Abstract
Chronic, iatrogenic movement disorders such as Tardive Dyskinesia (TD) have been known to arise with the usage of both 1st and 2nd generation antipsychotic agents during the long-term treatment of schizophrenia (SCZ) and bipolar disorder. However, the underlying mechanisms by which these extrapyramidal symptoms (EPS) are produced have not been conclusively determined. Thus, this meta-analysis aims to investigate the effect of antipsychotic-induced reductions in dopaminergic neurotransmission on blood-based oxidative stress values, consequently assessing the potential role of oxidative stress in the development of movement disorders. 894 studies on PubMed were screened for data pertaining to the effect of 1st and 2nd generation antipsychotics on common enzymatic components of the antioxidant defense system as well as metabolites of oxidative stress induced lipid peroxidation. Studies were filtered by specific exclusion and inclusion criteria to maintain data accuracy. A total of 13 studies met the inclusion criteria, in which a total of 1627 patients’ data was obtained and analyzed. A random-effects model was used for aggregate data analysis, and subgroup analyses were performed. At alpha (α) = 0.05, malondialdehyde (MDA) and glutathione peroxidase (GPx) had statistically significant one-tailed p-values, whereas superoxide dismutase (SOD) did not. SOD and GPx had moderate to high negative effect sizes; MDA had a high positive effect size (in which a positive effect size indicates an increase in enzyme activity). These results indicate that antipsychotics do cause a statistically significant degree of change in the blood-based levels of some oxidative stress biomarkers, indicating that oxidative stress may play some role in the development of movement disorders.
Keywords: schizophrenia; oxidative stress; antioxidants; antipsychotics; tardive dyskinesia; dopamine neurotransmission; data extraction; meta-analysis
Introduction
Antipsychotic medications are the primary treatment for schizophrenia (SCZ), a chronic mental disorder characterized by hallucinations, delusions, and impaired cognition1. Although the precise etiology of SCZ has not been conclusively determined, one explanation of its origin is the hyperactivity of dopaminergic neurons in certain pathways of the brain1. First-generation antipsychotic agents, also known as typical antipsychotics, aim to address this by inhibiting dopaminergic neurotransmission. Second-generation antipsychotic agents—atypical antipsychotics—inhibit dopaminergic neurotransmission to a lesser extent, while also altering the transmission of other neurotransmitters, typically 5-hydroxytryptamine (serotonin) along with other noradrenergic, cholinergic, and histamine-binding receptors2.
While both typical and atypical antipsychotics effectively reduce psychotic symptoms, long term usage of either has been shown to correlate with the development of movement disorders. Such disorders are characterized by chronic extrapyramidal symptoms (EPS), the most significant of which being excessive involuntary and/or repetitive movement or paucity of voluntary movement3. Despite extensive research on antipsychotic function, there is an absence of research pertaining to the specific biological mechanisms that prompt the development of EPS. Numerous hypotheses to bridge these uncertainties have been proposed, including the hypersensitivity of dopamine receptors, neuronal degeneration, and maladaptive synaptic plasticity4.
This study aims to investigate one potential hypothesis by which these movement disorders and EPS develop: oxidative stress induced ferroptosis as a consequence of dopaminergic hypersensitization in the basal ganglia, a brain region containing dopamine pathways that govern movement. Oxidative stress is a phenomenon characterized by an imbalance between reactive oxygen species (ROS) and the antioxidant defense system’s capacity to neutralize them in the body5. Though ROS in certain quantities are vital in cell signaling and inflammatory responses, their systemic excess production can oxidize cellular components, causing cellular dysfunction and death5. Basal ganglia-specific oxidative damage induced by antipsychotic use, therefore, is one possible explanation for the pathogenesis of TD, as damage to this region may produce similar motor dysfunction6.
To validate the first part of this hypothesis, the scope of this study is to determine if antipsychotic use produces oxidative stress by measuring the changes in associated biomarkers. This is done by collecting and analyzing data about the influence of antipsychotics on metabolites of lipid peroxidation and common enzymes with antioxidant properties. Malondialdehyde (MDA), glutathione peroxidase (GPx), and superoxide dismutase (SOD) levels were measured for this reason. MDA is a product of lipid peroxidation (one common manner of oxidative stress induced cell death) commonly used to assess oxidative stress. SOD and GPx are two of the primary components of the antioxidant defense system, neutralizing free radicals by different, but crucial methods7. These three specific biomarkers were selected also because they are the most commonly reported enzymes in measuring oxidative stress in humans.
This study does not establish causation; it validates a singular mechanistic link in the whole proposed explanation of TD development. If such a relationship between antipsychotic usage and oxidative stress exists, it helps support the aforementioned explanation, contributing to the future development of novel interventions and targeted treatment methods. Intervention strategies might include the formation of antioxidant enzyme regimens or dietary/lifestyle change recommendations to address excess ROS, potentially reducing or preventing TD and other movement disorder symptoms in patients undergoing long-term treatment. Lastly, understanding the pathophysiology of antipsychotic-induced movement disorders could not only contribute to the mitigation of EPS, but also provide valuable insights into the mechanisms of other movement disorders like dopamine-responsive dystonia or Parkinson’s disease.
Methodology
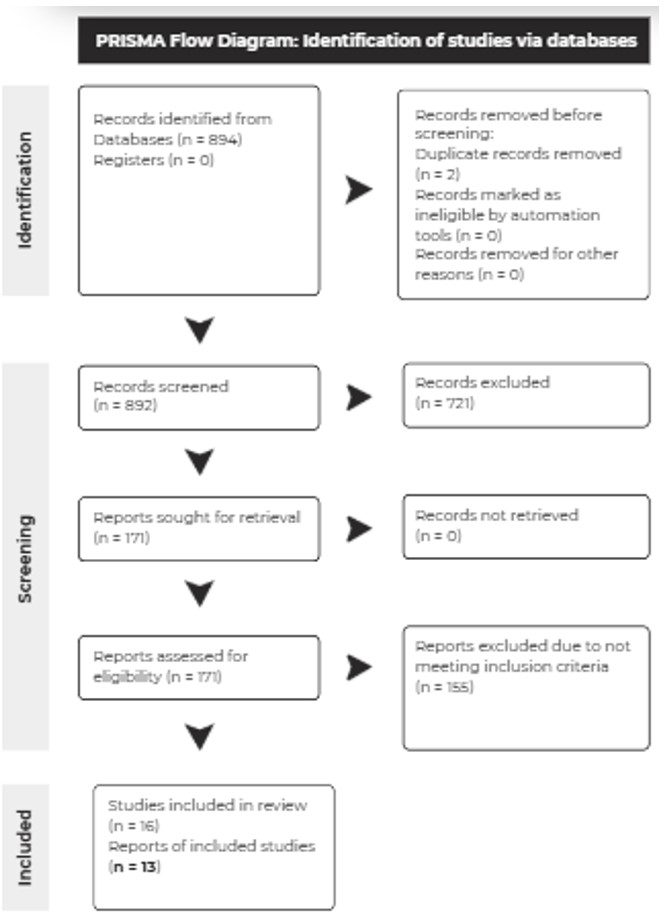
Articles from the PubMed database were searched based on specific search criteria and 894 were screened based on their title and abstract (for pertinence). Upon assessing congruency to the inclusion/exclusion criteria, they were selected for secondary review. Then, they were searched through for relevant and easily extractable data. During the literature search, studies with non-human subjects, duplicate publications, meta-analyses, case studies/reports, and non-English texts were excluded. The inclusion criteria included clinical trials evaluating the effect of typical (1st generation), atypical (2nd generation), or unspecified antipsychotic regimens on the levels of either biomarkers of oxidative stress (malondialdehyde in nanomoles/milliliter) or enzymatic components of the antioxidant defense system (superoxide dismutase and glutathione peroxidase in units/milliliter) in SCZ patients. A total of 13 studies with drug-naïve first episodic psychosis patients and chronically antipsychotic-treated SCZ patients were incorporated, with subgroup analyses performed. Relevant data values, namely levels of antioxidant enzymes and oxidative stress biomarkers, were converted to uniform units if possible and consolidated. If values were stratified based on gender, weight gain, or any other factors, they were compounded with the appropriate statistical procedures (combined mean and combined standard deviation formulas). A random-effects model was used due to high between-study heterogeneity, in workbook 3 of the Meta-Essentials software. Control group data and post-treatment experimental group data were assigned to groups 1 and 2 respectively within the workbook. To assess the quality of the incorporated studies, a funnel plot was graphed in order to detect publication bias; Cochran’s Q test and the I2 statistics were calculated to determine between-study heterogeneity.
Results
13 out of 894 studies met the inclusion criteria detailed in the methodology. Data pertaining to the control values and antipsychotic post-treatment values of superoxide dismutase (SOD), glutathione peroxidase (GPx), and malondialdehyde (MDA) was extracted. All data were converted to uniform units of u/ml for SOD and GPx, with units of nmol/ml for MDA. The probabilities of the experimental values occurring by random chance were lower than the significance value (α = 0.05) for 2 out of 3 of the variables; the one-tailed p values (hypothesis indicates directional changes) were: MDA (p = 0.046), and GPx (p = 0.006), SOD (p = 0.102). Hedge’s g effect sizes were calculated for each biomarker, with the following results: MDA (0.83 ± 0.49), GPx (-0.72 ± 0.29), and SOD (-0.57 ± 0.45). Subgroup analyses for drug-naïve first episode psychosis patients (AA) were performed, with the following results: MDA (0.58), GPx (-0.62), and SOD (0.09). Subgroup analyses for chronically treated SCZ patients (BB) were performed as well, with the following results: MDA (1.22), GPx (-0.9), and SOD (-1.24). Subgroup analyses and effect size data is illustrated in the table below.
The following 13 studies were incorporated in data analysis: Bai et al., Bošković et al., Chien et al., Gama et al., Khan et al., Liu et al., Li et al., M. Al-chalabi et al., Sarandol 1 et al., Sarandol 2 et al., Tsai et al., Zhang 1 et al., Zhang 2 et al.8,9,10,11,12,13,14,15,16,17,18,19,20
Discussion
Statistically significant differences were observed for MDA and GPx, but not SOD. Furthermore, the µ values for SOD and GPx were lower than the mean control values, with effect sizes of -0.72 and -0.57 respectively, indicating that antipsychotics correlate with reductions in the presence of these antioxidant enzymes in hemoglobin. Moreover, the µ value for MDA was greater than the mean control value; the increase of lipid peroxidation byproducts in the bloodstream indicates that antipsychotic usage correlates with oxidative stress-mediated cell apoptosis. MDA’s effect size substantiates, an effect size of 0.83 demonstrates a strong increasing effect that increases MDA levels.
However, some limitations do exist within the framework of the study. There may exist a potential bias in the sample of patients, as the inclusion criteria for articles written in English excludes studies and therefore patients based in non-English speaking countries. Furthermore, all studies included in the meta-analysis have different control variables: inherently different treatment timeframes and dose sizes, so the consolidated data’s accuracy decreases. Lastly, the analysis does not stratify results by drug type (which have distinct mechanisms of action), due to the dearth of existing and relevant data available for analysis. These confounders do impair the quality of the results.
Some biological explanations exist that justify this study’s findings, but current explanations are theoretical in nature and presume the operation of certain biological processes, lacking the scientific rigor and experimental evidence necessary to arrive at a conclusion. Thus, further studies should be conducted in order to conclusively determine the relationship between antipsychotic use and movement disorder development and how other presently unaccounted variables influence it. This study does indirectly hold patient implications: gaining additional insights into oxidative stress hypothesis of TD development would allow for the future creation of strategies to mitigate the symptoms of TD, significantly improving patient outcomes while retaining the benefits of antipsychotic usage.
Conclusion
In summary, this meta-analysis and its findings indicate that antipsychotic use is correlated with changes in oxidative stress biomarkers, confirming the guiding research conjecture. It helps validate a mechanism in the oxidative stress hypothesis of TD etiology, helping elucidate the convoluted relationship between antipsychotic agents and movement disorder development. The study’s small sample size and inability to stratify analyses by drug type, dose size, and other confounders is its primary limitation, but it does build upon current knowledge on the mechanisms of movement disorder development. The next step to slowing and eventually preventing movement disorders is the organization of nuanced, scrupulous research and clinical trials that bridge molecular mechanisms with clinical observations. By doing so, we can slowly begin crafting targeted therapeutic regimens that improve patient health outcomes now and in the future.
References
- B. Ralf, S. Arthur, W. Rainer, B. Hendrik, B. Hans-Gert, S. Johann, B. Bernhard, B. Katharina, J. Zbigniew, K. Jaliya, H. Maciej. The role of dopamine in schizophrenia from a neurobiological and evolutionary perspective: old fashioned, but still in vogue. Front Psychiatry. (2014). [↩] [↩]
- L. Peng, L. S. Gretchen, E. V. Kimberly. Dopamine targeting drugs for the treat of schizophrenia: past, present, and future. Curr Top Med Chem. (2016). [↩]
- M. Maju, G. Sylvia, A. Babatunde, G. Vinu, M. Manu, B. Biju. Antipsychotic-induced movement disorders: evaluation and treatment. Psychiatry (Edgemont). (2005). [↩]
- T. T. James, J. E. Mark, B. Kailash. Tardive dyskinesia is caused by maladaptive synaptic plasticity: a hypothesis. Movement Disorders. (2012). [↩]
- Pizzino, Gabriele, et al. “Oxidative Stress: Harms and Benefits for Human Health.” Oxidative Medicine and Cellular Longevity, edited by Victor M., vol. 2017, no. 1, Wiley, Jan. 2017. Crossref, doi:10.1155/2017/8416763. [↩] [↩]
- Dash, Umesh Chandra, et al. “Oxidative Stress and Inflammation in the Pathogenesis of Neurological Disorders: Mechanisms and Implications.” Acta Pharmaceutica Sinica B, vol. 15, no. 1, Elsevier BV, Jan. 2025, pp. 15–34. Crossref, doi:10.1016/j.apsb.2024.10.004. [↩]
- Ighodaro, O. M., & Akinloye, O. A. (2018). First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid. Alexandria Journal of Medicine, 54(4), 287–293. https://doi.org/10.1016/j.ajme.2017.09.001 [↩]
- Bai, Zhi-Le et al. “Serum Oxidative Stress Marker Levels in Unmedicated and Medicated Patients with Schizophrenia” Journal of molecular neuroscience: MN vol. 66,3 (2018): 428-436. doi:10.1007/s12031-018-1165-4 [↩]
- Bošković, Marija et al. “Oxidative stress in schizophrenia patients treated with long-acting haloperidol decanoate.” Psychiatry research vol. 210,3 (2013): 761-8. doi:10.1016/j.psychres.2013.08.035 [↩]
- Chien, Yi-Ling et al. “Clinical implications of oxidative stress in schizophrenia: Acute relapse and chronic stable phase” Progress in Neuro-Psychopharmacology and Biological Psychiatry vol. 99 (2020): 0278-5846. https://doi.org/10.1016/j.pnpbp.2020.109868. [↩]
- Gama, Clarissa Severino et al. “Elevated serum superoxide dismutase and thiobarbituric acid reactive substances in schizophrenia: A study of patients treated with haloperidol or clozapine” Progress in Neuro-Psychopharmacology and Biological Psychiatry vol. 30,2 (2006): 512-515. https://doi.org/10.1016/j.pnpbp.2005.11.009. [↩]
- Khan, Fatema Zerin. “Effect of Olanzapine and Risperidone on Oxidative Stress in Schizophrenia Patients.” IBBJ, International Biological and Biomedical Journal, 8 Apr. 2018, ibbj.org/article-1-170-en.pdf. [↩]
- Liu, Haixia et al. “Antioxidant Enzymes and Weight Gain in Drug-naive First-episode Schizophrenia Patients Treated with Risperidone for 12 Weeks: A Prospective Longitudinal Study.” Current neuropharmacology vol. 20,9 (2022): 1774-1782. doi:10.2174/1570159X19666210920090547 [↩]
- Li, Xi Rong et al. “Altered Antioxidant Defenses in Drug-Naive First Episode Patients with Schizophrenia Are Associated with Poor Treatment Response to Risperidone: 12-Week Results from a Prospective Longitudinal Study.” Neurotherapeutics : the journal of the American Society for Experimental NeuroTherapeutics vol. 18,2 (2021): 1316-1324. doi:10.1007/s13311-021-01036-3 [↩]
- M. Al-chalabi, Basil et al. “Potential Effect of Olanzapine on Total Antioxidant Status and Lipid Peroxidation in Schizophrenic Patients.” Neuropsychobiology 1 March 2009; 59 (1): 8–11. https://doi.org/10.1159/000202823 [↩]
- Sarandol, Alsi et al. “Oxidative–antioxidative systems and their relation with serum S100 B levels in patients with schizophrenia: Effects of short term antipsychotic treatment” Progress in Neuro-Psychopharmacology and Biological Psychiatry vol. 31,6 (2007): 1164-1169. https://doi.org/10.1016/j.pnpbp.2007.03.008 [↩]
- Sarandol, Alsi et al. “First-episode psychosis is associated with oxidative stress: Effects of short-term antipsychotic treatment” Psychiatry Clin Neurosci, vol 69 (2015): 699-707. https://doi.org/10.1111/pcn.12333 [↩]
- Tsai, Meng-Chang et al. “Changes in oxidative stress markers in patients with schizophrenia: the effect of antipsychotic drugs.” Psychiatry research vol. 209,3 (2013): 284-90. doi:10.1016/j.psychres.2013.01.023 [↩]
- Zhang, Xiang Yang et al. “Antioxidant enzymes and lipid peroxidation in different forms of schizophrenia treated with typical and atypical antipsychotics.” Schizophrenia research vol. 81,2-3 (2006): 291-300. doi:10.1016/j.schres.2005.10.011 [↩]
- Zhang, Xiang Yang, et al. “Effects of Risperidone and Haloperidol on Superoxide Dismutase and Nitric Oxide in Schizophrenia.” Neuropharmacology, vol. 62, no. 5–6, Elsevier BV, Apr. 2012, pp. 1928–1934. Crossref,doi:10.1016/j.neuropharm.2011.12.014. [↩]