Abstract
Parkinson’s disease (PD) is a progressive neurodegenerative disorder affecting over 10 million people worldwide, primarily caused by the loss of dopamine-producing neurons in the brain. This neuronal loss leads to significant impairments in motor control and cognitive function. This review aims to summarize recent research on induced pluripotent stem cell-derived dopaminergic neurons, transplantation outcomes, and emerging tissue engineering strategies for Parkinson’s disease. Recent advances in regenerative medicine have focused on the use of stem cell–derived dopaminergic neurons as a novel therapeutic approach. These neurons are generated through the directed differentiation of induced pluripotent stem cells (iPSCs), which can be reprogrammed from adult somatic cells and guided to develop into mature midbrain dopaminergic neurons that express key markers such as TH and Nurr1. In vitro studies demonstrate that these neurons exhibit dopamine production and electrophysiological activity comparable to native neurons. When implanted in vivo, stem cell–derived neurons successfully engraft, integrate with host tissue, and improve both dopamine levels and behavioral outcomes, highlighting their therapeutic potential. To address the limitations of current cell injection techniques, biofabrication and bioprinting methods are being developed to engineer structured, implantable neural tissues that better support cell survival and integration. While challenges such as incomplete neuronal maturation and large-scale tissue manufacturing remain, ongoing research is working to overcome these barriers. If successful, this approach could not only transform treatments for PD, but also lay the foundation for regenerative therapies targeting a wide range of neurological diseases. Overall, the current findings suggest that while stem cell-based approaches represent a promising, yet still experimental, direction for the future treatment of PD and other neurological disorders.
Introduction
Parkinson’s disease (PD) is a progressive neurodegenerative disorder that impacts over 10 million people worldwide, making it one of the most common neurological disorders1. It is characterized primarily by the loss of dopamine-producing neurons in a specific brain region called the substantia nigra. These neurons mainly produce and utilize dopamine, a neurotransmitter that plays a crucial role in regulating movement, cognitive control, and emotional regulation. A key pathological feature of PD is the presence of Lewy bodies, abnormal aggregates of the protein alpha-synuclein, in affected neurons. This neuronal degeneration leads to motor symptoms such as tremors, rigidity, and bradykinesia, as well as non-motor symptoms like sleep disturbances and cognitive decline.
Currently, treatment options for PD include medications such as dopamine precursors and dopamine agonists, as well as surgical interventions like deep brain stimulation2 Dopamine precursors are turned into dopamine through chemical reactions in the brain, whereas dopamine agonists bind to dopamine receptors to mimic the effects of dopamine. Conversely, deep brain stimulation works by implanting an electric pulse generator near the brain, releasing electric shocks in order to relieve some symptoms of PD. While these therapies can alleviate symptoms, they do not stop disease progression or reverse neuronal loss, and their efficacy may diminish over time2. These limitations have driven interest in regenerative medicine approaches aimed at replacing lost dopaminergic neurons rather than solely managing symptoms3.
Early regenerative efforts involved the transplantation of fetal ventral mesencephalon (fVM) tissue, which demonstrated that grafted dopaminergic neurons could survive and provide motor benefit in some patients. However, these trials produced highly variable outcomes and, in some cases, resulted in graft-induced dyskinesia, a severe complication characterized by involuntary movements. Ethical concerns, the limited availability of tissues, and inconsistent efficacy ultimately restricted the scalability of fetal tissue-based approaches, highlighting the need for more standardized and ethically viable cell sources4,5.
Stem cell-based therapies offer the potential to generate patient-specific dopamine-producing neurons, which could replace those lost in PD and restore function. In parallel, advances in tissue engineering have introduced the possibility of biomanufacturing structured neural tissue constructs that may better support long-term graft survival and integration than isolated cell injections6,7. Together, these strategies represent a promising but still experimental direction for addressing dopaminergic neuron loss in PD3.
Stem cell differentiation and maturation of dopamine-producing neurons
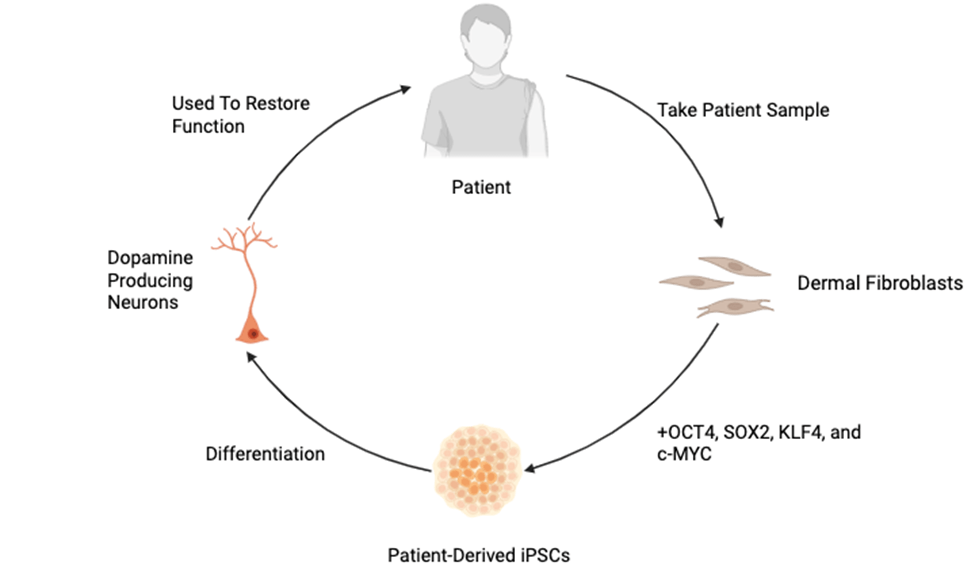
Current therapies for PD focus mainly on symptom management and do not address the underlying loss of dopaminergic neurons. These limitations have led to growing interest in regenerative strategies, particularly those involving stem cells. Induced pluripotent stem cells (iPSCs) are adult cells, most commonly dermal fibroblasts or blood cells, that have been genetically reprogrammed into an embryonic-like, pluripotent state through the upregulation of key transcription factors—OCT4, SOX2, KLF4, and c-MYC8. Through directed differentiation, scientists can guide iPSCs to become specific cell types, like midbrain dopaminergic neurons, by using carefully chosen signals and growth factors. This breakthrough allows scientists to generate almost any cell type in the body from a patient’s own cells, offering powerful tools for disease modeling and regenerative medicine.
A key application of iPSC technology for neurological diseases is their differentiation into dopamine-producing neurons, which are particularly relevant for studying, and ultimately treating, PD—a disorder caused by the degeneration of midbrain dopaminergic neurons (Figure 1). The process of directing the differentiation of iPSCs into neurons mimics early brain development and occurs in multiple stages: starting from pluripotent iPSCs (expressing OCT4 and NANOG), progressing to ectodermal cells (marked by PAX6), then forming neural rosettes and neural progenitor cells (expressing NESTIN), and finally differentiating into mature midbrain dopaminergic neurons identified by markers such as LMX1A, FOXA2, and tyrosine hydroxylase (TH)9.
The development from neural progenitor cells into mature midbrain dopaminergic neurons is critical because only mature neurons can effectively produce dopamine, integrate into brain circuits, and restore the motor functions impaired in PD. Patterning toward a ventral midbrain fate is typically achieved through sustained exposure to signaling molecules such as sonic hedgehog (SHH) and fibroblast growth factor 8 (FGF8), often over several days to weeks, with the duration and concentration of these cues playing a key role in determining cell identity9.
Markers like TH and Nurr1 help identify developing dopamine-producing neurons, with TH playing a key role in dopamine production.
Suboptimal patterning can result in heterogeneous cell populations containing off-target neuronal subtypes, which may compromise graft safety and efficacy. To address this issue, Doi et al. demonstrated that CORIN-based sorting of iPSC-derived dopaminergic progenitors improved graft purity and reduced tumorigenic risk10.
Beyond the presence of these essential markers, additional in vitro studies have assessed the electrophysiological activity of these iPSC-derived neurons, confirming proper neural activity and dopamine signaling11. To confirm the proper differentiation of iPSCs into neurons, scientists employ methods such as immunofluorescence staining to detect key proteins, biochemical assays to measure dopamine production, and electrophysiological techniques to assess whether the neurons are electrically active. Culturing these neurons typically involves growing them on a supportive extracellular matrix such as Matrigel, which promotes cell adhesion and development. However, even under extended culture conditions, many iPSC-derived neurons retain immature phenotypes, and neuronal maturation and dopamine production may plateau over time11,9.
To overcome these limitations, researchers are exploring advanced systems such as microfluidic platforms and brain-on-a-chip technologies12. These systems aim to better replicate the biomechanical and chemical microenvironment of the brain, potentially improving the maturation of neurons and their long-term viability, although their translational relevance remains under investigation12.
Implantation of dopamine-producing neurons for PD patients
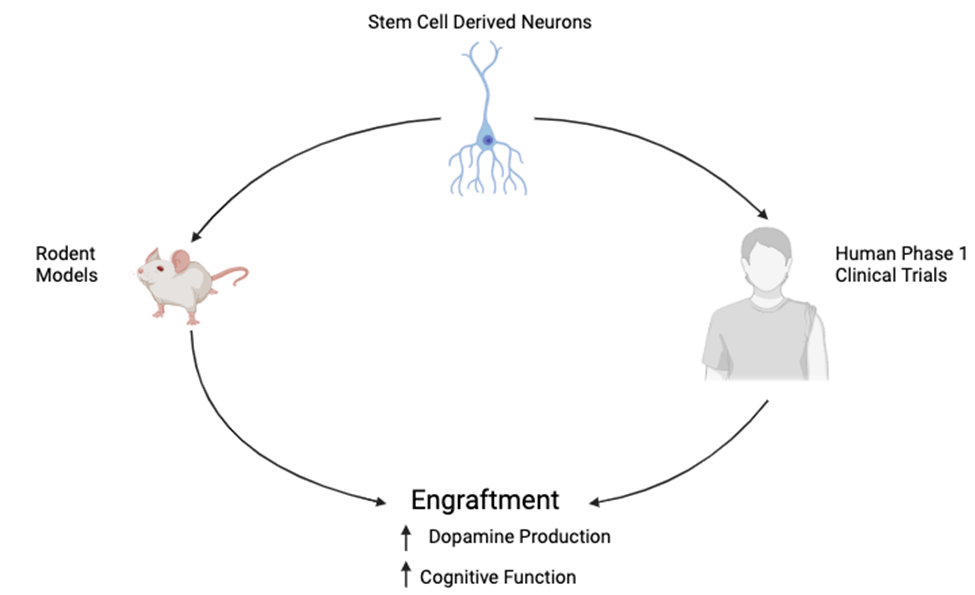
In vivo implantation of stem cell–derived neurons has emerged as one of the most promising strategies for treating the progressive loss of dopaminergic neurons observed in patients with PD13. Preclinical studies using rodent models of PD have demonstrated that human pluripotent stem cells can be differentiated into midbrain dopaminergic progenitors and successfully implanted into the striatum, where they survive, integrate, and restore dopamine production10. These neurons, often labeled with green fluorescent protein (GFP) to allow for in vivo tracking, have shown robust engraftment and synaptic integration with host tissue. In a widely cited study, GFP-labeled neurons implanted into 6-hydroxydopamine (6-OHDA)-lesioned rats not only survived for over 16 weeks but also released dopamine and significantly reduced motor deficits such as apomorphine-induced rotational behavior10.
The functional restoration observed in these models has been further validated by biochemical assays and behavioral testing14. However, the 6-OHDA model primarily reflects dopaminergic neuron loss and does not fully recapitulate the key features of human PD, including alpha-synuclein aggregation, progressive multisystem degeneration, or nonmotor symptoms14. Additionally, graft heterogeneity remains a challenge, as transplanted populations may contain mixed neuronal and non-neuronal cell types that could influence both safety and efficacy outcomes15,16.
Encouraged by these results and to see how they would translate to human models, several human clinical trials have recently progressed to early-phase testing. One notable trial involves bemdaneprocel, a therapy derived from human embryonic stem cells (hESCs), in which dopaminergic progenitors are surgically implanted into the putamen of PD patients17. Interim results from this Phase I trial indicated increased uptake of 18F-DOPA in the implanted regions—an imaging marker of dopaminergic activity—along with good safety and tolerability. In a separate study, intranasal administration of human neural stem cells (ANGE-S003) was tested in patients with moderate to advanced PD, leading to significant reductions in MDS-UPDRS scores over a 12-month follow-up, resulting in better cognitive and motor function without any major adverse effects18. These findings suggest that stem cell–based therapies may provide functional benefits even in the absence of direct intracerebral implantation (Figure 2), though the mechanisms behind this remain under investigation.
While these early trials offer optimism, they also underscore key limitations that must be addressed before stem cell therapies can be widely adopted. Current implants typically deliver 100,000 to 300,000 dopaminergic neurons per hemisphere, which may be sufficient for moderate PD but likely fall short in treating more advanced cases characterized by widespread neurodegeneration and cognitive decline19,18. Importantly, none of the current trials have demonstrated a reduction in underlying PD pathology such as Lewy bodies or alpha-synuclein aggregation, which are central to disease progression. Although these advances mark a critical step in positioning stem cell–based neural implantation as a core component of next-generation PD therapy, for broader and more sustained therapeutic impact, future strategies may require the development of larger, multi-regional neural grafts or tissue-engineered constructs capable of restoring complex neural circuits20.
Biomanufacturing of brain tissue for replacing regions of the brain
Creating functional lab-grown brain tissue using iPSCs offers a promising new direction for treating Parkinson’s disease. Early approaches towards utilizing stem cell-derived neurons for PD therapy often involved injecting large numbers of neural cells—between 100,000 and 300,000—directly into the brain. Poor cell retention, mechanical stress during injection, and lack of structural and biochemical support at the implantation site have been identified as key contributors to low graft survival21,22. As a result, researchers are increasingly focusing on engineering structured, implantable neural tissues that provide mechanical support and biochemical cues to enhance cell survival, maturation, and functional integration23,22.
A major aspect of making these tissues is choosing the right biomaterials—substances that help cells grow in the lab. These materials act as scaffolds, giving cells a supportive matrix to attach to and grow on. To support neural cells, especially those meant to become dopaminergic neurons (the type lost in Parkinson’s), the scaffold must closely mimic the environment of the brain. This includes matching the brain’s softness, which is ~10 kilopascals (kPa). Reported elastic moduli for brain tissue typically range from ~0.1 to 10 kPA depending on the brain region and measurement method, and neural cells are sensitive to deviations from this range23,24. Commonly used biomaterials like gelatin, matrigel, collagen, and gelatin methacrylate (GelMA) have been shown to support healthy neural growth. For example, GelMA can be adjusted to have the same stiffness as brain tissue, helping cells survive and function more naturally23. In their study, Banerjee and colleagues found that hydrogels with stiffness close to brain tissue significantly increased neural stem cell proliferation and differentiation. Softer gels promoted greater neurite outgrowth and higher expression of neuronal markers compared to stiffer gels, suggesting that matching brain-like mechanical properties encourages better neuronal maturation and network formation. These mechanical cues have also been shown to influence dopaminergic lineage commitment and synaptic development in stem cell-derived neurons25.
Bioprinting is an innovative technique that combines biomaterials and cells to create complex, custom-designed brain tissues by precisely placing cells and scaffolds in specific patterns. This allows researchers to replicate structures similar to real brain tissue, arranging iPSC-derived neurons and glial cells to enhance their interactions and connectivity (Figure 3). Bioprinting offers improved control over cell density, spatial organization, and scaffold architecture compared to traditional cell suspension approaches26,27. In the study by Gu et al. (2016), researchers successfully printed 3D neural mini-tissues using a gel-based bioink containing human neural stem cells26. The printed tissues showed high cell viability, maintained neural progenitor markers, and demonstrated early neural activity, including neurite outgrowth and synapse formation, indicating functional maturation27. Similarly, Lozano et al. (2015) used peptide-modified hydrogels to bioprint layered neural structures that supported the proliferation and differentiation of neural cells. Their printed constructs exhibited organized cell networks and robust neurite extension, mimicking the microarchitecture of brain tissue. Together, these studies highlight that bioprinting can produce living neural tissues with improved structural organization and functional properties compared to traditional cell culture methods. This precise control over tissue architecture opens the door for creating personalized implants tailored to a patient’s unique neural environment. However, these findings are limited to preclinical models, and no bioprinted neural constructs have yet been tested in human PD patients23.
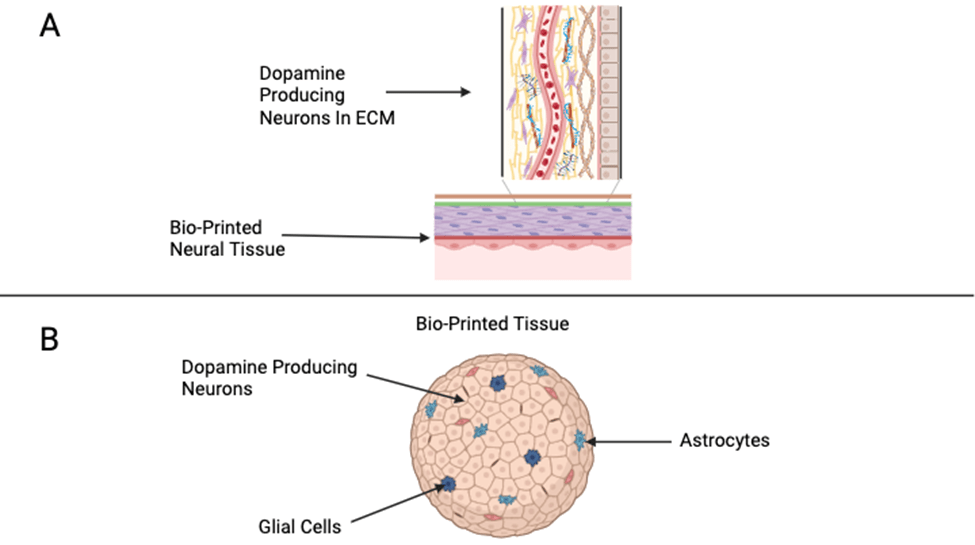
In the future, these bioengineered brain tissues could find applications for treating many other conditions beyond PD. For example, they could be used to help treat traumatic brain injuries, strokes, epilepsy, and other neurodegenerative diseases like Alzheimer’s disease. The combination of stem cells, advanced materials, and precise manufacturing techniques opens the door to new therapies that could repair or even replace damaged brain regions in a wide range of neurological disorders. Significant translational challenges remain, including the vascularization of larger constructs, long-term graft survival, immune capability, and reliable large-scale manufacturing21,5.
Limitations and Future Directions
A current limitation of stem cell–based therapies for PD is that stem cell–derived neurons often remain immature and lack the full functionality of adult dopaminergic neurons. This limits their ability to integrate into the brain and sustain long-term therapeutic benefits. Enhancing neuronal maturation remains a key focus, with researchers refining differentiation protocols and in vitro culture systems to better mimic the natural brain environment.
Incomplete maturation not only affects functional integration, but it may also increase safety risks, as partially differentiated or heterogeneous graft populations have been associated with variable efficacy and potential adverse outcomes in transplantation studies28,29.
Additionally, differentiating these neurons at scale is costly and technically complex, making large-scale, reproducible manufacturing a major barrier to clinical use. To address this, bioreactors are being developed to support the scalable differentiation of neurons in controlled environments. At the same time, bioprinting technologies are improving the consistency and reproducibility of tissue fabrication. Together, these innovations could make high-quality neural implants more accessible and effective for treating Parkinson’s and other neurological disorders.
However, increased scale and manufacturing complexity also impose stricter regulatory requirements for quality control, batch consistency, and traceability, which are particularly stringent for pluripotent stem cell-derived products.
Beyond biological and engineering challenges, safety, ethical, and regulatory considerations represent central limitations for the clinical translation of pluripotent stem cell-based therapies for Parkinson’s disease. A major safety concern is the risk of tumor formation arising from residual undifferentiated pluripotent cells or off-target proliferative populations, necessitating rigorous purification strategies and long-term post-transplant monitoring28,30. Immune rejection remains a further challenge, particularly for allogeneic hESC- or iPSC-derived grafts, often requiring immunosuppression that introduces additional clinical risk31,4. Ethical debates surrounding the use of fetal tissue and embryonic stem cells have always influenced funding, regulation, and public acceptance of these therapies and continue to shape clinical trial design and approval pathways4,32. From a regulatory perspective, these therapies often require a constant influx of money, laboratory equipment, and human donors, leading to concerns regarding the long term viability for these sorts of treatments32,5.
Conclusion
As the limitations of current Parkinson’s disease treatments become increasingly evident, regenerative strategies are gaining momentum as potential disease-modifying therapies. Among these, stem cell-based approaches offer the ability to restore lost function by directly addressing the degeneration of dopaminergic neurons. Preclinical and early clinical studies have shown that transplanted neurons can survive, integrate, and produce measurable improvements in motor function, supporting the feasibility of this approach. However, the majority of supporting evidence comes from animal models and early-phase human trials that are primarily designed to evaluate feasibility and safety rather than long-term efficacy or disease modification31,4.
However, challenges remain. Many implanted neurons continue to exhibit immature phenotypes, and the scale of cell loss in advanced PD may exceed what current grafting strategies can address. Moreover, while symptomatic improvements have been observed, these interventions have yet to demonstrate a significant impact on the underlying pathophysiology, such as alpha-synuclein aggregation and Lewy body formation. Accordingly, current findings should be interpreted as evidence of potential symptomatic benefit in specific experimental contexts rather than proof of sustained disease-modifying effects28,31. To move beyond symptomatic relief and toward long-term disease modification, future efforts must focus on enhancing the functional maturation of grafted cells, improving their integration into host circuitry, and expanding the scale and complexity of engineered neural tissues. These biological advances must proceed alongside rigorous safety evaluation, standardized manufacturing protocols, and extended clinical follow-up to identify the delayed adverse effects, such as graft overgrowth or immune-mediated complications5.
The integration of stem cell-derived neurons into structured, biomanufactured tissue constructs represents a critical next step. By combining advances in cellular reprogramming, tissue engineering, and scalable manufacturing, the field is steadily moving toward the development of implantable neural systems capable of replacing not only isolated neurons but entire damaged regions. Such innovations mark a pivotal shift in the therapeutic landscape of neurodegenerative diseases, laying the groundwork for more personalized, durable, and effective treatments. At present, however, these approaches remain experimental and their translation into routine clinical practices will depend on resolving outstanding biological, ethical, and regulatory challenges associated with pluripotent stem cell-based therapies26.
References
- M.F. Mendez, R.C. Doss, & J.L. Taylor, The frequency and clinical characteristics of Parkinson’s disease in an ethnically diverse population. Journal of Neurology, 255(10), 1559–1564, 2008, https://doi.org/10.1007/s00415-008-0950-6. [↩]
- M.S.B. Faheem, M. Haroon-Ul-Rasheed, R. Aftab, Q. Faheem, F. Feroze, H.Q.U. Ain, & S. Samadi, The Next Frontier in Parkinson’s Disease Treatment: A Narrative Review of Innovative Neurosurgical and Gene Therapy Approaches. Health Science Reports, 8(9), e71197, 2025, https://doi.org/10.1002/hsr2.71197. [↩] [↩]
- T. Cardoso, A.F. Adler, & D. Agalliu, Stem cell transplantation for Parkinson’s disease: Current clinical trials and advances in neural grafting. Stem Cell Research & Therapy, 9(1), 299, 2018, https://doi.org/10.1186/s13287-018-1035-7. [↩] [↩]
- S. Moradi, H. Mahdizadeh, T. Šarić, et al. Research and therapy with induced pluripotent stem cells (iPSCs): social, legal, and ethical considerations. Stem Cell Research & Therapy, 10, 341, 2019, https://doi.org/10.1186/s13287-019-1455-y. [↩] [↩] [↩] [↩]
- V. Volarevic, B. Ljujic, et al. Ethical and safety issues of stem cell-based therapy. Journal of Biomedical Science, 25, 57, 2018, https://doi.org/10.1186/s12929-018-0471-z. [↩] [↩] [↩] [↩]
- S. Maheshwari, Y.M. Tan, & C. Wang, Engineering functional brain tissue from stem cells: Challenges and opportunities. Tissue Engineering Part B: Reviews, 30(3), 145–159, 2024, https://doi.org/10.1089/ten.teb.2024.0035. [↩]
- A. Castiglione, S. Lai, & E. Monni, Brain-on-a-chip models for neurodegenerative disease: A review of recent advances. Frontiers in Neuroscience, 16, 891233, 2022, https://doi.org/10.3389/fnins.2022.891233. [↩]
- K. Takahashi, K. Tanabe, M. Ohnuki, M. Narita, T. Ichisaka, K. Tomoda, & S. Yamanaka, Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell, 131(5), 861–872, 2007, https://doi.org/10.1016/j.cell.2007.11.019. [↩]
- S. Kriks, J. W. Shim, J. Piao, Y. M. Ganat, D. R. Wakeman, Z. Xie, … & L. Studer. Dopamine neurons derived from human ES cells efficiently engraft in animal models of Parkinson’s disease. Nature, 480(7378), 547–551, 2011, https://doi.org/10.1038/nature10648. [↩] [↩] [↩]
- T. Kikuchi, A. Morizane, & D. Doi. Human iPS cell-derived dopaminergic neurons function in a primate Parkinson’s disease model. Nature, 548(7669), 592–596, 2017, https://doi.org/10.1038/nature23664. [↩] [↩] [↩]
- S.C. Zhang, M. Wernig, Duncan, I. D., Brustle, O., & Thomson, J. A. In vitro differentiation of transplantable neural precursors from human embryonic stem cells. Nature Biotechnology, 19(12), 1129–1133, 2001, https://doi.org/10.1038/nbt1201-1129. [↩] [↩]
- Y. Zhang, C. Pak., Y. Han, H. Ahlenius, Z. Zhang,S. Chanda, … & T.C. Südhof. Rapid single-step induction of functional neurons from human pluripotent stem cells. Neuron, 78(5), 785–798, 2013, https://doi.org/10.1016/j.neuron.2013.05.029. [↩] [↩]
- R.A. Barker, J.Barrett, S.L.Mason, & A. Björklund. Fetal dopaminergic transplantation trials and the future of neural grafting in Parkinson’s disease. The Lancet Neurology, 12(1), 84–91, 2013, https://doi.org/10.1016/S1474-4422(12)70290-4. [↩]
- S. Grealish, A. Heuer, T. Cardoso, A. Kirkeby, & M. Parmar. Monosynaptic tracing of human stem cell-derived dopaminergic neurons in a rat Parkinson’s disease model. Stem Cell Reports, 5(5), 869–880, 2015, https://doi.org/10.1016/j.stemcr.2015.09.009. [↩] [↩]
- S. Grealish, E. Diguet, A. Kirkeby, B. Mattsson, A. Heuer, Y. Bramoulle, … & M. Parmar (2014). Human ESC-derived dopamine neurons show similar preclinical efficacy and potency to fetal neurons when grafted in a rat model of Parkinson’s disease. Cell Stem Cell, 15(5), 653–665. https://doi.org/10.1016/j.stem.2014.10.003 [↩]
- D. Doi, A. Morizane, & T. Kikuchi. Isolation of human induced pluripotent stem cell-derived dopaminergic progenitors by cell sorting for successful transplantation. Stem Cell Reports, 2(3), 337–350, 2014, https://doi.org/10.1016/j.stemcr.2014.01.003. [↩]
- J. Takahashi, K. Tanabe, & S. Yamanaka. iPS cell-based therapy for Parkinson’s disease: A Kyoto trial. NPJ Regenerative Medicine, 5(1), 15, 2020, https://doi.org/10.1038/s41536-020-0094-1. [↩]
- S. Zhang, X. Ma, & Y. Liu. Intranasal delivery of human neural stem cells improves motor and cognitive functions in Parkinson’s disease patients. Stem Cells Translational Medicine, 9(12), 1605–1616, 2020, https://doi.org/10.1002/sctm.20-0198. [↩] [↩]
- J. Takahashi, K. Tanabe, & S. Yamanaka, iPS cell-based therapy for Parkinson’s disease: A Kyoto trial. NPJ Regenerative Medicine, 5(1), 15, 2020, https://doi.org/10.1038/s41536-020-0094-1. [↩]
- R. A. Barker, J. Barrett, S.L. Mason, & A. Björklund, Fetal dopaminergic transplantation trials and the future of neural grafting in Parkinson’s disease. The Lancet Neurology, 12(1), 84–91, 2013, https://doi.org/10.1016/S1474-4422(12)70290-4. [↩]
- S. Maheshwari, Y. M. Tan, & C. Wang. Engineering functional brain tissue from stem cells: Challenges and opportunities. Tissue Engineering Part B: Reviews, 30(3), 145–159, 2024, https://doi.org/10.1089/ten.teb.2024.0035 [↩] [↩]
- A. Banerjee, M. Arha, S. Choudhary, R. S. Ashton, S. R. Bhatia, D. V. Schaffer, & R. S. Kane. The influence of hydrogel modulus on the proliferation and differentiation of encapsulated neural stem cells. Biomaterials, 30(27), 4695–4699, 2009, https://doi.org/10.1016/j.biomaterials.2009.05.002 [↩] [↩]
- S. Maheshwari, Y. M. Tan, & C. Wang. Engineering functional brain tissue from stem cells: Challenges and opportunities. Tissue Engineering Part B: Reviews, 30(3), 145–159, 2024, https://doi.org/10.1089/ten.teb.2024.0035 [↩] [↩] [↩] [↩]
- Z. Zhang, X. Wang, & S. Wang. Safety and efficacy of cell transplantation on improving motor symptoms in patients with Parkinson’s disease: A meta-analysis. Frontiers in Human Neuroscience, 16, 849069, 2022, https://doi.org/10.3389/fnhum.2022.849069 [↩]
- Z. Zhang, X. Wang, & S. Wang. Safety and efficacy of cell transplantation on improving motor symptoms in patients with Parkinson’s disease: A meta-analysis. Frontiers in Human Neuroscience, 16, 849069, 2022, https://doi.org/10.3389/fnhum.2022.849069 [↩]
- Q. Gu, E. Tomaskovic-Crook, R. Lozano, Y. Chen, R. M. Kapsa, Q. Zhou, & J. M. Crook. Functional 3D neural mini-tissues from printed gel-based bioink and human neural stem cells. Advanced Healthcare Materials, 5(12), 1429–1438, 2016, https://doi.org/10.1002/adhm.201600095 [↩] [↩] [↩]
- R. Lozano, L. Stevens, B. C. Thompson, K. J. Gilmore, R. Gorkin, E. M. Stewart, & G. G. Wallace. 3D printing of layered brain-like structures using peptide-modified gellan gum hydrogels. Biomacromolecules, 16(2), 587–595, 2015, https://doi.org/10.1021/bm501930w [↩] [↩]
- S. Kriks, J. W. Shim, J. Piao, Y. M. Ganat, D. R. Wakeman, Z. Xie, … L. Studer. Dopamine neurons derived from human ES cells efficiently engraft in animal models of Parkinson’s disease. Nature. Vol. 480, no. 7378, pp. 547–551, 2011, https://doi.org/10.1038/nature10648. [↩] [↩] [↩]
- H. E. Marei. Stem cell therapy: a revolutionary cure or a pandora’s box. Stem Cell Research & Therapy. Vol. 16, p. 255, 2025, https://doi.org/10.1186/s13287-025-04334-1. [↩]
- H. E. Marei. Stem cell therapy: a revolutionary cure or a pandora’s box. Stem Cell Research & Therapy. Vol. 16, p. 255, 2025, https://doi.org/10.1186/s13287-025-04334-1. [↩]
- V. Tabar, H. Sarva, A. M. Lozano, A. Fasano, S. K. Kalia, K. K. H. Yu, … C. Henchcliffe. Phase I trial of hES cell-derived dopaminergic neurons for Parkinson’s disease. Nature. Vol. 641, no. 8064, pp. 978–983, 2025, https://doi.org/10.1038/s41586-025-08845-y. [↩] [↩] [↩]
- Y.-C. Lin, K. K. Yokoyama, et al. Prevention of tumor risk associated with the reprogramming of human pluripotent stem cells. Journal of Experimental & Clinical Cancer Research. Vol. 39, p. 100, 2020, https://doi.org/10.1186/s13046-020-01584-0. [↩] [↩]