Abstract
Background/objective Clinical trials have been instrumental in improving human health. Despite the diversity of global populations, historical and current evidence show that minorities by ethnicity, race, sexual orientation, and socioeconomic status have been underrepresented in clinical trials. To address this issue, Diversity, Equity and Inclusion (DEI) is an emerging principle which prioritizes the recruitment of minority groups in clinical trials, however its utility is uncertain. To this aim, a literature review was performed evaluating DEI methods and their success in clinical trials.
Methods PubMed and Google Scholar were indexed from 2000 to 2024 to identify relevant studies. Specifically, this study used search terms “DEI in clinical trials”, “Diversity, Equity, and Inclusion in clinical trials”, “Efficacy of DEI in clinical trials”, etc. These terms resulted in a collection of 3,253 papers; this collection was further filtered to 47 papers, using abstract screening for relevance (filtering for primary sources/review papers, papers in English, studies done in the US, remote and in-person settings).
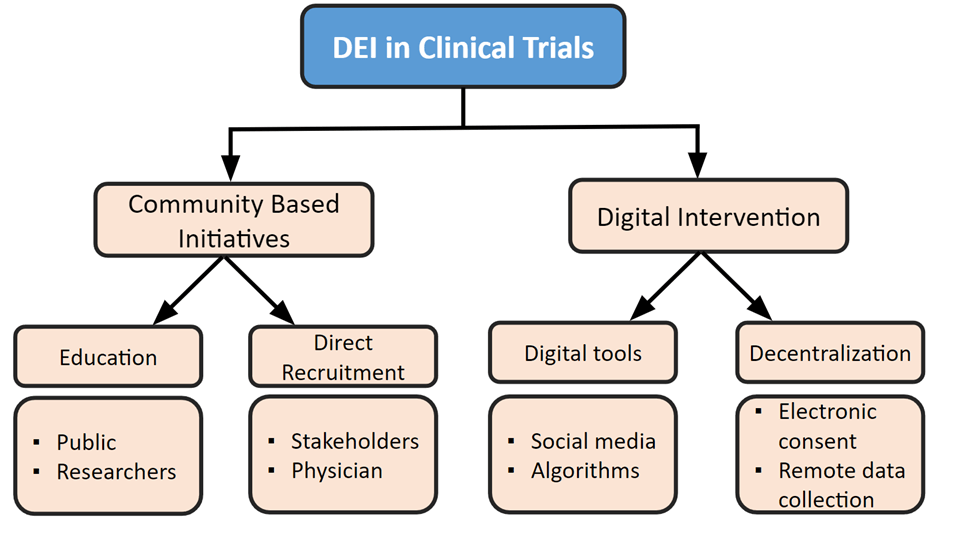
Results The review found that the two broad groups of methods to increase DEI were community-based initiatives, which specifically targeted minority populations through education and direct recruitment, and digital interventions, which used digital tools and decentralization to increase accessibility.
Conclusions Our study shows that when implemented, DEI practices can improve minority recruitment and the overall health of populations. Future trials should aim to adopt DEI principles in trial design stages to ensure representative studies.
Keywords: Diversity equity and inclusion, clinical trials, equity
Introduction
Clinical trials have been performed in healthcare since before the beginning of modern medicine and have been critical in advancing global healthcare1. The importance of clinical trials lies in their ability to evaluate if a new treatment or method of treatment is both safe and effective for the general population. Researchers and developers use clinical trials to obtain approval for new drug therapies for a variety of diseases. Due to cost restraints and limited resources, clinical trials rely on evaluating therapies in a smaller sample of the overall population with the assumption that the sample is representative of the overall population. However, with the increasing diversity of global populations, evidence shows that many minorities by ethnicity, race, gender, sexual orientation, and socioeconomic status have been underrepresented in clinical trials. This underrepresentation undermines the generalizability of trial results. For example, past clinical trials evaluating the efficacy of beta-blockers to manage cardiac arrhythmias, primarily included white patients. However, studies show that African American patients with heart failure do not have the same response to the medications as the white patients; thus, the studies that solely include white patients, do not ensure generalizability2. To overcome this, efforts have been made to intentionally diversify clinical trials using principles related to Diversity, Equity, and Inclusion (DEI).
DEI refers to the practices of intentionally diversifying various domains of medicine with the hope of creating equitable environments. Specifically, in the context of clinical trials, DEI refers to the intentional recruitment of minority groups in studies. When there are minorities which go unseen and underrepresented in clinical trials, health care disparities drastically increase and minorities are disproportionately represented3. Especially, with the increasing diversity of the American population, clinical trials must move towards having more representation to have reliable and equitable healthcare for all populations.
The National Institutes of Health (NIH) Revitalization Act was passed by the US Congress in 1993 with the purpose of increasing the enrollment and participation of minority groups in NIH sponsored clinical trials4. Despite these policies, evidence suggests that the enrollment of minorities in clinical trials has not drastically improved. Although, NIH funded studies had increases in minority participation by 2019 including an increase of 0.97% to 2.23% in African Americans, 0.18% to 1.24% in Hispanic/latinos, and 0.14% to 1.04% in Asian Americans, these increases are modest relative to demographic makeup of the population5. A 2018 Census Bureau report estimates that 60.7% of the population are non-Hispanic white Americans, but they represent above 90% of participants in clinical trials5.This supports the idea that there is not enough inclusion of minorities in clinical trials to be representing the diverse population of the US. A more recent Food & Drug Administration (FDA) initiative issued a draft guidance to enroll more underrepresented minorities, both racial and ethnic, into biomedical research3. The FDA sought to create better treatment options for minorities groups who are disproportionately harmed by various diseases. Of all randomized clinical trials (RCTs) organized in the United states, the total percentage of minority inclusion was only 3.95% with more than half being NIH funded studies5. The disproportionate representation exists in the status quo of America’s healthcare and continues to contribute to racially based healthcare inequalities.
Over the last two decades, various DEI efforts have been implemented including community-based initiatives and digital interventions. However, since its inception, there has been conflicting evidence regarding its utility and efficacy in recruiting minority patients into trials. This literature review aims to summarize and evaluate the value of the current DEI efforts. The current study will discuss the current state of DEI in clinical trials and methods of increasing DEI that are currently being used or show promise for success in the future.
Methods
A literature search was conducted using PubMed and Google Scholar to identify studies that evaluated DEI methods in clinical trials. The literature was then canvassed from the years 2000 to 2024. Specifically, combinations of search terms were used such as “Diversity, equity and inclusion in clinical trials”, “History of DEI”, “History of minority inclusion in clinical trials”, “Methods to improve DEI in clinical trials”, “Efficacy of DEI in clinical trials”, “Community-based initiatives in clinical trials to improve DEI”, “Barrier to minority enrollment in clinical trials”, “Education to improve DEI in clinical trials”, “Direct recruitment in clinical trials”, “Digital interventions in clinical trials”, “Digital interventions to recruit remote populations” and “Decentralization of clinical trials”.
Terms resulted in a collection of roughly 3253 papers from 2000 to 2024; the papers were then further filtered using abstract screening for relevance. This filtering included (1) filtering for primary sources and conducted studies (2) papers in the English language (3) studies done in the United States (4) setting of both remote and in-person; hospitals, clinical care centers, online, social media, etc (5) English as the language of publication (6) exclusion of duplicate publications. After applying these inclusion/exclusion criteria, the collection was put through thematic and content analysis to categorize the relevant publications into the following subcategories: current state of DEI, community based interventions, education, direct recruitment, digital interventions, digital tools, and decentralization. After the abstract screening for relevance and the thematic and content analysis, the collection was brought to 47 final papers. No other automation tools were used in the screening process.
Results
Current State of DEI in Clinical Trials
Over the past decades, the role of DEI in clinical trials has substantially increased: NIH-funded studies had about a 4-fold increase in minority inclusion from 2.78% (22 clinical trials with minority inclusion out of 791 total NIH trials in 1993) to 11.1% (187 clinical trials with minority inclusion out of 1684 total NIH trials in 2018) and non-NIH-funded studies has a 2.2-fold increase in minority inclusion from 1.23% (45 clinical trials with minority inclusion out of 3648 total Non-NIH trials in 1993) to 2.78% (177 clinical trials with minority inclusion out of 5822 total Non-NIH trials in 2018)5. However, this modest increase is not representative of the population considering the rapidly increasing diversity of the United States; the total minority inclusion in all clinical trials (NIH and Non-NIH) in the US during the past 25 years was only 3.95% (10057 trials included minority inclusion out of the 254535 total trials), while 41.6% of the country was classified as non-white in 2023 by the US Census Bureau6. Furthermore, the 2022 National Academies report displays an increase in the inclusion of white women, but not for individuals of underserved racial and ethnic backgrounds7 Gender equality and women’s rights are being prioritized in healthcare, as shown by their increase in inclusion, however, individuals of underserved racial and ethnic backgrounds are not being prioritized in the same way and additional efforts must be included to prioritize these groups.
Not only is DEI in clinical trials increasing at a slower pace than it should be, it is often not a priority within researchers and organizations. In fact, out of the 20,692 trials done in the U.S. from 2000 to 2020, including trials which were unpublished, only 43% of the trials reported data related to the race or ethnicity of the patients4. When organizations conducting clinical trials fail to report the identity of their patients, it indicates that they may not be representative of the diverse population4. While there is limited data on how many clinical trials prioritize the recruitment of minority groups, the statistics of the minority representation clearly shows that recruitment is lacking and there must be further action taken. However, it is important to note that there are underlying root causes of DEI increasing at a slow pace, including institutional and cultural barriers. It is crucial that recruitment initiatives consider these barriers and work to target both the root causes and the current logistical and economic challenges in order to effectively work towards enhancing DEI in clinical trials.
The diversity of societies by ethnicity, race, gender, sexual orientation, and socioeconomic status must be represented in clinical trials to ensure their generalizability; without this representation, the results of clinical trials will not be relatable to all populations, increasing the disparities. In addition, the inclusion of minority groups is critical for better quality of life and economic reasons. Clinical trials recruitment is commonly an additional cost to the study; studies show that only 55% of trials recruit the target sample size that was originally targeted and racial and ethnic recruitment is even more limited in trials8. While there are other reasons that trials are unable to recruit their target sample size including accessibility and practical challenges, the additional cost and effort needed is a common excuse for not having minority representation in clinical trials. However, the economic benefit of including minority patients outweighs the negative cost burden of recruitment. A recent analysis of the Food and Drug Administration (FDA) drug approvals from 2014 to 2021 found that there is a large economic cost of having populations underrepresented in clinical trials9. A model developed by the USC Schaeffer Center found the economic cost of the next 25 years due to various medical concerns such as reduced life expectancy, shortened disability-free lives, and lack of working with underrepresented populations in clinical trials: if only 1% of health disparities were alleviated by increasing diversity within clinical trials, there would be over $40 million and $60 billion gains in diabetes and heart disease, respectively10. Thus, with a goal of either increased generalizability for a stronger public health system or national economic growth, DEI in clinical trials is crucial and must be prioritized.
The current methods of increasing DEI in clinical trials can be categorized into two major groups: (1) Community initiatives that rely on targeting the specific populations of interest using educational tools and direct recruitment interventions (2) Digital interventions that use technology for both recruitment specialists and devices accessible to the patients to further the reach of minority recruitment and allow for remote data collection.
Overview of Key Studies
| Key Study Title | Section | Summary |
| Community-based interventions for health promotion and disease prevention in noncommunicable diseases: a narrative review | Current State of DEI in Clinical Trials | A model developed by the USC Schaeffer Center found that there is an economic benefit to incorporating DEI in clinical trials. |
| Training needs of clinical and research professionals to optimize minority recruitment and retention in cancer clinical trials | Education | Researchers created a Massive Open Online Course (MOOC) which was implemented on Coursera and, after using pre-tests and post-tests, found that the mean number of correct answers relating to DEI rose from 15.4 to 18.7. Additionally, 71.4% of the respondents said that they were extremely likely to make positive and equitable changes to their recruitment practice. |
| Barriers to clinical trial enrollment in racial and ethnic minority patients with cancer | Education | A study looking at the negative attitudes regarding clinical trials that patients may have found that attitudes improved by 6% using a multimedia psychoeducational intervention and 4% using a printed education material approach. |
| Design and implementation of a massive open online course on enhancing the recruitment of minorities in clinical trials – faster together | Direct Recruitment | Project DC-Hope, one NIH-DC Initiative to Reduce Infant Mortality in Minority Populations in the District of Columbia evaluating the relationship between smoking, depression and intimate partner violence in African American populations, used community based initiatives. By specifically recruiting women presenting for prenatal care at six local clinics by going to various sites and explaining the study along with answering their questions, 85% consented to the study and it had a 79% retention rate. |
| Race, ethnicity, and nih research awards | Direct Recruitment | RECRUIT, a cluster randomized recruitment trial, promoted relationships between physician-investigators and minority-serving physicians and found that 75% of their trials had an increase in minority referrals compared to the control trials. |
| Using mobile technology to engage sexual and gender minorities in clinical research | Digital Tools: Social Media/Internet | A study aimed at seeing the difference in recruitment of African American women with untreated prehypertension between using online outreach and traditional methods found that of 176 inquiries, 44% were from advertisements posted on the University website and 13% were from Facebook or Craigslist (both online outreach methods). |
| Strategies to improve diversity, equity, and inclusion in clinical trials | Decentralization (Geographic) | An Apple Heart Study, studying the identification of atrial fibrillation, 400,000 participants were recruited and enrolled within 9 months through applications on the iPhone. |
Community Based Intervention
Community-based interventions are those that specifically target a given minority population in a community11. One of the major reasons that there is a lack of minority participation in clinical trials is the failure of considering their situation in terms of invitation, accessibility, and other barriers12. Community based initiatives work to address these issues directly and target these minority groups.
Education
An important community based intervention that can increase clinical trial representation is education of researchers, their teams, and the public. Underrepresentation of racial and ethnic minorities has commonly been attributed solely to the distrust of researchers or lack of education. However, recent findings suggest that the research teams contribute to underrepresentation because of their lack of knowledge regarding the importance of DEI and lack of skills associated with recruitment and retention of minority groups13. For example, a study investigating the limited enrollment of minorities in cancer clinical trials among five national institutions part of the consortium for Enhancing Minority Participation in Clinical Trials (EMPaCT) sought to understand the impact of education on recruitment14. Using qualitative research findings, they found that the four key stakeholder groups, principal investigators, research staff, referring clinicians, and cancer center leaders, were not receiving training on the recruitment and retention of minority groups; specifically, the training they were receiving was focused on federal guidelines and ethical principles rather than a focus on minority populations14. This emphasizes the need for education methods for healthcare specialists so that they can actively prioritize recruitment.
One study that looked into educating researchers consisted of an interdisciplinary team with clinical researchers, community engagement specialists, minority clinical trial recruitment/retention educators, and knowledge management information scientists. The group created an evidence-based Massive Open Online Course (MOOC) which was implemented on Coursera, an online learning platform13. MOOC included 8 modules aimed to improve understanding of minority recruitment such as understanding the need for increased minority recruitment in clinical trials; key principles of community engagement; educating potential research participants; outreach with community healthcare providers; effective screening, education, and decision support13. They used a pretest and a posttest aimed at improving the knowledge and skills required to enhance the recruitment and retention of racial and ethnic minorities to see the impact of the education material13. The questions on the tests were split into two major groups of attitudes and intentions; for example, the test-takers were asked about using their thoughts on community relationships to improve participation of ethnic/racial minorities, via communication with community providers to increase clinical trial referrals, their intention to educate potential participants and members of the community, etc.13. They found that the mean number of correct answers increased from 15.4 to 18.713. Furthermore, 71.4% of the respondents said that they were extremely likely to make positive and equitable changes to their recruitment practices13. This study demonstrates that by educating the primary recruiters in clinical trials, minority recruitment can be enhanced. In fact, another study that used community-based academic sites to improve cultural competence among health providers, found a 62% increase in the enrollment of African-American patients in non-therapeutic cancer clinical trials from 2012-201415.
In addition to education for recruitment specialists, educating the public regarding their eligibility for trials and the benefits of clinical trials to their community can increase minority participation. A major barrier to minority enrollment is that patients are unaware of their eligibility for trials16. Furthermore, they may hold negative attitudes towards clinical trials stemming from their distrust in the healthcare system, leading to lack of willingness to participate in the studies17. For example, a systematic review by Manne et al. evaluating the factors that influence participation among African American candidates reports that the strongest inhibitors participation are low levels of knowledge and lack of awareness of clinical trials. However, it is important to note that the lack of education for the public is not the only reason for under enrollment in clinical trials; in fact, studies show that, when offered, minorities are just as likely to join clinical trials18. Even so, the prioritization of educating the public about their eligibility and benefits of participating in clinical trials can help decrease the enrollment gap.
One study aimed to improve the knowledge and attitudes regarding clinical trials to increase participation among racial and ethnic minorities with cancer19. A multicenter, randomized trial of a web-based and interactive educational tool was created to improve preparation for making decisions about the clinical trials by improving attitudes and knowledge of the patients19. The control group received general texts about clinical trials from the National Cancer Institute while the intervention group watched individually tailored videos regarding knowledge and attitude barriers for clinical trial participation, which are obstacles that result from patients preconceived notions and beliefs of clinical trials19. The results were taken from a survey containing questions about the barriers; for example, the knowledge barrier questions included information about the use of placebos and informed consent, and the attitude barrier questions included information about side effects of trial participation and health insurance19. The results showed that both groups had improved knowledge compared to baseline levels but the intervention group had a significantly greater increase19.
Another study looked at the negative attitudes regarding clinical trials that patients may have; this intervention did not specifically use minority participants but found that the results can be applied to different patient demographics20. It assigned 472 adults with cancer to either a multimedia psychoeducational intervention or a printed education material20. When the attitude of an individual at baseline was compared to his/her secondary attitude taken 7-28 days later, patients of the intervention group had more positive attitudes towards clinical trials20. Specifically, attitudes improved by 6% for the multimedia psychoeducational intervention compared to 4% in the printed education material; both groups had a greater willingness to participate in clinical trials20. These interventions can decrease the knowledge barrier relating to clinical trials and their purpose to encourage minority participation, by using cultural awareness.
Direct recruitment
Direct recruitment interventions are a common type of community-based solution which work to directly and intentionally target patients of minority groups or specific minority communities. As stated earlier, oftentimes the lack of DEI in clinical trials can be attributed to the lack of effort in acquiring patients of minority groups; direct recruitment interventions prioritize intentional recruitment so that more minority groups are able to be recruited and retained in various studies21.
Directly inviting minority populations into clinical trials by going to their specific locations can greatly improve their representation within the study. Project DC-Hope, one NIH-DC Initiative to Reduce Infant Mortality in Minority Populations in the District of Columbia evaluating the relationship between smoking, depression and intimate partner violence in African American populations, used community based initiatives22. This project specifically recruited women presenting for prenatal care at six local clinics; rather than physicians recommending specific patients to given clinical trials or choosing populations that are more accessible to their physical location, the trial recruiters went to various sites and explained the study along with answering their questions22. Out of 4,213 women approached for the first part of the trial, 2,913 were screened for demographic and risk eligibility and it was found that there would be 1,398 eligible women; 85% consented to the study and it has a 79% retention rate; about 98% of the women in the trial were African American, showing that if specific groups are targeted by recruiters and an effort is made to recruit them into the trial, there will be many individuals who will voluntarily get involved22. In fact, minorities are as likely to participate in trials as whites when offered; an assessment of approximately 4,000 racially diverse cancer patients saw no association between race or ethnicity and willingness to participate or refuse a trial23. Furthermore, using key location points such as religious centers, community clinics and other stakeholder locations is a strategic and beneficial way to involve more minority individuals who concentrate in specific locations.
Another type of direct recruitment is using strategic planning. In this method, key stakeholders and vital organizations, selected based upon their racial and ethnic diversity, are contacted and invited to conferences and meetings in order to increase minority enrollment21. The National Lung Screening Trial (NLST) used this approach in order to increase their minority recruitment. Out of the 18,842 participants that were enrolled, 8.4% of them were minority participants; 77.6% of these minority participants were accounted for by the institutions used for strategic planning24. The authors found that institutions that used strategic planning, including trusted community clinicians, social and religious organizations, businesses, and local medical providers, enrolled more minority patients than those who did not use these efforts24. Thus, in addition to coming to the location where more minority participants are located, focusing on community stakeholder institutions can greatly increase minority recruitment.
While specifically going into minority communities and targeting stakeholders are prominent examples of direct recruitment interventions, physician-patient interactions can also help directly recruit more minority patients in two main ways: (1) when investigators and local physicians develop strong relationships or (2) specialists are part of minority populations. Although there are no specific studies that investigate the implicit biases of healthcare professions as barriers to recruitment, studies show that some physicians may have negative attitudes toward specific racial and ethnic groups leading them to have a distorted vision of their candidacy for clinical trials21. Focusing on direct recruitment starting from physicians and recruitment specialists can be an effective method of directly increasing minority enrollment.
Studies find that well-established community relationships built on trust are commonly the most successful in the recruitment and retention of minority populations25. Furthermore, physician referral is one of the most effective means of recruiting minority patients. However, when there is not a strong relationship between the physicians and the recruitment specialists, physician referral can be a potential barrier rather than a tool to increase recruitment. Evidence supports that when promoting relationships between the physician and patient, the referrals increase for minority patients. RECRUIT, a cluster randomized recruitment trial, was conducted between July 2013 and April 201726. By promoting relationships between physician-investigators and minority-serving physicians, RECRUIT found that 75% of their trials had an increase in minority referrals compared to the control trials26.
In addition, cultural representation within recruitment specialists can have a positive impact on minority inclusion. Racial and ethnic minorities are underrepresented not only in clinical trials, but among investigators in clinical research. Out of 600,000 faculty and academic institutions, less than 5% are African American, 3% Hispanic, and 1% Native American27. The same underrepresentation is present for women; only 17.9% of women were lead authors in randomized controlled trials in oncology between 2003 and 201828. When minorities are underrepresented in the healthcare field itself, including recruitment specialists, there is underrepresentation of the minority groups of interest. For example, heart failure trials that were driven by a female investigator, had a 50% increase within the proportion of female patients29. Furthermore, a study done by the Duke Cancer Institute called the Office of Health Equity and Disparities demonstrated that racial and ethnic minority participation rates were higher in centers with a diverse workforce30. Thus, increased hiring of specialists from underrepresented groups or providing increased support for these individuals throughout training, can increase direct recruitment of minority patients.
Digital Interventions
Digital interventions focus on the use of technology and artificial intelligence (AI) to increase the recruitment of minority candidates31.They focus on addressing the goal to increase DEI in clinical trials by specifically targeting minority patients and focusing on decentralization of trials to increase accessibility using new and innovative technology.
Digital tools: Social Media/Internet
Online outreach is a form of digital intervention which utilizes internet and social media platforms for recruitment of minority candidates32. This form of outreach can use filtering tools to exclude individuals who are not eligible or not interested in studying, thus increasing the ability to specifically focus on target demographics32. One specific study aimed to see the difference in recruitment of African American women with untreated prehypertension between using online outreach and traditional methods33. Online outreach methods included posting advertisements on Facebook, Craigslist and the University website; traditional methods included promoting with flyers, health fairs, and clinics33. The study found that of 176 inquiries, 44% were from advertisements posted on the University website and 13% were from Facebook or Craigslist33. The online outreach methods were more successful that the traditional methods, emphasizing their benefits and potential to recruit minority candidates more effectively. Furthermore, recruiting LGBTQ+ minorities has proved to be more successful if done digitally. The PRIDE (Population Research in Identity and Disparities for Equality) Study is the largest online study of the health of sexual and gender minorities; this study recruited 20,000 sexual and gender minorities through the use of mobile applications and social media34. Additionally, a study which compared online and in-person recruitment of LGBT individuals for baseline survey data found that the survey completion rate of individuals who clicked on the social media ad was higher than those asked to complete a survey in person at locations like nightclubs and bars35. Therefore, the use of social media and digital recruitment can be more effective and save time and effort for both the patient and the recruitment specialists.
In addition to websites and social media, online outreach includes targeted advertisement tools. These tools include the “Honest Broker methods”, research registries and proprietary algorithms. Honest broker entities are health information integration systems that use AI to recognize de-identified patient databases and health records, increasing the participant pool by identifying larger numbers of potential participants36. Examples of these systems include the Michigan Depression Outreach and Collaborative Care (M-DOCC), Michigan Cardiovascular Outcomes Research and Reporting Program (M-CORRP)36. These methods use electronic health record data to increase outreach and patient participation and show potential to be utilized for minority candidates, however, there is limited evidence of them targeting minorities36. Research registries refer to a database of potential patients who have agreed to be contacted regarding their eligibility in given clinical trials. These databases exist for many minority groups including LGBTQ+, Asians, African Americans, Hispanics, etc37. There are no such studies comparing the use of research registries compared to other methods, however, they provide a pool of willing participants to increase minority recruitment and make it more efficient. Lastly, proprietary algorithms can be developed by tech companies that allow researchers to set target demographics. These targets can be in terms of age, location, education, relationship status, interests, and income38. These algorithms can improve the reach of recruitment materials by reaching rural and remote populations. Furthermore, a significant benefit is that the study teams can tailor their searches so they use their budget efficiently rather than wasting resources on patients that would not qualify for the trials38.
Decentralization (Geographic)
Decentralization of clinical trials refers to conducting them in locations/spaces that are not “typical” for randomized control trials (RCTs)39. Decentralization includes using new technology to increase recruitment of minority groups in various spaces, most commonly remotely and online. Decentralization addresses various different barriers including less accessibility due to location, less accessibility due to low socioeconomic status (SES), language barriers and more. Decentralizing clinical trials can help various different minority groups in their ability to participate in the trial itself. Digital tools that support decentralization are found in both the clinics and in patients’ homes40. Researchers have begun to use these tools in order to allow clinical studies to continue regardless of geographic limitations of conventional randomized controlled trials41.
A critical barrier to enrollment is the consenting procedure for patients joining a clinical trial. For minority patients, the large burden comes in the form of a language barrier; in fact, patients with limited English proficiency (LEP) face health care issues including the under enrollment in trials42. A US Census Bureau’s American Community Survey (2014-2018) reported that 21.6% of the US population spoke a language other than English at home and 8.4% spoke English “less than very well”43. Communication in patients’ primary languages is essential to recruitment in clinical trials in regards to discussion about trials, medical appointments, etc.44‘45. Enrolling LEP patients in clinical trials potentially can be increased through the use of digital tools in decentralizing the consent process because consent forms are not able to be converted into every language46. Although there is limited evidence of increased recruitment of minority candidates due to electronic consenting, the possibility provides a way to address both transportation and language barriers.
Digital health technology that is accessible to patients can further address transportation issues and access rural populations. There are many participation barriers to patients who want to join clinical trials but face logistical and SES barriers. A 2016 study found that for cancer clinical trials, patients with an annual household income of below 50,000 USD were 32% less likely to participate in the clinical trials compared to patients with an annual household income of above 50,000 USD47. Often the lack of transportation, limited childcare availability, language barriers, financial difficulty and burdensome procedures decrease minority enrollment48. In clinical trials, low SES patients have substantial issues finding affordable and efficient transportation to clinical trial sites49. In terms of transportation, 5.8 million persons in the US delayed medical care in 2017 because of transportation difficulty; of these people Hispanics, people living below the poverty threshold and people with functional limitations were more likely to face a transportation barrier50. This enrollment gap in clinical trials enrollment is uniquely problematic because clinical trial participation can be an opportunity for patients of low SES to receive medications which may not be otherwise affordable49. Furthermore, other than SES, 22% of the rural population is non-White; this means that one person in every 25 in the US is a non-White rural person51. The geographic barrier along with other minority specific barriers including distrust and uneducation can deter minority enrollment. This issue is seen both in the recruitment and the process of the clinical trials. Thus, decentralization can be a major advantage in minority recruitment.
Use of digital technology helps decentralization of clinical trials because more patients that are eligible for the trial can be contacted rather than those who are closer to the study area. Furthermore, for tools in patients homes, 97% of all U.S. adults owned mobile phones, including 98% of Black adults, 92% of high school graduates and 95% of those making less than $30,000 a year52. Thus, using these technologies, which are common throughout households, can be a way to conduct clinical trials remotely. For example, in an Apple Heart Study, studying the identification of atrial fibrillation, 400,000 participants were recruited and enrolled within 9 months through applications on the iPhone41. This successful study provides evidence that remote data collection is possible and may be extremely beneficial in the future. Furthermore, mobile clinical trials have been proven to include more minorities and women in trials because of the increased accessibility53. These types of remote data collection approaches can help reduce transportation barriers for minority groups of low SES status. Furthermore, they can help reach remote populations; meaning they can reach the 22% of the rural population which are minority candidates51. Although there is limited research in digital tools in patients homes increasing minority recruitment, the reach of remote data collection has been shown to be successful.
Overview of Methods to Increase DEI in Clinical Trials
Discussion
Incorporation of DEI methods in clinical trials has become an increasingly important conversation. Although the implementation of DEI methods has increased recently, the rate of implementation is not comparable to the increasing diversity of the United States. As a result, minority groups by ethnicity, race, gender, sexual orientation, and socioeconomic status are still underrepresented in clinical studies5. This is problematic for two major reasons: (1) without diversity in clinical trials, the results are not generalizable to the entire population (2) there are economic harms of not having equitable healthcare for all. In this review, we identified and evaluated several important methods of improving DEI in clinical trials and their effectiveness in order to improve minority quality of life.
The two major groups of methods found were community-based initiatives and digital interventions. First, community-based initiatives include initiatives that directly target minority populations13. These included education for patient recruitment specialists and the general public; these also consisted of direct recruitment interventions in terms of specific location, strategic planning, directly interacting with stakeholders, bettering physician relationships and diversification of patient recruitment specialists. Second, digital interventions include using new technology in order to recruit and allow minority groups to participate in clinical trials32. These included digital tools like social media, the internet, and electronic health records; they also included the use of devices accessible to patients such as phones and smartwatches. It is important to note there are other methods being currently used including improving healthcare like policy in terms of DEI and financial incentives. However, we were unable to find extensive evidence of their efficacy, and thus, focused on the two major sections of community based initiatives and digital intervention. Additionally, it is important to note that all the approaches included in the manuscript have the potential for bias based upon the individuals conducting the study and the research methods used. These challenges of bias can be addressed through careful study designs, transparent reporting, following of ethical guidelines and ongoing evaluation.
We first noticed that community-based initiatives were the most frequently used in clinical trials to improve DEI because it was specific targeting. Furthermore, it addressed some of the root causes of less minority inclusion in trials including previous implicit and explicit biases and negative perspective 18. Using the education of physicians and patient recruitment specialists, they were able to further explore the importance of DEI in clinical trials, which was successful in the given studies. Using this education as a baseline, the other interventions such as direct recruitment based on location and stakeholders were also effective. These types of approaches are less difficult to do in comparison to some digital interventions because many do not require extensive development. For example, talking to community stakeholders may take time, but it has extreme benefits in recruitments and is relatively straightforward25. With education, it is a similar case because many education materials are available online including sites like Coursera14. However, the major drawback of this type of intervention is that it requires accessible resources and a time commitment. In order to educate specialists and the public, there must be reputable research and time dedicated to these tasks; in direct recruitment, traveling to different populations and communicating with stakeholders may be a burden and create extra costs in the long run. Additionally, direct recruitment methods may include having unintended consequences like excluding certain individuals from recruitment. For example, the two largest figures in direct recruitment are stakeholders and physicians, so recruitment depends on the specific locations or individuals they choose to target, which can inadvertently exclude certain groups of patients25. Furthermore, educational approaches must be created with the goal of sustainability because as the patient population grows more diverse, the education methods and materials must be adapted to continue to serve their purpose of enhancing DEI in clinical trials. Nonetheless, community-based initiatives have been proven successful and are currently effective methods being used to increase DEI in clinical trials.
In terms of digital interventions, they are also very common in the recruitment of minority candidates. In this century, social media is very popular and is often a useful tool in order to attract certain populations; many studies used this narrative in order to recruit specific ethnic, racial, and sexual minorities in their clinical trials successfully33. Digital interventions using the internet and social media have the potential to exponentially grow because of the increased dependence upon and use of social media. Furthermore, this intervention does not have many transportation or development barriers because of the accessibility of social media and the internet. The other type of digital intervention is decentralization through the use of tools in patients homes. Like social media, phones have been proven to be a common device in the majority of adults homes in the United States, thus they allow remote data collection41. These solutions address minorities living in remote populations or those who cannot cover transportation costs. However, there are still limitations in accessibility with digital tools since some patients and communities do not have access to cellular devices and the technology used in these methods may not be accessible for all individuals of a rural population. Furthermore, for both digital interventions and decentralization methods, ethical concerns surrounding data security may arise based on individual studies, an issue which can take significant time and money to solve. Regardless, digital interventions seem to be the future of DEI in clinical trials: they take development but they increase the accessibility of trials greatly to many different minority groups. Especially in the age of the internet, many barriers can be overcome using digital tools including geographical, financial, attitudinal, and language barriers.
Conclusion
The current literature shows that DEI methods can be effective in recruiting diverse populations, and barriers to their implementation negatively impact clinical trial validity. Importantly, many clinical trials are not held accountable for patient recruitment through a DEI lens. Even with various methods to increase DEI in clinical trials, recruiters and recruitment specialists must actively prioritize DEI in order to create equitable results for the trials. Using the methods reviewed in this study more frequently with more purpose will increase DEI in clinical trials, thus setting a standard for American healthcare.
Acknowledgements
Riya Mehrotra was the primary author for this literature review. She conducted the primary literature review, drafted and wrote the paper, and created the figures. Frank Glover was the mentor for Riya Mehrotra. He assisted with the literature review and guided Riya throughout the editing process.
References
- C. A. Umscheid, D. J. Margolis, C. E. Grossman, Key concepts of clinical trials: a narrative review. Postgraduate Medicine. 123,194–204 (2011). [↩]
- S. Goldstein, Beta blocker therapy in african american patients with heart failure. Heart Failure Reviews. 9, 161-167 (2004). [↩]
- E. Y. Adashi, I. G. Cohen, The fda initiative to assure racial and ethnic diversity in clinical trials. Journal of the American Board of Family Medicine. 36, 366–368 (2023). [↩] [↩]
- B. E. Turner, J. R. Steinberg, B. T. Weeks, F. Rodriguez, M. R. Cullen, Race/ethnicity reporting and representation in us clinical trials: a cohort study. Lancet Regional Health Americas. 11,100252 (2022). [↩] [↩] [↩]
- M. A. Ma, D. E. Gutiérrez, J. M. Frausto, W. K. Al-Delaimy, Minority representation in clinical trials in the united states: trends over the past 25 years. Mayo Clinic Proceedings. 96, 264–266 (2021). [↩] [↩] [↩] [↩] [↩]
- United States Census Bureau. QuickFacts. (2023). [↩]
- National Academies of Sciences, Engineering, and Medicine; Policy and Global Affairs; Committee on Women in Science, Engineering, and Medicine; Committee on Improving the Representation of Women and Underrepresented Minorities in Clinical Trials and Research; Improving representation in clinical trials and research: building research equity for women and underrepresented groups. (2022). [↩]
- K. Allison, D. Patel, R. Kaur, Assessing multiple factors affecting minority participation in clinical trials: development of the clinical trials participation barriers survey. Cureus. 14, e24424 (2022). [↩]
- A. K. Green, N. Trivedi, J. J. Hsu, N. L. Yu, P. B. Bach, S. Chimonas, Despite the fda’s five-year plan, black patients remain inadequately represented in clinical trials for drugs: study examines fda’s five-year action plan aimed at improving diversity in and transparency of pivotal clinical trials for newly-approved drugs. Health Affairs. 41, 368–374 (2022). [↩]
- D. Goldman, E. Perez, C. del Rio, Lack of diversity in clinical trials costs billions of dollars. incentives can spur innovation. https://healthpolicy.usc.edu/article/lack-of-d iversity-in-clinical-trials-costs-billions-of-dollars-incentives-can-spur-innovation/ (2022). [↩]
- P. M. Philip, S. Kannan, N. A. Parambil, Community-based interventions for health promotion and disease prevention in noncommunicable diseases: a narrative review. Journal of Education and Health Promotion. 7, 141 (2018). [↩]
- M. N. El-Khorazaty, A. A. Johnson, M. Kiely, A. A. El-Mohandes, S. Subramanian, H. A. Laryea, K. B. Murray, J. S. Thornberry, J. G Joseph, Recruitment and retention of low-income minority women in a behavioral intervention to reduce smoking, depression, and intimate partner violence during pregnancy. BMC public health. 7, 233 (2007). [↩]
- S. V. Kusnoor, V. Villalta-Gil, M. Michaels, Y. Joosten, T. L. Israel, M. I. Epelbaum, P. Lee, E. T. Frakes, J. Cunningham-Erves, S. A. Mayers, S. C. Stallings, N. B. Giuse, P. A. Harris, C. H. Wilkins, Design and implementation of a massive open online course on enhancing the recruitment of minorities in clinical trials – faster together. BMC medical research methodology. 21, 44 (2021). [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- S. J. Niranjan, R. W. Durant, J. A. Wenzel, E. D. Cook, M. N. Fouad, S. M. Vickers, B. R. Konety, S. B. Rutland, Z. R. Simoni, M. Y. Martin, Training needs of clinical and research professionals to optimize minority recruitment and retention in cancer clinical trials. Journal of Cancer Education: The Official Journal of the American Association for Cancer Education. 34, 26–34 (2019). [↩] [↩] [↩]
- S. F. Wallington, C. Dash, V. B. Sheppard, T. D. Goode, B. A. Oppong, E. E. Dodson, R. N. Hamilton, L. L. Adams-Campbell, Enrolling minority and underserved populations in cancer clinical research. American Journal of Preventive Medicine. 50, 111–117 (2016). [↩]
- J. G. Ford, M. W. Howerton, G. Y. Lai, T. L. Gary, S. Bolen, M. C. Gibbons, J. Tilburt, C. Baffi, T. P. Tanpitukpongse, R. F. Wilson, N. R. Powe, E. B. Bass, Barriers to recruiting underrepresented populations to cancer clinical trials: a systematic review. Cancer. 112, 228–242 (2008). [↩]
- S. Manne, D. Kashy, T. Albrecht, Y. N. Wong, A. Lederman Flamm, A. B. Benson, S. M. Miller, L. Fleisher, J. Buzaglo, N. Roach, M. Katz, E. Ross, M. Collins, D. Poole, S. Raivitch, D. M. Miller, T. G. Kinzy, T. Liu, N. J. Meropol, Attitudinal barriers to participation in oncology clinical trials: factor analysis and correlates of barriers. European Journal of Cancer Care. 24, 28–38 (2015). [↩]
- D. Wendler, R. Kington, J. Madans, G. Van Wye, H. Christ-Schmidt, L. A. Pratt, O. W. Brawley, C. P. Gross, E. Emanuel, Are racial and ethnic minorities less willing to participate in health research?. PLoS medicine. 3, e19 (2006). [↩] [↩]
- N. J. Meropol, Y. Wong, T. Albrecht, S. Manne, S. M. Miller, A. L. Flamm, A. B. Benson, J. Buzaglo, M. Collins, B. Egleston, L. Fleisher, M. Katz, T. G. Kinzy, T. M. Liu, S. Margevicius, D. M. Miller, D. Poole, N. Roach, E. Ross, M. D. Schluchter, Randomized trial of a web-based intervention to address barriers to clinical trials. Journal of Clinical Oncology: Official Journal of the American Society of Clinical Oncology. 34, 469–478 (2016). [↩] [↩] [↩] [↩] [↩]
- P. B. Jacobsen, K. J. Wells, C. D. Meade, G. P. Quinn, J. Lee, W. J. Fulp, J. E. Gray, R. C. Baz, G. M. Springett, R. M. Levine, M. Markham, G. M. Schreiber, T. H. Cartwright, J. M. Burke, R. D. Siegel, M. P. Malafa, D. Sullivan, Effects of a brief multimedia psychoeducational intervention on the attitudes and interest of patients with cancer regarding clinical trial participation: a multicenter randomized controlled trial. Journal of Clinical Oncology: Official Journal of the American Society of Clinical Oncology. 30, 2516–2521 (2012). [↩] [↩] [↩] [↩]
- L. M. Hamel, L. A. Penner, T. L. Albrecht, E. Heath, C. K. Gwede, S. Eggly, Barriers to clinical trial enrollment in racial and ethnic minority patients with cancer. Cancer Control: Journal of the Moffitt Cancer Center. 23, 327–337 (2016). [↩] [↩] [↩]
- M. N. El-Khorazaty, A. A. Johnson, M. Kiely, A. A. El-Mohandes, S. Subramanian, H. A. Laryea, K. B. Murray, J. S. Thornberry, J. G Joseph, Recruitment and retention of low-income minority women in a behavioral intervention to reduce smoking, depression, and intimate partner violence during pregnancy. BMC public health. 7, 233 (2007). [↩] [↩] [↩]
- A. T. Langford, K. Resnicow, E. P. Dimond, A. M. Denicoff, D. Germain, W. McCaskill-Stevens, R. A. Enos, A. Carrigan, K. Wilkinson, R. S. Go, Racial/ethnic differences in clinical trial enrollment, refusal rates, ineligibility, and reasons for decline among patients at sites in the national cancer institute’s community cancer centers program. Cancer. 120 ,877–884 (2014). [↩]
- C. Duda, I. Mahon,M. H. Chen, B. Snyder, R. Barr, C. Chiles, R. Falk, E. K. Fishman, D. Gemmel, J. G. Goldin, K. Brown, R. F. Munden, K. Vydareny, D. R. Aberle, Impact and costs of targeted recruitment of minorities to the national lung screening trial. Clinical Trials (London, England). 8, 214–223 (2011). [↩] [↩]
- A. K. Yancey, A. N. Ortega, S. K. Kumanyika, Effective recruitment and retention of minority research participants. Annual Review of Public Health. 27, 1–28 (2006). [↩] [↩] [↩]
- B. C. Tilley, A. G. Mainous, R. P. Amorrortu, D. M. McKee, D. W. Smith, R. Li, S. M. DeSantis, S. W. Vernon, G. Koch, M. E. Ford, V. Diaz, J. Alvidrez, Using increased trust in medical researchers to increase minority recruitment: the recruit cluster randomized clinical trial. Contemporary Clinical Trials. 109, 106519 (2021). [↩] [↩]
- D. K. Ginther, W. T. Schaffer, J. Schnell, B. Masimore, F. Liu, L. L. Haak, R. Kington, Race, ethnicity, and nih research awards. Science (New York, N.Y.). 333, 1015–1019 (2011). [↩]
- E. B. Ludmir, W. Mainwaring, A. B. Miller, T. A. Lin, A. Jethanandani, A. F. Espinoza, E. B. Holliday. Women’s representation among lead investigators of clinical trials in oncology. JAMA oncology. 5, 1501–1502 (2019). [↩]
- N. Reza, A. S. Tahhan, N. Mahmud, E. M. DeFilippis, A. Alrohaibani, M. Vaduganathan, S. J. Greene, A. H. Ho, G. C. Fonarow, J. Butler, C. O’Connor, M. Fiuzat, O. Vardeny, I. L. Piña, J. Lindenfeld, M. Jessup, Representation of women authors in international heart failure guidelines and contemporary clinical trials. Circulation. Heart Failure. 13, e006605 (2020). [↩]
- N. J. Barrett, T. V. Hawkins, J. Wilder, K. L. Ingraham, W. Worthy, X. Boyce, R. Reyes, M. Chirinos, P. Wigfall, W. Robinson, S. R. Patierno, Implementation of a health disparities & equity program at the duke cancer institute. Oncology Issues. 31, 48–57 (2016). [↩]
- J. B. Levin, M. Sajatovic, M. Rahman, M. E. Aebi, C. Tatsuoka, C. Depp, C. Cushman, E. Johnston, K. A. Cassidy, C. Blixen, L. Eskew, P. J. Klein, E. Fuentes-Casiano, D. J. Moore, Outcomes of psychoeducation and a text messaging adherence intervention among individuals with hypertension and bipolar disorder. Psychiatric Services (Washington, D.C.). 70, 608–612 (2019). [↩]
- J. K. Igwe, T. Wangdak Yuthok, E. Cruz, A. Mueller, R. H. Lan, C. Brown-Johnson, M. Idris, F. Rodriguez, K. Clark, L. Palaniappan, M. Echols, P. Wang, A. Onwuanyi, P. Pemu, E. F. Lewis, Opportunities to increase science of diversity and inclusion in clinical trials: equity and a lack of a control. Journal of the American Heart Association. 12, e030042 (2023). [↩] [↩] [↩]
- B. A. Staffileno, J. Zschunke, M. Weber, L. E. Gross, L. Fogg, C. C. Tangney, The feasibility of using facebook, craigslist, and other online strategies to recruit young african american women for a web-based healthy lifestyle behavior change intervention. The Journal of Cardiovascular Nursing. 32, 365–371 (2017). [↩] [↩] [↩] [↩]
- M. R. Lunn, M. R. Capriotti, A. Flentje, K. Bibbins-Domingo, M. J. Pletcher, A. J. Triano, C. Sooksaman, J. Frazier, J. Obedin-Maliver, Using mobile technology to engage sexual and gender minorities in clinical research. PloS One. 14, e0216282 (2019). [↩]
- J. Guillory, K. F. Wiant, M. Farrelly, L. Fiacco, I. Alam, L. Hoffman, E. Crankshaw, J. Delahanty, T. N. Alexander, Recruiting hard-to-reach populations for survey research: using facebook and instagram advertisements and in-person intercept in lgbt bars and nightclubs to recruit lgbt young adults. Journal of Medical Internet Research. 20, e197 (2018). [↩]
- A. D. Boyd, D. A. Hunscher, A. J. Kramer, C. Hosner, P. Saxman, B. D. Athey, J. F. Greden, D. C. Clauw, The “honest broker” method of integrating interdisciplinary research data. AMIA … Annual Symposium proceedings. AMIA Symposium. 2005, 902 (2005). [↩] [↩] [↩]
- J. K. Igwe, T. Wangdak Yuthok, E. Cruz, A. Mueller, R. H. Lan, C. Brown-Johnson, M. Idris, F. Rodriguez, K. Clark, L. Palaniappan, M. Echols, P. Wang, A. Onwuanyi, P. Pemu, E. F. Lewis, Opportunities to increase science of diversity and inclusion in clinical trials: equity and a lack of a control. Journal of the American Heart Association. 1 v2, e030042 (2023). [↩]
- L. Akers and J. S. Gordon, Using facebook for large-scale online randomized clinical trial recruitment: effective advertising strategies. Journal of Medical Internet Research. 20, e290 (2018). [↩] [↩]
- L. Sacks, The evolving role of decentralized clinical trials and digital health technologies. (2023). [↩]
- D. M. Harmon, P. A. Noseworthy, X. Yao, The digitization and decentralization of clinical trials. Mayo Clinic Proceedings. 98, 1568–1578 (2023). [↩]
- M. V. Perez, K. W. Mahaffey, H. Hedlin,J. S. Rumsfeld, A. Garcia, T. Ferris, V. Balasubramanian, A. M. Russo, A. Rajmane, L. Cheung, G. Hung, J. Lee, P. Kowey, N. Talati, D. Nag, S. E. Gummidipundi, A. Beatty, M. T. Hills, S. Desai, C. B. Granger, S. Desai, M. P. Turakhia, Apple Heart Study Investigators, Large-scale assessment of a smartwatch to identify atrial fibrillation. The New England Journal of Medicine. 381, 1909–1917 (2019). [↩] [↩] [↩]
- J. M. Kahn, D. M. Gray, J. M. Oliveri, C. M. Washington, C. R. DeGraffinreid, E. D. Paskett, Strategies to improve diversity, equity, and inclusion in clinical trials. Cancer. 128, 216–221 (2022). [↩]
- D. A. Evangelista, A. Goodman, M. K. Kohli, S. Bondocgawa Maflamills, M. Samuel-Foo, M. S. Herrera, J. L. Ware, M. Wilson, Why diversity matters among those who study diversity. American Entomologist. 66, 42–49 (2020). [↩]
- J. S. Lee, E. J. Pérez-Stable, S. E. Gregorich, M. H. Crawford, A. Green, J. Livaudais-Toman, L. S. Karliner, Increased access to professional interpreters in the hospital improves informed consent for patients with limited english proficiency. Journal of General Internal Medicine. 32, 863–870 (2017). [↩]
- M. Ju. Addressing health inequities for limited english proficiency patients: interpreter use and beyond. Pediatrics. 147, e2020032383 (2021). [↩]
- J. M. Kahn, D. M. Gray, J. M. Oliveri, C. M. Washington, C. R. DeGraffinreid, E. D. Paskett, Strategies to improve diversity, equity, and inclusion in clinical trials. Cancer. 128, 216–221 (2022). [↩]
- J. M. Unger, J. R. Gralow, K. S. Albain, S. D. Ramsey, D. L. Hershman, Patient income level and cancer clinical trial participation: a prospective survey study. JAMA oncology. 2, 137–139 (2016). [↩]
- D. R. Brown, M. N. Fouad, K. Basen-Engquist, G. Tortolero-Luna, Recruitment and retention of minority women in cancer screening, prevention, and treatment trials. Annals of Epidemiology. 10, S13-21 (2000). [↩]
- K. N. Price, A. B. Lyons, I. H. Hamzavi, J. L. Hsiao, V. Y. Shi, Facilitating clinical trials participation of low socioeconomic status patients. Dermatology. 237, 843–846 (2021). [↩] [↩]
- M. K. Wolfe, N. C. McDonald, G. M. Holmes. Transportation barriers to health care in the united states: findings from the national health interview survey, 1997–2017. American Journal of Public Health. 110, 815–822 (2020). [↩]
- W. E. Zahnd, C. Murphy, M. Knoll, G. A. Benavidez, K. R. Day, R. Ranganathan, P. Luke, A. Zgodic, K. Shi, M. A. Merrell, E. L. Crouch, H. M. Brandt, J. M. Eberth. The intersection of rural residence and minority race/ethnicity in cancer disparities in the united states. International Journal of Environmental Research and Public Health. 18, 1384 (2021). [↩] [↩]
- O. Sidoti. Mobile fact sheet. https://www.pewresearch.org/internet/fact-sheet/mobile/ (2024). [↩]
- Office of Research on Women’s Health. Review of the literature: primary barriers and facilitators to participation in clinical research. https://orwh.od.nih.gov/sites/ orwh/files/docs/orwh_outreach_toolkit_litreview.pdf 1-15. [↩]