Abstract
Lipoprotein A (Lp(a)) is a significant lipoprotein that lives in the human body. Lp(a) has been recognized as one of the main risk factors for cardiovascular diseases. It has been learned that Lp(a) is also an independent risk factor for heart disease. In addition to containing ApoB-100 and Apo(a), Lp(a) is unique as compared to other lipoproteins. It is able to bind to LDL receptors and promote the formation of clots and inflammatory responses along the vascular wall. As Lp(a) levels increase, there is a higher risk of developing heart disease and other cardiovascular issues. The focus of this review is uncovering how Lp(a) can affect the cardiovascular system and how it is possible to lower Lp(a) through lifestyle changes and/or drugs. To fully comprehend this analysis, it is essential to realize the strong relationship between Lp(a) and cardiovascular disease.
Keywords: Lipoprotein A (Lp(a)), Cardiovascular Disease (CVD), Wound Healing, Endothelial Cells, Therapeutic Development, Lifestyle Factors, Inflammation
Introduction
Lipids perform physiological and biochemical functions for humans such as acting as stores of energy, providing structural components of cell membranes, signaling molecules, and acting as a precursor to hormones. Having too much or under-regulated storage of these compounds results in many negative pathological conditions. When there is excessive lipid buildup, cell structure and function gets negatively impacted. There are disruptions in cellular signalling pathways which can cause apoptosis (programmed cell death) of damaged cells and interference with normal functioning organelles within those cells, leading to the spread of dysfunction.
Elevated levels of lipids are positively associated with cardiovascular disease (CVD) and mortality. This is due to the fact that when there are too many lipids in the bloodstream, the blood cannot process them resulting in lipid buildup in blood vessel walls. With these high levels, an individual has a risk of getting all types of CVD. It is important to watch over and monitor lipid levels in the body and the amount of lipids consumed to try to lower the risk of getting CVD.
One function of lipoproteins is to transport lipids through the bloodstream. Lipoproteins produce hormones and aid in digestion. One of the most well-known lipoproteins is cholesterol. It is produced in the liver. The human body naturally produces cholesterol as needed, however, blood cholesterol is influenced more by saturated fat intake and overall diet pattern rather than dietary cholesterol alone.
A number of studies support that hypercholesterolomic effects are strongly controlled by reducing the amount of saturated fat consumed, rather than dietary cholesterol itself. For example, red meat such as beef and pork is viewed as a risk factor for developing cardiovascular disease (CVD) since it has high levels of saturated fat. Hu et al. (2024) examined the relationship between red meat and the human body1. This method utilizes genetic variants associated with meat intake as indicators for dietary patterns combining UK Biobank meat-intake GWAS data with cardiovascular GWAS data sets. The results showed no significant association between meat intake and many cardiovascular events, suggesting that previous links between red meat and CVD may be influenced by confounding factors instead of direct causal effect. Eggs are also viewed as a concern for CVD because they contain dietary cholesterol. Drouin-Chartier et al. (2020) evaluated this relationship using Nurses’ Health Study, Nurses’ Health Study II, and Health Professional Follow-Up Study with 32 years of follow ups2. Participants described the amount of times they ate eggs through a questionnaire. This data demonstrated that eating one egg per day will not lead to a high CVD risk. However, when replacing eggs with red meats, eggs proved to be a much more balanced protein source.
In a study done by Clinicaltrials.gov, a randomized feeding trial was created to understand independent effects of dietary cholesterol and saturated fats (NCT05267522)3. Participants ate three different diets. LDL cholesterol was significantly lower when participants under the low saturated fat diet (having 600mg/day cholesterol and 6% energy from fats) ate two eggs each day, however, for participants with a high saturated fat diet (600 mg/day cholesterol and 12% energy from saturated fats), LDL remained constant. Consuming saturated fat was positively associated with LDL cholesterol, surprisingly, dietary cholesterol was not as strongly correlated. This demonstrates that saturated fat causes LDL to rise. These findings relate to epidemiologic patterns where dietary cholesterol could possibly relate to cardiovascular risk, but this may reflect broader dietary patterns that happen with cholesterol-filled foods. Zhong et al. (2019) used data from 29, 615 US adults and found that adding 300mg/day of dietary cholesterol was associated with higher risk for CVD event4. These studies support that hypercholesterolomic effects are strongly controlled by reducing the amount of saturated fat consumed, rather than dietary cholesterol on its own.
When an individual has excessive dietary intake, it can lead to high lipid levels. This could possibly result in developing hyperlipidemia and other vascular conditions. The probability of getting CVD while having hyperlipidemia increases dramatically with roughly 16.5% of hyperlipidemia patients getting CVD. Though hyperlipidemia itself may not seem as dangerous as some other types of vascular complications, it can lead to the buildup of plaque in the arteries. This poses many concerns because blood flow becomes very restricted. When this happens, there is additional strain on the heart and even the rest of the vascular system, showing how hyperlipidemia ends up affecting the entire body. This condition increases the chance of having heart attacks, strokes, and even peripheral artery disease.
Lipoprotein Measurements
| LDL | VLDL | HDL | Triglycerides | Total Cholesterol | |
| Healthy Range | <100mg/dL | <30mg/dL | >40mg/dL | <150 mg/dL | <200mg/dL |
| Pathological Range | 160-189 mg/dL | >39mg/dL | <40mg/dL | >150 mg/dl | >200mg/dL |
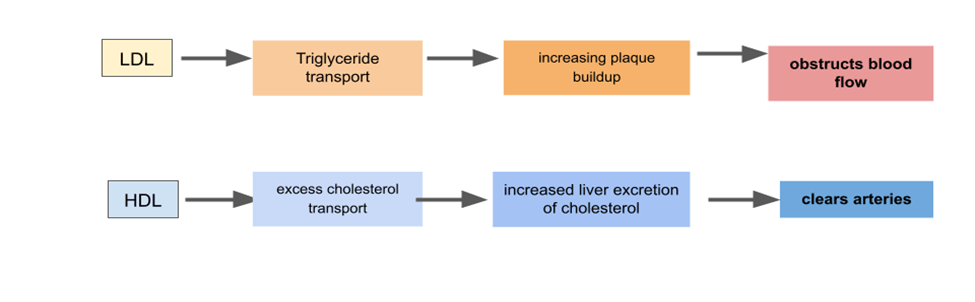
There are three types of cholesterol, one “good” cholesterol, and two “bad” cholesterols. First, High-density Lipoproteins (HDL). HDL’s job as being the “good” cholesterol is to transport any excess cholesterol to the liver for excretion, helping with clearing the arteries. Conversely, very low-density lipoproteins (VLDL) transport triglycerides through-out the human body. However, VLDL is considered bad because it is a factor behind the buildup of plaque and the obstruction of blood flow. When increasing the amount of low-density lipoproteins (LDL), there is an increased risk for plaque forming in someone’s arteries as shown by Table 1 and this also contributes to the formation of plaque in the arteries. The figure below is an illustration of lipid transport and how it relates to plaque building on the artery walls as well as an illustration of the different pathophysiologic effects of VLDL, HDL and LDL, as shown in Figure 1.
It is important to have a healthy balance of lipids (cholesterol and fats in one’s bloodstream). These are the target ranges:
Total Cholesterol < 200 mg/dL;
Low Density Lipid (LDL) < 100 mg/dL; and
Triglycerides < 150 mg/dL.
Routine screening is used to ensure healthy levels of lipids. Some ways of accomplishing this is by monitoring blood pressure and lipid panels to identify potential risks for CVD.
Lipoproten A
There are two main types of lipoproteins that everyone knows about (low-density lipoproteins – LDL & high-density lipoproteins – HDL), however there is another type of lipoprotein called Lipoprotein a (Lp a). Lp a is very similar in composition to LDL except it also contains a protein. While LDL and Triglycerides can be lowered with diet and exercise, Lp a’s level is largely influenced by genetics and cannot be easily changed by making lifestyle adjustments. High Lp a has been shown to increase one’s risk of having a cardiovascular event, including: Atherosclerosis, Heart Attack, Stroke, Aortic Valve Stenosis. Because of its strong genetic influence and lack of responsiveness to traditional ways of lowering LDL, Lp a is being recognized as an independent risk factor for Cardiovascular Disease. Although the incidence of Cardiovascular Disease is increasing, it is still often under diagnosed. Research is currently underway to develop therapeutic agents that will reduce Lp a levels.
Lp a is involved in the formation of atherosclerotic plaques and has been identified as an independent risk factor for Cardiovascular Disease. Lp a has multiple functions depending on the location where it is found, including; promotion of wound healing for endothelial cells and promotion of plaque development. Based on the association between Lp a and Cardiovascular Disease, screening tests and therapeutic options should be considered to avoid individuals from achieving high levels of Lp a. This review article addresses the pathophysiology behind increased levels of Lp a and discusses current therapeutic approaches to managing disease using drugs, and other strategies to reduce the likelihood of a variety of Cardiovascular Disease events.
Lp(a) Measurement
Lp(a) can be expressed in terms of mass concentration (mg/dL), or molar concentration (nm/L). The two measurements are not equivalent due to differences in apo(a) size among individuals. Several professional organizations define an Lp(a) level of 50 mg/dl (125 nm/L) as a positive test. Studies based on data from UK Biobank demonstrated that cardiovascular events increase as Lp(a) increases5. It is generally accepted that at least one adult measurement should be obtained since adult Lp(a) levels do not appear to vary significantly during the individual’s lifetime. Testing Lp(a) multiple times is only recommended for people with a high risk of increased Lp(a) or known CVD in the family.
Methods
To conduct this research, a variety of electronic databases were used including PubMed, Google Scholar, etc. The topics covered included lipids, CVD, and Lp(a). Articles spanned from years 1990 to 2025. Some search terms were (“lipoprotein(a)” OR “Lp(a)”) and (“cardiovascular disease” OR “lipids” OR “cholesterol” OR “treatment”). These were used to find information on Lp(a): its physiology; the relationship between Lp(a) and CVD; and treatment options for dangerous Lp(a) levels. Articles were selected for inclusion in this study based on their inclusion in a peer reviewed publication in English. All articles had to provide direct evidence regarding Lp(a).
All articles included were written in English and provided important information on Lp(a). Articles were not included if it wasn’t from a peer-reviewed source.
The initial database searches produced a wide range of results (about 100 articles were found). Articles were eliminated if the abstracts weren’t following the exclusion criteria (about 50 were eliminated). The remaining 50 articles cycled through a full read-through and review. Only 29 articles were suitable for this literature review.
Key findings were recorded, including study design, population, major outcomes, and clinical relevance. The review was structured to describe lipids, Lp(a), demographic and risk factors, Lp(a)’s function in health, and current emerging treatment options for elevated Lp(a).
Demographics and Risk Factors
CVD has grown to become the biggest threat to human mortality in the United States, accounting for roughly 30% of total deaths each year. CVD affects 48.6% of the adult population over the age of 20, this means roughly 170 million people within the U.S. CVD is a leading factor for various diseases and disorders within the heart and bloodstream. Many of these events are driven by atherosclerosis which prompts plaque buildup in the arteries.
A heart attack (scientifically known as myocardial infarction) is a common CVD event. This happens when the blood vessels in the heart are blocked by blood clots, closing up the arteries and preventing oxygen from going through. This also hinders the delivery of nutrients to the heart. Similarly, a stroke, in particular an ischemic stroke, results from a blockage in the blood vessels that supply the brain, leading to potential brain damage or death. Heart failure is another major CVD event. It is where the heart is unable to pump enough blood to allow proper functions for organs. Heart attacks are sudden blockage of bloodflow, while heart failure is more gradual.
Hypertension, also called high blood pressure, is another contributor to CVD. Hypertension is caused by an increased force of blood against artery walls, which is known to damage the endothelial lining of blood vessels, frequently leading to coronary artery disease and other issues. Over the span of a person’s life, blood pressure is expected to increase. However, when severe sudden spikes are spotted, they can be quite dangerous and consulting a medical professional is recommended.
There are many symptoms that demonstrate potential CVD, including chest pain, nausea, dizziness, and cold sweats. Luckily, around 90% of vascular events can be mitigated or prevented by taking part in healthy habits and lifestyles. Poor diet, lack of exercise, and smoking are a few examples of risk factors for obtaining CVD, and some factors thata person is not able to control (i.e. biological factors).
Lp(a), like LDL cholesterol, is a lipoprotein and is therefore usually located in tissues (healing areas) in the liver and arterial walls. The higher your Lp(a) level, the greater your risk of experiencing a vascular event. Despite what is known about how high levels of Lp(a) increase the likelihood of a vascular event, elevated Lp(a) remains a significantly underdiagnosed and undertreated condition worldwide. Elevated Lp(a) is thought to occur in approximately 20-30% of the global population, however it is rarely discussed as being one of the greatest concerns. Due to the fact that Lp(a) is genetically based and does not respond well to most of the traditionally recommended ways to modify lifestyle habits, Lp(a) is an important, although often overlooked, risk factor for heart disease.
Lipoprotein(a) Function in Health and Disease
Lp(a), has many roles throughout the body, and there are many ways Lp(a) contributes to the risk of cardiovascular events. An important function of Lp(a) is the manner in which it deposits cholesterol into artery walls, forming plaques that may hinder blood flow to major organs, such as the heart and liver. Intact and fragment types of these cholesterol deposits cause an increased risk of vascular complications which can later result in heart attacks, strokes, or the narrowing of arteries.
The structural components of Lp(a) include apo(a) and apolipoprotein B (apoB). An increase in apo(B) is associated with an increase in the risk for heart disease. Apo(B) is primarily responsible for LDL cholesterol metabolism and the formation of plaque in common pathologies. Lp(a) has a unique structure. This is attributed to the inclusion of apo(a) which increases the inflammatory and prothrombotic properties of Lp(a). Simantris et al. (2023) noticed that apo(a) contains oxidized phospholipids (OxPLs), stimulating endothelial dysfunction and activating immune cells including macrophages, accelerating plaque progression6. Hancock et al. (2003) found that apo(a) is structurally similar to plasminogen and interestingly can interfere with the plasminogen activator, reducing someone’s ability to break down clots, promoting thrombosis7. These combined properties help demonstrate how elevated Lp(a) increases cardiovascular risk.
There are certain health conditions and some demographic characteristics that can be associated with increased levels of Lp(a). These include familial hypercholesterolemia (an excessive amount of cholesterol), and chronic kidney disease; both of these are conditions which increase an individual’s cardiovascular risk as they have been associated with increased Lp(a) levels. Race/ethnicity has been shown by research to also play a role in how individuals’ Lp(a) levels will impact them in terms of their health status. For example, populations from South Asia and Africa who have no known medical issues are at a much greater likelihood for having elevated Lp(a) levels than are Caucasian populations. This variation is believed to be due to genetic variation and specifically is believed to be related to the number of copies of the kringle IV type 2 repeat contained within the LPA gene, and the existence of single nucleotide polymorphisms (SNPs) which control the amount of Lp(a) that is synthesized. The LPA gene controls the number of kringle IV type 2 repeats within the protein apo(a); it is this number of repeats which will determine the size of apo(a) isoforms.
In their study Brunner et al. (1996) discovered that when there was less of the kringle IV type 2 repeat present on the apo(a) isoform, then it would be produced and secreted into circulation at a faster rate, thus the amount of Lp(a) present in circulation would be greater8. Larger isoforms are produced and secreted more slowly and in lower amounts. Boomsma et al. (1993) realized that because of repeat-based size variation and other LPA variants, Lp(a) levels are hereditable, with genetic factors accounting for variability among most people9.
Interestingly, Lp(a) is not necessary for human survival, as many people have little to no detectable signs of Lp(a) in their body, leaving minimal consequences. Though its absence doesn’t pose a risk, when Lp(a) concentrations rise above 50 mg/dL, there are major risks including inflammation, thrombosis, and atherosclerosis increases substantially. The mg/dL measurement is generally used clinically to decide if you need to do something about it or just watch it. Because Lp(a) increases the potential for heart disease by promoting plaque formation and increasing the likelihood of blood clots, elevated levels are indicative of increased risk for those individuals who have LDL cholesterol under control.
Lp(a) and Calcific Aortic Valve Stenosis
In addition to its contribution to cardiovascular disease through plaque formation and blood clotting, elevated Lp(a) has been associated with calcific aortic valve stenosis (CAVS). As such, thanassoulis et al. (2013) found that genetic variation at the LPA locus determined the degree of aortic valve calcification and that long-term exposure to high levels of Lp(a) had an effect on this condition10.
Since Lp(a) carries OxPLs in the plasma, both Lp(a) and OxPLs have been detected in diseased aortic valve tissue. OxPL linked to Lp(a) is important for calcific remodeling, giving a pathway for leaflet thickening and stiffening.
Elevated Lp(a) and OxPLs have been linked to presence of CAVS and faster disease progression. The study by Capoulade et al. (2015) as part of the ASTRONOMER substudy determined that subjects within the highest tertile of Lp(a) showed significantly faster hemodynamic progression of their aortic stenosis and were at a greater risk of needing to undergo either valve replacement or cardiac death11. Arsenault et al. (2024) has also confirmed that individuals with elevated Lp(a) levels will show a significantly faster echocardiographic progression of their stenosis severity12.
High Lp(a) may justify echocardiographic monitoring in certain people, particularly those with CAVS. Recognition of Lp(a) as a driver for CAVS provides rational for future trials.
Targeting Lipoprotein(a) for Diagnostic Criteria and Therapeutics
The American Heart Association recommends testing for Lp(a) in all adults less than 65 years of age with a history of acute myocardial infarction, hyperlipidemia or known familial history of premature cardiovascular disease. The sooner high levels are identified, the better chance that future CVD risk can be intervened and prevented.
There are no medications at the moment which can completely eliminate Lp(a) from the bloodstream. However, there are several therapeutic options available to help lower Lp(a). PCSK9 inhibitors meet this requirement. In addition to lowering LDL-C, these inhibitors are able to lower Lp(a) by 20-30% through reducing PCSK9 activity. Some other kinds of medicines such as niacin, estrogens, and other lipid-lowering drugs have seemed to decrease Lp(a) levels.
Niacin was previously utilized as a lipid lowering agent. The addition of Niacin to treatment plans improved both LDL-C and triglycerides, while also improving HDL-C. With further research, Niacin has been observed reducing Lp(a) levels in some patients. However, with current statin patients, the biomarker changes haven’t decreased cardiovascular events. In the AIM-HIGH trial, Boden et al. (2011) gave 3414 patients with known CVD, who take simvastatin, 1500-2000 mg/day of niacin or placebo13. After three years, there was no difference in cardiovascular events even though LDL and triglycerides lowered and HDL increased. In the HPS2-THRIVE trial, Haynes et al. found that the addition of niacin to a statin lowered LDL while also lowering Lp(a), however, it did raise safety concerns due to a higher risk of myopathy and diabetes related effects14. Although niacin can lower Lp(a), these large clinical trials have not shown any clear reduction in cardiovascular events, suggesting it has a limited role in routine treatment.
Estrogen therapies have lowered Lp(a) levels likely through hepatic effects and mediation of lipoprotein metabolism. In a trial with postmenopausal women with venous thrombosis (formation of blood clots), Gregersen et al. (2019) found hormonal replacement therapy lowered Lp(a) within three months15. The relationship of this reduction in Lp(a) is directly inverse to estradiol indicating that there is a link of lowering Lp(a) through estrogen; however, the use of estrogen is contraindicated as an agent to reduce Lp(a), since the risk of thrombosis is elevated by exposure to estrogen. Abou-Ismail et al. (2020) provided evidence that showed estrogen shifting hemostasis which increased clotting and arterial thrombotic events16. Estrogen is effective in decreasing Lp(a) levels, however, it is not used often since thrombosis is a possible result.
An emerging therapy is Lipoprotein apheresis (LA) which eliminates apoB-containing lipoproteins. This includes LDL cholesterol and Lp(a). LA is an effective strategy for patients with severe dyslipidemia or extremely high levels of Lp(a), and is recommended for those with progressive CVD even with the use of medical therapies. Besides lowering lipid levels, LA can reduce blood viscosity, improve kidney function, and enhance microvascular myocardial perfusion.Leebmann et al. (2013) found that LA produces immediate reductions in circulating atherogenic particles, and as a single treatment can lower Lp(a) by up to 60-75%17. Although levels rebound between sessions, regular treatment (1-2 times per week) creates sustained time-averaged low Lp(a). Clinical outcome data suggests that cardiovascular events are reduced in selected high-risk patients. The Pro(a)LiFe study evaluated 170 patients with high Lp(a) and CVD and found that Lp(a) lowered 70% immediately after the sessions. There was also a reduction in cardiovascular events with the mean annual major adverse coronary events (MACE) decline from 0.41 to 0.09. Jaeger et al. (2009) performed a study of 120 patients with coronary artery disease (CAD) and extremely high Lp(a), and found that initiation of LA reduced mean annual MACE from 1.056 to 0.14418. Schumann et al. (2024) followed patients for 7 years and found LA reduced median Lp(a) by 67% (95.0 to 35.1 mg/dL) and MACE rates dropped from 0.34 to 0.00619. Klingel et al. (2025) conducted a 12 year follow up and demonstrated that regular LA was associated with sustained reduction of cardiovascular events20. The consistency of event reduction supports that LA is currently the only approved intervention that reliably and sustainably reduces Lp(a). The limitations of this method include the long duration it takes to do each session (2 to 4 hours); the number of sessions done per week (1 to 2); and the cost associated with doing each session ($2000-$4000).
Familial hypercholesterolemia
Familial Hypercholesterolemia (FH) is a well-known inherited cardiac disease characterized by elevated levels of LDL cholesterol throughout an individual’s lifetime and an increased risk of developing CVD. Goldberg and Gidding (2016) realized that FH affects 1 in 200-300 people around the world21.
FH does not represent a uniform-risk population, but CVD risk is strongly modified by additional genetic and lifestyle factors. Elevated Lp(a) is also considered to be a significant risk factor for developing CVD. Skoumas et al. (2025) conducted a study on adults who had familial dyslipidemia for ten years and concluded that individuals who were diagnosed with FH were at twice the risk for developing cardiovascular events than those with familial combined hypercholesterolemia22.Lp(a) acts as a prognostic modifier in FH, increasing cardiovascular risk even when LDL is treated.
In addition, Lp(a) acts as a prognostic modifier in FH; therefore, if your LDL is being managed, then the presence of high Lp(a) will increase your cardiovascular risk. Goldberg and Gidding also indicated that if LDL cholesterol and Lp(a) remain high after maximum medical therapy, LA should be considered21.
Lifestyle Modifications
Lp(a) levels are dependent on genetics and may not be affected greatly by a healthy diet or exercise. However, an active lifestyle still has great benefits. withIt is important to participate in cardiorespiratory fitness (CRF), a healthy heart diet, not smoking, and sufficient sleep to reduce future CVD risk in individuals with high Lp(a). A study of 750,302 US veterans by Kokkinos et al. (2022) found that increasing CRF resulted in a 23% decrease in all-cause mortality risk23. The lowest mortality risk in this study was achieved at a level of 14 METS and participants in the least fit category were at four times greater risk of death than participants in the extremely fit category. Improved fitness results in a decreased cardiovascular risk although Lp(a) levels remain present. A second study showed that excessive sitting during the day has been associated with increased mortality risk. Ekelund et al. (2016) demonstrated that replacing 60-75 minutes of sitting per day with either exercise or other forms of physical activity can eliminate the increased mortality risk associated with prolonged sitting24. Both studies support the concept of decreasing mortality risk through increased physical activity.
The studies by Kokkinos et al. and Ekelund et al. provided evidence supporting the fact that highly active individuals have a reduced mortality risk when they combine statin therapy with their physical activity routine. For example, Kokkinos et al. (2020) found that patients that used statins in combination with regular exercise had a 60-70% reduction in mortality risk as compared to sedentary statin users25. Highly fit people had a 60-70% reduction in mortality risk as compared to sedentary statin users. Similarly, Ekelund et al. reported that individuals who engaged in frequent physical activity had a 12-59% lower mortality rate compared to those who have a sedentary lifestyle24.
RNA-based therapies
RNA-based treatments are up and coming approaches to reduce Lp(a) levels directly since these medicines target the Lp(a) gene pathway that produces apo(a). Olpasiran is an siRNA (small-interfering RNA) which reduces the synthesis of apo(a) by degrading apo(a)-mRNA. This ends up reducing production in the liver. A study conducted by O’Donoghue et al. (2022) studied 281 patients with CVD and Lp(a) levels >150 nmol/L and found that certain amounts of olpasiran caused Lp(a) to reduce drastically; it lowered 70.5% with a 10 mg dose, 97.4% with a 75 mg dose, and 101.1% with a 225 mg dose as compared to a placebo26‘27. Side effects were similar among all treated patients and included local injection site reactions. Though olpasiran can lower Lp(a) extremely, it is not yet recommended for clinical practices because more trials need to be done to determine if cardiovascular events will be reduced.
Another emerging treatment for lowering Lp(a) is antisense oligonucleotide (ASO), which targets the Lp(a) gene in an attempt to prevent production of apo(a) and subsequently Lp(a). Pelacarsen is a second-generation, hepatocyte-targeted ASO designed to bind to apo(a)-mRNA in the nucleus. The ASO binds to the apo(a)-mRNA and promotes degradation via RNase H-mediated cleavage; thus preventing the production of Lp(a) in the liver. Tsimikas et al. (2020) studied 286 patients with CVD and >60mg/dL Lp(a), finding that pelacarsen greatly reduced Lp(a) for dose-dependent patients over six months28‘29. It was also seen that pelacarsen lowered Lp(a) by 35% with 20mg dose every 4 weeks, 56% with 40mg does every 4 weeks, 72% with 60mg dose every 4 weeks, and 80% with 20mg dose every week, compared to placebos. There were side effects including the site of injection, headaches, and myalgia. This therapy is not yet recommended as well because there are ongoing phase 3 outcome trials to determine if this helps lower cardiovascular event risk.
RNA-based therapies are continually proving itself to the medical world as it reduces Lp(a) more than PCSK9 inhibitors. These therapies may become a more common medicine when confronting high Lp(a).
Conclusion
Lipoproteins play helpful roles in human biology. Some vital lipoproteins to take note of include LDL, VLDL, and HDL. Lipoprotein(a) (Lp(a)) is a lesser known but equally or possibly more dangerous lipoprotein. It does aid the body in different ways; Lp(a) repairs tissue and assists in vascular remodeling. However, as an independent risk factor for CVD, this lipoprotein can lead to many complications. Adults should have their Lp(a) tested at least one time in adult years to establish a baseline level. Lp(a) generally doesn’t change over the course of someone’s lifetime. Repeated testing is suggested for those with known CVD, familial hypercholesterolemia, or other high-risk conditions.
High Lp(a) levels (>50mg/dL) can have roughly 30-50% increase in cardiovascular event risk, proving how dangerous unideal levels can be. Lp(a) is only one cause of cardiovascular risk. This risk is also determined by LDL cholesterol, blood pressure, and diabetes to name a few.
The global population needs to learn the importance of making conscious decisions when it comes to health. This is vital to prevent CVD and other heart conditions. Predictably, forming habits that provide many benefits to the human body minimizes the risk of developing other, potentially life-threatening, issues. Some habits to form are: reducing stress levels with meditation and music, staying active with cardio, and eating a heart-healthy diet with foods like beans and dairy. These practices may not impact Lp(a) levels (as it is determined mostly by genetics), however, it allows people to prevent other complications from arising.Though it is difficult to lower naturally, there are medications and therapies that can help. Procedures such as PCSK9 and Lipoprotein Apheresis can reduce Lp(a) levels greatly. As research advances, early identification of Lp(a) is important in improving cardiovascular risk.
References
- B. Hu, X. He, H. Sun, Y. Hu, F. Li, Y. Sun, J. Sun, L. Feng. Red and processed meat intake and risk of cardiovascular disease: a two-sample mendelian randomization study. Clinical Nutrition ESPEN. Vol. 60, pg. 289–297, 2024 https://doi.org/10.1016/j.clnesp.2024.02.014 [↩]
- J.-P. Drouin-Chartier, S. Chen, Y. Li, A. L. Schwab, M. J. Stampfer, F. M. Sacks, B. Rosner, W. C. Willett, F. B. Hu, S. N. Bhupathiraju. Egg consumption and risk of cardiovascular disease: three large prospective us cohort studies, systematic review, and updated meta-analysis. BMJ. pg. m513, 2020 https://doi.org/10.1136/bmj.m513 [↩]
- P. J. Buckley. Independent Effects of High-cholesterol (High-egg) and High-saturated Fat Diets on LDL-cholesterol. https://clinicaltrials.gov/study/NCT05267522 2023 [↩]
- Associations of dietary cholesterol or egg consumption with incident cardiovascular disease and mortality | dyslipidemia | jama | jama network. https://jamanetwork.com/journals/jama/fullarticle/2728487 [↩]
- J. C. Hopewell, R. Clarke, H. Watkins. Lp(a) (lipoprotein[a]), an exemplar for precision medicine. Arteriosclerosis, Thrombosis, and Vascular Biology. Vol. 41, pg. 475–477, 2021 https://doi.org/10.1161/ATVBAHA.120.315549 [↩]
- S. Simantiris, A. S. Antonopoulos, C. Papastamos, G. Benetos, N. Koumallos, K. Tsioufis, D. Tousoulis. Lipoprotein(a) and inflammation- pathophysiological links and clinical implications for cardiovascular disease. Journal of Clinical Lipidology. Vol. 17, pg. 55–63, 2023 https://doi.org/10.1016/j.jacl.2022.10.004 [↩]
- M. A. Hancock, M. B. Boffa, S. M. Marcovina, M. E. Nesheim, M. L. Koschinsky. Inhibition of plasminogen activation by lipoprotein(a): critical domains in apolipoprotein(a) and mechanism of inhibition on fibrin and degraded fibrin surfaces*. Journal of Biological Chemistry. Vol. 278, pg. 23260–23269, 2003 https://doi.org/10.1074/jbc.M302780200 [↩]
- C. Brunner, E.-M. Lobentanz, A. Pethö-Schramm, A. Ernst, C. Kang, H. Dieplinger, H.-J. Müller, G. Utermann. The number of identical kringle iv repeats in apolipoprotein(a) affects its processing and secretion by hepg2 cells *. Journal of Biological Chemistry. Vol. 271, pg. 32403–32410, 1996 https://doi.org/10.1074/jbc.271.50.32403 [↩]
- D. I. Boomsma, A. Kaptein, H. J. M. Kempen, J. A. Gevers Leuven, H. M. G. Princen. Lipoprotein(a): relation to other risk factors and genetic heritability. results from a dutch parent-twin study. Atherosclerosis. Vol. 99, pg. 23–33, 1993 https://doi.org/10.1016/0021-9150(93)90047-X [↩]
- G. Thanassoulis, C. Y. Campbell, D. S. Owens, J. G. Smith, A. V. Smith, G. M. Peloso, K. F. Kerr, S. Pechlivanis, M. J. Budoff, T. B. Harris, R. Malhotra, K. D. O’Brien, P. R. Kamstrup, B. G. Nordestgaard, A. Tybjaerg-Hansen, M. A. Allison, T. Aspelund, M. H. Criqui, S. R. Heckbert, S.-J. Hwang, Y. Liu, M. Sjogren, J. Van Der Pals, H. Kälsch, T. W. Mühleisen, M. M. Nöthen, L. A. Cupples, M. Caslake, E. Di Angelantonio, J. Danesh, J. I. Rotter, S. Sigurdsson, Q. Wong, R. Erbel, S. Kathiresan, O. Melander, V. Gudnason, C. J. O’Donnell, W. S. Post. Genetic associations with valvular calcification and aortic stenosis. New England Journal of Medicine. Vol. 368, pg. 503–512, 2013 https://doi.org/10.1056/NEJMoa1109034 [↩]
- R. Capoulade, K. L. Chan, C. Yeang, P. Mathieu, Y. Bossé, J. G. Dumesnil, J. W. Tam, K. K. Teo, A. Mahmut, X. Yang, J. L. Witztum, B. J. Arsenault, J.-P. Després, P. Pibarot, S. Tsimikas. Oxidized phospholipids, lipoprotein(a), and progression of calcific aortic valve stenosis. Journal of the American College of Cardiology. Vol. 66, pg. 1236–1246, 2015 https://doi.org/10.1016/j.jacc.2015.07.020 [↩]
- B. J. Arsenault, K. Loganath, A. Girard, S. Botezatu, K. H. Zheng, E. Tzolos, K. Abdoun, L. Tastet, R. Capoulade, N. Côté, N. Craig, K. L. Chan, J. W. Tam, K. K. Teo, C. Couture, M.-A. Clavel, P. Mathieu, S. Thériault, E. S. G. Stroes, D. E. Newby, S. Tsimikas, P. Pibarot, M. R. Dweck. Lipoprotein(a) and calcific aortic valve stenosis progression: a systematic review and meta-analysis. JAMA Cardiology. Vol. 9, pg. 835–842, 2024 https://doi.org/10.1001/jamacardio.2024.1882 [↩]
- Niacin in patients with low HDL cholesterol levels receiving intensive statin therapy | new england journal of medicine. https://www.nejm.org/doi/full/10.1056/NEJMoa1107579 [↩]
- HPS2-THRIVE Collaborative Group. HPS2-thrive randomized placebo-controlled trial in 25 673 high-risk patients of er niacin/laropiprant: trial design, pre-specified muscle and liver outcomes, and reasons for stopping study treatment. European Heart Journal. Vol. 34, pg. 1279–1291, 2013 https://doi.org/10.1093/eurheartj/eht055 [↩]
- I. Gregersen, E. Høibraaten, K. B. Holven, L. Løvdahl, T. Ueland, M.-C. Mowinckel, T. B. Dahl, P. Aukrust, B. Halvorsen, P. M. Sandset. Effect of hormone replacement therapy on atherogenic lipid profile in postmenopausal women. Thrombosis Research. Vol. 184, pg. 1–7, 2019 https://doi.org/10.1016/j.thromres.2019.10.005 [↩]
- M. Y. Abou-Ismail, D. C. Sridhar, L. Nayak. Estrogen and thrombosis: a bench to bedside review. Thrombosis Research. Vol. 192, pg. 40–51, 2020 https://doi.org/10.1016/j.thromres.2020.05.008 [↩]
- J. Leebmann, E. Roeseler, U. Julius, F. Heigl, R. Spitthoever, D. Heutling, P. Breitenberger, W. Maerz, W. Lehmacher, A. Heibges, R. Klingel. Lipoprotein apheresis in patients with maximally tolerated lipid-lowering therapy, lipoprotein(a)-hyperlipoproteinemia, and progressive cardiovascular disease: prospective observational multicenter study. Circulation. Vol. 128, pg. 2567–2576, 2013 https://doi.org/10.1161/CIRCULATIONAHA.113.002432 [↩]
- B. R. Jaeger, Y. Richter, D. Nagel, F. Heigl, A. Vogt, E. Roeseler, K. Parhofer, W. Ramlow, M. Koch, G. Utermann, C. A. Labarrere, D. Seidel. Longitudinal cohort study on the effectiveness of lipid apheresis treatment to reduce high lipoprotein(a) levels and prevent major adverse coronary events. Nature Reviews Cardiology. Vol. 6, pg. 229–239, 2009 https://doi.org/10.1038/ncpcardio1456 [↩]
- F. Schumann, U. Kassner, D. Spira, F. F. Zimmermann, T. Bobbert, E. Steinhagen-Thiessen, T. Hollstein. Long-term lipoprotein apheresis reduces cardiovascular events in high-risk patients with isolated lipoprotein(a) elevation. Journal of Clinical Lipidology. Vol. 18, pg. e738–e745, 2024 https://doi.org/10.1016/j.jacl.2024.04.134 [↩]
- R. Klingel, U. Julius, W. M. Bernhardt, F. Heigl, R. Spitthoever, J. Leebmann, V. J. J. Schettler, W. Lehmacher, B. G. Nordestgaard, F. Kronenberg, A. Heibges, R. Klingel. Lipoprotein apheresis for lipoprotein(a)-associated progressive atherosclerotic cardiovascular disease: 12-years follow-up. Atherosclerosis. Vol. 410, pg. 120508, 2025 https://doi.org/10.1016/j.atherosclerosis.2025.120508 [↩]
- A. C. Goldberg, S. S. Gidding. Knowing the prevalence of familial hypercholesterolemia matters. Circulation. Vol. 133, pg. 1054–1057, 2016 https://doi.org/10.1161/CIRCULATIONAHA.116.021673 [↩] [↩]
- I. Skoumas, I. Andrikou, S. Simantiris, K. Grigoriou, I. Dima, D. Terentes-Printzios, A. Papanikolaou, K. Akinosoglou, K. Tsioufis, C. Vlachopoulos. Lipoprotein(a) in familial dyslipidemias: the effect on cardiovascular prognosis in patients with familial hypercholesterolemia or familial combined hyperlipidemia. Nutrition, Metabolism and Cardiovascular Diseases. Vol. 35, pg. 103867, 2025 https://doi.org/10.1016/j.numecd.2025.103867 [↩]
- P. Kokkinos, C. Faselis, I. B. H. Samuel, A. Pittaras, M. Doumas, R. Murphy, M. S. Heimall, X. Sui, J. Zhang, J. Myers. Cardiorespiratory fitness and mortality risk across the spectra of age, race, and sex. Journal of the American College of Cardiology. Vol. 80, pg. 598–609, 2022 https://doi.org/10.1016/j.jacc.2022.05.031 [↩]
- U. Ekelund, J. Steene-Johannessen, W. J. Brown, M. W. Fagerland, N. Owen, K. E. Powell, A. Bauman, I.-M. Lee, Lancet Physical Activity Series 2 Executive Committe, Lancet Sedentary Behaviour Working Group. Does physical activity attenuate, or even eliminate, the detrimental association of sitting time with mortality? a harmonised meta-analysis of data from more than 1 million men and women. Lancet (London, England). Vol. 388, pg. 1302–1310, 2016 https://doi.org/10.1016/S0140-6736(16)30370-1 [↩] [↩]
- P. F. Kokkinos, C. Faselis, J. Myers, D. Panagiotakos, M. Doumas. Interactive effects of fitness and statin treatment on mortality risk in veterans with dyslipidaemia: a cohort study. Lancet (London, England). Vol. 381, pg. 394–399, 2013 https://doi.org/10.1016/S0140-6736(12)61426-3 [↩]
- M. L. O’Donoghue, R. S. Rosenson, J. A. G. López, N. E. Lepor, S. J. Baum, E. Stout, D. Gaudet, B. Knusel, J. F. Kuder, S. A. Murphy, H. Wang, Y. Wu, T. Shah, J. Wang, T. Wilmanski, W. Sohn, H. Kassahun, M. S. Sabatine. The off-treatment effects of olpasiran on lipoprotein(a) lowering: ocean(a)-dose extension period results. Journal of the American College of Cardiology. Vol. 84, pg. 790–797, 2024 https://doi.org/10.1016/j.jacc.2024.05.058 [↩]
- M. L. O’Donoghue, R. S. Rosenson, B. Gencer, J. A. G. López, N. E. Lepor, S. J. Baum, E. Stout, D. Gaudet, B. Knusel, J. F. Kuder, X. Ran, S. A. Murphy, H. Wang, Y. Wu, H. Kassahun, M. S. Sabatine. Small interfering rna to reduce lipoprotein(a) in cardiovascular disease. New England Journal of Medicine. Vol. 387, pg. 1855–1864, 2022 https://doi.org/10.1056/NEJMoa2211023 [↩]
- H. S. Bhatia, A. Bajaj, S. N. Goonewardena, P. M. Moriarty. Pelacarsen: mechanism of action and lp(a)-lowering effect. Journal of Clinical Lipidology. Vol. 19, pg. 1208–1222, 2025 https://doi.org/10.1016/j.jacl.2025.06.004 [↩]
- L. Cho, S. J. Nicholls, B. G. Nordestgaard, U. Landmesser, S. Tsimikas, M. J. Blaha, E. Leitersdorf, A. M. Lincoff, A. Lesogor, B. Manning, P. Kozlovski, H. Cao, J. Wang, S. E. Nissen. Design and rationale of lp(a)horizon trial: assessing the effect of lipoprotein(a) lowering with pelacarsen on major cardiovascular events in patients with cvd and elevated lp(a). American Heart Journal. Vol. 287, pg. 1–9, 2025 https://doi.org/10.1016/j.ahj.2025.03.019 [↩]