Abstract
Eczema and psoriasis are common inflammatory skin disorders, and each has distinct immune mechanisms that cause the disease and significantly impact the patients’ lives. Despite their high prevalence, there are many gaps in understanding regarding their differences and similarities. This study’s aim is to clarify the immune-mediated causes, symptoms, and treatments of the diseases and their impact on the skin. It will also discuss the topic of eczema’s classification as a hypersensitivity disease compared to an autoimmune disease like psoriasis. A literature review was conducted for this study that focused on existing knowledge about the diseases’ pathogenesis as well as recent findings in microbiome research, immunology, and treatments. Literature was collected using Google Scholar and was chosen based on content, relevance to the study, and date. Key findings of the study were that eczema is primarily driven by IgE-mediated reactions, cytokine and signal reactions, and skin barrier dysfunction. Psoriasis is associated with Th1 and Th17 cell pathways, antimicrobial peptides (AMPs), and the subsequent immune reactions, contributing to chronic inflammation and keratinocyte proliferation. Additionally, the role of other skin microbes was explored as a cause in both diseases, influencing severity and developing treatments. These findings benefit understandings of the pathophysiology of eczema and psoriasis, and highlight the need for further research into immune mechanisms.
Keywords: “the skin in eczema and psoriasis”, “pathogenesis of eczema”, “pathogenesis of psoriasis”, “filaggrin in eczema”, “filaggrin in psoriasis”, “the skin microbiome in eczema”, “the skin microbiome in psoriasis”, “hypersensitivity reactions in eczema”, “psoriasis autoimmunity”, “autoimmunity in eczema”, “IgE in eczema”, “symptoms and treatments for eczema”, “symptoms and treatments for psoriasis”
Introduction
The body’s largest and one of the most important organs, the skin, has many layers to it, both physically and in its functioning. The skin is composed of three main layers: the hypodermis, the dermis, and the epidermis, listed from the deepest to the most superficial layer1. Various skin conditions, such as eczema and psoriasis, can develop as a result of damage to the skin or predisposed genetic conditions.
Eczema, which refers to atopic dermatitis in this paper, is the most common inflammatory skin disease in the world, with a global cumulative lifetime prevalence of about 7-10%, which is dependent on location, environmental conditions, and population characteristics2. It is characterized by red, inflamed, and often itchy skin or eczematous lesions2. A condition distinct from eczema is psoriasis, another inflammatory skin disease that has overlapping symptoms with eczema, but is significantly less common, with a prevalence around 2 to 3%, though the prevalence fluctuates based on country. Psoriasis is chronic and can be identified by itching, burning skin, and red or white skin plaques, which often appear during flare-ups, similar to eczema3. These two diseases are often mixed up for many reasons, including their similar manifestations. However, their treatments and pathogenesis, especially the immune mechanisms that cause the diseases, make them very distinct. Eczema is an atopic and allergic disease, while psoriasis is an autoimmune disease. Their classifications are one of their major differences; however, some of the ways eczema is mediated resemble those of an autoimmune disease. This brings about another question regarding eczema’s classification as an allergic disease.
Under the Coombs and Gell classifications for hypersensitivity reactions, a type 1 hypersensitivity reaction is immunoglobulin E (IgE)-mediated and occurs when the body is exposed to an antigen and responds with several immune reactions. A common reaction is the degranulation of mast cells and basophils, which leads to the release of mediators such as histamine2,4. Atopic dermatitis is not a classic type I hypersensitivity. Type 1 hypersensitivity reactions can be seen as a result of the immune system’s overreaction to an allergen or trigger, which can contribute to characteristics of eczema, such as pruritus and inflammation. However, other symptoms of eczema, such as barrier dysfunction, involve other immune pathways and type 2 inflammation. Additionally, comorbidities of eczema, like allergic rhinitis and asthma, often have mechanisms that fall under the type 1 hypersensitivity category, but also involve other pathways2,5. A type 4 hypersensitivity reaction is delayed and typically T-cell mediated. Allergic contact dermatitis (ACD) is another condition that typically falls under the term “eczema”, but is separate from atopic dermatitis, which is what this paper mainly addresses. ACD is commonly associated with type 4 hypersensitivity reactions.
Psoriasis, however, is not a hypersensitivity reaction. It is an autoimmune disease caused by the excessive production of keratinocytes, the cells that make up most of the epidermis, the top layer of the skin. Psoriasis has a strong genetic link and involves both the innate and adaptive immune system, especially T-cells6.
This paper will outline the similarities and differences between eczema and psoriasis in their pathogenesis, symptoms, and treatments. The paper will also analyze how eczema’s overlapping immune mechanisms complicate its traditional frameworks.
Classifications and implications of eczema and psoriasis
The complexities around the classification of eczema and its relationship to psoriasis and autoimmunity emphasize gaps in our understanding of these diseases. Eczema (AD in this paper) is primarily categorized as an atopic, allergic condition associated with type 2 inflammation and, commonly, IgE-mediated sensitization. However, emerging connections can be made between its pathogenesis and autoimmunity because of its autoimmune-like mechanisms. This challenges traditional classifications of eczema, especially taking into account the presence of IgE autoantibodies and other autoimmune-like factors that may contribute to eczema. However, eczema should not be considered an autoimmune disease. Additionally, ACD is another condition that falls under the broader “eczema” term but is separate from AD, and ACD is associated with type 4 delayed hypersensitivity reactions. The overlapping immune pathways in eczema and psoriasis suggest common etiological factors, particularly in the skin microbiome and in systemic inflammation. Despite the proposed autoimmune-like mechanisms in some cases of eczema, eczema is still a mainly atopic allergic disease.
The dermis and layers of the epidermis
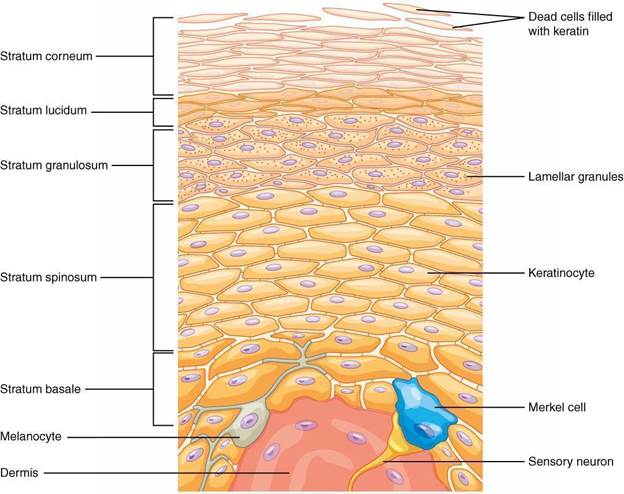
File:502 Layers of epidermis.jpg. (2025, August 24). Wikimedia Commons. Retrieved September 5, 2025, from https://commons.wikimedia.org/w/index.php?title=File:502_Layers_of_epidermis.jpg&oldid=1077204057.
Pathogenesis of eczema and psoriasis
The pathogenesis of eczema and psoriasis illustrates two very different mechanisms, yet in some places, they do overlap. While it has been difficult to know exactly what is occurring underneath the surface to cause these conditions, it is widely known that they are caused by overreactions in the immune system and skin barrier dysfunction.
The three layers of the skin form an intricate and irreplaceable network and barrier. It fights against allergens, pathogens, ultraviolet (UV) lights, chemicals, injury, and other external threats. The hypodermis is the deepest layer of skin and is mostly made up of fat and tissues. It contains sensory neurons, glands, and hair roots. On top of the hypodermis is the dermis, which has two layers of connective tissue and is made up of tough collagen fibers that make the skin strong and stretchy7. The topmost layer, the epidermis, is separated into five layers of its own (see Figure 1): the stratum basale, the stratum spinosum, the stratum granulosum, the stratum lucidum, and the stratum corneum. The basement membrane separates the stratum basale from the top of the dermis.
Stem cells in the stratum basale produce keratinocytes, which are the predominant cells present in the epidermis. Keratinocytes produce keratin and lipids that form the skin’s epidermal barrier and enable ultraviolet B (UVB) light absorption, which contributes to the activation of vitamin D.
Keratinocytes can be found in the stratum spinosum, along with dendritic cells, including Langerhans cells, which act as the skin’s first line of defense and work to drive inflammation. In the skin of individuals with eczema, there is an increased amount of these cells2. Dendritic cells are also major contributors to the uncontrolled inflammation seen in psoriasis3. The stratum granulosum is composed of several layers of cells, flattened keratinocytes, keratohyalin granules, and lamellar granules. The process where the skin begins to become waterproof and produce keratin is keratinization, and continues throughout the rest of the layers of the skin. However, keratinization and its results can be affected by gene mutations related to eczema and psoriasis or keratinocyte damage, which may lead to inflammation and problems with the skin1,8.
The thickest and most superficial layer of the epidermis, the stratum corneum, is made up of corneocytes, which are anucleated keratinocytes that have completed keratinocyte differentiation. Corneocytes still retain keratin filaments within a filaggrin matrix. This layer serves a crucial role, acting as more than just the first physical barrier, but also the first line of defense in the skin. To defend the skin, keratinocytes deeper in the skin release defensins, anti-microbial peptides (AMPs) that are a part of the innate immune system that keeps the skin healthy1,9,10
The skin’s role in eczema
The pathogenesis of eczema is driven by various factors, which include skin defects, microbiome, IgE autoantibodies, and the itch–scratch cycle. Eczema is caused by defects in the skin barrier and extreme immune reactions. In healthy skin, the stratum corneum is effective in keeping out intruders such as allergens and pathogens. However, in eczema, the skin barrier is compromised, and the allergens and pathogens can get through the skin, leading to increased immune reactions11. Mutations in the FLG gene (filaggrin gene), which encodes the protein filaggrin, cause reduced expression of filaggrin12. A deficiency in filaggrin causes increased permeability in the epidermal barrier and, therefore, increased immune activation and responses. The balance of fat in the skin is also disregulated, causing the skin to lose moisture13. However, a lack of filaggrin does not always cause eczema, and eczema is not always the result of a filaggrin deficiency. Filaggrin-2 (FLG2) is also linked to the skin barrier, and its presence is minimized in some dermatitic skin, but it is not proven to be as prominent in eczema specifically14. Keratinocytes and a family of serine proteases they secrete called kallikreins (KLK) are also thought to be involved in the pathogenesis of eczema, one specific kallikrein, KLK7, being required in eczema’s chronic itching15.
The itch-scratch cycle of eczema is a vicious loop that damages the skin and is a significant challenge in managing eczema. The structure of eczematous skin is disrupted in different ways, therefore causing problems in the skin barrier. When keratinocytes in the skin release cytokines, interleukins, and proteins, nerve fibers in the skin are activated and send messages to the brain through the spinal cord. When the brain receives these messages, it causes the hands to scratch as a reaction to the discomfort. This, in turn, activates the part of the brain responsible for reward mechanisms, providing temporary relief and satisfaction from itching. Although scratching is damaging to the skin and can cause symptoms of eczema to worsen, the brain has already associated itching with a feeling of satisfaction16.
Typically, mast cells are the culprit of causing itching, especially in IgE-dependent, allergic conditions such as eczema. When mast cells degranulate as a response to stimuli, they release histamine, which triggers inflammatory responses, especially itching17. Antihistamines block the histamine receptor (H1R), relieving some itch. However, antihistamines are not as efficient in blocking eczema itch, suggesting that different nerves and receptors are involved in eczema than in typical histamine itch reactions. Two nerves in the brain, gamma-aminobutyric acid (GABA) nerves and dopaminergic (DA) nerves, are associated with causing the uncomfortable and satisfactory feelings in the eczema itch17.
A more recently recognized aspect of eczema and psoriasis is the role the skin’s microbiome plays. When unbalanced, the skin microbiome can cause eczema and psoriasis, making it a promising target for new treatments. Both diseases involve harmful bacteria found in the microbiome: Staphylococcus aureus in eczema and Streptococcaceae in psoriasis.
The skin is more than just a barrier that keeps out intruders like bacteria—it has its own ecosystem of bacteria, fungi, viruses, and other microbiota, the microbiome. Symbiotic microbes found on the skin, or commensals, support the skin barrier by helping it break down natural products and supporting its repair and formation mechanisms. Microbial diversity is critical in healthy skin; a greater balance of microbial diversity can be an indicator of healthy skin. The skin is able to limit bacterial growth through its high salt concentration, low pH, and production of antimicrobial peptides (AMPs), which are produced by keratinocytes and sebocytes.
When imbalances to the skin’s microbiota, dysbiosis, occur, especially in skin with eczema, there is a lower microbial diversity and therefore less healthy skin. While S. aureus is a bacterium found commonly on the skin, a higher abundance of S. aureus is associated with a higher severity of eczema. It can weaken the skin barrier by releasing superantigens, enzymatic toxins, adhesins, and proteases. S. aureus also forms biofilms, which protect the bacteria against immune responses and external factors that might be able to harm the microbe, such as antibiotics and antifungal treatment. This creates a challenge when using antibiotics or antifungals as a treatment against S. aureus; however, scientists are exploring other methods involving other factors in eczema18. These biofilms are important in eczema, and their production is positively correlated with the severity of eczema. Additionally, it is found that the strain variations of Staphylococcus bacteria, such as Staphylococcus epidermis, influence the formation of biofilms and the severity of eczema19.
Regular skin versus skin in eczema
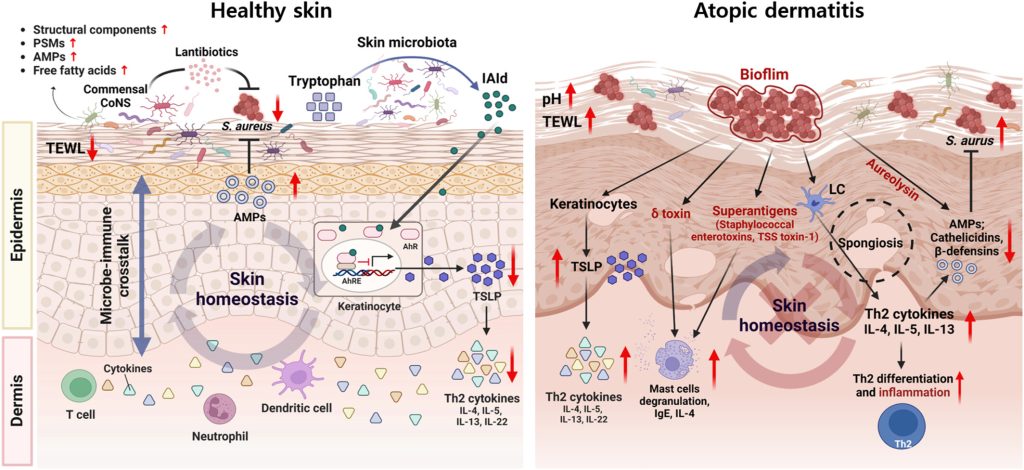
Diagram comparing the healthy skin microbiome and the mechanisms in the microbiome that regulate the pathogenesis of atopic dermatitis. Adopted from Figure 2 of Kim et al., 2023. On the left is an image of reactions occurring in healthy skin, with proper skin homeostasis. Levels of S. aureus, thymic stromal lymphopoietin (TSLP, an alarmin), and Th2 cytokines are lower, and the levels of anti-microbial peptides (AMPs) are up. The layers of the skin are even and form a barrier against pathogens, with a healthy skin microbiota. On the right side is an image of what may occur in the skin of an individual with atopic dermatitis and dysbiosis in the skin. The levels of S. aureus are up, and the skin microbiota and S. aureus are penetrating a damaged skin barrier. Biofilm from S. aureus points to keratinocytes, the delta toxin, superantigens, Langerhans cells (LCs), and aureolysin, which contributes to the virulence of S. aureus. The keratinocytes have an arrow pointing to TSLP, which has a red arrow signaling upregulation. This symbolizes the release of alarmins like TSLP when keratinocytes are damaged, leading to the upregulation of Th2 cytokines (pictured in the bottom left of the right image, with a red arrow pointing up). Additionally, the delta toxin and superantigens are pointing to the mast cell degranulation, which is upregulated, contributing to inflammation, and the LCs are pointing to the Th2 cytokines, which have an upregulated activity leading to the downregulation of AMPs, CAMPs, and beta defensins, which are part of the innate immune response and are important in keeping the skin healthy. The LCs also point to increased Th2 differentiation and inflammation, which are commonly seen in atopic dermatitis.
The skin and gut microbiome in psoriasis
In contrast to eczema, the relationship between the pathogenesis of psoriasis and the skin’s microbiome remains inconclusive. Despite many studies conducted and much data gathered, conflicting findings and different sampling methods contribute to discrepancies and limit reliability and reproducibility in research about the role of microbiota in psoriasis.
However, through association studies and examining the possible mechanisms behind psoriasis, tentative links can be made between an increase in the abundance of the Streptococcus microbe on the skin and the development of psoriasis20. It is found that there were slightly higher rates of Streptococcus in psoriatic lesions compared to normal skin20,21,22. In another study, it was found that there was a slightly greater frequency of Streptococcaceae in lesional psoriatic skin than in lesional eczematous skin and normal skin. On the other hand, there was also a much greater frequency of Staphylococcaceae found in eczematous skin than in psoriatic lesional skin23. Even these associations, though, may also be attributed as a consequence of the inflammatory microenvironment of a skin lesion in psoriasis.
The gut microbiome plays a role in the pathogenesis of psoriasis as well and helps regulate the immune system. Dysbiosis in the gut affects how microbes can work together and can also result in increased gut permeability. This allows more foreign substances to enter the body and the dislocation of microbial antigens and their byproduct metabolites into the blood. This triggers an immune reaction, causing inflammation in the gut and throughout the body. Inflammation in the skin worsens psoriasis, making dysbiosis in the gut microbiome part of psoriasis’s pathogenesis24.
Immune-mediated responses in eczema and psoriasis
Eczema and psoriasis are not just defects in the skin barrier; however, immune-mediated responses cause and sustain chronic inflammation, driving long-term symptoms for both diseases.
Once the skin barrier is already impaired, damaged keratinocytes release alarmins, a protein and subgroup of DAMPs (damage-associated molecular patterns), that cause inflammation in response (See Figure 2). Significant alarmins in eczema’s immune response include IL-1β, IL-25, IL-33, and thymic stromal lymphopoietin (TSLP). TSLP activates dendritic cells and induces expression of OX40 ligand (OX40L), driving naïve T-cells to become Th2 cells. Activated Th2 cells produce cytokines such as IL-4, IL-5, and IL-13, which amplify the immune responses. In parallel, dendritic cells process and present environmental antigens, further reinforcing Th2 skewing.
Innate lymphoid cells (ILC-2s) produce IL-5 and IL-13, which recruit eosinophils and are involved in stimulating nerves to cause itching, respectively. This reaction strengthens the type 2 immunity response and boosts IgE production.
As inflammation continues in the skin, additional T helper cells (Th1, Th17, and Th22) and their respective interleukins contribute to long-term inflammation and symptoms, though the exact role and activation of Th1 and Th17 cells are not fully clear2.
Many patients with eczema have IgE autoantibodies that bind to self-antigens. When the autoantibodies bind to self-antigens, histamine and cytokines are released and cause inflammation25. A study reported in 2009 found that there are at least 140 proteins that can act as IgE-binding self-antigens, and there are almost certainly more to be found. This proves that eczema may have autoimmune-like mechanisms, and also contributes to the idea of autoallergy, an autoimmune reaction that involves IgE. However, there is no proven causation to back this, only correlation and an overlap with autoimmunity. This gap in data is an important area to focus on for future research25,26. Future studies could explore more about how IgE autoantibodies affect eczema and how they interact with other antigens to contribute to the disease’s pathology.
The specific cause that triggers the activation of faulty immune cells is not fully known or understood. However, many people with psoriasis also have a family history of psoriasis, suggesting genetics as a probable factor. Some genes are already known to be involved in psoriasis’s development; psoriasis is strongly associated with the HLA-C locus3,27.
Keratinocytes, dendritic cells, and T-cells have key roles in the pathogenesis of psoriasis. As seen in Figure 2, damaged keratinocytes release antimicrobial peptides (AMPs), proteins that are part of the innate immune system. Cathelicidin antimicrobial peptides (CAMPs) are a type of AMP whose production is upregulated when there is epithelial damage.
CAMPs cause dendritic cells to activate an immune response. Cytokines like IL-12 and IL-23 are released, leading to prolonged inflammatory responses. These cytokines encourage T helper cells to become Th1 and Th17 cells. Cytokines increase inflammation by calling in more immune cells, and cause skin cells to divide and grow more rapidly than usual, leading to thickened skin. Keratinocytes further contribute to inflammation by creating more inflammatory signals. This creates a loop of more immune activation and, consequently, inflammation.
The result of these reactions is the clinical manifestations of psoriasis: acanthosis, or thickened skin, the Auspitz sign, or pinpoint bleeding, and other symptoms of psoriasis3,28.
Immune components in psoriatic skin
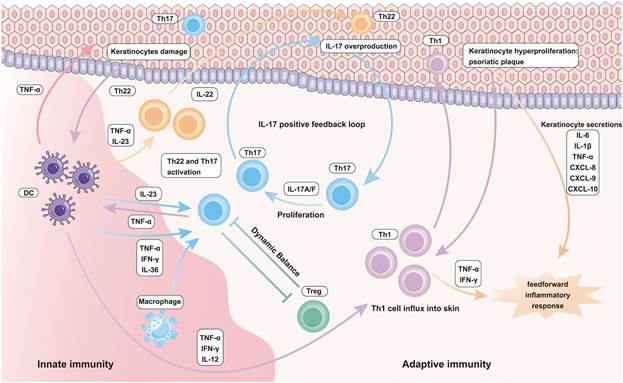
Immune cells in the epithelial immune microenvironment of psoriasis: emerging therapeutic targets – Scientific Figure on ResearchGate. Available from: https://www.researchgate.net/figure/Inflammatory-role-of-innate-and-adaptive-immune-cells-in-Psoriasis_fig2_377528087 [accessed 16 Aug 2025]. This figure shows the immune responses involved in psoriatic skin. Various factors can start the reactions found in psoriasis. In this figure, damaged keratinocytes (top left) activate dendritic cells and macrophages, leading to the production of IL-23 and TNF-ɑ. These activate the Th17 and Th22 cells. In the center, Th17 cells producing IL-17A/F and the excessive signaling of IL-17 form a positive feedback loop. Th22 cells contribute to keratinocyte proliferation and the formation of psoriatic plaques. This, plus TNF-α and IFN-γ, feeds into the inflammatory response.
| Eczema | Psoriasis | Similarities |
| Red and dry skin29 | Thick, cracked, and red skin that may bleed27 | Itchy skin27,28 |
| Rashes (may ooze fluid or bleed when scratched)29 | Erythematous patches with silvery plaques27,30 | Lichenification29 |
| Erythematous papules29 | Pus-filled lesions (in pustular psoriasis)30 | Poor sleep quality3,27 |
| Swelling31,32 | Fissured tongue (in oral psoriasis)30 | |
| Hyperlinear palms33 | Thick, pitted nails27 | |
| Dennie-Morgan lines33 | Auspitz sign (pinpoint bleeding)30 | |
| Discolored skin 31,32 | ||
| Inflamed skin28 |
In eczematous skin, red, dry, and itchy skin is very common. Some more severe cases of eczema might show swollen and inflamed skin, oozing or bleeding rashes, discolored skin, and lichenification, which is when the skin becomes hardened and leathery. Erythematous papules are often found in infants and are red, small bumps on the skin. Dennie-Morgan lines are an extra fold of skin that can be found below the lower lid. An increased number and depth of skin creases (hyperlinear palms) can also be observed27,28,29.
Symptoms of eczema (atopic dermatitis) are usually found in flexural surfaces, such as the inner elbow, back of the knees, and the neck. It can also be found in the hands (hand eczema), on the ankles and feet, and around the eyes27. Comorbidities of eczema include allergies (food allergy), allergic rhinitis, and asthma2. In addition, eczema is associated with mental health diseases (e.g., attention-deficit hyperactivity disorder (ADHD), depression, and anxiety) and other immune-mediated diseases (e.g., inflammatory bowel disease, rheumatoid arthritis, alopecia areata, and vitiligo)2.
Inflamed red skin with white skin plaques is a well-known symptom of psoriasis, and other symptoms include thick, cracked, red, and bleeding skin, itchy skin, lichenification, thick, pitted nails, and poor sleep quality. In specific types of psoriasis, different symptoms might present31,32,33.
Types of psoriasis are plaque psoriasis, guttate psoriasis, pustular psoriasis, inverse psoriasis, and erythrodermic psoriasis. Psoriasis can present on the tongue, on the scalp, on the nails, on the feet, and most commonly, on extensor surfaces such as the knees and elbows. Plaque psoriasis is the most common type of psoriasis, presenting as red skin patches with white plaques. Guttate psoriasis is characterized by small red dots on the limbs and trunk. Pustular psoriasis causes pus-filled spots on the skin, usually appearing on the hands and feet, but sometimes across the body. Inverse psoriasis is red and smooth psoriasis found in intertriginous areas that have folds of the skin. Erythrodermic psoriasis is a very severe and painful type of psoriasis that causes red and scaly skin all across the body31,32.
Psoriasis is also associated with rheumatoid arthritis, hypocalcemia, psoriatic arthritis, other autoimmune diseases, sleep apnea, and metabolic disease3. People with psoriasis are at a higher risk of developing cardiovascular diseases3. Obesity is a risk factor for psoriasis3.
Similarities and differences between the symptoms of eczema and psoriasis can also be seen in Figure 4. Both diseases share common symptoms like itchy skin, lichenification, and poor sleep quality. However, more distinct symptoms of eczema, but not psoriasis, include hyperlinear palms and Dennie-Morgan lines. Some symptoms seen in psoriasis, like the Auspitz sign, fissured tongue, silver plaques, and thick, pitted nails, are not seen in eczema.
Treatments
Significant differences and similarities can be seen in the treatments for eczema and psoriasis, showing how treatments and therapies are influenced by the distinct immune-mediated causes of each disease (reference Figures 5 and 6).
Treatments for eczema range from topical creams to drugs to therapies. Most commonly used, moisturizers and topical corticosteroids help maintain healthy skin in eczema. Emollients, including ointments and creams, are all topical treatments that are encouraged to be implemented in daily routines. Topical corticosteroids range in strength and can be over-the-counter or prescription medications, and target the immune system. New research is helping develop steroids and drugs that target the microbiome. Immunosuppressants like calcineurin, JAK, and TNF inhibitors are also effective treatments. A drug called dupilumab was found to have the ability to block IL-4 and IL-13 and reverse molecular disease pathways in the skin, proving that eczema is more than just a barrier defect. Newer or currently evolving treatments include phototherapy, UV treatments, and pre-/pro/post-biotics2,20,34,35.
Similar to eczema, psoriasis’s treatments involve corticosteroids, emollients, calcineurin inhibitors, and phototherapy. Other treatments include therapies, oral treatments, and vitamin D analogs. Biologic therapies are more common in psoriasis than in eczema, with there being at least 10 effective, approved drugs for treating psoriasis, and more being experimented with3,30. Biologic drugs are considered to be much more developed and effective for treating psoriasis.
Treatments can have negative effects as well. In eczema, some treatments may cause burning, skin atrophy, flu-like symptoms, and striae36. In treatments for psoriasis, adverse effects such as cutaneous atrophy, telangiectasia, and striae were found37.
| Eczema | Psoriasis |
| Biotics (pre/pro/post-biotics) | Vitamin D analogs |
| Phototherapy | Topical corticosteroids |
| Topical corticosteroids | Phototherapy |
| Immunosuppresants | Immunosuppresants |
| Biologic drugs | Biologic drugs |
| Corticosteroid | Potency |
| Hydrocortisone 1% | Mild |
| Hydrocortisone butyrate 0.1% | Mild-moderate |
| Betamethasone valerate 0.1% | Moderate |
| Triamcinolone acetonide 0.5% | Moderate-potent |
| Betamethasone dipropionate 0.05% | Potent |
| Halobetasol propionate 0.05% | Very potent |
| Clobetasol propionate 0.05% | Very potent |
Phototherapy as a treatment for eczema can be effective when topical corticosteroids are insufficient, depending on the severity of the disease. The concentration of each corticosteroid used in the treatments also impacts how potent the treatment is, and the order of potency in the list of corticosteroids is a generalized list. Additionally, moisturizers, ointments, and creams are all examples of common vehicles of drug delivery, and the type of vehicle used can impact the effectiveness of the treatment39. It can be seen that eczema and psoriasis have similar general treatments, but the specific treatments used differ for each. The exact comparison of efficacy between specific treatments is a gap in research. Generally, topical treatments are most helpful in mild cases, while therapies and biologics are most effective in more severe eczema and psoriasis.
Comparison of eczema and psoriasis
Both eczema and psoriasis derive from damaged skin barriers and involve inflammation. Eczema is more connected to skin barrier dysfunction and environmental triggers, while psoriasis is more connected with immune dysregulation. In eczema, allergens are able to penetrate the damaged skin, which leads to cytokine-induced inflammatory reactions. Additionally, mutations in the FLG gene, which lead to the loss of filaggrin in the skin, and dysbiosis in the skin microbiome can also contribute to damage to the skin and contribute to the development of eczema. The Streptococcaceae bacteria in the skin are associated with some types of psoriasis. Dysbiosis in the gut microbiome is also being studied as a factor in psoriasis.
In the pathogenesis of atopic dermatitis, damaged keratinocytes release alarmins to begin a reaction that leads to T-cell differentiation and type 2 inflammation. In other pathways relating to other types of eczema, such as contact dermatitis, reactions lead to immune responses associated with type 4 hypersensitivity. Th2 is the most involved in eczema’s pathogenesis, but Th1, Th17, and Th22 also contribute. Eczema is characterized more by itchy and inflamed skin. In psoriasis’s pathogenesis, keratinocytes, dendritic cells, and T-cells are important. Many factors, such as AMPs, viral DNA, and cytokine signaling cascades, lead to inflammation and keratinocyte hyperproliferation. These contribute to the appearance of psoriasis: scaling and thickened skin.
Eczema’s comorbidities include allergic rhinitis, asthma, and other allergic diseases. Rheumatoid arthritis is a comorbidity shared by both diseases, though it is more strongly associated with psoriasis than eczema. Both are also linked to an increased risk for other inflammatory diseases. Other comorbidities of psoriasis include psoriatic arthritis and metabolic diseases.
Their treatments both include corticosteroids, emollients, immunosuppressants, and biologic drugs, though there are more biologic therapies approved for psoriasis.
Effects of eczema and psoriasis on the skin
Eczema and psoriasis have significant impacts on the skin and how it is able to function because of both its short-term and long-term impacts. Immune reactions in the skin and bacterial infections cause the skin barrier to become damaged, which allows bacteria, fungi, and viruses to enter the skin more easily, increasing the risk of getting infections. The prolonged dryness of the skin leads to the skin having a decreased ability to keep moisture33. Skin texture and appearance can also change, leading to hyperpigmentation or discoloration, and in some cases, scarring, especially where scratching has occurred.
Methods
Differences and similarities between the pathogenesis, classifications, symptoms, and treatments in eczema and psoriasis were discussed through a literature review
Key terms such as “the skin in eczema and psoriasis”, “pathogenesis of eczema”, “pathogenesis of psoriasis”, “filaggrin in eczema”, “filaggrin in psoriasis”, “the skin microbiome in eczema”, “the skin microbiome in psoriasis”, “hypersensitivity reactions in eczema”, “psoriasis autoimmunity”, “autoimmunity in eczema”, “IgE in eczema”, “symptoms and treatments for eczema”, and “symptoms and treatments for psoriasis” were used.
Databases such as Google Scholar and PubMed were searched using key terms to find research papers, papers covering experiments, peer-reviewed government articles, and literature reviews covering eczema and psoriasis only. The date of publication for the pieces of literature used was limited to 1983 at the earliest, and onward to ensure more recent, relevant, and accurate information was being used, while also including literature covering the foundations of a topic when it was first introduced or discovered. For information about the effect of the skin’s microbiome in eczema and psoriasis, as well as developing treatments and new ideas, only literature from 2019 onward was used. Websites, blogs, and non peer-reviewed sources were excluded.
Information relevant to the topics of the literature review being written were recorded and organized into categories to be used in specific parts of the literature review (the skin, pathogenesis of both eczema and psoriasis, filaggrin, the skin’s microbiome in both diseases, hypersensitivity reactions, autoimmune diseases, treatments, symptoms, and diagnosis for eczema and psoriasis, comparisons and classifications).
Gaps in data or information, along with new approaches to treatments for eczema and psoriasis based on new ideas in areas such as the skin microbiome, were also identified and included.
| Paper | Key Findings |
| Atopic Dermatitis by Weidinger, S., et al.2 | This paper provided information on all aspects of atopic dermatitis, including prevalence, risk factors, comorbidities, pathophysiology, symptoms, treatments, and prevention. For the pathophysiology of atopic dermatitis, the paper found that a mutation of the FLG gene is the strongest genetic risk factor. The paper includes connections between atopic dermatitis and epidermal barrier dysfunction, immune pathways including cytokines and T-cells, and skin microbiota dysbiosis. Symptoms, treatments, and prevention are all explained thoroughly in the paper and cover multiple different symptoms and options for treatment and prevention. |
| Psoriasis by Greb, J. E., et al.3 | This paper provided information about the prevalence, risk factors, comorbidities, pathophysiology, symptoms, and treatments of psoriasis. Included was the immune responses behind psoriasis, the inflammatory responses, and T-cells as mediators. Treatments discussed included various therapies and medications. The paper also highlights the importance of further research on psoriasis. |
| Exploring the repertoire of IgE-binding self-antigens associated with atopic eczema by Zeller, S., et al.26 | The key finding of this paper was the identification of at least 140 possible IgE self-binding antigens associated with eczema. |
Conclusion
This paper has provided a review of the pathogenesis of eczema and psoriasis and a comparison of the two. Additionally, important aspects regarding the classification and immune-mediated mechanisms underlying eczema and psoriasis were discussed. Both diseases have similar inflammatory pathways involving T-cells and cytokine signaling. Eczema is more driven by skin barrier dysfunction and environmental allergens, whereas psoriasis is driven by immune dysregulation, classifying it as an autoimmune disease. This overall supports the fact that eczema is primarily an allergic, atopic disease with some overlapping factors with autoimmune diseases that do not fit strictly into one category. These findings address the main research goal, though not with completely solid conclusions, because of many gaps in knowledge in some areas. Nuances discovered suggest more complex relationships between the overlap of immune mechanisms and emphasize the importance of continued research. Several limitations of the study include limited research on the skin microbiome, specifically in psoriasis, the limited scope of the literature review, and the inability to cover all relevant studies that could provide additional insights. Relying on existing literature, experimental information, and data limits the ability to draw definitive conclusions. The exploration of eczema and psoriasis, and skin microbiome treatments can be a great benefit to research and for patients. This multidisciplinary approach combines dermatology, immunology, and the study of the microbiome. It will help decode the complexities of skin disorders like eczema and psoriasis and bring positive outcomes for patients.
Acknowledgements
I would like to express my sincere gratitude to my research mentor, Dr. Marah Chibwana, because with her guidance and encouragement, I was able to find what I wanted to research about and do it successfully, and learn about my interests. I would also like to thank Lumiere Research for allowing me to participate in their program and have this awesome opportunity to work with such a great mentor for my research.
References
- Yousef, H., Alhajj, M., & Sharma, S. (2024, June 8). Anatomy, Skin (Integument), Epidermis. PubMed; StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK470464/ [↩] [↩] [↩]
- Weidinger, S., Beck, L. A., Bieber, T., Kabashima, K., & Irvine, A. D. (2018). Atopic dermatitis. Nature Reviews Disease Primers, 4(1), 1–20. https://doi.org/10.1038/s41572-018-0001-z [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- Greb, J. E., Goldminz, A. M., Elder, J. T., Lebwohl, M. G., Gladman, D. D., Wu, J. J., Mehta, N. N., Finlay, A. Y., & Gottlieb, A. B. (2016). Psoriasis. Nature Reviews Disease Primers, 2(1). https://doi.org/10.1038/nrdp.2016.82 [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- Tanaka, S., & Furuta, K. (2021). Roles of IgE and Histamine in Mast Cell Maturation. Cells, 10(8), 2170. https://doi.org/10.3390/cells10082170 [↩]
- Fernandez, J. (2024, August 2). Overview of Allergic and Atopic Disorders. Merck Manual Professional Edition; Merck Manuals. https://www.merckmanuals.com/professional/immunology-allergic-disorders/allergic-autoimmune-and-other-hypersensitivity-disorders/overview-of-allergic-and-atopic-disorders#Classification-of-Hypersensitivity-Reactions_v994886 [↩]
- Ayala-Fontánez, N., Soler, D. C., & McCormick, T. S. (2016). Current knowledge on psoriasis and autoimmune diseases. Psoriasis (Auckland, N.Z.), 6, 7–32. https://doi.org/10.2147/PTT.S64950 [↩]
- In brief: How does skin work? (2025, April 22). Nih.gov; Institute for Quality and Efficiency in Health Care (IQWiG). https://www.ncbi.nlm.nih.gov/books/NBK279255/ [↩]
- Freeman, S. C., & Sidharth Sonthalia. (2023, May). Histology, Keratohyalin Granules. Nih.gov; StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK537049/ [↩]
- Europe PMC. (2016). Europe PMC. Europepmc.org. https://europepmc.org/article/NBK/nbk513299 [↩]
- Falconer, A., Ikram, M., Bissett, C. E., Cerio, R., Quinn, A. G., & Ali, R. S. (2001). Expression of the Peptide Antibiotics Human β Defensin-1 and Human β Defensin-2 in Normal Human Skin. Journal of Investigative Dermatology, 117(1), 106–111. https://doi.org/10.1046/j.0022-202x.2001.01401.x [↩]
- Yosipovitch, G., & Alexandru D. P. Papoiu. (2008). What causes itch in atopic dermatitis? Current Allergy and Asthma Reports, 8(4), 306–311. https://doi.org/10.1007/s11882-008-0049-z [↩]
- Rawlings, A. V., Scott, I. R., Harding, C. R., & Bowser, P. A. (1994). Stratum Corneum Moisturization at the Molecular Level. Journal of Investigative Dermatology, 103(5), 731–740. https://doi.org/10.1111/1523-1747.ep12398620 [↩]
- Overview: Eczema. (2021, February 11). Nih.gov; Institute for Quality and Efficiency in Health Care (IQWiG). https://www.ncbi.nlm.nih.gov/books/NBK279399/ [↩]
- Seykora, J., Dentchev, T., & Margolis, D. J. (2015). Filaggrin‐2 barrier protein inversely varies with skin inflammation. Experimental Dermatology, 24(9), 720–722. https://doi.org/10.1111/exd.12749 [↩]
- Guo, C. J., Mack, M. R., Oetjen, L. K., Trier, A. M., Council, M. L., Pavel, A. B., Guttman-Yassky, E., Kim, B. S., & Liu, Q. (2019). Kallikrein 7 Promotes Atopic Dermatitis-Associated Itch Independently of Skin Inflammation. Journal of Investigative Dermatology, 140(6), 1244-1252.e4. https://doi.org/10.1016/j.jid.2019.10.02214 [↩]
- Ferda Cevikbas, & Lerner, E. A. (2019). Physiology and Pathophysiology of Itch. Physiological Reviews, 100(3), 945–982. https://doi.org/10.1152/physrev.00017.2019 [↩]
- Su, X.-Y., Chen, M., Yuan, Y., Li, Y., Guo, S.-S., Luo, H.-Q., Huang, C., Sun, W., Li, Y., Zhu, M. X., Liu, M.-G., Hu, J., & Xu, T.-L. (2019). Central Processing of Itch in the Midbrain Reward Center. Neuron, 102(4), 858-872.e5. https://doi.org/10.1016/j.neuron.2019.03.030 [↩] [↩]
- Hülpüsch, C., Rohayem, R., Reiger, M., & Traidl-Hoffmann, C. (2024). Exploring the skin microbiome in atopic dermatitis pathogenesis and disease modification. Journal of Allergy and Clinical Immunology, 154(1), 31–41. https://doi.org/10.1016/j.jaci.2024.04.029 [↩]
- Gonzalez, T., Stevens, M. L., Baatyrbek kyzy, A., Alarcon, R., He, H., Kroner, J. W., Spagna, D., Grashel, B., Sidler, E., Martin, L. J., Biagini Myers, J. M., Khurana Hershey, G. K., & Herr, A. B. (2020). Biofilm propensity of Staphylococcus aureus skin isolates is associated with increased atopic dermatitis severity and barrier dysfunction in the MPAACH pediatric cohort. Allergy, 76(1), 302–313. https://doi.org/10.1111/all.14489 [↩]
- Lewis, D. J., Chan, W. H., Hinojosa, T., Hsu, S., & Feldman, S. R. (2019). Mechanisms of microbial pathogenesis and the role of the skin microbiome in psoriasis: A review. Clinics in Dermatology, 37(2), 160–166. https://doi.org/10.1016/j.clindermatol.2019.01.01126 [↩] [↩] [↩]
- Fahlén, A., Engstrand, L., Baker, B. S., Powles, A., & Fry, L. (2011). Comparison of bacterial microbiota in skin biopsies from normal and psoriatic skin. Archives of Dermatological Research, 304(1), 15–22. https://doi.org/10.1007/s00403-011-1189-x [↩]
- Gao, Z., Tseng, C., Strober, B. E., Pei, Z., & Blaser, M. J. (2008). Substantial Alterations of the Cutaneous Bacterial Biota in Psoriatic Lesions. PLoS ONE, 3(7), e2719–e2719. https://doi.org/10.1371/journal.pone.0002719 [↩]
- Drago, L., Grandi, R. D., Altomare, G., Paolo Pigatto, Rossi, O., & Toscano, M. (2016). Skin microbiota of first cousins affected by psoriasis and atopic dermatitis. Clinical and Molecular Allergy, 14(1). https://doi.org/10.1186/s12948-016-0038-z [↩]
- Chen, L., Li, J., Zhu, W., Kuang, Y., Liu, T., Zhang, W., Chen, X., & Peng, C. (2020). Skin and Gut Microbiome in Psoriasis: Gaining Insight Into the Pathophysiology of It and Finding Novel Therapeutic Strategies. Frontiers in Microbiology, 11. https://doi.org/10.3389/fmicb.2020.589726 [↩]
- Holmes, J., Fairclough, L. C., & Todd, I. (2019). Atopic dermatitis and autoimmunity: the occurrence of autoantibodies and their association with disease severity. Archives of Dermatological Research, 311(3), 141–162. https://doi.org/10.1007/s00403-019-01890-4 [↩] [↩]
- Zeller, S., Rhyner, C., Meyer, N., Schmid-Grendelmeier, P., Akdis, C. A., & Reto Crameri. (2009). Exploring the repertoire of IgE-binding self-antigens associated with atopic eczema. Journal of Allergy and Clinical Immunology, 124(2), 278-285.e7. https://doi.org/10.1016/j.jaci.2009.05.015 [↩] [↩]
- NIAMS. (2017, April 12). Psoriasis. National Institute of Arthritis and Musculoskeletal and Skin Diseases. https://www.niams.nih.gov/health-topics/psoriasis [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- Lande, R., Gregorio, J., Facchinetti, V., Chatterjee, B., Wang, Y.-H., Homey, B., Cao, W., Wang, Y.-H., Su, B., Nestle, F. O., Zal, T., Mellman, I., Schröder, J.-M., Liu, Y.-J., & Gilliet, M. (2007). Plasmacytoid dendritic cells sense self-DNA coupled with antimicrobial peptide. Nature, 449(7162), 564–569. https://doi.org/10.1038/nature06116 [↩] [↩] [↩] [↩]
- NIAMS. (2017, April 10). Atopic Dermatitis. National Institute of Arthritis and Musculoskeletal and Skin Diseases. https://www.niams.nih.gov/health-topics/atopic-dermatitis [↩] [↩] [↩] [↩] [↩]
- Nair, P. A., & Badri, T. (2023, April 3). Psoriasis. Nih.gov; StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK448194/ [↩] [↩] [↩] [↩] [↩]
- Hanifin, J. M., & Reed, M. L. (2007). A Population-Based Survey of Eczema Prevalence in the United States. Dermatitis, 18(2), 82–91. https://doi.org/10.2310/6620.2007.06034 [↩] [↩] [↩] [↩]
- Lönndahl, L., Abdelhadi, S., Holst, M., Lonne-Rahm, S.-B., Nordlind, K., & Johansson, B. (2023). Psychological Stress and Atopic Dermatitis: A Focus Group Study. Annals of Dermatology, 35(5), 342. https://doi.org/10.5021/ad.22.035 [↩] [↩] [↩] [↩]
- Nemeth, V., Syed, H. A., & Evans, J. (2024, March). Eczema. Nih.gov; StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK538209/ [↩] [↩] [↩] [↩]
- D’Ippolito, D., & Pisano, M. (2018). Dupilumab (Dupixent): An Interleukin-4 Receptor Antagonist for Atopic Dermatitis. Pharmacy and Therapeutics, 43(9), 532. https://pmc.ncbi.nlm.nih.gov/articles/PMC6110636/ [↩]
- Fabbrocini, G., Napolitano, M., Megna, M., Balato, N., & Patruno, C. (2018). Treatment of Atopic Dermatitis with Biologic Drugs. Dermatology and Therapy, 8(4), 527–538. https://doi.org/10.1007/s13555-018-0258-x [↩]
- Lee, J. H., Son, S. W., & Cho, S. H. (2016). A Comprehensive Review of the Treatment of Atopic Eczema. Allergy Asthma and Immunology Research, 8(3), 181–181. https://doi.org/10.4168/aair.2016.8.3.181 [↩]
- Bruner, C. R., Feldman, S. R., Madhuri Ventrapragada, & Fleischer, A. B. (2003). A Systematic Review of Adverse Effects Associated with Topical Treatments for Psoriasis. Dermatology Online Journal, 9(1). https://doi.org/10.5070/d30w66c5g [↩]
- Gabros, S., Nessel, T. A., & Zito, P. M. (2025, April 26). Topical Corticosteroids. Nih.gov; StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK532940/ [↩]
- Barnes, T. M., Dalibor Mijaljica, Townley, J. P., Spada, F., & Harrison, I. P. (2021). Vehicles for Drug Delivery and Cosmetic Moisturizers: Review and Comparison. Pharmaceutics, 13(12), 2012–2012. https://doi.org/10.3390/pharmaceutics13122012 [↩]