Abstract
Semaglutide, a GLP-1 receptor agonist widely prescribed for obesity, has recently been linked to central nervous system (CNS) side effects, including mood alterations and sleep disturbances, as reported in clinical and post-mortem studies. These findings suggest the possibility of neuronal inhibition mediated via the gut–brain axis, underscoring the need to investigate the underlying mechanisms and explore non-invasive therapeutic alternatives. We hypothesized that semaglutide may indirectly affect brain function through intestinal signaling and that such effects could be reversed using a natural product-based strategy that is both non-invasive and low in side effects. To test this hypothesis, we established a transwell co-culture model using human intestinal epithelial cells (Caco-2) and neuronal cells (SH-SY5Y). Semaglutide was applied to the gut cells, and downstream effects on neuronal gene expression were analyzed using RNA sequencing, qPCR, and Western blotting. The results revealed a significant reduction in the expression of GABA-related genes (GAD1, GABRA1) and circadian rhythm genes (BMAL1, CLOCK). Subsequently, L-theanine, a GABA pathway activator, and hesperidin, an antioxidant with circadian regulatory properties, were administered individually and in combination. Both treatments showed restorative effects at the gene and protein levels, with the combined treatment group exhibiting the most pronounced recovery. This study provides evidence, using a human intestinal–neuronal co-culture model, that semaglutide exposure to intestinal epithelial cells is associated with downstream neuronal transcriptional changes. Unlike prior rodent or microbiota-focused gut–brain axis studies, our model enables direct examination of human epithelial–neuronal signaling. These findings may provide a valuable foundation for improving drug safety and developing alternative treatments to alleviate CNS-related side effects of semaglutide.
Introduction
Semaglutide, a widely prescribed obesity treatment, contains the active ingredient, glucagon-like peptide-1 (GLP-1) receptor agonist1‘2‘3. Semaglutide mimics the action of GLP-1 in the body, stimulating insulin secretion, suppressing appetite, and promoting sustained weight loss. GLP-1 receptor agonists are currently used not only for glycemic control in type 2 diabetes but also for obesity management and reducing cardiovascular disease-related mortality4‘5.
However, recent clinical and post-marketing reports have raised concerns about a wide range of adverse effects. The most commonly reported adverse effects include gastrointestinal symptoms such as nausea, vomiting, constipation, and diarrhea6‘7. In fact, several studies have shown that semaglutide carries a higher relative risk of serious gastrointestinal complications compared to other obesity treatments. The risk of intestinal obstruction was found to be 4.22-fold higher, gastroparesis 3.67-fold higher, and pancreatitis 9.09-fold higher8‘9‘10
In addition to gastrointestinal symptoms, several observational and post-marketing reports have described mood changes, sleep disturbances, or cognitive symptoms in some GLP-1RA users11. However, large randomized controlled trials and pooled safety analyses generally report similar rates of anxiety, depression, and sleep-related events between semaglutide and placebo groups, and no confirmed causal relationship has been established12‘13. Therefore, while CNS-related symptoms have been noted in certain patient populations, their mechanistic basis remains uncertain and warrants careful investigation. This is particularly noteworthy given that GLP-1 receptors are expressed not only in the gastrointestinal tract but also in several CNS regions, including the hypothalamus14, brainstem15, and hippocampus, which are involved in appetite regulation, emotional regulation, and circadian rhythm maintenance16‘17‘18.
Although several observational studies have reported mood changes, sleep disturbances, or cognitive symptoms in GLP-1RA users, large randomized controlled trials show inconsistent findings and do not establish a confirmed causal link. Therefore, potential CNS-related effects remain mechanistically unexplained and require molecular-level investigation. In this study, we hypothesized that semaglutide may indirectly modulate neural gene expression via gut-derived signals, thereby disrupting multiple pathways underlying the observed CNS-related side effects. Prior studies investigating GLP-1RA actions on the gut–brain axis have primarily used rodent models19 or microbiota-focused approaches20. While these studies suggest that peripheral GLP-1 signaling can influence brain function, they do not isolate the contribution of human intestinal epithelial signaling independent of neural, immune, or microbial confounders.
To address this gap, we established a gut-brain axis co-culture model using human intestinal epithelial cells (Caco-2) and human neuroblastoma cells (SH-SY5Y) in a transwell system. Semaglutide (50 nM) was administered to Caco-2 cells, and its subsequent effects on SH-SY5Y cells were evaluated using RNA sequencing, RT-qPCR, Western blot, and ELISA.
To date, no human cell-based study has examined whether semaglutide-exposed intestinal epithelial cells can modulate neuronal gene expression through epithelial-derived signaling. Therefore, a reductionist human gut–neuron co-culture system offers a unique opportunity to dissect epithelial-to-neuronal communication in isolation.
Materials and Methods
Cell Culture and Transwell Co-culture Setup
Human intestinal epithelial cells (Caco-2) and human neuroblastoma cells (SH-SY5Y) were obtained from ATCC and cultured in DMEM (Gibco) supplemented with 10% fetal bovine serum (FBS), 1% nonessential amino acids, and 1% penicillin-streptomycin at 37°C in a humidified incubator containing 5% CO₂. For co-culture experiments, Caco-2 cells were seeded onto 0.4 µm pore-size polyester membrane Transwell inserts (Corning) and differentiated for 10 days to form a confluent monolayer. SH-SY5Y cells were plated in the lower well one day before treatment. Semaglutide (50 nM) was applied to Caco-2 cells for 48 hours. Where indicated, natural compounds (L-theanine and/or hesperidin) were co-administered.
Extraction and Purification of Hesperidin from Citrus Peel
To obtain naturally occurring esperidin, fresh citrus peel was collected, thoroughly washed with distilled water, and dried at 50°C for 48 hours. The dried peel was ground into a fine powder using a mechanical grinder. For extraction, 10 g of citrus peel powder was mixed with 100 mL of 70% ethanol and heated in a 60°C water bath with continuous stirring for 2 hours. The resulting mixture was filtered through Whatman No. 1 filter paper, and the filtrate was concentrated using a rotary evaporator at 40°C under reduced pressure.
The concentrated extract was then subjected to liquid-liquid partitioning using ethyl acetate and water to isolate the flavonoid-rich fraction. The ethyl acetate layer was collected and further dried to obtain a crude flavonoid extract. For purification, the extract was dissolved in methanol and loaded onto a silica gel column chromatography system. Hesperidin was eluted using a methanol:chloroform (3:7) gradient, and the fractions were analyzed by thin layer chromatography (TLC) against a hesperidin standard (Sigma-Aldrich). The purified fractions were pooled, concentrated, and quantified using UV-Vis spectroscopy at 285 nm. The final hesperidin extract was stored at -20°C and dissolved in DMSO before use.
Preparation of L-theanine and Semaglutide
L-theanine (Sigma-Aldrich, 3081-61-6) was dissolved in sterile water to prepare a 100 mM stock solution. Semaglutide (Sigma-Aldrich, 910463-68-2) was prepared as a 1 mM stock solution in DMSO and stored at -20°C. Working concentrations (L-theanine 100 µM, semaglutide 50 nM) were freshly prepared in cell culture medium before treatment.
RNA Extraction and RT-qPCR
After 48 hours of treatment, SH-SY5Y cells were harvested and total RNA was extracted using TRIzol reagent (Invitrogen). RNA concentration and purity were measured using a NanoDrop spectrophotometer. Reverse transcription was performed using a High-Capacity cDNA Reverse Transcription Kit (Applied Biosystems). Quantitative PCR was performed using SYBR Green Master Mix (Thermo Fisher) and primers specific for GAD1, GABRA1, BMAL1, and CLOCK genes (Table 1). Relative gene expression was normalized to GAPDH and calculated using the 2−ΔΔCt method. All reactions were performed in triplicate.
| Gene | Forward (5’ -> 3’) | Reverse (5’ -> 3’) |
| GAD1 | TGCTAGAGATCTGCTTCCGG | GTGGTCAGAGAGCTCCAAGT |
| GABRA1 | GACCTATGACAGTCCTCCGG | AGTCCTCCAAATGCATCGGA |
| BMAL1 | CCCTGGGCCATCTCGATTAT | TCATCCAGCCCCATCTTTGT |
| CLOCK | AAGTTCACTGCTGGCAATGG | CTGAGGCCTTCCCCAACTTA |
RNA Sequencing and Differential Expression Analysis
Total RNA from SH-SY5Y cells (control vs. semaglutide-exposed co-culture conditions) was extracted using TRIzol reagent and quantified with a NanoDrop spectrophotometer. RNA integrity was assessed using an Agilent 2100 Bioanalyzer, and samples with RIN ≥ 8.0 were used for library preparation. mRNA libraries were prepared using the Illumina TruSeq Stranded mRNA Library Prep Kit according to the manufacturer’s protocol.
Sequencing was performed on an Illumina NovaSeq 6000 platform to generate 150 bp paired-end reads. Raw sequencing data were processed using FastQC for quality assessment and TrimGalore for adaptor removal. Reads were aligned to the human reference genome (GRCh38) using STAR aligner (version 2.7). Gene-level counts were generated with featureCounts.
Differential expression analysis was performed using DESeq2 (version 1.40.0) in R (version 4.3.1). Genes with an adjusted p-value < 0.05 and |log2 fold change| > 1 were classified as significantly differentially expressed. Gene Ontology (GO) and KEGG pathway enrichment analyses were conducted using clusterProfiler (version 4.8). Volcano plots and heatmaps were generated using ggplot2 and pheatmap packages in R.
Protein Quantification Using ELISA
To measure GAD1 protein levels, cell lysates were prepared using RIPA buffer supplemented with protease inhibitors. Total protein concentration was measured using the BCA assay (Thermo Fisher). GAD1 protein levels were quantified using a human GAD1-specific ELISA kit (Abnova, KA2880) according to the manufacturer’s instructions. Absorbance was measured at 450 nm using a microplate reader, and concentrations were determined using a standard curve.
Statistical Analysis
All statistical analyses were performed using GraphPad Prism version 10.0.3 (GraphPad Software, San Diego, CA). For all experiments, n refers to independent biological replicates (n = 3), and each biological sample was measured in technical triplicate. Data distribution was evaluated using the Shapiro–Wilk normality test prior to statistical comparisons.
For experiments involving more than two groups, one-way analysis of variance (ANOVA) was performed followed by Tukey’s post hoc test to assess pairwise differences. When only two groups were compared, a two-tailed unpaired Student’s t-test was used. All data are presented as mean ± standard deviation.
Results
Establishing an in vitro model of the gut–brain axis via transwell co-culture
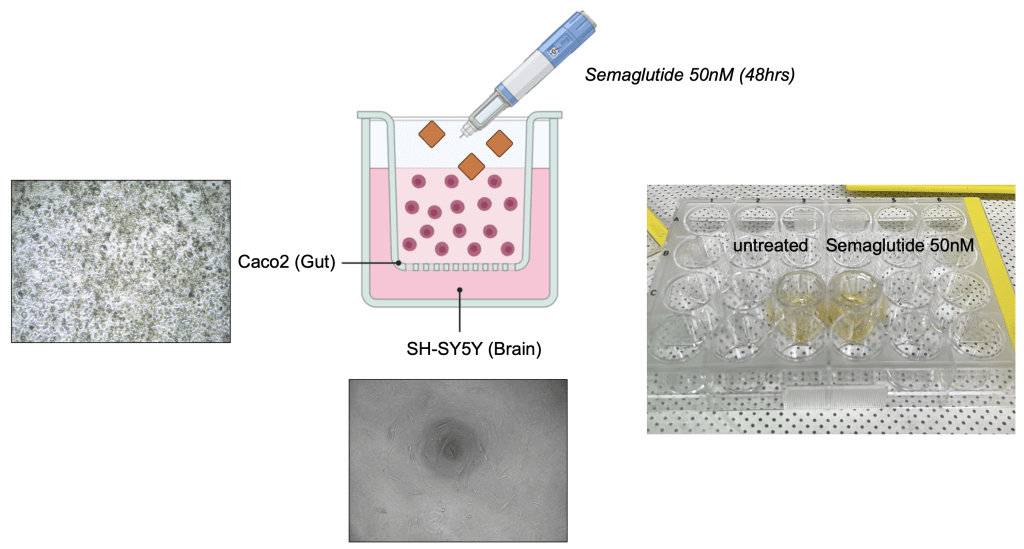
To investigate whether semaglutide exerts indirect neuromodulatory effects through gut-derived signaling, we first established a transwell-based co-culture system to simulate the gut–brain axis (Figure 1). This setup enabled paracrine communication between intestinal epithelial cells and neurons without direct contact.
Caco-2 cells (intestinal epithelial) were seeded on the apical insert of a transwell plate, while SH-SY5Y neuroblastoma cells were cultured in the basolateral compartment. After 10 days of differentiation for barrier formation, semaglutide (50 nM) was administered to the Caco-2 cells for 48 hours. The SH-SY5Y cells were then harvested to assess downstream molecular changes.
Transcriptome analysis reveals semaglutide-induced suppression of neuronal inhibitory and circadian signaling
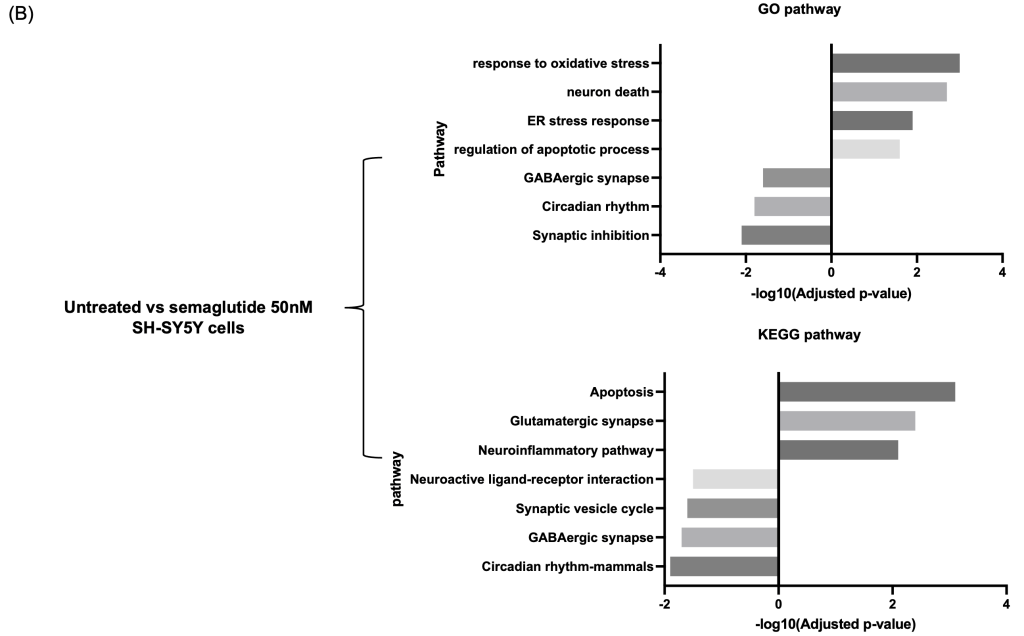
(c) Relative mRNA expression levels of GAD1, GABRA1, BMAL1, and CLOCK were significantly reduced in semaglutide-treated groups compared to untreated controls, as measured by RT-qPCR. Data are presented as mean ± SEM (n=3); (**p < 0.01, ***p < 0.001).
To further investigate the downstream molecular effects of semaglutide-mediated gut signaling on neuronal cells, we performed transcriptome analysis of SH-SY5Y cells following 48 hours of co-culture with either untreated or semaglutide-treated Caco-2 cells. This approach allowed us to identify differentially expressed genes (DEGs) and affected signaling pathways resulting from gut-to-neuron communication.
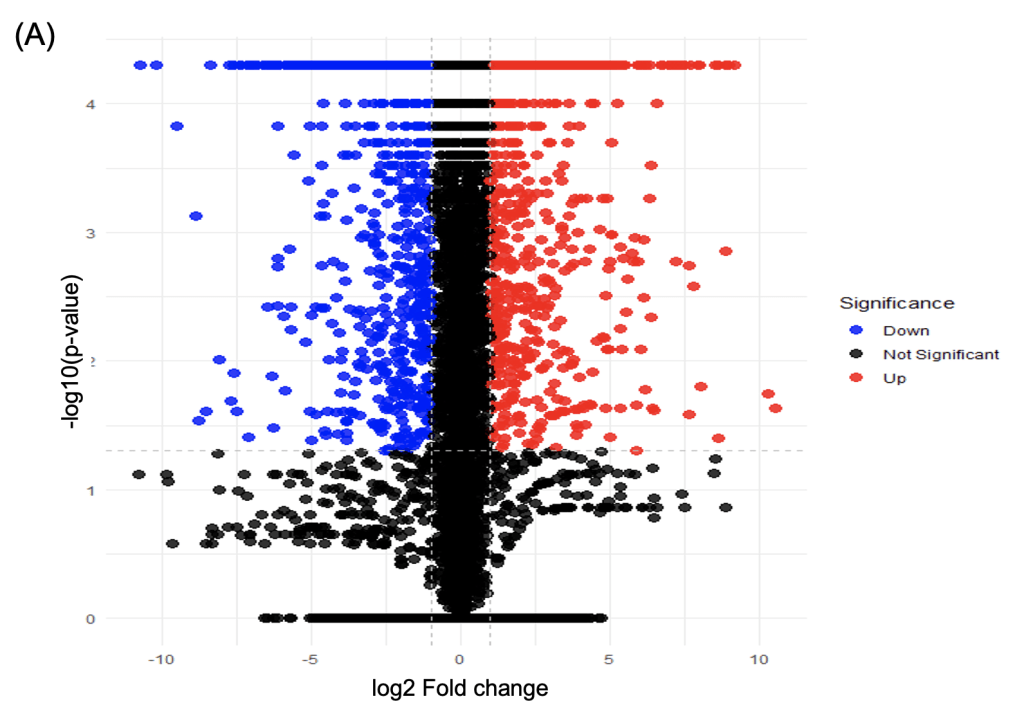
Total RNA was extracted from SH-SY5Y cells and subjected to RNA sequencing. DEG analysis revealed significant transcriptional changes between control and semaglutide-treated conditions. Specifically, 268 genes were upregulated and 315 genes were downregulated (adjusted p < 0.05, fold change > 1.5) (Figure 2A).
Gene Ontology (GO) enrichment analysis showed that upregulated pathways were primarily related to cellular stress and death, including response to oxidative stress, endoplasmic reticulum (ER) stress response, regulation of apoptotic processes, and neuronal cell death. In contrast, downregulated pathways included GABAergic synaptic transmission, synaptic inhibition, and circadian rhythm (Figure 2B).
Consistent with GO findings, KEGG pathway analysis revealed increased enrichment in pro-apoptotic and excitatory signaling pathways, such as apoptosis, glutamatergic synapse, and neuroinflammation. In contrast, the GABAergic synapse and circadian rhythm – mammals pathways showed decreased enrichment scores (Figure 2B).
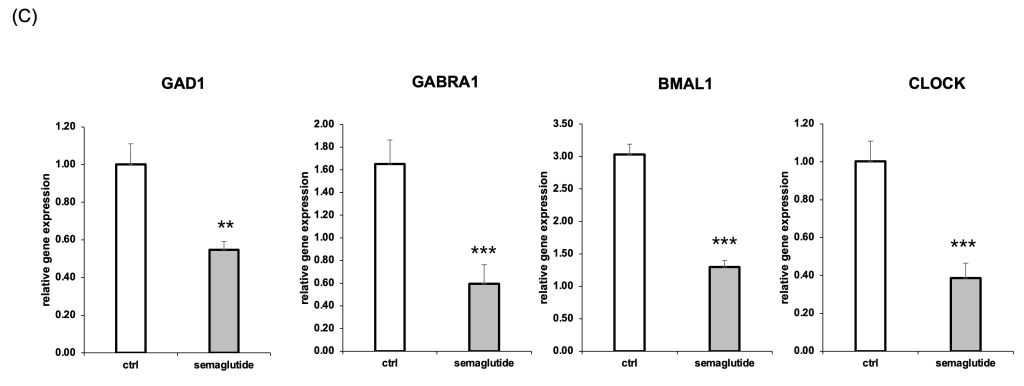
To validate key genes identified in the RNA-seq dataset, we performed RT-qPCR analysis on four representative targets. Relative to control, GAD1 and GABRA1 expression levels decreased by 45% (**p < 0.01) and 35.7% (***p < 0.001), respectively. Circadian genes BMAL1 and CLOCK were also significantly reduced by 42.9% (***p < 0.001) and 46.0% (***p < 0.001), respectively (Figure 2C).
These data suggest that semaglutide exposure—via gut epithelial signaling—downregulates key genes involved in inhibitory neurotransmission and circadian regulation in neurons, providing a mechanistic explanation for the CNS-related symptoms observed in clinical contexts.
Natural Compound Treatment Restores Semaglutide-Induced Neuronal Depression

To assess whether the downregulation of GABAergic and circadian genes observed after semaglutide exposure could be reversed, we investigated two natural compounds, L-theanine and hesperidin, based on their known bioactivities. L-theanine, a green tea-derived amino acid that enhances GABA receptor signaling21‘22, whereas hesperidin is a citrus flavonoid with reported antioxidant and circadian regulatory effects23‘24. In this study, we hypothesized that administering these compounds alone or in combination would alleviate the semaglutide-induced neuronal depression observed in a gut-brain co-culture model.
To test this hypothesis, five experimental groups were established in a Caco-2/SH-SY5Y transwell system. Group A: Untreated control; Group B: Semaglutide alone (50 nM); Group C: Semaglutide + L-theanine (100 µM); Group D: Semaglutide + hesperidin (100 µM); Group E: Semaglutide + L-theanine + hesperidin (100 µM each) (Figure 3).
After 48 hours of treatment, SH-SY5Y cells were harvested and the following experiments were performed.
Natural compound co-treatment restores GABAergic and circadian gene expression suppressed by semaglutide
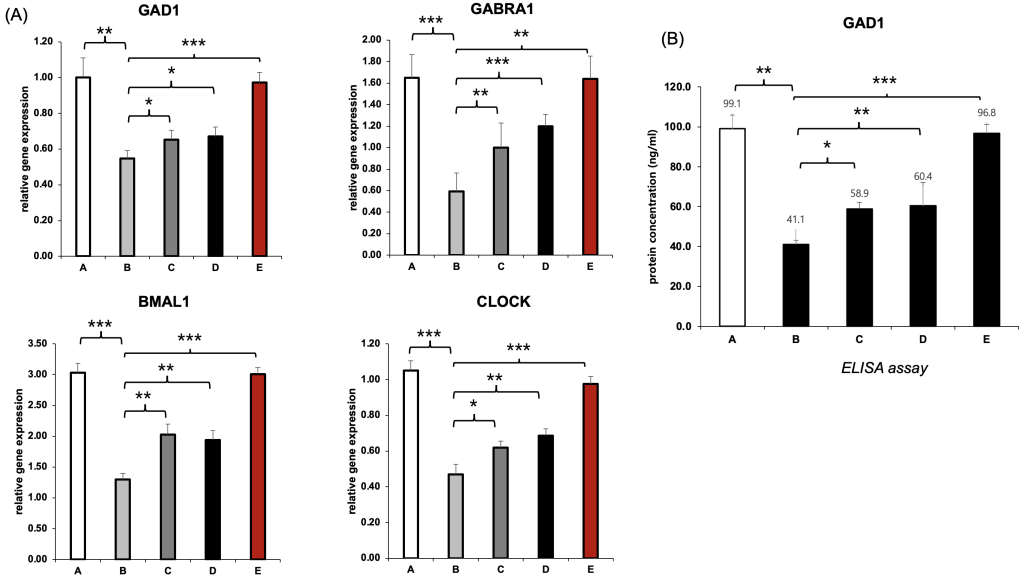
To determine whether the suppression of GABAergic and circadian genes induced by semaglutide could be reversed, we performed RT-qPCR analysis of GAD1, GABRA1, BMAL1, and CLOCK in SH-SY5Y neuronal cells following 48 hours of treatment (Figure B). We compared five groups: untreated control (A), semaglutide-only (B), semaglutide with L-theanine (C), semaglutide with hesperidin (D), and a combination treatment with both natural compounds (E).
In the semaglutide-only group (B), all four genes were significantly downregulated relative to the control. For example, GAD1 expression decreased from 1.00 to 0.43, GABRA1 from 1.65 to 1.01, BMAL1 from 3.03 to 1.74, and CLOCK from 1.00 to 0.52. These reductions suggest that semaglutide impairs both inhibitory neurotransmission and circadian regulation at the transcriptional level. Treatment with either L-theanine (C) or hesperidin (D) partially restored expression of specific genes: L-theanine showed stronger effects on GAD1 and GABRA1, while hesperidin more effectively restored BMAL1 and CLOCK. Notably, the combination group (E) demonstrated near-complete recovery of all four genes, with values closely approximating those of the control group, indicating a possible synergistic interaction between the two compounds (Figure 4A).
To confirm these effects at the protein level, we measured GAD1 protein concentration using ELISA (Figure 4B). Semaglutide alone reduced GAD1 from 99.1 ng/mL in the control group to 41.1 ng/mL. Partial recovery was observed in the L-theanine and hesperidin groups, which reached 58.9 ng/mL and 60.4 ng/mL, respectively. The combination treatment group (E), however, showed almost complete recovery at 96.8 ng/mL, aligning closely with the untreated control.
These results collectively demonstrate that semaglutide impairs both the transcription and translation of GABAergic and circadian regulatory components in neuronal cells, and that co-treatment with L-theanine and hesperidin can effectively reverse these effects in a gut–brain axis in vitro model.
Comparison of Direct Versus Epithelial-Mediated Effects of Semaglutide on Neuronal Gene Expression

To determine whether semaglutide-induced neuronal changes resulted from direct exposure or required epithelial-mediated signaling, we compared gene expression profiles between an “empty-insert” condition and the Caco-2 co-culture system. Direct exposure of SH-SY5Y cells to semaglutide through an empty insert led to a modest reduction in inhibitory neurotransmission and circadian-related genes, with GAD1, GABRA1, BMAL1, and CLOCK (Table 2).
In contrast, semaglutide treatment in the presence of a differentiated Caco-2 monolayer produced a more pronounced and consistent downregulation of the same genes (Table 2).
These results indicate that while direct neuronal exposure to semaglutide can partially suppress transcription of these pathways, the presence of intestinal epithelial cells amplifies this effect, supporting a contributory role of epithelial-derived signaling in shaping neuronal responses.
Discussion
We asked whether semaglutide exposure of human intestinal epithelial cells in a gut-neuron co-culture system could induce transcriptional changes in underlying neuronal cells. A transwell system of co-cultured Caco-2 and SH-SY5Y cells was established. Both GABAergic neurotransmission and core circadian genes (such as GAD1, GABRA1, BMAL1, and CLOCK) were downregulated following exposure to semaglutide-exposed Caco-2 cells, in the gut-neuron transwell co-culture model. These findings indicate that signals from epithelium may be involved in neural transcription patterning and may be implicated in the reported variability in mood or sleep-related adverse events with GLP-1 receptor agonists, observed in clinical and post-marketing settings, but a causal link has not been demonstrated in human clinical studies.
Our results corroborate and mechanistically explain previous studies of GLP-1RA actions through the microbiota-gut-brain axis, showing that both microbiota-dependent and -independent mechanisms exist. Previous studies also show that peripheral GLP-1 signaling can affect central nervous system signaling, but were not able to distinguish the effects of human intestinal epithelial signaling from those of the microbiota. We addressed this question in an innovative way using what we believe to be the first human epithelial-neuronal transwell co-culture model with semaglutide to study the role of the epithelium free from the potentially confounding influences of systemic, microbial, hormonal and neural circuitry inputs that preclude definitive interpretation in vivo.
Finally, to explore whether the semaglutide-induced transcriptional program could be modified, we tested natural compounds with known effects on GABA and/or circadian rhythms, the amino acid L-theanine and the flavonoid hesperidin. Each compound partially reversed the transcriptional pattern, with a greater effect being observed with the combination treatment. While these results show putative molecular interactions, we do not show functional rescue, therapeutic efficacy or direct mechanistic reversal but rather show the utility of this model for interrogating how epithelial-neuronal communication is modulated.
Though the results are exciting, some caution in interpretation should be exercised. First, the study was performed entirely in vitro and transcriptional changes identified may not be representative of in vivo alterations. The model of the gut-brain axis used in the study did not include other layers of regulation including microbial metabolites, enteroendocrine signaling, immune signaling and the neural tract. Secondly, only four genes representative of two major improved pathways were verified by qPCR. While an approach of this type is common for mechanistic studies using RNA-seq, confirming additional genes and pathways can provide more information. Third, we only used one dose (50 nM) and one time point (48 h), so we cannot assess whether there is a dose-response or time-response relationship. Additional analyzes, including dose response analyzes, other time points, and physiologically based concentration modeling, would help clarify these results. Finally, we did not assess downstream neuronal functional endpoints, such as electrophysiological activity or neurotransmitter release, that may help support the role of transcriptional changes in mediating the physiological effects we observed.
Future directions include the assessment of semaglutide in other in vivo models of behavior or physiology (e.g., circadian rhythm, affective behaviors or sleep) or models that create a microbiota-modulating condition, epithelial-specific manipulations or receptor inhibition or agonism. Such an approach may help us to identify the signaling mechanisms and cell types that semaglutide acts through within the gut-brain axis. Further investigations of other molecular endpoints for L-theanine and hesperidin may reveal whether they are acting transcriptionally, post-transcriptionally, or through antioxidant or metabolic modes of action.
In summary, we show that epithelial exposure to semaglutide alters neuronal gene expression, and that the effect can be modulated by naturally occurring compounds with complementary biological effects. Confirmation of this finding in more complex biological systems is warranted. Furthermore, our human gut-neuron co-culture model can be used to investigate the role of epithelial cells in gut-brain signaling in response to GLP-1 receptor agonist exposure.
References
- Alorfi, N. M. & Algarni, A. S. Clinical Impact of Semaglutide, a Glucagon-Like Peptide-1 Receptor Agonist, on Obesity Management: A Review. Clin Pharmacol 14, 61-67, 2022. [↩]
- Chao, A. M., Tronieri, J. S., Amaro, A. & Wadden, T. A. Semaglutide for the treatment of obesity. Trends Cardiovasc Med 33, 159-166, 2023. [↩]
- Garvey, W. T. et al. Two-year effects of semaglutide in adults with overweight or obesity: the STEP 5 trial. Nature Medicine 28, 2083-2091. 2022. [↩]
- Nachawi, N., Rao, P. P. & Makin, V. The role of GLP-1 receptor agonists in managing type 2 diabetes. Cleveland Clinic Journal of Medicine 89, 457-464, 2022. [↩]
- Turkistani, Y. Glucagon-like peptide-1 receptor agonists: a review from a cardiovascular perspective. Frontiers in Cardiovascular Medicine Volume 12 – 2025, 2025. [↩]
- Shu, Y. et al. Gastrointestinal adverse events associated with semaglutide: A pharmacovigilance study based on FDA adverse event reporting system. Front Public Health 10, 996179, 2022. [↩]
- Sodhi, M., Rezaeianzadeh, R., Kezouh, A. & Etminan, M. Risk of Gastrointestinal Adverse Events Associated With Glucagon-Like Peptide-1 Receptor Agonists for Weight Loss. JAMA 330, 1795-1797, 2023. [↩]
- Aneke-Nash, C., Hung, K. S., Wall-Wieler, E., Zheng, F. & Sharaiha, R. Z. Comparing the risk of gastroparesis following different modalities for treating obesity: semaglutide versus bupropion-naltrexone versus sleeve gastrectomy – a retrospective cohort study. BMJ Open Gastroenterol 12, 2025. [↩]
- Chaudhry, A., Gabriel, B., Noor, J., Jawad, S. & Challa, S. R. Tendency of Semaglutide to Induce Gastroparesis: A Case Report. Cureus 16, e52564, 2024. [↩]
- Hughes, K., Sumaruth, Y. R. K., Mohammed, E. & Sant Bakshsingh, V. Acute Pancreatitis Likely Due to Semaglutide. Cureus 16, e69844, 2024. [↩]
- Chen, W., Cai, P., Zou, W. & Fu, Z. Psychiatric adverse events associated with GLP-1 receptor agonists: a real-world pharmacovigilance study based on the FDA Adverse Event Reporting System database. Frontiers in Endocrinology Volume 15 – 2024, 2024. [↩]
- Wadden, T. A. et al. Psychiatric Safety of Semaglutide for Weight Management in People Without Known Major Psychopathology: Post Hoc Analysis of the STEP 1, 2, 3, and 5 Trials. JAMA Internal Medicine 184, 1290-1300, 2024. [↩]
- Ebrahimi, P. et al. Suicide and Self-Harm Events With GLP-1 Receptor Agonists in Adults With Diabetes or Obesity: A Systematic Review and Meta-Analysis. JAMA Psychiatry 82, 888-895, 2025. [↩]
- Sandoval, D. A., Bagnol, D., Woods, S. C., D’Alessio, D. A. & Seeley, R. J. Arcuate Glucagon-Like Peptide 1 Receptors Regulate Glucose Homeostasis but Not Food Intake. Diabetes 57, 2046-2054, 2008. [↩]
- Heppner, K. M. et al. Expression and Distribution of Glucagon-Like Peptide-1 Receptor mRNA, Protein and Binding in the Male Nonhuman Primate (Macaca mulatta) Brain. Endocrinology 156, 255-267, 2015. [↩]
- Melson, E., Ashraf, U., Papamargaritis, D. & Davies, M. J. What is the pipeline for future medications for obesity? International Journal of Obesity 49, 433-451, 2025. [↩]
- Wang, J.-Y. et al. GLP−1 receptor agonists for the treatment of obesity: Role as a promising approach. Frontiers in Endocrinology Volume 14 – 2023, 2023. [↩]
- Smits, M. M. & Van Raalte, D. H. Safety of Semaglutide. Frontiers in Endocrinology Volume 12 – 2021, 2021. [↩]
- Lachey, J. L. et al. The Role of Central Glucagon-Like Peptide-1 in Mediating the Effects of Visceral Illness: Differential Effects in Rats and Mice. Endocrinology 146, 458-462, 2005. [↩]
- Kanbay, M. et al. Gut Microbiota Modulation in GLP-1RA and SGLT-2i Therapy: Clinical Implications and Mechanistic Insights in Type 2 Diabetes. Clinical Kidney Journal, 2025. [↩]
- Dasdelen, M. F. et al. A Novel Theanine Complex, Mg-L-Theanine Improves Sleep Quality via Regulating Brain Electrochemical Activity. Frontiers in Nutrition Volume 9 – 2022, 2022. [↩]
- Kim, S., Jo, K., Hong, K. B., Han, S. H. & Suh, H. J. GABA and l-theanine mixture decreases sleep latency and improves NREM sleep. Pharm Biol 57, 65-73, 2019. [↩]
- Buzdağlı, Y., Eyipınar, C. D., Kacı, F. N. & Tekin, A. Effects of hesperidin on anti-inflammatory and antioxidant response in healthy people: a meta-analysis and meta-regression. Int J Environ Health Res 33, 1390-1405, 2023. [↩]
- Khan, A., Ikram, M., Hahm, J. R. & Kim, M. O. Antioxidant and Anti-Inflammatory Effects of Citrus Flavonoid Hesperetin: Special Focus on Neurological Disorders. Antioxidants (Basel) 9, 2020. [↩]