Abstract
Toxic heavy metals in industrial wastewater present immediate environmental and health risks, urging scientists to explore different remediation techniques (the process of purifying pollutants from a medium). Conventional remediation methods are often costly, inefficient, and produce secondary waste in the form of sludge. In contrast, bioremediation via laccases—enzymes naturally found in fungi, plants, and bacteria—is an eco-friendly alternative that removes heavy metals through various mechanisms. However, laccases suffer from limited stability under industrial conditions, narrow optimal pH and temperature ranges, and low natural yields that prevent their implementation in industrial scales. Scientists have developed genetic engineering techniques like directed evolution, site-directed mutagenesis, and recombinant expression to combat these shortcomings. Yet, full industrial adoption is still limited by key barriers, including high large-scale production costs, lack of long-term performance data under continuous-flow industrial conditions, and incomplete analyses of economic feasibility1. This review discusses the structural and functional properties of laccases, their mechanism for heavy metal remediation, and recent genetic engineering advancements. Additionally, it assesses scalability for industrial use and outlines future research directions needed to translate these technologies to an industrial scale.
Introduction
Industrial operations – including mining, electroplating, and textile manufacturing – regularly discharge wastewater containing toxic heavy metals into aquatic systems. Industrial wastewater is defined as effluent discharged from manufacturing and processing facilities, with composition, pH, metal speciation, and contaminant concentrations varying by industrial sector. Mining wastewater typically contains substantial concentrations of arsenic, lead, and copper at highly acidic pH, often exceeding 400mg/L of total dissolved metals2. Electroplating effluents are characterized by hexavalent chromium Cr(VI), nickel and zinc, with chromium concentrations frequently reaching 200mg/L3. Similarly, tannery operations produce acidic, chromium-laden effluents (~600mg/L total Cr from a tannery in Sialkot) from leather tanning processes4. This review examines laccase-based remediation developed primarily for electroplating, tannery and textile wastewater as they are three of the most extensively studied sources of hexavalent chromium pollution. However, the findings are potentially generalizable to other heavy metal contaminated industrial effluents with compatible physiochemical characteristics, such as matching pH profiles, temperature, and the absence of laccase-inhibiting co-contaminants.
Studies from regions with concentrated industrial activity shows that heavy metal concentrations in wastewater often exceed permissible limits set by the World Health Organization (WHO) and other regulatory bodies. A recent example is the 2025 Sino-Metals Leach Zambia dam disaster, where approximately 50 million liters of heavy metal-laden acidic effluent were released into the Kafue River basin. This incident resulted in massive fish deaths, ecosystem damage, and heavy metal poisoning on local communities relying on the river5.
On a smaller scale, just one gas company (LOUISVILLE GAS & ELECTRIC CO,) has released significant heavy metal pollutants into sources providing drinking water for surrounding communities, even though heavy metals can cause allergy and carcinogenicity at high concentrations6. Unlike organic pollutants that degrade over time, heavy metals persist indefinitely, accumulating in sediments and undergoing biomagnification through aquatic food chains – a process where toxins increase in concentration as they move up trophic levels. Due to their inherent harmful nature, researchers have developed different methods to address heavy metal contamination in wastewater7.
Conventional physiochemical methods for wastewater include chemical filtration, ion exchange, membrane filtration and electrocoagulation. While they can achieve high removal efficiencies (89 – 99% for lead, copper nickel and cadmium), these methods face many operational challenges. Chemical precipitation generates substantial metal hydroxide sludge requiring specialized disposal. Ion exchange suffers from high regeneration costs and fouling issues8. Considering these shortcomings, scientists have turned to enzymatic bioremediation, a process that uses living organisms or their enzymes to accelerate degradation reactions. Enzymes offer unique advantages for environmental applications and can exhibit extremely high efficiency for the specialized reactions they do. Some enzymes can process thousands of metal ions per catalyst molecule while operating under “regular” temperature and pressure conditions. Among the most promising are laccases (EC 1.10.3.2, p-diphenol:dioxygen oxidoreductases), due to their ability to catalyze the oxidation of a broad spectrum of substrates like phenols, amines and certain heavy metals ions9.
Given their potential, researchers have experimented on genetically improving these enzymes. Newly developed genetically modified microorganisms (GMM’s) have enhanced capabilities for heavy metal detoxification: engineered bacteria have demonstrated removal efficacies exceeding 90% for lead and cadmium under optimized conditions, demonstrating their potential for scalability in environmental applications10.
However, challenges in enzyme stability and environmental adaptability still limit the effectiveness of laccase-based bioremediation11. This problem is compounded by the complex composition of industrial wastewater, which often contains pollutants that interfere with enzyme activity. Therefore, a knowledge gap currently exists in understanding how genetically engineered enzymes perform long-term under these industrial conditions. While some studies praise laccases for their broader-spectrum pollutant degradation, others mention concerns regarding their stability, economic feasibility and suboptimal remediation outcomes, hindering deployment of these biotechnologies at industrial scales12.
This review situates laccases within the broader framework of microbial enzymatic bioremediation. It compares different mechanisms of bioremediation and explores how genetic engineering methods can improve efficiencies and performances of these enzymes for heavy metal remediation. Furthermore, this review evaluates the scalability of implementing microbial technologies on a larger, industrial scale, focusing on factors like enzyme stability, operational costs and environmental conditions. By synthesizing recent advances and addressing operational challenges, this review provides a comprehensive evaluation of laccases’ suitability for heavy metal remediation in industrial wastewater.
Laccase Structure and Mechanisms of Heavy Metal Removal
Laccases are multi-copper oxidases that catalyze the oxidation of a variety of phenolic and non-phenolic compounds. Widely distributed in fungi, plants and bacteria, they are particularly prominent in bioremediation due to their high redox potential and effectiveness in degrading a wide variety of compounds. These compounds include synthetic dyes, polycyclic aromatic hydrocarbons (PAHs), and pharmaceutical residues12.
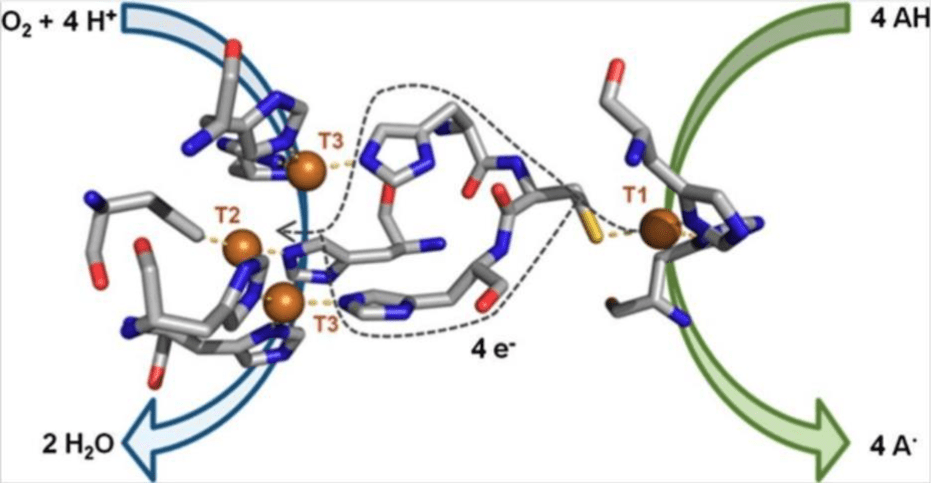
Structurally, laccases are glycoproteins that can exist as either single units (monomeric), pairs (dimeric), or clusters of four (tetrameric). Each monomer contains four copper atoms arranged in three distinct copper centers (as shown in Figure 1).
There are two main mechanisms through which laccases transform heavy metals: direct oxidation or indirect reduction mediated by organic co-substrates.
Although the direct oxidation mechanism is not as common, a well-documented exception is the conversion of Manganese (Mn(II) to Mn(III)). In a foundational study by Schlosser and Höfer, a purified laccase from Trametes versicolor directly oxidizes the soluble Mn2+ ion in the presence of Na-pyrophosphate. The oxidation occurs at the type 1 (T1) copper in the laccase active site, which is the primary electron-accepting site in the enzyme. The reaction occurs optimally at a pH of 5.0. However, this process requires chelators such as oxalate, malonate, or pyrophosphate to stabilize the Mn3+ product13.
In the indirect reduction mechanism, the process also begins with the Type 1(T1) copper site. Often called the “blue copper” site due to its distinct spectroscopic properties, the T1 copper is responsible for oxidizing substrates by abstracting an electron from the organic substrate.
The type 2(T2): acts as an electron transfer intermediary, shuttling electrons from the T1 site to the nuclear center (T3).
Type 3 (T3) is a binuclear center that reduces molecular oxygen (O2) to water (H2O), completing a four-electron reduction of oxygen (equation 1). Together, the T2 and T3 copper centers comprise the trinuclear cluster (TNC). Laccases catalyze this reaction without generating harmful reactive oxygen species (e.g. superoxide radicals or hydrogen peroxide), which significantly reduces oxidative damage to non-target compounds during the bioremediation processes14.
(1) ![]()
The combination of a strong oxidative potential at the T1 site and the safe reduction of oxygen to water allows laccases to efficiently oxidize a broad range of substrates, while minimizing secondary pollution, making them ideal for various environmental applications. An example of this type of mechanism is hexavalent chromium, Cr(VI). It is a carcinogen characterized by its high bioavailability, meaning it dissolves easily (more soluble) and can be readily absorbed by living organisms. Chromium’s trivalent form Cr(III) is more stable, less soluble and exhibits significantly lower toxicity16. Thus, it is desirable to convert Cr(VI) to Cr(III) through the following reduction reaction. (equation 2)
(2) ![]()
One study was able to deploy purified laccase from G. multipleum to reduce >94% of Cr(VI) with 100µg/ml concentration under acidic conditions (pH 3) and a temperature of 70˚C17. This percentage value often varies, depending on the laccases’ original source and complexity of the system. Here, “complexity of the system” refers to the composition of the contaminated medium beyond a single contaminant. It encompasses factors that interfere with laccase activity, such as the presence of competing ions or fluctuating pH levels. Essentially, complex systems can reduce the overall efficiency of chromium reduction. For instance, laccase from the fungal species Aspergillus flavus and isolates of dark septate endophytic fungi reduced chromium by 89.1% and 99%, respectively at the concentration 50 mg/L in a simple environment with no potential interference, while Trametes versicolor from a white rot fungus achieved a relatively low 32.2% removal efficiency in more complex systems (Whole cell)18,19,20.
| Fungal Source | Initial Conc. | Removal Efficiency (%) | Optimal pH | Optimal Tempreature | Reaction time (h) | Waste water type |
| Ganoderma multipleum | 100 pg/mL | 94.0 | 2.5-3.0 | 25-30 | 24 | Synthetic / Lab |
| Aspergillus flavus | 50 mg/mL | 89.1 | 4.5 | 30 | 48 | Efectroplatin g |
| Pseudomona s aeruginosastrain ZM130 | 25 mg/L | 76.6-98.7 | Not reported | Not reported | 180 | Simulated Textile |
| Dark septateendophytic fungi (multiple) | 10 — 50 mg/L | Up to 99.0 | 5.6 | Room Temperature Generally 20 – 25 | Not specified | Synthetic /Lab |
| Trametes Versicolor | 30 mg/mL | 32.29 | 4.0 (May not be optimal) | Not reported | 288 | Dye-bearing |
Analysis of table 1 shows several patterns regarding operational parameters of fungal systems for Cr(VI) remediation. First, the environments for optimal activity are restrictive. Most systems require acidic conditions (pH of 3.0 – 5.0), which misalign with many neutral or alkaline industrial streams. Optimal temperatures values are relatively low, ranging from 25 – 30°C while some purified enzymes like that from G. multipileum are thermotolerant (70°C.) Second, performance is highly context dependent. In single-pollutant systems particularly those tested in synthetic wastewater, removal efficiencies are consistently high, ranging from 89.1% – 99.0%. However, as demonstrated by the T. versicolor in a dye-bearing co-contaminant mixture, efficiency can plummet by over 60%. Removal rates follow the same pattern, varying widely from 24 hours in single cell pollutants to 288 hours in co-contaminant mixtures. These findings on the potential and limitations of fungal bioremediation were identified through a systematic review of the literature. The methodology for this analysis is detailed in the following section.
Methods
A literature review was conducted using the electronic databases of PubMed and Google Scholar. Additional information was incorporated from government organizations, including the U.S Environmental Protection Agency. Searches were done through the keywords (“laccases” OR “laccase”) AND (“heavy metals OR chromium) AND (“biodegradation” OR “remediation” OR “removal”) AND (“wastewater” OR industrial effluent”).
Inclusion and Exclusion Criteria
Inclusion Criteria: Original Research Articles (both in vivo and in vitro) that a) specifically investigated laccase-based remediation of heavy metals b) provided quantitative data related to the main search terms.
Exclusion Criteria: Studies were excluded if a) focused on organic pollutants without heavy metal involvement b) where full text was not accessible
Potential Risk Bias
Several limitations and potential biases should be considered when interpreting the findings of this review. For instance, a) selection bias: studies were chosen to convey a clearer narrative regarding the comparative efficacy of laccase-based remediation. b) publication bias: journals preferentially publish studies with positive results as opposed to negative ones.
Thematic Analysis Approach
The final included studies were analyzed using a thematic synthesis approach. Key findings, mechanisms, and limitations were grouped into themes, allowing for the development of a coherent narrative that structures the review.
Results
While laccases demonstrate exceptional capabilities in heavy metal remediation, several mechanistic and operational flaws restrict their practical application11. For one, despite their high redox potential, laccases often cannot directly reduce certain heavy metals like Cr(VI) due to mismatches in redox potential. To overcome this issue, scientists use redox mediators such as 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) or 1-hydroxybenzotriazole (HBT) to shuttle electrons between the enzyme and target pollutants. Mediators can introduce several drawbacks, such as additional costs and the potential toxicity of its radicals21. Furthermore, laccases generally lose their activity over time. When oxidizing heavy metals, they often interact with co-substrates or mediators to produce free radicals (an atom, molecule, or ion that has at least one unpaired electron in its outer shell, making it highly reactive and unstable.) Free radicals reportedly inactivate laccases, though the reason why is relatively unexplored10. Additionally, since it is difficult to recover laccases from aqueous samples, they are often considered a one-time use biocatalyst22.
Practical Limitations:
The reduction of heavy metals generally requires specific conditions (optimal pH, temperature and absence of inhibitors) to maintain activity. The same study that showed laccases reduced >94% of Cr(VI) with 100µg/ml concentration on purified laccase from Ganoderma multipileum noted that optimal conditions played a significant role in laccase activity, specifically requiring an acidic pH of 3 and a temperature of less than 70°C. When deviating from these optimal parameters, laccase activity decreased: at neutral to alkaline pH levels (above pH 6), enzyme activity declined by 50 – 70% compared to its maximum activity. Similarly, temperatures exceeding 70°C caused near inactivation of the enzyme17.
Furthermore, industrial wastewater typically contains a wide variety of inorganic and organic contaminants that potentially inhibit laccase’ catalytic function, exacerbating the problem of laccase efficiency in a “real-world” setting. These compounds either directly interact with the enzyme’s active site or alter their conformation (essentially changing its shape), affecting how well it can work or even deactivate it entirely. Wastewater streams also frequently exhibit wide fluctuations in pH and temperature—parameters that influence laccase stability and activity. As laccases generally have narrow optimal pH and temperature windows, any deviations from optimal conditions, including those typical of many industrial settings, cause reduced degradation efficiencies17.
Limited Yield + Production Cost:
The industrial scale production of laccase at a competitive cost is the biggest challenge behind it’s application in bioremediation. A 2025 tecno-economic analysis for producing Trametes hirsuta laccase provides a clear breakdown of these costs. The cost structure shows that operational expenditures (OpEx,) from labor accounted for 51.6% of the total cost. Capital expenditures (CapEx) for equipment depreciation were also significant at 42.2%, while raw materials for the culture medium constituted only 5.0%. This production cost is 7 to 12 times cheaper than some commercial laccases, which can range from 19.27 to 31.02 CAD kU⁻¹ (kilounit). However, for the vast quantities needed for large-scale wastewater remediation, this cost is still prohibitive compared to conventional methods23.
Genetic Engineering Advancements
To improve their applicability in heavy metal remediation, scientists have attempted to remedy laccases’ limitations through a variety of genetic engineering techniques, as compared in table 2.
| Engineering Strategy | Laccase Variant / Source | Modification | Key Properties Enhanced | Outcome | Reference |
| Directed Evolution | POXA1b variant (Pleurotus ostreatus) | Multiple rounds of mutagenesis and screening | Thermostability, pH tolerance | Increased half-life at 70°C; enhanced activity retention across pH 3-5. | (Pardo & Camarero, 2015) |
| Site-Directed Mutagenesis | POXA1b variant (Pleurotus ostreatus) | Substitution of axial methionine ligand at T1 copper site with Leu or Phe. | Mediator Kinetics, Redox Potential, Thermostability | ~60 mV increase in redox potential; however, stability of the T1 copper center was reduced. | (Zhu et al., 2019) |
| Computer-Guided Mutagenesis | “GreeDo” Variant (Fungal laccase) | Hydrophobic substitutions near the T1Cu site. | Redox Potential, Catalytic Efficiency, Thermostability | 7.5-fold improvement in catalytic efficiency for high-redox mediators (e.g., | (Mateljak et al., 2019) |
| Recombinant Expression | Laccase1 from Cryphonectria parasitica | Heterologous expression in Saccharomyces cerevisiae with optimized culture conditions. | Production Yield | Laccase activity reached 2.2 U/mL, a 6.5-fold increase over conventional media. | (So et al., 2025) |
| Immobilization | Trametes versicolor laccase | Enzyme immobilized on magnetic nanoparticles. | Reusability, Operational Stability | Allows for enzyme recovery and reuse. | (Muslim et al., 2024) |
Directed Evolution
Directed evolution is a laboratory technique that simulates natural selection to produce biological molecules with desirable traits. In the context of laccases, this approach focuses on properties related to industrial application, including pH and thermal stability, catalytic activity, and substrate specificity. The process begins with mutagenesis methods (DNA shuffling or error-prone PCR) to introduce random mutations toward specific regions likely to influence stability. Subsequently, thousands of mutants are screened for improved thermostability, tolerance to alkaline or acidic pH ranges, or resistance to denaturing solvents. The best-performing mutants are selected for further screening rounds. Through this process, beneficial mutations are accumulated, resulting in enzymes better suited for remediation applications24.
Researchers from the Spanish CSIC institution found that through multiple rounds of directed evolution, POXA1b variants (a gene encoding one of the laccase isoenzymes from the fungus Pleurotus ostreatus) showed increased half-life at 70°C and enhanced activity retention across a broader pH range (3-5)15. This enables laccases to be utilized in industrial wastewater environments, where heat and pH fluctuations are common.
While directed evolution relies on random mutations, another strategy for genetic engineering is site-directed mutagenesis. This involves precisely engineering laccases based on structural and functional insights. Knowing the role of each copper site is important here, as a laccases’ catalytic efficiency is directly linked to its copper centers, particularly the Type 1 (T1) site responsible for substrate oxidation.
The T1 copper site is the primary electron acceptor from substrates and determines the enzyme’s redox potential. Experimental substitutions of the axial methionine ligand with hydrophobic residues such as leucine (Leu) or phenylalanine (Phe) have been shown to increase redox potentials by ~60mV, enhancing the enzyme’s oxidative capacity toward high-redox-potential substrates (Figure 2.) The results were nuanced, however, as substitutions altered coordination geometry, leading to reduced stability of the T1 copper center, showing that computational design only demonstrates laboratory scale success and that there is more research needed to be done before industrial application.
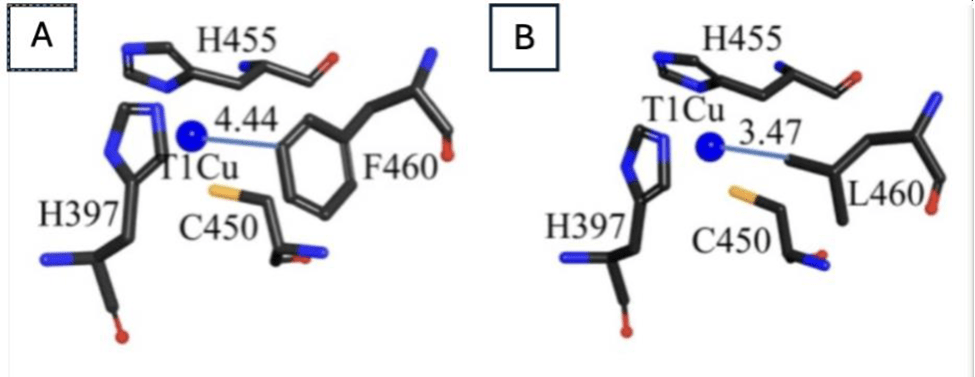
Novel approaches have also integrated computational design with targeted mutagenesis to successfully engineer high-redox-potential laccases (HRPLs’) with enhancements in catalytic efficiency and operational stability. Similarly, the study introduced hydrophobic amino acid substitutions near the T1Cu site in an evolved variant, dubbed GreeDo. GreeDo displayed a near 7.5-fold improvement in catalytic efficiency toward high-redox mediators, which act as electron shuffles in laccase mediator systems. Additionally, GreeDo’s stability improved. Its thermal half-life at 70˚C more than doubled – from 23 minutes to 60 minutes – showing the necessary robustness for sustained industrial processes26.
Finally, a third method of genetic engineering is called recombinant expression systems. Recombinant laccase refers to laccase enzymes produced via a specific type of genetic engineering, where the laccase gene from a source organism is inserted into a different host organism, to achieve higher expression and optimized production of the enzyme compared to their native source. Host organisms commonly used for recombinant laccase expression include bacteria like Escherichia coli and yeast species such as Pichia pastoris. These hosts are favored due to their rapid growth, adaptable metabolism (they can grow and metabolize a wide range of carbon and energy sources), and production of active protein. However, achieving optimal yields and protein quality in these systems often requires fine-tuning of production conditions. The choice of host depends on the target yield, protein quality, and key production parameters such as growth medium composition, induction protocols, pH, temperature, and feeding strategies11.
One example of successful recombinant expression of laccase is the expression of Laccase1 from Cryphonectria parasitica in Saccharomyces cerevisiae. The gene encoding for extracellular laccase from C. parasitica was cloned into a yeast episomal vector and transformed into S. cerevisiae. After successfully optimizing culture conditions, the recombinant yeast produced laccase with activity reaching 2.2 U/mL, a 6.5-fold increase compared to conventional media27.
Industrial Application
As established before, laccases possess intrinsic limitations that constrain their direct application in industrial wastewater bioremediation. Theoretically, genetic and protein engineering approaches have been shown to effectively enhance catalytic efficiency, pH thermostability, and production yield, thereby addressing these constraints. However, a large part of industrial applications has to do with operational factors: production cost and scalability.
Production Cost
Production cost remains one of the biggest drawbacks. Although advances in genetic engineering have enabled higher yields of recombinant laccases in recombinant hosts, the cost of producing and optimizing these enzymes at an industrial scale is still substantial. Bulk enzyme production costs generally range from $10 – 320 / kg, varying significantly by process scale and purity28.
A comprehensive laccase-based treatment considers costs across several categories: costs for cultivation, fermentation media and purification.
Recombinant expression in hosts does increase yield, but necessitates the complicated optimization of conditions, adding to bulk enzyme costs8.
Extra Costs: Immobilization + Mediators
While not genetic engineering per se, immobilization modifies how enzymes are used by confining them to a solid support. Research shows that immobilization improves reusability: immobilized laccase on activated carbon derived from pomegranate peels retained about 51% activity after 7 reuse cycles in pharmaceutical contaminant removal. The immobilized enzyme also showed better storage stability than free laccase, maintaining >90% of initial activity over a month at 25°C22. Essentially, immobilization is necessary for industrial scale remediation, ensuring that laccases can be reused more than once. The price for materials used for immobilization need to be taken into account. Supports such as polymeric beads, nanomaterials, and activated carbons add significant material costs, but specific percentage costs are context-dependent29.
As mentioned before, laccase-based remediation may include mediators for enhancing substrate range. Mediator use introduces additional operational costs and raises toxicity concerns30.
Conventional remediation costs range from about $0.30 – 0.70 per cubic meter of treated wastewater31. For sludge disposal alone, costs of roughly 500 euros per ton of dry mass, according to the type of treatment and disposal have been reported in European contexts, significantly higher than the original treatment cost32.
One cost-related factor where enzymes do have an advantage over conventional methods is energy consumption. Enzyme processes run under mild temperature and pressure; hence they take up lower energy needs relative to thermal or chemical treatments.
Another advantage compared to physicochemical methods is that laccase-based treatments produce significantly less secondary sludge, reducing disposal costs and environmental impact33.
Although there is no exact estimate on how much laccase treatment would cost for the remediation of heavy metals, it is very likely that with all the required modifications mentioned above, laccase-based wastewater treatment costs exceed those of conventional remediation methods, as summarized in Table 3.
| Cost Category | Engineered Enzyme | Conventional Approach |
| Production Cost | High due to enzyme optimization | Moderate: varies depending on method |
| Enzyme Immobilization | Adds material cost but improves reusability | Not applicable |
| Mediator Usage | Adds cost and potential toxicity | Not applicable |
| Energy Consumption | Low: operates under mild temperatures and pressure | High: thermal and chemical inputs required |
| Sludge Generation | Low sludge production | High sludge production requiring costly disposal |
| Overall Operational Cost | Currently high but can decrease with tech improvements | Generally lower but with significant environmental costs |
Scalability
To implement laccases in industrial settings, researchers need to establish a definitive way to produce large amounts of laccases while keeping in mind cost. Recombinant expression has made large-scale bioremediation seem feasible, yet producing these enzymes cost- effectively at large scale is still complex, which is why most applications with recombinant laccases are commercial: the first usage of recombinant laccase was recorded in 1996 for denim fishing, then for the construction of biosensors for detection of acid. Since then, laccases have been applied for bioremediation purposes, especially for the treatment of synthetic dyes, and recently, PAH’s, PCB’s and heavy metals34.
However, Enzyme quantities needed for these industrial treatments are much larger. For example, the Soktas project requires treating a wastewater volume of approximately 7,000 cubic meters per day35. There is no definitive method to quantify the amount of laccase needed for projects like Soktas, since enzyme expression is measured in activity units per liter (U/L). An interesting yet speculative benchmark can be made, however. Researchers found that the estimated cost of treating 1L of wastewater using commercial and crude grade laccase was $2 – 60 and $1 – 60 respectively36. This implies that to treat 7000 cubic meters of wastewater, the equivalent of seven million liters, it would cost around $7 million on the low end and $420 million on the high end. Granted, this estimation is quite dubious and should be taken with a grain of salt, and it should only serve to illustrate the potential financial burden associated with large-scale treatment. This issue of production cost is prevalent in studies, as several authors cite the lack of cost-effective laccase production to be the primary bottleneck preventing industrial application37.
Semi-industrial studies have validated the use of enzyme-based treatments in real wastewater streams. For example, engineered laccase from T. versicolor immobilized on magnetic nanoparticles has been used to treat dye and heavy metal contaminated textile effluents, achieving over 90% removal of azo dyes and significant reduction of metals like Pb, Cr, and Cd. Similarly, recombinant laccase-expressing yeast has enabled treated electroplating wastewater to meet discharge standards while producing lower sludge volumes than chemical methods33. These are some of the few notable examples that utilize engineered laccases in industrial settings, bringing us to the most pressing issue: the limited data availability on long-term operational data.
Discussion
The biggest gap in laccase research is the lack of studies that evaluate the stability of laccase-based systems in continuous-flow industrial settings. A large majority of published work on laccase-based wastewater bioremediation remains at the laboratory or pilot scale. While these smaller-scale models highlight exemplary removal efficiencies under controlled conditions, there remains little published evidence documenting enzyme behavior over extended operational periods. Consequently, we do not yet know the impact of factors like the frequency and cost of re-immobilization or enzyme replenishment, or the impacts of real industrial wastewater fluctuations on laccases’ lifespan. For example, most available studies report laccase performance over several hours to a few days, whereas industrial effluent treatment requires consistent function for months or more.38.
Currently, the only use cases for laccases consist of reactions under milder conditions, with applications ranging from the food industry and a select few bleaching processes to cosmetics36. Application on industrial wastewater still requires further research on economic feasibility considering enzyme production, immobilization, and replenishment costs and full-scale continuous operation studies under realistic industrial conditions.
When compared quantitatively with physicochemical methods, laccase-based bioremediation offers lower energy consumption due to operation at mild temperatures and pressures and produces less secondary waste, reducing disposal costs, and environmental concerns. However, the higher enzyme production and immobilization costs render laccase treatments more expensive overall. Popular alternatives, particularly electrocoagulation, deliver variable removal efficiencies but benefit from scalability advantages31. A comprehensive cost-benefit analysis is necessary to convincingly position laccase bioremediation as a competitive industrial solution.
In sum, popular conventional methods, most notably electrocoagulation and filtration processes still hold an overall advantage due to their established scalability and often lower cost despite their own limitations. Further research advances must be made regarding the cost effective production of large quantities of laccase, and long term studies on laccase performance are key for their future implementation.
Conclusion
Current research on laccases for bioremediation for industrial wastewater consistently highlights limitations of native enzymes, primarily their instability in harsh wastewater conditions, reliance on mediators, and low native production yields. Genetic engineering approaches have been crucial in addressing these challenges, with techniques like directed evolution, site-directed mutagenesis and rational design demonstrating noteworthy improvements in catalytic efficiency, thermal and pH stability. However, the transition from laboratory success to industrial-scale application remains the primary bottleneck. Scientists must prioritize research on economically viable large-scale production and validate laccases in continuous-flow industrial settings. If these limitations are addressed, genetically engineered enzymes can become a cost-effective solution for heavy metal remediation in industrial wastewater.
Acknowledgements
Thank you for the guidance of Sabrina Hu from Cambridge University in the development of this research paper.
References
- P. Giardina, V. Faraco, C. Pezzella, A. Piscitelli, S. Vanhulle, G. Sannia. Laccases: a never-ending story. Cell Mol Life Sci. 67(3): 369-85, 2009, https://doi.org/10.1007/s00018-009-0169-1. [↩]
- K. K. Kefeni, T. A. M. Msagati, B. B. Mamba. Acid mine drainage: Prevention, treatment options, and resource recovery: A review. Journal of Cleaner Production. Vol. 151, pg. 475–493, 2017, https://doi.org/10.1016/j.jclepro.2017.03.082. [↩]
- G. Srisuwan, P. Thongchai. Removal of heavy metals from electroplating wastewater by membrane. Songklanakarin Journal of Science and Technology. Vol. 24, pg. 965–976, 2002. [↩]
- Z. Ali, R. N. Malik, A. Qadir. Heavy metals distribution and risk assessment in soils affected by tannery effluents. Chemistry and Ecology. Vol. 29, pg. 676–692, 2013, https://doi.org/10.1080/02757540.2013.810728. [↩]
- U.S. Embassy Lusaka. Health Alert – U.S. Embassy, Lusaka, Zambia: Acute Heavy Metal Poisoning, 2025. [↩]
- U.S. Environmental Protection Agency. National Primary Drinking Water Regulations. EPA.gov, 2024. [↩]
- T.E. Oladimeji, M. Oyedemi, M.E. Emetere, O. Agboola, J.B. Adeoye, O.A. Odunlami. Review on the impact of heavy metals from industrial wastewater effluent and removal technologies. Heliyon. 10(23), e40370, 2024, https://doi.org/10.1016/j.heliyon.2024.e40370. [↩]
- S. Charazińska, E. Burszta-Adamiak, P. Lochyński. The efficiency of removing heavy metal ions from industrial electropolishing wastewater using natural materials. Scientific Reports. 12, Article number: 17766 (2022.) https://doi.org/10.1038/s41598-022-22466-9. [↩] [↩]
- P. K. Robinson. Enzymes: principles and biotechnological applications. Essays in Biochemistry. 59, 1-41, 2015, https://doi.org/10.1042/bse0590001. [↩]
- Muslim, O. A., Bakr, H. M., Jwaziri, A. K., Ali, K. M. R., & Al-Yassiry, K. Removal of toxic heavy metals using genetically engineered microbes: Molecular tools, risk assessment, and management strategies. ACOPEN Journal – Section Microbiology, 9(2) ,2024, https://doi.org/10.21070/acopen.9.2024.10300. [↩] [↩]
- V. Chandra, K. Arpita, P. Yadav, V. Raghuvanshi, A. Yadav, S. Ali, V.M. Tripathi, S. Prajapati. Recombinant Laccase: A Promising Tool for Industrial Effluent Bioremediation. Science Publishing Group. 3(2), 2023, https://doi.org/10.11648/j.reports.20230302.12. [↩] [↩] [↩]
- P. Diep, R. Mahadevan, A.F. Yakunin. Heavy Metal Removal by Bioaccumulation Using Genetically Engineered Microorganisms. Frontiers in Bioengineering and Biotechnology. 6, 157, 2018, https://doi.org/10.3389/fbioe.2018.00157. [↩] [↩]
- D. Schlosser, C. Höfer. Laccase-Catalyzed Oxidation of Mn2+ in the Presence of Natural Mn3+ Chelators as a Novel Source of Extracellular H2O2 Production and Its Impact on Manganese Peroxidase. Applied and Environmental Microbiology. Vol. 68, pg. 3514–3521, 2002, https://doi.org/10.1128/AEM.68.7.3514-3521.2002. [↩]
- L. Arregui, M. Ayala, X. Gómez-Gil, G. Gutiérrez-Soto, C.E. Hernández-Luna, M. Herrera de los Santos, L. Levin, A. Rojo-Domínguez, D. Romero-Martínez, M.C.N. Saparrat, M.A. Trujillo-Roldán, N.A. Valdez-Cruz. Laccases: structure, function, and potential application in water bioremediation. Springer Nature. 18, 2019, https://doi.org/10.1186/s12934-019-1248-0. [↩]
- I. Pardo, S. Camarero. Laccase engineering by rational and evolutionary design. Cell Mol Life Sci. 72, 897–910, 2015, https://doi.org/10.1007/s00018-014-1824-8. [↩] [↩]
- U.S. Environmental Protection Agency. Chromium compounds hazard summary. EPA.gov ,2000. [↩]
- M.A. Alsheikheid, A. Umar, F. Ameen, S.A. Alyahya, L. Dufossé. Biodegradation of chromium by laccase action of Ganoderma multipileum. Journal of King Saud University – Science. 35, 102948, 2023, https://doi.org/10.1016/j.jksus.2023.102948. [↩] [↩] [↩]
- V. Kumar, S.K. Dwivedi. Hexavalent chromium reduction ability and bioremediation potential of Aspergillus flavus CR500 isolated from electroplating wastewater. Chemosphere. 237, 124567 ,2019, https://doi.org/10.1016/j.chemosphere.2019.124567. [↩]
- I. Melati, G. Rahayu, H. Effendi, C. Henny, E. Susanti, S. Surono. Chromium (VI) bioremediation potential of dark septate endophytic (DSE) fungi. IOP Conference Series: Earth and Environmental Science. 1201(1), 012077 , 2023, https://doi.org/10.1088/1755-1315/1201/1/012077. [↩]
- Z. Aksu, N. K. Kılıç, S. Ertuğrul, G. Dönmez. Inhibitory effects of chromium(VI) and Remazol Black B on chromium(VI) and dyestuff removals by Trametes versicolor. Enzyme and Microbial Technology. Vol. 40, pg. 1167–1174, 2007, https://doi.org/10.1016/j.enzmictec.2006.08.024. [↩]
- A. Chiadò, F. Bosco, M. Bardelli, L. Simonelli, M. Pedotti, L. Marmo, L. Varani. Rational engineering of the lccβ T. versicolor laccase for the mediator-less oxidation of large polycyclic aromatic hydrocarbons. Computational and Structural Biotechnology Journal. 19, 2021, https://doi.org/10.1016/j.csbj.2021.03.017. [↩]
- O.J. Al-Sareji, M. Meiczinger, R.A. Al-Juboori, R.A. Grmasha, M. Andredaki, V. Somogyi, I.A. Idowu, C. Stenger-Kovács, M. Jakab, E. Lengyel, K.S. Hashim. Efficient removal of pharmaceutical contaminants from water and wastewater using immobilized laccase on activated carbon derived from pomegranate peels. Scientific Reports. 13,12345, 2023, https://doi.org/10.1038/s41598-023-38821-3. [↩] [↩]
- K. Alokpa, H. Cabana. Techno-economic analysis of laccase production, immobilization and use of the immobilized biocatalyst in hospital wastewater bioremediation. Environmental Challenges. Vol. 20, pg. 101230, 2025, https://doi.org/10.1016/j.envc.2025.101230. [↩]
- D. Maté, E. Garcia-Ruiz, S. Camarero, M. Alcalde. Directed Evolution of Fungal Laccases. Current Genomics. 12(2), 113-122, 2011, https://doi.org/10.2174/138920211795564322. [↩]
- Y. Zhu, Y. Zhang, J. Zhan, Y. Lin, X. Yang. Axial bonds at the T1 Cu site of Thermus thermophilus SG0.5JP17-16 laccase influence enzymatic properties. FEBS Open Bio. 9, 482–492 , 2019, https://doi.org/10.1002/2211-5463.12633. [↩]
- I. Mateljak, et al. Increasing Redox Potential, Redox Mediator Activity, and Stability in a Fungal Laccase by Computer-Guided Mutagenesis and Directed Evolution. ACS Catalysis 9, 4561-4572, 2019, https://doi.org/10.1021/acscatal.9b00531 [↩]
- K.-K. So, F.A. Hernandez Alvarado, G.-H. Han, J.-W. Kim, T.-G. Kim, D.-H. Kim. Heterologous Expression of Laccase1 from Cryphonectria parasitica in Saccharomyces cerevisiae. Mycobiology. 53(1), 36–46, 2025, https://doi.org/10.1080/12298093.2024.2439646. [↩]
- R. d. Gama Ferreira, A. R. Azzoni, and S. Freitas. Techno-economic analysis of the industrial production of a low-cost enzyme using E. coli: the case of recombinant β- glucosidase. Biotechnology for Biofuels,11:81, 2018, https://doi.org/10.1186/s13068-018-1077-0. [↩]
- J. M. Bolivar, J. M. Woodley, R. Fernandez-Lafuente. Is enzyme immobilization a mature discipline? Some critical considerations to capitalize on the benefits of immobilization. Chem. Soc. Rev. 51, 6251-6290, 2022, https://doi.org/10.1039/d2cs00083k. [↩]
- A. Chiadò, F. Bosco, M. Bardelli, L. Simonelli, M. Pedotti, L. Marmo, L. Varani. Rational engineering of the lccβ T. versicolor laccase for the mediator-less oxidation of large polycyclic aromatic hydrocarbons. Computational and Structural Biotechnology Journal. 19, 2021, https://doi.org/10.1016/j.csbj.2021.03.017. [↩]
- O. A. Shaker, S. M. Safwat, and M. E. Matta. Nickel removal from wastewater using electrocoagulation process with zinc electrodes under various operating conditions: performance investigation, mechanism exploration, and cost analysis. Environ. Sci. Pollut. Res. Int. 30 (10), 26650–26662, 2022, https://doi.org/10.1007/s11356-022-24101-6. [↩] [↩]
- P. Foladori, G. Andreottola, and G. Ziglio. Sludge Reduction Technologies in Wastewater Treatment Plants. IWA Publishing, 2010. [↩]
- K.H. Hama Aziz, F.S. Mustafa, K.M. Omer, S. Hama, R.F. Hamarawf, K.O. Rahman. Heavy metal pollution in the aquatic environment: efficient and low-cost removal approaches to eliminate their toxicity: a review. RSC Advances. Vol 13, 17595-17610, 2023, https://doi.org/10.1039/D3RA00723E. [↩] [↩]
- C. Pezzella, L. Guarino, A. Piscitelli. How to enjoy laccases. Cellular and Molecular Life Sciences. Vol. 72, pg. 923–940, 2015, https://doi.org/10.1007/s00018-014-1823-9. [↩]
- Wastewater Treatment for Textile Industry, egesis. [↩]
- P. Mathur, D. Sanyal, P, Dey. Optimization of growth conditions for enhancing the production of microbial laccase and its application in treating antibiotic contamination in wastewater. Biotech. 11(2), 2021, https://doi.org/10.1007/s13205-020-02627-1. [↩]
- A.S. Sodhi, S. Bhatia, N. Batra. Laccase: Sustainable production strategies, heterologous expression and potential biotechnological applications. International Journal of Biological Macromolecules. 280 (Part 1), 135745 , 2024, https://doi.org/10.1016/j.ijbiomac.2024.135745. [↩]
- M. Maryskova, M. Vrsanska, A. Sevcu, V. Novotny, A. Blahutova, S. Voberkova. Laminated PAA nanofibers as a practical support for crude laccase: A new perspective for biocatalytic treatment of micropollutants in wastewaters. Environmental Technology & Innovation. 26, 102316, 2022. [↩]