Abstract
Nowadays, global warming induces growing environmental stresses, such as heat and drought, which significantly inhibit plant growth and threaten global food security. This study investigated the potential of plant hormones to enhance stress resilience in Arabidopsis thaliana ecotypes Landsberg erecta (Ler) and Columbia-0 (Col-0). The plant responses under simulated drought (50 mM and 250 mM D-mannitol) and mild heat (26°C and 30°C) stresses were studied, separately, by measuring the growth of the roots and some of the shoots. The influences of abscisic acid (ABA), 6-benzylaminopurine (6-BA), gibberellins (GA), and (2,4-Dichlorophenoxy) acetic acid (24D) were first assessed under 250 mM D-mannitol induced drought. The influence of GA on Ler and Col-0 was further investigated and compared under drought and mild heat stresses. In addition, advanced 3D micro-Computed Tomography (micro-CT) technology was employed and demonstrated to be effective in plant research as a high-resolution, non-destructive tool. In addition, quantitative real-time polymerase chain reaction (PCR) analyzed the expression level of key genes ent-kaurene oxidase (KO) and ent-kaurenoic acid oxidase (KAO), and further provided evidence to confirm the effect of the GA. Our results demonstrated the importance of hormone GA in stress adaptation of the Arabidopsis thaliana, though its effect may vary between ecotypes and plant types. These findings still highlight the potential of hormones to improve plant resilience under climate change and plant tolerance to global warming.
Keywords:Drought stress, mild heat stress, plant hormone, gibberellins, gene expression, root, shoot
Introduction
Global warming refers to the gradual increase in Earth’s overall temperature due to the cumulative greenhouse gases1. Research indicates that with the continuous rise of temperature, plants would have to grow under elevated atmospheric temperature and prolonged drought stresses. Concurrently, global warming is projected to cause a continued reduction in precipitation. It is predicted to negatively impact the global crop productivity2.
Both mild heat and drought conditions can significantly inhibit plant growth and may lead to global food shortages3. Study has found that elevated temperature decreases biological activity of wheat directly, thereby reducing yields and posing challenges to agricultural development4. Drought is another main abiotic stress, which causes loss of crop yields5. Numerous studies have been conducted to address this critical issue. However, an effective and low-cost solution remains challenging. Recently, new approaches have been explored, such as plant growth regulators or plant hormones, whose knowledge could be transferred from the laboratory to the real crop production. This work studied how plant hormones stimulate Arabidopsis thaliana growth under drought or mild heat stresses and compared the impact of four major hormones on two different ecotypes, which haven’t been studied systematically before.
Plant hormones, also called phytohormones, are organic compounds synthesized by plants. Though those compounds exist in the plants at an extremely low concentration (ppm level), they play crucial roles in controlling plant growth, development, and their stress response. They function in complex ways by coordinating the physiological processes such as seed germination, root and shoot elongation, flowering, fruit ripening, and defense mechanisms against stresses. Also, different hormones influence plant growth differently, and their impacts also vary with concentration6. Among them, abscisic acid (ABA), 6-benzylaminopurine (6-BA), gibberellins (GA), and (2,4-Dichlorophenoxy) acetic acid (24D) were widely studied and have been demonstrated to be effective in promoting plant growth but with different mechanisms7,8,9,10. Abscisic acid (ABA) is a key plant hormone to help plants adapt the environmental challenges. It affects not only seed dormancy, cell growth, and leaf abscission, but also regulates stomatal closure and root development under unfavorable conditions7. The cytokinin 6-Benzyladenine (6BA) modulates plant growth and development by stimulating cell division and promoting cell enlargement11. Gibberellins (GA), a large family of tetracyclic diterpenoid phytohormone, has also been demonstrated to be crucial for promoting stem elongation, seed germination, and fruit development, etc.9. 2,4-dichlorophenoxyacetic acid (24D), a synthetic auxin, promotes plant growth and wound healing at low concentrations9,10. Although the influence of those four hormones has been studied, a systematic comparison of those hormones on the same plant is absent. In addition, Arabidopsis thaliana was selected as the model plant for two reasons. First, it has a short life cycle, small size, and hardiness, making it excellent to serve as a model plant species. Second, the completion of its genome sequencing in 2000 by the international Arabidopsis research community established a solid foundation for in-depth studies of this model organism and further application of results to other plants12. Landsberg erecta (Ler) and Columbia-0 (Col-0) are two typical ecotypes of Arabidopsis thaliana with the same life cycle. However, Col-0 is a standard, reference model as a wild type, while Ler is the results of an erecta mutant. It has been previously reported that Ler shows an escape strategy while Col shows a tolerance strategy to drought stress13. This difference allows the study of understanding how the phytohormones stimulate the two ecotypes’ response to stresses. The results will provide important evidence on how the gene variation impacts the plant response differently to the environment.
This work primarily investigated the root growth of the plants as it controls the total nutrition and water uptake of the plant and are particularly vulnerable to stressful environments, thereby inhibiting the overall plant growth. Hence, a detailed investigation of root development under stressful environment and exploration of improvement strategies are essential for maintaining and elevating plant productivity.
Nevertheless, the underground nature of roots brings substantial observational challenges compared to aboveground tissues, making root studies more difficult and less common. Therefore, the use of novel methods and technologies for root observation is crucial for further advancing root growth research. The micro-CT (Micro Computed Tomography) technique employed in this study represents a groundbreaking 3D non-destructive imaging technology with submicron resolution for the root study14. This method enables observation of root systems without disturbing the plant growth, signifying a milestone in study of root development.
Overall, this research compared the influence of four major different hormones on the growth of two Arabidopsis ecotypes (Ler and Col-0) under simulated drought or mild heat stresses and revealed the effectiveness of gibberellin (GA) in enhancing plant growth under such stresses. The effects of GA were further confirmed by studying the relationships between the gene KO and KAO expression levels, and plant development. It provided a solution in addressing plants’ adaptation disabilities to drought or mild heat stress. As the research was conducted on a model system of Arabidopsis thaliana, our results are valuable for studying how the hormone regulates plants and for investigating potential solutions to future crises in possible food shortage. However, because of the observed slight difference in the influences of phytohormone on Ler and Col-0 originating from the gene variation, more research is required to achieve its final application on other plants. This work also has two primary limitations. First, only two different concentrations of hormones were investigated, and the two concentrations were selected based on previously literature15,16,17,18. Second, all experiments were conducted under controlled environment. No experiments were conducted in the fields or other plants.
Methods
Seed treatment and plant cultivation
Arabidopsis thaliana Columbia-0 (Col-0) and Landsberg erecta (Ler) were planted as following. Approximately 200 seeds for each type were surface sterilized by 1 mL 75% ethanol for 15 min and then washed three times by deionized water and kept at 2-4°C for 2 days. The seeds were planted in a half-strength Murashige & Skoog (1/2 MS) medium, which contains 2.2 g/L MS salt, 0.5 g/L 2-morpholinoethanesulfonic acid, and 10 g/L sucrose with a pH value of 5.8, adjusted by 1M NaOH. 10 g agar was added to the mixture. The mixture was autoclaved at 121°C for 20 min and marked as 1/2 MS. Under sterile conditions, the 1/2 MS medium was poured into Petri dishes and solidified. When the D-mannitol and hormones were applied, they were mixed with the 1/2 MS medium at the denoted concentrations before the medium was poured into the dishes. All ingredients used above were purchased from Sigma Aldrich. The surface-sterilized seed was planted in two rows at 10 seeds/row on the medium. Sealed plates were vertically incubated under 4000-5000 Lux light intensity under conditions as below:
All seeds were grown on 1/2 MS medium for 14 days with a light/dark cycle of 16 hours/8 hours per day. Drought stress was simulated by adding D-mannitol into the medium at 22°C, and the concentration of mannitol in the medium were controlled at 50 mM or 250 mM (9.1 g/L or 45.5 g/L, respectively)19. Mild heat stress was simulated by growing the plant at 26°C or 30°C without adding mannitol in the medium. The control samples grew at 22°C on the 1/2 MS medium. The concentration of the hormone in this work corresponds the final hormone concentration in the medium. Four biological replicates were conducted and placed randomly in the chamber.
Measurement of shoots and roots
In the experiment, the longest root was defined as the primary root. The other roots with a length not shorter than 1 mm were counted and defined as lateral roots. The length of the primary roots and the number of lateral roots were measured on the plants after 14-day growth. The morphology of the shoots and roots was recorded by photos. The fresh weight of the shoots was measured with a balance. All results are the average of 20 plants total, which were randomly selected from 4 biological replicates, 5 plants each. Three technical replicates were conducted.
Measurement of KAO and KO gene expression level
For quantitative reverse transcription-polymerase chain reaction (qRT-PCR), the extraction of RNAs and reverse transcription was conducted as being described previously20. The procedures were as following: the total RNA was extracted from Arabidopsis thaliana tissues using Trizol reagent. liquid nitrogen frozen tissue (50-100 mg) was homogenized in 1 mL Trizol and then sat for 5 min, followed by centrifugation at 12,000 rpm under 4°C for 5 min. The supernatant was mixed with 200 μL of chloroform and centrifuged again. The liquid was mixed with 500 μL of isopropanol. The mixture was stabilized for 20-30 min at -20°C. Then, the precipitate was mixed with 1.5 mL of 75% ethanol, centrifuged at 10000 rpm for 5 min. The precipitate was collected and dissolved in 25 μL RNase-Free water (DERC-treated water)21,22.
The complementary DNA (cDNA) was synthesized by mixing the reverse transcription solution and the genomic DNA (gDNA) removal solution, followed by treatments at 42°C for 15 min and 93°C for 3 min. The reverse transcription solution was prepared through mixing 2 μL 10× Fast RT Buffer, 1 μL RT Enzyme Mix, 2 μL FQ-RT Primer Mix, and 5 μL RNase-Free ddH2O. The gDNA removal solution is obtained by mixing 2 μL 5x gDNA Buffer and 8 μL RNA mixture as prepared above and treated at 42°C for 3 min.
KAO and KO gene expression was quantified by Bio-Rad CFX96 using 20 μL PCR reagent, TOYOBO SYBR® Green Real-time PCR Master Mix (Cat# QPK-201) containing 10 μL Master Mix, 0.8 μL 10 μM Forward Primers, 0.8 μl 10 μM Reverse Primers, and 8.4 μL the synthesized cDNA23. The qRT-PCR results are shown as relative transcript levels, which were normalized by the ACTIN primers. The ACTIN genes were ACTIN-QF: TGGCATCAYACTTTCTACAA and ACTIN-QR: CCACCACTDAGCACAATGTT. The reference gene used for KO measurements are LW1662-K0-QRT-F1: ATGAAACGTCAGATCTTCAC and LW1664-KO-QRT-R1: TGATGCAATCTCAGAGGAT. The reference gene used for KAO measurements are LW1665-KA0-QRT-F1: ATGATCTGGCTAAGCTCGA and LW1667-KAO-QRT-R1: AACAGAGTATGTAAGTCTG. All experiments included three biological replicates were analyzed and three technical replicates. The tested cycle threshold (CT) for each measurement was then normalized by the CT of the corresponding reference genes, marked as DD CT, and the gene expression level was the average of 2-DD CT.
Micro-computed tomography scanning of Arabidopsis thaliana
High-resolution micro-computed tomography (micro-CT; Xradia 520 Versa) was employed to analyze the Arabidopsis thaliana growth, using a voltage of 50 kV, a current of 100 mA and exposure time of 0.4 mS. It is a non-destructive 3D imaging technique with a submicron resolution. This approach enabled in situ scanning of plants in soil while maintaining the plant structural integrity, thereby yielding comprehensive 3D morphological data for multiaxial growth analysis. In this work, the 1/2 MS medium was solidified in a 50 mL centrifugal tube. The seeds were planted on the medium and the roots grew in the tube for observation.
Results
Arabidopsis thaliana growth under simulated drought or mild heat stress
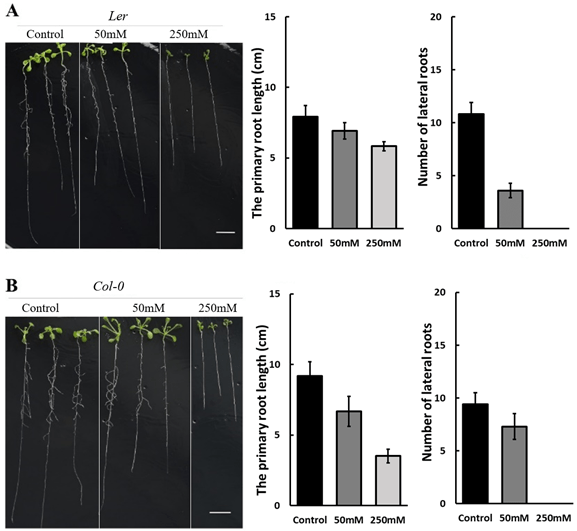
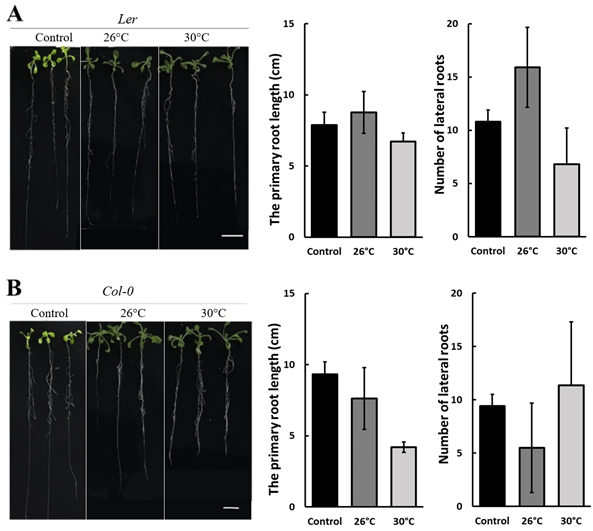
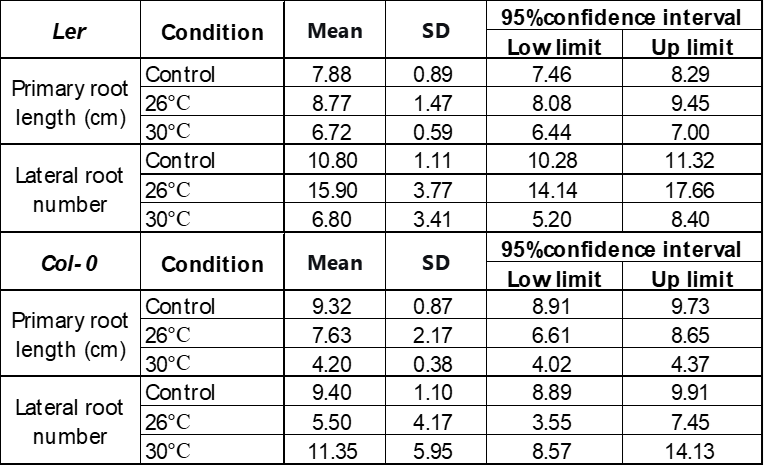
D-mannitol (mannitol) has been demonstrated to be an effective chemical to simulate drought stress by its osmotic effect24. The stress level can be controlled by the mannitol concentration. This work studied the growth of Arabidopsis thaliana under drought induced by 50 mM and 250 mM mannitol in the 1/2 MS medium. Figure 1 compares the growth of Ler and Col-0 under concentrations of 0 mM (control), 50 mM, and 250 mM D-mannitol at 22 °C, suggesting that the primary root length and number of lateral roots were inhibited with the increasing concentration of mannitol. The inhibition is more significant on Col-0 than Ler in the primary root length, while more significant on Ler in the number of lateral roots. The photos on the left in Figure 1 further show that the shoots grown under 50 mM mannitol have similar size as the control and is significantly larger than that under 250 mM-induced stress, suggesting a more severe growth inhibition by 250 mM mannitol.
The mild heat stress was studied by growing the Arabidopsis thaliana in a constant temperature incubator. Ler and Col-0 grew at temperatures of 22℃ (control), 26℃, and 30℃ with no mannitol. Figure 2 shows that compared to the control, the primary root length of Ler increases slightly at 26℃ but decreases about 20% at 30℃, while the number of lateral roots increases by 30% at 26℃ and decreases by 40% at 30℃. The shoots grow stronger at 26℃ than control, suggesting 26℃ is favored by Ler. The primary root length of Col-0 reduced to about 75% and 50% at 26℃ and 30℃ compared to 22℃, but no clear temperature dependence was observed for the number of lateral roots. The shoot of Col-0 is larger when growing at elevated temperatures, as shown by the photos on the left in Figure 2B.
Hormone influences on the growth of Arabidopsis thaliana under drought simulated by 250 mM D-mannitol
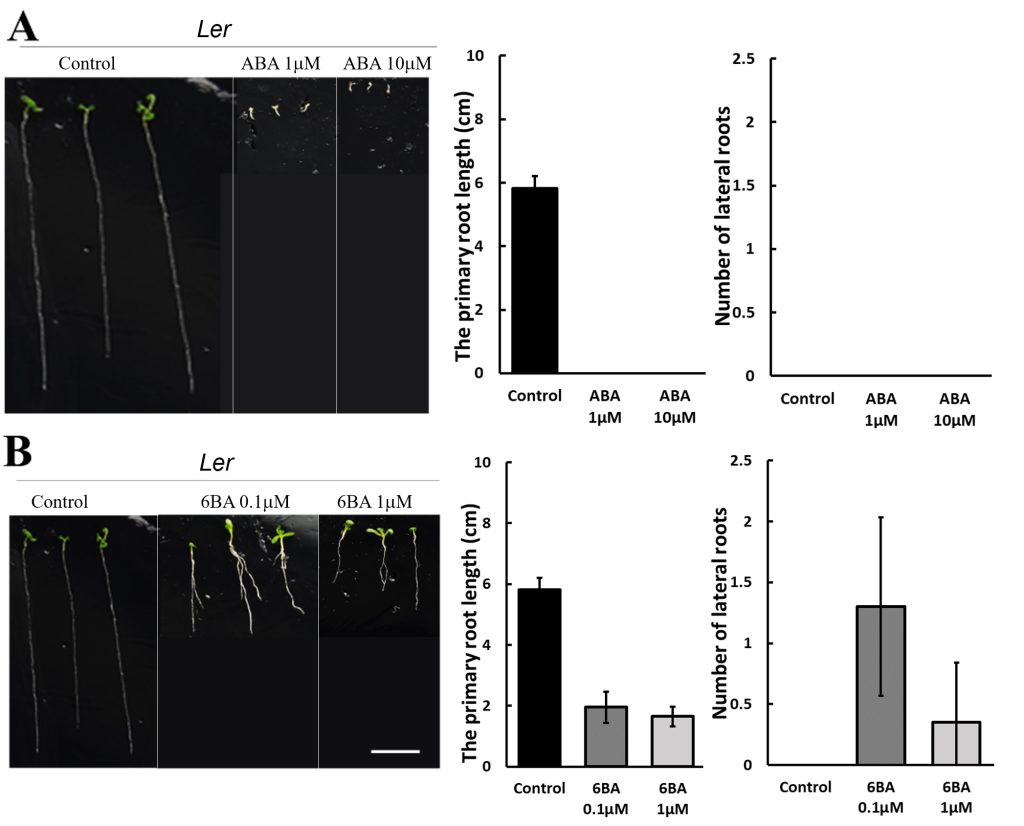
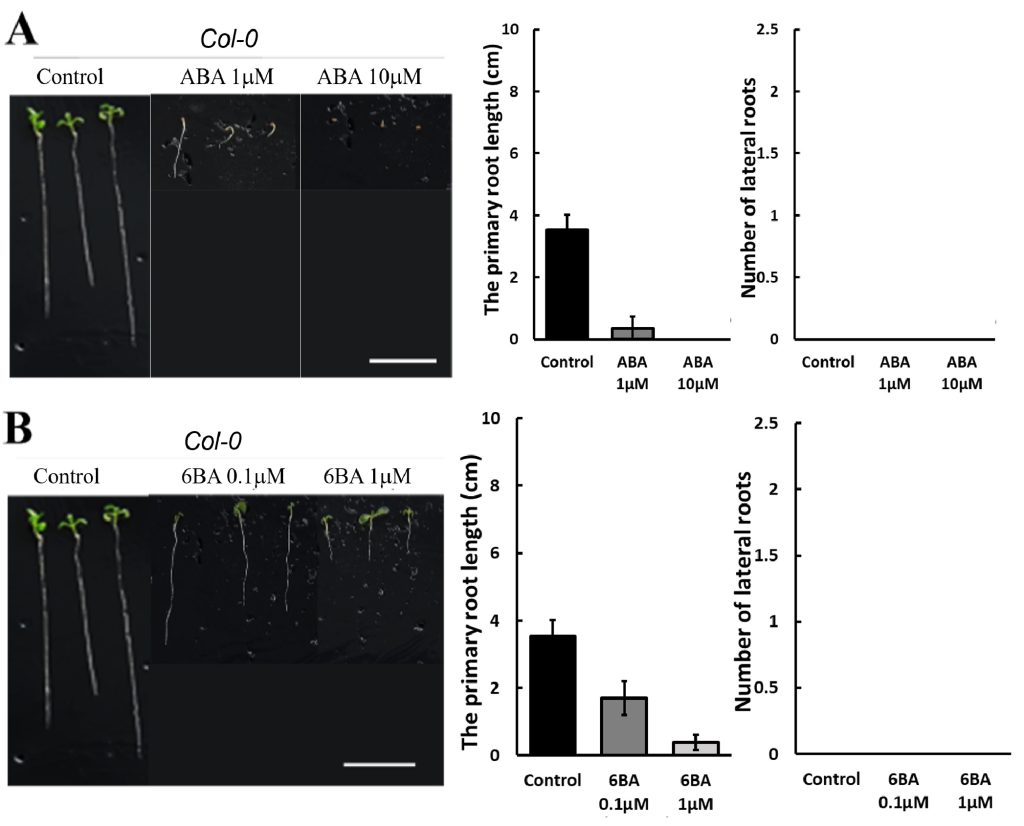
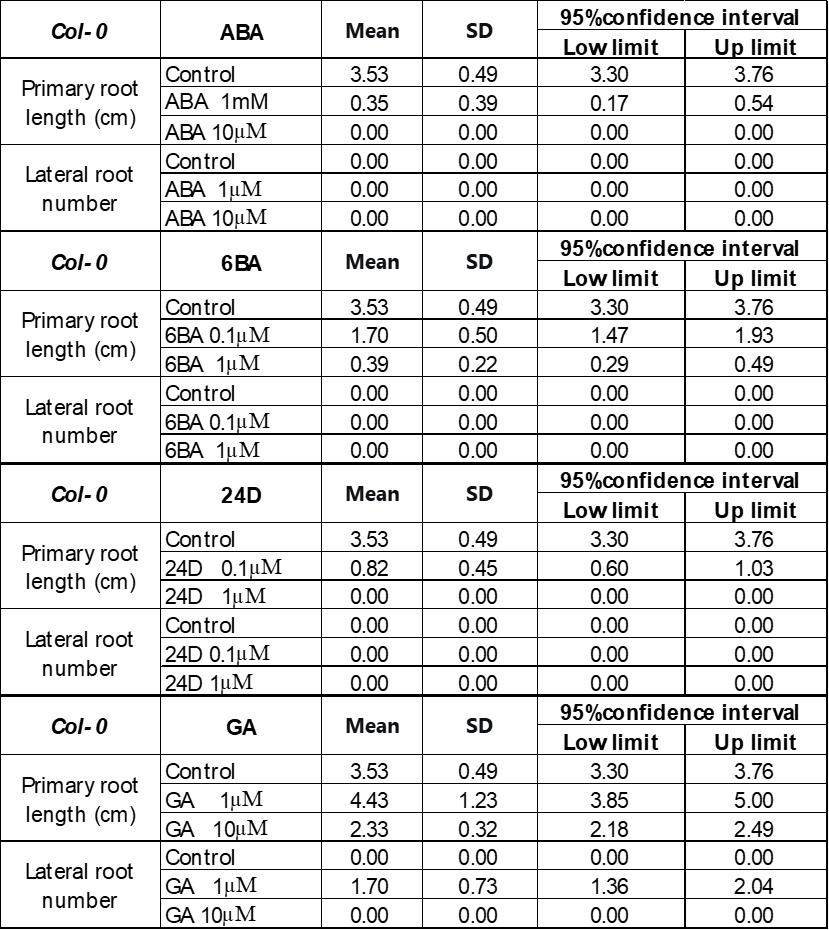
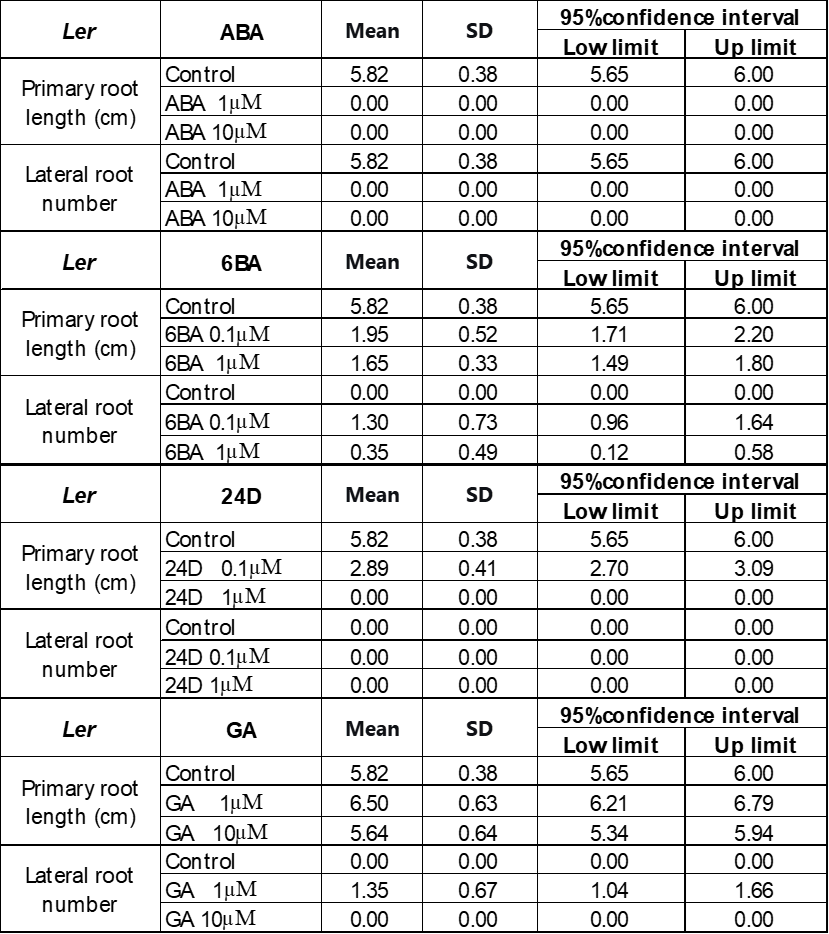
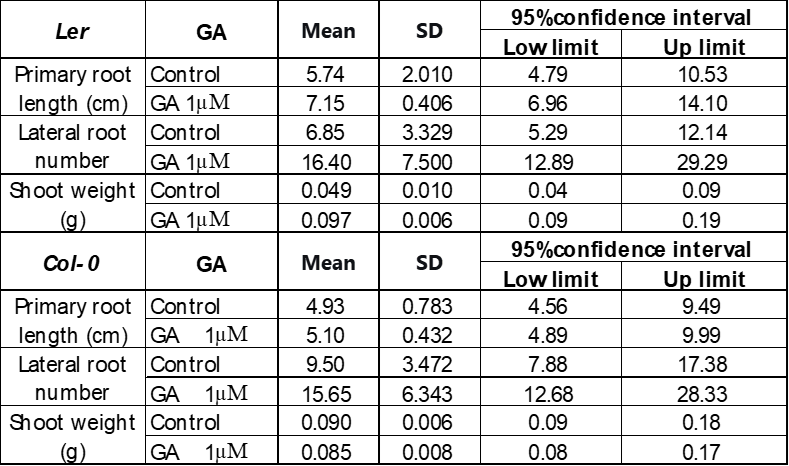
250 mM D-mannitol simulated drought was selected as the stress for the hormone screening. Abscisic Acid (ABA), 6-Benzylaminopurine (6BA), 2,4-Dichlorophenoxyacetic acid (24D), and Gibberellins (GA) were selected because they have been demonstrated to be effective in enhancing plant growth by other studies 6-11. The growth of the primary roots and lateral roots of Ler and Col-0 were further inhibited by 1 μM and 10 μM ABA (Figure 3A & 4A) or 0.1 μM and 1 μM 24D (Figure 3C & 4C) under 250 mM D-mannitol induced drought. The growth of the shoots was inhibited significantly by ABA and 24D. In addition, the 0.1 μM and 1 μM of 6BA also inhibited the primary root growth of Ler and Col-0 (p < 0.05), but it stimulated the lateral root growth of Ler (p < 0.05) (Figure 3B). In addition, the 6BA maintained the shoot growth of Ler but inhibited the shoot growth of Col-0.
1 μM of GA slightly increased the primary root length of Arabidopsis and notably enhanced the number of lateral roots in comparison with the control (Figure 3D & 4D). 10 μM of GA has no significant effects (p > 0.05) on their growth except for about 25% reduction of the primary root length in Col-0. We concluded that low concentrations of GA (1 μM) effectively improved the root length and lateral root quantity, together with an improved growth of the shoots under simulated drought conditions (250 mM D-mannitol).


Non-destructive 3D micro-CT to monitor the growth of Ler under stress
3D X-ray micro-CT technology has been an important tool in medical and biological studies25. It records 2D project images by transmitting X-rays through the sample and processes the 2D into 3D models. It quantitatively measures the morphology of a living organ and enables in-vivo monitoring of the sample growth at a sub-micron meter scale. However, it has been rarely used in plant and agricultural research. In this study, we have demonstrated its effectiveness in measuring the growth of Arabidopsis thaliana without disturbing its growth.
The Arabidopsis thaliana Ler was cultivated under 30°C mild heat or drought simulated by 50 mM mannitol. We compared its root growth with and without 1 μM GA, as shown in Figure 5. After adding GA, Ler’s root volume significantly increased under either simulated drought or mild heat stresses, and more lateral roots grew than those having no GA added. The effect of GA was more effective for plants under mild heat than drought. The results agree well with what shown in section 3.2 and confirmed that the plant hormone is capable to boost the growth of the plant. In addition, the micro-CT technology has also been demonstrated to be effective on studying the growth of plants with no destruction.

Influence of GA on the growth of Ler and Col-0 under stress
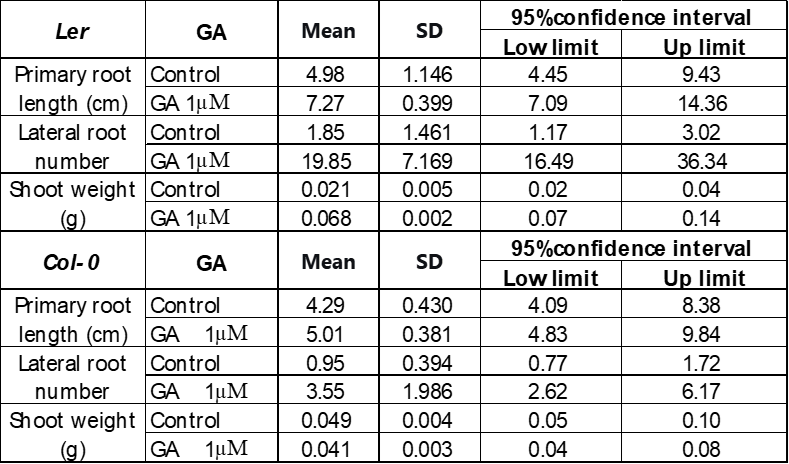
Sections above have proved that 1μM of GA effectively enhanced both the root and shoot growth of Arabidopsis thaliana under 250 mM D-mannitol-induced drought. Its influence was further studied under 50 mM D-mannitol-induced drought and 30℃ stresses, respectively.
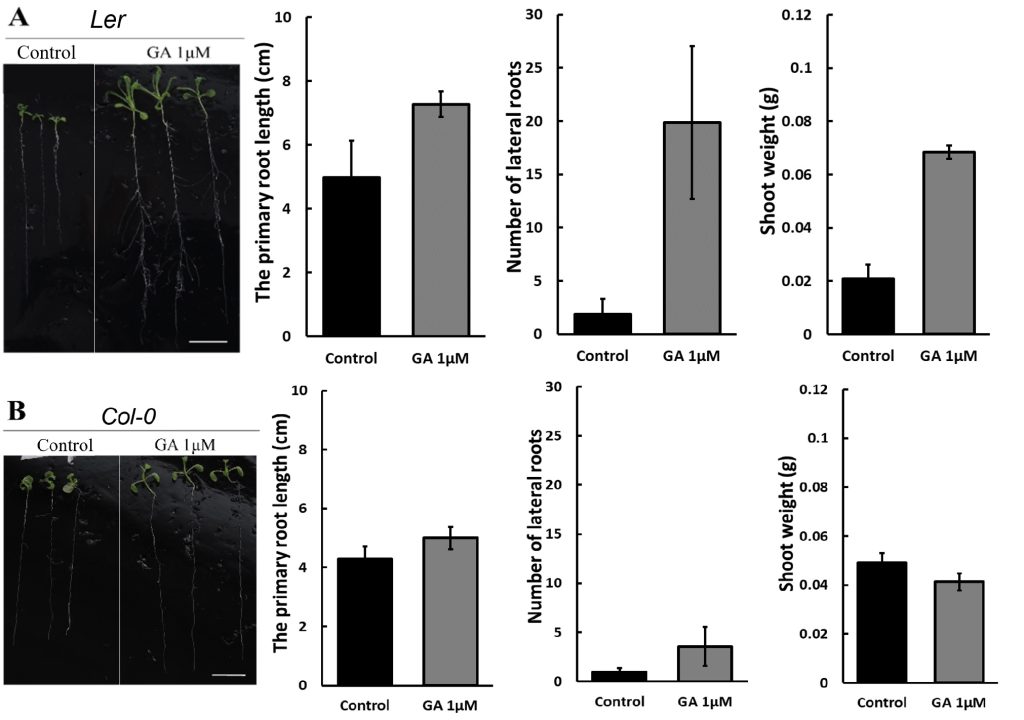
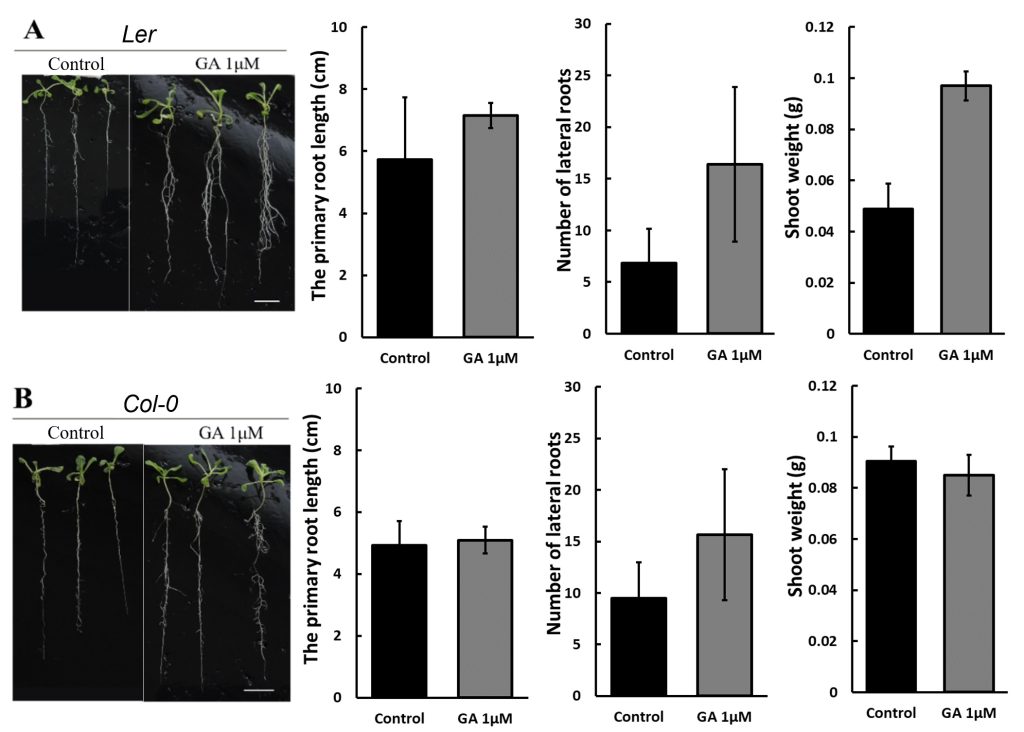
Figure 6 showed that 1 μM GA enhanced the growth of Ler more effectively than Col-0 under 50 mM D-mannitol simulated drought. The primary root length of Ler increases about 40%, and the number of lateral roots increases more than 10 times, which is much higher than the increase in Col-0, 20% and 4 times. The photos on the left in Figure 4 also suggest a significant increase in the Ler shoot, with a weight increase of three times. Though the shoot dimension of the Col-0 seems larger with GA on the photo, the shoot weight remained after adding 1 μM GA, suggesting GA has no impact on the Col-0 shoot growth.
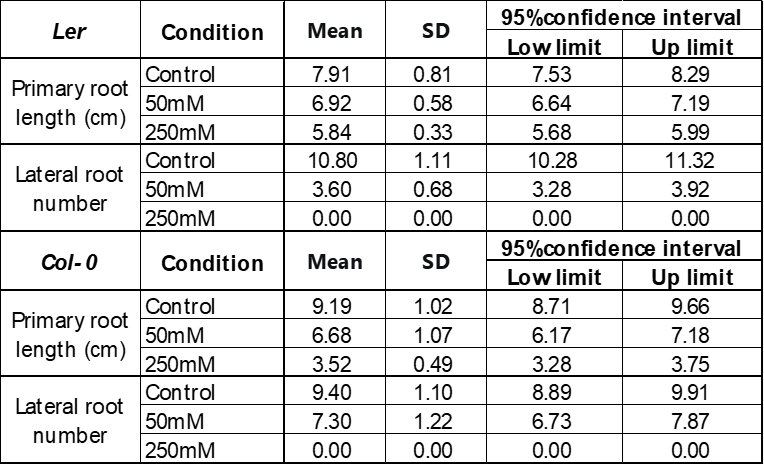
The impact of 1 μM GA on the growth of Ler and Col-0 under 30℃ heat stress is also studied as summarized in Figure 7. 1 μM of GA enhanced the root growth of Ler and Col-0 similarly, with an increase in the primary root length by 20-30%, the number of lateral roots by about 2 times, and a similar increase in the shoot morphology visually. However, the lateral roots were observed to be stronger and longer in Ler. The shoot weight of Ler grown under 1 μM GA is about two times larger than the control, while GA didn’t show an increase in the weight of Col-0 shoot, although its morphology seems larger than the control in Figure 7B.
Overall, the addition of GA enhances the growth of Ler and Col-0. This is consistent with the established model which suggests that GA promotes the plant growth by triggering the degradation of the DELLA repressor proteins. The DELLA proteins were accumulated when the plant experienced stress conditions. Therefore, the proteins inhibited the plant growth. Its degradation recovered the plant growth again26.
The KO and KAO gene expression levels of Arabidopsis thaliana under stresses
We also measured the gene expression level of the KO and KAO in Ler and Col-0 grown under drought or mild heat stress, respectively. Both KO and KAO genes are key synthetic enzyme genes involved in the GA (gibberellin) synthesis in plants. The results, as shown in Figure 8, indicate that the KAO gene expression level of Ler is doubled and KO is tripled compared with the control when growing under drought stress. Under mild heat, the expression level of the KAO gene was also doubled, while the KO gene expression level increased about seven times. For Col-0, the expression levels of the KAO and KO genes also showed significant increases under drought stress, which are about 6 times and 3 times, respectively. Under heating conditions, the KAO gene exhibited less than a two-fold increase in expression level, whereas the KO gene showed a slightly more than two-fold increase. The increase in KO and KAO expression level is an indication of the improved amount of GA in the plant, which improves the plant resilience to the stress27. Our results confirmed that the production of GA is an important process for plant adaption to stress and provided evidence for the statement that the GA enhances the growth of Arabidopsis thaliana under stresses effectively. The work also indicated the different response of Ler and Col-0 to GA under that same concentration. It could be attributed to the phenotypic variation, which results in the variations in the response of hormone concentration and signaling in plants28. This finding provides a theoretical explanation for GA in enhancing the plant adaption to stressful environments and the differences in various plants.

Discussion
Global warming becomea a worldwide problem today. Studies have predicted that in about 100 years, the global average temperature would increase by 1.0 to 3.9 °C; and in approximately 300 years, the increase could reach 0.6 to 7.8 °C or even higher. If greenhouse gas emissions rise further, the increase in the average temperature would become more significant29.
Global warming is regarded as a major threat to agriculture as it will bring abiotic stresses to plant growth. The threat from drought stress and mild heat stress to crop growth has attracted much attention30, as they would substantially limit the crop growth and reduce their yields, therefore affecting the global food supply31.
In this work, we explored the possibility of using phytohormone to alleviate this issue and concluded that GA should be one solution to help crops adapt to the stresses and resume their productivity.
Plants are considered to grow under stress when the environment is not ideal. The adverse conditions include drought, mild heat, cold, salinity, and flooding, etc. This research studied the growth of Arabidopsis thaliana, a model plant, under simulated drought and mild heat stresses. The results showed that drought stress simulated either 50 mM or 250 mM mannitol inhibited the growth of Ler and Col-0 significantly. The inhibition becomes more severe at a higher mannitol concentration, representing a harsher stress. There are no lateral roots under 250 mM mannitol. This could be understood by a higher osmotic stress induced by the higher mannitol concentration32.
The growth of Arabidopsis thaliana was also observed at 26°C and 30°C to simulate the mild heat stress with a control at 22°C. At 26°C, there was a light increase in the primary root length or the number of lateral roots for Ler, suggesting that 26°C is favorable for Ler. At 30°C, the root growth of Ler was significantly suppressed, suggesting that 30°C is a stressful environment for Ler. The average root length of Col-0 grown at 26°C and 30°C was reduced by 25% and 50%. No obvious inhibition was observed on the lateral root growth. The results indicate that the mild heat stress caused by global warming negatively impacts plant root growth, and the impact becomes more severe at higher temperatures. Meanwhile, as Arabidopsis thaliana is a model plant, the response of Ler and Col-0 shared the same trend but still with little difference.
To explore solutions to accelerate the plant adaptation to stress and improve their productivity, four plant hormones were investigated, including GA (gibberellin), ABA (abscisic acid), 6-BA (a synthetic cytokinin), and 24D (2,4-dichlorophenoxyacetic acid. Bharath et al. have found that ABA, as a stress hormone, accumulates in plant under stress to defend against the environment33. However, this study found that the 1 µM and 10 µM ABA completely inhibited the root growth of Arabidopsis thaliana Ler and Col-0 and reduced the shoot growth significantly while Ler and Col-0 still grew under control conditions. This is different from what Miao et al. observed, that the addition of ABA improves the root growth of Col-0 under water strength34. Hormone ABA functions as either a promoter or an inhibitor, depending on the type of stress, its concentration, and other influencing factors35,36. Our results suggested that under drought or mild heat stress, several µM ABA inhibits the growth of Ler and Col-0. 0.1 µM and 1 µM 6BA hormone also inhibit the shoot and the primary root growth of Ler and Col-0 under drought or mild heat stress, as shown in Figure 3 and 4. The 6BA did stimulate the lateral root of Ler grow more than control under drought as the function of 6BA observed in tortisepalum, where the 6BA promotes the growth of the lateral buds37. The effect is more prominent at 0.1 µM, suggesting that an overwhelming high concentration of phytohormone may suppress the plant growth.
Hormone 2,4-dichlorophenoxyacetic acid (24D) is a synthetic auxin, used to promote root and shoot growth at low concentration and is normally used to regulate the plant response to environmental stresses. Comparing to control in Figure 3 and 4, the 0.1 µM and 1 µM hormone 24D didn’t show any impact on the lateral root growth of Ler and Col-0 under simulated drought conditions. They even inhibited the primary root growth by 50% at 0.1 µM and 100% at 1 µM. The results indicate that though 24D is used as an herbicide-free growth enhancer for crops like wheat and rice38. It doesn’t help to stimulate the Ler and Col-0 growth under drought stress at similar concentration. That means when we select plant hormone, both the hormone concentration, type and the plant species need to be considered.
In contrast, GA is proved to be very helpful for root growth under stresses. Figure 3 and 4 showed that 1 μM GA significantly promoted both the shoot, the primary root length and the number of lateral roots of Ler and Col-0, while 10 μM GA inhibits their growth comparing to control under drought when simulated by 250 mM mannitol. Results suggested 1 μM GA is an appropriate concentration to stimulate the plant grow under stresses. 1 μM GA is also demonstrated to be effective to enhance the root growth of Ler and Col-0 under stresses as simulated by 50 mM mannitol and 30°C mild heat. It is also effective for the shoot growth of Ler but the shoot growth of Col-0 at 1 μM GA under drought and mild heat is like the control. The positive effects of GA on the growth of Arabidopsis thaliana is further confirmed by the increase in the expression levels of KO and KAO. KO and KAO are two critical gene existing in the GA synthesis pathway39. The increase in the KO and KAO gene expression level indicates an improved amount of GA is synthesized. Our study showed that under simulated drought and mild heat stresses, the expression levels of KO and KAO genes in both Col-0 and Ler significantly increased, indicating to adapt the stress, the plant synthesized more GA. This means more GA would help the plant to grow better under stress. Moreover, the results agree well with our results that the introduction of GA promotes the growth of plant under drought and mild heat stresses.
Among all four hormones measured in this study, only GA greatly enhanced the growth of Col-0 and Ler under simulated stresses. Comparing to control, the addition of GA improves a significant amount of the root growth and the aboveground shoot biomass. It restores the growth of Ler and Col-0 under stresses to that in normal environments.
In addition, the Micro CT technology was confirmed our conclusion further by scanning the vivo-plants grown under simulated mild heat or drought stresses. The Arabidopsis thaliana was compared with and without the addition of 1 μM GA, resulting in complete 3D views of the roots. The results clearly showed that plants treated with GA had better root growth, and both the number of lateral roots and root length increased significantly, with more pronounced effects under mild heat stress.
The hormone ABA, 6BA, 24D and GA worked differently on the growth of Arabidopsis thaliana, and the same hormone also worked differently on Ler and Col-0, indicating the importance of the natural variation. The results revealed that only GA improves the Arabidopsis thaliana growth significantly under the drought or mild heat stresses. The enhancement in the Ler and Col-0 ecotype growth are slightly different, possibly due to the different genetic background. The improved KO and KAO gene expression levels further confirmed the positive effect of adding GA on the plant growth as they are key gene for GA biosynthesis in plants. The finding provides valuable insights for studying the function of hormones in plant growth. Appropriate utilization of GA could provide a potential solution to mitigate the risk of reduced plant growth under environmental stresses.
Appendix:
References
- S. Pappas What is global warming? https://www.livescience.com/37003-global-warming.html (2022). [↩]
- E. Hull, G. Head, G. T. Tzotzos Appendix B – Climate change and global warming: Impacts on crop production. Genetically Modified Plants (Second Edition), 283 (2021). [↩]
- Aalto University A food crisis is brewing – Climate change could put half of the world’s crops at risk https://scitechdaily.com/a-food-crisis-is-brewing-climate-change-could-put-half-of-the-worlds-crops-at-risk (2025). [↩]
- S. Asseg, I. A. N. Foster, N. C. Turenr The impact of temperature variability on wild heat yields. Global Change Biology 17 (2), 997 (2011). [↩]
- X. Liu, Y. Pan, X. Zhu, T. Yang, J. Bai, Z. Sun Drought evolution and its impact on the crop yield in the North China Plain Journal of Hydrology 564, 984 (2018). [↩]
- M. Iqbal, R. Khan, A. Singh, P. Poór Plant hormones in crop improvement Elsevier Inc. (2023). [↩]
- W. Mo, X. Zheng, Q. Shi, X. Zhao, X. Chen, Z. Yang, Z. Zuo Unveiling the crucial roles of abscisic acid in plant physiology: implications for enhancing stress tolerance and productivity Front Plant Science 21(15), 1437184(2024). [↩] [↩]
- J. Wang, D. Wang, M. Zhu, F. Li Exogenous 6-benzyladenine improves waterlogging tolerance in maize seedlings by mitigating oxidative stress and upregulating the Ascorbate-Glutathione cycle Frontiers in Plant Science 12, 680376 (2021). [↩]
- R. Castro-Camba, C. Sánchez, N. Vidal, J. M. Vielba Plant development and crop yield: The Role of Gibberellins Plants (Basel) 11(19), 2650(2022). [↩] [↩] [↩]
- Revista Cultivar Plant Health 2,4-D https://revistacultivar.com/plant-health/2-4-d. [↩] [↩]
- J. Wang, D. Wang, M. Zhu, F. Li Exogenous 6-benzyladenine improves waterlogging tolerance in maize seedlings by mitigating oxidative stress and upregulating the Ascorbate-Glutathione cycle Frontiers in Plant Science 12, 680376 (2021). [↩]
- The Arabidopsis genome initiative Analysis of the genome sequence of the flowering plant Arabidopsis thaliana Nature, 408, 796 (2000). [↩]
- D. Meyre, A. Leonardi, G. Brisson, N. Vartanian Drought-adaptive mechanisms involved in the escape / tolerance strategies of Arabidopsis Landsberg erecta and Columbia ecotypes and their F1 reciprocal progeny J Plant Physiol. 158, 1145 (2001). [↩]
- J. Xie, L. Zhang The current situation and prospects of the development, industrialization and application of MicroCT China Medical Device Information 2 (2016). [↩]
- P. E. Verslues, E. A. Bray Role of abscisic acid (ABA) and Arabidopsis thaliana ABA-insensitive loci in low water potential-induced ABA and proline accumulation J. Experimental Botany 57 (1), 201 (2006). [↩]
- E. M. Savelieva, V. E. Oslovsky, D. S. Karlov, N. N. Kurochkin, I. A. Getman, S. N. Lomin, G. V. Sidorov, S. N. Mikhailov, D. I. Osolodkin, G. A. Romanov Cytokinin activity of N6-benzyladenine derivatives assayed by interaction with the receptors in planta, in vitro, and in silico Phytochemistry 149, 161(2018). [↩]
- D. dos S. Costa, C. C. A. Silva, A. J. R. D. Silva, N. Albarello Residual 2,4-D in plant tissue culture discarded media: a neglected source of environmental pollution Brazilian J. Development 6(5), 30046 (2020). [↩]
- M. Mahmoody, M. Noori1 Effect of gibberellic acid on growth and development plants and its relationship with abiotic stress IJFAS ISSN 2322-4134 (2014). [↩]
- Z. Fu, F. Wang, X. Bo Comparison of PEG6000 and D-mannitol simulated drought stress Anhui Agriculture Science Bulletin 25(10), 19 (2019). [↩]
- C. He, X. Chen, H. Huang, L. Xu Reprogramming of H3K27me3 is critical for acquisition of pluripotency from cultured Arabidopsis tissues PLoS Genet 8 (8), e1002911 (2012). [↩]
- S. Zhao, Y. Wu, G. Xia Introduction of a simple and effective method for plant total RNA isolation Hereditas 24(3), 337(2002). [↩]
- H. Yu, S. Peng, Y. Xie, Y. Hu Study on Improvement of RNA isolating reagent kit—TRIZOL Food Science 11, 39 (2005). [↩]
- C. Zhang, S. Xu, X. Huang A novel and convenient relative quantitative method of fluorescence real time RT-PCR Assay Progress In Biochemistry and Biophysics 9, 883(2005). [↩]
- Q. Li, W. Jiang, Z. Jiang, W. Du, J. Song, Z. Qiang, B. Zhang, Y. Pang, Y. Wang Transcriptome and functional analyses reveal ERF053 from Medicago falcata as key regulator in drought resistances Frontier Plant Science13, 995754 (2022). [↩]
- Micro Photonic Inc. What is Micro-CT? An introduction https://www.microphotonics.com/what-is-micro-ct-an-introduction/. [↩]
- T. Sun, F. Gubler Molecular mechanism of Gibberellin Signaling in Plants Annu. Rev. Plant Bio 55, 197 (2024). [↩]
- T. Regnault, J. Davière, M. Wild, L. Sakvarelidze-Achard, D. Heintz, E. C. Bergua, I. L. Diaz, F. Gong, P. Hedden, P. Achard The gibberellin precursor GA12 acts as a long-distance growth signal in Arabidopsis Nature Plants 1, 15073 (2015). [↩]
- L. Barboza, S. Effgen, C. Alonso-Blanco, R. Kooke, J. J. B. Keurentjes, M. Koornneef, R. Alcázar Arabidopsis semidwarfs evolved from independent mutations in GA20ox1, ortholog to green revolution dwarf alleles in rice and barley PNAS 110 (39) 15818 (2013). [↩]
- NASA Earth Observatory World of change: global temperatures https://earthobservatory.nasa.gov/world-of-change/global-temperatures. [↩]
- F. Shah, A. A. Bajwa, U. Nazir, S. A. Anjum, A. Farooq, A. Zohaib, S. Sadia, W. Nasim, S. Adkins, S. Saud, M. Z. Ihsan, H. Alharby, C. Wu, D. Wang, J. Huang Crop production under drought and mild heat stress: plant responses and management options Frontier Plant Science 8, 1147 (2017). [↩]
- M. Beniston Climate change and its impacts: growing stress factors for human societies International Review of the Red Cross 92, 879 (2010). [↩]
- H. Claeys, S. V. Landeghem, M. Dubois, K. Maleus, D. Inze What is stress? Dose-response effects in commonly used in vitro stress assays Plant Physiology 165(2), 519 (2014). [↩]
- P. Bharath, S. Gahir, A. S. Raghavendra Abscisic acid-induced stomatal closure: an important component of plant defense against abiotic and biotic stress Frontier Plant Science Section Plant Pathogen Interactions 12, 615114 (2021). [↩]
- R. Miao, W. Yuan, Y. Wang, I. Garcia-Maquilon, X. dang, Y. Li, J. Zhang, Y. Zhu, P. L. Rodriguez, W. Xu Low ABA concentration promotes root growth and hydrotropism through relief of ABA INSENSITIVE 1-mediated inhibition of plasma membrane H+-ATPase 2 Science advances 7(12), 4113 (2021). [↩]
- B. P. Brookbank, J. Patel, S. Gazzarrini, E. Nambara Role of Basal ABA in plant growth and development Genes (Basel) 12(12), 1936 (2021). [↩]
- X. Li, L. Chen, B. G. Forde, W. J. Davies The biphasic root growth response to abscisic acid in Arabidopsis involves interaction with ethylene and auxin signalling pathways Front Plant Sci. 25 (8), 1493 (2017). [↩]
- G. Wang, T. Xie, L. Fu, S. Qu, J. Li Research on the Cymbidium tortisepalum var. longibracteatum growth and non-tube rapid propagation based on response surface methodology Phyton – International Journal of Experimental Botany 94(3), 953 (2025). [↩]
- V. V. Butova, T. V. Bauer, V. A. Polyakov, V. D. Rajput, T. M. Minkina Analyzing the benefits and limitations of advanced 2,4-D herbicide delivery systems Crop Protection 184, 106865 (2024). [↩]
- S. Salazar-Cerezo, N. Martínez-Montiel, J. García-Sánchez, R. Pérez-y-Terrón, R. D. Martínez-Contreras Gibberellin biosynthesis and metabolism: A convergent route for plants, fungi and bacteria Microbiological Research 208, 85 (2018). [↩]