Abstract
Osteoporosis is a progressive disease of the bone that causes decreased bone density and impaired osteogenesis, ultimately resulting in fractures and limited mobility in the aging population. Bone Morphogenetic Protein 2 (BMP-2) is a critical regulator of osteoblast differentiation and a part of a series of growth factors that maintains bone homeostasis. The purpose of this study was to identify BMP-2 mutations that could enhance osteoblast activity, specifically mutant D375K, on recombinant protein expression levels in a bacterial system. A synthetic BMP-2 gene was cloned into the pET-15b vector, in addition to performing site directed mutagenesis, transformation, and protein expression in E. coli BL21(DE3). Of the variants performed, D375K had the highest protein yield, which could account for either enhanced structural stability or translational efficiency. While these findings suggest that the D375K mutation may improve structural stability, this study is limited to bacterial expression. The findings of this work present the exciting opportunity to bioengineer BMP-2 for stability in BMP-2 levels. Future work will involve functional assays in mammalian osteoblast cells and lysine-targeting delivery systems to determine the therapeutic relevance of BMP-2 mutants.
Keywords: Osteoporosis, BMP-2, site-directed mutagenesis, D375K, osteoblast, protein expression, bioengineering.
Introduction
Osteoporosis and Its Clinical Relevance
Osteoporosis is a chronic and progressive disorder of the bone that insidiously erodes bone strength over time, making individuals—particularly older individuals—more vulnerable to fractures. It arises when the equilibrium between bone formation and resorption is lost, leading to a net bone mineral density deficit1. Commonly referred to as the “silent thief,” osteoporosis usually goes undiagnosed until a fracture occurs, most commonly at the hip, spine, or wrist. With the aging of the world’s population, the incidence of osteoporosis is growing very fast and is recognized as a significant public health issue. Both women and men are affected, but postmenopausal women are especially at risk because of hormonal changes that favor bone loss2. In addition to the physical morbidity of fractures—pain, deformity, and loss of mobility—osteoporosis can drasticallyaffect quality of life and independence and even contribute to mortality from major fractures such as the hip. The treatment modalities that are available are primarily anabolic drugs or anti-resorptive drugs, lifestyle modification, and nutritional supplements. These are not without drawbacks, however. Long-term medication usage such as bisphosphonates has adverse effects such as gastrointestinal discomfort and extremely rare but serious complications such as those of atypical femoral fractures3 . Furthermore, compliance of patients with treatment regimens is still an issue. Considering these challenges, more advanced and sustainable treatments are receiving increased attention, including next-generation drug delivery systems, bone-targeting biologics, and gene therapies to control bone regeneration4. These technologies have the promise of delivering more effective and safer osteoporosis treatments in the future.
Role of Osteoblasts in Bone Homeostasis
Osteoblasts are vital builders of our skeleton. Osteoblasts construct and repair the bone structural framework through the synthesis and mineralization of the extracellular matrix. In the healthy body, they cooperate with osteoclasts—cells responsible for resorbing old bone—to maintain our bones strong and responsive to stress and trauma. Bone remodeling, this active process, is vital throughout life. But in conditions like osteoporosis, this balance is lost. Osteoblast activity and numbers decrease, but bone resorption by osteoclasts is uncontrolled. Progressive bone loss and increased fragility result5. This decreased osteoblast activity comes to the forefront with increasing age or with postmenopausal women experiencing hormonal changes, making the bones prone to fractures6. Therefore, stimulation of osteoblast activity has been a central approach in the development of novel osteoporosis treatments. Novel advances have included the use of anabolic medications like teriparatide and abaloparatide, that directly stimulate osteoblast differentiation and bone formation7. Another approach that may be considered is the use of anti-sclerostin antibodies like romosozumab that stimulate osteoblast activity by blocking the Wnt signaling pathway6.
In addition, research into the signaling communication between osteoclasts and osteoblasts is revealing the promise for bone balance reconstruction in the future, with increasingly targeted and effective treatments to follow8.
Bone Morphogenetic Protein 2 (BMP-2)
Bone Morphogenetic Protein 2 (BMP-2) is a strong signaling protein highly regarded for use in bone regeneration and repair. A TGF-β superfamily member, BMP-2 is a master controller of mesenchymal stem cell differentiation into osteoblasts—the very osteoblasts that create and mineralize new bone tissue9. BMP-2 is thereby an essential element in skeletal system health and injury response.
In normal conditions of a healthy body, BMP-2 participates in organized bone remodeling by osteoblast differentiation and activity. It triggers Smad and MAPK pathways, which induce the genetic and structural remodeling for osteoblast maturation and function10. Under clinical conditions—most notably in osteoporosis or bone trauma—native BMP-2 is insufficient to induce bone regeneration.
To reverse this, researchers have attempted to enhance the therapeutic activity of BMP-2. Prolonged BMP-2 stimulation has been found to not only enhance osteoblast differentiation but also trigger overall remodeling procedures, including transdifferentiation of bone cells11. Encapsulation of BMP-2 in biodegradable nanoparticles also allows for controlled release, making it more effective with fewer side effects12.
These results translate to increased potential for BMP-2 in regenerative medicine. By amplifying and concentrating this potent protein, scientists believe they can harness more effective therapies for osteoporosis and other bone diseases.
Study Rationale
Osteoporosis is a result of reduced osteoblast activity, leading to decreased bone formation and increased susceptibility. While Bone Morphogenetic Protein 2 (BMP-2) is a well-established osteoblast differentiation and new bone formation inducer, it remains limited to clinical applications. Supraphysiological concentrations of recombinant BMP-2 have been associated with unwanted side effects, including inflammation, over-resorption of bone, and even the creation of suboptimal bone11. The central question is whether the efficacy of BMP-2 can be enhanced by modifying the protein’s structure itself, rather than solely increasing its quantity.
The premise of this study is that amino acid mutations in BMP-2 specifically would render it more potent with increased capacity for inducing osteoblast activity without side effects. Since BMP-2 plays such a pivotal role in bone remodeling by virtue of its capability to induce such signaling pathways as Smad1/5/8 and activate transcription factors such as RUNX2, even slight changes in its form would increase its osteogenic activity13.
Notably, human population studies like the Rotterdam Study found BMP-2 gene polymorphisms in humans but with no substantial correlation with bone density, implying that natural variants may be neutral14. This leaves the door open for engineered mutations—engineered to be rationally improved in function and not to have resulted by chance.
Through the correction of these mutations, this study aims to discover a better, more biologically sensitive way to treat osteoporosis by redirecting the body’s own ability to create healthy, strong bones. The current investigation focuses strictly on the feasibility of producing these mutants and quantifying their expression yields.
Objectives of the Study
- To understand the role of BMP-2 in osteoblast differentiation and bone development.
- To identify and engineer specific amino acid substitutions in the BMP-2 gene that will enhance its osteogenic activity.
- To produce mutated BMP-2 constructs and compare with the wild-type construct.
- To compare protein and expression levels of BMP-2 mutants in bacterial expression systems.
- To measure the osteoblast-stimulating activity of BMP-2 mutants in bacterial systems by protein quantitation and DNA analysis.
- To establish if BMP-2 mutations can enhance bone-forming activity in bacterial systems, and thus provide a foundation for future research regarding BMP-2 mutations as therapeutic targets for osteoporosis.
Methodology
Gene Synthesis and Vector Preparation
For the efficient expression and mutational analysis of Bone Morphogenetic Protein 2 (BMP-2), a synthetic BMP-2 gene was designed and cloned into the pET-15b expression vector. With its high levels of transcription under the T7 promoter and N-terminal His-tag designed for purification purposes, the pET-15b vector was ideal for the expression of this gene. The gene was also codon-optimized for E. coli expression, synthesized by GenScript, which means the sequence fidelity could be confirmed and will be seamless for downstream applications.
The pET-15b expression system is appropriate for this study as it has a great performance with bacterial hosts with protein expression that is directed to the periplasmic space after a signal sequence was added to the synthetic BMP-2 protein, which improved disulfide bond formation and protein folding15. Prior literature has shown that BMP-2 produced from E. coli will biologically function if processed properly16.
Once the cloning of the wild-type construct was completed successfully, site-directed mutagenesis was executed to produce specific amino acid substitutions in order to enhance the osteogenic potential of BMP-2. Using PCR-based techniques with rationally designed primers, all mutants were sequenced-verified and cloned into bacterial expression systems. The mutagenesis tactic utilized strategies like DiRect17 and allowed us to look specifically at how mutations to BMP-2 may augment osteoblast activity, which is the foundation of new osteoporosis therapies with mutations to BMP-2.
Bacterial Transformation and Culture
For cloning, mutagenesis, and expression of BMP-2 and its variants, a two-strain bacterial system was utilized with E. coli DH5-α for plasmid amplification and BL21(DE3) for protein expression. DH5-α was selected to facilitate transformation because of high transformation efficiency and genetic stability with recA1 (recombinase) and endA1 (endonuclease) activity which aids in the preservation of plasmids during DNA amplification18.
Verification of wild-type and mutant BMP-2 constructs was performed by sequencing, and the plasmids were then transformed into BL21(DE3) cells. This strain has IPTG-inducible T7 RNA polymerase, which is capable of robust transcription of BMP-2 under the T7 promoter, and also has reduced protease activity which is beneficial for the stability of expressed recombinant proteins19.
Transformed colonies were grown in LB broth with the appropriate antibiotics at 37 °C, shaking at 200-250 rpm. Protein expression was induced at 25-28 °C to facilitate protein folding and solubility after reaching mid-log phase with OD600 of approximately 0.6-0.8.
Mutagenesis and Primer Design
To probe whether some specific amino acid changes might improve the osteogenic activity of BMP-2, we aimed for mutations like D375K that could affect the protein’s structure, binding to its receptor, or stability. We chose this region of the protein because mutations in this region are found to have high yields of results in E. Coli bacterial systems. We experimented with a deletion mutation and altered amino acids based on opposing amino acid properties (Fig 1). Plasmid DNA and Miniprep kits were ordered from GenScript. These mutations were made using polymerase chain reactions. We made changes in residue spatially close to BMP-2’s active site, paying attention to positions that could act as receptors or dimers. Charge, size, or hydrophobic changes were made, for example, substituting aspartic acid for lysine, to assess whether beneficial changes were possible.
Primers were designed according to the established protocols of mutagenesis, with the targeted nucleotide change in the middle of the oligonucleotide and the 12–15 base pairs of the complementary sequences on both sides.
With the introduction of mutations, a PCR-based site mutagenesis strategy was used and DpnI digestion was completed to eliminate the parental methylated DNA. The mutated plasmids were transformed to E. coli DH5-α and were propagated to confirm the sequence. For some specific situations, we applied efficiency and verification20,21 restriction-based insertions. This approach was the first step in the construction of BMP-2 variants.
Confirmation of DNA and Mutants Verification
For the verification of BMP-2 constructs cloning and mutagenesis, a plasmid miniprep was done for the isolation of plasmid DNA from transformed E. coli DH5-α colonies. The extracted DNA was analyzed by the means of agarose gel where sharp bands at the expected plasmid size indicated successful transformation and plasmid DNA integrity.
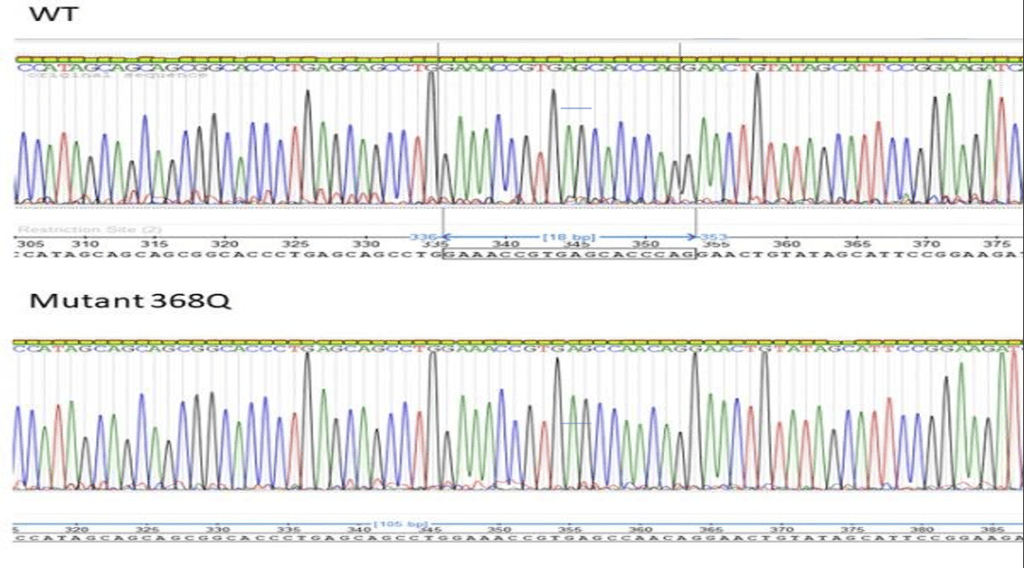
With this, mutagenesis was validated using the Sanger sequencing method after two trials. The sequencing primers which flank the mutation sites provide high-resolution reads of the modified areas. The resulting chromatograms were analyzed to ensure all expected base substitutions were made without any off-target mutations, and frameshifts which would indicate loss of sequence fidelity22. Confirming high-accuracy sequence is pivotal prior to expression as sequence damage, no matter how small, is likely to create functional, interpretational errors for the protein23. The wild type and its variants were found to be folded correctly and functional within a bacterial system through a functional assay.
In some cases, parallel primer approaches alongside a modified mutagenesis workflow were implemented to improve mutation verification and efficiency21. This guaranteed that all validated constructs were the only ones progressed for recombinant protein expression in bacterial systems.
Protein Expression and Quantification
After successful cloning and mutagenesis, wild-type and mutant BMP-2 genes were transformed to E. coli BL21(DE3) for expression of recombinant proteins. These cells were chosen due to their synthesis of T7 expression system-compatible proteins as well as their low levels of protease activity, which would provide high yield with low degradation24. Protein expression was induced with IPTG at mid-log phase (OD₆₀₀ ~0.6–0.8) and was sustained at 25–28 °C for better folding.
For purification, all BMP-2 constructs contained an N-terminal 6×His-tag which made purification possible through Ni-NTA affinity chromatography. Purification was accomplished through selective binding of BMP-2 to the His-tagged nickel resin, and was eluted after the addition of high concentrations of imidazole25.
Quantification of proteins was achieved through spectrophotometry at 280 nm, which measures the absorption of proteins with tyrosine and tryptophan. Expression was confirmed through NanoDrop readings. Interestingly, the D375K mutant demonstrated an increased absorbance compared to the wild-type, which suggested that it was better at being expressed26.
Results
The experimental research provided the successful construction, validation, and expression of wild type (WT) and mutant BMP-2 genes and subsequent quantification of expressed proteins. The research reported the successful designed mutations that resulted in alterations of the expressed protein levels, including the D375K mutant.
Confirmation of Mutant Constructs
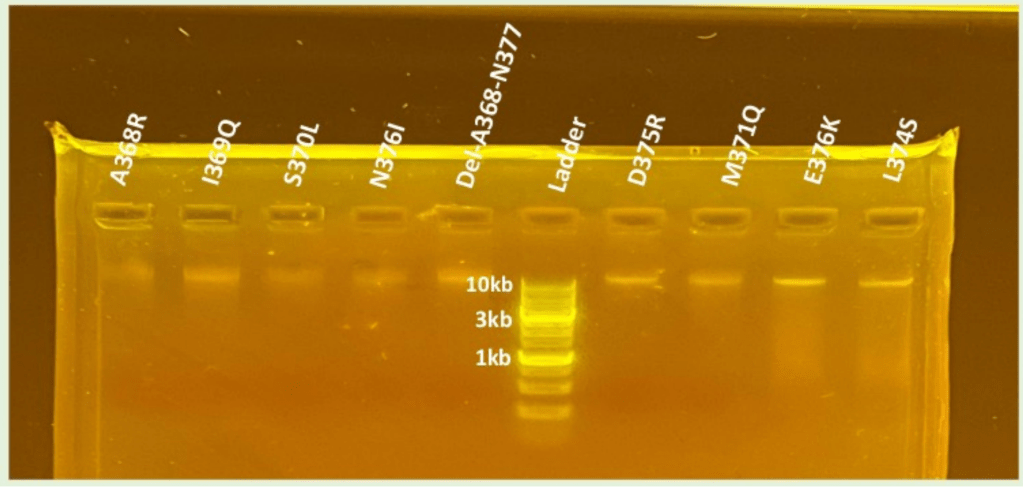
Following site-directed mutagenesis, gel electrophoresis confirmed successful amplification of the wild-type and mutant BMP-2 plasmids. As shown in Figure 2, bands were observed for all plasmid DNA constructs verifying amplification of the DNA. Given the consistently strong bands observed for WT, D375K, N376I, and Del-A368-N377, it was sufficient to conclude that both the mutant plasmids were intact and had the appropriate size.
In addition, validation of mutation and wild-type sequences were confirmed by Sanger sequencing. Figure 3 shows a representative chromatogram displaying a successful mutation (368Q) with the wild-type sequence ensuring that the desired amino acid mutations were introduced and that the PCR (site-directed mutagenesis) did not introduce additional off-target mutations. Using Chi-Square analysis, we determined that the wild type’s and D375K’s absorbance and protein concentration are independent of each other, showing a significant difference between the two protein variations.
Bacterial Growth and Protein Induction
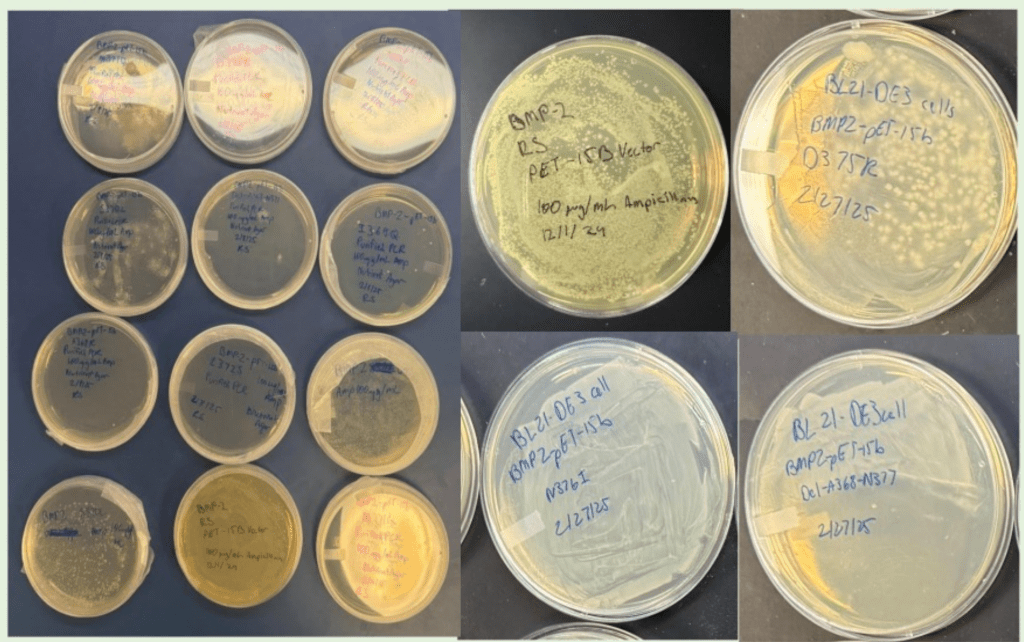
The transformed plasmids were plated onto agar plates to evaluate bacterial colony formation (Figure 4). All of the constructs (WT, D375K, N376I, and Del-A368-N377) had very robust colonies indicating that the mutations withstood plasmid stability and bacterial viability. However, it was important to verify neither bacterial growth nor transformation was affected by the mutations.
The colonies were then transferred to LB broth and IPTG was added to induce BMP-2 protein expression in E. coli BL21 (DE3) cells. It became evident that the level of expression was different between the constructs, as will be discussed by spectroscopy results.
Protein Quantification by Spectrophotometry
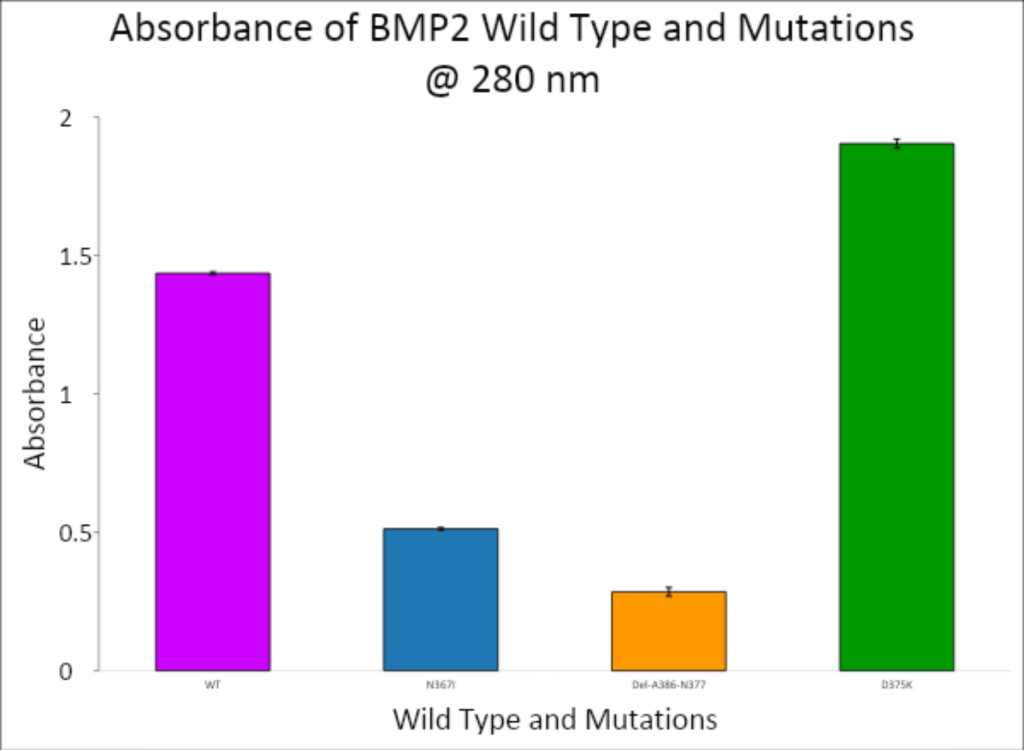
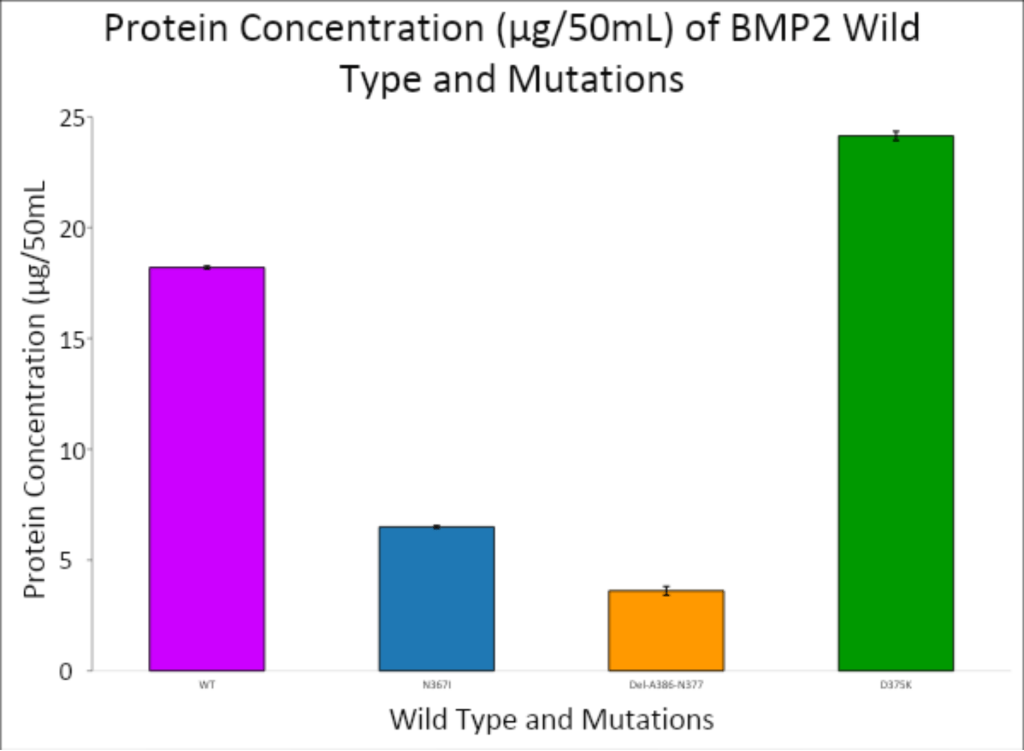
Protein expression was quantified using a spectrophotometer at 280 nm, targeting aromatic residues that correlate with protein concentration. The data, visualized in Figures 5 and 6, demonstrated a range of expression levels among the BMP-2 constructs.
- Wild-type BMP-2 showed a baseline absorbance of 1.435.
- N376I mutant had an average absorbance of 0.512, indicating lower protein production compared to WT.
- Del-A368-N377 mutant showed the lowest expression, with an average absorbance of 0.28475.
- D375K mutant, however, exhibited the highest protein yield, with an absorbance of 1.90325, surpassing the wild-type.
The corresponding protein concentrations reflected the absorbance values and suggested that the D375K mutation may enhance protein expression or stability. This observation is noteworthy, as increased expression could be linked to improved osteogenic function—a key aim of the study.
Observational Trends and Comparative Analysis
Several trends emerged from the data:
- Mutants with deletions or significant structural changes (e.g., Del-A368-N377) resulted in diminished protein expression, potentially due to impaired folding or instability.
- Conservative point mutations like N376I also produced lower expression, indicating limited functional enhancement.
- In contrast, the D375K mutation, which involved a charge and size shift (aspartate to lysine), not only retained bacterial growth but also increased protein expression.
This trend suggests that the D375K substitution might improve translation efficiency or protein folding, making it a promising candidate for further osteoblast-based functional assays.
Discussion
The results of this study show the promise of engineered BMP-2 mutations, specifically D375K, to have a positive increase in protein expression, and possibly, osteogenic function. D375K had the greatest absorbance at 280 nm (1.90325) of the three BMP-2 mutants and more than wild-type BMP-2 (1.435), which likely indicates both greater protein expression or structural stability. Any increase in protein yield is likely a factor for greater biological activity along osteoblast-inductive pathways11.
The D375K mutation has a lysine, a positively charged amino acid, instead of an aspartate, a negatively charged amino acid, due to the substitution of amino acids of different characteristics. This shift in charge may drive differentiation of how BMP-2 folds or interacts with receptors, potentially contributing to dimerizing or activation of the Smad signaling pathway critical for osteoblast differentiation. There is support in the literature to the notion that amino acid changes polarity or charge, specifically in the functionality, can alter protein signaling and thereby, cellular outcomes17.
Engineered BMP-2 variants, especially those with augmented protein intrinsic activity, are more directed in the treatment of osteoporosis as compared to currently available therapies (bisphosphonates; hormone therapy, etc.) that also pose risks for gastrointestinal distress as well as very rare bone issues. With greater protein intrinsic activity, potential doses could be lowered with reduced risk of side effects.
This study has its limitations; within E. coli as a heterologous system, we were unable to examine mammalian post-translational modifications, which meant that we could only measure protein yield, not the osteogenic function of the protein. Future experiments should include expressing D375K in mammalian or human osteoblast precursor cells followed by functional assays including, ALP activity measurements, mineralization, and the activation of the Smad pathway, in order to evaluate D375K’s performance in systems that are clinically relevant.
Conclusion
In summary, this study showed that careful mutations of BMP-2, specifically D375K, can enhance protein yield in a bacterial system. The highest yield of any of the mutants was D375K. This suggests that certain substitutive amino acids can alter the structural or translational efficiency of BMP-2 protein. Previous studies performed by Sebald et al. and Siverino et al. have demonstrated that BMP-2 variants have potential to alter osteogenic activity in vivo27,28. The advantage of a more active or stable form of BMP-2 is that with the anticipated reduction in dosages, side effects may be reduced, and bone regeneration can be improved. More validation is needed. Functional testing of the D375K mutant has to be done in mammalian or human osteoblast precursor cells to examine common markers of BMP-2 induction, such as ALP activity and Smad signaling. Additionally, using lysine-targeting delivery systems may provide a useful strategic method for enhancing tissue-specific delivery of BMP-2, which is key to facilitating clinical outcomes of BMP-2 delivered following restoration of bone repair and osteoporosis.
References
- Liang, B., Burley, G., Lin, S., & Shi, Y. C. (2022). Osteoporosis pathogenesis and treatment: existing and emerging avenues. Cellular & Molecular Biology Letters, 27(1), 72. [↩]
- Elahmer, N. R., Wong, S. K., Mohamed, N., Alias, E., Chin, K. Y., & Muhammad, N. (2024). Mechanistic insights and therapeutic strategies in osteoporosis: a comprehensive review. Biomedicines, 12(8), 1635. [↩]
- Song, S., Guo, Y., Yang, Y., & Fu, D. (2022). Advances in pathogenesis and therapeutic strategies for osteoporosis. Pharmacology & Therapeutics, 237, 108168. [↩]
- Ma, M., Zeng, H., Yang, P., Xu, J., Zhang, X., & He, W. (2023). Drug delivery and therapy strategies for osteoporosis intervention. Molecules, 28(18), 6652. [↩]
- Xu, J., Yu, L., Liu, F., Wan, L., & Deng, Z. (2023). The effect of cytokines on osteoblasts and osteoclasts in bone remodeling in osteoporosis: a review. Frontiers in Immunology, 14, 1222129. [↩]
- Rauner, M., Taipaleenmäki, H., Tsourdi, E., & Winter, E. M. (2021). Osteoporosis treatment with anti-sclerostin antibodies—mechanisms of action and clinical application. Journal of Clinical Medicine, 10(4), 787. [↩] [↩]
- Kostenuik, P. J., Binkley, N., & Anderson, P. A. (2023). Advances in osteoporosis therapy: focus on osteoanabolic agents, secondary fracture prevention, and perioperative bone health. Current Osteoporosis Reports, 21(4), 386–400. [↩]
- Daponte, V., Henke, K., & Drissi, H. (2024). Current perspectives on the multiple roles of osteoclasts: Mechanisms of osteoclast–osteoblast communication and potential clinical implications. Elife, 13, e95083. [↩]
- Martini, F., Pellati, A., Mazzoni, E., Salati, S., Caruso, G., Contartese, D., & De Mattei, M. (2020). Bone morphogenetic protein-2 signaling in the osteogenic differentiation of human bone marrow mesenchymal stem cells induced by pulsed electromagnetic fields. International Journal of Molecular Sciences, 21(6), 2104. [↩]
- Zhou, L., Wang, J., & Mu, W. (2023). BMP-2 promotes fracture healing by facilitating osteoblast differentiation and bone defect osteogenesis. American Journal of Translational Research, 15(12), 6751. [↩]
- Ingwersen, L. C., Frank, M., Naujokat, H., Loger, K., Bader, R., & Jonitz-Heincke, A. (2022). BMP-2 long-term stimulation of human pre-osteoblasts induces osteogenic differentiation and promotes transdifferentiation and bone remodeling processes. International Journal of Molecular Sciences, 23(6), 3077. [↩] [↩] [↩]
- del Castillo-Santaella, T., Ortega-Oller, I., Padial-Molina, M., O’Valle, F., Galindo-Moreno, P., Jódar-Reyes, A. B., & Peula-García, J. M. (2019). Formulation, colloidal characterization, and in vitro biological effect of BMP-2 loaded PLGA nanoparticles for bone regeneration. Pharmaceutics, 11(8), 388. [↩]
- Valcourt, U., & Moustakas, A. (2005). BMP signaling in osteogenesis, bone remodeling and repair. European Journal of Trauma, 31(5), 464–479. [↩]
- Medici, M., van Meurs, J. B., Rivadeneira, F., Zhao, H., Arp, P. P., Hofman, A., … & Uitterlinden, A. G. (2006). BMP-2 gene polymorphisms and osteoporosis: The Rotterdam Study. Journal of Bone and Mineral Research, 21(6), 845–854. [↩]
- Oliveira, J. E., Suzuki, M. F., Damiani, R., Lima, E. R., Amaral, K. C., Santos, A. M., … & Bartolini, P. (2021). Synthesis of human bone morphogenetic protein-2 (hBMP-2) in E. coli periplasmic space: its characterization and preclinical testing. Cells, 10(12), 3525. [↩]
- Lee, J. H., Jang, S. J., Koo, T. Y., Suh, C. W., Lee, E. N., Lee, K. M., … & Baek, H. R. (2011). Expression, purification and osteogenic bioactivity of recombinant human BMP-2 derived by Escherichia coli. Tissue Eng Regen Med, 8(1), 8–15. [↩]
- Watanabe, S., Ito, M., & Kigawa, T. (2021). DiRect: Site-directed mutagenesis method for protein engineering by rational design. Biochemical and Biophysical Research Communications, 551, 107–113. [↩] [↩]
- Phue, J. N., Lee, S. J., Trinh, L., & Shiloach, J. (2008). Modified Escherichia coli B (BL21), a superior producer of plasmid DNA compared with Escherichia coli K (DH5α). Biotechnology and Bioengineering, 101(4), 831–836. [↩]
- Heyde, S. A., & Nørholm, M. H. (2021). Tailoring the evolution of BL21 (DE3) uncovers a key role for RNA stability in gene expression toxicity. Communications Biology, 4(1), 963. [↩]
- Rouached, H. (2010). Efficient procedure for site-directed mutagenesis mediated by PCR insertion of a novel restriction site. Plant Signaling & Behavior, 5(12), 1547–1548 [↩]
- Rouached, H. (2010). Efficient procedure for site-directed mutagenesis mediated by PCR insertion of a novel restriction site. Plant Signaling & Behavior, 5(12), 1547–1548. [↩] [↩]
- Crossley, B. M., Bai, J., Glaser, A., Maes, R., Porter, E., Killian, M. L., … & Toohey-Kurth, K. (2020). Guidelines for Sanger sequencing and molecular assay monitoring. Journal of Veterinary Diagnostic Investigation, 32(6), 767–775. [↩]
- Gallegos, J. E., Rogers, M. F., Cialek, C. A., & Peccoud, J. (2020). Rapid, robust plasmid verification by de novo assembly of short sequencing reads. Nucleic Acids Research, 48(18), e106–e106 [↩]
- Agrawal, S., Padmaswari, M. H., Stokes, A. L., Maxenberger, D., Reese, M., Khalil, A., & Nelson, C. E. (2024). Optimizing Recombinant Cas9 expression: insights from E. coli BL21 (DE3) strains for enhanced protein purification and genome editing. Biomedicines, 12(6), 1226. [↩]
- Khan, F., He, M., & Taussig, M. J. (2006). Double-hexahistidine tag with high-affinity binding for protein immobilization, purification, and detection on Ni− nitrilotriacetic acid surfaces. Analytical Chemistry, 78(9), 3072–3079. [↩]
- Berrow, N. S., Büssow, K., Coutard, B., Diprose, J., Ekberg, M., Folkers, G. E., … & Busso, D. (2006). Recombinant protein expression and solubility screening in Escherichia coli: a comparative study. Biological Crystallography, 62(10), 1218–1226. [↩]
- Sebald HJ, Klenke FM, Siegrist M, Albers CE, Sebald W, Hofstetter W. Inhibition of endogenous antagonists with an engineered BMP-2 variant increases BMP-2 efficacy in rat femoral defect healing. Acta Biomater. 2012 Oct;8(10):3816-20. doi: 10.1016/j.actbio.2012.06.036. Epub 2012 Jun 29. PMID: 22750247. [↩]
- Siverino C, Fahmy-Garcia S, Mumcuoglu D, Oberwinkler H, Muehlemann M, Mueller T, Farrell E, van Osch GJVM, Nickel J. Site-Directed Immobilization of an Engineered Bone Morphogenetic Protein 2 (BMP2) Variant to Collagen-Based Microspheres Induces Bone Formation In Vivo. Int J Mol Sci. 2022 Apr 1;23(7):3928. doi: 10.3390/ijms23073928. PMID: 35409290; PMCID: PMC8999711. [↩]