Abstract
Gliomas are one of the most common brain tumors, affecting around 20,000 individuals. Specifically, they grow in the brain or spinal cord, classifying them as central nervous system (CNS) tumors. Fortunately, a new medical innovation called CAR T-cell therapy bears the potential for selectively targeting these tumor cells, improving prognosis. This study ultimately establishes the current status and progress of CAR T-cell therapy to assess its viability as a cancer treatment option. To qualify for inclusion in this literature review, papers had to be peer-reviewed and discuss mechanisms of CAR T-cell therapy or its applications in clinical trials. After compiling this data, we identify broad trends with CAR T-cell therapy as well as general patient responses across different institutions. Due to initial clinical trial successes, we ultimately conclude that CAR T-cell therapy is a promising potential treatment strategy, with the important caveat that physicians must carefully monitor patients receiving CAR T-cell therapy for potential toxic side effects. However, given that standard medications and procedures exist to manage these side effects, clinical trials indicate that CAR T-cell therapy bears potential as a complement to current glioma treatment methods. In order to further advance the progress that has been made with CAR T-cell therapy, researchers ought to continue pursuing modifications to CAR T-cells that improve specificity of tissue targeting and address later-stage tumors with heterogeneous surface phenotypes.
Keywords: glioma; CAR T; CAR T-cells; brain tumor; CAR T-cell therapy
Introduction
Each year, hundreds of thousands of patients suffer from aggressive gliomas means that proliferate quickly from their loci of origin in the brain and spinal cord1’2. CAR T-cell therapy is a strong candidate treatment for both efficient and highly specific immune attack against tumors3. As our medical capabilities to characterize cancer cells become more advanced, physicians are focusing on strategies like CAR T-cell therapy that enhance the specificity of cancer therapeutics in targeting malignant cells while sparing healthy tissue. This specificity is especially critical for gliomas given that they arise within populations of glia, cells that provide support for neurons and are thus critical to preserve as much of as possible4. Symptoms of gliomas include seizures, nausea, fatigue, and cognitive problems5.
Defining CAR T-cell therapy
Simply put, CAR T-cells are immune cells from a patient’s body that have been modified to specifically target tumors6. Essentially, researchers take advantage of the ability of T-cells to identify harmful, pathogenic entities in the body by their surface protein markers.
In order to engineer CAR T-cells, scientists extract a patient’s T-cells from their blood and insert artificially engineered genes into the T-cell DNA. These implanted genes encode for cell-surface receptors capable of binding to and recognizing proteins commonly expressed on the face of cancer cells7. These newly added cell-surface receptors are referred to as chimeric antigen receptors or CARs. Different varieties of CARs can be leveraged to target a vast array of unique, heterogeneous surface protein markers on different types of cancer cells. Given that researchers can modify T-cells to express receptors for any antigen that appears on cancer cell surfaces, CAR T-cell therapy represents a new avenue towards specializing therapeutics for each patient’s unique cancer profile3.
Objective of this Review
This paper ultimately aims to assess the viability of CAR T-cell therapy by accounting for its unique advantages and shortcomings in treating gliomas. Despite current treatments, such as surgical removal and chemotherapy, poor prognosis is common amongst patients8. Emerging developments in CAR T-cell therapy have provided a potentially improved outlook for patients suffering from gliomas, making this research extremely pertinent to the current medical landscape. Additionally, relatively few studies have explored CAR T-cell therapy for gliomas, and current trials are predominantly early-phase or have small sample sizes9’10’11’12. Also, there is currently a lack of agreement in the medical community about the optimal antigen for CAR T-cells to target. While the targets IL-13Rα2, EGFRvIII, and HER2 have been investigated prominently, clinical results have been largely inconsistent or remain in early phases (Table 1)13’12. This question of identifying candidate target antigens for CAR T-cells to recognize is also complicated by the fact that surface protein expression on tumor cells can be heterogeneous between different patients and even within a single patient’s tumor.
An initial review of the existing literature was conducted using academic databases to explore current research on CAR T-cell therapy in glioma treatment. Peer-reviewed studies that offered insights into clinical applications and biological mechanisms of CAR T-cell therapy were prioritized. This search provided a preliminary understanding of CAR T-cell therapy in glioma treatment while highlighting gaps for further research.
A summary of the key features of clinical trials discussed in this review can be found in the table below. Although preclinical data also plays a role in our analysis of the costs and benefits of CAR T-cells, below we only focus on data taken with human subjects. Furthermore, although this review article assesses the potential of CAR T-cell therapy for glioma treatment, trials investigating CAR T-cell therapy for hematological cancers are included in the table below because toxicities reported in such trials are significant considerations for physicians applying similar procedures for glioma therapy.
| Trial Ref. # | Tumor Type and Sample Size | Design | Delivery Method | Toxicities | Initial Response | Survival Time |
| [8] | Recurrent high-grade glioma (n=65) | IL-13Rɑ2 targeted | Locoregional Infusion | 2 cases of grade 3+ toxicities: one case of ataxia and one case of cognitive decline | Halted tumor growth in 29/58 patients | Median survival time of 7.7 months* |
| [12] | Multiple myeloma (n=50) | Bispecific CAR T-cells engineered to recognize BCMA and CD19 | Infusion | Cytokine release syndrome, neurotoxic side effects, neutrophil level decline, low white blood cell levels, anemia, low platelet levels | 46/50 subjects exhibited initial tumor shrinkage | Median overall survival of 19.7 months |
| [13] | GBM (n=18) | Bivalent CAR T-cells engineered to recognize IL-13Rα2 and EGFR | Intracerebro-ventricular Infusion | Neurotoxic side effects | 8/13 patients with MRI visible tumors exhibited tumor shrinkage | Median tumor growth free survival of 1.9 months. Median overall survival not reached by end of study. |
| [14] | GBM (n=10) | EGFRvIII directed CAR T-cells | Intravenous Infusion | None reported | Decline of EGFRvIII tumor cells in 5/7 subjects that received surgery and CAR T-cell therapy | Not reported. One patient with halted tumor growth after 18 months. |
| [9] | Recurrent GBM (n=3) | CARv3- TEAM-E T-cells utilizing bispecific antibodies | Intrathecal Infusion | Transient cognitive side effects and fevers | Significant tumor shrinkage in all 3 patients | Not reported. Tumor returned in all patients. |
| [15] | Leukemia (n=45) | CD19 CAR T-cells transduced with lentiviral vector | Infusion | Neurotoxic side effects or cytokine release syndrome in 23% of patients | 93% initial remission rate. 7 cases of later CD19 tumor growth. 11 cases of later CD19– tumor growth. | Not reported |
| [16] | Leukemia (n=30) | CD19 CAR T-cells transduced with lentiviral vector | Infusion | 100% rate of cytokine release syndrome | Complete remission in 27 (90%) of subjects one month after treatment | 78% survival rate after 7 months |
| [17] | Leukemia (n=53) | CD19 CAR T-cells | Infusion | Severe cytokine release syndrome in 14/53 subjects and neurotoxic side effects | 83% of subjects exhibited initial remission | Median overall survival time of 12.9 months |
| [18] | Large B-cell Lymphoma (n=111) | CD19 CAR T-cells | Intravenous Infusion | Neutrophil level declines, anemia, low platelet levels, neurotoxic side effects, cytokine release syndrome | 42% of subjects continued to respond to therapy after 15.4 months. | 52% survival rate after 18 months |
| [19] | DIPG subtype of glioma (n=21) | B7-H3 targeting CAR T-cells | Intracerebro-ventricular Infusion | None reported that were due to CAR T-cell therapy | 1 patient exhibited some tumor shrinkage. 15 patients exhibited halted tumor growth. 2 patients exhibited continued tumor growth. | Median survival time of 10.7 months |
| [20] | GBM (n=7) | EGFRvIII targeted CAR T-cells | Peripheral Infusion | None reported | Initial median tumor growth free survival of 5.2 months | Median overall survival of 11.8 months |
| [21] | DIPG or spinal Diffuse Midline Glioma (n=13) | GD2-CAR T-cells transduced with retroviral vector | Intravenous and Intracranial Infusions | Cytokine release syndrome, neurotoxic swelling, ICANS | Initial tumor growth stagnation as seen on MRI | Median overall survival of 17.6 months for DIPG patients |
*This study report included median survival time for the cohort of patients specifically diagnosed with rGBM, a specific type of high-grade glioma. This cohort included the majority of the 65 patients.
Methods
The PubMed and Google Scholar databases were searched using combinations of the following keywords: CAR T-cell therapy, CAR T, CAR T-cells, brain tumor, and glioma. Peer-reviewed studies written in English that specifically examined the impact of CAR T-cell therapy on gliomas were included. Articles that explored the biological mechanisms of CAR T-cell therapy, its usage in ongoing clinical trials, or alternative glioma treatments were also considered.
For data extraction, we used Google Scholar to search for articles regarding CAR T-cell therapy published since 2020. We searched specifically for clinical trials utilizing CAR T-cell therapy in order to collect information about the most recent advancements in this treatment’s application to human patients. We also explored other review articles published from earlier years to gain an understanding of the broader background surrounding CAR T-cells, as well as more recent studies about the new advancements made in CAR T-cell therapy.
Information was first organized by background information about the topic; for example, studies were read about glioma pathophysiology, current glioma therapies and resistance, clinical trials, mechanisms of CAR T-cells, delivery strategies, and limitations. Summaries were written about articles, along with a short paragraph about important findings and topics that either the researchers did not address or suggested should be explored in the future. This organization helped uncover gaps in the existing literature that would be helpful for future studies to explore while highlighting useful content for inclusion in the paper.
The papers were gathered from reputable journals, such as Nature and Frontiers in Immunology. Databases such as PubMed and ScienceDirect were also searched using the keywords mentioned above.
By gathering and analyzing the data of multiple clinical trials, this paper was able to meet its objectives. Our findings support the importance of personalized immunotherapy and the growing potential of CAR T-cell therapy, although more clinical trials and research are needed to address limitations such as cytokine release, cost, and limited effectiveness in solid tumors.
Evaluating Different CAR T-Cell Delivery Methods
One practical issue that researchers and physicians must address for CAR T-cells to become useful in a real-world therapeutic context is the matter of which delivery method maximizes localization of CAR T-cells at the tumor site. Various methods exist for administering CAR T-cells to subjects with solid tumors, and these methods can generally be divided into two categories: locoregional and systemic delivery14. While locoregional delivery involves administering CAR T-cells directly to the tumor site, systemic delivery results in treatments circulating throughout the body. Some examples of locoregional delivery strategies include intraventricular and intratumoral dosing, both of which administer treatment directly to the brain, the former by administering therapy to a site with higher volumes of cerebrospinal fluid and the latter by placing therapies directly into tumor cells15. Meanwhile, one systemic procedure known as intravenous delivery involves inserting a needle into a patient’s vein such that treatment directly enters the bloodstream and circulates the whole body16.
A common challenge when delivering drugs to the brain is bypassing the blood-brain barrier, a sheet of cells lining neural capillaries that selectively prevents certain compounds from entering the brain17.
Preclinical studies have already supported the benefits of locoregional drug delivery methods over systemic treatment16. A study led by scientists at the City of Hope clinic demonstrated the advantages of locoregional treatment delivery over systemic delivery using mouse models. These researchers implanted cells of brain metastases from breast cancer patients into mice and programmed CAR T-cells expressing receptors for the HER2 antigen, a common cell-surface protein of breast cancer cells. Subsequently, they separated the mice into different groups that were assigned different methods of CAR T-cell delivery. Mice that received a form of locoregional delivery, such as intracerebral or intraventricular delivery, exhibited similar degrees of tumor shrinkage. However, while mice in the intraventricular group achieved full remission, mice that received intravenous treatment demonstrated a lesser extent of tumor shrinkage18. These findings suggest that locoregional CAR T-cell delivery procedures are superior to systemic strategies in promoting localization of CAR T-cells in tumor sites.
Potential of CAR T-cell therapy
Multi-Specific and Logic-Gated CAR T-cell therapy
One notable advancement occurred in a recent study where researchers designed special types of CAR T-cells called bivalent CAR T-cells that have the ability to target glioma cells with high specificity through dual recognition of multiple cell-surface antigens19. In this clinical trial, the bivalent cells were programmed to recognize and attack cells expressing both the IL-13Rɑ2 and EGFR cancerous surface protein markers. Essentially, the main advantage of this technique is that the CAR T-cells bear CARs equipped with multiple antigen-binding domains that can attach to multiple different cancer surface antigens.20. These CAR T-cells were administered intracerebroventricularly, meaning they were injected into the cerebrospinal fluid (CSF) produced in a pocket of the brain called the lateral ventricles. Once successfully placed in the CSF, the CAR T-cells can circulate the brain and broadly attack any detected tumor cells.This study demonstrates the incredible potential of emerging new-generation CAR T-cell therapies – of the thirteen patients whose tumors could be visualized with imaging techniques, eight exhibited tumor shrinkage following CAR T-cell therapy administration. Granted, only one patient continued exhibiting stagnated tumor growth after 16 months, suggesting the need for strategies to improve CAR T-cell durability21.
In addition to dual-targeting, logic-gated constructs in CAR T-cells are emerging. Many CAR T-cell therapies focus on one specific antigen. However, some healthy cells may express a similar antigen, causing the CAR T-cell to attack normal, healthy cells. Due to oncogenic mutations, antigens on cancer cell surfaces can also change, resulting in the CAR T-cells no longer being able to recognize those cells as cancerous. Logic-gated constructs provide a possible solution to this problem. This new design uses the Boolean operators “AND”, “OR,” and “NOT” to target different antigens22. The AND gate is activated only when two different specific antigens are present. One derivation of the AND logic-gate involves the SynNotch, or synthetic Notch system, which is a conditional logical type. In other words, when this mechanism detects one antigen, it will as a result express the receptor for another. The OR gate is activated if either of the two antigens are present, while the NOT gate is activated when one antigen is present, but the other is not23.
Logic-gated constructs in CAR T-cells are still in very early clinical phases24. Therefore, there is no clear evidence that guarantees the success of this new mechanism. Specifically, trials are currently still in phase 1 and focus on the safety and efficacy of these treatments in patients. For instance, a phase 1 trial of EGFRvIII CAR T-cells employing the Synthetic Notch (synNotch) logic-gating method in glioblastoma is underway. With the synNotch method, T-cells recognizing the tumor antigen EGFRvIII triggers the transcription of CARs that bind IL13Rɑ2, a second target antigen This particular strategy exemplifies the advantages of AND-gating, with the recognition of EGFRvIII, and subsequently IL13Rɑ2, being the two necessary conditions for the initiation of a CAR T-cell attack against a cell25. Results of this trial have not been published yet, but it is important to remember that glioblastoma’s aggressiveness and complexity make it a difficult cancer to control and treat effectively.
Armored CARs
Another advancement in CAR T therapy is armoured CAR T-cells. Unlike regular CAR T-cells, these cells are modified to secrete molecules, like cytokines, that improve treatment potency. One type of inflammatory cytokine that some armored CAR T-cells release is IL-1226. Preclinical data done on mice show that this cytokine can contribute to cancer cell death27. However, possible toxic side effects following cytokine administration–including advanced fatigue, difficulty respirating, and pain induced by swelling–remain a concern28.
A newer approach combines both armoured CARs and a strategy called dual-targeting, which involves targeting more than one antigen29. There is not enough detailed human clinical trial data for this new mechanism; however, animal trials on mice have been conducted. One trial included developing CAR T-cells that targeted two glioblastoma stem cell antigens: CD44 and CD133. The researchers then engineered the cells with a IL-7 receptor alpha intracellular domain, which is the “armouring” part. When these CAR T-cells were delivered locoregionally, the results showed improved tumor control. Researchers are currently investigating other candidate cytokines for armoured CAR T-cell secretion as well, including IL-15.30. Again, although these results sound promising, more clinical data and evidence on humans is needed to validate this particular treatment.
Allogeneic CAR T-Cells
Allogeneic CAR T-cells are T-cells donated from a healthy individual to patients. Healthy T-cells can be produced in advance, improving treatment efficiency, which is crucial for improving patient outcomes31. Despite this advantage, allogeneic CAR T-cells have challenges of their own. For instance, a patient’s immune system may reject the donor cells, leading to graft versus host disease, or GVHD32. This occurs when the donor T-cells attack healthy cells in the patient’s body, leading to life-threatening conditions. Currently, researchers are exploring gene-editing tools, like CRISPR, to improve immune compatibility33.
Challenges and Toxicities of CAR T-Cells
Although CAR T-cell therapy has made significant progress and is already being applied in clinical trials, there are several challenges that must be overcome before they can be broadly prescribed in a clinical setting. We will proceed to highlight some of the most prominent issues that must be addressed in CAR T-cell therapy, as well as outline some potential solutions as future directions for research.
Antigen Loss
The ability of cancer cells to rapidly evolve through a process that mimics natural selection in the tumor microenvironment serves as an obstacle to treatments like CAR T-cell therapy that heavily rely on recognition of a certain aspect of a cancer cell’s phenotype34. As CAR T-cells target and eradicate cancer cells with a specific antigen, tumor cells lacking alleles encoding for that antigen will survive and proliferate. This microevolution can result in a phenomenon known as antigen loss, in which the proportion of tumor cells expressing the target antigen decreases such that the tumor has evolved immunity and invisibility against the CAR T-cells35‘36.
Antigen loss has been reported in several CAR T-cell therapy clinical trials, including one documented by O’Rourke et al. in which peripherally administered CAR T-cells targeting the EGFRvIII antigen eventually induced varying degrees of antigen loss in 5 out of 7 available subjects with glioblastoma, a particularly aggressive grade of glioma. Although researchers did observe successful accumulation of CAR T-cells around the tumor site, the rise of resistant cancer cells suggests that tumor phenotype heterogeneity–even within a single patient–remains a challenge for CAR T-cell therapy in later stages of treatment37.
To overcome such challenges, physicians conducting another phase 1 clinical trial at Mass General Hospital engineered and intraventricularly delivered CARv3-TEAM-E T cells capable of binding multiple variations of the EGFR antigen on glioblastoma cell surfaces. This team of researchers specifically chose to target variations of the EGFR antigen because of its consistency as a marker in mutated forms on tumor cells. Although the group achieved initial success in tumor shrinkage, they then observed a resurgence of tumor cells in two subjects10. However, the initial treatment success suggests that engineering CAR T-cells capable of targeting common variants of a single cell-surface protein presents a promising solution to issues of tumor heterogeneity.
Off-Target Cytotoxicity
One main concern with the injection of CAR T-cells is that they may exhibit off-target cytotoxicity and attack normal, healthy cells in addition to glioma cells38. This could occur if the CAR T-cells are engineered to recognize antigens that also happen to be expressed on the surface of healthy cells as well as cancer cells. Unfortunately, this proves a rather difficult problem to circumvent because many of the cancer cells that survive in the tumor microenvironment have evolved to express a cell-surface protein profile similar to that of healthy cells. This phenomenon occurs so that cancer cells can closely resemble normal cells and avoid being recognized as foreign by the immune system.
Risks of Viral Delivery Systems
Another risk when introducing foreign or genetically modified cells into a patient’s body is that those cells may fail to acclimate to their new environment and could potentially develop into secondary tumors. According to the CDC in 2023, twenty-two reports of T-cell malignancies related to CAR T-cell therapy for leukemia and lymphoma were placed under investigation to assess the possibility that retroviral vectors introduced into T-cells resulted in these malignancies.39. Then, as of 2024, the FDA issued a warning regarding the risk of tumors developing from infused CAR T-cells40. As Yin and Wei noted, “While retroviral vectors offer durable CAR T efficacy, they pose risks like replication-competent retroviruses (RCRs) and insertional mutagenesis, potentially activating oncogenes or causing secondary malignancies”41 Specifically, lentiviral insertion within certain T-cell homeostasis genes could theoretically cause T-cell lymphomas after CAR T-cell therapy42. However, it is important to note that there is no clear evidence or clinical trial data that proves this; this risk is currently preliminary and speculative, but it is still an important risk to take into account. To this date, no published glioma CAR T-cell human trial has confirmed cases of T-cell malignancy yet.
Cytokine Release Syndrome (CRS)
Following the injection of CAR T-cells–modified immune cells–into a patient’s body, patients can exhibit various forms of immune system dysregulation. As a result, patients can exhibit various forms of immune system dysregulation. One observed form of such immune dysfunction is cytokine release syndrome (CRS). With CRS, patients exhibit a spike of inflammation after CAR T-cells are injected into the body and multiply rapidly, releasing inflammatory chemicals called cytokines43.
This response can endanger patients’ lives because blood vessels become dilated and more permeable in response to inflammation, leading to dangerous drops in blood pressure and potentially life-threatening shock43.
Although treatment varies based on the causes of each patient’s CRS, standard management includes medication to target specific cytokines, including anakinra and siltuximab. Corticosteroids can also be administered to reduce inflammation caused by the cytokines. Ventilation and medication for low blood pressure may also be incorporated into treatment44.
Another standard procedure following the onset of CRS is the administration of tocilizumab, a protein that blocks receptors for interleukins, signals that initiate an inflammation response44.
However, one important consideration to note is that although some degree of CRS can occur frequently, the number of cases that develop into severe inflammation cases is much lower. For example, among a series of four clinical trials involving CAR T-cell therapy, CRS occurred in >85% of patients, but severe CRS only occurred in 13-27% of patients45’46’47’48. Granted, these clinical trials treated subjects with leukemia and lymphoma, rather than glioma, but similar concerns exist for glioma patients given that such inflammatory signaling could occur with CAR T-cells targeting brain cancer.
Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS)
An additional toxicity that has been documented following the administration of CAR T-cell therapy is Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS). It is hypothesized that this condition induced blood-brain barrier dysfunction through the excessive release of inflammatory signals that promote the opening of capillaries to allow for movement of larger compounds in and out of blood vessels49.
However, once a patient develops ICANS, multiple medications can be prescribed to manage each of the array of symptoms, which include cognitive decline, seizures, and severe neural inflammation. Management of worsening cases of ICANS typically involves infusions of corticosteroids and methylprednisolone50. However, ambiguity exists regarding the optimal dosages and timings to administer these medications for patients afflicted with ICANS51. Additionally, when seizures associated with ICANS occur, physicians commonly prescribe benzodiazepines to manage electrical misfiring52.
Tumor Lysis Syndrome (TLS)
Tumor lysis syndrome, or TLS, is a life-threatening medical condition that is caused by the rapid death of swaths of cancer cells whose contents then enter and accumulate in the bloodstream. CAR T-cell therapy can directly cause TLS in patients and has been linked to abnormally high levels of serum creatinine as well as severe cytokine release syndrome (CRS). However, TLS can be managed using standard guidelines applied with chemotherapy treatments along with frequent hydration and the use of uric acid-lowering substances53.
Reduction of Blood Cell Count
Cytopenia, an abnormally low number of one or more types of blood cells, occurs often following CAR T-cell therapy54. Factors such as gender, disease, age, previous therapy, and the CAR’s target can affect cytopenia after CAR T-cell therapy. Cytopenia can increase risk of severe infections and bleeding in patients undergoing CAR T-cell therapy54’55.
Concluding Thoughts on Challenges and Toxicities of CAR T-cell therapy
Current research on CAR T-cell therapy demonstrates that its beneficial effects may be short-lived in certain cases. For example, Table 1 includes some clinical trials in which cases showed initial tumor regression followed by sudden tumor progression. When prescribing cancer treatments, physicians must consider the possibility that the treatment will prove more destructive than the illness itself. Thus, going forward, a primary focus of clinical trials investigating CAR T-cell therapy includes determining treatment dosages patients can tolerate. A phase 1 clinical trial conducted earlier this year, for example, focused on titrating doses and confirming the safety of B7-H3-targeting CAR T-cells for treating pediatric diffuse intrinsic pontine glioma (DIPG). This particular study established acceptable doses of B7-H3-targeting CAR T-cells, work that will surely be replicated by future studies as various brands and types of CAR T-cell therapies emerge56.
Addressing Possible Toxicities of CAR T-cell therapy
Medications for Inflammation
Researchers ought to be cautious of potentially dangerous levels of inflammation that could arise from the introduction of CAR T-cells. Neurotoxic levels of inflammation have been reported in past clinical trials with glioma patients, with 2 out of 65 patients in an aforementioned phase 1 clinical trial exhibiting neural edema–an inflammation caused by buildup of liquids–following CAR T-cell therapy57. While these side effects can be managed with medication, alternative anti-inflammatory medications should be considered in emergency cases57.
Additionally, should cytokine release syndrome occur, physicians can treat the afflicted patients with proteins–namely, antibodies–that block binding of interleukins, a type of inflammatory cytokine, to their receptors. Such measures reduce the inflammatory response and have been effective in treating life-threatening symptoms of CRS58.
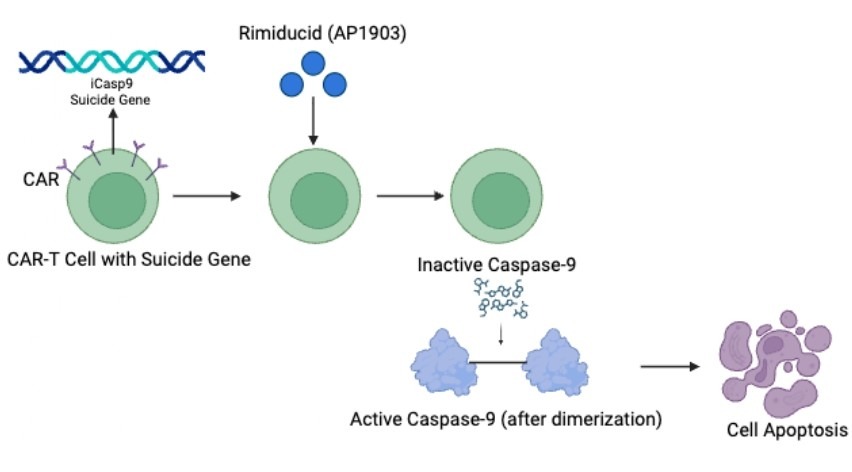
Suicide Genes in CAR T Cells
The introduction of engineered “suicide genes” into CAR T-cells allows for researchers to introduce drugs into the body that kill CAR T-cells should a patient exhibit unexpected, dangerous side effects. A “suicide gene” codes for proteins that will initiate CAR T-cell death in response to the injection of a specific drug or protein into the patient’s body59.
A common suicide gene used in CAR T-cells is inducible caspase-9 (iCasp9). When two caspase 9 proteins pair together, or “dimerize”, they trigger a downstream biochemical cascade that results in cell death via apoptosis. The drug AP1903 will induce the pairing of these caspase 9 proteins in CAR T-cells, leading to CAR T-cell death while preserving normal cells that do not possess the iCasp9 gene59’60. Thus, by introducing suicide genes, researchers can selectively eliminate CAR T-cells in emergency cases when patients experience severe side effects.
Preclinical trials involving suicide genes have already commenced. In particular, one group of researchers investigated the efficacy of apoptosis-inducing drugs with CAR T-cells in which iCasp9 suicide genes have been inserted61. Following the administration of a drug denoted as AP20187 at 16nM concentration, researchers observed complete programmed cell death of CAR T-cells. These results validate that suicide genes present a viable option for quickly eliminating CAR T-cells in cases of dramatic negative side effects, solidifying their potential for use in clinical trials.
Promoting Specificity of CAR T Cell Immune Attack
Inhibitory CAR T-Cells
The emergence of inhibitory CAR T (iCAR T)-cells is one promising solution to overcoming unintentional toxicity of these engineered immune cells. These are cells that possess binding sites for target antigens found on glioma cell surfaces but also possess “inhibitory domains” or inhibitory antigen receptors62. These inhibitory domains are specifically engineered to bind to cell-surface proteins that are found only on the surface of healthy cells. Essentially, when encountering a healthy cell that happens to express the same target antigen as cancer cells, one set of receptors on the CAR T-cell will bind to the target antigen labeled “cancerous” and another set of inhibitory receptors on the CAR T-cells will bind to antigens only expressed on healthy cells63. Upon the binding of this inhibitory receptor to an antigen labeled “healthy”, the iCAR T-cell will emit chemical signals that prevent immune attack against the cell. The use of these inhibitory domains is referred to as NOT-gating64.
Thus, even if a healthy cell expresses the target antigen found on cancer cells, if the healthy cell also expresses the marker that binds to the inhibitory domain, it will be recognized as a healthy cell and the CAR T-cells will not launch an immune attack against them65.
iCARs have been deployed in mouse models implanted with Burkitt’s lymphoma cells to investigate the ability of inhibitory domains to guide CAR T-cells to cancerous B cells while limiting attacks against normal B cells66. In this particular study, the research group adjoined a KIR/PD-1 conjugate domain to CAR T-cells–this protein enabled the recognition of HLA-C1, a surface marker found to be present solely on healthy B cells. Following binding of the external KIR2DL2 receptor to HLA-C1, CAR T-cells would initiate chemical signaling to suppress attacks from other T-cells, protecting normal B cells. The researchers compared activity levels of these iCARs in the presence of malignant versus healthy B cells and confirmed a statistically significant decrease in iCAR activity upon encountering normal B cells, a result that confirms the role of iCARs in improving cell selectivity.
Split-Signal CAR T Cells
Another approach that should be adopted in conjunction with inhibitory CAR T-cells are split-signal CAR T-cells. These are cells that can recognize a combination of antigens on the surface of cells67. Thus, one can increase the selectivity of CAR T-cells so they only launch an attack on cells that express a specific combination of protein markers, making the range of activity of these engineered cells narrower and much more specific to gliomas.
Despite its promise, this specific strategy of split-signaling requires further testing with clinical trials before claims can be made regarding their efficacy in addressing off-target toxicity.
Bispecific Adapters
The ability to mutate and change is one of the leading challenges in treating cancer. Fortunately, bispecific adapters may be a potential solution. Bispecific adapters can bind to two sites, one on the cancer cell and the other to a special marker that is recognized by the CAR T-cell68. This bridges the two, enabling the CAR T-cell to initiate a stronger immune response with more guided and efficient targeting of tumor cells69. A carefully selected combination of bispecific adapters aids physicians in targeting tumor cell populations in which varieties of mutations and surface phenotypes have arisen70.
Flow Cytometry for Precise Cancer Protein Marker Profiles
Many researchers are also in the midst of performing flow cytometry experiments to compile a list of protein markers that are present only on the surface of cancer cells71. Although this search is difficult given the similarity between healthy cells and glioma cells, continuing this effort will allow for the production of a broader arsenal of CAR T-cells with a larger scope of antigen receptors.
Discussion
CAR T-cell therapy has demonstrated potential for tumor shrinkage and increased patient survival rates for gliomas. While scientists are still determining the best target tumor antigens, notable advancements, such as multi-specific CAR T-cell therapy, armored CARs, and allogeneic CAR T-cells, have shown promising results. Of these three advancements, dual-target CAR T-cell therapy holds the highest potential as it is the only treatment that has undergone clinical trials on humans with successful results. Although armored CARs and allogeneic CAR T-cells are promising, they are still in very early clinical trials and face challenges such as immune rejection. Despite many advancements, the biggest barrier in CAR T-cell therapy is off-target cytotoxicity. Due to cancer cell microevolution, CAR T-cells may accidentally attack normal cells. Fortunately, innovations to promote CAR T-cell specificity, such as inhibitory and split-signal CAR T-cells, are undergoing research and studies. Future research should prioritize larger clinical trials and testing aforementioned improvements on CAR T-cells in order to further prove their potential.
Limitations of Review
One of the limitations of our review is that CAR T-cell therapy is still a relatively new, emerging treatment, and thus our ability to assess its benefits and downsides remains somewhat restricted.
Another limitation of our study is that a significant portion of data on the efficacy of CAR T-cells must be derived from clinical trials, and in many cases this information is kept confidential or takes multiple years to collect. Thus, our scope of clinical trials to cull information from for this review is relatively limited.
Conclusion
While CAR T-cell therapy has been effective in treating some malignancies, its application in gliomas is still limited by multiple challenges. Inconsistent results are found in clinical trials as a result, especially because of factors like the changing tumor microenvironment and immune evasion. However, continued research in disciplines like immunology, neurology, oncology, and bioengineering will be critical in overcoming current limitations. As CAR T-cell therapy evolves, it bears the potential to transform glioma treatments and offer hope for patients with an otherwise challenging diagnosis.
References
- K. M. Walsh, H. Ohgaki, M. R Wrensch. Epidemiology. Handbook of clinical neurology. 134, pg. 3–18, 2016, https://doi.org/10.1016/B978-0-12-802997-8.00001-3. [↩]
- M. Weller, P. Y. Wen, S. M. Chang, L. Dirven, M. Lim, M. Monje, G. Reifenberger. Glioma. Nature Reviews Disease Primers. 10, 2024, https://doi.org/10.1038/s41572-024-00516-y. [↩]
- S. Park, M. V. Maus, B. D. Choi. CAR T-cell therapy for the treatment of adult high-grade gliomas. Npj Precision Oncology. 8, 2024, https://doi.org/10.1038/s41698-024-00753-0. [↩] [↩]
- F. B. Mesfin, T. Karsonovich, M. A. Al-Dhahir. Gliomas. In StatPearls. StatPearls Publishing. 2025, http://www.ncbi.nlm.nih.gov/books/NBK441874/ [↩]
- M. IJzerman-Korevaar, T. J. Snijders, A. de Graeff, S. C. Teunissen, C. M. de Vos. Prevalence of symptoms in glioma patients throughout the disease trajectory: A systematic review. Journal of Neuro-Oncology, 140, pg. 485–496, 2018, https://doi.org/10.1007/s11060-018-03015-9. [↩]
- F. B. Mesfin, T. Karsonovich, M. A. Al-Dhahir. Gliomas. In StatPearls. StatPearls Publishing. 2025, http://www.ncbi.nlm.nih.gov/books/NBK441874/. [↩]
- D. M. Bedoya, V. Dutoit, D. Migliorini. Frontiers in Immunology. Immunotherapy for glioblastoma. 12, 2021, https://doi.org/10.3389/fimmu.2021.640082. [↩]
- Y. Zhou, F. Shi, J. Zhu, Y. Yuan. An update on the clinical trial research of immunotherapy for glioblastoma. Frontiers in Immunology. 16, 2025, https://doi.org/10.3389/fimmu.2025.1582296 [↩]
- C.E. Brown, J.C. Hibbard, D. Alizadeh, M. Suzette Blanchard, H. M. Natri, D. Wang, J. R. Ostberg, B. Aguilar, J. R. Wagner, J. A. Paul, R. Starr, R. A. Wong, W. Chen, N. Shulkin, M. Aftabizadeh, A. Filippov, A. Chaudhry, J. A. Ressler, J. Kilpatrick, P. Myers-McNamara, M. Chen, L. D. Wang, R. C. Rockne, J. Georges, J. Portnow, M. E. Barish, M. D’Apuzzo, N. E. Banovich, S. J., Forman, B. Badie. Locoregional delivery of IL-13Rα2-targeting CAR T-cells in recurrent high-grade glioma: a phase 1 trial. Nature Medicine. 30, 2024, https://doi.org/10.1038/s41591-024-02875-1. [↩]
- B. D. Choi, E. R. Gerstner, M. J. Frigault, M. B. Leick, C. W. Mount, L. Balaj, S. Nikiforow, B. S. Carter, W. T. Curry, K. Gallagher, M. V ts Maus. Intraventricular CARv3-TEAM-E T Cells in Recurrent Glioblastoma. New England Journal of Medicine. 390, 2024, 10.1056/NEJMoa2314390. [↩] [↩]
- N. Montano, T. Cenci, M. Martini, Q. G. D’Alessandris, F. Pelacchi, L. Ricci-Vitiani, G. Maira, R. De Maria, L. M. Larocca, R. Pallini. Expression of EGFRvIII in glioblastoma: Prognostic significance revisited. Neoplasia. 13, 2011, 10.1593/neo.111338. [↩]
- P. Sharma, W. Debinski. Receptor-Targeted Glial Brain Tumor Therapies. International Journal of Molecular Sciences. 19, 2018, https://doi.org/10.3390/ijms19113326. [↩] [↩]
- N. Montano, T. Cenci, M. Martini, Q. G. D’Alessandris, F. Pelacchi, L. Ricci-Vitiani, G. Maira, R. De Maria, L. M. Larocca, R. Pallini. Expression of EGFRvIII in glioblastoma: Prognostic significance revisited. Neoplasia. 13, 2011, 10.1593/neo.111338. [↩]
- G. D. Cha, S. Jung, S. H. Choi, D. H. Kim. Local Drug Delivery Strategies for Glioblastoma Treatment. Brain Tumor Research and Treatment. 10, 2022, https://doi.org/10.14791/btrt.2022.0017. [↩]
- M. Geurts, M. Preusser. Locoregional delivery of chimeric antigen receptor-T cells: Breaking the spell in glioblastoma?. Neuro-oncology. 26, 2024, https://doi.org/10.1093/neuonc/noae063. [↩]
- X. Gu, Y. Zhang, W. Zhou, F. Wang, F. Yan, H. Gao, W. Wang. Infusion and delivery strategies to maximize the efficacy of CAR T-cell immunotherapy for cancers. Experimental Hematology & Oncology, 13, 70, 2024, https://doi.org/10.1186/s40164-024-00542-2. [↩] [↩]
- W. M. Pardridge. Drug transport across the blood–brain barrier. Journal of Cerebral Blood Flow & Metabolism. 32, 1959–1972, 2012, https://doi.org/10.1038/jcbfm.2012.126. [↩]
- S. J. Priceman, D. Tilakawardane, B. Jeang, B. Aguilar, J. P. Murad, A. K. Park, W.C. Chang, J. R. Ostberg, J. Neman, R. Jandial, J. Portnow, S. J. Forman, C. E. Brown. Regional Delivery of Chimeric Antigen Receptor–Engineered T Cells Effectively Targets HER2+ Breast Cancer Metastasis to the Brain | Clinical Cancer Research. American Association for Cancer Research. 24, 2018, https://doi.org/10.1158/1078-0432.CCR-17-2041. [↩]
- H. Qin, S. Ramakrishna, S. Nguyen, T. J. Fountaine, A. Ponduri, M. Stetler-Stevenson, C. M. Yuan, W. Haso, J. F. Shern, N. N. Shah, T. J. Fry. Preclinical Development of Bivalent Chimeric Antigen Receptors Targeting Both CD19 and CD22. Molecular Therapy Oncolytics. 11, 2018, https://doi.org/10.1016/j.omto.2018.10.006. [↩]
- R. Havunen, J. M. Santos, S. Sorsa, T. Rantapero, D. Lumen, M. Siurala, A. J. Airaksinen, V. Cervera-Carrascon, S. Tähtinen, A. Kanerva, A. Hemminki. Abscopal Effect in Non-injected Tumors Achieved with Cytokine-Armed Oncolytic Adenovirus. Molecular Therapy Oncology. 11, pg. 109-121, 2018, 10.1016/j.omto.2018.10.005. [↩]
- S. J. Bagley, A. S. Desai, J. A. Fraietta, D. Silverbush, D. Chafamo, N. F. Freeburg, G. K. Gopikrishna, A. J. Rech, A. Nabavizadeh, L. J. Bagley, J. Park, D. Jarocha, R. Martins, N. Sarmiento, E. Maloney, L. Lledo, C. Stein, A. Marshall, R. M. Leskowitz, J. K. Jadlowsky, S. Mackey, S. Christensen, B. S. Oner, G. Plesa, A. Brennan, V. Gonzalez, F. Chen, D. Barrett, R. Colbourn, M. P. Nasrallah, Z. Mourelatos, W.-T. Hwang, C. Alanio, D. L. Siegel, C. H. June, E. O. Hexner, Z. A. Binder, D. M. O’Rourke. Intracerebroventricular bivalent CAR T cells targeting EGFR and IL-13Rα2 in recurrent glioblastoma: a phase 1 trial. Nature Medicine. 31, 2025, https://doi.org/10.1038/s41591-025-03745-0. [↩]
- A. Herrick, M. Belur. What is Logic-Gating and Why Does It Matter in CAR T Therapy? Premier Research. 2024, https://premier-research.com/perspectives/what-is-logic-gating-and-why-does-it-matter-in-car-t-therapy/. [↩]
- J. H. Choe, P. B. Watchmaker, M. S. Simic, R. D. Gilbert, A. W. Li, N. A. Krasnow, K. M. Downey, W. Yu, D. A. Carrera, A. Celli, J. Cho, J. D. Briones, J. M. Duecker, Y. E. Goretsky, R. Dannenfelser, L. Cardarelli, O. Troyanskaya, S. S. Sidhu, K. T. Roybal, W. A. Lim. SynNotch-CAR T-cells overcome challenges of specificity, heterogeneity, and persistence in treating glioblastoma. Science Translational Medicine. 13, 2021, https://doi.org/10.1126/scitranslmed.abe7378. [↩]
- A. Herrick, M. Belur. What is Logic-Gating and Why Does It Matter in CAR-T Therapy? Premier Research. 2024, https://premier-research.com/perspectives/what-is-logic-gating-and-why-does-it-matter-in-car-t-therapy/. [↩]
- H. Okada. Anti-EGFRvIII synNotch Receptor Induced Anti-EphA2/IL-13Ralpha2 CAR (E-SYNC) T Cells. 2025, https://clinicaltrials.gov/study/NCT06186401. [↩]
- E. R. Hawkins, R. R. D’Souza, A. Klampatsa. Armored CAR T-Cells: The Next Chapter in T-Cell Cancer Immunotherapy. Biologics: Targets & Therapy. 15, 95–105, 2021, https://doi.org/10.2147/BTT.S291768. [↩]
- W. E. Carson, J. E. Dierksheide, S. Jabbour, M. Anghelina, P. Bouchard, G. Ku, H. Yu, H. Baumann, M. H. Shah, M. A. Cooper, J. Durbin, M. A. Caligiuri. Coadministration of interleukin-18 and interleukin-12 induces a fatal inflammatory response in mice: critical role of natural killer cell interferon-gamma production and STAT-mediated signal transduction. Blood. 96, 2000. [↩]
- M. Golmohammadi, N. Noorbakhsh, M. Kavianpour. CAR-T Cell Therapy: Managing Side Effects and Overcoming Challenges. Advanced Biomedical Research. 14, 38, 2025, https://doi.org/10.4103/abr.abr_531_23. [↩]
- Y. Zhai, G. Li, C. Pan, M. Yu, H. Hu, D. Wang, Z. Shi, T. Jiang, W. Zhang. The development and potent antitumor efficacy of CD44/CD133 dual-targeting IL7Rα-armored CAR T-cells against glioblastoma. Cancer Letters. 614, 2025, https://doi.org/10.1016/j.canlet.2025.217541. [↩]
- Hawkins, E. R., D’Souza, R. R., & Klampatsa, A. (2021). Armored CAR T-Cells: The Next Chapter in T-Cell Cancer Immunotherapy. Biologics : targets & therapy, 15, 95–105. https://doi.org/10.2147/BTT.S291768. [↩]
- S. Depil, P. Duchateau, S. A. Grupp, G. Mufti, L. Poirot. “Off-the-shelf” allogeneic CAR T cells: Development and challenges. Nature Reviews: Drug Discovery. 19, 2020, https://doi.org/10.1038/s41573-019-0051-2. [↩]
- K. M. N. Farid, G. Bug, A. Schmitt, F. Lang, M.L. Schubert, U. Haberkorn, C. Müller-Tidow, P. Dreger, M. Schmitt. GVHD after CAR T-cell therapy post allogeneic hematopoietic cell transplantation—Successfully treated by extracorporeal photopheresis. Frontiers in Immunology. 15, 2024. [↩]
- C. Diorio, D. T. Teachey,, S. A. Grupp. Allogeneic chimeric antigen receptor cell therapies for cancer: Progress made and remaining roadblocks. Nature Reviews: Clinical Oncology. 22, 2025, https://doi.org/10.1038/s41571-024-00959-y. [↩]
- S. U. Khan, K. Fatima, S. Aisha, F. Malik. Unveiling the mechanisms and challenges of cancer drug resistance. Cell Communication and Signaling. 22, 2024, https://doi.org/10.1186/s12964-023-01302-1. [↩]
- L. Rasche, L. Vago, T. Mutis. Tumour Escape from CAR-T Cells. In N. Kröger, J. Gribben, C. Chabannon, I. Yakoub-Agha, & H. Einsele (Eds.), The EBMT/EHA CAR-T Cell Handbook. 2022, http://www.ncbi.nlm.nih.gov/books/NBK584178/. [↩]
- A. Mishra, R. Maiti, P. Mohan, P. Gupta. Antigen loss following CAR-T cell therapy: Mechanisms, implications, and potential solutions. European Journal of Haematology. 112, 2024, https://doi.org/10.1111/ejh.14101. [↩]
- O’Rourke, D. M., Nasrallah, M. P., Desai, A., Melenhorst, J. J., Mansfield, K., Morrissette, J. J. D., Martinez-Lage, M., Brem, S., Maloney, E., Shen, A., Isaacs, R., Mohan, S., Plesa, G., Lacey, S. F., Navenot, J.-M., Zheng, Z., Levine, B. L., Okada, H., June, C. H., … Maus, M. V. A single dose of peripherally infused EGFRvIII-directed CAR T cells mediates antigen loss and induces adaptive resistance in patients with recurrent glioblastoma. Science Translational Medicine. 9, 2017, https://doi.org/10.1126/scitranslmed.aaa0984. [↩]
- C. L. Flugel, R. G. Majzner, G. Krenciute, G. Dotti, S. R. Riddell, D. L. Wagner, M. Abou-El-Enein. Overcoming on-target, off-tumour toxicity of CAR T-cell therapy for solid tumours. Nature Reviews: Clinical Oncology. 20, 2023. [↩]
- T. Braun, F. Kuschel, K. Reiche, M. Merz, M. Herling. Emerging T-cell lymphomas after CAR T-cell therapy. Leukemia. 39, 2025, https://doi.org/10.1038/s41375-025-02574-x. [↩]
- FDA Requires Boxed Warning for T cell Malignancies Following Treatment with BCMA-Directed or CD19-Directed Autologous Chimeric Antigen Receptor (CAR) T cell Immunotherapies. Center for Biologics Evaluation and Research (CBER). 2024, https://www.fda.gov/vaccines-blood-biologics/safety-availability-biologics/fda-requires-boxed-warning-t-cell-malignancies-following-treatment-bcma-directed-or-cd19-directed. [↩]
- H. Yin, X. Wei. The design of retroviral vectors used in the CAR‐T products, risk management, and future perspective. MedComm. 6, 2025, https://doi.org/10.1002/mco2.70067. [↩]
- J. Hu, C. E. Dunbar. T-cell lymphomas in recipients of CAR-T cells: Assessing risks and causalities. Blood. 144, 2024, https://doi.org/10.1182/blood.2024025828. [↩]
- J. Feng, M. Shao, Y. Hu, H Huang. Profile of capillary-leak syndrome in patients received chimeric antigen receptor T cell therapy. Bone Marrow Transplantation. 57, 2022, https://doi.org/10.1038/s41409-022-01562-4. [↩] [↩]
- R. Q. Le, L. Li, W. Yuan, S. S. Shord, L. Nie, B. A. Habtemariam, D. Przepiorka, A. T. Farrell, R. Pazdur. FDA Approval Summary: Tocilizumab for Treatment of Chimeric Antigen Receptor T Cell‐Induced Severe or Life‐Threatening Cytokine Release Syndrome. The Oncologist. 23, 2018, https://doi.org/10.1634/theoncologist.2018-0028. [↩] [↩]
- R. A. Gardner, O. Finney, C. Annesley, H. Brakke, C. Summers, K. Leger, M. Bleakley, C. Brown, S. Mgebroff, K. S. Kelly-Spratt, V. Hoglund, C. Lindgren, A. P. Oron, D. Li, S. R. Riddell, J. R. Park, M. C. Jensen. Intent-to-treat leukemia remission by CD19 CAR T-cells of defined formulation and dose in children and young adults. Blood. 129, 2017, https://doi.org/10.1182/blood-2017-02-769208. [↩]
- S. L. Maude, N. Frey, P. A. Shaw, R. Aplenc, D. M. Barrett, N. J. Bunin, A. Chew, V. E. Gonzalez, Z. Zheng, S. F. Lacey, Y. D. Mahnke, J. J. Melenhorst, S. R. Rheingold, A. Shen, D. T. Teachey, B. L. Levine, C. H. June, D. L. Porter, S. A. Grupp. Chimeric Antigen Receptor T Cells for Sustained Remissions in Leukemia. New England Journal of Medicine. 371, 2014, 10.1056/NEJMoa1407222. [↩]
- J. H. Park, I. Rivière, M. Gonen, X. Wang, B. Sénéchal, K. J. Curran, C. Sauter, M. Sadelain. Long-Term Follow-up of CD19 CAR Therapy in Acute Lymphoblastic Leukemia. New England Journal of Medicine. 378, 2018, 10.1056/NEJMoa1709919. [↩]
- Neelapu, S. S., Locke, F. L., Bartlett, N. L., Lekakis, L., Miklos, D. B., Jacobson, C. A., Braunschweig, I., & Go, W. Y. Axicabtagene Ciloleucel CAR T-Cell Therapy in Refractory Large B-Cell Lymphoma. New England Journal of Medicine. 377, 2017, 10.1056/NEJMoa1707447. [↩]
- R. C. Sterner, R. M. Sterner. Immune effector cell associated neurotoxicity syndrome in chimeric antigen receptor-T cell therapy. Frontiers in Immunology. 13, 2022, https://doi.org/10.3389/fimmu.2022.879608. [↩]
- J. H. Rees. Management of Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS). In N. Kröger, J. Gribben, C. Chabannon, I. Yakoub-Agha, & H. Einsele (Eds.), The EBMT/EHA CAR-T Cell Handbook. 2022, http://www.ncbi.nlm.nih.gov/books/NBK584157/. [↩]
- R. C. Sterner, R. M. Sterner. Immune effector cell associated neurotoxicity syndrome in chimeric antigen receptor-T cell therapy. Frontiers in Immunology. 13, 2022, https://doi.org/10.3389/fimmu.2022.879608. [↩]
- J. H. Rees. Management of Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS). In N. Kröger, J. Gribben, C. Chabannon, I. Yakoub-Agha, & H. Einsele (Eds.), The EBMT/EHA CAR-T Cell Handbook. 2022, http://www.ncbi.nlm.nih.gov/books/NBK584157/. [↩]
- I. Puri, D. Sharma, K. S. Gunturu, A. A. Ahmed. Diagnosis and management of tumor lysis syndrome. Journal of Community Hospital Internal Medicine Perspectives. 10, 2020, https://doi.org/10.1080/20009666.2020.1761185. [↩]
- S. Shaikh, H. Shaikh. CART-cell therapy toxicity. StatPearls. 2023, http://www.ncbi.nlm.nih.gov/books/NBK592426/. [↩] [↩]
- N. Sharma, P. M. Reagan, J. L. Liesveld. Cytopenia after CAR T-cell therapy: A brief review of a complex problem. Cancers. 14, 1501, 2022, 10.3390/cancers14061501. [↩]
- N. A. Vitanza, R. Ronsley, M. Choe, K. Seidel, W. Huang, S. D. Rawlings-Rhea, M. Beam, L. Steinmetzer, A. L. Wilson, C. Brown, A. Beebe, C. Lindgren, J. A. Gustafson, A. Wein, S. Holtzclaw, C. Hoeppner, H. E. Goldstein, S. R. Browd, J. S. Hauptman, M. C. Jensen. Intracerebroventricular B7-H3-targeting CAR T-cells for diffuse intrinsic pontine glioma: A phase 1 trial. Nature Medicine, 31, 2025, https://doi.org/10.1038/s41591-024-03451-3. [↩]
- C.E. Brown, J.C. Hibbard, D. Alizadeh, M. Suzette Blanchard, H. M. Natri, D. Wang, J. R. Ostberg, B. Aguilar, J. R. Wagner, J. A. Paul, R. Starr, R. A. Wong, W. Chen, N. Shulkin, M. Aftabizadeh, A. Filippov, A. Chaudhry, J. A. Ressler, J. Kilpatrick, P. Myers-McNamara, M. Chen, L. D. Wang, R. C. Rockne, J. Georges, J. Portnow, M. E. Barish, M. D’Apuzzo, N. E. Banovich, S. J., Forman, B. Badie. Locoregional delivery of IL-13Rα2-targeting CAR T-cells in recurrent high-grade glioma: a phase 1 trial. Nature Medicine. 30, 2024, https://doi.org/10.1038/s41591-024-02875-1. [↩] [↩]
- A. Shimabukuro-Vornhagen, P. Gödel, M. Subklewe, H. J. Stemmler, H. A. Schlößer, M. Schlaak, M. Kochanek, B. Böll, M. S. von Bergwelt-Baildon. Cytokine release syndrome. Journal for Immunotherapy of Cancer. 6, 2018, https://doi.org/10.1186/s40425-018-0343-9. [↩]
- B. S. Jones, L. S. Lamb, F. Goldman, A. D. Stasi. Improving the safety of cell therapy products by suicide gene transfer. Frontiers in Pharmacology. 5, 2014, 10.3389/fphar.2014.00254. [↩] [↩]
- T. Gargett, M. P. Brown. The inducible caspase-9 suicide gene system as a “safety switch” to limit on-target, off-tumor toxicities of chimeric antigen receptor T-cells. Frontiers in Pharmacology. 5, 2014, 10.3389/fphar.2014.00235. [↩]
- W. Xiao, L. Xu, J. Wang, K. Yu, B. Xu, Y. Que, J. Zhao, Q. Pan, C. Gao, P. Zhou, X. Zhang. FGFR4-specific CAR-T cells with inducible caspase-9 suicide gene as an approach to treat rhabdomyosarcoma. Cancer Gene Therapy. 31, 2024, https://doi.org/10.1038/s41417-024-00823-2. [↩]
- P. Celichowski, M. Turi, S. Charvátová, et al. Tuning CARs: Recent advances in modulating chimeric antigen receptor (CAR) T-cell activity for improved safety, efficacy, and flexibility. J. Transl. Med. 21, 197 (2023). [↩]
- S. Sun, H. Hao, G. Yang, Y. Zhang, Y. Fu. Immunotherapy with CAR-Modified T Cells: Toxicities and Overcoming Strategies. Journal of Immunology Research. 2018, https://doi.org/10.1155/2018/2386187. [↩]
- R. C. Abbott, H. E. Hughes-Parry, M. R. Jenkins. To go or not to go? Biological logic gating engineered T-cells. Journal for ImmunoTherapy of Cancer. 10, 2022, https://doi.org/10.1136/jitc-2021-004185. [↩]
- P. Celichowski, M. Turi, S. Charvátová, D. Radhakrishnan, N. Feizi, Z. Chyra, M. Simíček, T. Jelínek, J. R. Bago, R. Hájek, M. Hrdinka. Tuning CARs: Recent advances in modulating chimeric antigen receptor (CAR) T-cell activity for improved safety, efficacy, and flexibility. Journal of Translational Medicine. 21, 2023, https://doi.org/10.1186/s12967-023-04041-6. [↩]
- L. Tao, M. A. Farooq, Y. Gao, L. Zhang, C. Niu, I. Ajmal, Y. Zhou, C. He, G. Zhao, J. Yao, M. Liu, W. Jiang. CD19-CAR T Cells Bearing a KIR/PD-1-Based Inhibitory CAR Eradicate CD19+HLA-C1− Malignant B Cells While Sparing CD19+HLA-C1+ Healthy B Cells. Cancers. 12, 2020, https://doi.org/10.3390/cancers12092612. [↩]
- M. Huang, J. Deng, L. Gao, J. Zhou. Innovative strategies to advance CAR T-cell therapy for solid tumors. American Journal of Cancer Research. 10, pg. 1979-1992, 2020, https://pmc.ncbi.nlm.nih.gov/articles/PMC7407347/. [↩]
- M. J. Borrok, Y. Li, P. B. Harvilla, B. V. Maruthachalam, N. Tamot, C. Prokopowitz, J. Chen, S. Venkataramani, I. S. Grewal, R. Ganesan, S. Singh. Conduit CAR: Redirecting CAR T-cell specificity with a universal and adaptable bispecific antibody platform. Cancer Research Communications. 2, 2022, 10.1158/2767-9764.CRC-21-0150. [↩]
- M. J. Borrok, Y. Li, P. B. Harvilla, B. V. Maruthachalam, N. Tamot, C. Prokopowitz, J. Chen, S. Venkataramani, I. S. Grewal, R. Ganesan, S. Singh. Conduit CAR: Redirecting CAR T-cell specificity with a universal and adaptable bispecific antibody platform. Cancer Research Communications. 2, 2022, 10.1158/2767-9764.CRC-21-0150. [↩]
- Y. G. Lee, I. Marks, M. Srinivasarao, A. K. Kanduluru, S. M. Mahalingam, X. Liu, H. Chu, P. S. Low. Use of a single CAR T-cell and several bispecific adapters facilitates eradication of multiple antigenically different solid tumors. Cancer Research. 79, 2019, 10.1158/0008-5472.CAN-18-1834. [↩]
- H. Røgenes, K. Finne, I. Winge, L. A. Akslen, A. Ostman, V. Milosevic. Development of 42 marker panel for in-depth study of cancer associated fibroblast niches in breast cancer using imaging mass cytometry. Frontiers in Immunology. 15, 2024, https://doi.org/10.3389/fimmu.2024.1325191. [↩]