Abstract
Diabetes mellitus remains a global health crisis, affecting over 500 million individuals, with insulin therapy being essential for type 1 and advanced type 2 diabetes patients. Traditional subcutaneous insulin injections, though effective, pose challenges such as pain, needle anxiety, and poor adherence, driving the need for non-invasive alternatives. Oral insulin has been proposed as a potential alternative to subcutaneous injections, with theoretical advantages in mimicking the physiological path of insulin secretion. By mimicking the physiological path of pancreatic insulin, oral insulin is expected to have a distinct effect on hepatic insulin resistance, while reducing hyperinsulinemia and hypoglycemia risk compared to subcutaneous delivery. However, the gastrointestinal tract (GIT) presents significant barriers, including enzymatic degradation, poor intestinal permeability, and patient-specific physiological variability. Among currently investigated strategies, nanoparticle-based systems such as polymeric PLGA nanoparticles and liposomes have demonstrated promising preclinical results, but clinical translation remains limited. Permeation enhancers like bile salts and glucose-responsive “smart” systems also show strong translational potential, though long-term safety data remains under investigation. Mucoadhesive polymers (e.g., chitosan) and protease inhibitors face greater challenges due to variability in patient absorption and potential gastrointestinal side effects. While nanoparticle platforms lead to clinical progress, their scalability and cost-effectiveness require further optimization. Successful development of oral insulin could revolutionize diabetes care, offering a patient-friendly, physiologically aligned treatment that improves adherence, reduces complications, and lowers healthcare costs. This review highlights the current advancements, persistent hurdles, and future directions in oral insulin research, underscoring its transformative potential in diabetes therapy.
Keywords: oral insulin, diabetes mellitus, nanoparticle drug delivery, PLGA nanoparticles, liposomes, permeation enhancers, mucoadhesive polymers, glucose-responsive systems
Introduction
Diabetes remains one of the most pressing global health challenges, affecting over 500 million people worldwide. This chronic metabolic disorder is characterized by impaired blood glucose regulation, with two primary forms: Type 1 diabetes, an autoimmune condition requiring lifelong insulin therapy due to the destruction of insulin-producing pancreatic beta cells, and Type 2 diabetes, which involves progressive insulin resistance and beta-cell dysfunction, often necessitating insulin therapy in advanced stages.
Insulin, a peptide hormone secreted by pancreatic beta cells, is central to glucose homeostasis—it promotes cellular glucose uptake, inhibits hepatic glucose production, and facilitates glycogen storage. Without proper insulin function, chronic hyperglycemia leads to devastating complications including neuropathy (nerve damage causing pain and disability), retinopathy (vision loss from retinal damage), and cardiovascular disease (heart attacks and strokes), which collectively account for significant patient morbidity and over $300 billion in annual global healthcare costs.
For individuals with type 1 diabetes and many with advanced type 2 diabetes, insulin therapy is essential to regulate blood glucose levels and prevent these complications. However, the current standard of insulin delivery—subcutaneous injections—poses significant challenges. Frequent injections can cause pain, needle anxiety, and psychological distress, leading to poor adherence and suboptimal glycemic control. Alternative non-invasive insulin options include inhalable insulin (e.g., Afrezza), which was shown in clinical studies to achieve non-inferior HbA1c reductions compared with injectable insulin while offering patient preference for ease of use1. Additionally, non-insulin agents like GLP‑1 receptor agonists and SGLT2 inhibitors have dramatically changed type 2 diabetes care, reducing cardiovascular and kidney outcomes—with SGLT2 inhibitors lowering major adverse cardiovascular events by about 20% and GLP‑1 agonists offering further cardiorenal protection2.
The development of oral insulin has long been a goal in diabetes research, given its potential to provide a non-invasive, convenient alternative. Despite its promise, creating an effective oral insulin formulation has proven to be a formidable challenge due to the harsh environment of the GIT. Enzymatic degradation, poor permeability across the intestinal lining, and patient-specific variability have hindered progress. More recent clinical advances, such as the oral insulin candidate ORMD-0801 (developed by Oramed Pharmaceuticals) demonstrated modest HbA1c reductions (~0.5%) in Phase II trials, but its Phase III study did not meet primary efficacy endpoints, underscoring persistent challenges for oral insulin translation, suggesting incremental progress towards a viable oral insulin therapy3. While ORMD-0801 uses a proprietary enteric coating to protect insulin from gastric degradation, the exact polymer composition has not been fully disclosed in peer-reviewed literature, highlighting a common challenge in industrial transparency for oral peptide delivery systems. Similarly, preclinical studies of Hepatic-Directed Vesicle Insulin (HDV-I) show that it can stimulate hepatic glucose uptake at doses less than 1% of the regular insulin dose required for liver stimulation and can normalize blood glucose curves during oral glucose tolerance tests in pancreatectomized dogs4, indicating the potential for improved physiological testing. However, recent advancements in nanotechnology, enzyme inhibitors, and innovative delivery systems may contribute to the future development of oral insulin therapy, though further clinical validation.
Review Methodology
This review was conducted using a targeted literature search of the PubMed database, focusing on peer‑reviewed articles published between 2000 and 2024. Search terms included “oral insulin,” “diabetes therapy,” “nanoparticle insulin delivery,” and “insulin permeation enhancers.” Studies were selected based on their direct relevance to advances in oral insulin formulations, with an emphasis on clinical trials, preclinical studies, and regulatory guidance documents. Exclusion criteria comprised review articles lacking primary data, case studies, and papers unrelated to insulin delivery or its associated barriers. While this review was not conducted as a formal systematic review following PRISMA guidelines, it aimed to provide a comprehensive and balanced assessment of the literature, highlighting key advances and ongoing challenges in the development and clinical translation of oral insulin formulations.
The Rationale of Oral Insulin
Diabetes mellitus, a chronic metabolic disorder, requires effective and patient-friendly treatment options. Traditional subcutaneous insulin therapy, while effective, is associated with challenges such as patient discomfort, needle phobia, and the risk of injection site infections. Oral insulin has emerged as a promising alternative, offering a non-invasive and physiologically aligned approach to diabetes management.
The primary rationale for oral insulin lies in its ability to mimic the natural pathway of insulin secretion5. In non-diabetic individuals, insulin is released by pancreatic beta cells into the portal vein, which carries blood directly from the intestines to the liver. This first-pass hepatic delivery is critical because the liver—the body’s primary glucose storage and production site—requires higher insulin concentrations than peripheral tissues to properly regulate postprandial glucose metabolism. Subcutaneous insulin bypasses this pathway, often leading to peripheral hyperinsulinemia and hypoglycemia. Oral insulin, if successfully delivered, could restore this physiological process, improving glucose control and reducing side effects (Figure 1).
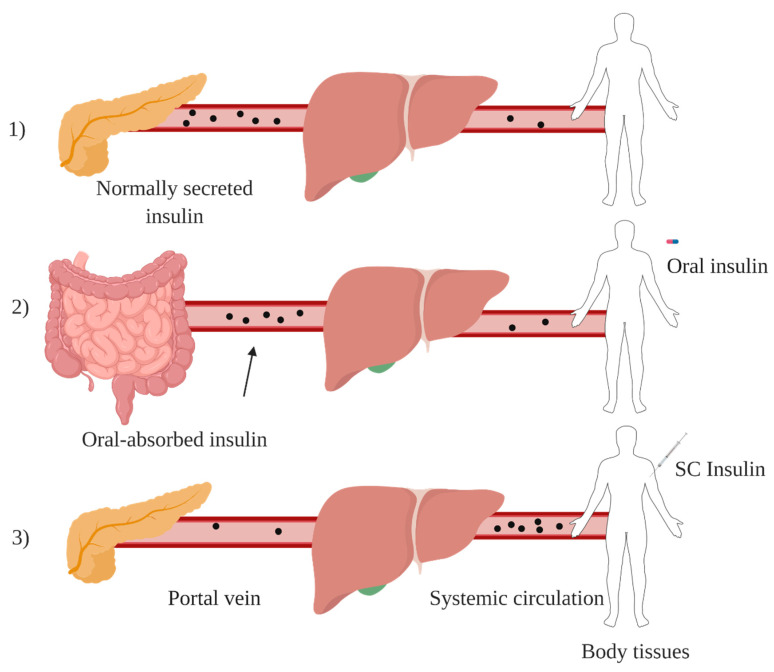
Additionally, oral insulin addresses practical challenges associated with injections, particularly for pediatric and geriatric populations. Its non-invasive nature could enhance patient adherence, simplify diabetes management, and improve quality of life. By aligning with the principles of patient-centered care, oral insulin represents a significant step forward in diabetes therapy, combining scientific innovation with patient convenience.
The Challenges of Developing Oral Insulin
One of the primary obstacles to oral insulin delivery is the harsh environment of the GIT, which is designed to break down complex molecules like proteins. Insulin, a peptide hormone composed of 51 amino acids, is particularly vulnerable to enzymatic degradation7 that dramatically reduces its bioavailability to less than 2% when administered orally8. Proteolytic enzymes such as pepsin in the stomach and trypsin and chymotrypsin in the intestines target insulin’s peptide bonds, breaking it into smaller, inactive fragments. For example, pepsin, which is active in the highly acidic environment of the stomach (pH 2–3)5, cleaves insulin at specific aromatic amino acid residues, rendering it biologically inactive. Similarly, trypsin and chymotrypsin in the small intestine further degrade insulin by hydrolyzing peptide bonds at lysine and arginine residues. In vitro studies indicate that insulin is nearly completely degraded by these pancreatic enzymes within one hour9.
Even if insulin survives enzymatic degradation, its absorption is hindered by the intestinal epithelium, a tightly packed layer of cells that acts as a barrier to large molecules. Insulin’s large molecular size (approximately 5808 Da) and hydrophilic nature make it difficult to cross this barrier10. Clinical data from a crossover study showed that oral insulin formulations achieved maximum fraction absorbed (Fa) of only ~3 %, compared to ~19 % with subcutaneous insulin; the effective permeability coefficient (Peff) for oral insulin was approximately 0.084 × 10⁻⁴ cm/sec versus 0.179 × 10⁻⁴ cm/sec for subcutaneous reference 1 U/kg formulations, illustrating quantitatively poorer intestinal absorption11. Moreover, broad analyses estimate that clinically tested oral peptide/protein drugs generally exhibit bioavailabilities of <1 %, underscoring the physical barriers posed by the intestinal epithelium and mucus layer12. The intestinal epithelium is composed of enterocytes joined by tight junctions, which restrict the paracellular transport of large molecules like insulin. Additionally, the mucus layer coating the intestinal lining further traps insulin, preventing its absorption. This mucus layer, which is composed of glycoproteins and water, acts as a physical barrier that limits the diffusion of insulin to the epithelial surface. Unlike small molecules, which can passively diffuse through the intestinal lining, insulin lacks efficient transport mechanisms in the GIT. These foundational barriers are further magnified by physiological variability: differences in gastric pH and gastrointestinal transit times significantly influence oral peptide dissolution and absorption kinetics13. Moreover, the gut microbiome also plays a critical role—its composition varies widely between individuals and can markedly alter drug metabolism and bioavailability through microbial enzymes that deactivate or transform drugs and by modulating intestinal epithelial function (e.g., drug transporters or barrier integrity)14. From a regulatory perspective, all included studies report that, as of their publication dates, no oral insulin product has received approval from the Food and Drug Administration (FDA) or European Medicines Agency (EMA). Maher and Brayden (2021) is the only study to mention recent FDA approvals for oral peptide drugs (semaglutide and octreotide), but these do not include insulin15. Across the studies, low and variable bioavailability, along with physiological and structural barriers, are consistently identified as major obstacles to regulatory approval and market entry for oral insulin.
The effectiveness of oral insulin is also influenced by patient-specific factors, such as gastric emptying time, intestinal motility, and pH levels16. For example, delayed gastric emptying, a potential issue in patients with diabetes, can alter insulin absorption kinetics. Gastric emptying time can vary significantly between individuals, ranging from 1 to 4h, and is influenced by factors such as meal composition and blood glucose levels. High-fat meals, for instance, can delay gastric emptying, leading to unpredictable insulin absorption and therapeutic outcomes. Additionally, variations in gut microbiota composition can impact insulin absorption and metabolism. The gut microbiota, which consists of trillions of microorganisms, plays a critical role in modulating the intestinal environment. For example, certain bacterial species can produce enzymes that degrade insulin, while others can alter the permeability of the intestinal epithelium. These factors underscore the need for delivery systems that can adapt to individual physiological differences and ensure consistent insulin absorption.
Moreover, the acidic pH of the stomach not only destabilizes insulin’s molecular structure but also makes it more susceptible to enzymatic degredation. The complicated pH environments in the GIT tract could lead to conformational alterations of insulin that expose its peptide bonds to proteolytic enzymes, further reducing its efficacy5. In vitro studies using pH-responsive poly(acrylic acid)-based hydrogel nanocomposites demonstrated over 26 % relative bioavailability compared to subcutaneous insulin and approximately 96 % controlled insulin release over 24 h, reflecting significant protection from acidic degradation17. In vivo, diabetic rodents treated orally with these hydrogel systems showed up to a 52.6 % drop in blood glucose and ~26 % relative bioavailability, indicating effective gastric protection and sustained release17.This harsh environment necessitates the use of protective strategies, such as pH-sensitive coatings or encapsulation, to ensure that insulin remains stable and intact until it reaches the small intestine, where absorption can occur.
Innovative Strategies to Overcome Barriers
Nanoparticle-Based Delivery Systems
Nanoparticles have emerged as a promising solution to protect insulin from enzymatic degradation and enhance its stability in the GIT18. Polymeric nanoparticles, such as those made from poly (lactic-co-glycolic acid) (PLGA), provide a protective barrier against proteolytic enzymes18. Oral and intravenous administration have lessened success as a result of proteolytic degradation. To overcome these issues, attempts have been made to administer greater concentrations to overwhelm proteolysis rates. However, even if enzymes manage to survive till systemic circulation, 100% of large therapeutic molecules fail to traverse the blood brain barrier (BBB)19. These results emphasize the use of PLGA nanoparticles as drug delivery vehicles. Moreover, in vivo studies in diabetic rats showed that pH-sensitive PLGA‑Hp55 nanoparticles achieved a relative bioavailability of 6.27 ± 0.42 % compared to subcutaneous insulin, with a sustained blood glucose reduction lasting up to 8 h post-oral dosing, whereas standard PLGA NPs achieved only ~3.7 % bioavailability20. In another study, in vivo studies in diabetic rats showed that TPGS-emulsified, PEG-capped PLGA nanoparticles produced about a twofold reduction in serum glucose compared with free insulin, whereas an insulin-loaded PLGA (70/30) formulation produced about a threefold reduction; the hypoglycaemic effect persisted for ~24h21. PLGA nanoparticles are particularly effective because they are biodegradable and biocompatible, making them safe for use in drug delivery systems18. However, challenges such as batch-to-batch variability in polymer synthesis and potential inflammatory responses to degradation products must be considered. These nanoparticles can encapsulate insulin within their core, shielding it from the acidic environment of the stomach and proteolytic enzymes in the intestines, though premature drug release in the stomach remains a limitation for some formulations.
Recent clinical trials of oral insulin candidates provide critical insights into translational potential. In a 12-week Phase II trial, ORMD-0801 was safe, well tolerated, and not associated with weight gain or hypoglycemia. The 8 mg twice-daily dose at bedtime demonstrated a statistically significant reduction in HbA1c compared with placebo3. In contrast, Oral HDV-I, evaluated in a single-blind, placebo-controlled dose-escalation study in type 2 diabetes patients, significantly lowered postprandial plasma glucose and incremental postprandial glucose area under the curve at all tested doses (0.05–0.4 U/kg), with 0.05 U/kg identified as the minimum effective dose. Oral HDV-I was safe and well tolerated22.
Liposomes, which are lipid-based nanoparticles, are another promising delivery system18. Liposomes are composed of phospholipid bilayers that mimic cell membranes, allowing them to fuse with intestinal epithelial cells and deliver insulin directly into the bloodstream. While generally well-tolerated, liposomes may face stability issues in the GIT and could trigger immune responses with repeated administration. Preclinical studies in rats showed that sodium-glycocholate–containing liposomes (SGC-liposomes) produced substantially higher oral bioavailability of insulin — about 11.0% (diabetic rats; ≈8.5% in non-diabetic rats) when referenced to subcutaneous insulin — compared with conventional liposomes, and elicited significant hypoglycemic effects after oral dosing, showcasing improved pharmacokinetics while still falling short of subcutaneous administration23. In another study using Caco‑2 cell monolayers, liposomes containing sodium taurocholate increased transepithelial permeability (log k = –1.95 cm/h vs –2.22 cm/h for insulin solution), and animal studies confirmed a significant hypoglycemic effect correlating with in vitro permeability data24. Additionally, liposomes can be modified with targeting ligands, such as lectins, to enhance their specificity for intestinal cells25, though the potential for off-target binding and lectin-induced gut irritation warrants further investigation.
Liposomal systems, along with other nanocarriers, can be engineered to release insulin in a controlled manner, ensuring targeted delivery26. However, inter-patient variability in intestinal pH may affect their consistency. pH-sensitive polymeric nanoparticles, such as those made from Eudragit or chitosan, can enhance insulin absorption by improving mucosal adhesion and controlled release properties27, though chitosan’s cationic nature may disrupt tight junctions and increase intestinal permeability to harmful substances. When choosing between platforms, key criteria must include biocompatibility (both Eudragit and chitosan are FDA-approved excipients, though chitosan derived from crustacean shells carries allergen concerns), cost and scalability (Eudragit’s synthetic production ensures lower batch variability vs. variable chitosan raw material costs), and long-term stability (Eudragit-coated systems show ≥12‑month shelf stability under Internation Council for Harmonizatio (ICH) conditions, while chitosan formulations often require refrigeration and cross-linking to prevent deacetylation)28. Preclinical in vivo studies provide evidence for their potential efficacy: for instance, insulin-loaded alginate/chitosan nanoparticles administered orally in diabetic rats achieved pharmacological bioavailability of approximately 6.8% at 50 IU/kg and 3.4% at 100 IU/kg doses, significantly higher than the ~1.6% from insulin solution, and reduced basal serum glucose by over 40%, sustaining hypoglycemia for up to 18 h in one study29. Similarly, dextran sulfate/chitosan nanoparticles showed around 5.6% bioavailability at a 50 IU/kg dose and lowered glucose levels by approximately 35%, with effects lasting over 24h30. While both polymers have demonstrated promise in preclinical settings, current clinical trials involving oral insulin platforms have more frequently focused on synthetic polymer-based systems. Notably, ORMD-0801, which uses a protective enteric coating to shield insulin from gastric degradation, has advanced through Phase II, but the Phase III trial did not meet its primary endpoint3. In contrast, chitosan-based oral insulin systems remain largely in preclinical or early Phase I stages.
Despite their therapeutic promise, nanoparticle-based systems also pose potential immunogenicity risks. PEGylation, commonly used to improve the stability and circulation time of nanoparticles, can lead to the development of anti-PEG antibodies, which may reduce treatment efficacy and cause hypersensitivity reactions with repeated exposure31. Synthetic polymers like PLGA may generate degradation byproducts that trigger low-grade inflammatory responses or local immune activation at the intestinal mucosa32. Furthermore, repeated oral exposure to nanoparticulate formulations may lead to the formation of mucosal immune memory, potentially contributing to long-term immune reactivity or antigen sensitization33. Although most preclinical studies report good biocompatibility in short-term experiments, long-term studies assessing chronic exposure to nanocarriers—particularly those involving PEG, polyacrylate derivatives, or thiolated polymers—are limited.
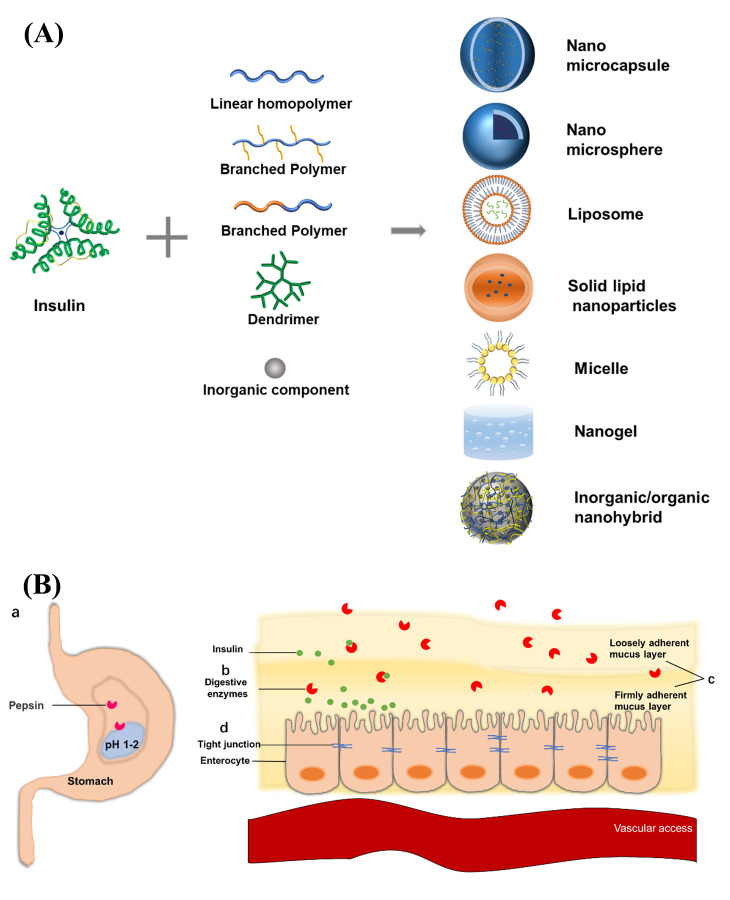
Permeation Enhancers
Permeation enhancers are compounds that increase the permeability of the intestinal epithelium, facilitating insulin absorption35. Bile salts, such as sodium taurocholate, temporarily disrupt epithelial tight junctions, allowing insulin to pass through. Sodium taurocholate works by activating protein kinase C, which phosphorylates tight junction proteins and causes them to contract, creating temporary gaps in the epithelial barrier36. Surfactants like sodium lauryl sulfate enhance membrane fluidity, improving insulin uptake. Sodium lauryl sulfate interacts with the lipid bilayer of epithelial cells, increasing membrane permeability and allowing insulin to diffuse more easily37. However, these enhancers must balance efficacy with safety, as long-term use may cause mucosal irritation, inflammation, or systemic toxicity. For example, bile salt concentrations above 0.3% are known to cause mucosal irritation in nasal applications38 and have detergent-like effects that may damage epithelial membranes. Studies on self-emulsifying systems report that surfactants like sodium taurocholate can reduce systemic toxicity compared to harsher surfactants, but gastrointestinal irritation remains a concern when used chronically39. Preclinical rat studies show that sodium taurocholate and related bile salts can cause transient epithelial villi tip damage and increased lactate dehydrogenase release—though mucosal integrity typically recovers within 1–3 hours40’41. Chronic daily dosing in mice using sodium caprate (a similar fatty acid enhancer) over 30 days did not increase baseline intestinal permeability or histological damage, supporting its safety profile under controlled conditions42. To address this, researchers are exploring safer alternatives, such as chitosan-based permeation enhancers, which have shown similar efficacy with low toxicity and minimal mucosal irritation in preclinical studies6. Preclinical in vivo data demonstrate that chitosan‑coated solid lipid nanoparticles significantly improve oral insulin delivery: oral insulin-loaded chitosan‑coated SLN administered to diabetic rats achieved a relative pharmacological bioavailability of 17%, compared with 8% for uncoated SLN, and produced a pronounced hypoglycemic effect lasting up to 24h, highlighting both efficacy and gastrointestinal safety of chitosan‑based enhancers43. Despite these advances, the included studies consistently report that neither the FDA nor the EMA has approved any oral insulin formulation or permeation enhancer for chronic use44. Regulatory agencies require robust, reproducible efficacy in well-controlled clinical trials, comprehensive long-term safety data, and excipient approval for chronic pharmaceutical use45. Most candidates have only demonstrated efficacy in preclinical or early clinical studies, and long-term safety data—especially in humans—are lacking46. Additionally, concerns about immunogenicity and unintended absorption of antigens remain insufficiently addressed47. As a result, regulatory barriers and the absence of large-scale clinical trial data continue to prevent market approval of oral insulin candidates46.
While mucoadhesive polymers like chitosan and thiolated chitosan provide foundational benefits for oral insulin delivery, their combination with permeation enhancers represents a critical advancement. The integration of sodium taurocholate with chitosan-based nanoparticle systems exemplifies this synergistic approach, simultaneously enhancing mucosal adhesion (through chitosan’s electrostatic interactions or thiolated derivatives’ disulfide bonds) and epithelial permeability6. Notably, this combination has demonstrated improved insulin bioavailability, which is crucial given that oral administration typically suffers from less than 2% bioavailability due to the gastrointestinal environment’s harshness6. Moreover, recent studies indicate that such mucoadhesive-enhanced delivery systems can significantly increase the residence time of insulin in the GIT, allowing for better absorption and overall metabolic control48. Such combinatorial strategies address both the absorption barrier and residence time challenges inherent to oral insulin delivery, offering a more comprehensive solution than mucoadhesion alone.
Protease Inhibitors
Another strategy involves co-administering protease inhibitors with insulin to prevent enzymatic degradation. Aprotinin, a protease inhibitor, has shown promise in protecting insulin from trypsin and chymotrypsin49. Aprotinin works by binding to the active sites of these enzymes, preventing them from breaking down insulin. Aprotinin has been shown to enhance the stability of orally administered insulin by inhibiting proteolytic enzymes that degrade it50. Soybean trypsin inhibitor (SBTI) is another example that has been used in preclinical studies. SBTI inhibits trypsin and chymotrypsin by forming stable complexes with these enzymes, rendering them inactive. However, the use of protease inhibitors may carry potential side effects, including allergic reactions, interference with normal protein digestion, and possible pancreatic feedback mechanisms that could alter enzyme production. Combining enzyme inhibitors with other strategies, such as nanoparticle encapsulation, can further enhance insulin stability and bioavailability51. Nanoparticles co-loaded with insulin and protease inhibitors have been shown to enhance insulin bioavailability by protecting it from enzymatic degradation and improving its absorption, highlighting the potential of this combined approach for oral insulin delivery27. Recent studies developed chitosan/γ-PGA nanoparticles functionalized with a chelator acting as protease inhibitor; in diabetic rats this system achieved an approximately 20% relative oral bioavailability of insulin, accompanied by sustained hypoglycemic responses and confirmed intestinal uptake of insulin up to 4h post-administration, demonstrating both efficacy and bioavailability benefits in vivo52. This synergistic approach not only protects insulin from degradation but also enhances its absorption, making it a promising strategy for oral insulin delivery.
Mucoadhesive Polymers
In addition to these strategies, researchers are exploring the use of mucoadhesive polymers to prolong insulin’s contact with the intestinal mucosa48. Chitosan, a natural polysaccharide derived from chitin, has been widely studied for its mucoadhesive properties. Chitosan adheres to the mucosal layer, increasing residence time and improving bioavailability48. Thiolated polymers, such as thiolated chitosan, form disulfide bonds with mucus, further prolonging contact53. Thiolated chitosan has been reported to enhance drug absorption, including insulin, due to its ability to form disulfide bonds with mucus glycoproteins, thereby improving mucoadhesion and prolonging mucosal contact time54. A controlled in vivo study in rats demonstrated that thiolated chitosan–insulin tablets resulted in a significant hypoglycemic effect lasting 24 h, with a pharmacological efficacy equivalent to ~1.7% of a subcutaneous dose—levels not achieved by unmodified chitosan tablets or insulin solution—highlighting markedly improved residence time and bioavailability attributed to thiolation and mucoadhesion55.
Glucose-Responsive Systems
Emerging technologies, such as glucose-responsive systems, are also being developed to improve oral insulin delivery56. These systems mimic the body’s natural insulin release, improving glycemic control56. For example, orally administered hydrogels that swell in response to high glucose levels in the GIT can release insulin on demand, reducing the risk of hypoglycemia56. In a comprehensive in vivo study using nanoporous stimuli-responsive hydrogel microparticles, diabetic rats demonstrated a 7.45-fold increase in oral insulin bioavailability compared to insulin solution, accompanied by enhanced hypoglycemic effects and no histological toxicity in gastrointestinal tissues55. A study demonstrated that glucose-responsive hydrogels could release insulin in a controlled manner, maintaining blood glucose levels within the normal range for up to 12h57. However, despite over a decade of promising preclinical research, glucose-responsive formulations have not entered clinical trials due to several translational hurdles. Common barriers include slow response kinetics—many platforms undergo hydrogel swelling on the scale of tens of minutes to hours, which fails to match postprandial glucose surges—and inconsistent performance in complex in vivo environments with variable pH and interfering metabolites58. Manufacturing challenges also persist: ensuring reproducible incorporation of glucose-sensitive moieties like glucose oxidase or phenylboronic acid at scale without enzyme denaturation or stability loss remains problematic59. Regulatory concerns additionally arise regarding long-term safety—such as residual H₂O₂ generated by enzyme-based systems, off‑target swelling triggering hypoglycemia, and the absence of rigorous Good Manufacturing Practices -compliant production protocols—all of which must be addressed before human testing can proceed.
Smart insulin delivery systems represent another groundbreaking approach to oral insulin delivery. pH-sensitive systems, such as those using Eudragit polymers, are designed to release insulin in response to the alkaline environment of the intestines. Eudragit L100 and S100, for instance, are methacrylic acid copolymers that dissolve at pH levels above 6.0 and 7.0, respectively60. These polymers protect insulin from degradation in the acidic stomach and ensure targeted release in the small intestine, where absorption is optimal. In a preclinical study using Eudragit L30D-coated PLGA microparticles (analogous to L100/S100 functionality), alloxan-diabetic rats given a single oral dose (25 IU/kg insulin equivalent) exhibited a 62.7 ± 3.8 % reduction in blood glucose at 2 h—compared to 37.3 ± 11 % with uncoated PLGA particles—while maintaining hypoglycemia for up to 24 h, demonstrating markedly enhanced efficacy and pharmacodynamics of pH-triggered delivery61. Eudragit-based nanoparticles are recognized for their potential to enhance insulin bioavailability by protecting the drug from gastric degradation and facilitating targeted intestinal release27.
Glucose-responsive systems take this concept further by mimicking the body’s natural insulin release mechanism. These systems use hydrogels or nanoparticles that respond to changes in glucose levels. For example, glucose oxidase (GOx)-loaded hydrogels can swell in the presence of high glucose concentrations, releasing insulin in a controlled manner58. When glucose levels rise, GOx converts glucose into gluconic acid, lowering the local pH and causing the hydrogel to swell58. This swelling triggers the release of encapsulated insulin, providing a feedback loop that mimics pancreatic beta cell function. Recent studies have shown that GOx-based hydrogels could maintain blood glucose levels within the normal range for up to 10h in diabetic rats, significantly reducing the risk of hypoglycemia62. In preclinical models, orally administered pH-responsive bacterial cellulose‑g‑poly(acrylic acid) hydrogel microparticles encapsulating insulin showed a 7.45‑fold increase in relative oral bioavailability compared to insulin solution, with significantly enhanced hypoglycemic effects and no observed gastrointestinal toxicity in diabetic rats63.
Combining these technologies with advanced nanotechnology has further enhanced their efficacy. For instance, pH-sensitive nanoparticles coated with mucoadhesive polymers can provide dual protection and targeted release. Chitosan-coated PLGA nanoparticles that released insulin in response to both pH and glucose levels have been shown in preclinical models to significantly enhance pharmacological effectiveness: in diabetic rats, orally administered N-trimethyl chitosan–coated PLGA nanoparticles achieved a 2-fold higher relative pharmacological availability and produced stronger hypoglycemic effects compared with uncoated PLGA nanoparticles, underscoring the functional benefits of mucoadhesive, pH-responsive design64.
| Delivery System | Key Formulation | Encapsulation Efficiency / Bioavailability | Efficacy (Glucose Reduction) | Safety Observations | Ref |
|---|---|---|---|---|---|
| PLGA Nanoparticles | pH-sensitive PLGA Hp55 | 6.27% relative bioavailability | Sustained reduction for 8h | Biocompatible; batch variability concerns | (Cui, 2007) |
| PEG-capped, TPGS-emulsified PLGA | 2× hypoglycemic effect vs. free insulin | Effects lasting 24h | Potential inflammatory degradation products | (Malathi, 2015) | |
| Liposomes | Sodium glycocholate-stabilized | 12% bioavailability | Significant glucose reduction | Stability issues in GIT; immune responses | (Zhang, 2014) |
| Sodium taurocholate-modified | Increased transepithelial permeability | Hypoglycemic effect confirmed | Off-target binding risks | (Degim, 2004) | |
| Chitosan Nanoparticles | Alginate/chitosan | 6.8% (50 IU/kg), 3.4% (100 IU/kg) | >40% glucose reduction, 18h | Mucoadhesive; allergen concerns | (Sarmento, 2007) |
| Dextran sulfate/chitosan | 5.6% bioavailability | ~35% glucose reduction, 24h | Long-term stability challenges | (Sarmento, 2007) | |
| Glucose-Responsive Hydrogels | pH-responsive poly(acrylic acid) | 26% relative bioavailability | 52.6% glucose drop | No observed GI toxicity | (Shabir, 2022) |
| Bacterial cellulose/poly(acrylic acid) | 7.45× bioavailability vs. solution | Enhanced hypoglycemic effect | No histological toxicity | (Ahmad, 2016) | |
| Protease Inhibitors | Chitosan/γ-PGA + chelator | ~20% relative bioavailability | Sustained hypoglycemia (4h) | Allergenicity concerns (e.g., aprotinin) | (Su, 2012) |
Regulatory Barriers for Oral Insulin Approval
Regulatory agencies impose stringent requirements for oral peptide drugs—oral insulin must satisfy these before gaining approval. Agencies like the FDA, EMA, and ICH mandate thorough physicochemical and stability characterization, including demonstration of identity, purity, potency, and degradation profiles throughout shelf life65. Manufacturers must provide data confirming well-characterized excipients, validated analytical methods, and maintenance of insulin integrity under storage conditions.
From a clinical pharmacology standpoint, the FDA’s December 2023 draft guidance on peptide drugs requires robust assessment of pharmacokinetics (PK), drug–drug interactions, hepatic impairment, and most notably immunogenicity risk as part of the New Drug Application submission. For instance, early-stage trials must include anti-drug antibody testing to evaluate immunogenicity profiles and monitor potential reductions in efficacy or safety signals.
Additionally, regulatory bodies expect future oral insulin candidates to demonstrate consistent bioavailability and exposure in diverse populations, with clear mitigation strategies for variability linked to gut pH, motility, or microbiota composition—a known challenge for oral peptides66. Agencies also emphasize that reproducible long-term safety data, especially regarding mucosal integrity, immune responses, and chronic gastrointestinal exposure, will be crucial during late-stage, pivotal trials. Without such evidence, approval remains unlikely despite promising preclinical findings.
The Future of Oral Insulin and Its Implications
Oral insulin has the potential to reduce the need for injections20 reducing pain and inconvenience. Patients are more likely to adhere to a non-invasive treatment regimen, leading to better glycemic control and improved long-term health outcomes, although long-term data on adherence and outcomes remain limited. By reducing the risk of complications such as neuropathy and retinopathy, oral insulin may have the potential to enhance the quality of life for millions of people with diabetes.
The widespread adoption of oral insulin may offer economic and social benefits. By improving glycemic control, oral insulin could reduce healthcare costs associated with diabetes complications, including hospitalizations for end-organ damage such as kidney failure, blindness, and amputations, as well as emergency room visits. Additionally, oral formulations are generally considered easier to distribute and administer than injectable insulin, which may improve accessibility in low-resource settings.
Despite its promise, the development of oral insulin faces several challenges. Ensuring the safety and efficacy of new delivery systems requires rigorous clinical trials and long-term studies to assess potential side effects. Regulatory hurdles and scalability for mass production must also be addressed. However, with continued innovation and collaboration, these challenges can be overcome, bringing oral insulin closer to reality.
Conclusion
The development of oral insulin has emerged as a promising area of research in diabetes treatment. By addressing the challenges of enzymatic degradation, poor permeability, and patient-specific variability, innovative strategies such as nanoparticle encapsulation, permeation enhancers, and smart delivery systems have shown potential in advancing oral insulin formulations. If future research continues to yield positive outcomes, oral insulin has the potential to revolutionize diabetes care, offering a more patient-friendly and effective treatment option. As research progresses, the integration of these technologies will be critical in overcoming current limitations and informing the viability of oral insulin as a therapeutic option. Continued innovation and rigorous evaluation will help clarify the role oral insulin may play in future diabetes care.
References
- Greene, Nikula, Poulin, McInally and Reynolds. (2021). Long-Term Nonclinical Pulmonary Safety Assessment of Afrezza, a Novel Insulin Inhalation Powder, 49(2), 334-348. http://www.ncbi.nlm.nih.gov/pubmed/33043802 [↩]
- Brown, Heerspink, Cuthbertson and Wilding. (2021). SGLT2 inhibitors and GLP-1 receptor agonists: established and emerging indications, 398(10296), 262-276. http://www.ncbi.nlm.nih.gov/pubmed/34216571 [↩]
- Eldor, Francis, Fleming, Neutel, Homer, Kidron and Rosenstock. (2023). Oral insulin (ORMD-0801) in type 2 diabetes mellitus: A dose-finding 12-week randomized placebo-controlled study, 25(4), 943-952. http://www.ncbi.nlm.nih.gov/pubmed/36281496 [↩] [↩] [↩]
- Geho, Geho, Lau and Gana. (2009). Hepatic-directed vesicle insulin: a review of formulation development and preclinical evaluation, 3(6), 1451-1459. http://www.ncbi.nlm.nih.gov/pubmed/20144401 [↩]
- Zhu, Chen, Paul, Lu, Wu and Qi. (2021). Oral delivery of proteins and peptides: Challenges, status quo and future perspectives, 11(8), 2416-2448. http://www.ncbi.nlm.nih.gov/pubmed/34522593 [↩] [↩] [↩]
- Seyam, Nordin and Alfatama. (2020). Recent Progress of Chitosan and Chitosan Derivatives-Based Nanoparticles: Pharmaceutical Perspectives of Oral Insulin Delivery, 13(10), http://www.ncbi.nlm.nih.gov/pubmed/33066443 [↩] [↩] [↩] [↩]
- Weiss, Steiner and Philipson. (2000). Insulin Biosynthesis, Secretion, Structure, and Structure-Activity Relationships, http://www.ncbi.nlm.nih.gov/pubmed/25905258, Zhu, Chen, Paul, Lu, Wu and Qi. (2021). Oral delivery of proteins and peptides: Challenges, status quo and future perspectives, 11(8), 2416-2448. http://www.ncbi.nlm.nih.gov/pubmed/34522593 [↩]
- Renukuntla, Vadlapudi, Patel, Boddu and Mitra. (2013). Approaches for enhancing oral bioavailability of peptides and proteins, 447(1-2), 75-93. http://www.ncbi.nlm.nih.gov/pubmed/23428883 [↩]
- Baral and Choi. (2025). Barriers and Strategies for Oral Peptide and Protein Therapeutics Delivery: Update on Clinical Advances, 17(4), http://www.ncbi.nlm.nih.gov/pubmed/40284395 [↩]
- Oyama, Matayoshi, Kanetaka, Nitta, Koide, Minami and Asai. (2025). Enhanced oral insulin delivery with charge-reversible lipid nanoparticles, 750(151420. http://www.ncbi.nlm.nih.gov/pubmed/39892057 [↩]
- Badwan, Remawi, Qinna, Elsayed, Arafat, Melhim, Hijleh and Idkaidek. (2009). Enhancement of oral bioavailability of insulin in humans, 30(1), 74-78. http://www.ncbi.nlm.nih.gov/pubmed/19300394 [↩]
- Zhang, Zhu, Song, Shi and Cao. (2024). Recent advances in oral insulin delivery technologies, 366(221-230. http://www.ncbi.nlm.nih.gov/pubmed/38161033 [↩]
- Abuhelwa, Foster and Upton. (2016). A Quantitative Review and Meta-Models of the Variability and Factors Affecting Oral Drug Absorption-Part I: Gastrointestinal pH, 18(5), 1309-1321. http://www.ncbi.nlm.nih.gov/pubmed/27495120 [↩]
- Zhang, Han, Huang, Jin and Gao. (2021). The influence of the gut microbiota on the bioavailability of oral drugs, 11(7), 1789-1812. http://www.ncbi.nlm.nih.gov/pubmed/34386321 [↩]
- Maher and Brayden. (2021). Formulation strategies to improve the efficacy of intestinal permeation enhancers(), 177(113925. http://www.ncbi.nlm.nih.gov/pubmed/34418495 [↩]
- Cil, Anderson, Yen, Kelleher, Huynh, Seo, Nilsen, Turner and Verkman. (2019). Slowed gastric emptying and improved oral glucose tolerance produced by a nanomolar-potency inhibitor of calcium-activated chloride channel TMEM16A, 33(10), 11247-11257. http://www.ncbi.nlm.nih.gov/pubmed/31299174 [↩]
- Shabir, Mahmood, Zafar, Zaman, Sarfraz and Ijaz. (2022). Novel Black Seed Polysaccharide Extract-g-Poly (Acrylate) pH-Responsive Hydrogel Nanocomposites for Safe Oral Insulin Delivery: Development, In Vitro, In Vivo and Toxicological Evaluation, 15(1), http://www.ncbi.nlm.nih.gov/pubmed/36678691 [↩] [↩]
- Caturano, Nilo, Nilo, Russo, Santonastaso, Galiero, Rinaldi, Monda, Sardu, Marfella and Sasso. (2024). Advances in Nanomedicine for Precision Insulin Delivery, 17(7), http://www.ncbi.nlm.nih.gov/pubmed/39065795 [↩] [↩] [↩] [↩]
- Pardridge. (2005). The blood-brain barrier: bottleneck in brain drug development, 2(1), 3-14. http://www.ncbi.nlm.nih.gov/pubmed/15717053 [↩]
- Cui, Tao, Cun, Zhang and Shi. (2007). Preparation of insulin loaded PLGA-Hp55 nanoparticles for oral delivery, 96(2), 421-427. http://www.ncbi.nlm.nih.gov/pubmed/17051590 [↩] [↩]
- Malathi, Nandhakumar, Pandiyan, Webster and Balasubramanian. (2015). Novel PLGA-based nanoparticles for the oral delivery of insulin, 10(2207-2218. http://www.ncbi.nlm.nih.gov/pubmed/25848248 [↩]
- Geho, Rosenberg, Schwartz, Lau and Gana. (2014). A single-blind, placebo-controlled, dose-ranging trial of oral hepatic-directed vesicle insulin add-on to oral antidiabetic treatment in patients with type 2 diabetes mellitus, 8(3), 551-559. http://www.ncbi.nlm.nih.gov/pubmed/24876619 [↩]
- Zhang, Qi, Lu, He, Li and Wu. (2014). Biotinylated liposomes as potential carriers for the oral delivery of insulin, 10(1), 167-176. http://www.ncbi.nlm.nih.gov/pubmed/23891617 [↩]
- Degim, Unal, Essiz and Abbasoglu. (2004). The effect of various liposome formulations on insulin penetration across Caco-2 cell monolayer, 75(23), 2819-2827. http://www.ncbi.nlm.nih.gov/pubmed/15464833 [↩]
- He, Lu, Qi, Zhu, Chen and Wu. (2019). Adapting liposomes for oral drug delivery, 9(1), 36-48. http://www.ncbi.nlm.nih.gov/pubmed/30766776 [↩]
- Gao, Chan and Farokhzad. (2010). pH-Responsive nanoparticles for drug delivery, 7(6), 1913-1920. http://www.ncbi.nlm.nih.gov/pubmed/20836539 [↩]
- Souto, Souto, Campos, Severino, Pashirova, Zakharova, Silva, Durazzo, Lucarini, Izzo and Santini. (2019). Nanoparticle Delivery Systems in the Treatment of Diabetes Complications, 24(23), http://www.ncbi.nlm.nih.gov/pubmed/31756981 [↩] [↩] [↩]
- Li, Zhang, Zhao and Zhang. (2022). Advances in oral peptide drug nanoparticles for diabetes mellitus treatment, 15(392-408. http://www.ncbi.nlm.nih.gov/pubmed/35386357 [↩]
- Sarmento, Ribeiro, Veiga, Sampaio, Neufeld and Ferreira. (2007). Alginate/chitosan nanoparticles are effective for oral insulin delivery, 24(12), 2198-2206. http://www.ncbi.nlm.nih.gov/pubmed/17577641 [↩]
- Sarmento, Ribeiro, Veiga, Ferreira and Neufeld. (2007). Oral bioavailability of insulin contained in polysaccharide nanoparticles, 8(10), 3054-3060. http://www.ncbi.nlm.nih.gov/pubmed/17877397 [↩]
- Zhang, Sun, Liu and Jiang. (2016). Anti-PEG antibodies in the clinic: Current issues and beyond PEGylation, 244(Pt B), 184-193. http://www.ncbi.nlm.nih.gov/pubmed/27369864 [↩]
- Xu, Kim, Saylor and Koo. (2017). Polymer degradation and drug delivery in PLGA-based drug-polymer applications: A review of experiments and theories, 105(6), 1692-1716. http://www.ncbi.nlm.nih.gov/pubmed/27098357 [↩]
- Lotfipour, Shahi, Farjami, Salatin, Mahmoudian and Dizaj. (2021). Safety and Toxicity Issues of Therapeutically Used Nanoparticles from the Oral Route, 2021(9322282. http://www.ncbi.nlm.nih.gov/pubmed/34746313 [↩]
- Wang, Wang, Ren, Pan, Wang, Shen, Zeng, Cui and Zhao. (2022). Versatile Oral Insulin Delivery Nanosystems: From Materials to Nanostructures, 23(6), http://www.ncbi.nlm.nih.gov/pubmed/35328783 [↩]
- Nicze, Borowka, Dec, Niemiec, Buldak and Okopien. (2024). The Current and Promising Oral Delivery Methods for Protein- and Peptide-Based Drugs, 25(2), http://www.ncbi.nlm.nih.gov/pubmed/38255888 [↩]
- Johansson, Hjertberg, Eirefelt, Tronde and Hultkvist Bengtsson. (2002). Mechanisms for absorption enhancement of inhaled insulin by sodium taurocholate, 17(1-2), 63-71. http://www.ncbi.nlm.nih.gov/pubmed/12356421 [↩]
- Sabra, Kirby, Chouk, Malgorzata and Mohammed. (2024). Buccal Absorption of Biopharmaceutics Classification System III Drugs: Formulation Approaches and Mechanistic Insights, 16(12), http://www.ncbi.nlm.nih.gov/pubmed/39771541 [↩]
- Moghimipour, Ameri and Handali. (2015). Absorption-Enhancing Effects of Bile Salts, 20(8), 14451-14473. http://www.ncbi.nlm.nih.gov/pubmed/26266402 [↩]
- Gao, Wang, Sun, Sun, Sun, Liu and Guan. (2017). Evaluation of the cytotoxicity and intestinal absorption of a self-emulsifying drug delivery system containing sodium taurocholate, 106(212-219. http://www.ncbi.nlm.nih.gov/pubmed/28591563 [↩]
- Legen, Kracun, Salobir and Kerc. (2006). The evaluation of some pharmaceutically acceptable excipients as permeation enhancers for amoxicillin, 308(1-2), 84-89. http://www.ncbi.nlm.nih.gov/pubmed/16330171 [↩]
- McCartney, Gleeson and Brayden. (2016). Safety concerns over the use of intestinal permeation enhancers: A mini-review, 4(2), e1176822. http://www.ncbi.nlm.nih.gov/pubmed/27358756 [↩]
- Fein, Gleeson, Cochran, Lamson, Doerfler, Melamed and Whitehead. (2023). Long-term daily oral administration of intestinal permeation enhancers is safe and effective in mice, 8(1), e10342. http://www.ncbi.nlm.nih.gov/pubmed/36684095 [↩]
- Fonte, Nogueira, Gehm, Ferreira and Sarmento. (2011). Chitosan-coated solid lipid nanoparticles enhance the oral absorption of insulin, 1(4), 299-308. http://www.ncbi.nlm.nih.gov/pubmed/25788364 [↩]
- Ma, Ma, Liu, Zheng and Duan. (2006). In vitro and in vivo evaluation of a novel oral insulin formulation, 27(10), 1382-1388. http://www.ncbi.nlm.nih.gov/pubmed/17007747 [↩]
- 2012). Overcoming poor permeability: translating permeation enhancers for oral peptide delivery, 9(2), e71-e174. http://www.ncbi.nlm.nih.gov/pubmed/24064271 [↩]
- Fonte, Araujo, Reis and Sarmento. (2013). Oral insulin delivery: how far are we?, 7(2), 520-531. http://www.ncbi.nlm.nih.gov/pubmed/23567010 [↩] [↩]
- Maher, Leonard, Jacobsen and Brayden. (2009). Safety and efficacy of sodium caprate in promoting oral drug absorption: from in vitro to the clinic, 61(15), 1427-1449. http://www.ncbi.nlm.nih.gov/pubmed/19800376 [↩]
- Banerjee, Lee and Mitragotri. (2016). Intestinal mucoadhesive devices for oral delivery of insulin, 1(3), 338-346. http://www.ncbi.nlm.nih.gov/pubmed/29313019 [↩] [↩] [↩]
- Ivachtchenko, Ivashchenko, Shkil and Ivashchenko. (2023). Aprotinin-Drug against Respiratory Diseases, 24(13), http://www.ncbi.nlm.nih.gov/pubmed/37446350 [↩]
- Dan, Samanta and Almoazen. (2020). An Update on Pharmaceutical Strategies for Oral Delivery of Therapeutic Peptides and Proteins in Adults and Pediatrics, 7(12), http://www.ncbi.nlm.nih.gov/pubmed/33352795 [↩]
- Zhang, Ting, et al. [2021]. Can nanoparticles and nano-protein interactions bring a bright future for insulin delivery? Acta Pharmaceutica Sinica B, 11[3], 651-667. https://doi.org/10.1016/j.apsb.2020.12.015 [↩]
- Su, Lin, Sonaje, Wey, Yen, Ho, Panda, Chuang, Maiti and Sung. (2012). Protease inhibition and absorption enhancement by functional nanoparticles for effective oral insulin delivery, 33(9), 2801-2811. http://www.ncbi.nlm.nih.gov/pubmed/22243802 [↩]
- Millotti, Laffleur, Perera, Vigl, Pickl, Sinner and Bernkop-Schnurch. (2014). In vivo evaluation of thiolated chitosan tablets for oral insulin delivery, 103(10), 3165-3170. http://www.ncbi.nlm.nih.gov/pubmed/25139279 [↩]
- Federer, Kurpiers and Bernkop-Schnurch. (2021). Thiolated Chitosans: A Multi-talented Class of Polymers for Various Applications, 22(1), 24-56. http://www.ncbi.nlm.nih.gov/pubmed/32567846 [↩]
- Krauland, Guggi and Bernkop-Schnurch. (2004). Oral insulin delivery: the potential of thiolated chitosan-insulin tablets on non-diabetic rats, 95(3), 547-555. http://www.ncbi.nlm.nih.gov/pubmed/15023465 [↩] [↩]
- Wang, Wang, Kahkoska, Buse and Gu. (2021). Developing Insulin Delivery Devices with Glucose Responsiveness, 42(1), 31-44. http://www.ncbi.nlm.nih.gov/pubmed/33250274 [↩] [↩] [↩]
- Mansoor, Kondiah and Choonara. (2021). Advanced Hydrogels for the Controlled Delivery of Insulin, 13(12), http://www.ncbi.nlm.nih.gov/pubmed/34959394 [↩]
- Pal, Rakshit, Saha and Jinagal. (2025). Glucose-Responsive Materials for Smart Insulin Delivery: From Protein-Based to Protein-Free Design, 5(2), 239-252. http://www.ncbi.nlm.nih.gov/pubmed/40093833 [↩] [↩] [↩]
- Mohanty, Ravikumar and Peppas. (2022). Recent advances in glucose-responsive insulin delivery systems: novel hydrogels and future applications, 9(rbac056. http://www.ncbi.nlm.nih.gov/pubmed/36072265 [↩]
- Bando and McGinity. (2006). Relationship between drug dissolution and leaching of plasticizer for pellets coated with an aqueous Eudragit S100:L100 dispersion, 323(1-2), 11-17. http://www.ncbi.nlm.nih.gov/pubmed/16815652 [↩]
- Naha, Kanchan, Manna and Panda. (2008). Improved bioavailability of orally delivered insulin using Eudragit-L30D coated PLGA microparticles, 25(4), 248-256. http://www.ncbi.nlm.nih.gov/pubmed/18465311 [↩]
- Yu, Zhang, Bomba and Gu. (2016). Stimuli-Responsive Delivery of Therapeutics for Diabetes Treatment, 1(3), 323-337. http://www.ncbi.nlm.nih.gov/pubmed/29147685 [↩]
- Ahmad, Mohd Amin, Ismail and Buang. (2016). Enhancement of oral insulin bioavailability: in vitro and in vivo assessment of nanoporous stimuli-responsive hydrogel microparticles, 13(5), 621-632. http://www.ncbi.nlm.nih.gov/pubmed/26943455 [↩]
- Sheng, Han, Qin, Ru, Li, Wu, Cui, Yang, He and Wang. (2015). N-trimethyl chitosan chloride-coated PLGA nanoparticles overcoming multiple barriers to oral insulin absorption, 7(28), 15430-15441. http://www.ncbi.nlm.nih.gov/pubmed/26111015 [↩]
- Elsayed, Kuhl and Imhof. (2025). Regulatory Guidelines for the Analysis of Therapeutic Peptides and Proteins, 31(3), e70001. http://www.ncbi.nlm.nih.gov/pubmed/39921384 [↩]
- Peng, Wang, Chen, Peng, Wang, Chen, Kaplan and Wang. (2023). Challenges and opportunities in delivering oral peptides and proteins, 20(10), 1349-1369. http://www.ncbi.nlm.nih.gov/pubmed/37450427 [↩]