Abstract
Cardiovascular disease remains the leading cause of death worldwide, contributing to over 17 million deaths annually and affecting millions more through chronic illness and disability. These disorders often result in irreversible damage to the heart that current treatments cannot fully repair. Current medications can only help manage the symptoms without treating the underlying condition itself, while surgical interventions come with complications such as immune rejection and limited durability. To address these shortcomings, researchers are exploring stem cell-derived cardiomyocytes, which are functional heart cells that can exhibit the same contractile behavior and express the same chemical markers as those found in the human heart. These cells can be cultured in heart-on-chip systems, which are microfluidic platforms that mimic the heart’s physiologic conditions and enhance cardiomyocyte maturation. This can enable more accurate disease modeling and drug testing. Another promising approach is bioprinting, a tissue engineering technique that uses 3D printing to create functional cardiac tissue by layering biomaterials and patient-derived cells. This technology has successfully manufactured subsections of the heart, such as valves, demonstrating its potential for regenerative medicine. However, bioprinting a fully functional human heart remains challenging due to the complexity of replicating the heart’s intricate structure and conditions, particularly the vascular networks needed to sustain cells. Advancements in methods for bioprinting and incorporating functional blood vessels are necessary to pave the way for bioengineered human hearts, which will offer more effective and lasting solutions for cardiovascular disease patients.
Introduction
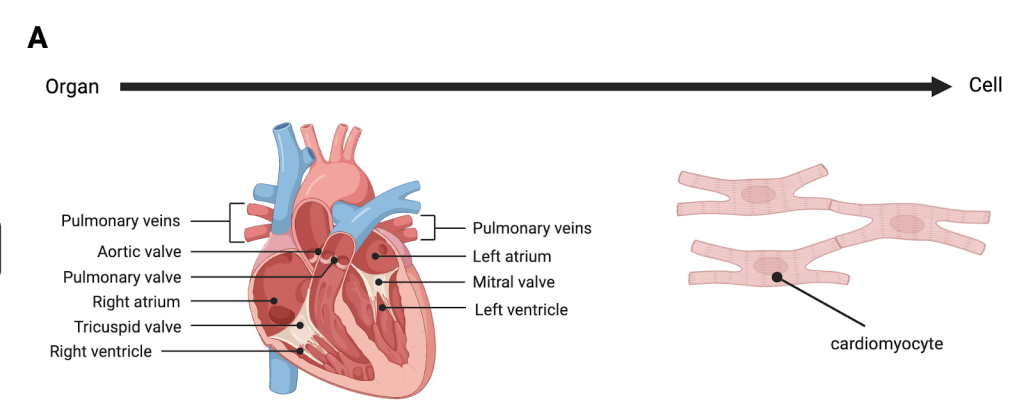
Each year, an estimated 17.9 million people die from cardiovascular disease, making it the leading cause of death worldwide1. Despite affecting populations across all income levels, four in every five of these deaths occur in low- and middle-income countries2. These regions often face limited access to long-term care and advanced treatments and medical technologies. As a result, the burden of cardiovascular disease continues to grow, both in terms of mortality and long-term disability. These diseases weaken the muscle of the heart and damage its structural parts, disrupting proper blood flow, depriving the body of oxygen and vital resources, and allowing for the accumulation of cellular waste. When left untreated, cardiovascular disease can lead to life-threatening repercussions. One example is a myocardial infarction, where reduced blood flow causes the death of heart muscle tissue. In more severe cases, this can progress to heart failure3. Cardiovascular diseases are a primary public health concern due to the importance of the human heart as a vital organ. The heart circulates blood throughout the body, transporting oxygen and nutrients to tissues while removing waste products. To perform these functions, it relies on an intricate system of ventricles, arteries, veins, and valves (Fig. 1a).
Two out of the four chambers of the heart are called ventricles and control the organ’s pumping function. The right ventricle pumps deoxygenated blood to the lungs, while the left ventricle pumps oxygenated blood to the rest of the body. Arteries and veins serve as pathways for blood flow. Arteries transport blood away from the heart to the rest of the body, whereas veins transport blood to the heart. In order to move blood against gravity, veins rely on valves. The heart has four valves—the mitral, tricuspid, aortic, and pulmonary valves—that open and close to facilitate the flow of blood in the necessary direction. When any of these components malfunction, the consequences can be devastating for human health.
Currently, many of the treatment options available, including medications and lifestyle changes, are not curative and instead only alleviate symptoms. In their most extreme cases, cardiovascular diseases can only be addressed via surgical replacement of the affected part or even the entire heart. However, engineered subsections of the heart face biocompatibility issues and other limitations that can lead to poor patient outcomes4. For example, when surgically treating valvular diseases, the defective valve is replaced with either a mechanical valve or a biological valve composed of cow or pig tissue. Tissue valves deteriorate rapidly, creating the need for another replacement operation. On the other hand, mechanical valves, though more durable, raise the risk of the formation of blood clots on their metal surfaces4. Additionally, the five year survival rate for heart transplant recipients is around 72 percent, with recipients often facing post-surgical complications and requiring lifelong immunosuppressive treatments5.
These limitations have driven the exploration of stem cell technology in regenerating cardiac tissue. Stem cells are uniquely capable of developing into many types of cells in the body. Early stem cell research utilized embryonic stem cells. However, much of this work was limited by cell availability and ethical concerns6. Since 2006, the focus has shifted to induced pluripotent stem cells (iPSCs), which are created by reprogramming patient-derived adult cells into a pluripotent state through the upregulation of specific transcription factors; thus eliminating the need to harvest embryonic stem cells and creating patient-specific stem cells7. iPSCs can be differentiated into cardiomyocytes by introducing differentiation reagents that activate cell signaling pathways to guide iPSCs through developmental stages that mimic natural heart cell formation, leading to the formation of functioning cardiac cells8.
Stem cell-derived cardiomyocytes not only provide opportunities for regenerative treatment of cardiovascular disease, but also serve as a means of drug screening and disease modeling that is better aligned with physiological conditions. This includes heart-on-chip systems, in which the differentiated cardiomyocytes are cultured in microfluidic chip systems. These systems simulate the physiological environment of the heart, allowing researchers to test drug responses and study the progression of cardiac diseases9,10,11.
Differentiated cardiomyocytes also lead to advancements in 3D bioprinting for subsets of the heart as well as whole organ regeneration. This technique couples biomaterials and living cells, such as cardiomyocytes, to create tissue structures, enabling the creation of organs and their components. As stem cell and bioprinting technologies continue to advance, they hold the potential to revolutionize cardiovascular medicine by providing more effective, personalized, and long-term solutions for heart disease patients.
Cardiomyocyte differentiation
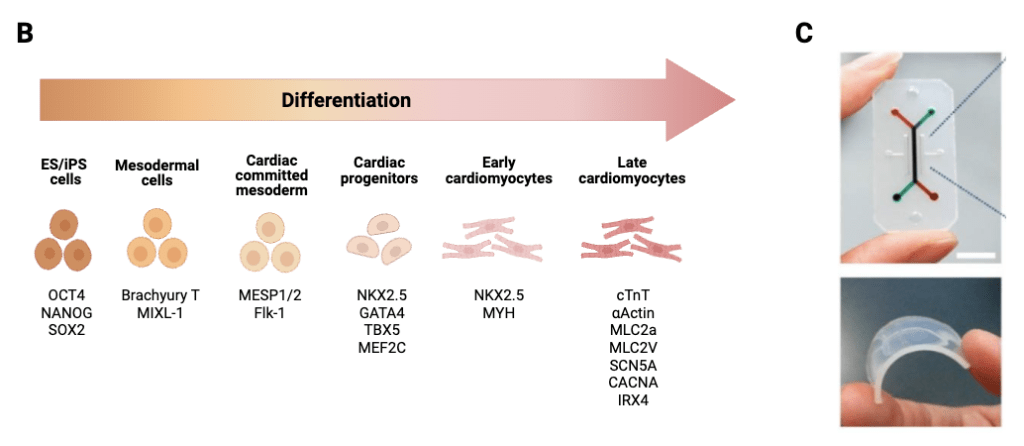
iPSCs differentiate into cardiomyocytes following several key stages in which cell signaling pathways are activated by reagents in order to drive cardiac development (Fig. 1b). First, the iPSCs are signaled to induce the formation of the “primitive streak mesoderm,” a layer of cells in early embryos capable of developing into a variety of bodily tissues. Next, the primitive streak mesoderm’s trajectory of development is specified as the cardiac mesoderm; this is the group of embryonic cells that form the endocardium (the inner lining of the heart that provides structural support for the valves and allows for blood circulation) and the myocardium (the muscular tissue of the heart that does the pumping)12. Then, cardiac progenitors, which are the specialized stem cells for the development of cardiac cell types, will be formed. These progenitors mature into functional cardiomyocytes. These cells express markers also seen in adult cardiomyocytes such as cardiac troponin-T (cTNT), myosin light chain 2 (MLC2a), and sodium channel protein type 5 subunit alpha (SCN5a).
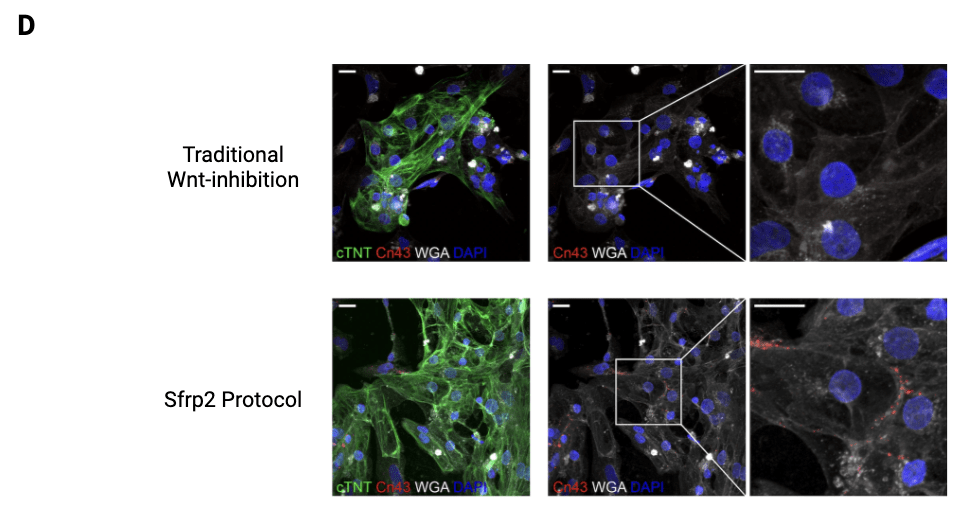
Unfortunately, many of the early approaches to differentiation protocols struggled to produce sufficiently mature cardiomyocytes with the proper structural and functional properties, such as cell size and beating frequencies. The induced cardiomyocytes often stagnate in the fetal stage and are unable to survive implantation. Hsueh et al. (2023)13 investigated this problem contributing to cardiomyocyte immaturity and implicated the pharmacological inhibitors used to mature progenitors into cardiomyocytes. Specifically, at the cardiac progenitor stage, previous protocols relied on inhibitors of the Wnt activation pathway for differentiation into early cardiomyocytes. Although this was successful in driving early cardiomyocyte differentiation, the indiscriminate targeting of all Wnt proteins stunted certain downstream pathways necessary for complete cardiomyocyte maturation. To overcome this, Hsueh et al.13 used secreted frizzled-related protein 2 (Sfrp2), which more selectively binds to and inhibits specific Wnt proteins rather than the entire Wnt pathway, allowed for differentiation into early cardiomyocytes while permitting later stages of cardiomyocyte differentiation and maturation. Throughout this new differentiation process, successfully differentiated and maturing cardiomyocytes were identified by tracking certain characteristics within the cells. In addition to the presence of cardiac markers such as cTNT, further characteristics indicating maturation were also observed after evaluating several structural and functional features of the cardiomyocytes. For example, sarcomere length was significantly increased in Sfrp2-treated cells, showing greater structural maturity as longer sarcomeres are essential for the strong contractions of adult cardiomyocytes. Additionally, these cells showed lower circularity indices, meaning they had adopted a more elongated shape typical of adult cardiomyocytes, rather than the rounded shape seen in immature cells. In terms of functionality, the spontaneous beating frequency was also significantly reduced in the Sfrp2 group, with a mean of ~25 Hz compared to ~40 Hz in WntC59-treated cells (p < 0.05), consistent with a more mature phenotype. Electrophysiological measurements further supported this: Sfrp2-treated cells showed a longer action potential duration at 90 percent repolarization (APD90), increasing from a median of ~45 ms in the control to ~75 ms with Sfrp2 treatment. This suggests improved electrical maturity and stability, characteristics of fully developed ventricular cardiomyocytes. Finally, gap junctions—structures only seen in mature cardiomyocytes that allow electrical signals to travel between cells—were not only present, but also polarized, reflecting coupling between cardiomyocytes13 (Fig. 1d).
A table comparing Sfrp2-based differentiation to traditional Wnt inhibition is shown below to clarify differences in mechanism and outcomes:
| Feature | Traditional Wnt-inhibition | Sfrp2 Protocol |
| Mechanism of action | Non-selective inhibition of all Wnt proteins | Selective inhibition of Wnt proteins |
| Sarcomere length (μm) | Shorter (~1.5–1.8 μm) | Longer (~2.2–2.5 μm) |
| Circularity index | ~0.5 (more rounded) | ~0.25 (more elongated, adult-like) |
| Beating frequency (Hz) | ~40 Hz | ~25 Hz |
| APD90 | ~45 ms (shorter and more immature) | ~75 ms (prolonged and more mature) |
| Gap junctions | Sparse or absent | Present and polarized |
While promising, the Sfrp2 protocol requires further testing. Hsueh et al.13 observed stable maturation over the span of 40 days, but longer-term studies are needed to confirm whether the observed maturation effects are stable over time and adaptable to varied culture conditions. The study does not yet explore practical aspects like cost, efficiency, or scalability, which are important for translating this method beyond the proof-of-concept stage. Continued progress may depend on recreating aspects of the embryonic heart environment, such as extracellular matrix composition, nutrient delivery, and oxygen gradients, to support full tissue development.
Current developments in and applications of heart-on-chip technology
Heart-on-chip technology couples induced cardiomyocytes with microfluidic chip systems in order to more accurately recapitulate the physiologic conditions of the human heart14. These systems consist of four main parts: the microfluidic chip, the cardiac microtissues, the microactuators, and the microsensors. The two most vital components are the microfluidic chip and the cardiac microtissues. The chip serves as the environment in which the cells can grow and is most popularly made of polydimethylsiloxane (PDMS), a polymer organic silicon compound, but can also be made of other materials including glass, papers, and, more recently, printable materials such as thermoplastic polyurethane (TPU) and polycaprolactone (PCL) (Fig. 1c). The second key component, the cardiac microtissues, are the constructs that mimic the human heart. These tissues are primarily composed of induced cardiomyocytes, as well as human cardiac fibroblasts (hCFs), which are supportive cells that provide connective tissue and structure for the heart, and endothelial cells, which line blood vessels.
The final two parts of microfluidic chip systems are more recent additions resulting from improvements in fabrication methods like 3D bioprinting. First, there are microactuators, which act as external stimuli—mechanical, electrical, or chemical—on the microtissues in order to create certain behaviors or induce and catalyze processes like maturation. For example, electrical stimulation via electrodes can increase the quantity of beating cells and improve cell synchronization. Second, there are microsensors, which monitor the status of the cells and assess how well the tissue is functioning, often by measuring the force of cardiomyocyte contraction. This can be done by using a bendable elastic material. When the cardiomyocytes contract, they exert a force on the elastic material, causing it to bend or deform. The contraction force can then be calculated based on how significant the visible deformation of the material is under a microscope15.
Overall, by providing a replica of the cellular conditions of the human heart and the means to observe the microtissues in real time, heart-on-chip technology is highly advantageous to disease modeling and drug screening. To start, the specific mechanisms and complications of a cardiac disease can be monitored using heart-on-chip systems. In a study of coronary heart disease, in which the narrowing or blockage of arteries impede the flow of blood and therefore oxygen, a heart-on-chip was developed to simulate coronary artery blockages and observe the heart muscle’s response of myocardial hypoxia, or oxygen deprivation, and its effects16. The model clearly showed observable symptoms that revealed the mass apoptosis that had occurred, including cell shrinkage, loss of mitochondrial membrane potential, and cytoskeletal breakdown. Other conditions that have been similarly studied include arrhythmia, cardiac fibrosis, and myocardial infarction17. In all three cases, the 3D heart-on-chip models provided a better assessment of the specific biological responses to these diseases and the efficacy of the drugs being tested; for example, the drug pirfenidone was observed to significantly reduce tissue stiffness resulting from cardiac fibrosis17.
Furthermore, coupling human-derived cells with the heart-on-chip system enables researchers to directly assess how the human body will react to a drug designed to treat a certain disease or particular symptoms. This expedites and reduces costs of the drug screening process as it removes the additional step of studying animal models, which are not ideal for modeling the human heart due to anatomical and physiological discrepancies18. To better contextualize these benefits, it is helpful to also compare heart-on-chip systems with traditional 2D culture models. Heart-on-chip systems provide a more physiologically relevant environment than 2D cultures by better mimicking the structure and conditions of real heart tissue. While 2D models confine cells to a flat surface, chip-based platforms enable three-dimensional organization, exposure to fluid flow, and interaction with multiple cell types. These conditions help the cells align better, beat more forcefully and regularly, and respond to drugs in a way that more closely reflects in vivo behavior.
Despite these advantages, heart-on-chip systems still face limitations. For example, current heart-on-chip models fall short in mimicking chronic cardiovascular diseases such as arrhythmias, fibrosis, and chronic ischemia. Most existing models cannot sustain viable cardiac tissue long enough to capture these slow-progressing diseases. Additionally, even with techniques like the Sfrp2 protocol improving cardiomyocyte maturation, stem cell-derived cardiomyocytes still typically show less developed physical structures and often exhibit immature electrophysiological properties. Recent innovations, such as the incorporation of primitive macrophages, have helped address these issues. In a study by Nature Cardiovascular Research, macrophages helped stabilize perfusable vasculature for over two weeks, countering the rapid vessel regression seen in earlier systems. These cells also secreted pro-angiogenic and cardioprotective factors that promoted cardiomyocyte maturation and tissue viability, effectively extending the model’s functional lifespan and enabling longer-term studies of disease progression19.
Ultimately, heart-on-chip technologies are still limited in their ability to directly treat the patient. This is especially apparent when diseases progress to life-threatening stages where immediate therapies are necessary. Heart-on-chip systems also face challenges in achieving consistent results, as variations between patient-derived cells can lead to unpredictable outcomes, complicating their use in personalized medicine. These differences, including genetic background, donor age, or disease state, can influence how cells respond to drugs or mimic disease phenotypes, making it harder to draw generalizable conclusions. As research on heart-on-chip systems continues to advance, other regenerative technologies such as bioprinting offer approaches that move beyond disease modeling to actively repairing or replacing damaged cardiac tissue.
Bioprinting subsections of the heart
Bioprinting, a tissue engineering technique in which 3D printing technology is used to create functional cellular structures, offers a promising advancement to regenerative medicine. In the context of bioprinting subsections of the heart, functionality refers to the ability of printed constructs to support viable cells that exhibit appropriate biological behavior, such as survival, organization, and tissue-specific activity such as contraction or vascular support.
In 3D bioprinting, biomaterials can mimic real human tissue by being precisely deposited layer by layer to build the desired structure. This method provides several unique advantages over traditional treatments for cardiovascular diseases. First, it offers the accurate and reproducible positioning of cells in a 3D space. Second, bioprinting is capable of generating customized structures from patient-derived cells, reducing the risks of transplant failure and immune rejection. Finally, an expansive variety of biomaterials can be used based on what is needed.
Bioprinting approaches can be classified based on the printing modality used to deposit cells and biomaterials, which directly influences resolution, cell viability, scalability, and material compatibility. Extrusion-based bioprinting is the most commonly used approach in cardiac applications because it can print viscous biomaterials and high cell densities, though shear forces during extrusion limit resolution and can stress cells. Laser-assisted bioprinting enables much finer spatial resolution and precise cell placement, but the localized energy used during printing can reduce the viability of sensitive cell types and makes the method less practical for fabricating large, three-dimensional cardiac structures. Inkjet bioprinting allows rapid deposition but is restricted to low-viscosity materials and low cell concentrations, making it less suitable for mechanically robust cardiac constructs. Light-based techniques such as stereolithography provide excellent structural accuracy but are constrained by a narrow range of photocurable materials and potential phototoxic effects20. As a result, different printing modalities are better suited to different cardiac structures, depending on whether fine spatial control, cell survival, mechanical integrity, or anatomical scale is prioritized.
Across these printing modalities, bioprinting strategies are often divided into two material-based categories: acellular printing, which involves using biomaterials like collagen and gelatin, and cellular printing, which uses bioinks. These are substances that combine living cells with biocompatible materials to support cell growth and tissue formation during and after printing21.
Acellular printing is most commonly used to generate scaffolds that mimic the extracellular matrix (ECM) that cells naturally inhabit, providing a foundation for cell maturation and tissue growth. A key component of the cardiac ECM is collagen, a protein that offers mechanical reinforcement and organization and that stores cell adhesion and signaling molecules. Lee et al. (2019)22 bioprinted collagen using a method called freeform reversible embedding hydrogels (FRESH), where bioink is extruded into a support bath of hydrogel that holds the material in place during the printing process and melts away at 37 degrees Celsius23 (Fig. 2a). With this approach, multiple components of the heart were successfully printed, including multiscale vasculature. This included a simplified model of a coronary artery, a left ventricle, and a tri-leaflet valve at an adult human scale (Fig. 2b). The collagen walls also demonstrated stability and structural integrity, meeting critical requirements for functionality.
It is important to note that despite the widespread use of collagen in cardiac bioprinting, it has key limitations when considered as a standalone biomaterial for functional myocardial tissue. Native cardiac muscle relies on rapid electrical signal propagation to coordinate contraction, yet collagen-based matrices are electrically insulating and do not actively facilitate electrical coupling between cardiomyocytes. As a result, collagen scaffolds alone may support cell attachment and organization but are insufficient for promoting synchronized electrical activity. To address the lack of electrical conductivity in collagen-based scaffolds, researchers have explored the incorporation of electrically conductive components—such as gold nanoparticles, carbon-based nanostructures, and conductive polymers—into biocompatible matrices to better approximate the native myocardial electrical microenvironment. These electroconductive biomaterials have been shown to support propagation of electrical signals and more coordinated activity among cardiomyocytes in engineered cardiac tissues compared with non-conductive scaffolds24. Decellularized extracellular matrix (dECM) has also emerged as a promising alternative biomaterial, as it preserves tissue-specific biochemical cues and structural complexity derived from native myocardium25. Together, these approaches illustrate that while collagen provides a useful foundation for cardiac bioprinting, additional material modifications or alternatives are often required to better support physiologically relevant cardiac function.
Furthermore, because cardiac vasculature requires hollow, perfusable channels rather than solid filaments, other extrusion-based approaches have been developed to address this. One example is coaxial extrusion, which uses concentric nozzles to print a core and surrounding shell simultaneously, allowing tube-like, vessel-mimicking structures to be fabricated directly within cardiac scaffolds26.
In addition to various parts of the heart, acellular printing can also be used to develop cardiac patches, or synthesized sections of heart tissue that can be used to replace damaged parts after myocardial infarction to prevent the deterioration of the entire organ18. In order to develop these patches beyond scaffold materials that temporarily support the impaired tissue, progenitor cells that can differentiate into cardiomyocytes are crucial for healing the tissue. Therefore, cellular printing takes the process a step further by incorporating living cells directly into the printed scaffold material (Fig. 2c). In the development of cardiac patches specifically, the cardiac progenitor cells work together alongside bioactive factors like intracellular signaling molecules and growth factors to help repair infarcted areas27.
Overall, cellular printing allows for the formation of constructs with living cells that can regenerate and integrate with a patient’s native tissues. Furthermore, to optimize the function of these printed tissues, the introduction of endothelial cells, which line blood vessels, is critical for establishing vascular networks that circulate oxygen and nutrients. However, different types of endothelial cells play distinct roles in the cardiovascular system. Arterial endothelial cells are specialized for high-pressure environments and tend to align with blood flow, while venous endothelial cells are more permeable and respond differently to signaling cues. Microvascular endothelial cells, which make up capillaries, are best suited for fine-scale nutrient and gas exchange. These distinctions affect how blood vessels form and function within engineered tissue28. Principally, the incorporation of endothelial cells with the bioprinted tissue constructs facilitates tissue engraftment by promoting the formation of blood vessels, ensuring that the implanted tissue receives adequate nutrient supply and oxygenation. By incorporating endothelial cells suited to their intended vascular role and printing biologically functional heart components such as contractile muscle tissue or vessels capable of carrying blood, researchers can better support tissue engraftment, ensure adequate perfusion, and develop more targeted therapies for conditions like myocardial infarction or valve disease.
This growing ability to engineer functional heart tissue raises important considerations about the types of bioprinting strategies best suited for different clinical goals. Both cellular and acellular bioprinting come with specific benefits and trade-offs. Acellular scaffolds offer mechanical strength and structural precision, making them ideal for recreating the physical architecture of heart tissue. They also offer high resolution, achieving ~50 µm resolution with pure biomaterials, enabling anatomically precise constructs ideal for structural support28. However, because they lack living cells, these scaffolds cannot actively contribute to tissue regeneration and must instead rely on the body to populate the structure with host cells over time. In contrast, cellular bioprinting embeds living cells directly into the printed material, offering the potential for immediate biological activity and integration with surrounding tissue. This approach comes with challenges in printability, long-term survival, and high resolution. In particular, maintaining uniform cell distribution and preventing stress-induced apoptosis during printing remain key challenges. Furthermore, when high cell densities are used, extrusion-based systems often struggle to maintain fine feature resolutions, dropping to 200–500 µm29.
For example, Papaioannou et al. compared cellular and acellular printing strategies for vascular grafts. Collagen-based acellular scaffolds demonstrated greater mechanical strength, as shown by higher burst pressures, making them well-suited for structural applications like arterial repair. In contrast, grafts printed with endothelial and smooth muscle cells showed enhanced remodeling and endothelial coverage after implantation, supporting better long-term integration with host tissue30.
This comparison highlights that mechanical properties play an important role in determining how acellular and cellular bioprinted constructs perform. Cardiac tissue is elastic and undergoes repeated stretching with each heartbeat, but it is also anisotropic, meaning its stiffness differs depending on direction because cardiac muscle fibers are aligned. These mechanical features affect how cardiomyocytes orient themselves and coordinate contraction. In an experimental study by Engler et al.31, cardiomyocytes were cultured on materials engineered to span a range of stiffness values. Substrates with stiffness similar to native heart tissue provided enough resistance for cells to anchor and generate force, allowing them to form organized contractile structures and beat rhythmically. When the material was more compliant, meaning it deformed too easily under cellular forces, cells were unable to generate sufficient tension and showed weaker, poorly coordinated contractions. Conversely, when the material was much stiffer than native myocardium, cells exhibited disorganized contractile structures and irregular beating. These results demonstrate that both excessively soft and excessively stiff environments can impair cardiac cell function. In bioprinting, this helps explain why acellular constructs with higher mechanical strength are suitable for structural support, while cellular constructs require a carefully tuned balance of stiffness and compliance to support tissue remodeling and long-term integration.
A comparative summary of the benefits and trade-offs of acellular and cellular bioprinting is shown below:
| Feature | Acellular Bioprinting | Cellular Bioprinting |
| Printing resolution | Higher; achieves ~50 µm resolution with pure biomaterials | Lower; typically ranges from 200–500 µm |
| Maturation capacity | Lower; relies on post-printing cell seeding, so tissue development and specialization happen slower | Higher; living cells are printed directly, allowing for earlier tissue growth and maturation |
| Mechanical strength | Stronger; printed scaffolds can support physical loads and maintain shape | Weaker; softer bioinks result in more fragile constructs |
| Vascularization | Limited; depends on host blood vessels growing in after implantation | Can be designed with endothelial cells to help form blood vessels and improve nutrient delivery |
| Ideal use case | Structural implants (e.g., heart valves, vascular grafts) | Regenerative patches for damaged heart tissue |

Beyond mechanical and vascular considerations, immune compatibility remains a significant challenge for bioprinted cardiac constructs. Although patient-derived induced pluripotent stem cells are often used to reduce the likelihood of rejection, experimental evidence shows that autologous origin does not fully eliminate immune responses. As reviewed by Scheiner et al.32, transplantation studies in genetically matched animal models found that iPSCs and some iPSC-derived tissues could still trigger immune cell infiltration and fail to persist after implantation. These effects were linked to changes introduced during reprogramming, such as abnormal gene expression or incomplete resetting of epigenetic marks, which can cause transplanted cells to display proteins recognized as foreign by the immune system32. Because these immune responses were observed in small grafts and localized implants, they raise concern that immune challenges may become more pronounced as bioprinted constructs increase in size and complexity, particularly in the context of whole-organ engineering.
While printing cardiac patches and individual structures is a major step forward, these partial solutions aren’t enough for patients with end-stage heart failure. As bioprinting technology advances, the next challenge lies in scaling these constructs into whole organs capable of supporting life.
Organ-scale 3D cardiac bioprinting
The ultimate goal of regenerative medicine is the fabrication of full-size, functional organs—a necessity for patients with end-stage cardiovascular diseases, where heart transplantation has become the only viable treatment plan. In the context of organ-scale cardiac bioprinting, functionality refers to physiological performance rather than structural resemblance alone. Specifically, functional cardiac constructs are expected to contain viable cardiomyocytes capable of coordinated, tissue-level contraction and electrical coupling, as well as sufficient vascular support to maintain cell survival over time.
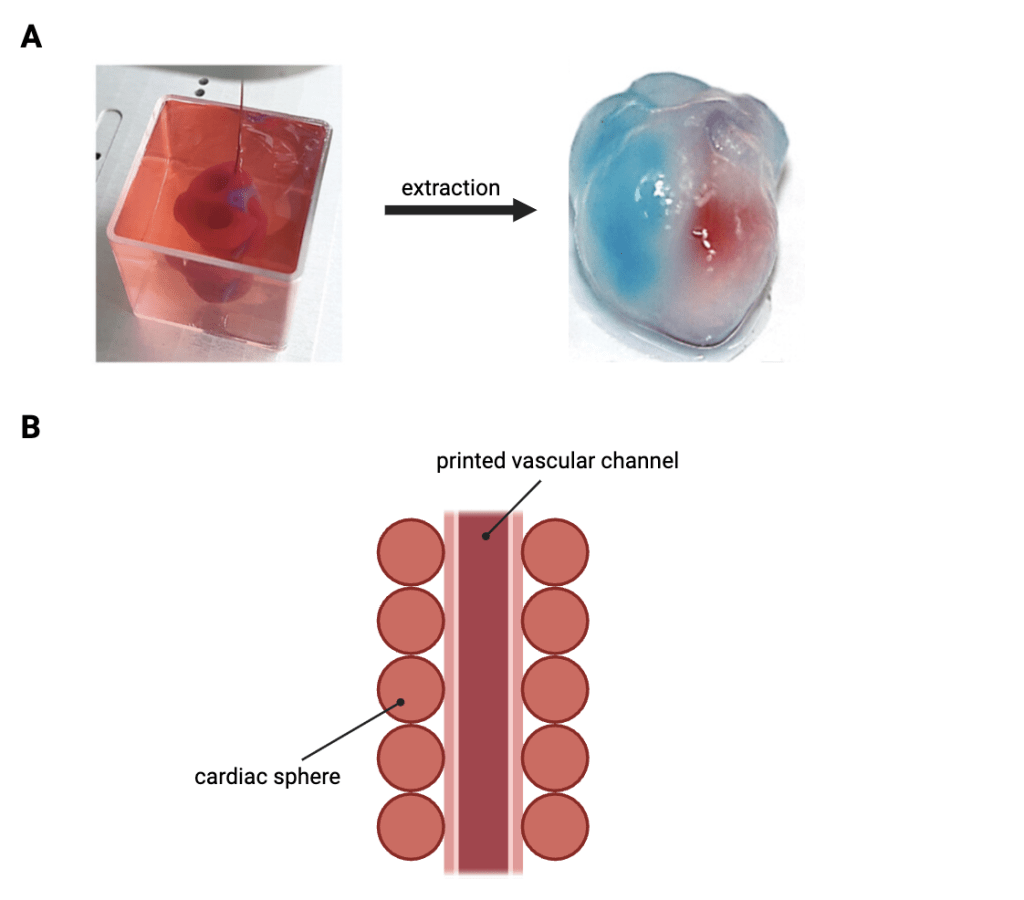
So far, the majority of bioprinted models of the heart lack cells and blood vessels. Even those with cells incorporated are limited in physical size, largely due to the challenge of achieving sufficient and sustainable vascularization for larger structures. In the absence of effective vascularization, cells deeper within the layers of the tissue rapidly experienced oxygen deprivation, making it impossible for the printed heart to survive and remain stable following implantation. Noor et al. (2019)33 bioprinted the first vascularized model of a heart, complete with chambers and other essential components required for cardiac function. In order to create this model, a patient’s omentum tissue was extracted and the fat cells were reprogrammed into iPSCs, which were subsequently differentiated into both cardiomyocytes and endothelial cells. The matrix from the extracted tissue was processed into a hydrogel, in which the differentiated cells were then placed into to develop a bioink tailored to the patient. The completed heart was incapable of beating and was significantly smaller than a human heart (Fig. 3a), instead resembling the size of a rabbit heart as it measured 14 millimeters in diameter and 20 millimeters in height33.
To overcome the limitations of previous bioprinting approaches and generate viable, perfusable cardiac tissues with the required cell density for human organ engineering, other researchers have used cardiac spheres—self-assembled spherical clusters of cardiomyocytes—as building blocks for printing cardiac tissues34. For this, temporary structures called sacrificial pathways were printed into a granular cardiac tissue matrix and evacuated, leaving behind perfusable channels within the tissue that allow for the flow of nutrients and oxygen (Fig. 3b). Over the span of a week, these tissues fused together in in vitro culture and beat in a synchronous manner. The printing approach was also demonstrated to generate branching print pathways, capable of keeping larger tissues alive, possibly presenting a path forward for generating human heart tissues.
Sacrificial pathways represent only one strategy for addressing the broader vascularization challenge at the organ scale. Other approaches have focused on designing scaffolds with predefined, perfusable channel networks that support continuous fluid flow and reduce reliance on passive diffusion for nutrient and oxygen delivery in thick tissues35. Microfluidic scaffold designs, in particular, have been shown to improve long-term tissue viability by maintaining more uniform transport conditions throughout engineered constructs. In addition to physical perfusion strategies, biochemical approaches have been explored to promote vascular development. Spatially controlled presentation of angiogenic growth factors has been used to guide endothelial cell migration and encourage the formation of vascular networks that can connect with host vasculature after implantation36. More recently, emerging 4D bioprinting concepts have proposed the use of materials that undergo programmed changes over time, allowing printed constructs to adapt their structure or properties as tissues mature37. Rather than relying on a single solution, these strategies suggest that successful vascularization of organ-scale cardiac constructs will likely depend on combining engineered perfusion pathways with biochemical signaling and time-dependent tissue adaptation.
Therefore, while current bioprinted heart models are limited in size and function, advancements in vascularization and tissue fusion techniques are bringing the field closer to overcoming these setbacks. By using new and refined methods to improve cell viability and integrate functional blood vessels, larger, more sustainable cardiac constructs can be developed in order to bridge the gap between engineered tissues and human organs.
Limitations and Future Directions
One of the major challenges in bioprinting a human heart involves generating the appropriate number of cells for such a large and complex organ. However, the use of bioreactors, which are specialized devices that help to optimize the setting for biological reactions, can greatly accelerate the culturing of large volumes of cells. By creating an environment that mimics bodily conditions, generally through regulating the temperature, pH, and oxygen levels, bioreactors allow for cells to more efficiently differentiate and proliferate38.
In addition, current biomaterials are insufficient, especially in scaffold printing, struggling to replicate the ECM and supporting cell growth. Biomaterials need to emulate the properties of the ECM to provide essential support and signaling for cells, while also maintaining biocompatibility to prevent immune rejection and biodegradability to safely break down as the tissue regenerates. While commonly used materials, such as collagen, meet the chemical requirements for biocompatibility and biodegradability, they lack the necessary architectural complexity and the ability to adapt to the changing needs of cells during implantation and regeneration39.
Future scaffolds need to replicate not only the proteins of the ECM, but also the specific structural features and interactions between components of the matrix. Bioprinting is also limited by the struggle to keep vast quantities of cells alive through both the printing process and implantation; this calls for advanced vascularization and printing techniques. To manage this, new bioprinting approaches like multi-nozzle printing are being explored. These new approaches may enable the printing of larger volumes of cells and the simultaneous use of multiple materials, creating more complex structures with varying cell types and ECM components, while also increasing speed and precision40. Greater speed reduces the amount of time in which cells are exposed to potentially damaging conditions, and enhanced precision helps ensure the accurate placement of materials to construct intricate microvascular networks necessary for cardiac tissue survival.
While advances in bioreactors, scaffold design, and overall printing technologies are addressing the biological and engineering challenges of cardiac bioprinting, technical feasibility alone does not determine whether these therapies can be translated into clinical practice. Progress is also shaped by broader systemic factors, including access to research infrastructure, regulatory hurdles, and ethical considerations related to safety and equity.
Global differences in biomedical research capacity create unequal conditions for the development of stem cell-based cardiac therapies. High-income countries generally possess the funding and facilities required for advanced regenerative medicine research, such as specialized bioprinting equipment and large-scale bioreactors. In contrast, many low- and middle-income countries lack the infrastructure and resources needed to support complex cell-based manufacturing or early-phase clinical trials41. Seeing as cardiovascular disease places a particularly heavy burden on these regions, limited access to regenerative medicine research raises concerns that early clinical applications of cardiac bioprinting may benefit only a narrow subset of patients in well-resourced healthcare systems42.
Regulatory and ethical oversight also presents a major barrier to the clinical translation of stem cell-based cardiac therapies. In the United States, these therapies are regulated by the Food and Drug Administration (FDA) as biological products. When stem cell-derived cardiomyocytes are combined with biomaterial scaffolds or implantable constructs, they are reviewed as combination products, meaning that both the cellular component and the material scaffold must meet regulatory standards for safety and quality43. Before human trials can begin, extensive preclinical testing is required to establish safety and reproducibility. This includes verifying that the cells are free of microbial contamination, maintain stable genetic characteristics, and exhibit predictable behavior over time. For cardiac applications specifically, regulators focus on risks such as abnormal electrical activity that could induce arrhythmias, uncontrolled cell proliferation, and long-term stability of the implanted tissue44. Only after these criteria are met can a therapy enter phased clinical trials.
All stages of clinical development must be conducted under Good Manufacturing Practice (GMP) conditions. GMP refers to a set of FDA-enforced standards designed to ensure that biological products are produced in a controlled and reproducible manner. In practice, this requires the use of validated manufacturing protocols, strict environmental controls, detailed documentation of every step in production, and rigorous quality control testing of each batch43. For stem cell-based therapies, GMP compliance also involves monitoring cell identity, purity, viability, and potency to ensure consistency between batches. These requirements are particularly challenging for personalized therapies, where each patient-specific product must meet the same regulatory standards despite being individually manufactured.
In addition to the United States, other regulatory agencies have established frameworks to evaluate stem cell-based therapies, but these frameworks differ in structure and emphasis. In the European Union, for example, stem cell-derived products are regulated as Advanced Therapy Medicinal Products (ATMPs) by the European Medicines Agency (EMA). Therapies classified as ATMPs—which include gene therapies, cell therapies, and tissue-engineered products—undergo centralized authorization, meaning a single approval by the EMA is valid across all EU member states. Like the FDA, the EMA requires extensive quality, safety, and efficacy data before granting market authorization. This includes evidence of manufacturing consistency, absence of contaminants, and functional performance in relevant preclinical models44. . The EMA also requires long-term follow-up plans to monitor patients after treatment, particularly for products that involve living cells capable of engraftment or long-term integration into tissues. These requirements are designed to detect late-emerging risks, such as immune reactions or unintended tissue changes.
Regulatory pathways outside of the United States and European Union vary considerably, largely due to differences in regulatory capacity and experience with advanced biological therapies. In many low- and middle-income countries, national regulatory agencies do not yet have specialized frameworks tailored to stem cell–based or tissue-engineered products. As a result, these therapies are often evaluated using regulatory standards originally designed for conventional drugs or biologics, which are not well suited to address the unique challenges posed by living cells or combination products. This mismatch can lead to uncertainty in the approval process, inconsistent oversight, and delays in authorizing clinical trials, further limiting opportunities for global clinical translation.
Ethical considerations are closely linked to these regulatory challenges. Stem cell-based cardiac therapies involve living cells and, in some cases, long-lasting or irreversible interventions, which places a strong emphasis on patient protection. Regulatory authorities therefore require rigorous informed consent procedures, careful risk assessment, and long-term follow-up to monitor possible adverse outcomes, such as abnormal tissue growth, immune responses, or delayed functional complications45. Ethical oversight also extends to issues of fairness and access. When advanced therapies are developed and approved primarily within regulatory systems that have substantial technical expertise, funding, and manufacturing oversight, patients in regions with limited infrastructure may be excluded from potential benefits, reinforcing existing inequalities in access to regenerative medicine41,42. These ethical considerations highlight that regulatory evaluation is not solely a technical process, but also a mechanism for balancing innovation with patient safety, autonomy, and equity.
Overall, the limitations of cardiac bioprinting arise from a combination of biological, engineering, and systemic challenges rather than a single unresolved problem. Although advances in bioreactor design, scaffold materials, and printing strategies continue to improve the feasibility of engineered cardiac tissues, progress toward clinical use will require improvements in manufacturing scalability, regulatory clarity, and ethical oversight. Future work in this field will therefore need to focus not only on refining bioprinting technologies, but also on developing standardized production processes and regulatory pathways that support safe, consistent, and broadly accessible clinical translation.
Conclusion
Together, stem cell-derived cardiomyocytes, heart-on-chip technology, and 3D bioprinting of the heart and its subsections represent a notable progression toward the future of regenerative medicine and cardiovascular disease treatment. Stem cell technologies supply patient-specific cardiomyocytes, heart-on-chip systems provide a physiologically relevant environment to study disease mechanisms and evaluate functional performance, and bioprinting translates these validated components into structured tissues capable of repairing or replacing damaged cardiac regions. Collectively, these approaches could restore cardiac function in ways that current pharmacological and surgical treatments cannot.
At the same time, bringing these technologies closer to clinical use reveals several important challenges. Many engineered cardiac tissues still struggle to achieve full maturation, long-term viability, and reliable vascularization, and immune responses remain a concern even when patient-derived cells are used. Beyond the biological and engineering barriers, translation is also shaped by regulatory requirements, manufacturing constraints, and ethical considerations related to safety, cost, and equitable access. These factors make it clear that progress will depend not only on improving individual technologies, but also on developing standardized production methods and regulatory pathways that can support safe and reproducible therapies.
Even with these limitations, the combined use of stem cell biology, microphysiological systems, and bioprinting marks a meaningful shift in how cardiovascular disease can be studied and treated. Rather than focusing solely on symptom management, this integrated framework enables more accurate disease modeling, functional testing, and tissue-level repair using human-relevant systems. As these approaches continue to advance, they offer a path toward more long-lasting and personalized treatments for cardiovascular disease, with the potential to improve outcomes for patients who currently have limited therapeutic options.
References
- World Health Organization. Cardiovascular diseases (CVDs). https://www.who.int/health-topics/cardiovascular-diseases. [↩]
- World Heart Federation. Deaths from cardiovascular disease surged 60% globally over the last 30 years. https://world-heart-federation.org/news/deaths-from-cardiovascular-disease-surged-60-globally-over-the-last-30-years-report/ (2023). [↩]
- Ojha, N. & Dhamoon, A. Myocardial Infarction. Anal Chem (2013). [↩]
- Frankel, W. C. O. C. M. et al. 2020 ACC/AHA Guideline for the Management of Patients With Valvular Heart Disease. J Am Coll Cardiol (2021). [↩] [↩]
- Wilhelm, M. J. Long-term outcome following heart transplantation: Current perspective. J Thorac Dis (2015). [↩]
- Singh, V. K., Saini, A., Kalsan, M., Kumar, N. & Chandra, R. Describing the stem cell potency: The various methods of functional assessment and in silico diagnostics. Frontiers in Cell and Developmental Biology (2016). [↩]
- Stem Cell Basics. STEM Cell Information. [↩]
- Hsueh, Y. C., Pratt, R. E., Dzau, V. J. & Hodgkinson, C. P. Novel method of differentiating human induced pluripotent stem cells to mature cardiomyocytes via Sfrp2. Sci Rep (2023). [↩]
- Yang, Q., Xiao, Z., Lv, X., Zhang, T. & Liu, H. Fabrication and Biomedical Applications of Heart-on-a-chip. Int J Bioprint 7, 54–70 (2021). [↩]
- Deir, S., Mozhdehbakhsh Mofrad, Y., Mashayekhan, S., Shamloo, A. & Mansoori-Kermani, A. Step-by-step fabrication of heart-on-chip systems as models for cardiac disease modeling and drug screening. Talanta vol. 266 Preprint at https://doi.org/10.1016/j.talanta.2023.124901 (2024). [↩]
- Abulaiti, M. et al. Establishment of a heart-on-a-chip microdevice based on human iPS cells for the evaluation of human heart tissue function. Sci Rep 10, (2020). [↩]
- Gittenberger-De Groot, A. C., Bartelings, M. M., Deruiter, M. C. & Poelmann, R. E. Basics of cardiac development for the understanding of congenital heart malformations. Pediatric Research 57 (2005). [↩]
- Hsueh, Y. C., Pratt, R. E., Dzau, V. J. & Hodgkinson, C. P. Novel method of differentiating human induced pluripotent stem cells to mature cardiomyocytes via Sfrp2. Sci Rep (2023). [↩] [↩] [↩] [↩]
- Abulaiti, M. et al. Establishment of a heart-on-a-chip microdevice based on human iPS cells for the evaluation of human heart tissue function. Sci Rep 10 (2020). [↩]
- Yang, Q., Xiao, Z., Lv, X., Zhang, T. & Liu, H. Fabrication and Biomedical Applications of Heart-on-a-chip. International Journal of Bioprinting 7 (2021). [↩]
- Ren, L. et al. Investigation of hypoxia-induced myocardial injury dynamics in a tissue interface mimicking microfluidic device. Analytical Chemistry 85 (2013). [↩]
- Mourad, O., Yee, R., Li, M. & Nunes, S. S. Modeling Heart Diseases on a Chip: Advantages and Future Opportunities. Circulation Research 132 (2023). [↩] [↩]
- Deir, S., Mozhdehbakhsh Mofrad, Y., Mashayekhan, S., Shamloo, A. & Mansoori-Kermani, A. Step-by-step fabrication of heart-on-chip systems as models for cardiac disease modeling and drug screening. Talanta 266 (2024). [↩] [↩]
- Korda, M. Macrophages enable heart-on-a-chip longevity. Nat Cardiovasc Res 3, 776. https://doi.org/10.1038/s44161-024-00519-8 (2024). [↩]
- Derakhshanfar, S. et al. 3D bioprinting for biomedical devices and tissue engineering: A review of recent trends and advances. Bioact Mater 8, 144–156. https://pmc.ncbi.nlm.nih.gov/articles/PMC5935777/ (2023). [↩]
- Kačarević, Ž. P. et al. An introduction to 3D bioprinting: Possibilities, challenges and future aspects. Materials 11 (2018). [↩]
- Lee, A. et al. 3D Bioprinting of Collagen to Rebuild Components of the Human Heart. Science (2019). [↩]
- Lee, A. et al. 3D Bioprinting of Collagen to Rebuild Components of the Human Heart. Science (n.d.). [↩]
- Esmaeili, H. et al. Electroconductive biomaterials for cardiac tissue engineering. Acta Biomater 139, 118–140 (2021). [↩]
- Pati, F., Jang, J., Ha, DH. et al. Printing three-dimensional tissue analogues with decellularized extracellular matrix bioink. Nat Commun 5, 3935. (2014). [↩]
- Mohan, T. S. et al. 3D coaxial bioprinting: Process mechanisms, bioinks and applications. Prog Biomed Eng (Bristol) 4(2):022003. (2022). [↩]
- Zhang, Y. et al. Recent Advances in Cardiac Patches: Materials, Preparations, and Properties. ACS Biomaterials Science and Engineering vol. 8 3659–3675. (2022). [↩]
- dela Paz, N. G. & D’Amore, P. Arterial versus venous endothelial cells. Cell Tissue Res 335(1):5–16. (2008). [↩] [↩]
- You, S. et al. High cell density and high-resolution 3D bioprinting for fabricating vascularized tissues. Sci Adv 9(8):eade7923. (2023). [↩]
- Papaioannou, T. G., Manolesou, D., Dimakakos, E., Tsoucalas, G., Vavuranakis, M. & Tousoulis, D. 3D bioprinting methods and techniques: Applications on artificial blood vessel fabrication. Acta Cardiol Sin 35, 284–289. (2019). [↩]
- Engler, Adam J et al. “Embryonic cardiomyocytes beat best on a matrix with heart-like elasticity: scar-like rigidity inhibits beating.” Journal of cell science vol. 121,Pt 22: 3794-802. (2008). [↩]
- Scheiner, Z. S., Talib, S. & Feigal, E. G. The potential for immunogenicity of autologous induced pluripotent stem cell-derived therapies. J Biol Chem 289(8):4571–4577. (2013). [↩] [↩]
- Noor, N. et al. 3D Printing of Personalized Thick and Perfusable Cardiac Patches and Hearts. Advanced Science 6, (2019). [↩] [↩]
- Skylar-Scott, M. A. et al. Biomanufacturing of organ-specific tissues with high cellular density and embedded vascular channels. Sci Adv (2019). [↩]
- Miller, J., Stevens, K., Yang, M. et al. Rapid casting of patterned vascular networks for perfusable engineered three-dimensional tissues. Nature Mater 11, 768–774. (2012). [↩]
- Rouwkema, J. & Khademhosseini, A. Vascularization and angiogenesis in tissue engineering: Beyond creating static networks. Trends Biotechnol 34(9):733–745. (2016). [↩]
- Arif, Z. U., Khalid, M. Y., Zolfagharian, A. & Bodaghi, M. 4D bioprinting of smart polymers for biomedical applications: Recent progress, challenges, and future perspectives. React Funct Polym. (2022). [↩]
- Grayson, W. & Stephenson, M. Recent advances in bioreactors for cell-based therapies. F1000Research 7 (2018). [↩]
- Williams, D. F. Challenges with the development of biomaterials for sustainable tissue engineering. Frontiers in Bioengineering and Biotechnology 7 (2019). [↩]
- Zhou, H. et al. Simultaneous multimaterial multimethod bioprinting. Bio-Design and Manufacturing 5 (2022). [↩]
- World Health Organization. Global Observatory on Health R&D: Bridging the gap in global health research and development. https://www.who.int/news/item/21-11-2023-global-observatory-on-health-r-d–bridging-the-gap-in-global-health-research-and-development (2023). [↩] [↩]
- Yap, K. K. Inequality issues in stem cell medicine. Stem Cells Transl Med 5(2):265–266 (2016). [↩] [↩]
- U.S. Food and Drug Administration. Guidance for Industry: Considerations for the Design of Early-Phase Clinical Trials of Cellular and Gene Therapy Products. https://www.fda.gov/media/87564/download (2013). [↩] [↩]
- Lysaght, T., Kerridge, I. H., Sipp, D., Porter, G. & Capps, B. J. Ethical and regulatory challenges with autologous adult stem cells: A comparative review of international regulations. J Bioeth Inq 14(2):261–273 (2017). [↩] [↩]
- Madrid, M. et al. Considerations for the development of iPSC-derived cell therapies: A review of key challenges by the JSRM-ISCT iPSC Committee. Cytotherapy 26(11):1382–1399. (2024). [↩]