a 1000 mL beaker. Calculated values of liquid ACTH(6-9) PGP were added to each batch of fly food to achieve the following concentrations of the treatment within the fly food: 5 mg/mL, 10 mg/mL, 20 mg/mL, and 25 mg/mL. Each vial containing fly food was filled up to a height of 0.5 inches. Each of the two control groups (wildtype and disease) and four experimental groups consisted of ten vials of flies per group, with ten flies per vial. The different fly groups were given their respective fly food. The four experimental groups of flies were given 48 hours (2 days) in their vials of the altered fly food with ACTH(6-9) PGP to allow for proper consumption and digestion of the peptide. This time frame was used as, in the case of standard methodological processes that test for locomotor capability and utilize locomotor-based assays, 48 hours is a sufficient time period for a stable physiological state in response to a new treatment fed to Drosophila in their fly food14. Furthermore, this limit was decided as, due to feasibility reasons regarding available equipment in the lab setting, the individual intake of each fly or vial of flies could not be directly measured. The 48-hour period was just long enough to ensure all the flies in each vial (that was fed the treatment) consumed the treatment, while being short enough to be within the bounds of the flies’ lifespan and the experimental procedure’s timeline.
Regarding variables, appropriate experimental, wild-type, and disease controls were developed and readied (as previously established). A vehicle control was inapplicable in this experiment, as the solvent the ACTH(6-9) PGP was dissolved in was distilled water, which is part of the recipe for fly food. For these reasons, there was no vehicle control used in the experiment.
Climbing Assays
After being given 48 hours in their respective vials with their respective type of fly food, a climbing assay was conducted for data collection to assess the effect of ACTH(6-9) PGP on the locomotor capability of diseased flies that consumed the altered fly food with the treatment. Climbing assays have been shown in previous studies to serve as strong indicators of locomotor behavior and capability. A 2011 study was able to model neurodegeneration as well as locomotor and learning capability in Drosophila using a type of climbing assay known as negative geotaxis15. For each vial of flies (ten vials per group, ten flies per vial), the flies were tapped out of their respective vial and into an empty, sealed vial. The ten flies were given approximately two minutes (120 seconds) to adjust to their new environment, while the 5 cm (approx. 2 in) mark from the bottom of the vial was marked using a marker and ruler. A 5 cm mark was used as the threshold for determining the pass/fail status of each of the flies because a climbing capability of 5 cm within three seconds (time frame can be altered for different experiments, especially when using different strains of flies) is well within the capabilities of healthy, wildtype Drosophila16. Since this research aims to determine whether the climbing capabilities of Aβ-expressing flies treated with ACTH6-9 PGP can be restored to that of healthy, regular flies, this cutoff and the use of a pass/fail system are most appropriate for the climbing assay. The use of a climbing assay is most appropriate as the approach, in addition to assessing the motor capability of diseased flies, provides insights into early detection of Alzheimer’s in fly models through progressive locomotor deficits; furthermore, it provides insights into the phenotypic severity of fly models with Alzheimer’s-associated genes (which may be applied to Alzheimer’s research with alternative angles) and can be applied to a breadth of disease models in model organisms17. The ability of climbing assays to provide fine-scale behavioral data as well as gauge locomotor phenotypes of Drosophila makes this method ideal for assessing flies in the context of neurodegenerative diseases such as Alzheimer’s18.
Next, in the climbing assay procedure, the empty vial, containing the flies, was then tapped on a surface such that all the flies fell to the bottom before the vial was placed still. As soon as all the flies reached the bottom of the vial, a timer of 10 seconds was started, while the flies would try to climb the inside of the vial towards the top, where the light source was shining. After ten seconds, the number of flies, out of the ten in the vial, that passed the 5-cm mark was counted. A single trial of this process consisted of doing this for one vial from the wildtype (+) control, disease (-) control, and each of the four experimental groups (5 mg/mL, 10 mg/mL, 20 mg/mL, and 25 mg/mL of ACTH(6-9) PGP). Per trial, each vial is tested once. Each vial is the statistical unit for analysis. A total of ten trials were completed across the research, with a two-day (48 hours) rest interval for the flies between trials. Per previous research in this field, the minimum rest interval between trials is three minutes19. However, due to time constraints in the research environment where the experiment was conducted, trials were conducted on a 48-hour interval.
To ensure the integrity and reliability of data, as well as minimize any potential for random and/or systematic error, the following were kept constant throughout the experiment: time of day, temperature of the research setting, the dimensions of the vial, and the wall texture of the vials. Regarding the time of day, all research was completed between the hours of nine and eleven in the morning, ensuring the flies’ behaviors based on daytime were consistent. The research environment was maintained at room temperature with an exact temperature of 25 degrees Celsius. All vials used had the same glass-like wall texture and dimensions of 10 centimeters in height and roughly 2 centimeters in diameter. Cotton flugs were used as caps for the vials.
Pass Rate = (# of flies that passed climbing assay mark) / (total # of flies) x 100
Equation 1: Formula for calculating the pass rate of flies in a single climbing assay trial (by percentage).
An example calculation where 4 out of 10 flies passed the climbing assay mark provides a 40 percent pass rate, as can be calculated below. Ex:
Pass Rate = ( 4 / 10) * 100
Pass Rate = (0.4) * 100
Pass Rate = 40 %
After this was done for all the vials across all the control and experimental groups, the collected data was analyzed for a statistical analysis test to be conducted. To ensure reliable consistency of data and account for any potential phototaxis-based stimulus, the illumination of the research setting where the experiment took place was kept constant. This was done to avoid any potential discrepancies or inconsistencies in the data that could have been due to phototaxis stimulus, should the research environment’s room illumination have been inconsistent. The room was kept illuminated at a constant level across the entirety of the research project and its trials.
Statistical Analysis
Following the collection of data on the different fly groups’ motor capability in the climbing assay, a Kruskal-Wallis statistical test was run on the compiled data using the DataClassroom software, with a Post-Hoc test being used to compare the p-values of the experimental groups of flies to those of the wildtype (+) and disease (-) controls. This would help determine, from a statistical standpoint, if the concentrations of ACTH(6-9) PGP were able to improve climbing capability in the Drosophila melanogaster. Should this be supported, it would strongly support that ACTH(6-9) PGP can improve the locomotor capabilities of Drosophila melanogaster models of Alzheimer’s Disease, paving the way for treatments that mitigate the motor-related impairments that human patients with Alzheimer’s disease face.
Data
| # of Flies out of 10 That Climbing Assay Mark Per Control/Experimental Group | ||||||
| Trial | Wildtype (+) | Disease (-) | 5 mg/mL ACTH(6-9) PGP | 10 mg/mL ACTH(6-9) PGP | 20 mg/mL ACTH(6-9) PGP | 25 mg/mL ACTH(6-9) PGP |
| 1 | 7 | 3 | 2 | 4 | 0 | 4 |
| 2 | 8 | 7 | 3 | 5 | 4 | 8 |
| 3 | 7 | 5 | 2 | 0 | 1 | 6 |
| 4 | 10 | 5 | 4 | 4 | 4 | 6 |
| 5 | 8 | 7 | 6 | 5 | 6 | 4 |
| 6 | 9 | 7 | 4 | 3 | 7 | 4 |
| 7 | 9 | 5 | 7 | 4 | 6 | 7 |
| 8 | 7 | 5 | 6 | 7 | 6 | 7 |
| 9 | 10 | 3 | 7 | 6 | 5 | 8 |
| 10 | 8 | 2 | 4 | 5 | 7 | 7 |
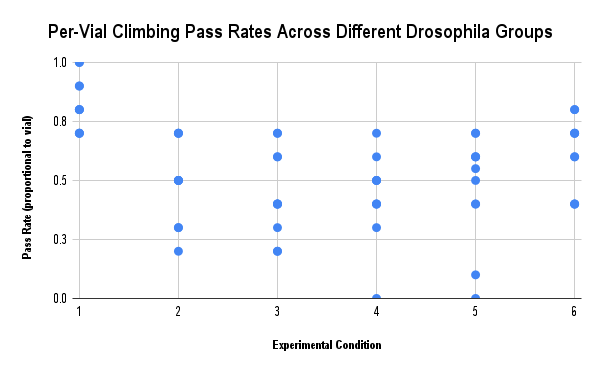
Data Availability Statement
The above data was collected during experimentation by the student researcher and author under the research mentors at the Academies of Loudoun. The author affirms that all included data (raw and calculated) have been included in this paper along with any results and statistical analysis. For the direct acquisition of raw data, statistically analyzed data, graphs, and/or supplementary materials, a considerable and reasonable request may be made directly to the corresponding author of this paper.
Results
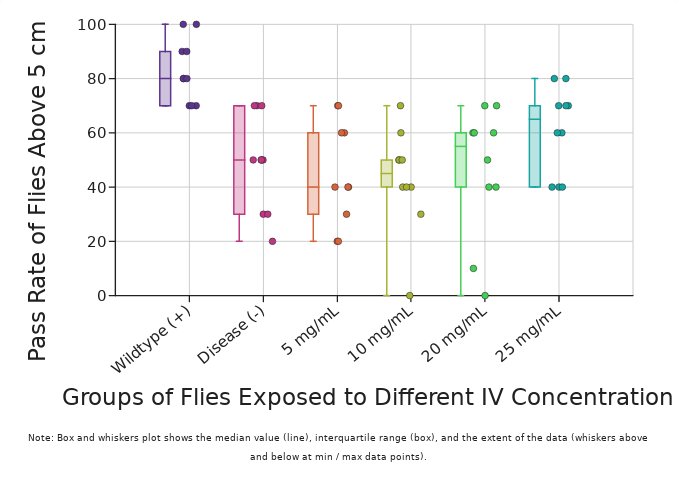
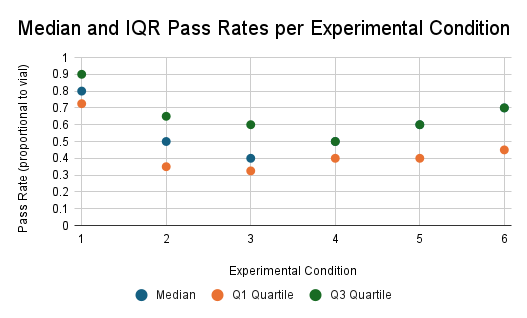
| Compiled Data of 10 Trials on Each Group of Flies Receiving (or Not Receiving) IV Treatment of ACTH(6-9) PGP (Units in %) | ||||||
| Wildtype (+) | Disease (-) | 5 mg/mL ACTH(6-9) PGP | 10 mg/mL ACTH(6-9) PGP | 20 mg/mL ACTH(6-9) PGP | 25 mg/mL ACTH(6-9) PGP | |
| Average | 83 | 49 | 45 | 43 | 46 | 61 |
| Median | 80 | 50 | 40 | 45 | 55 | 65 |
| Kruskal-Wallis Post-Hoc’s P-Value Results Between Wildtype/Disease and Groups | ||||
| 5 mg/mL ACTH(6-9) PGP | 10 mg/mL ACTH(6-9) PGP | 20 mg/mL ACTH(6-9) PGP | 25 mg/mL ACTH(6-9) PGP | |
| Wildtype (+) | <0.01 | <0.01 | <0.01 | 0.37 |
| Disease (-) | 1.00 | 1.00 | 1.00 | 0.63 |
Discussion
Hypothesis and Experimental Research Findings
This research originally hypothesized that the neuroprotective peptide ACTH(6-9) PGP could improve the locomotor capabilities of a Drosophila melanogaster model of Alzheimer’s Disease, which was observed by measuring the climbing assay pass rates of the Drosophila when administered with concentrations of the peptide. Flies were given four concentrations of ACTH(6-9) PGP in their diet (5, 10, 20, and 25 mg/mL) and had their overall motor capabilities observed via a climbing assay and calculated based on their pass rate percentages. The results reflected no statistically significant or detectable improvement in climbing capability via treatment of ACTH(6-9) PGP, since there was no detectable improvement in the motor performance of diseased flies treated with ACTH(6-9) PGP. This was determined by observing how the climbing pass rates of the treated flies continued to remain consistent with those of the disease (-) control, rather than having values statistically closer or similar to those of the wildtype (+) control.
Interpretations of Statistical Analysis
The Kruskal-Wallis statistical test was run with a Post-Hoc test to analyze the data. The results revealed low p-values (<0.01 to 0.37) between the experimental groups and the wildtype (+) control, highlighting a high statistical difference. In great contrast to this, the results also revealed high p-values, 1.00 for all except 25 mg/mL, which resulted in 0.63, between the experimental groups and the disease (-) control, reflecting a low statistical difference between the 5, 10, and 20 mg/mL values compared to the disease control, and a slightly higher statistical difference between the 25 mg/mL pass rates and disease control. These findings do indicate no statistically significant improvement of climbing capability in Drosophila treated with the ACTH(6-9) PGP for the first three concentrations, but also suggest that higher concentrations of the neuroprotective peptide could potentially show greater capability of restoring motor function and locomotor ability in the Alzheimer’s model of Drosophila melanogaster. Further studies and research of ACTH6-9 PGP in the field of Alzheimer’s research, from its effects on locomotor capability to its mechanisms in the amyloid branch of Alzheimer’s development and more, will need to be conducted to fully grasp the breadth of the potential for ACTH6-9 PGP and other neuroprotective treatments.
Limitations and Potential Improvements
An array of certain limitations pertaining to the lab setting, as well as experimental parameters and procedural shortcomings, likely contributed to the lack of a statistically significant or favorable result. Such limitations include the singular dependency on the climbing assay and the relatively limited scope to analyze locomotor capability solely on its own. These parameters limit the breadth of Alzheimer’s research as they are unable to account for or provide insights into alternative mechanisms and pathways: amyloid-beta, Tau proteins, oxidative stress, inflammation, and/or more.
To address these limitations, certain improvements can be made to the structural procedure of the experiment. Incorporating additional assays such as survival to track lifespan, immunostaining to track the development of pertinent proteins such as amyloid-beta and Tau, and the WAFO (wall-following) assay to track oxidative stress. These assays can provide deeper insights into the mechanisms of Alzheimer’s in a Drosophila melanogaster model as well as the effect different treatments have on these mechanisms and proteins. Incorporating combination therapies (CT) can also provide more favorable results, as such methods consistently improve overall health and performance in Drosophila with Alzheimer’s Disease20. Additionally, incorporating newly developed models of Drosophila melanogaster expressing the amyloid-beta gene and utilizing built-in apoptotic sensors that emit fluorescence signals from the green fluorescence protein may provide deeper insights into the amyloid pathway of Alzheimer’s disease as well as identify substances that may limit amyloid proteotoxicity, thus mitigating the progression of Alzheimer’s disease21. In the focus of assessing locomotor activity, the incorporation of a rapid iterative negative geotaxis (RING) assay could provide deeper insights into the consistency of the effect different therapies and treatments have on impaired locomotor activity. The use of a RING assay, which utilizes digital photography to document negative geotaxis in multiple groups of flies simultaneously, is highly favorable as it eliminates potential effects such as time of day, repeated testing, and the density of flies being tested, while providing more detailed insights into the locomotor capability of flies22. Furthermore, tracking locomotor activity can be improved and done more efficiently as well as more effectively by incorporating an AnimalTracker application programming interface (API). This method, which requires only a high-definition camera and computer peripheral hardware integration to analyze and record behavior, is an affordable and effective approach to systematically recording the locomotor performance of both larvae and adult flies via image processing of recorded video23.
In future Alzheimer’s research utilizing Drosophila models, it would be ideal to explore multiple pathways of Alzheimer’s pathogenesis beyond the amyloid pathway. This would be ideal, as in the case of Alzheimer’s, the progressive neurodegeneration and locomotor deficits that characterize this disease are the result of both amyloid-beta and non-amyloid aggregates building up in the brain as observed with Drosophila models24. Developing experimental procedures that can address multiple pathways may allow for accurate testing of treatment therapies against multiple pathways of Alzheimer’s pathogenesis, potentially paving the way for more versatile and effective treatment therapies for Alzheimer’s.
Future Work
While there does lie potential for higher concentrations of ACTH(6-9) PGP in the advancement of Alzheimer’s research, future research should explore alternative methods, therapies, and/or substances that highlight a greater level of success in the improvement of locomotor and motor-based functions in Alzheimer’s disease models such as Drosophila melanogaster and more. This may include using other neuroprotective compounds and utilizing methods such as combination therapy to apply more than one therapy while targeting and emphasizing specific mechanisms of Alzheimer’s (e.g, the amyloid/Aβ pathway, oxidative stress, inflammation, Tau proteins, etc.). Additionally, the addition of a survival assay alongside a climbing assay would likely increase the efficacy and comprehensive reliability of assessing the effect of different drugs and treatment therapies on Alzheimer-based Drosophila.
In the focus of the amyloid pathway, where different treatments and therapies would be tested on their ability to mitigate amyloid formation and peptide aggregation, the use of immunostaining and electron microscopy (EM) can help determine the efficacy of different treatment therapies in the amyloid pathway of Alzheimer’s pathogenesis25. The same methods may be applied to studying the Tau protein pathway, which focuses on the neurodegenerative effect of Tau proteins, observing how different treatment therapies may mitigate the internal and external factors of Tau proteins through scanning electron microscopy (SEM) and light microscopy26. Additionally, incorporating Drosophila models with different systems can help strengthen the efficacy and accuracy of studying the amyloid gene expression within Drosophila models of Alzheimer’s, as well as the effect different treatment therapies have on such gene expression. In particular, the Q-system of Drosophila, which is a recently-developed and repressible binary system that promotes widespread opportunities in genetic manipulation and transgene expression, can be used to study gene expression, including but not limited to amyloid and Tau27. In genetic manipulation, to express genes such as amyloid and Tau that are pertinent in Alzheimer’s pathogenesis, CRISPR-Cas9 models of Drosophila can also be applied as the CRISPR system can catalyse specific genome modification via homology-directed repair28. With more comprehensive and refined methodologies, more effective therapeutics, and focused mechanisms and properties of the disease, more effective and statistically significant treatments can be developed to better combat Alzheimer’s Disease.
Conclusion
The following study supported the notion that ACTH(6-9) PGP did not significantly improve motor capability in Drosophila melanogaster with Alzheimer’s disease at lower concentrations, but may have potential to do so at higher concentrations within fly food, such as 25 mg/mL. Despite this gray lining, the neuroprotective compound was best supported to have inefficacy in this goal as shown by statistical analysis, which confirmed that treated flies performed closer to disease (-) control flies than wildtype (+) flies. While this particular compound failed to be a significant step for Alzheimer’s research, expanding methodologies to include comprehensive methods, such as a survival assay, and exploring stronger treatment therapies while targeting specific mechanisms and pathways in the development of Alzheimer’s, could enhance future research. This could ultimately lead to treatments that play a crucial role and serve as the next step in developing pertinent therapies to combat Alzheimer’s disease.
References
- M. M. Rahman, C. Landel. Extracellular protein components of amyloid plaques and their roles in Alzheimer’s disease pathology. Mol Neurodegeneration. Vol. 16, pg. 59, 2021, DOI: https://doi.org/10.1186/s13024-021-00465-0. [↩]
- L. Gu, Z. Guo. Alzheimer’s Aβ42 and Aβ40 peptides form interlaced amyloid fibrils. Journal of Neurochemistry. Vol. 126(3), pg. 305-311, 2013, DOI: https://doi.org/10.1111/jnc.12202. [↩]
- P. Fernandez-Funez, L. de Mena, D.E. Rincon-Limas. Modeling the complex pathology of Alzheimer’s disease in Drosophila. Experimental Neurology. Vol 274(A), pg. 58-71, 2015, DOI: https://doi.org/10.1016/j.expneurol.2015.05.013. [↩]
- W.J. Huang, X. Zhang, W.W. Chen. Role of oxidative stress in Alzheimer’s disease. Biomedical Reports. Vol. 4(5), pg. 519-522, 2016, DOI: https://doi.org/10.3892/br.2016.630. [↩]
- J.W. Kinney, S.M. Bemiller, A.S. Murtishaw, A.M. Leisgang, A.M. Salazar, B.T. Lamb. Inflammation as a central mechanism in Alzheimer’s disease. Alzheimer’s & Dementia: Translational Research & Clinical Interventions. Vol. 4(1), pg. 575-590, 2018, DOI: https://doi.org/10.1016/j.trci.2018.06.014. [↩]
- A. Association. 2019 Alzheimer’s disease facts and figures. Alzheimer’s & Dementia. Vol. 15(3), pg. 321-387, 2019, DOI: https://doi.org/10.1016/j.jalz.2019.01.010. [↩]
- A. Association. 2025 Alzheimer’s disease facts and figures. Alzheimer’s & Dementia. Vol. 24(4), pg. e70235, 2025, DOI: https://doi.org/10.1002/alz.70235. [↩]
- A. Association. 2020 Alzheimer’s Disease Facts and Figures. Alzheimer’s & Dementia. Vol. 16(3), pg. 391-460, 2020, DOI: https://doi.org/10.1002/alz.12068. [↩]
- S. A. Dodonova, I. I. Bobyntsev, A. E. Belykh, A. O. Vorvul’. ACTH6-9 PGP improves memory consolidation processes in rats. Research Results in Pharmacology/. Vol. 7(1), pg. 27-32, 2021, DOI: https://doi.org/10.3897/rrpharmacology.7.62479. [↩]
- L. V. Dergunova, I. B. Filipennkov, S. A. Limborska, N. F. Myasoedov. Neuroprotective peptides and new strategies for ischemic stroke drug discoveries. Genes. Vol. 14(5), pg. 953, 2023, DOI: https://doi.org/10.3390/genes14050953. [↩]
- Y. Ding, Y. Zhong, A. Baldeshwiler, E. L. Abner, B. Bauer, A. M. S. Hartz. Protecting P-glycoprotein at the blood-brain barrier from degradation in an Alzheimer’s disease mouse model. Fluids Barriers CNS. Vol. 18, pg. 10, 2021, DOI: https://doi.org/10.1186/s12987-021-00245-4. [↩]
- K. Prüβing, A. Voight, J.B. Schulz. Drosophila melanogaster as a model organism for Alzheimer’s disease. Molecular Neurodegeneration. Vol. 8, pg. 35, 2013, DOI:https://doi.org/10.1186/1750-1326-8-35. [↩]
- H. Colinet, D. Renault. Metabolic effects of CO2 anaesthesia in Drosophila melanogaster. Biology Letters. Vol. 8(6), pg. 1050-1054, 2012, DOI: https://doi.org/10.1098/rsbl.2012.0601. [↩]
- J. Berlandi, F.J. Lin, O. Ambrée, D. Rieger, W. Paulus, A. Jeibmann. Swing Boat: Inducing and Recording Locomotor Activity in a Drosophila melanogaster Model of Alzheimer’s Disease. Front Behav Neurosci. Vol. 11, pg. 159, 2017, DOI: https://doi.org/10.3389/fnbeh.2017.00159. [↩]
- Y. O. Ali, W. Escala, K. Ruan, R. G. Zhai. Assaying locomotor, learning, and memory deficits in Drosophila models of neurodegeneration. Journal of Visualized Experiments. Vol. 49, 2504, 2011, DOI: https://dx.doi.org/10.3791/3795. [↩]
- C.D. Nichols, J Becnel, U.B. Pandey. Methods to assay Drosophila behavior. J Vis Exp. Vol. 61, 3795 (2012), DOI: https://dx.doi.org/10.3791/3795. [↩]
- T.R. Jahn, K.J. Kohlhoff, M. Scott, G.G. Tartaglia, D.A. Lomas, C.M. Dobson, M. Vendruscolo, D.C. Crowther. Detection of early locomotor abnormalities in a Drosophila model of Alzheimer’s disease. Journal of Neuroscience Methods. Vol. 197(1), pg. 186-189, 2011, DOI: https://doi.org/10.1016/j.jneumeth.2011.01.026. [↩]
- A. Aggarwal, H. Reichart, K. VijayRaghavan. A locomotor assay reveals deficits in a heterozygous Parkinson’s disease model and proprioceptive mutants in adult Drosophila. Proceedings of the National Academy of Sciences of the United States of America. Vol. 116(49), pg. 24830-24839, 2019, DOI: https://doi.org/10.1073/pnas.1807456116. [↩]
- K. Rakshit, R. Wambua, T.M. Giebultowicz, J.M. Giebultowicz. Effects of exercise on circadian rhythms and mobility in aging Drosophila melanogaster. Experimental Gerontology. Vol. 48(11), pg. 1260-1265, 2013, DOI: https://doi.org/10.1016/j.exger.2013.07.013. [↩]
- M.T. Kabir, M.S. Uddin, A.A. Mamun, P. Jeandet, L. Aleya, R.A. Mansouri, G.M. Ashraf, B. Mathew, M.N. Bin-Jumah, M.M. Abdel-Daim. International Journal of Molecular Sciences. Vol. 21(9), pg. 3272, 2020, DOI: https://doi.org/10.3390/ijms21093272. [↩]
- G. Elovsson, T. Klingstedt, M. Brown, K.P.R. Nilsson, A-C. Brorsson. A Novel Drosophila Model of Alzheimer’s Disease to Study Aβ Proteotoxicity in the Digestive Tract. International Journal of Molecular Sciences. Vol. 25(4), pg. 2105, 2025, DOI: https://doi.org/10.3390/ijms25042105. [↩]
- J.W. Gargano, I. Martin, P. Bhandari, M.S. Grotewiel. Rapid iterative negative geotaxis (RING): a new method for assessing age-related locomotor decline in Drosophila. Experimental Gerontology. Vol. 40(5), pg. 386-395, 2005, DOI: https://doi.org/10.1016/j.exger.2005.02.005. [↩]
- X. Long, H. Du, M. Jiang, H. Meng. A Simple Technique to Assay Locomotor Activity in Drosophila. Journal of Visualized Experiments. Vol. 192, pg. 65092, 2023, DOI: https://doi.org/10.3791/65092. [↩]
- D.C. Crowther, K.J. Kinghorn, E. Miranda, R. Page, J.A. Curry, A.I. Duthie, D.C. Gubb, A. Lomas. Intraneuronal Abeta, non-amyloid aggregates and neurodegeneration in a Drosophila model of Alzheimer’s disease. Neuroscience. Vol. 132(1), pg. 123-135, 2005, DOI: https://doi.org/10.1016/j.neuroscience.2004.12.025. [↩]
- K. Carmine-Simmen, T. Proctor, J. Tschäpe, B. Poeck, T. Triphan, R. Strauss, D. Kretzschmar. Neurotoxic effects induced by the Drosophila amyloid-β peptide suggest a conserved toxic function. Neurobiology of Disease. Vol. 33(2), pg. 274-281, 2009, DOI: https://doi.org/10.1016/j.nbd.2008.10.014. [↩]
- M.B. Trotter, T.D. Stephens, J.P. McGrath, M.L. Steinhilb. The Drosophila model system to study Tau action. Methods in Cell Biology. Vol 141, pg. 259-286, 2017, DOI: https://doi.org/10.1016/bs.mcb.2017.06.006. [↩]
- O. Riabinina, C.J. Potter. The Q-System: A Versatile Expression System for Drosophila. Methods in Molecular Biology. Vol 1478, pg. 53-78, 2016, DOI: https://doi.org/10.1007/978-1-4939-6371-3_3. [↩]
- S.J. Gratz, C.D. Rubinstein, M.M. Harrison, J. Wildonger, K.M. O’Connor-Giles. CRISPR-Cas9 Genome Editing in Drosophila. Current Protocols in Molecular Biology. Vol. 111, pg. 31.2.1-31.2.20, 2015, DOI: https://doi.org/10.1002/0471142727.mb3102s111. [↩]






