Abstract
Natural Killer (NK) cells are crucial regulators of anti-tumor immunity, but their activity is significantly shaped by the tumor microenvironment across diverse cancer types. This study investigates how variations in cytokine and chemokine presence, reactive oxygen species (ROS) dynamics, and effector-to-target (E:T) ratios influence NK cell phenotype and cytotoxic function in Lung, Breast, Ovarian, Gastric, Renal, Colon & Colorectal cancers. By analyzing previously published studies of experimental killing assays and patient data, this narrative review demonstrates that higher E:T ratios and a predominance of mature NK cells are closely linked to increased cancer cell death. This data also suggests that pro-inflammatory cytokines (IL-12, IFN-γ) enhance NK cell function while immunosuppressive cytokines (IL-10) and elevated ROS levels suppress cytotoxicity. Single-cell and pathway data highlight how the balance of these molecules vary across tumor microenvironments. The findings support the development of NK cell based therapies aimed at restoring NK cell number, maturity, cytotoxicity and anti-tumor immunity, providing insight into the interplay between cytokines, ROS, and NK cell activity. This research is extremely important because it demonstrates why tumors often evade the body’s natural immune defenses, even when NK cells are present. By identifying how factors like cytokines, chemokines, and reactive oxygen species in the microenvironment influence NK cell function, this study points towards new strategies for improving cancer treatments.
Keywords: Natural Killer (NK) cells, effector to target (E:T), cytokines, reactive oxygen species (ROS), cytotoxicity, tumor microenvironment, apoptosis
Introduction
Natural Killer (NK) cells are a vital part of the immune system and play a critical role in controlling tumor progression. Unlike other lymphocytes such as T-cells, which require antigen specific receptors to act, NK cells can directly kill abnormal cells which have the capacity to become cancerous. T-cells need these receptors to precisely identify which specific antigens presented on Major Histocompatibility Complex (MHC) molecules to bind to, ensuring a targeted response. NK cells, part of the innate immune system, are instead regulated through a balance of activating and inhibitory receptors to detect abnormal cells, and often are in direct interaction with these cells, triggering cell death through distinct cytotoxic mechanisms. This gives NK cells a unique advantage of providing a rapid defense mechanism before the T-cell mediated response develops1.
The core process by which NK cells enact their killing mechanisms happens through a process called cytotoxicity, which refers to the capacity of immune cells to kill cells. In the context of cancer, cytotoxicity is especially important because it allows NK cells to kill growing tumor cells and suppress tumor growth. However, NK cell activity is often inhibited due to a suppressive tumor microenvironment and immune evasion strategies by tumor cells to avoid death. Studies have shown that reduced NK cell activity in certain cancers correlate with increased tumor growth and increased cancer development, highlighting the importance of NK cell cytotoxicity2. This raises the question: How does NK cell activity shape cancer tumor progression across different tissues or environments?
Understanding NK cell behavior is significant because it not only advances our knowledge of immune cell regulation and tumor progression, but also lays the potential framework for new approaches to cancer therapy that restore or enhance immune function. The purpose and objective of this research is to investigate the mechanisms by which NK cells mediate cytotoxicity and identify the factors that influence their effectiveness in the tumor microenvironment, leading to potential breakthroughs for cancer treatments. While this review uses publicly available data and published figures, it is limited by the availability of detailed data for these specific cancer types and varying methodologies across data sets. Nonetheless, this analysis is grounded on ideas about how the immune system works, including the effector to target ratio, and how changes in the tumor microenvironment create a large impact.
By drawing on NCBI resources, literature from PubMed, the Journal of Immunology, and integrating curated pathway and single-cell datasets from biological databases such as the Human Cell Atlas, this study aims to clarify the critical role of NK cells in shaping tumor progression. To understand how NK cell activity influences this progression, it is crucial to examine their primary cytotoxic mechanisms. First, this review will explore the primary cytotoxic mechanisms employed by NK cells, particularly apoptosis and necrosis, and their implications for tumor progression.
Apoptosis: Programmed Cell Death
A main mechanism through which NK cells eliminate their target cells is apoptosis, a programmed intracellular self-destruction that removes abnormal cells without causing damage or inflammation. This NK cell-mediated apoptosis occurs through two main pathways, the perforin/granzyme pathway or the Fas/Fas ligand (FasL) pathway, and results in the shrinking of cells controllably.
When NK cells recognize a target cell, they activate the perforin/granzyme cytotoxic pathway, in which NK cells release perforin, a protein that inserts into the target cell membrane, to form pores, allowing other molecules to pass through. These pores allow the entry of granzymes: enzymes released by NK cells that trigger apoptosis by activating caspases or caspase-independent pathways, leading to DNA damage and mitochondrial dysfunction. Caspase activation directly drives cell death and dismantling of the cell, while other functions within the apoptotic pathway help ensure the process remains contained, preventing damage to surrounding tissue1,3.
NK cells can also induce apoptosis extrinsically through the Fas/Fas ligand (FasL) pathway. The Fas receptor, also known as a death receptor, is found on the surface of lymphocytes and other immune cells. When FasL binds to Fas, the proteins, FADD and procaspase-8 are recruited, forming the death-inducing signaling complex (DISC). Within the DISC, procaspase-8 is cleaved to form caspase-8 , which then triggers a cascade of activating additional caspases. This cascade results in features of apoptosis, dismantling the target cell without any further inflammation1,3.
Necrosis: Cell Tissue Death
When apoptosis is evaded, NK cells can engage regulated necrotic cell death pathways, such as necroptosis or pyroptosis in some contexts, which leads to the rupture of the cell membrane, causing a release of intracellular content and triggerinh inflammation that can cause damage to tissues and neighboring cells. This process involves a destruction of cell membrane integrity, following a release of DAMPs (Damage-Associated Molecular Patterns), like ATP or HMGB1. Necrotic, DAMP driven inflammation can activate phagocytosis to eliminate target cells, but persistent and dysregulated necrosis in the tumor microenvironment can also contribute to excessive tissue damage and in certain settings, support tumor growth and progression4,5,6.
Together, apoptosis and necrosis represent two distinct strategies (see Figure 1) for eliminating harmful cells: apoptosis provides a controlled non-inflammatory technique to preserve tissue health, whereas necrosis involves a release of intracellular contents that can potentially damage tissue when controlled death is evaded.
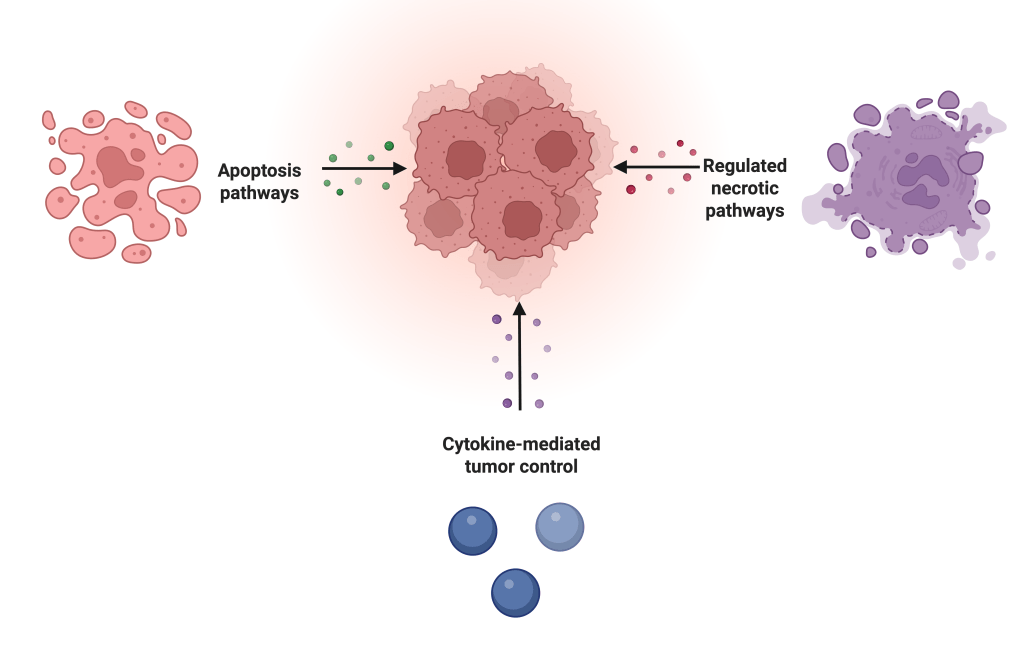
Ultimately, these cytotoxic functions are directly related to the cell cycle and regulation of tumor cells. Within the cell cycle, those that are dividing and growing rapidly are labeled as cancer cells. These cells cause stress signals like MICA or MICB which can activate NK cells or reduce MHC-I molecules, causing NK cells to attack tumor cells. NK cells arrest tumor growth, as they can target cells in the G1 or S phase, inhibiting further growth and removing these cancerous cells. The cell cycle is regulated not just by killing cells, but also through the secretion of cytokines (as shown in Figure 1) which also induce arrest, and stimulate immune responses. However, as tumors evolve, they may evade NK cell recognition and bypass checkpoints through altering their ligands or other methods, leading to the complex relationship between NK cells and cancer7,8.
Cancer Progression: ROS, Cytokines, Tumors
As tumors progress and evolve, their microenvironment changes accordingly, often causing harmful conditions for NK cells. One critical factor shaping NK behavior is the presence of ROS (reactive oxygen species). ROS, generated by tumors and immune cells, act as important molecules, helping to regulate cell growth and immune activity. In balanced amounts, ROS promotes NK and T cell activation9. However, once ROS levels exceed a threshold, they begin to have harmful effects, including cell death through apoptosis, necrosis, and promoting tumor development.
In cancer cells, these ROS levels are normally elevated due to increased metabolic activity and hypoxic conditions within the tumor microenvironment. Higher levels of ROS contribute to DNA damage, triggering the start of unchecked tumor proliferation. At the same time, ROS modulate signaling pathways, such as NF-κB, HIF-1α, and STAT3, which promote the survival of tumor cells. On the immune side, excess ROS can suppress NK cell function by downregulating activation receptors and inducing NK cell apoptosis, inhibiting the ability to kill target cells10,11. This not only lowers the clearance of tumor cells, but also promotes immune evasion and metastasis, as tumor cells continue to create an immunosuppressive environment (see Figure 2).
The relationship between ROS and NK cells is complex, but essential for understanding tumors. While NK cells are essential for eliminating abnormal or stressed cells, they are extremely sensitive to changes and stresses in the tumor microenvironment. At controlled levels, this makes ROS act as a positive regulator by enhancing NK cell activation and cytotoxicity. Elevated levels of ROS, however, shifts the balance toward tumor progression. Adaptive responses which normally help microenvironments, such as dendritic cell activation or T cell recruitment, are also impaired by the tumor microenvironments. These roles demonstrate the importance of ROS as a regulator between tumor progression and mediating the immune system12,13.
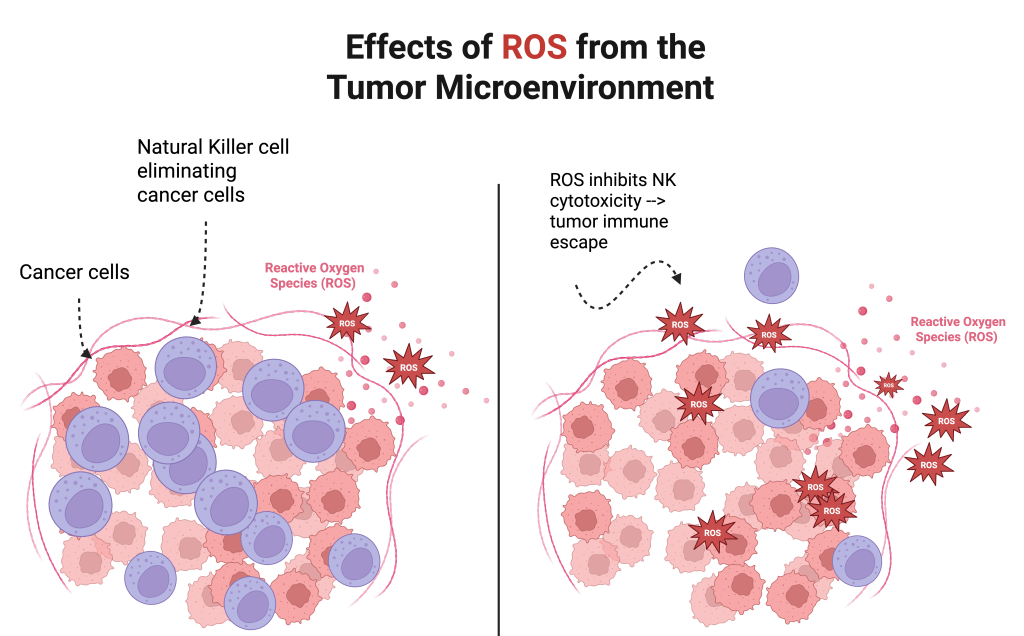
Elevated ROS levels promote an immunosuppressive microenvironment that favors tumor progression, as tumor cells begin to multiply (right). However, the left depicts an environment where ROS levels are normal, and NK cells are effectively eliminating cancer cells, suppressing tumor progression (left). Made in www.biorender.com.
Beyond ROS, NK cell function is also profoundly regulated by another class of signaling molecules: cytokines and chemokines, which influence NK cell behavior and activation. Cytokines such as IL-2, IL-15, TNF-α, IFN-γ play important roles when it comes to cytotoxicity (See Figure 3). They are signaling molecules that help facilitate the body’s immune and inflammation responses, influencing the function of NK cells and their ability to recognize and eliminate target cells. Chemokines, such as CXCL9 and CXCL10, help guide cytotoxic T cells and NK cells toward tumor sites, ensuring communication between the cell and enhancing anti-tumor immune responses. While these cytokines and chemokines facilitate NK function, tumors can resist NK attack by altering these molecules by secreting inhibitory cytokines or downregulating activating ligands14,15. This causes a reduced number of NK cell subsets and can suppress receptors on NK cells, therefore inhibiting their function and causing tumor progression1.
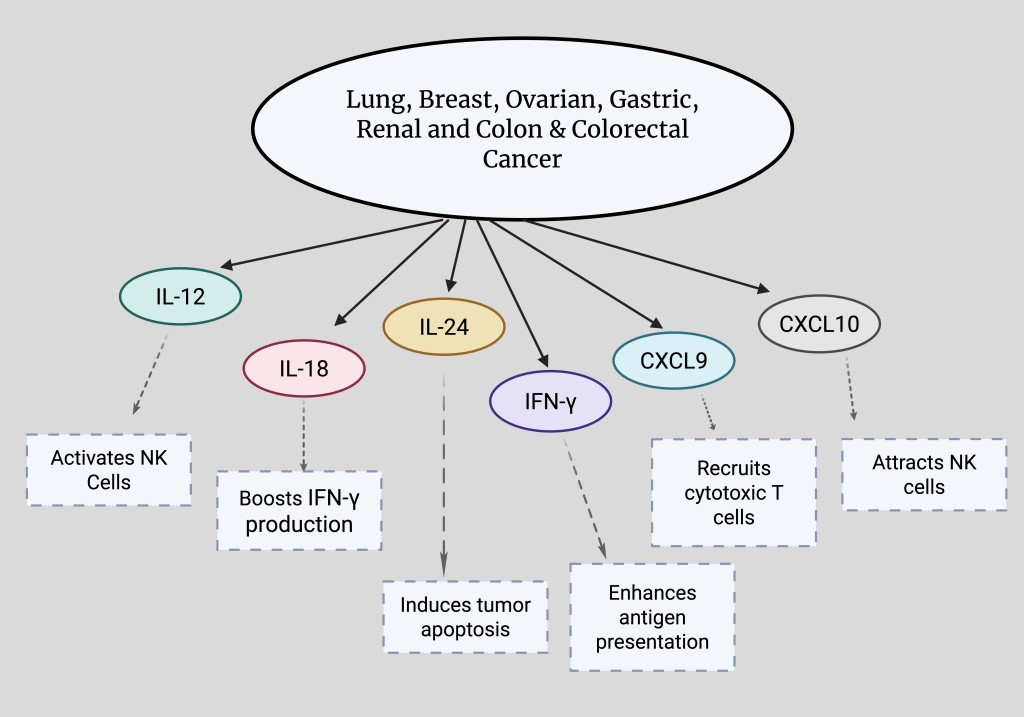
Positive roles of various cytokines (IL-12, IL-18, IL-24, IFN-γ) and chemokines (CXCL9, CXCL10) in lung, breast, ovarian, gastric, renal and colon & colorectal cancers. Cytokines regulate NK cell activity, while chemokines drive NK cell movement. Both interact with ROS pathways to influence cancer progression. Made with www.biorender.com.
NK Cell Numbers and Subsets Across Cancer Types
The abundance and state of NK cells play an important role in cancer progression and survival. Examining NK cell numbers and their subsets across different cancers provides insight into how these immune cells can either suppress or promote tumor growth.
Across cancers and progressive stages, reduced NK cell numbers are associated with poor survival rates and enhanced tumor growth. In contrast, higher NK cell numbers have led to improved survival in lung, colorectal, and many other cancers. However, there is one exception: triple-negative breast cancer (TNBC). In TNBC, a higher number of NK cells correlates with worse outcomes, as NK cells actually aid tumor progression. This paradox is because of an immature subset of CD56^bright NK cells. In healthy conditions, CD56^bright NK cells demonstrate reduced cytotoxic activity and are important for secreting cytokines such as TNF-α and IFN-γ, which help promote immune responses16,17. Within the TNBC tumor microenvironment, however, these cells fail to perform their role. Their immaturity exhibits reduced cytotoxicity due to low granzyme and perforin expression and also secrete cytokines in a way that favors tumor growth and immune evasion. While normally regulatory, this NK subset becomes one of the main factors in poor survival for TNBC.
This complex relationship between NK subsets and tumor outcomes highlights the importance of not just NK cell abundance, but NK cell maturity and function. In regards to therapeutic strategies, approaches that target ROS pathways or modulate cytokine signaling may help shift NK cells toward a more tumor-suppressive and cytotoxic phenotype.
Tissue Microenvironments
Key Cancer Types: Lung, Breast, Ovarian, Gastric, Renal, Colon & Colorectal
The study of major solid tumors, including lung, breast, ovarian, gastric, renal and colon/colorectal cancers, highlights how distinct tissue microenvironments influence NK cell activity and disease progression. Lung cancer, the leading cause of cancer-related deaths, develops within a highly immunosuppressive microenvironment with factors that impair immune cells and NK cells that are critical for early tumor clearance18,19. In breast cancer, particularly hormone receptor-positive and HER2-positive subtypes, NK cells often show reduced cytotoxicity and altered states20. Colorectal cancer progression reflects both genetic mutations and a microenvironment enriched in suppressive cytokines and metabolic stress, limiting NK maturation and infiltration21. Gastric and ovarian cancers, frequently diagnosed at advanced stages, feature hypoxic and cytokine rich niches that limit NK response, though ovarian tumors paradoxically show high NK cell activity along with suppression. Lastly, in renal cell carcinoma (kidney cancer) inflammatory and stress-related signals combine with immune cell infiltration to shape NK cell function. As explored in this paper, each of these cancerous functions depends heavily on the microenvironment’s ability to sustain or suppress NK cell activity22,23.
Despite their distinct origins, these diverse cancer tissue microenvironments share several critical features that collectively undermine the immune system and NK cell function. Some commonalities are increased levels of ROS, nutrient deprivation, acidic pH, and immunosuppressive cytokines, such as IL-10 (See Figure 4). These factors collectively work to suppress NK cell activity by reducing resources necessary for cytotoxicity and altering the balance within the cell22.
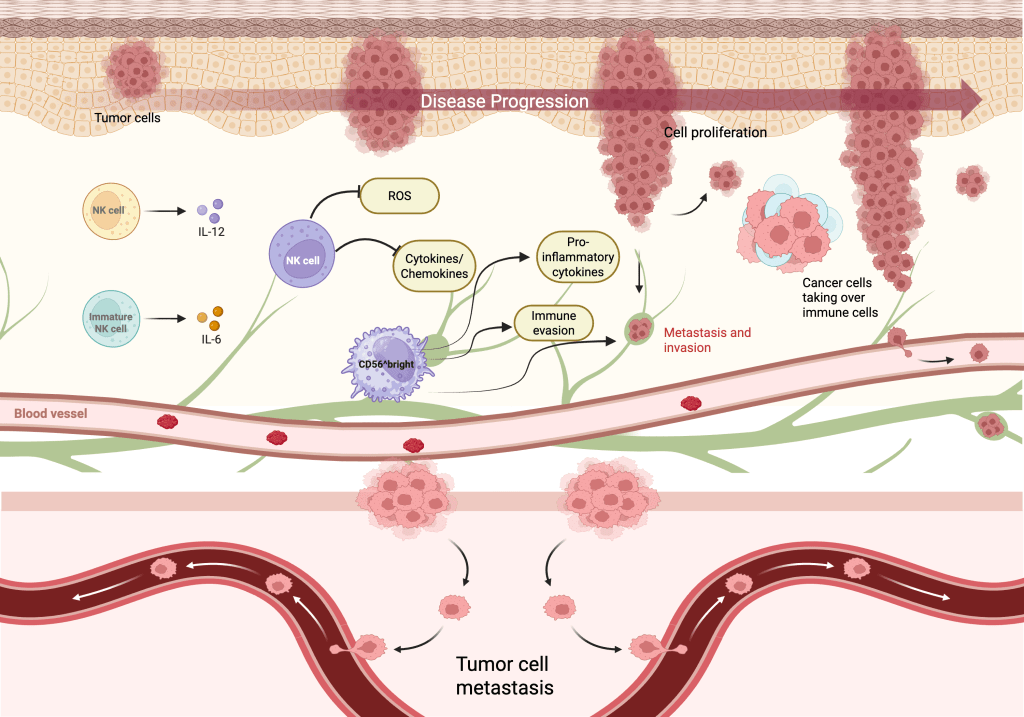
The diverse tumor microenvironments profoundly influence the dynamics of NK cells, impacting their activation, proliferation, and maturation at different stages of progression. In early stages, NK cells are more effectively recruited with higher cytotoxicity due to a higher number of cytokines and activating ligands on malignant cells. However, in later stages, as the tumor progresses, the environment becomes increasingly suppressive and reductions in NK cells start to appear, causing impairment and less mature phenotypes24,25,26. The complex interactions between NK cells, cytokines, ROS, and tumor cells is depicted in Figure 4, which shows how changes in concentrations of these molecules collectively shapes tumor cell proliferation, immune evasion, and metastasis across tumor stages.
Within these varying tumor microenvironments, the ratio of NK cells to target cells is a critical determinant of patient survival rates, and this ratio is significantly influenced by the microenvironmental conditions. High NK to target ratios are often associated with a controlled early stage, yet diminish as tumors begin to evade detection and change their microenvironment. The difference in this ratio is driven by processes involving cytokines and chemokines, and involves differing rates of NK cell expansion and proliferation. In more aggressive cancers, factors like TGF-β can reduce overall NK cell numbers and create immature subsets with reduced cytotoxicity27.
As mentioned before, immature subsets can be marked by high expression of CD56^bright. Other features of immature subsets include low CD16, receptors like NKG2A, or a lack of killer immunoglobulin receptors (KIRs). These immature subsets are more likely to have decreased cytotoxicity, however they excel at releasing cytokines and help to regulate the immune system16,28.But, with tumor progression, the prevalence of these subsets are enhanced. Increased immature NK cells across tumor microenvironments generally correlates with reduced antitumor immunity, impaired direct killing of cancerous cells, increased immune evasion, and poorer outcomes with more aggressive tumor behavior.
The top panel illustrates cell proliferation, where tumor cells multiply to form a larger mass. Mature NK cells, supported by IL-12 (in purple), kill tumor cells and help constrain tumor growth, while immature NK cells (CD56^bright) and IL-6 (in orange). The involvement of IL-6 is immunosuppressive in this case, as its role is highly context-dependent since IL-6 operates as a mediator for cytokine shifting microenvironments to reduce cytotoxicity. ROS and cytokines/chemokines form a signaling network that can initially promote NK cell activation, but when elevated, they suppress NK cytotoxicity and can evade the immune system (immune evasion), taking over immune cells. This causes metastasis and the invasion of surrounding tissue and blood vessels. The bottom panel, labeled “Tumor cell metastasis” depicts cancer cells spreading to a new location in the body, making it more harmful. Made in www.biorender.com.
Therapeutic Implications
As shown in several key studies8,29,30,31, NK cell-based therapies are emerging as promising cancer treatments, offering potential advantages over traditional immunotherapies because of their potent cytotoxic capabilities and inherent safety profile. CAR-NK cells (NK cells with Chimeric Antigen Receptors) can be used effectively, as clinical trials have shown fewer adverse side effects compared to CAR T-cell therapy. The CAR enables NK cells to recognize tumor-specific antigens directly, activating NK cell killing through perforin/granzyme release and cytokine secretion, enhancing tumor clearance. Unlike CAR T-cell therapy, CAR-NK therapy is associated with lower rates of side effects like cytokine release syndrome and neurotoxicity, making it a safer option. Recent strategies also focus on enhancing NK cell function directly through genetic engineering to improve tumor recognition and resistance to suppression. Furthermore, optimizing NK cell therapies involves combining them with antibodies or other immunostimulatory agents to activate dendritic and T cells, thereby fostering lasting immune protection.
Cytokine-Based Therapies and Checkpoint Blockade in NK Cell Immunotherapy
NK cell immunotherapy also leverages distinct strategies to enhance NK cell activity, notably through cytokine-based therapies and immune checkpoint blockade.Cytokine therapy aims to enhance NK cell proliferation, activation, and cytotoxic function by restoring activation signals and growth that may have been impaired in cancer patients. Key cytokines include IL-2, IL-12, IL-15, and IL-2, which act through various mechanisms: IL-2 promotes NK cell survival and proliferation, IL-12 stimulates IFN-γ production, IL-15 is important for NK cell development and cytotoxic activity, and IL-21 enhances granzyme and perforin expression, boosting tumor killing. Among these potential therapies, IL-2 and IL-15 have advanced into early phase clinical trials, while IL-12 and IL-21 remain primarily in pre-clinical stages14,30.
Immune checkpoint blockade, effective for T cell activation in cancer, is now being extended to target NK cell specific inhibitory pathways that dampen NK cytotoxicity. Blocking these pathways releases NK cells from inhibitory signals, restoring their ability to kill tumor cells. Some targets include PD-1/PD-L1, a pathway that suppresses T and NK cell activity; KIR blockade, which dampens NK cytotoxicity; and NKG2A, which limits NK cell activation. Other inhibitory receptors include TIM-3, TIGIT, and CD96, which contribute to immune evasion and NK cell exhaustion. While most of these approaches remain in pre-clinical trials, some anti–PD-1/PD-L1 and anti-NKG2A antibody trials have advanced and are being further evaluated for specific benefits related to NK cells31.
Methods
This study analyzed publicly available datasets to evaluate the relationship between NK cell activity, cytokine and chemokine presence, and tumor progression across multiple cancer types. A structured literature search was conducted on the National Center for Biotechnology Information (NCBI) database and the Web of Science to identify studies examining NK cells. Additionally, online academic journals (Journal of Immunology and PubMed) and the Human Cell Atlas provided additional data on NK cell ratios and cytokines. To ensure a collection of relevant material, searches were conducted using keywords such as “NK cell,” “cell cycle”, “progression,” “cytokines,” and the names of specific cancer types, including: breast, colorectal, lung, and ovarian. These keywords were chosen to focus on studies that investigated the role of natural killer (NK) cells in cancer progression, and their involvement in cell cycle regulation and cytokine signaling.
Datasets were evaluated and selected based on several criteria. These datasets were required to focus on at least one of the specific cancer types (breast, colorectal & colon, lung, ovarian, gastric, or renal). Additionally, datasets were only selected if they included information on survival versus death data, as it provided the opportunity to link NK cell presence to patient outcomes. Finally, datasets were prioritized if they contained specific information on NK cells like frequency, activity, and their relationship with cytokine presence. Studies which did not meet these criteria or lacked detail on NK cells were excluded from this paper and further analysis.
Human studies of NK cells were gathered from reputable journals, such as Frontiers in Immunology, focusing on variables which included NK cell abundance, activity level (categorized as high or low), tumor size, progression, patient survival outcomes, and cytokine and chemokine presence. Eligible studies provided quantitative data on NK cells and patients with breast, colorectal, lung, ovarian, gastric, and renal cancers. Key datasets from one study (“Killer cell Ig-like receptors ligand-mismatched, alloreactive natural killer cells lyse primary solid tumors”) included samples from gastric, ovarian, renal carcinoma, and colon cancer patients, reported alloreactive NK cell-mediated lysis of tumor cells and provided effector-to-target (E:T) killing percentages at multiple ratios. Additionally, data extracted from a separate study (“Prognostic significance of tumor infiltrating natural killer cells subset CD57 in patients with squamous cell lung cancer”) included CD57+ NK cells in squamous cell lung cancer that related NK cell abundance to overall survival. Studies of impaired or immature NK subsets in colorectal and triple-negative breast cancer were used (“Impact of mitochondrial dysfunction on the antitumor effects of immune cells” and “Immature natural killer cells promote progression of triple-negative breast cancer”) to report NK receptor expression, cytotoxicity, and its association with survival rates. For each paper, we focused on cancer type, NK cell phenotype and function, cytotoxicity data (E:T ratios where available), cytokine/chemokine measurements, and survival or progression outcomes. Furthermore, using the Human Cell Atlas, information on the role of cytokines and their association with the microenvironment was obtained, providing additional data. Together, these sources strengthened the validity and findings, and confirmed observed patterns across data types, but do not include cross-cancer claims since different conditions were present in each cohort.
Data from the included individual studies were synthesized to evaluate relationships between NK cell activity and tumor progression across cancer types. Within each dataset, NK cell activity was categorized as high or low, and compared to determine how these categories relate to tumor size, disease progression, and patient survival. (High vs. Low NK cell activity was not determined, but was taken from established thresholds within the various studies). Additionally, reported associations between NK cells and the presence of specific cytokines (e.g., IL-15, IFN-γ) were recorded, and examined to assess their correlation with tumor size and progression. The narrative synthesis focused on the number of NK cells, or lack thereof, and the influence on tumor growth across various cancer types. By comparing NK cell activity and cytokine presence across multiple datasets, a consistent pattern was identified regarding the role of NK cells in regulating cancer progression providing further support for determining critical contextual factors to consider in future experiments.
Higher NK Cell Ratios Lead to Increased Tumor Cell Death Across Cancers
Across experimental killing assays of gastric, renal, ovarian, and colorectal carcinomas, the results demonstrate that effector to target (E:T) ratios are a strong determinant of cancer cell lysis32. Generally, high E:T ratios are associated with increased killing and greater NK-cell mediated lysis of tumor targets (See Table 1). At a 40:1 and 20:1 ratio, ligand-mismatched NK cells lysed approximately 80% of gastric cancer cells, with only a slight reduction in cytotoxicity at 10:1. However, ovarian and renal cancer targets showed more pronounced declines in percent lysis as the E:T ratio decreased. Colon cancer cells also showed a decline as the E:T ratio decreased, but presented the lowest rates of lysis across ratios, exhibiting either greater resistance or more immune evasion (See Figure 5). This consistent decrease in cancer cell killing as the E:T ratio is lowered shows that higher numbers of NK cells relative to tumor cells enhance overall cytotoxicity. These patterns indicate that the best treatment outcomes occur when there is a higher percentage of mature and effective NK cells relative to cancer cells.
| Cancer | E:T Ratio | Rate of Lysis |
| Gastric Cancer | 40:1 | 0.8200 |
| Gastric Cancer | 20:1 | 0.8200 |
| Gastric Cancer | 10:1 | 0.8000 |
| Ovarian Cancer | 40:1 | 0.7900 |
| Ovarian Cancer | 20:1 | 0.6500 |
| Ovarian Cancer | 10:1 | 0.4300 |
| Renal Carcinoma | 40:1 | 0.7500 |
| Renal Carcinoma | 20:1 | 0.5700 |
| Renal Carcinoma | 10:1 | 0.5000 |
| Colon Cancer | 40:1 | 0.4000 |
| Colon Cancer | 20:1 | 0.3200 |
| Colon Cancer | 10:1 | 0.2900 |
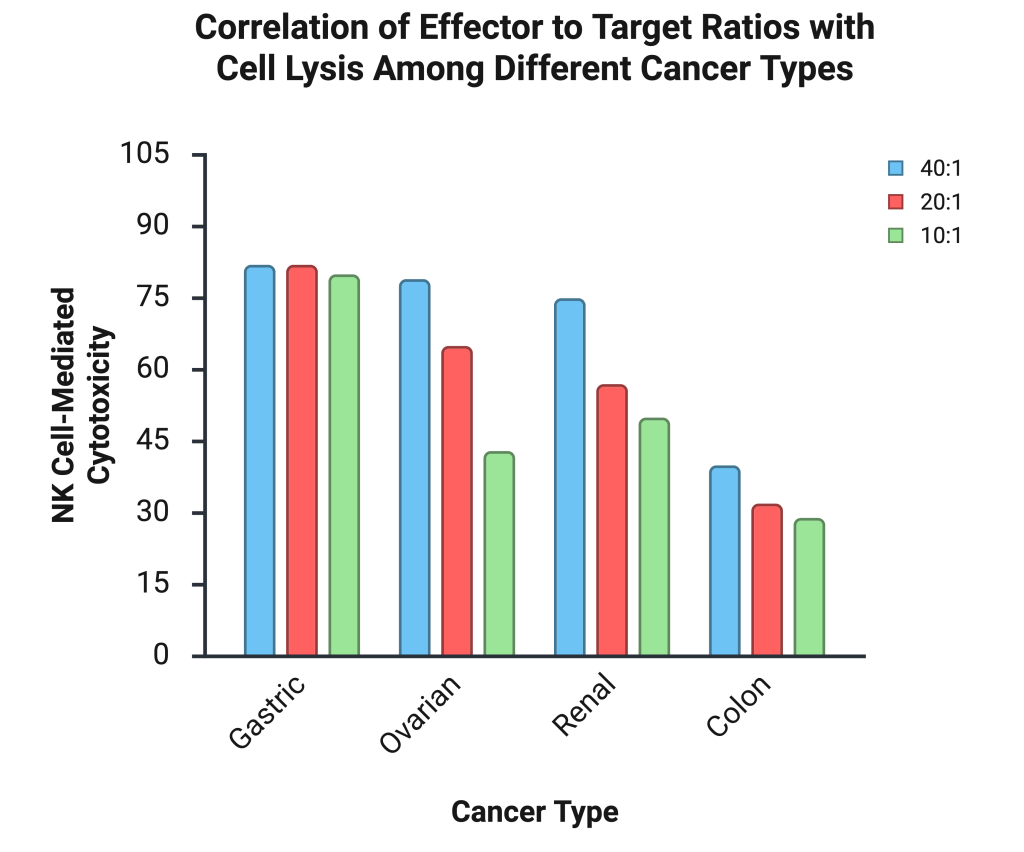
The graph displays the percentage of cell death induced by NK Cells (effector) across four different cancer cell lines: Gastric, Ovarian, Renal, and Colon. The cytotoxic percentage was measured at three different effector: target (E:T) ratios: 40:1, 20:1, and 10:1. This data suggests NK cytotoxicity is most effective against gastric cancer, and less effective against colon cancer. Additionally, NK cell cytotoxicity generally decreases as E:T cell decreases. Made in biorender.com
| Cancer Type | NK Cell Subset | Survival Correlation |
| Lung Cancer16 | CD57 | Patients with >5 TINK (Tumor infiltrating NK) had better survival, risk of death higher with <5 TINK |
| Triple Negative Breast Cancer17 | CD56^bright | Higher levels of CD56^bright NK cells correlate with poor survival |
Mature Subsets of NK Cells Correlate with Higher Survival Rates
Table 2 summarizes findings from separate published clinical studies33,34 that examined associations between specific NK cell subsets and patient survival in different cancers. In this context, ‘survival correlation’ refers to whether higher levels of a given NK subset were associated with better or worse overall survival within each original cohort, as reported by the original authors. In lung cancer, the referenced study found that patients with higher levels of mature, cytotoxic CD57+ NK cells had significantly better survival, echoing findings from other trials that emphasize the importance of NK cell phenotype. By contrast, in triple-negative breast cancer, although NK cell infiltration is increased, most cells are CD56^bright, an immature, cytokine-secreting subset with low granzyme and perforin expression, which contributes to decreased immune regulation and less direct tumor lysis. In colorectal cancer, a different dynamic appeared: although mature NK cells were present, their effectiveness was dulled by metabolic dysfunctions, most notably mitochondrial impairment leading to reduced granzyme levels. These observations illustrate that survival is not just tied to NK cell presence, but to their maturity and cytotoxicity within the specific tumor tissue as well.
Cytokines and Chemokines Across Cancers/Stages
Across the cancers included in this review, individual studies (e.g. References 31 and 34) report a recurring pattern in which higher levels of NK-supportive cytokines in early stage tumors, such as IL-12, IL-18, IL-24, IFN-γ, are associated with enhanced cytotoxicity or better outcomes in their respective cohorts. In breast cancer cohorts, higher IL-15 and IFN-γ expression correlated with signatures of NK activation and more favorable outcomes, whereas enrichment of IL-10 driven pathways coincided with reduced NK-cell cytotoxic signatures and poorer outcomes in those cohorts. Single-cell resources, such as the Human Cell Atlas (Figure 6), are used to illustrate populations which can produce these cytokines, measured by RNA sequencing, and show how changes to their environment can potentially cause lesser outcomes22,35. As shown from the data in Figure 6, as cancers progressed or adapted to pressure, the landscape of cytokines began to change, shifting towards a weaker immune system. Some signs of this include: rising TGF-β and IL-10 concentrations, more attraction of immature subsets, and inhibition of NK cell growth and cytotoxicity. The transition from working to suppressive cytokines and chemokines was especially present in advanced breast cancer and late stage colorectal tumors, where prolonged exposure led to impaired growth, function, and an immature subset of NK cells. These altered phenotypes further affected the environment by releasing immunomodulatory cytokines, thus promoting tumor evasion and further immunosuppression. These conclusions are based on published clinical and translational studies summarized in Figure 6 and in the previously mentioned studies, which report cytokine concentrations with NK cell infiltration in defined patient cohorts.
IL-10 Expression and Immune Cell Diversity
Figure 6 illustrates how IL-10 expression is distributed across major immune and stromal cell populations in a representative single-cell RNA-sequencing dataset from the Human Cell Atlas. After downloading processed single-cell data and selecting malignant and non-malignant cells from solid tumor samples, data were then projected into a two-dimensional UMAP space using the cell type labels provided. Figure 6B below (left UMAP) shows the quantitative expression of IL-10 across different cell clusters, highlighting the key role of IL-10 as an immunosuppressive cytokine. IL-10 suppresses NK cell function and affects the tumor microenvironment by depending on immune response in many cancer types, not just renal cell carcinoma. Figure 6C (right UMAP) shows the diversity of immune and stromal cells within the tumor, illustrating the complexity of the microenvironment and emphasizing the importance of understanding which cells produce or respond to cytokines for interpreting NK cell activity.
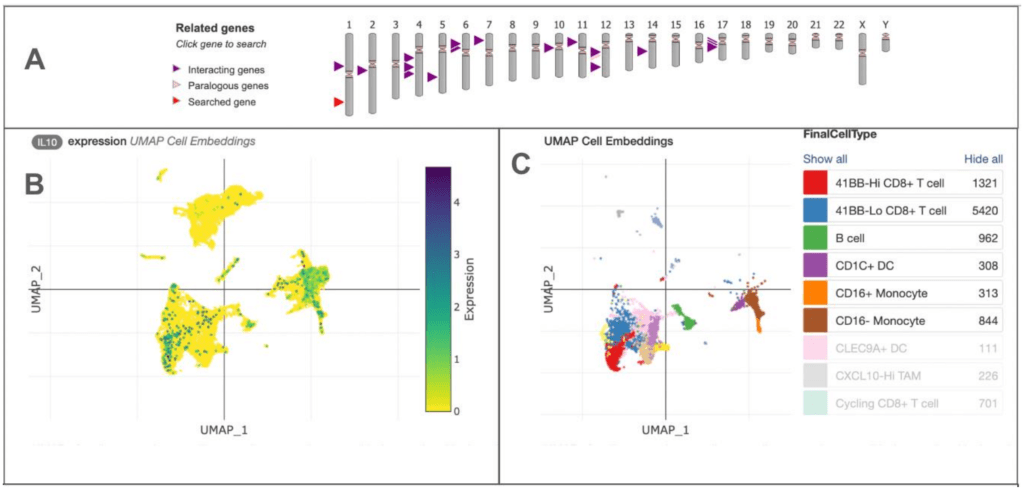
Granular cell types and states were discerned through iterative reprojection and unsupervised clustering of lymphoid myeloid and tumor compartments and merged into broader cell type categories for visualization of expression of genes of interest (6A). Quantitative UMAP plot showing expression levels of IL-10 across different cell clusters within the tumor microenvironment. Each point represents an individual cell, colored with the according magnitude of expression. High IL-10 levels mark higher immunosuppressive areas that can dampen NK cell activity and cytotoxicity as illustrated in 6B. UMAP plot illustrates the diversity of immune and stromal cell types present in the tumor microenvironment. Each color corresponds to a different cell type (CD8+T cells, B cells, dendritic cells, and various monocyte populations) as shown in the chart. This highlights the complexity in cytokine dynamics and their impact on NK cell states and tumor evasion (6C).
A Balance of ROS in the Tumor Microenvironment increases NK Cell Function
In experiments with analyzed tumor samples and model systems, ROS levels were found to be increased, particularly in areas of rapid growth and inflammation. In normal conditions, this ROS activity enhanced NK cell activation and supported the release of effector molecules, boosting anti-tumor responses. However, when ROS accumulation passed a certain threshold, NK cells became susceptible to damage. This led to the downregulation of activating receptors (e.g. NKG2D), lowered cytokine production, and ultimately apoptosis of NK cells themselves. In tumors with ROS suppression, both T cell and NK cell function were impaired, highlighting the importance of a finely tuned environment and state. In another example, in advanced colorectal and lung tumors, excessive ROS was associated with a decline in NK cell numbers and function, further supporting that fluctuating levels of ROS may lead to disruption in the immune system and tumor escape. Thus, the balance and levels of ROS within the tumor microenvironments not only dictate NK cell fate, but are also important factors in the overall immune pressure exerted on cancer progression.
Discussion
These findings reinforce that the function and makeup of NK cells within the tumor environment are important determinants of cancer cell death and survival outcomes. As shown in Figure 5, Table 1, and Table 2, higher E:T ratios of mature NK cells has led to greater tumor cell lysis, supporting their role as immune regulators. Their cytotoxic responses mirror the expected survival correlation of higher NK cell infiltration with improved clinical results in active tumors, such as lung and colorectal cancers. This also explains the variability in outcomes when NK cell maturity and other factors shift in microenvironments which are suppressive like breast or melanoma cancers. Additionally, this data demonstrates that high NK cell presence does not necessarily translate to similar outcomes across all tissues; instead, the function and local environment dictate the outcome. The analysis of cytokine profiles further shows the interplay between immune activation and suppression in the tumor microenvironment. Pro-inflammatory cytokines in this context, such as IL-15 and IFN-γ, correlate with robust NK cell function, driving cytotoxicity. By contrast, cytokines with immunosuppressive effects on NK cells, including IL-10, as visualized in the Human Cell Atlas figure, can dampen NK effector functions and promote exhaustion or conversion toward less cytotoxic phenotypes. This highlights the critical role of cytokines in shaping NK cell phenotype and effectiveness. (Note: The Human Cell Atlas was used to map pathways and regional expression patterns aggregated from current understanding of immune signaling). Similarly, the role of ROS as a regulator and switch for immune activation, capable of both activating and inhibiting NK cell function depending on concentration and duration, further supports findings indicating that a balanced microenvironment may be a target for restoring NK cell activity in tumors. Together, these results, when compared and evaluated using both literature review and aggregated atlas sources, suggest that the balance between cytokine function and control of ROS production is a complex biological process dependent on experimental conditions and biological differences across cells, tissues, and organs. Thus, consideration of how the modulation of these factors impacts signaling networks is essential to developing effective cytokine-based therapies.
Goals and Limitations
Reflecting on the research objectives, the findings support the goals mentioned at the start of the study: to evaluate how NK cell phenotype, maturity, and tumor microenvironment conditions impact tumor progression and patient outcomes. The study achieved its main objectives by demonstrating that NK cell maturity, abundance, and the surrounding environment are critical factors in controlling progression across cancers. There were also several limitations to this study. The data analysis relied on publicly available datasets, which may not represent all patient populations or capture all variances in NK cell behavior. Moreover, variations in how NK cell activity or cytokine levels were measured across datasets may introduce potential bias. Additionally, the evidence synthesized in this paper is drawn from separate studies conducted in different cancer types and cohorts, therefore findings should be interpreted within each disease context rather than direct comparisons across cancers. Limited access to data and rare cancer subtypes also restricted the scope of this study, and these conclusions providing additional opportunities to investigate the role of NK cells and associated cytokines in these cases.
Ultimately, these insights point towards the need to develop more refined therapeutic strategies. Further research should focus on not only methods to increase NK cell numbers, but also means to promote maturation, persistence, and resistance to suppressive areas of the cancer microenvironment. These results suggest that cytokine-based therapies, immune checkpoint blockade, and genetically engineered NK cell products appear to be promising tools for enhancing NK cell activity and tumor control, especially in varied microenvironments. As our understanding of NK cells deepens, future therapies may move beyond cell counts and focus on empowering the most effective immune cells, unlocking new possibilities for cancer research.
Conclusion
In summary, this research paper investigates how the tumor microenvironment, across Lung, Breast, Ovarian, Gastric, Renal, Colon & Colorectal cancers are correlated with NK cell presence, abundance, phenotype, and function within but not across cancer types. This paper evaluated the current literature on the roles of cytokines, chemokines, and ROS dynamics across cancers in relation to NK cell features as described. Using quantitative analyses of functional assays and patient data, this study demonstrates that survival outcomes in addition to tumor cell death are closely linked to the higher abundance and maturity of NK cells, with higher effector to target ratios and mature phenotypes correlating with improved outcomes. This paper highlights the functional role of cytokines, in particular how immunosuppressive cytokines such as IL-10 and TGF-β, as well as elevated levels of ROS impair NK cell cytotoxicity. Analysis from this review provides novel insight into the dynamics of NK cells and its relationship with the tumor microenvironment, providing potential temporal-spatial frameworks necessary to evaluate fundamental immune cell signaling processes to develop targeted immunotherapies appropriate across tissues and disease progression to improve patient survival outcomes.
Acknowledgements
Thank you to Jacqueline Howells for her support and mentorship during my process of writing this paper. I would also like to thank Luke Fisher and Alexandra Massa for their edits to this paper. Lastly, thank you to my family for their support.
References
- L. Coënon, M. Geindreau, F. Ghiringhelli, M. Villalba, M. Bruchard. Natural Killer cells at the frontline in the fight against cancer. Cell Death & Disease 15, 1-14 (2024). [↩] [↩] [↩]
- L. Coënon, M. Geindreau, F. Ghiringhelli, M. Villalba, M. Bruchard. Natural Killer cells at the frontline in the fight against cancer. Cell Death & Disease 15, 1-14 (2024). [↩]
- S. Paul, G. Lal. The Molecular Mechanism of Natural Killer Cells Function and Its Importance in Cancer Immunotherapy. Frontiers Immunology 8, 1-15 (2017). [↩] [↩]
- D. Pisetsky. Cell death in the pathogenesis of immune-mediated diseases: the role of HMGB1 and DAMP-PAMP complexes. Swiss Med Wkly 141 (2011). [↩]
- E. Vénéreau, C. Ceriotti, M.E. Bianchi. DAMPs from cell death to new life. Frontiers in immunology 6, 422 (2015). [↩]
- D. Bertheloot, E. Latz, B.S. Franklin. Necroptosis, pyroptosis and apoptosis: an intricate game of cell death. Cell Mol Immunol 18, 1106–1121 (2021). [↩]
- I. Prager, C. Watzl. Mechanisms of natural killer cell-mediated cellular cytotoxicity. Journal of leukocyte biology 105, 1319-1329 (2019). [↩]
- T. Bald, M.F. Krummel, M.J. Smyth, K.C. Barry. The NK cell-cancer cycle: advances and new challenges in NK cell-based immunotherapies. Nature Immunology 21, 835-847 (2020). [↩] [↩]
- S. Liu, B. Huang, J. Cao, Y. Wang, H. Xiao, Y. Zhu, H. Zhang. ROS fine-tunes the function and fate of immune cells. International Immunopharmacology 119, 1-14 (2023). [↩]
- R. Shah, B. Ibis, M. Kashyap, VA. Boussiotis. The role of ROS in tumor infiltrating immune cells and cancer immunotherapy. Metabolism 151 (2024). [↩]
- M.J. Tavassolifar, M. Vodjgani, Z. Salehi, M. Izad. The Influence of Reactive Oxygen Species in the Immune System and Pathogenesis of Multiple Sclerosis. Autoimmune Diseases 2020, 1-14 (2020). [↩]
- R.R. Manoharan, A. Prasad, P. Pospíšil, J. Kzhyshkowska. ROS signaling in innate immunity via oxidative protein modifications. Frontiers in Immunology 15, 1-15 (2024). [↩]
- A. Kotsafti, M. Scarpa, I. Castagliuolo. Reactive Oxygen Species and Antitumor Immunity-From Surveillance to Evasion. Cancers 12, 1-16 (2020). [↩]
- Z. Shen, X. Meng, J. Rautela. M. Chopin, N.D. Huntington. Adjusting the scope of natural killer cells in cancer therapy. Cellular & Molecular Immunology 22, 699–711 (2025). [↩] [↩]
- A.V. Kuznetsova, X.A. Glukhova, I.P. Beletsky, A.A. Ivanov. NK cell activity in the tumor microenvironment. Frontiers in Cell and Developmental Biology 13, 1-11 (2025). [↩]
- S. Lopez-Vergès, JM. Milush, S. Pandey, VA. York, J. Arakawa-Hoyt, H. Pircher, PJ. Norris, DF. Nixon, LL. Lanier. CD57 defines a functionally distinct population of mature NK cells in the human CD56dimCD16+ NK-cell subset. Blood, The Journal of the American Society of Hematology 116,3865-3874 (2010). [↩] [↩] [↩]
- G. Thacker, S. Henry, A. Nandi, R. Debnath, S. Singh, A. Nayak, B. Susnik, M.M. Boone, Q. Zhang, S.B. Kesmodel, S. Gumber, G.M. Das, T. Kambayashi, C.O. Dos Santos, R. Chakrabarti. Immature natural killer cells promote progression of triple-negative breast cancer. Science translational medicine 15, 1-15(2023). [↩] [↩]
- H. Zhang, J. Wang, F. Li. Modulation of natural killer cell exhaustion in the lungs: the key components from lung microenvironment and lung tumor microenvironment. Frontiers in immunology 14, 1-10 (2023). [↩]
- Y. Zeng, X. Lv, J. Du. Natural killer cell‑based immunotherapy for lung cancer: Challenges and perspectives (Review). Oncology Reports 46,1-14 (2021). [↩]
- F. Li, C. Gao, Y. Huang, Y. Qiao, H. Xu, S. Liu, H. Wu. Unraveling the breast cancer tumor microenvironment: crucial factors influencing natural killer cell function and therapeutic strategies. International Journal of Biological Sciences 21, 2606-2628 (2025). [↩]
- A. Coppola, R. Arriga, D. Lauro, MI. Del Principe, F. Buccisano, L. Maurilio, P. Palomba, A. Venditti, G. Sconocchia. NK Cell Inflammation in the Clinical Outcome of Colorectal Carcinoma. Frontiers in Medicine 2, 1-6 (2015). [↩]
- H. Jia, H. Yang, H. Xiong, K.Q. Luo. NK cell exhaustion in the tumor microenvironment. Frontiers in immunology 14, 1-8 (2023). [↩] [↩] [↩]
- Y. Du, Y. Wei. Therapeutic potential of natural killer cells in gastric cancer. Frontiers in immunology 9, 1-12 (2019). [↩]
- C. Chen, Z. Wang, Y. Ding, Y. Qin. Tumor microenvironment-mediated immune evasion in hepatocellular carcinoma. Frontiers in immunology 14, 1-19 (2023). [↩]
- O. Melaiu, V. Lucarini, L. Cifaldi, D. Fruci. Influence of the Tumor Microenvironment on NK Cell Function in Solid Tumors. Frontiers in immunology 10, 1-18 (2020). [↩]
- A. Stojanovic, MP. Correia, A. Cerwenka. Shaping of NK cell responses by the tumor microenvironment. Cancer Microenvironment 6, 135-146 (2013). [↩]
- S. Nersesian, S. Schwartz , S. Grantham, L. MacLean, S. Lee, M. Pugh-Toole, J. Boudreau. NK cell infiltration is associated with improved overall survival in solid cancers: A systematic review and meta-analysis. Translational oncology 14 (2021). [↩]
- S.K. Larsen, Y. Gao, PH. Basse. NK cells in the tumor microenvironment. Critical Reviews in Oncogenesis 19,91-105 (2014). [↩]
- K. Rezvani, R. Rouce, E. Liu, E. Shpall. Engineering Natural Killer Cells for Cancer Immunotherapy. Molecular Therapy 25, 1769-1781 (2017). [↩]
- M. Gergues, R. Bari, S. Koppisetti, A. Gosiewska, L. Kang, RJ. Hariri. Senescence, NK cells, and cancer: navigating the crossroads of aging and disease. Frontiers in Immunology 16, 1-21 (2025). [↩] [↩]
- J.A. Myers, JS. Miller. Exploring the NK cell platform for cancer immunotherapy. Nature Reviews Clinical Oncology 18, 85-100 (2021). [↩] [↩]
- F. Re, C. Staudacher, L. Zamai, V. Vecchio, M. Bregni. Killer cell Ig-like receptors ligand-mismatched, alloreactive natural killer cells lyse primary solid tumors. Cancer 107, 640-648 (2006). [↩] [↩]
- S. Lopez-Vergès, JM. Milush, S. Pandey, VA. York, J. Arakawa-Hoyt, H. Pircher, PJ. Norris, DF. Nixon, LL. Lanier. CD57 defines a functionally distinct population of mature NK cells in the human CD56dimCD16+ NK-cell subset. Blood, The Journal of the American Society of Hematology 116, 3865-3874 (2010). [↩]
- G. Thacker, S. Henry, A. Nandi, R. Debnath, S. Singh, A. Nayak, B. Susnik, M.M. Boone, Q. Zhang, S.B. Kesmodel, S. Gumber, G.M. Das, T. Kambayashi, C.O. Dos Santos, R. Chakrabarti. Immature natural killer cells promote progression of triple-negative breast cancer. Science translational medicine 15, 1-15 (2023). [↩]
- I. Terrén, A. Orrantia, J. Vitalle, O. Zenarruzabeitia, F. Borrego. NK cell metabolism and tumor microenvironment. Frontiers in immunology 10, 1-6 (2019). [↩]