Abstract
Parkinson’s disease (PD) is a progressive neurodegenerative disorder characterized by motor impairments and other non-motor symptoms. Its global prevalence is increasing, with men displaying higher incidence than women. Notably, earlier menopause in women is associated with elevated PD risk, implicating estrogen in a neuroprotective role. Given the widespread use of estrogenic compounds as part of hormone replacement therapy in women undergoing menopause—when PD risk rises—estrogen and other similar estrogenic hormones have been proposed as potential agents for symptom alleviation or risk reduction. This review synthesizes evidence from human studies on the effects of estrogenic compounds on Parkinson’s disease, complemented by data-science analysis of published findings. Human studies yield inconsistent results: some indicate heightened risk with exposure to estrogenic compounds, while others suggest neuroprotection, with ethnicity as the only consistent modulator. These divergent outcomes highlight the need for further investigation to clarify the therapeutic potential of estrogenic compounds, as well as the underlying mechanisms in Parkinson’s disease.
Introduction
Background
Parkinson’s disease is the second most prevalent neurodegenerative disease in the world, with a European study noting that around 1–2% of individuals above the age of 65 are affected1. In addition, Parkinson’s disease is increasing in prevalence as life expectancy increases worldwide, especially in the developed world; Parkinson’s disease is the fastest-growing neurodegenerative disease in the world1.
There are numerous risk factors for Parkinson’s disease. One of the most significant risk factors for Parkinson’s disease is sex; studies suggest that males are 1.5 to 2 times more likely to get Parkinson’s disease than females are2,3. Unique risk factors exist for women; a higher number of pregnancies and a younger age of menopause are linked with an increased risk of Parkinson’s disease2. Other risk factors potentially attributable to Parkinson’s disease, based on a study done in the Southern United States, include pesticide exposure, chemical exposures from military activities, and blows to the head from sports or combat; the former was somewhat more prevalent in women, while the latter two were strongly associated with men4.
The symptoms for Parkinson’s disease can be divided into motor symptoms and non-motor symptoms (such as memory problems, taste loss, and fatigue), with non-motor symptoms sometimes appearing several years before the more obvious motor symptoms of Parkinson’s arise5. This adds a unique challenge in diagnosing and treating Parkinson’s disease, especially if the mental conditions induced by Parkinson’s disease are misdiagnosed.
This study focuses on estrogen, a common compound in hormonal medicine. One way in which estrogen is affected is in hormone replacement therapy for menopausal women, where estrogen levels are increased to alleviate symptoms of menopause. Estrogen therapy’s effect on non-menopausal health conditions has been mixed so far, with some observations from the United Kingdom, Australia, and New Zealand showing that starting estrogen therapy many years after the start of menopause may increase health risks, especially circulatory conditions such as Venous thromboembolism6. It is also possible that other estrogenic hormones, such as progestin, can provide altered but nevertheless related effects as well, based on observations in the United Kingdom7.
In women, estrogen levels constantly fluctuate during the menstrual cycle and decline after menopause. However, these estrogen cycles can be substantially influenced by geographical origin, ethnicity, and other personal factors such as smoking and alcohol consumption, based on observations in Utah and California8.
Since women have a lower incidence of Parkinson’s disease and since women who have an earlier onset of menopause have an increased risk of developing Parkinson’s disease, it is possible that estrogen provides some protective effect against Parkinson’s disease9. In addition, estrogen may have a long-term protective effect on neurons, potentially through interactions with neurotransmitter systems and other parts of the brain, especially in regions of the brain with many estrogen receptors, such as the hippocampus and amygdala10. Interestingly, a study performed on Californian teachers found that women with higher lifetime levels of estrogen may have lower Parkinson’s disease rates than men or women with lower lifetime levels of estrogen11. This supports the notion that estrogen, and potentially other estrogenic compounds, provide a more chronic protective effect on neurons.
Regardless, it is very likely that the effectiveness of these hormones as a treatment for Parkinson’s disease is at least somewhat dependent on other factors. One study performed internationally reported that estrogens may increase the risk of short-term and long-term mechanical complications and revisions12. Due to the old age of many Parkinson’s patients, as well as the debilitating motor symptoms caused by Parkinson’s that may increase the risk of bone breakage, a further weakening of the skeletal system from treatments may be deemed as unacceptable, meaning that estrogenic treatments may be unable to be used feasibly within the context of Parkinson’s disease.
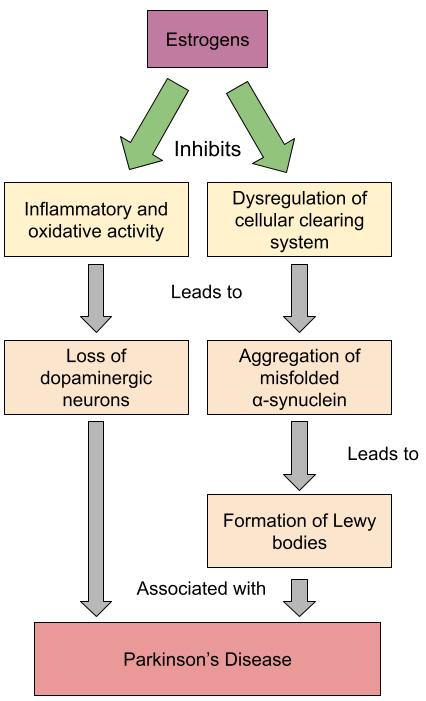
In Model 1, we illustrated how estrogen may decrease the risk of Parkinson’s disease through inhibiting processes that ultimately lead to the loss of dopaminergic neurons and the formation of Lewy bodies. This graph is conceptual in nature.
Significance
No treatment can currently halt the progression of Parkinson’s disease1. Levodopa, a dopamine precursor, is the main pharmacological treatment for alleviating the symptoms of Parkinson’s disease13. However, Levodopa administration is associated with several side effects, such as Levodopa-induced dyskinesia (unusual movements induced by Levodopa treatment) and anxiety14. In addition, Levodopa seems to only treat the motor symptoms of Parkinson’s, while Parkinson’s disease also contains many debilitating non-motor symptoms as well5. Gut bacteria such as Clostridium sporogenes may break down and deactivate Levodopa residues, which may inhibit Levodopa’s effectiveness15.
Objectives
The objectives of this systemic review of human data are to understand the effects of estrogenic compounds, namely estrogen but also progesterone and other compounds that either act as estrogens or activate estrogen receptors, on the prevalence and severity of Parkinson’s disease with regards to humans, especially human women. We chose to focus more on women because women are the primary recipients of estrogenic compounds as part of post-menopausal treatment, when their estrogen production rapidly declines and when their risk of Parkinson’s disease increases. The studies that we collected serve to provide us data about not only how estrogenic compounds may impact Parkinson’s disease, but also about what other factors may alter the impact that these estrogenic compounds may have.
Literature Review
Yoo (2020) discussed how a later age of menarche was associated with increased Parkinson’s disease risk; an increased age of menopause and an increased fertility length were both associated with reductions in Parkinson’s disease risk; and hormone replacement therapy, oral contraceptives, and female reproductive factors were associated with increases in Parkinson’s disease risk2. Payami (2023) identified repeated blows to the head and pesticide exposure as preventable risk factors for Parkinson’s disease, and race, sex, and genetics as intrinsic risk factors for Parkinson’s disease4. Pesce (2022) discussed how early and late age at menarche, a higher number of children, artificial menopause (especially at a younger age), and some fertility treatments were associated with an increase in Parkinson’s disease risk, and how hormonal therapy was associated with a decrease in Parkinson’s disease risk9. These three papers all discuss various factors that increase the risk for Parkinson’s disease, and all of them include sex as a risk factor. Two of them identify hormonal therapy as a risk factor, but their conclusions are contradictory, with Yoo (2020) associating hormonal therapy with an increase in Parkinson’s disease risk, and Pesce (2022) associating hormonal therapy with a decrease in Parkinson’s risk2,9.
Six more papers directly analyze how estrogenic compounds affect Parkinson’s disease risk. Currie (2004) associated postmenopausal estrogen therapy with a decrease in Parkinson’s disease risk16. Simon (2009), on the other hand, found no such beneficial effect from any hormone replacement therapy17. Yuk (2023) found an elevated Parkinson’s disease risk with tibolone and estrogen that was most apparent in the first years of treatment, as well as a much more consistent increase in risk with regards to an estrogen-progesterone combination18. Liu (2020) found little correlation between hormone replacement therapy use and Parkinson’s disease risk, even when analyzing both the type of hormone and the duration of hormone usage19. Rao (2023) found no correlation between hormone replacement therapy and Parkinson’s disease20. Lundin (2014) found that esterified estrogen was the only formula of estrogenic hormone replacement therapy associated with Parkinson’s disease, and that conjugated estrogen was not associated with Parkinson’s disease21. All six of these papers focus on a similar subject but tend to differ in terms of formulations of hormones studied, focus on the effect duration of on Parkinson’s disease, and conclusions about the overall effect of estrogenic hormones on Parkinson’s disease.
Overall, existing literature extensively surveys estrogenic compounds and their effects on Parkinson’s disease, but the fairly low level of agreement between the studies and the widely differing focuses on duration and formulation necessitates an organized, systematic review of data that aggregates scattered data to form a more comprehensive view on the effects of estrogenic compounds on Parkinson’s disease, as well as a more organized look on how duration of use and formulation may alter their effect. Our systematic review intends to provide this organized view on estrogenic compounds’ effect on Parkinson’s disease.
Data Collection and Synthesis
Search Strategy and Data Collection
We used databases such as PubMed, ProQuest Research Library, and Medline, along with other academic tools such as Consensus, to find our articles. The main keywords we used were “Parkinson’s”, “estrogen”, and “women”. Inclusion criteria included a publication date from the year 2000 or later, with priority being given to more recently written articles; being primary research articles; and having been peer reviewed in order to ensure that the studies that we used are high quality. Exclusion criteria included being irrelevant to the topic of the effects of estrogenic compounds on Parkinson’s disease; focusing on Alzheimer’s disease; and only containing marginal information about Parkinson’s disease. For all of the studies that met our criteria, the methods, results, and discussion within the studies were used to find the data that was needed. We then used this data to make observations about each study. The data that we collected was also organized into tables and figures.
Data Synthesis
After collecting and filtering studies, we organized our quantitative data into three figures in the form of forest plots: one study about the effect of estrogenic compounds on the risk of Parkinson’s disease, one about how duration of use of estrogenic hormones affects the risk of Parkinson’s disease, and one about how the type of estrogenic hormone affects the risk of Parkinson’s disease. All three of these forest plots involve analyzing odds ratios, hazard ratios, and/or risk ratios; the type of ratio depicted depends on the studies being analyzed. These figures were made using Copilot. We also recorded qualitative data about each study, such as the main ethnicity studied, whenever it was feasible. Finally, we recorded our results, both quantitative and qualitative, in an organized manner.
Results of Comparative Analyses
Use of HRT: Out of the eleven studies that we collected, one study indicated a correlation between hormone replacement therapy and a reduction in the risk of Parkinson’s disease16; three studies indicated a correlation between hormone replacement therapy and an increase in the risk of Parkinson’s disease2,17,18; and four studies indicated no correlation or an inconsistent correlation between the two factors9,19,20,21. Overall, the effect that estrogenic hormones have on the risk of Parkinson’s disease is mixed, with some studies reporting an increase in risk while others report a decrease in risk.
Table 1 shows the different conclusions that each study on humans reached regarding the effects of hormone replacement therapy on Parkinson’s disease, as well as additional information regarding each study. Only two of the eight studies were case-control studies. The one study that associated estrogenic hormone usage with a reduced risk of Parkinson’s disease also had the lowest number of participants16. The study where all of its participants had Parkinson’s disease found no correlation between estrogenic hormone usage and Parkinson’s disease20. Three out of the four studies with more than 100,000 participants found a correlation between estrogenic hormone usage and an increased risk of Parkinson’s disease; these three studies also found that less than 1% of their participants had PD cases2,17,18.
| Study | Risk Association | Study Design Type | Total Participants | PD Cases Identified | Follow-Up Duration | Adjustment Variables Used |
|---|---|---|---|---|---|---|
| Currie (2004) | Decrease | Case-control | 140 | 68 | None | Age at menarche and menopause, menses duration, number of pregnancies, birth control pill use and duration |
| Simon (2009) | Cohort | 121,701 | 244 | 4 years* | Age, pack-years of smoking | |
| Yuk (2023) | Cohort | 303,260 | 0** | 7.9 years*** | Age and calendar year at inclusion, socioeconomic status, Charlson Comorbidity Index, region of residence, hypertension, diabetes, hyperlipidemia, uterine fibroids, endometriosis, previous hysterectomy, previous adnexal surgery | |
| Increase | ||||||
| Yoo (2020) | Cohort | 4,729,546 | 20,816 | 2 years**** | Age at menarche, age at menopause, number of parities, duration of breastfeeding, duration of HRT, duration of oral contraceptive use, alcohol consumption, smoking, regular exercise, income, BMI, hypertension, diabetes mellitus, dyslipidemia, cancer† | |
| Liu (2014) | Cohort | 119,166 | 410 | 8–11 years | Age in years at baseline, race, smoking status, caffeine intake | |
| Lundin (2014) | No effect | Case-control | 364 | 137 | None | Duration of enrollment in Group Health Cooperative pharmacy database†† |
| Pesce (2022) | Cohort | 96,862 | 1,165 | 22 years | Age, rural living††† | |
| Rao (2023) | Cohort | 304 | 304 | None | Age, disease duration, medication |
*: Or date of either Parkinson’s disease diagnosis, death, stroke diagnosis, loss to follow-up, or end of study 26 years later
**: Study assessed risk and systematically excluded people with Parkinson’s disease
***: Between 6.9 and 7.9 years
****: Or until date of incident Parkinson’s disease
†: For Model 2; Model 1 was unadjusted, and Model 3 was adjusted for duration for fertility instead of age of menarche and menopause
††: When covariates modified risk estimates by more than 10% and when chi-square statistic was statistically significant, covariates such as smoking, race, coffee consumption, education, history of estrogen-related cancers, and family history of PD were adjusted
†††: As a baseline; models that included characteristics of menopause and hormonal therapy were adjusted for menopausal status; models including the number of children and breastfeeding were adjusted for nulliparity; in model A1, data regarding age at menarche was adjusted for rural living and childhood silhouette; in model B1, data regarding parity was adjusted for rural living, age at menarche, childhood silhouette, silhouette at 20 years, smoking status, time-varying nulliparity, and parity; in model C1, data regarding menopause was adjusted for rural living, age at menarche, childhood silhouette, silhouette at 20 years, time-varying nulliparity, parity, smoking status, BMI, physical activity, menopausal status, type of menopause, and age of menopause; data for fertility treatments was further adjusted for rural living, age at menarche, childhood silhouette, silhouette at 20 years, and smoking status; post hoc composite score adjusted for baseline rural living
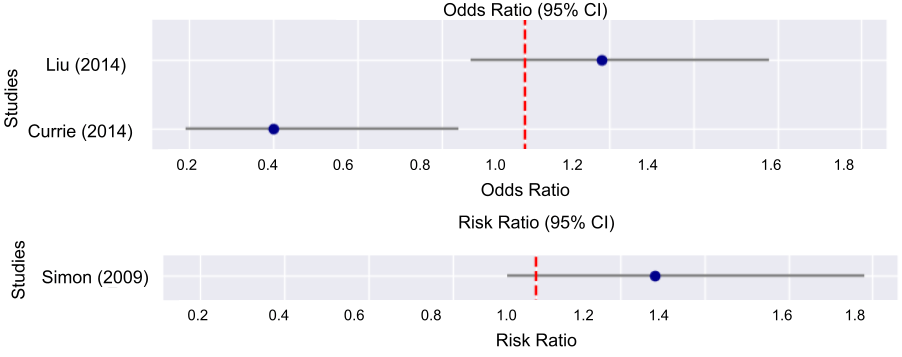
In Figure 1, we plotted the odds and risk ratios regarding estrogen’s impact on Parkinson’s disease from 3 studies done on humans. Each study reported a specific odds or risk ratio, along with the 95% CI ranges of each ratio. For Liu (2014), we used Model 1 adjustment, which means that its odds ratio is adjusted for age, race, smoking, and caffeine. This figure shows that there is an inconsistent correlation between administration of estrogen and the development of Parkinson’s disease.
Type of HRT: Overall, the effect of hormones on Parkinson’s disease depends on the hormone type; some hormones, such as estrogen, have an inconsistent effect on Parkinson’s disease risk, while others, such as progesterone, more consistently affect Parkinson’s disease risk in a certain way.
Table 2 describes the hormones used by five of the studies conducted on humans, as well as the distinct effects that these hormones had. Progestin and Progesterone were grouped together for our data analysis, as Progestin is a synthetic hormone that mimics Progesterone. The hormones used in the experiments were all different in terms of amount, kind, and duration, so the data will be affected by the heterogeneity of formulas.
| Study | Type of Hormone | Risk Correlation |
|---|---|---|
| Currie (2004) | Estrogen only | Decrease |
| Liu (2014) | Estrogen | |
| Estrogen + Progesterone | Increase* | |
| Yuk (2023) | Estrogen | |
| Estrogen + Progesterone | No effect** | |
| Tibolone | ||
| Yoo (2020) | Estrogen, Progesterone (Distinguishment was not made in data) | Decrease*** |
| Simon (2009) | Estrogen | |
| Progesterone | Increase | |
| Unknown Hormone | ||
| Estrogen + Progesterone | Decrease |
*: For all categories except “Formerly used Estrogen”, where a decrease in risk was noted
**: Increase or decrease in risk dependent on duration
***: Later changes to “Increase” as time goes on
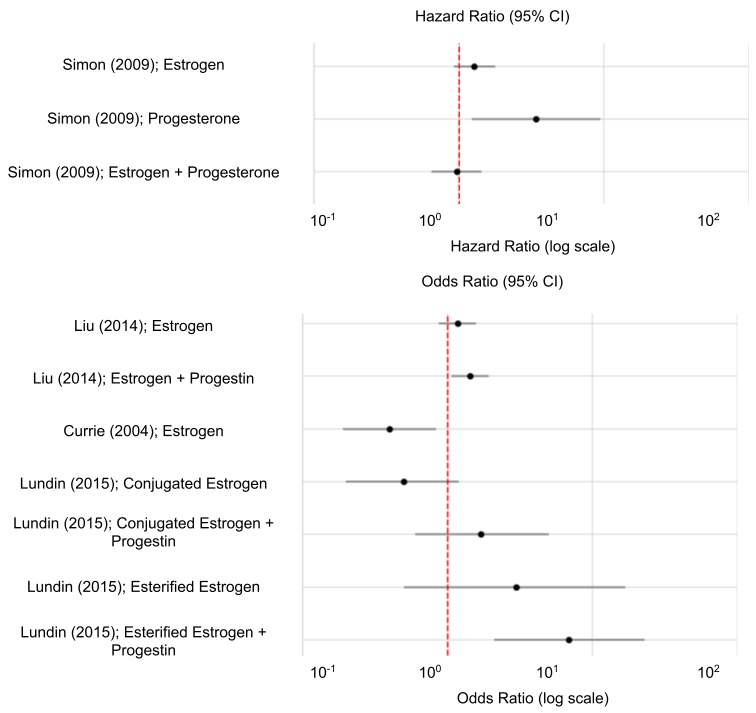
In Figure 2, we plotted the hazard and odds ratios associated with the types of estrogen used from four studies along with their 95% CI ranges. We used the Model 1 adjustment for the data from Liu (2014), which means that the data is adjusted for continuous age, race, smoking, and caffeine. The data indicates that estrogen and a combination of estrogen and progesterone have an inconsistent effect on Parkinson’s disease, while progesterone consistently increases the risk of Parkinson’s disease. Lundin (2014) also indicates that conjugated estrogen decreases the risk of Parkinson’s disease, and that a combination of conjugated estrogen and progestin, esterified estrogen, and a combination of esterified estrogen and progestin increase the risk of Parkinson’s disease21. Therefore, Figure 2 shows a correlation that implies that estrogen and a combination of estrogen and progesterone have an unclear effect on Parkinson’s disease; conjugated estrogen decreases the risk of Parkinson’s disease; and progesterone, esterified estrogen, and a combination of esterified or conjugated estrogen and progestin consistently increase the risk of Parkinson’s disease.
Duration of HRT: Overall, the longest and shortest durations tend to result in the highest risk increases for Parkinson’s disease, although this tendency is not absolute.
Table 3 shows the results obtained from four of the studies conducted on humans regarding the effects of duration of hormone replacement therapy on the risk level of Parkinson’s disease.
| Study | Duration | Risk Change |
|---|---|---|
| Simon (2009) | Fewer than 5 years | No effect |
| More than 5 years | ||
| Liu (2014) | Used in the past for fewer than 5 years | Decrease |
| Used in the past for greater than 5 years | Increase | |
| Currently using for less than 5 years | ||
| Currently using for more than 5 years | ||
| Yuk (2023) | Fewer than 1 year, 1–2.9 years | Increase* |
| 3–4.9 years, more than 5 years | Increase** | |
| Yoo (2020) | 2 or fewer years | |
| 2–5 years | Increase*** | |
| 5 or fewer years |
*: Estrogen and Tibolone showed a greater increase in risk for durations of less than 3 years than Estrogen + Progesterone
**: Estrogen + Progesterone showed a greater increase in risk for of durations 3 years of greater than Estrogen or Tibolone
***: Decreased before adjustment; increased after adjustment
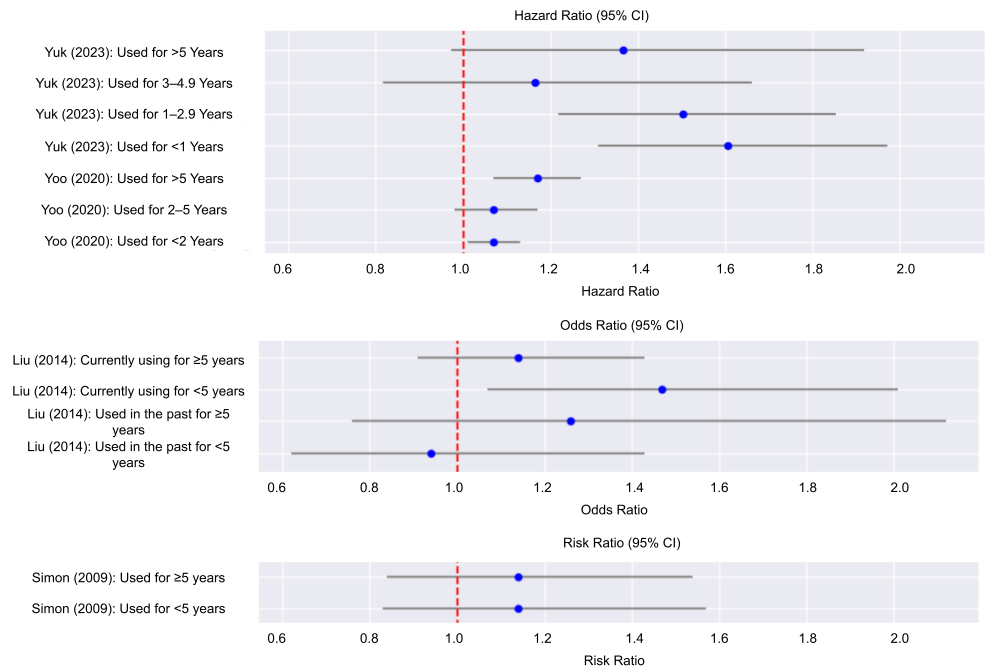
In Figure 3, we plotted the hazard ratios, odds ratios, and risk ratios associated with specific time intervals from four studies, along with their 95% CI ranges. We used Model 1 for data adjustment of Liu (2014), which means that its data is adjusted for continuous age, race, smoking, and caffeine. We used Model 3 for data adjustment of Yoo (2020), which means that its data is adjusted for duration of fertility, parity, duration of breast feeding, duration of HRT, duration of oral contraceptive use, alcohol consumption, smoking, regular exercise, income, body mass index, hypertension, diabetes mellitus, dyslipidemia, and cancer. The data shows that while hormone use generally increases the risk of Parkinson’s disease, the highest risks tend to appear at the shortest and longest periods of use.
Impact of Ethnicity: Out of 8 studies, 3 of them reported data on ethnicity. Liu (2014) reported that 91.0% of its non-PD subjects were non-Hispanic White, while 93.2% of its PD subjects were non-Hispanic White; 7.9% of its non-PD subjects were of another ethnicity, while 5.9% of its PD subjects were of another ethnicity19. Lundin (2014) reported that 93% of both its case and control groups were non-Hispanic White; this study did not provide data for other ethnicities21. Pesce (2022) noted that data on skin color may indicate that it used a mostly White study base, but due to restrictions on collecting ethnicity data due to French law, the study was unable to precisely verify ethnic data; as such, we did not count it as having reported data on ethnicity9. Both Liu (2014) and Lundin (2014) concluded that there was no overall effect on risk from Parkinson’s disease. Yuk (2023) reported in its discussion that all of its subjects were of Korean ancestry18. It concluded that there was some increase in the risk of Parkinson’s disease when estrogenic hormones were administered. Yuk (2023) found an increase in risk for the first 3 years, and cited in its discussion a study that indicated that Asian women may have different susceptibilities to Parkinson’s disease18. These factors suggest that ethnicity may affect how estrogenic compounds affect Parkinson’s disease, and ultimately their effectiveness in mitigating the risk and severity of Parkinson’s disease.
Conclusions and Discussion
Key Findings
Our analysis reveals complex and often contradictory effects of estrogenic hormones on PD risk and severity.
Human studies show inconsistent outcomes: of 8 reviewed, one reports reduced risk with exposure to estrogenic hormones, three indicate increased risk, and four show no effect or mixed results. Ethnicity emerges as the only consistent modulator, with divergent findings between reported East Asian and European populations. Hormone type also influences severity; progesterone, esterified estrogen, and esterified or conjugated estrogen combined with progestin generally exacerbate symptoms; conjugated estrogen alone is linked with alleviating symptoms; and estrogen and a combination of estrogen and progesterone have an unclear effect.
Implications
The observed ethnic divergence between East Asian populations and non-Hispanic White populations represents an informative finding that is supported by existing observations. Brakedal (2022) noted in its introduction that lower male/female sex ratios for Parkinson’s disease are reported from Asian populations1. Liu (2004) found in its study in Utah and California that Asian women had a much longer menstrual cycle than Caucasian women, which implies that Asians and non-Hispanic Whites may have different responses to certain drugs such as hormone replacement therapies8. However, the effect of ethnicity on the effectiveness of treatments for Parkinson’s disease is understudied, largely because non-White ethnicities are not well-represented in many studies of Parkinson’s disease. For example, Park (2023) found that Genome-wide association studies (GWAS), which apply to diseases such as Parkinson’s disease, are disproportionately conducted among White populations; at the time of the study, there was no GWAS data for the Korean population afflicted with Parkinson’s disease despite Korea having the fastest-aging society in the world, paired with a rapidly rising prevalence of Parkinson’s disease22.
Hormone formulation also has practical implications; women at an elevated Parkinson’s disease risk who are considering hormone replacement therapy for menopause may benefit from estrogen-only regimens over those including progesterone, given the latter’s association with an increased risk and severity of Parkinson’s disease.
Additionally, timing of therapy appears at least somewhat influential, with evidence for a potential “critical window” where hormone administration yields protective effects, outside of which risk may rise. Lord (2008) noted how past studies indicated that using estrogen or a combination of estrogen and progesterone may provide beneficial effects on the brain if a certain duration was not exceeded; long-term usage of a combination of estrogen and progesterone was linked with negative effects on the brain10. These findings are supported by our data, which links the highest and lowest durations of usage with the largest increase in Parkinson’s disease risk, although the studies that we used to observe duration broadly linked estrogenic hormones with an increase in Parkinson’s disease risk. Liu (2004) states based on data from past studies how the “timing hypothesis” states that the timing of estrogen treatment relative to the stage or type of menopause may crucially affect estrogenic hormones’ effect on Parkinson’s disease19; our data provides some support for this hypothesis.
Collectively, these insights underscore factors—ethnicity, formulation, and timing—that likely modulate estrogen’s therapeutic potential in PD and identify promising directions for targeted interventions.
Future Directions
Current studies predominantly focus on European and East Asian cohorts, limiting generalizability to African, Hispanic, and other populations. As such, judging from the potential for estrogenic hormones to have different effects on Parkinson’s disease depending on the ethnicity of the patient, more investigations regarding ethnic differences in the effect of estrogenic hormones on Parkinson’s disease should be conducted, especially on underrepresented groups such as people of African and Hispanic ethnicity.
In addition, we recommend further studies that search for the validity of a “critical window” with regards to using estrogenic hormones in order to alleviate the symptoms of Parkinson’s disease, as while some evidence exists for this hypothesis, there is still a lack of definitive data or proof regarding the existence or lack thereof of this “critical window”.
Further studies on estrogenic compounds’ effect on Parkinson’s disease are essential to resolve existing contradictions and establish estrogenic hormone based strategies as viable PD interventions.
As a result of this research, we believe that our study would provide a useful comparison of results from a variety of studies that all observe the effect of estrogenic compounds on Parkinson’s disease for future studies studying estrogenic compounds and their effects on Parkinson’s disease to use and reference. Our paper currently only holds research value, but it could be used to suggest or to guide a suggestion for a standard guideline for estrogen therapy for people with Parkinson’s disease.
Acknowledgements
I would like to recognize Dr. Yuefan Deng from Stony Brook University for his priceless guidance throughout the entire process of writing this research paper, especially with regards to data analysis. His experience and patience greatly helped me write a more sophisticated and nuanced paper.
References
- Brakedal, B., Toker, L., Haugarvoll, K., & Tzoulis, C. (2022b). A nationwide study of the incidence, prevalence and mortality of Parkinson’s disease in the Norwegian population. Npj Parkinson’s Disease, 8(1). https://doi.org/10.1038/s41531-022-00280-4. [↩] [↩] [↩] [↩]
- Yoo, J. E., Shin, D. W., Jang, W., Han, K., Kim, D., Won, H. S., & Park, H. S. (2020). Female reproductive factors and the risk of Parkinson’s disease: a nationwide cohort study. European journal of epidemiology, 35(9), 871–878. https://doi.org/10.1007/s10654-020-00672-x. [↩] [↩] [↩] [↩] [↩] [↩]
- Lee, Y. H., Cha, J., Chung, S. J., Yoo, H. S., Sohn, Y. H., Ye, B. S., & Lee, P. H. (2019). Beneficial effect of estrogen on nigrostriatal dopaminergic neurons in drug-naïve postmenopausal Parkinson’s disease. Scientific reports, 9(1), 10531. https://doi.org/10.1038/s41598-019-47026-6. [↩]
- Payami, H., Cohen, G., Murchison, C. F., Sampson, T. R., Standaert, D. G., & Wallen, Z. D. (2023). Population fraction of Parkinson’s disease attributable to preventable risk factors. Npj Parkinson’s Disease, 9(1). https://doi.org/10.1038/s41531-023-00603-z. [↩] [↩]
- Pont‐Sunyer, C., Hotter, A., Gaig, C., Seppi, K., Compta, Y., Katzenschlager, R., Mas, N., Hofeneder, D., Brücke, T., Bayés, A., Wenzel, K., Infante, J., Zach, H., Pirker, W., Posada, I. J., Álvarez, R., Ispierto, L., De Fàbregues, O., Callén, A., … Tolosa, E. (2014). The Onset of Nonmotor Symptoms in Parkinson’s disease (The ONSET PD Study). Movement Disorders, 30(2), 229–237. https://doi.org/10.1002/mds.26077. [↩] [↩]
- Vickers, M. R., MacLennan, A. H., Lawton, B., Ford, D., Martin, J., Meredith, S. K., DeStavola, B. L., Rose, S., Dowell, A., Wilkes, H. C., Darbyshire, J. H., & Meade, T. W. (2007). Main morbidities recorded in the Women’s International Study of Long Duration Oestrogen after menopause (wisdom): A randomised controlled trial of hormone replacement therapy in Postmenopausal women. BMJ, 335(7613), 239. https://doi.org/10.1136/bmj.39266.425069.ad. [↩]
- Tannen, R. L., Weiner, M. G., Xie, D., & Barnhart, K. (2007). Estrogen affects post-menopausal women differently than estrogen plus progestin replacement therapy. Human Reproduction, 22(6), 1769–1777. https://doi.org/10.1093/humrep/dem031. [↩]
- Liu, Y., Gold, E. B., Lasley, B. L., & Johnson, W. O. (2004). Factors affecting menstrual cycle characteristics. American Journal of Epidemiology, 160(2), 131–140. https://doi.org/10.1093/aje/kwh188. [↩] [↩]
- Pesce, G., Artaud, F., Roze, E., Degaey, I., Portugal, B., Nguyen, T. T., Fournier, A., Boutron-Ruault, M.-C., Severi, G., Elbaz, A., & Canonico, M. (2022). Reproductive characteristics, use of exogenous hormones and Parkinson’s disease in women from the E3N study. Brain, 146(6), 2535–2546. https://doi.org/10.1093/brain/awac440. [↩] [↩] [↩] [↩] [↩]
- Lord, C., Buss, C., Lupien, S. J., & Pruessner, J. C. (2008b). Hippocampal volumes are larger in postmenopausal women using estrogen therapy compared to past users, never users and men: A possible window of opportunity effect. Neurobiology of Aging, 29(1), 95–101. https://doi.org/10.1016/j.neurobiolaging.2006.09.001. [↩] [↩]
- Gatto, N. M., Deapen, D., Stoyanoff, S., Pinder, R., Narayan, S., Bordelon, Y., & Ritz, B. (2014). Lifetime exposure to estrogens and parkinson’s disease in California teachers. Parkinsonism & Related Disorders, 20(11), 1149–1156. https://doi.org/10.1016/j.parkreldis.2014.08.003. [↩]
- Mousavi, S. Z., Harris, E. R., Agarwal, S., Saha, P., Glenn, E. R., Fox, H. M., & Srikumaran, U. (2025). Impact of estrogen replacement therapy on outcomes following total shoulder arthroplasty: A propensity-matched retrospective cohort study of 1,779 patients. JSES International, 9(4), 1345–1351. https://doi.org/10.1016/j.jseint.2025.04.022. [↩]
- Sosero, Y., Bandres‐Ciga, S., Ferwerda, B., Tocino, M., Belloso, D., Gómez‐Garre, P., Faouzi, J., Taba, P., Pavelka, L., Marques, T., Gomes, C., Kolodkin, A., May, P., Milanowski, L., Wszolek, Z., Uitti, R., Heutink, P., van Hilten, J., Simon, D., … Gan‐Or, Z. (2024). Dopamine pathway and Parkinson’s risk variants are associated with levodopa‐induced dyskinesia. Movement Disorders, 39(10), 1773–1783. https://doi.org/10.1002/mds.29960. [↩]
- Dias, C. M., Leal, D. A., & Brys, I. (2022a). Levodopa-induced dyskinesia is preceded by increased levels of anxiety and motor impairment in Parkinson’s disease patients. International Journal of Neuroscience, 133(12), 1319–1325. https://doi.org/10.1080/00207454.2022.2079501. [↩]
- van Kessel, S. P., de Jong, H. R., Winkel, S. L., van Leeuwen, S. S., Nelemans, S. A., Permentier, H., Keshavarzian, A., & El Aidy, S. (2020). Gut bacterial deamination of residual levodopa medication for Parkinson’s disease. BMC Biology, 18(1). https://doi.org/10.1186/s12915-020-00876-3. [↩]
- Currie, L. J., Harrison, M. B., Trugman, J. M., Bennett, J. P., & Wooten, G. F. (2004). Postmenopausal Estrogen Use Affects Risk for Parkinson Disease. Archives of Neurology, 61(6), 886. https://doi.org/10.1001/archneur.61.6.886. [↩] [↩] [↩]
- Simon, K. C., Chen, H., Gao, X., Schwarzschild, M. A., & Ascherio, A. (2009). Reproductive factors, exogenous estrogen use, and risk of Parkinson’s disease. Movement disorders: official journal of the Movement Disorder Society, 24(9), 1359–1365. [↩] [↩] [↩]
- Yuk, J.-S., & Jeong, S. H. (2023). Association between menopausal hormone therapy and risk for Parkinson’s disease. Journal of Parkinson’s Disease, 13(8), 1357–1367. https://doi.org/10.3233/jpd-230230. [↩] [↩] [↩] [↩] [↩]
- Liu, R., Baird, D., Park, Y., Freedman, N. D., Huang, X., Hollenbeck, A., Blair, A., & Chen, H. (2014). Female reproductive factors, menopausal hormone use, and Parkinson’s disease. Movement disorders: official journal of the Movement Disorder Society, 29(7), 889–896. https://doi.org/10.1002/mds.25771. [↩] [↩] [↩] [↩]
- Rao, S. C., Li, Y., Lapin, B., Pattipati, S., Ghosh Galvelis, K., Naito, A., Gutierrez, N., Leal, T. P., Salim, A., Salles, P. A., De Leon, M., & Mata, I. F. (2023). Association of women-specific health factors in the severity of Parkinson’s disease. NPJ Parkinson’s disease, 9(1), 86. https://doi.org/10.1038/s41531-023-00524-x. [↩] [↩] [↩]
- Lundin, J. I., Ton, T. G. N., LaCroix, A. Z., Longstreth, W. T., Franklin, G. M., Swanson, P. D., Smith‐Weller, T., Racette, B. A., & Checkoway, H. (2014). Formulations of hormone therapy and risk of Parkinson’s disease. Movement Disorders, 29(13), 1631–1636. https://doi.org/10.1002/mds.26037 [↩] [↩] [↩] [↩]
- Park, K. W., Ryu, H.-S., Shin, E., Park, Y., Jeon, S. R., Kim, S. Y., Kim, J. S., Koh, S.-B., & Chung, S. J. (2023). Ethnicity- and sex-specific genome wide association study on Parkinson’s disease. Npj Parkinson’s Disease, 9(1). https://doi.org/10.1038/s41531-023-00580-3. [↩]