Abstract
Microplastics (MPs), plastic particles less than 5 mm in size, are increasingly recognized as emerging contaminants in agricultural systems. However, the overall effect of MPs on agriculture health has not been fully understood. This review attempts to fill this void by integrating the mechanisms of the key effects recently studied across the major processes associated with the agricultural system. Once in the soil, MPs can alter soil structure, water retention, and nutrient cycling, while also disrupting plant-microbe interactions. Their influence hinders physiological processes including respiration, photosynthesis, vascular transport, and nutrient uptake. MPs have also been shown to reduce seed germination rates, delay sprouting velocity, and obstruct reproductive structures, leading to declines in yield and long-term agricultural health. Given the persistence of MPs in the environment and the large amount of plastics released to the environment serving as the source of MPs, these effects may cascade through the food chain, allowing MPs to persist across growing seasons and potentially pass to herbivores, threatening not only crop productivity but also broader ecosystem stability. Recent research on MP-induced plant toxicities was reviewed and compiled in the hope of highlighting the primary mechanisms to promote public awareness and education in the protection of agricultural health and food security.
Keywords: microplastics, plant health, soil health, plant physiology, agriculture, contamination
Introduction
Over the past decade, microplastics (MPs), plastic particles generally less than 5 millimeters in size, have emerged as a pervasive pollutant. They can be classified into two types: primary MPs and secondary MPs. Primary MPs are manufactured for certain purposes such as industrial raw materials, cosmetics, and resin pellets1. Secondary MPs are formed from degradation of larger waste through physical, chemical, and biological processes2. As MPs continue to degrade, through human and natural processes, they can be broken down further into the category of nanoplastics (NPs), which are particles between the size range 1 to 1000 nm3. Due to its miniscule size, NPs possess unique physical, chemical, and morphological traits which allow them easier access to organisms and biological systems. This review generally refers to MPs as MPs and NPs in combination unless NPs are specifically referred to. Recent studies have found the presence of MPs/NPs in soil can affect plant growth and wellbeing4,5,6.
When plants or produces impacted by MPs are consumed by herbivores, MPs may pass along in the food chain and possibly spread to humans due to their persistence in the environment, making MP-invasion of natural ecosystems a pressing and alarming topic. However, environmental regulations targeting MPs in agriculture for protection of plants and crops have not been identified in the US and are rare globally at the time of this review as of January 2026.
The objective of this review is to distill a variety of recent studies to find out the key information to support effective communication for public awareness and education, which could potentially promote development of environmental and agricultural policies and regulations on MPs in agriculture. This review inspects two central aspects of the impacts of MPs on crop health, which are equivalent to the two questions this review intends to answer: (1) what are the major significant mechanisms for MPs to enter and move into a plant? and (2) what are the most significant damages that MPs would bring to a plant’s life?
Materials and Methods
In this review, recent research pertinent to MP’s effect on plant health was compiled and evaluated. The databases used to identify pertinent research include Google Scholar, ResearchGate, and ScienceDirect. Searches were performed using English-language keywords, including combinations of microplastics, nanoplastics, plant health, crop yield, soil-plant interactions, nutrient uptake, and plant physiology. As a result, the studies synthesized largely reflect findings found in English-language publications.
Given the rapid advancement of research surrounding MPs, this review primarily focused on the studies published in the years since 2020, although earlier studies were also considered when relevant to mechanistic understanding. Articles that did not involve plant-based systems or lacked primary experimental or review-based data were excluded.
This review synthesizes the compiled studies to evaluate the key mechanisms of MP-induced plant stress, focusing on how MPs alter root-zone environment, enter plant tissues, move within the plant, and ultimately impair plant health across respiration, photosynthesis, vascular system and nutrition, as well as germination and reproduction.
Results
Microbe-Mediated Impacts
MPs in soil can disrupt the symbiosis between plants and root-associated microbes, which can constrain processes for plant nutrient acquisition. Once introduced in soil, MPs can bring changes to soil structure, porosity, and water-air balance, which in turn can obstruct hyphal networks and root-fungus contact zones, thus reducing mycorrhizal colonization. Reduced mycorrhizal fungi activities lower the effective absorptive surface area of root and their capacity to suppress pathogens, decreasing nutrient acquisition and increasing plant susceptibility to disease7.
MPs can also change bacterial community composition in the plant root zone, disrupting plant growth-promoting rhizobacteria (PGPR) and their associated functions. By modifying pH and nutrient bioavailability, MPs select microbial taxa capable of adapting to plastic surfaces and surrounding soil, disfavoring PGPR involved in nitrogen fixation, phytohormone production, and mineral acquisition7, 8. As a result, the presence of MPs in soil can suppress PGPR activity and weaken plant defenses and stress tolerance9, 10.
From the biogeochemical perspective, disrupted hyphal networks and PGPR communities can reduce rates of organic phosphorus mineralization and phosphate solubilization while shifts in nitrifier and denitrifier microbial populations affect nitrogen availability7. In a simple conceptual triad involving plant, microbiome, and MPs, MPs would be a destabilizing factor that changes soil physical-chemical conditions and microbial networks, thereby weakening nutrient fluxes and defense signaling back to the plant. In response, plants may adjust root architecture and exudation patterns in response to nutrient stress or pathogen pressure, potentially amplifying MP-induced microbiome shifts8, 10. This triad highlights that MP exposure affects mycorrhizal colonization, PGPR functions, and N/P cycling through interconnected processes that collectively constrain nutrient uptake and disease resistance.
Plant Uptake of MPs
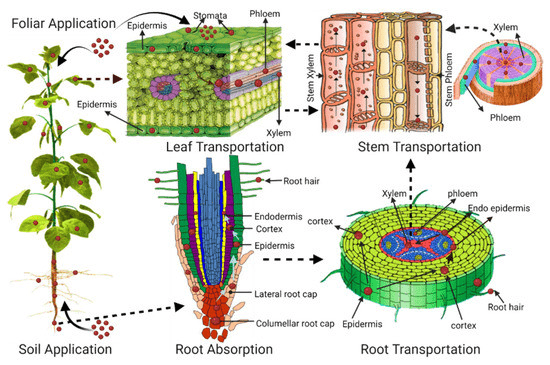
MPs can enter the organ system of a plant through a variety of mechanisms, as shown in Figure 1, which causes repercussions for the plant’s health, growth, and reproduction.
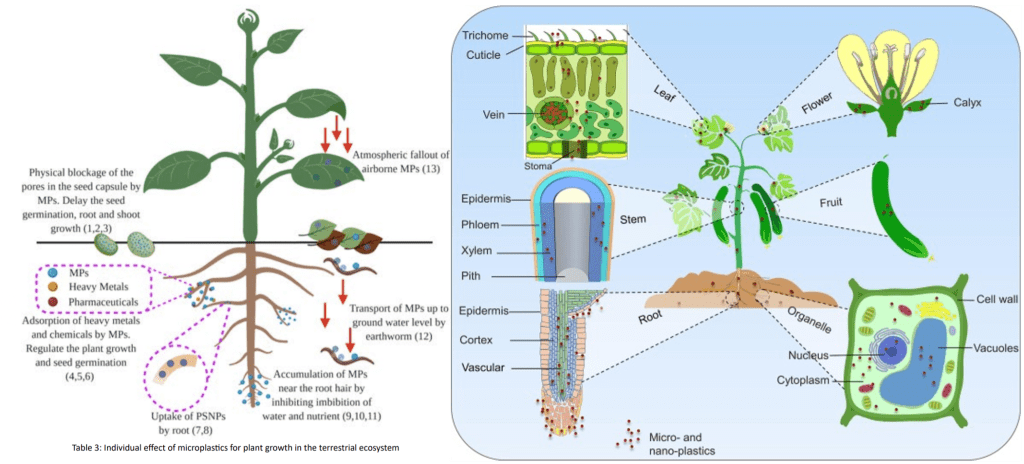
Primarily, there are three types of transport routes that facilitate uptake of MPs in plants, namely solid, aqueous, and gaseous vehicles13. Solid transport includes physical contact with MPs found in the soil, organismal interactions that include uptake from symbiosis with MPs-containing mutualists, and uptake through contaminated fertilizer14. Experimental work showed that liquid uptake was primarily via wastewater run-off which could be taken in by plants via normal root water uptake mechanisms such as passive osmosis or root drinking15. Air-transported uptake was mainly foliar, a process where MPs in the air were deposited onto plant surfaces and eventually entered the plant through stomatal pores16. Of these three routes, although solid transport was considered as the more significant pathway for MPs to enter plants because of the filtration of MPs by soil from the liquid matrix, aqueous transport is often the way through that the majority of the MPs are introduced to the local system supporting a plant.
MPs Transport in Plants
Particle sizes play an important role in controlling MP transport in plants. Larger MPs that entered are not expected to penetrate cell walls or be digested by plants’ tissue. In some cases, movement inside a plant is less common but can potentially occur through apoplastic pathway (between cells and intercellular spaces), symplastic pathway (through the cytoplasm and plasmodesmata), and xylem vessels (water conducting channels). As roots are the primary site for MP entry to plants, transpiration through xylem vessels is considered the primary pathway for the movement of MPs from plant roots to above ground tissues and plant parts, including stems, leaves, and fruits. When exposed to MPs (particularly NPs with size less than 1 micrometer) in air, internalization through stomatal openings can occur. Mobilization from leaf to stem to root as a secondary pathway appears possible based on studies with inorganic nanosized particles; however, this pathway needs to be confirmed with MPs and mechanisms need additional studies17.
When inflicted, MP toxicity is induced in plants through various routes, each causing various issues with different magnitudes and natures of damage, as well as long-term impacts. Adverse effects are evident in physical growth reduction (primarily affecting seed germination, roots, plant yields) and physiological growth reduction (primarily affecting shoot, leaf, and root physiology)18. Because MPs are generally non-biodegradable and chemically weather slowly, infected agriculture is expected to experience persistence of MP presence across seasons and their detriments to plant health would continue for an extended period. These detriments may be reflected in four aspects as discussed below, respectively.
Impeded Effect on Respiration
A significant ramification of MP-contamination is its adverse effects upon respiration in plants, which can result from direct interference with leaf organelles.
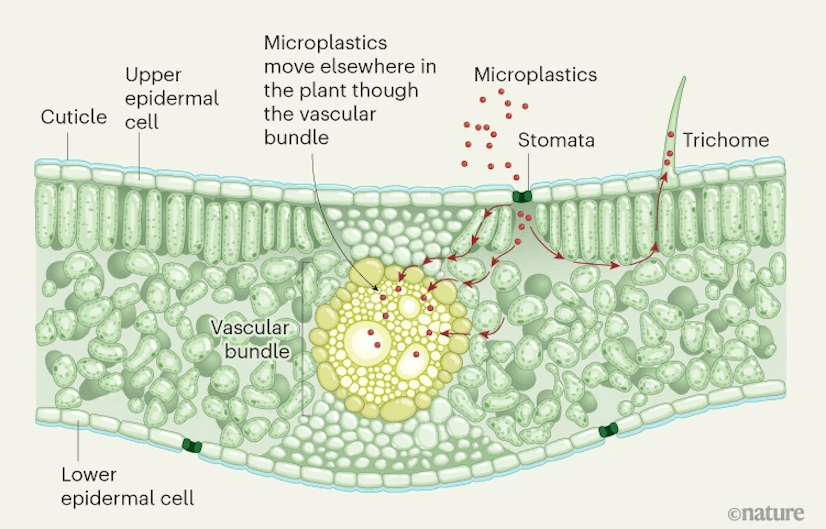
As shown in Figure 2, airborne MPs can accumulate within crops en masse via plant leaves during respiration16. Respiration is a vital process where plants cycle out carbon dioxide in exchange for oxygen intake, allowing for the breakdown of glucose into adenosine triphosphate (ATP), a fundamental energy-dense molecule that drives growth, reproduction, and other life functions. Studies have found that MPs contamination can hinder this process in two ways, with multiple mechanisms proposed but not all directly demonstrated. Firstly, when in the surrounding environment, as demonstrated, MPs can impede soil aeration19, by blocking soil pores20 and disrupting soil pore connectivity21. This reduces the breathability of the surrounding soil and the plant itself. Secondly, MPs in the air can accumulate on plant surfaces and block the stomata directly18, affecting gas exchange and water transportation (as the primary effects), two key factors in respiration which without, can induce oxidative stress (as a secondary response)22. Under oxidative stress, free radicals, in particular, reactive species of oxygen, can eventually saturate the plant and surpass its antioxidant defenses, risking damage to DNA integrity, as proposed (but mechanisms still being elucidated)23. Additionally, studies have found that while MPs do not directly sequester carbon, they can cause enough disruption in carbon cycling, especially in the ocean, that affects marine plants and other photosynthetic organisms24,25. Therefore, large-scale macro-ecosystem and biome effects can be caused.
Impeded Effect on Photosynthesis
Suspended MP concentrations in shallow water environments increase sunlight blockage, thereby reducing photosynthesis potential of submerged plant populations26. This is because MPs, due to their size, permit less light diffusion when layered, effectively shielding chloroplasts from the sun. MPs can also aggregate native sediments, creating larger clusters that may remain suspended whilst increasing their blockage, or precipitating masses that can envelope plants27.
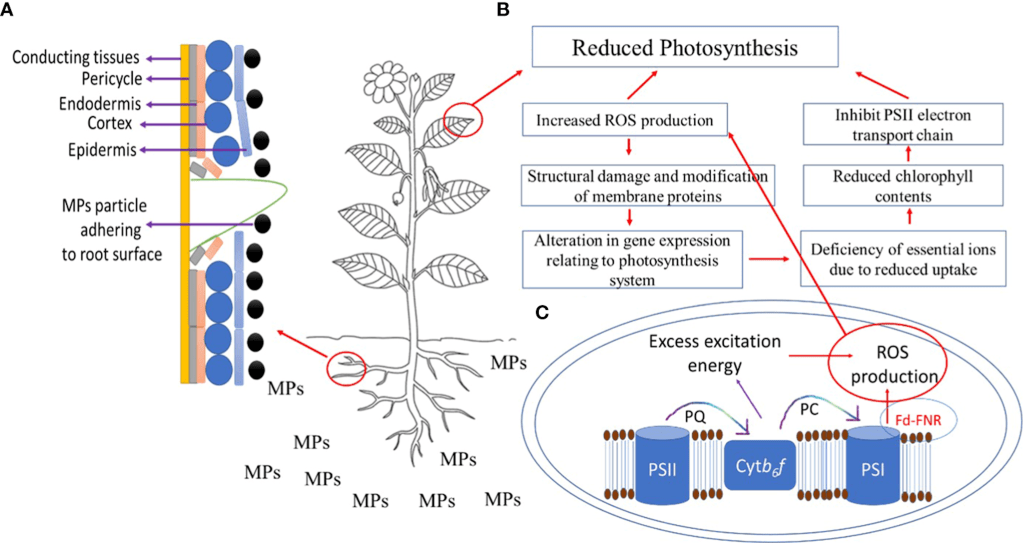
MPs have been found to hinder photosynthetic capabilities of plants through multiple pathways, as illustrated in Figure 3. Table 1 synthesizes the effects of MPs on the physiological process of photosynthesis, reported by several studies using different MP types, plants, and experimental conditions.
| Experimental Conditions | Plant | MP/NP Type and Concentration | Effects on Photosynthesis | Mechanistic Hypothesis |
| Hydroponic growth chamber exposure. Controlled temperature and light. (14 days)29 | Rice (Oryza sativa) | Polystyrene MPs; 3.0 mg/L nutrient solution | ~31% photosynthetic rate reduction against control | MPs induce oxidative stress and disrupt phosphorus translocation, causing chlorophyll loss and a decline in stomatal conductance. |
| Poly vinyl chloride MPs; 3.0 mg/L nutrient solution | ~44% photosynthetic rate reduction against control | MPs trigger chemical toxicity and ionic imbalance, leading to metabolic collapse and non-stomatal inhibition of CO2 fixation. | ||
| Growth chamber, foliar spray exposure (21 days)30 | Lettuce (Lactuca sativa) | Polystyrene NPs; 1 mg/L foliar spray | Reductions in: Chlorophyll a: ~9% Chlorophyll b: ~9% Carotenoids: ~13% | Foliar NPs cause oxidative stress and iron deficiency, leading to chloroplast damage and inhibited pigment biosynthesis. |
| Growth chamber, soil exposure, 28 days31 | Pumpkin (Cucurbita pepo) | Poly vinyl chloride MPs; 0.02–0.2% (w/w in soil) | Reduction in: Fv/Fm ~10%, PIABS ~50%, NDVI ~25%, and chlorophyll ~41%. | MPs induce iron deficiency, impairing chlorophyll synthesis, and photosynthetic efficiency. |
| Polyethylene (PE) MPs; 0.02–0.2% (w/w in soil) | Reduction in: Fv/Fm ~11%, PIABS ~14%, NDVI ~25%, and chlorophyll ~49%. | PE MPs decreased chlorophyll and photosynthetic efficiency by depleting essential leaf nutrients such as magnesium and increasing toxic nickel levels. | ||
| Polypropylene MPs; 0.02–0.2% (w/w in soil) | No significant effect | Not Applicable | ||
| Polyethylene terephthalate MPs; 0.02–0.2% (w/w in soil) | No significant effect | Not Applicable |
% w/w = g MP per 100 g
soilChl = total chlorophyll content
Fv/Fm = maximum quantum efficiency of photosystem II
NDVI = normalized difference vegetation index (proxy for plant greenness and vigor)
mg/L = milligrams per liter
PIABS = performance index on absorption basis (overall photosynthetic performance)
The results in Table 1 show that MPs can have negative effects on photosynthesis to different extents depending on testing parameters32. It remains unclear why the polypropylene and polyethylene terephthalate MPs did not significantly affect pumpkin photosynthesis at soil concentrations similar to those of other MP types that caused measurable declines. Other studies likewise showed that, in both algae and terrestrial plants, exposure to lower concentrations of MPs did not have discernible effects on chlorophyll and photosynthetic pigment content33. These limited responses suggest plants may tolerate MPs pollution up to a certain threshold. However, a universal threshold may not exist because responses would vary from plant to plant when exposed to different types and levels of MPs. Identifying which MP types are most damaging, and determining region-specific thresholds for critical crops, is therefore an important direction for further research.
In a multiecosystem meta-analysis of more than 3000 of published records34, MPs were estimated to have a mean reduction effect of 12%, 7%, and 9% on photosynthetic capabilities in terrestrial plants, marine algae, and freshwater algae, respectively. The corresponding input MP exposure levels in this meta-analysis were based on experimental doses of >1% of MPs in soil by weight, >72 micrograms MPs per liter water, and >100 micrograms MPs per liter water, respectively34. Although the results remain to be validated, these multiecosystem values show that MPs can have a negative effect on photosynthetic activity, whether the disruption occurs through causing physical blockage, inhibiting chemical activity, or altering photosynthetic pigment contents.
Photosynthesis makes up a large portion of nutritional value for plants. Disruptions in photosynthesis will decrease the outcomes of the process, such as glucose and oxygen, which sequentially will cause shortages in nutrient transport to other parts of the plant, disrupting the entire process and causing systemic problems.
Impeded Effect on Vascular System & Nutrition Transport
The vascular system in plants is key to a species’ long-term health and success, as it transports nutrients like water and minerals to all parts of the plant. It was shown that MPs, upon entry into a plant, can eventually end up in vascular tissue via tissue perforation or permeation35. This mobilization, illustrated in Figure 4, is limited by MP size, shape, and structure.
Intake of MPs (particularly NPs) by plants and their subsequent migration in the vascular system can impair plant health issues via three routes. The first route is physical obstruction. MPs can accumulate in apoplastic spaces or xylem vessels, impeding water or nutrient flow and thereby limiting the supply of essential materials for plant development and growth36. This route is supported by direct visualization, including fluorescence and electron microscopy showing particle deposition36. The second route involves chemical interactions. MPs can act as sorption media that bind plant nutrients or signaling molecules or they can release plastics-associated additives and pollutants. These processes can disrupt ion balance, hormone signaling, and redox chemistry, weakening tissue functions. Evidence for this route comes from chemical measurements, such as altered nutrient stoichiometry, leached plasticizers, and shifts in soil or tissue chemical profiles37. The third route reflects biological responses. MPs in plants can trigger oxidative stress and immune-like signaling. When these stress responses occur in leaves, they can suppress photosynthesis and supply less nutrition to circulate through the vascular network30. This pathway is supported primarily by inferred physiological changes, including elevated reactive oxygen species, altered antioxidant enzyme activity, and growth suppression38. Together, these routes illustrate that MPs uptake and vascular transport can cause multi-faceted adverse health outcomes.
Impeded Effect on Germination and Reproductive Potential
Germination
The accumulation of MPs in plant systems can eventually lead to reduced germination rates and disruptions in reproduction, as seen in Figure 5. A study by Bosker et al. showed significant reductions in germination rates over the course of 8 hours of MPs exposure40. Three sizes of MPs (50, 500, and 4800 nm) were tested on Lepidium sativum seeds, which consequently experienced reduced germination rates as MP size increased. L. sativum seeds in the control group had 78% germination success, and after being exposed to 4800 nm polystyrene MPs, the germination rate dropped to 17% when exposed to 107 MP items per milliliter, characterizing a significant swing in germination caused by MPs exposure and uptake. Continued presence of MPs can result in progressive clogging of pores and chemical toxicity that retard internal germination processes, marked by root growth rates, antioxidant enzyme fluctuations, and overall seedling viability41.
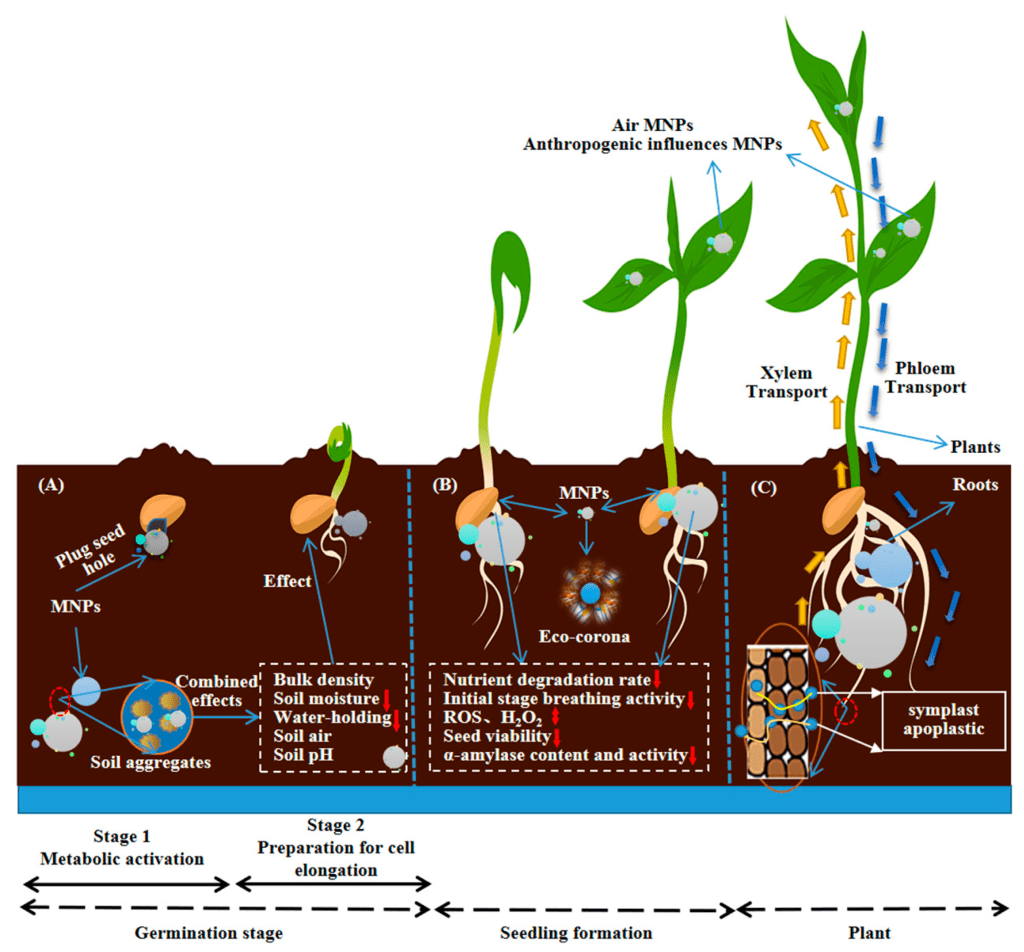
Plants exposed to MPs can decrease seed velocity, also known as the speed at which seeds transition from a dormant state into seedlings43. Decreased velocity can pose a problem for plant species as seedlings that sprouted later become more vulnerable to environmental stressors such as colder weather conditions or dryer climates. MPs were found to impact germination by changing soil porosity and structure. Through reducing the porosity of the soil, or the amount of air that is passed through, it increased the difficulty for seeds to obtain water and nutrients from the soil, leading to delayed germination44. MPs in the soil create other problems, as MPs will degrade soil structure, leading to a loss in water retention capabilities45. Consequently, the young plant will experience exacerbated water stress and hindered development in critical early stages of growth. Direct exposure to MPs to the seed surface can also lead to detrimental effects. Adhesion to seed surfaces will result in physical blockage to channels responsible for nutrient and water uptake46.
Research on MP effects in plant health has largely focused on the exposed parental generation, with very limited information available specifically comparing the transgenerational effects that would be reflected on offsprings. Targeted field studies that track MPs through several cropping cycles and into successive generations would greatly improve understanding in this aspect.
Reproductive Potential
The deposition of MPs on plant surfaces can result in significant consequences for reproductive potential and output in agriculture. MPs were observed to inhibit reproductive output by clogging reproductive organs. This blockage resulted in decreases in seed production and negatively affected the amount of pollen tubules able to reach ovaries47. Other primary effects involve the overall impairment to reproductive structures from residues of MPs contamination48. Additionally, reproductive potential can be inhibited as a secondary response if the plant must focus resources and energy upon other factors, especially those that pose significant threats to the safety and wellbeing of the plant. One such scenario is observed when MPs contamination creates oxidative stress, meaning the plant must invest resources into spurring the activity of antioxidant enzymes49. This scenario leaves the plant without sufficient resources to fully support reproductive processes, inhibiting the overall reproductive potential.
Plant Tolerance Mechanisms and Potential Mitigation Strategies
Plants affected by MPs appear to activate intrinsic tolerance responses similar to those used during abiotic stress. Physiological and molecular studies consistently show increased activity of antioxidant enzymes under MP exposure, indicating that plants attempt to counter MP-induced oxidative stress by strengthening redox homeostasis50. Plants also show morphological adjustments, including changes in root branching, greater production of fine roots, and shifts in root-shoot allocation. These changes likely represent compensatory strategies to maintain water and nutrient uptake when MPs alter soil structure or reduce root-soil contact51.
Microbiome mediated buffering is another plausible tolerance mechanism. Mycorrhizal fungi can stabilize soil aggregates, maintain hyphal nutrient foraging networks, and influence plant stress signaling. These functions may mitigate MP-related disruptions to nutrient uptake and root physiology52. However, it is still unclear whether microbial partners such as PGPR can directly reduce MP accumulation or toxicity. Pilot studies are needed to better understand these interactions.
Agronomic interventions may also mitigate MP impacts by altering their mobility, sorption behavior, distribution, or bioavailability in soil. Amendments, such as biochar and compost, can influence how plants are exposed to MPs52. Biochar can improve soil aggregation, aeration, and water-holding capacity in MP-contaminated soils while also supporting more diverse and resilient microbial communities. These changes can enhance plant growth and reduce oxidative damage53. Compost additions may stimulate microbial communities that help plant buffer stress. Cover crops can also modify soil structure and hydrological pathways, which may influence MP transport52. The potential of using agronomic practices to alter MP mobility and bioavailability warrants further investigation.
Together, these emerging tolerance mechanisms and mitigation strategies show that plant responses to MPs can be multifaceted. However, systematic testing under realistic agricultural conditions is limited. Targeted experiments are needed to identify which strategies effectively reduce MP impacts on plant health.
Discussion
Due to wide spread of MPs in the environment, the health detriments of MPs to plants can potentially cause large-scale environmental, agricultural, and ecosystem impacts because of the impairment of vital physiological functions. The more significant impairing effects are believed to be caused by direct contact with MPs that obstruct plant pores and respiratory organs, impede respiration, disrupt photosynthesis, reduce germination and reproduction rate. The persistence of MPs in the environment and the impairing effects suggest that the MP-resulting malnourishment can be proliferated across ecological members and other species when recycling of MPs keeps them locked in the ecosystem and risks biome expansion.
Secondary effects are believed to be attributable to MPs with smaller sizes that can be mobilized to various parts of a plant, block transport vessels, and result in suffocation of essential nutrients. Secondary effects include damage to vascular tissues, local and holistic oxidative stresses, disrupted digestive processes, and weakened antioxidant responses. Because there are multiple forms of MPs-induced toxicity, there is also a mosaic of less dangerous effects coupled with long term presence-encouraged probabilities of health complications. The continued recycling of particulates may enforce more adverse outcomes in time. This can be potentially compounded by the effect of continued novel exposure of MPs from the sources of ongoing industrial and public consumption practices.
A preliminary correlation of experimental MP levels in soil, MP-induced physiological impairments, and the observed or inferred plant yield is summarized in Table 2 based on limited published data to establish a quantitative evaluation of MP effect. Although the experimental MP levels do not reflect typical field MP levels in agricultural soil, the results provide insights into establishing limits for bounding risks and mapping typical physiological changes onto observed or hypothesized plant yield impacts.
| Experimental MP dose in soil (w/w) | Typical physiological change | Yield or biomass impact |
| 0.1% Polystyrene MPs | Photosynthesis: ↓~10-12% | Biomass: ↓~8-10%; (Wheat experimental observation)54 |
| 1% Polystyrene MPs | Photosynthesis: ↓~15-25%; Chlorophyll: ↓~10-15%; Root length/biomass: ↓~10-20% | Biomass / grain proxy: ↓~12-20%; (Wheat experimental observation)54 |
| 0.4% LowDensity Polyethylene or polyester MPs | Root biomass: ↓~5-10%; Shoot height: ↓~5-8% | Biomass: ↓~5-15% in some soil textures; near‑neutral in others; (Winter wheat experimental observation)55 |
| 0.1-10% mixed aged Polystyrene, Polyethylene, Polylactic Acid MPs | Chlorophyll: ↓~5-20%; Root/shoot length: ↓~5-25% | Biomass / marketable yield: ↓~5-25% with increasing MP dose; (Lettuce experimental observation)56 |
| 0.1-1% various MPs | Photosynthesis & chlorophyll: ↓~7-15%; Root traits & nutrient uptake: ↓~5-20% | Yield / biomass: ↓~5-20%; (Inferred based on meta‑analysis for multiple crops typically)57 |
These quantitative correlations suggest that serious attention shall be directed to the nature of MPs induced plant health issues for protection of environment and agriculture. Because of the bottom-up benefits of agriculture and wildlife plant dependencies, enhanced understanding of the key effect of MPs and more significant mechanisms on agriculture health is an important piece of the puzzle to achieve sustainable life and health across all ecologies. This enhanced understanding is critical for public awareness and education, and also instrumental in development of environmental protection policies and regulation.
This review also identified the following key research gaps linking MP contamination to crop health risks and potential agricultural impacts.
First, MP toxicity to plants depends strongly on the polymer material. Differences in crystallinity, surface chemistry, and additives (such as plasticizers, stabilizers, pigments, and flame retardants) influence how MP particles degrade, sorb co-contaminants, and interact with roots and soil. More oxidizable polymers, such as PLA or weathered PE, tend to form smaller, more polar and charged fragments, which increases their reactivity and contact with root surfaces. In contrast, more crystalline and persistent polymers, such as PET or some PP, degrade slowly but provide long-lasting surfaces for adsorbing organic contaminants (including pesticides) and metals. Additives can leach out as separate toxicants or alter surface charge and hydrophobicity, which changes sorption behavior and can indirectly affect plant health58, 59. However, cross-polymer comparisons using the same particle size, shape, dose, soil, and plant species are rare and often inconsistent. This makes polymer-specific conclusions for crop plants a major research gap that requires coordinated, multi-polymer experiments.
Secondly, the key health effects reported are largely based on laboratory studies which used model MPs with relatively high concentrations, low variability on structure and chemical compositions, and controlled testing conditions. Reported MP levels in agricultural soil are much lower, typically in the range of hundreds to thousands of MP items per kilogram soil worldwide, with hotspots reaching one to two orders of magnitude higher57,60. Field studies with real-world MPs contamination and control studies are very limited. Although the primary effects would likely be less pronounced, multi-season real-world field studies, including evaluation of possible transgenerational impacts, will be very valuable and highly anticipated in clarifying the actual extents of the effects caused by the MPs that have been released in the current environment.
Thirdly, confounding factors play an important role in the plant responses reported in MPs exposure studies. Adding MPs to soil can change soil structure, water retention, and nutrient intake, making it difficult to link plant effects to direct particle toxicity alone. Even in controlled experiments, it is difficult to separate causes because MPs can induce oxidative stress, modify soil physical and chemical properties, and shift microbial communities that influence nutrient availability and plant stress responses. To separate direct MP effects on plant health from nutrient‑mediated or microbiome‑mediated mechanisms, studies with more advanced experimental designs are needed.
Lastly, when plant tissues (leaves, fruits, seeds) containing MPs are consumed, it is still unclear how much of those MPs through food chain reach the human gastrointestinal tract or whether any are absorbed at the low levels found in the environment. Long‑term health risks from eating crops affected by MPs are possible, but they have not been demonstrated. Rigorous exposure assessment studies are needed before claims about threats to food security can be confidently supported.
References
- Yu, Z., Xu, X., Guo, L., Seyit Yuzuak and Lu, Y. Physiological and biochemical effects of polystyrene micro/nano plastics on Arabidopsis thaliana. Journal of Hazardous Materials 469, 133861 (2024). [↩]
- Lalrinfela, P., Vanlalsangi, R., Lalrinzuali, K. and Babu, P.J. Microplastics: Their Effects on the Environment, Human Health, and Plant Ecosystems. Environmental Pollution and Management 1, 248 (2024). [↩]
- Gigault, J., Halle, A. ter, Baudrimont, M., Pascal, P.-Y., Gauffre, F., Phi, T.-L., El Hadri, H., Grassl, B. and Reynaud, S. Current opinion: What is a nanoplastic? Environmental Pollution 235, 1030 (2018). [↩]
- Abbasi, S., Moore, F., Keshavarzi, B., Hopke, P.K., Naidu, R., Rahman, M.M., Oleszczuk, P. and Karimi, J. PET-microplastics as a vector for heavy metals in a simulated plant rhizosphere zone. Science of The Total Environment 744, 140984 (2020). [↩]
- Huang, Y., Zhao, Y., Wang, J., Zhang, M., Jia, W. and Qin, X. LDPE microplastic films alter microbial community composition and enzymatic activities in soil. Environmental Pollution 254, 112983 (2019). [↩]
- Iqbal, S., Xu, J., Allen, S.D., Khan, S., Nadir, S., Arif, M.S. and Yasmeen, T. Unraveling consequences of soil micro- and nano-plastic pollution on soil-plant system: Implications for nitrogen (N) cycling and soil microbial activity. Chemosphere 260, 127578 (2020). [↩]
- Singh, A. Microplastic Alters Rhizosphere Microbiome: A Review. In: Khanna, K., Kaur Kohli, S., Bhardwaj, R. (eds) Microplastics and Soil Microbiome. Springer, Singapore. (2025). [↩] [↩] [↩]
- Wankhade, A., Wilkinson, E., Britt, D., Kaundal, A. A Review of Plant-Microbe Interactions in the Rhizosphere and the Role of Root Exudates in Microbiome Engineering. Applied Science 15, 7127 (2025). [↩] [↩]
- Zeng, W., Xiang, D., et al. Effects of combined inoculation of arbuscular mycorrhizal fungi and plant growth-promoting rhizosphere bacteria on seedling growth and rhizosphere microecology. Frontiers in Microbiology 15, 1475485 (2024). [↩]
- Yang, L., Qian, X., Zhao, Z., Wang, Y., Ding, G., Xing, X. Mechanisms of rhizosphere plant-microbe interactions: molecular insights into microbial colonization. Frontiers in Plant Science 15, 1491495 (2024). [↩] [↩]
- De Silva, Y., Rajagopalan, U. and Kadono, H. Microplastics on the growth of plants and seed germination in aquatic and terrestrial ecosystems. Global Journal of Environmental Science and Managemen, 7(3), 347 (2021). [↩]
- Liu, Y., Ben, Y., Che, R., Peng, C., Li, J. and Wang, F. Uptake, transport and accumulation of micro- and nano-plastics in terrestrial plants and health risk associated with their transfer to food chain – A mini review. Science of The Total Environment 902, 166045 (2023). [↩]
- Liu, D. Microplastics: Notes on Recycling, Waste, and Environmental Prevalence. International Journal of Scientific Research and Management (IJSRM) 13(04), 262. (2025). [↩]
- Weithmann, N., Möller, J.N., Löder, M.G.J., Piehl, S., Laforsch, C. and Freitag, R. Organic fertilizer as a vehicle for the entry of microplastic into the environment. Science Advances 4(4), 8060 (2018). [↩]
- Seelig, A.H., Junghans, V., Thorsten, R. and Zahn, D. Plant Uptake of Persistent and Mobile Chemicals in Rocket (Eruca sativa)-A Greenhouse Study on Agricultural Wastewater Reuse. Environmental Science & Technology 59(18), 9265 (2025). [↩]
- Peijnenburg, W. Airborne microplastics enter plant leaves and end up in our food. Nature 641, 601 (2025). [↩] [↩] [↩]
- Boctor, J., Hoyle, F.C., Farag, M.A., Matta Ebaid, Walsh, T., Whiteley, A.S. and Murphy, D.V. Microplastics and nanoplastics: fate, transport, and governance from agricultural soil to food webs and humans. Environmental Sciences Europe 37, 68 (2025). [↩]
- Jia, L., J., Liu, L., Zhang, Y., Fu, W., Xing, L., Wang, Q., Tanveer, M. and Huang, L. Microplastic stress in plants: effects on plant growth and their remediations. Frontiers in Plant Science 14, 1226484 (2023). [↩] [↩]
- Hasan, M.M., Tarannum, M.N., Rafiqul Bari, G.A.K.M., Swapon, A.R., Kabir, M.S., Ahmmed, S. and Daraj Uddin Prodhan, Md. Impact of microplastics on terrestrial ecosystems: A plant-centric perspective. Environmental Pollution and Management 1, 223 (2024). [↩]
- Kim, S.W. and An, Y.-J. Soil microplastics inhibit the movement of springtail species. Environment International 126, 699 (2019). [↩]
- Jiang, X.J., Liu, W., Wang, E., Zhou, T. and Xin, P. Residual plastic mulch fragments effects on soil physical properties and water flow behavior in the Minqin Oasis, northwestern China. Soil and Tillage Research 166, 100 (2017). [↩]
- Jia, Y., Cheng, Z., Peng, Y. and Yang, G. Microplastics alter the equilibrium of plant-soil-microbial system: A meta-analysis. Ecotoxicology and Environmental Safety 272, 116082 (2024). [↩]
- Jia, Y., Cheng, Z., Peng, Y. and Yang, G. Microplastics alter the equilibrium of plant-soil-microbial system: A meta-analysis. Ecotoxicology and Environmental Safety 272, 116082 (2024). [↩]
- Shen, M., Ye, S., Zeng, G., Zhang, Y., Xing, L., Tang, W., Wen, X. and Liu, S. Can microplastics pose a threat to ocean carbon sequestration? Marine Pollution Bulletin 150, 110712 (2020). [↩]
- Li, K., Du, L., Qin, C., Bolan, N., Wang, H. and Wang, H. Microplastic pollution as an environmental risk exacerbating the greenhouse effect and climate change: a review. Carbon Research 3(1), 1 (2024). [↩]
- Yang, H., Niu, S., Guo, M. and Xue, Y. A critical review of the ecotoxic effects of microplastics on aquatic, soil and atmospheric ecosystems and current research challenges. Environmental Research 274, 121361 (2025). [↩]
- Khalid, N., Aqeel, M. and Noman, A. Microplastics could be a threat to plants in terrestrial systems directly or indirectly. Environmental Pollution 267, 115653 (2020). [↩] [↩]
- Ilyas, M., Liu, X., Yang, J. and Xu, G. Foliar implications of polystyrene nanoplastics on leafy vegetables and its ecological consequences. Journal of Hazardous Materials 480, 136346 (2024). [↩]
- Ma, J., Aqeel, M., et al., Effects of microplastics on growth and metabolism of rice (Oryza sativa L.). Chemosphere 307, 135749 (2022). [↩]
- Lian, J., Liu, W., et al., Foliar-applied polystyrene nanoplastics (PSNPs) reduce the growth and nutritional quality of lettuce (Lactuca sativa L.). Environmental Pollution 280, 116978 (2021). [↩] [↩]
- Colzi, I., Renna, L., et al., Impact of microplastics on growth, photosynthesis and essential elements in Cucurbita pepo L. Journal of hazardous Materials 423, 127238 (2022). [↩]
- Li, Y., Liu, X., Shinde, S., Wang, J., Zhang, P., Impacts of Micro- and Nanoplastics on Photosynthesis Activities of Photoautotrophs: A Mini-Review. Frontiers in Microbiology 12, 773226 (2021). [↩]
- Wang, F., Zhang, X., Zhang, S., Zhang, S. and Sun, Y. Interactions of microplastics and cadmium on plant growth and arbuscular mycorrhizal fungal communities in an agricultural soil. Chemosphere 254, 126791 (2020). [↩]
- Zhu, R., Zhang, Z., et al. A global estimate of multiecosystem photosynthesis losses under microplastic pollution. Proceedings of the National Academy of Sciences 122, 11 (2025). [↩] [↩]
- Li, Y., Zhang, J., et al. Leaf absorption contributes to accumulation of microplastics in plants. Nature 641, 666 (2025). [↩]
- Roy, T., Dey, T.K. and Jamal, M. Microplastic/nanoplastic toxicity in plants: an imminent concern. Environmental Monitoring and Assessment 195, 27 (2022). [↩] [↩]
- Chaudhary, H., Shah, G., Bhatt, U., Singh, H. and Soni, V. Microplastics and plant health: a comprehensive review of sources, distribution, toxicity, and remediation. npj Emerging Contaminants 1, 8 (2025). [↩]
- Wong, A., Taylor, G. Plants and microplastics: Growing impacts in the terrestrial environment. Frontiers in Plant Science 16, 1666047 (2025). [↩]
- Azeem, I., Adeel, M., et al. Uptake and Accumulation of Nano/Microplastics in Plants: A Critical Review. Nanomaterials 11(11), 2935 (2021). [↩]
- Bosker, T., Bouwman, L.J., Brun, N.R., Behrens, P. and Vijver, M.G. Microplastics accumulate on pores in seed capsule and delay germination and root growth of the terrestrial vascular plant Lepidium sativum. Chemosphere 226, 774 (2019). [↩]
- De Silva, Y.S.K., Rajagopalan, U.M., Kadono, H. and Li, D. Effects of microplastics on lentil (Lens culinaris) seed germination and seedling growth. Chemosphere 303, 135162 (2022). [↩]
- Li, X., Wang, R., Dai, W., Luan, Y. and Li, J. Impacts of Micro(nano)plastics on Terrestrial Plants: Germination, Growth, and Litter. Plants 12(20), 3554 (2023). [↩]
- Lozano, Y.M., Caesaria, P.U. and Rillig, M.C. Microplastics of different shapes increase seed germination synchrony while only films and fibers affect seed germination velocity. Frontiers in Environmental Science 10, 1017349 (2022). [↩]
- Wang, Z., Li, W., Li, W., Wu, Y. and Jing, S. Effects of microplastics on the water characteristic curve of soils with different textures. Chemosphere 317, 137762 (2023). [↩]
- de Souza Machado, A.A., Lau, C.W., Till, J., Kloas, W., Lehmann, A., Becker, R. and Rillig, M.C. Impacts of Microplastics on the Soil Biophysical Environment. Environmental Science & Technology 52(17), 9656 (2018). [↩]
- Liang, L., Wong, S.C. and Lisak, G. Effects of plastic-derived carbon dots on germination and growth of pea (Pisum sativum) via seed nano-priming. Chemosphere 316, 137868 (2023). [↩]
- Carvallo, G.O. and Muñoz-Michea, V. Polypropylene Fragments Block Pollen–Pistil Interactions and Reduce Seed Production in a Monkeyflower Species. Environmental Science & Technology Letters 11(5), 426 (2024). [↩]
- Qi, Y., Yang, X., Pelaez, A.M., Huerta Lwanga, E., Beriot, N., Gertsen, H., Garbeva, P. and Geissen, V. Macro- and micro- plastics in soil-plant system: Effects of plastic mulch film residues on wheat (Triticum aestivum) growth. Science of The Total Environment 645, 1048 (2018). [↩]
- Mészáros, E., Bodor, A., Kovács, E., Papp, S., Kovács, K., Perei, K. and Feigl, G. Impacts of Plastics on Plant Development: Recent Advances and Future Research Directions. Plants 12(18), 3282 (2023). [↩]
- Pehlivan, N., Terzi, Y., Gündoğdu, S. et al. Navigating microplastic-induced stress in plants: adaptations from physiology to gene regulation. Plant Molecular Biology 116, 5 (2026). [↩]
- Ren, F., Huang, J., Yang, Y. Unveiling the impact of microplastics and nanoplastics on vascular plants: A cellular metabolomic and transcriptomic review. Ecotoxicology and Environmental Safety 279, 116490 (2024). [↩]
- Das, R., Kumar, A., Kumar, R. Kumar, R., Singh, B. Impact of microplastics on plants: a transcriptome analysis perspective. Vegetos (2025). [↩] [↩] [↩]
- Ji, H., Zhou, C., Li, P. Applications of biochar in the remediation of soil microplastic pollution. Soil Science Society of America Journal 88, 1 (2024). [↩]
- Riaz, K., Yasmeen, T., Attia, K., Kimiko, I., Arif, M. Phytotoxic Effects of Polystyrene Microplastics on Growth Morphology, Photosynthesis, Gaseous Exchange and Oxidative Stress of Wheat Vary with Concentration and Shape, Toxics 13, 57 (2025). [↩] [↩]
- Neubert, K., Brüggemann, N. Soil texture modifies the impact of microplastics on winter wheat growth, Journal of Soils and Sediments 25, 1340 (2025). [↩]
- Fan, F., Liu, S., Jiang, Y., Zou, D., Zhang, Y., Zou, C. Studies on the impact of aged microplastics on agricultural soil enzyme activity, lettuce growth, and oxidative stress. Environmental Geochemistry Health 47, 213 (2025). [↩]
- Chen, Z.; Carter, L.; Banwart, S.; Kay, P. Microplastics in Soil–Plant Systems: Current Knowledge, Research Gaps, and Future Directions for Agricultural Sustainability. Agronomy 15, 1519 (2025). [↩] [↩]
- Menéndez-Pedriza, A.; Jaumot, J. Interaction of Environmental Pollutants with Microplastics: A Critical Review of Sorption Factors, Bioaccumulation and Ecotoxicological Effects. Toxics 8, 40 (2020). [↩]
- Li, W., Zu, B., Yang, Q., Guo, J., Li, J. Sources, distribution, and environmental effects of microplastics: a systematic review. Royal Society of Chemistry 13, 15566 (2023). [↩]
- Büks, F., Kaupenjohann, M. Global concentrations of microplastics in soils – a review, SOIL 6, 649 (2020). [↩]