Abstract
Human tears still lie on uncharted waters. Historically, tears have long served as one of the most significant mediums of emotional expression in humans. Holding the longstanding social notion that “emotional people cry, while rational ones don’t,”most people consider tears as a straightforward and transient expression. Nonetheless, recent research suggests that the process of crying may originate from a more profound psychobiological foundation: Oxytocin and neural mirroring systems have each been associated with affective empathy and tearful responses. Although previous studies have mapped these two mechanisms independently, no research has been done on exploring their connection in emotional tear generation empirically. Recognizing this gap, we suggested that these two systems-oxytocin and neural mirroring systems-are deeply connected. The study aimed to investigate whether short-term social bonding influences emotional tearfulness through the combined activation of these pathways. In this experiment, the randomly assigned experimental group (14 students) participated in bonding activities designed to evoke affective empathy prior to emotional stimuli. Both groups (experimental and control) then watched emotionally evocative videos, and we measured their tearful reactions through both subjective self-reports and external observational ratings. Stronger displays of tearfulness and affective empathy in the experiment supported our hypothesis that social interaction may intensify emotional tearfulness through oxytocin-related pathways and emotional resonance. This high-school-level study did not employ direct measurements because of limited access to neural tools, thus future studies using direct measures such as fMRI, heart rate variability, or EEG are recommended.
Keywords: Emotional Tears, Affective Empathy, Oxytocin-Related Mechanisms, Neural Mirroring Systems, Emotional Resonance
Introduction
Emotional Tears
Emotional tears, which are elicited by powerful emotional stimuli such as sadness, awe, or frustration, can be seen in only humans1,2. Some studies have revealed that brain regions of the limbic system, particularly the amygdala and prefrontal cortex, are activated by these emotional stimuli3,4. Then they send signals to the hypothalamus to trigger tear secretion through the brainstem circuits that control the lacrimal glands2,5,6. At the same time, other scientists have focused on the social roles of tears. They have suggested that the external expression of tears may serve not only to reflect internal emotional states but also to draw social attention and evoke affective empathy, which thus functions as nonverbal communicative tools that facilitate emotional sharing7. This study was conducted to empirically explore whether oxytocin pathways and neural mirroring systems may modulate emotional tear generation.
The Neural Mirroring Systems
One of the most possible mechanisms of empathy is the mirror neuron system. When observing someone cry, people often experience an affective resonance, as if they are sharing the same emotion with another person8,9. This phenomenon has been linked to the Mirror Neurons Theory: it was first identified in macaque monkeys9,10,11. According to the theory, the mirror neurons activate in the ventral premotor cortex both when executing specific actions and observing another person engage in the same behavior9,10,11. These imitative mechanisms, therefore, may support the idea that the observation of others’ emotional expressions recruit neural pathways similar to those engaged during direct emotional experience and thus facilitate social connection with others12.
Further studies emerged that suggested the Mirror Neurons Theory was increasingly viable8,13. While the Mirror Neurons theory only explains the activation during motor actions10, an additional theory known as the Shared Manifold of Intersubjectivity also encodes emotional and sensory experiences8. This idea refers to a form of “neural resonance” in which the observer’s nervous system mirrors another’s internal state14. Thus, the production of emotional tears may reflect complex psychobiological interactions between perception and affective empathy.
However, some researchers are skeptical about the presence of mirror neurons at the single-cell level in the human brain due to insufficient evidence15,16. Acknowledging this limitation, this study adopts the more inclusive term “neural mirroring systems” instead of the narrowly defined “mirror neuron” terminology17.
Oxytocin and the Neurobiological Basis of Emotional Bonding
Recent research has established a new perspective of empathic mechanisms that oxytocin also may play a crucial role in mechanisms of empathy. Being released by the posterior pituitary gland, oxytocin originates from hypothalamus, particularly the paraventricular and supraoptic nuclei, and functions as a neuropeptide involved in various processes18. It is known as a classical endocrine hormone that helps stimulate uterine contractions and lactation19,20. While oxytocin functions as an endocrine hormone, it influences the maintenance of emotional regulation, the establishment of trust, and social connection21,22. It binds to oxytocin receptors (OXTRs) located in several brain regions (located in the amygdala, prefrontal cortex, and nucleus accumbens) are all associated with social behavior and responsiveness23,24. Some animal studies have also detected oxytocin receptors in lacrimal glands, implying a potential relationship between oxytocin-related pathways and tear production5. By examining whether oxytocin-related mechanisms could contribute to generating more tear production, we attempted to close this critical gap in neurobehavioral research.
Scope and Methodology Overview
We recruited twenty-eight East Asian high school students as participants. We compared tearfulness between bonding and control groups after engaging short-term bonding activities, which was only for the bonding group, and emotional video stimuli. Although we did not include direct methods such as neuroimaging or hormonal assays, we instead did use self-report questionnaires and behavioral observation to indirectly explore psychobiological mechanisms.
Methods
Participants
| 1 | A Pre-Screening Questionnaire Measuring Emotional Instability I have experienced noticeable mood swings in the past week. I feel emotionally unstable today. I experienced intense emotional moments that led me to cry within the past 3 days. I have recently felt irritable due to lack of sleep or stress. |
| 2 | Low Baseline Empathy Quotient I can easily tell how someone else is feeling. I often get emotionally involved with characters in books or movies. I tend to respond sensitively to other people’s emotions. Seeing someone in pain makes me feel sad as well. I try to understand the feelings of those who struggle to express themselves. |
| 3 | Self-Responded Emotional Response Questionnaire Did you experience the tearfulness of shedding tears during the video? How much did you empathize with the characters in the video? Did you feel the urge to suppress your emotions during the video? How long did your emotional state persist after the video ended? Overall, how emotionally affected were you by this video? |
This experiment was conducted to explore the influence of short-term social bonding activities on emotional tearfulness and empathic response. A total of thirty high school students aged 15 to 19, who belonged to the same school cohort, participated in the study. Before beginning the experiment, we gave all participants a detailed explanation of the procedures involved and a document confirming that they had no history of psychological disorders. Emotional instability (as stated in Table 1-1) and low baseline empathy (as stated in Table 1-2) were assessed via a 5-point Likert scale: those scoring a 5 on any emotional instability items or averaging 4 or above were excluded because scores of 4 or 5 generally fall within the highest quartile of responses on Likert scale, reflecting an extreme pattern that can conceal meaningful variation in emotional reactivity. Based on emotional instability, we excluded two participants whose scores were 5 on some specific items, respectively. Low baseline empathy was also measured through five questions, and the average score served as each participant’s initial level of affective empathy. The low baseline empathy and emotional instability questionnaires used in this study were self-developed, though their item structures were guided by well-established psychological models such as the Big Five Inventory (BFI) and Interpersonal Reactivity Index (IRI). Given the limited time and scope of a high-school-level project, the design prioritized conceptual consistency and practical feasibility over fully replicating standardized tools. Although the Baron-Cohen 60-item Empathy Quotient (EQ) is a tool widely used to measure empathy, it was not implemented here for the same rationale (high-school-level research). Instead, we developed a shortened, self-constructed questionnaire that was generated to reflect the core EQ dimensions: affective empathy, emotional reactivity, and social awareness. This approach ensured theoretical consistency with established empathy models while offering the flexibility needed for data gathering. The final consistency was satisfactory (Cronbach’s α = 0.84 for low baseline empathy; 0.81 for emotional instability), successfully reflecting individual differences pertinent to the study’s objectives.
Design, Procedure, and Measure
We recognized that prior familiarity among participants could influence emotional responses and considered this possibility by avoiding selecting students known to be close peers during the recruitment phase. Completely eliminating existing friendships in advance was beyond our control, but we informed participants that the grouping for the study would be entirely random and not based on their social ties to minimize their pre-existing familiarity. By informing it in advance, we attempted to reduce the potential effects of prior familiarity, although some level of it may still have remained. Subsequent random allocation of twenty-eight participants was guided to maintain fairness and reduce potential bias. Although no block randomization was formally applied, we discovered that an adequate balance between those two groups was successfully achieved after the analysis of the distribution of gender and baseline empathy scores.
Following the assignment, an 8-minute social bonding task was conducted by only participants in the emotionally stimulated group, in pairs.
For the first 3 minutes, pairs exchanged verbal compliments they had previously wanted to share. For the next 5 minutes, each participant wrote and read aloud a handwritten letter recalling a moment they were thankful for their partner. After the activity, participants immediately completed a self-assessment questionnaire that evaluated their perceived level of emotional bonding.
All participants then watched three emotionally evocative video clips presented in a fixed order under controlled environments (dim lighting, minimal noise, and separated seating) to ensure consistency of this experiment.
The clips were:
- Military Family Reunion
- Unsung Hero
- Dear Future Mom
This study measured tearfulness through two methods : observer-based assessment and self-reported data. During the video viewing, two observers independently evaluated participants’ visible emotional responses using structured criteria:
- Tears formation
- 0 : No visible tears, neutral facial expression / 1 : Slight eye moisture / 2 : Noticeable tear pooling / 3 : Tears running down the face / 4 : Wiping away tears / 5 : Tears accompanied by notable changes in facial expressions
- Additional Determining factors : Nose blowing
Immediately following the video session, all participants completed a self-report; the Emotional Response Questionnaire measured overall intensity of emotional response, tear frequency, empathic engagement, emotional endurance, and suppression tendencies (as stated in Table 1-3). In order to record responses, we also used a five-point Likert scale.
To measure any changes in empathy levels after performing the social bonding activities, we asked participants to re-complete the identical five-item empathy index initially administered during the pre-screening phase. This eventually allowed for comparison between baseline and post-bonding empathy levels. Observer ratings and participant self-reports were collected and analyzed independently.
In this study, the following statistical methods were used:
- Independent-sample t-tests: to compare group differences in tearfulness and empathy scores
- Pearson Correlation coefficients: to figure the relationship between changes in empathy and tearful responses
- Inter-rater reliability: to accurate the value between the two observers assessed
- Sensitivity analysis: to remove a statistical outlier to confirm the robustness of the findings
Ethical guidelines were followed throughout all procedures. We took into account the emotional nature of the stimuli both before and after the experiment. Adverse reactions were not reported.
Results
Group Differences in Tearfulness
As we expected, participants in the experimental group tended to exhibit higher levels of tearfulness than those in the control group.
| Experimental | Control | |||
|---|---|---|---|---|
| Self-report | Observer-report | Self-report | Observer-report | |
| < 3.0 | 0 (0%) | 2 (14.28%) | 5 (35.72%) | 8 (57.14%) |
| 3.0 — 3.49 | 5 (35.71%) | 3 (21.43%) | 4 (28.57%) | 3 (21.43%) |
| 3.5 — 3.99 | 1 (7.15%) | 2 (14.28%) | 3 (21.43%) | 3 (21.43%) |
| 4.0 — 4.49 | 5 (35.71%) | 6 (42.86%) | 2 (14.28%) | 0 (0%) |
| ≥ 4.5 | 3 (21.43%) | 1 (7.15%) | 0 (0%) | 0 (0%) |
| Total | 14 (100%) | 14 (100%) | 14 (100%) | 14 (100%) |
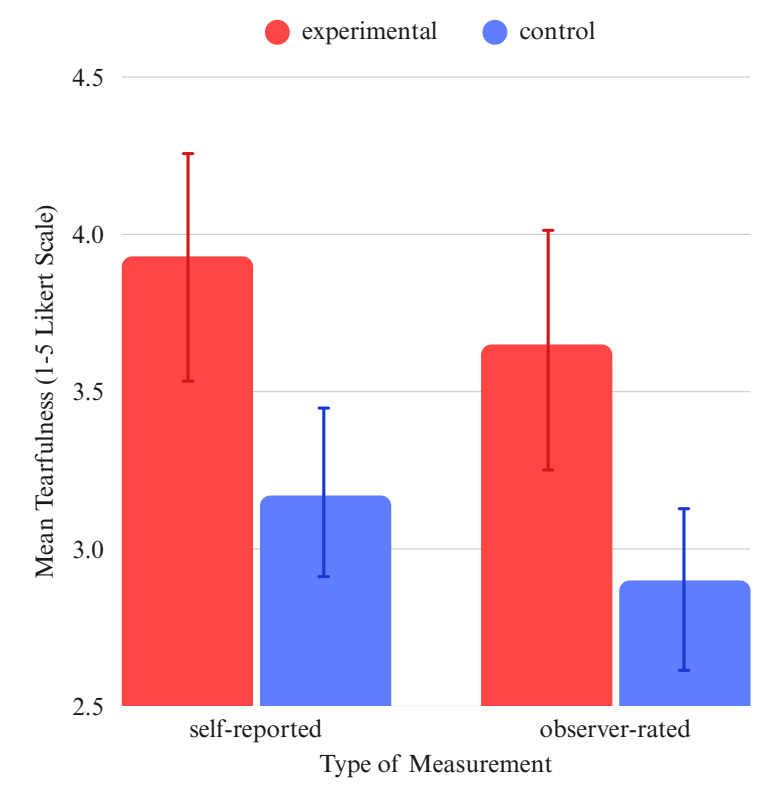
As illustrated in Figure 1, there is a similar trend in both self-reported and observer-rated tearfulness scores: While self-reported tearfulness averaged 3.93 (SD = 0.62) in the experimental group and 3.17 (SD = 0.50) in the control group, observer-rated tearfulness also showed mean values of 3.65 (SD = 0.63) for the experimental group and 2.90 (SD = 0.47) for the control. As shown in Table 2, the tearfulness scores among participants in the emotionally stimulated group were relatively much greater than those in the control group. To determine the mean difference in central tendency across the two groups, we ran an independent t-test (Welch’s t-test). The observed distinctions were statistically significant:
- t(24.88) = 3.5706, p = 0.0015 for self-reported tearfulness
- t(24.05) = 3.5703, p = 0.0015 for observer-rated tearfulness
In Welch’s t-test, when p < 0.05 (the typical alpha level), it is statistically significant. Since both p values are below significance level of 0.05, we conclude that these differences in tearfulness scores can be considered valid.
Change in Empathy Scores (EQ)
| Experimental | Control | |
|---|---|---|
| Pre-test | 3.46 | 3.28 |
| Pro-test | 3.94 | 3.44 |
| Experimental | Control | |
|---|---|---|
| < 0.5 | 6 (42.86%) | 13 (92.85%) |
| 0.5 — 0.99 | 8 (57.14%) | 1 (7.15%) |
| Total | 14 (100%) | 14 (100%) |
As described in Table 3, we found a notable increase in participants’ affective empathy scores after we compared pre- and post-test scores in the emotionally stimulated group: 3.46 to 3.94 (0.48-point gain). By contrast, the control group’s scores showed only a minor change, moving from 3.28 to 3.44 (0.16-point gain). According to Table 4, a substantial percentage of participants in the experimental group (57.14%) showed a change of empathy score increase of 0.5 or more compared to the control group. These findings suggest that even a short-term social bonding activity can temporarily elevate empathic sensitivity.
Correlation between Empathy Change and Tearfulness
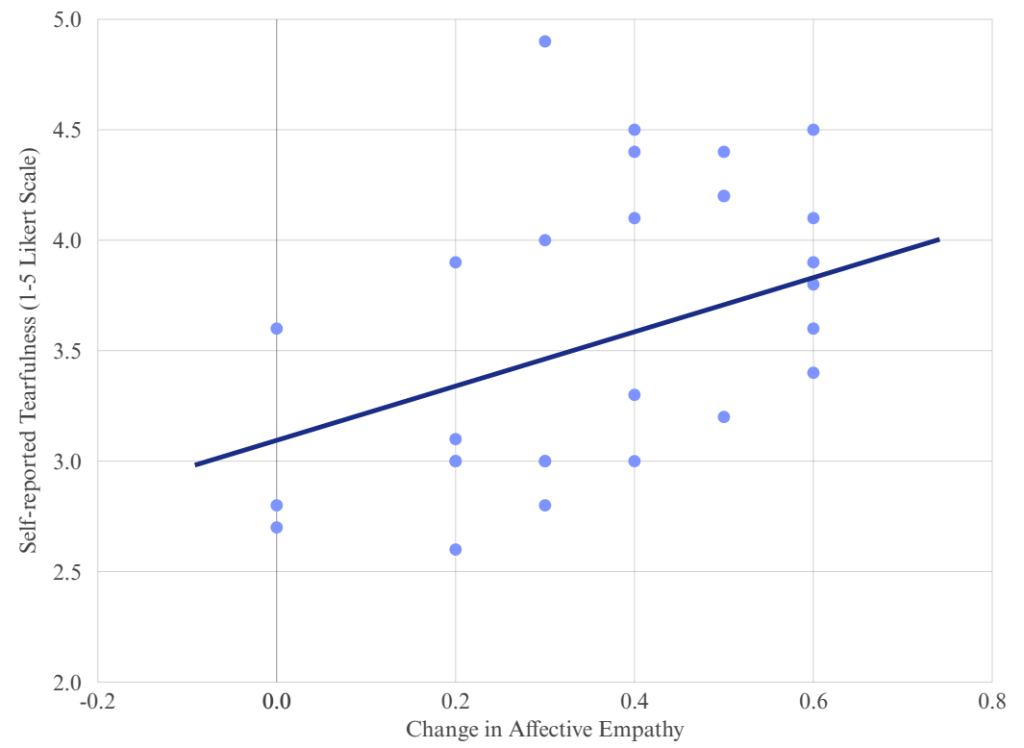
 0.05, 95% CI [-0.02, 0.82]). The regression line is illustrative, representing the positive trend based on observed data.
0.05, 95% CI [-0.02, 0.82]). The regression line is illustrative, representing the positive trend based on observed data.We conducted a correlation analysis to figure the relationship between changes in affective empathy and tearfulness and figured that there is a statistically meaningful relationship between empathy gains and self-reported tearfulness within the experimental condition. The correlation coefficient was r = 0.52. As illustrated in Figure 2, we got a moderate positive association, which is 95% CI [-0.02, 0.82], p <0.05. It indicates that individuals who experienced greater changes in affective empathy tended to exhibit stronger emotional reactions; therefore, this correlation may support the hypothesis that affective empathy can heighten emotional expression.
Inter-Rater Reliability of Observer Ratings
In order to maintain objectivity in the observed data, two independent observers were not able to have any information of group assignment. They evaluated each participant’s behavior separately. The inter-rater reliability assessed by Pearson correlation was high (r = 0.79), which indicates strong agreement between the two observers’ assessments.
Sensitivity Analysis
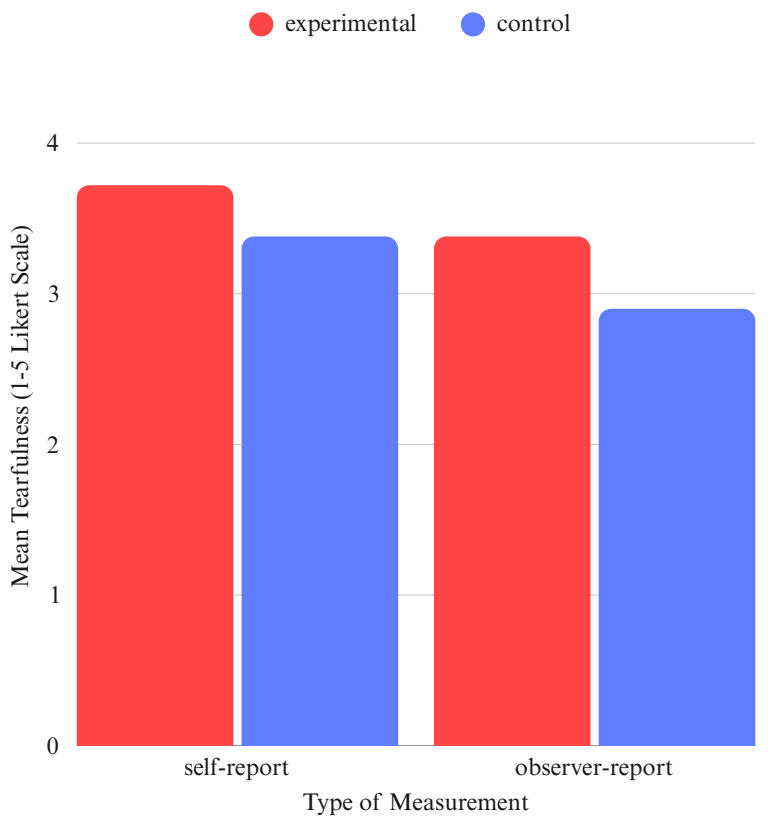
In the experimental group, a single participant showed exceptionally high scores across all measures: EQ = 4.6, Self-report = 4.9, Observer Score = 4.8, which all exceeded the group mean by over two standard deviations. We additionally analyzed the cases both including and excluding this score to confirm whether this outlier did skew the findings or not. As visualized in Figure 3, group averages in the experimental group remained stable even after this removal: 3.72 vs 3.17 for self-reports and 3.38 vs 2.90 for observer ratings. Based on this pattern, we confirmed that a single extreme case did not skew the main trends of the study, which thus reflects a wider pattern.
Discussion
In summary, this study suggests that even short-term social interaction may contribute to increased emotional tearfulness and affective empathy. The empathy referred to in this study represents affective empathy (the ability to share others’ feelings), as distinct from cognitive empathy (the ability to understand other’s mental state). Although most of the mechanisms are still unclear, the results support the idea that emotional crying may be related to deep psychobiological systems, including oxytocin pathways and neural mirroring systems2,8,22.
Neural Mirroring Systems and Empathic Responses
In this study, participants who engaged in social bonding activities prior to exposure to emotional stimuli showed greater tearfulness and empathic scores to emotional stimuli. This result may support the neural resonance theory, a concept hypothesizing that individuals simulate the emotional states of others when observing them8,9. These responses indicate that the neural mirroring system in humans may serve as a link between empathic processes and tearful responses, potentially helping activate shared neural circuits associated with empathy25. The overall behavioral patterns, measured through the use of self-assessment and direct observation, provide inferential support for our hypothesis, even though this study did not employ direct neuroimaging methods like fMRI.
Oxytocin as a Modulator of Emotional and Physiological Responses
The hormone Oxytocin, a neuropeptide released during social engagement, is another likely contributor to the tearing mechanisms in humans20,21,22. Previous research has identified oxytocin receptors not only in some brain regions (the prefrontal cortex and amygdala) but also in the lacrimal glands, potentially indicating oxytocin’s involvement in tear secretion5,24. The increased empathy and bonding scores (tearfulness) within the bonding group supports the hypothesis that short-term social interactions may activate oxytocin-related mechanisms, enhancing emotional responsiveness. The mechanisms behind emotional tears likely extend beyond oxytocin and suggest the possibility of a broader neuroendocrine framework that can modulate emotional expression. Hormones like cortisol and vasopressin are known to regulate affective arousal, stress management, and social affiliation26. Therefore, these hormonal systems likely interact with and modify oxytocin’s overall effect on emotional responses. For example, cortisol helps regulate emotional learning caused by stress, which may indirectly affect tearful expression through stress-responsive feedback systems26. Another factor we need to consider is the fact that hormonal secretion, including oxytocin secretion, varies across diurnal rhythms, which could also change emotional sensitivity. This introduces the idea that potential physiological fluctuations need to be discussed in later investigation. By integrating these temporal and hormonal factors in future studies, we could understand oxytocin’s role more accurately, within the complex neuroendocrine system.
Limitations of the Experiment
While the findings of this study are significant, this study has several notable limitations.
- Indirect Measurement and Vulnerability of Self-Report – the study relied solely on behavioral data measured by indirect measurements. This was due to the inaccessibility of advanced physiological and neural monitoring tools (e.g., fMRI, heart rate variability, or EEG). Therefore, these findings should be seen as exploratory; they primarily show a correlation between affective empathy and emotional tearfulness, falling short of establishing a causal connection. Furthermore, oxytocin levels and neural mirroring activity were not directly quantified. Given the reliance on self-reported data, the findings could bring another inaccuracy that introduces a social desirability bias27, particularly in collectivist contexts where showing affective empathy is often highly socially valued.
- Lack of Active Control Task – while the emotionally stimulated group was participating in an 8-minute social bonding activity, we did not give any structured task to the control group. Therefore, it is highly likely that we only captured the effect of performing an activity itself rather than the social nature of the task.
- Sampling Bias – the social bonding activity may reflect existing social relationships between participants instead of establishing new connections because all students were recruited from the same school. Despite our attempts to assign groups without prior close connections, a complete exclusion of existing friendships was not feasible, which could introduce inaccurate results.
- Small Sample Size – this study cannot yet be generalized to the public due to its small sample size (n=28) as it was difficult to enroll enough participants into the study given the limited time of this high-school-level study. All statistical outcomes (e.g., p-value) should be interpreted carefully since a small sample power may reduce the dependability of the observed effects.
- Lack of Long-term Tracking – we did not incorporate sustained observation or longitudinal measures because of practical challenges. Therefore, the findings may only include temporary results, not sustained behavioral shifts.
- Cultural Homogeneity – the fact that all participants shared a similar cultural background (East Asia) may yield different results because there is a possibility of cultural differences in empathic responses when expressing emotions.
- Confounders Not Controlled – confounding factors like general mood, social facilitation, affective priming, social identity reinforcement, or general arousal were not controlled for and may have influenced the findings. Moreover, adolescent developmental factors such as hormonal fluctuations, attachment anxiety, dispositional empathy, and alexithymia may have contributed to variability in emotional reactivity.
- Lack of Stratified Randomization – we did not employ block or stratified randomization, which could introduce minor imbalances in gender or baseline empathy between groups.
Future Directions
Various future directions are suggested based on the improvements derived from the limitations raised in Limitations of the Experiment.
- Future research should employ objective physiological indicators (e.g., EEG, fMRI, measuring tear volume, pupillometry, or hormonal assays) to formally test indirect effects and better understand the psychological processes. Furthermore, there is a need for the use of blind observation and anonymized reporting to minimize social desirability bias.
- Future studies should assign a non-social but equally engaging task (e.g., solving a puzzle or neutral shared video discussion) to the control group to more clearly differentiate the social component from other components of the bonding effect.
- To strengthen the robustness of the findings, future studies may benefit from larger, more representative samples.
- Future research using sustained observation or longitudinal measures, such as long-term volunteering, are needed to clarify how social interactions affect tear production.
- By exploring the effects of social bonding activities with unfamiliar individuals, future studies may provide more accurate perspectives on the impact of social interaction.
- Replicating these analyses in samples by using cross-cultural groups would provide whether the observed patterns hold true across different cultures.
- Other confounding factors such as general emotional arousal or social desirability effects could be considered as another explanation of increased tear production. In future studies, it should be isolated from the fundamental mechanisms of tear production through physiological assessments (e.g., EEG, fMRI, or heart variability).
- To balance gender or baseline empathy, future studies should employ block or stratified randomization and improve their validity.
- Future studies should examine how not only behavioral expression but also biochemical processes can reflect emotional tearfulness. In recent studies, researchers have discovered that tear fluid carries bioactive molecules such as lysozyme and lactoferrin28,29. They suggest the possibilities that emotional arousal or social connectedness could affect the concentration of these substances28,29. Therefore, future studies including these tear biomarkers could offer direct biochemical evidence connecting emotional crying to the physiological and socio-affective mechanisms.
Acknowledgements
I would like to give thanks to my parents for their constant support and everyone who participated in this study.
References
- A. J. J. M. Vingerhoets. Why only humans weep: unraveling the mysteries of tears. Oxford University Press (2013). [↩]
- L. M. Bylsma, A. Gračanin, A. J. J. M. Vingerhoets. The neurobiology of human crying. Clinical Autonomic Research 28, 63-80 (2018). [↩] [↩] [↩]
- J. E. LeDoux. Emotion circuits in the brain. Annual Review of Neuroscience 23, 155-184 (2000). [↩]
- F. C. Murphy, I. Nimmo-Smith, A. D. Lawrence. Functional neuroanatomy of emotions: a meta-analysis. Cognitive, Affective, & Behavioral Neuroscience 3(3), 207-233 (2003). [↩]
- D. F. Hawley, T. P. DeWall, C. M. Bentley, C. T. Toth. Neurobiology of crying and oxytocin pathways. Neuroscience Letters 682, 124-130 (2018). [↩] [↩] [↩]
- M. Garriz, R. K. Varghese, H. T. Shimada, A. Patel, N. S. Malik. Role of the plc pathway in emotional expression. Neuroscience Research 168, 88-97 (2021). [↩]
- M. C. P. Hendriks, A. J. J. M. Vingerhoets. Social messages of crying faces. Social Behavior and Personality 34(4), 377-386 (2006). [↩]
- V. Gallese. The roots of empathy: the shared manifold hypothesis and the neural basis of intersubjectivity. Psychopathology 36, 171-180 (2003). [↩] [↩] [↩] [↩] [↩]
- B. Wicker, C. Keysers, J. Plailly, J. P. Royet, V. Gallese, C. Frith. Both of us disgusted in my insula: the common neural basis of seeing and feeling disgust. Neuron 40(3), 655-664 (2003). [↩] [↩] [↩] [↩]
- G. di Pellegrino, L. Fadiga, L. Fogassi, V. Gallese, G. Rizzolatti. Understanding motor events: a neurophysiological study. Experimental Brain Research 91, 176-180 (1992). [↩] [↩] [↩]
- V. Gallese, L. Fadiga, L. Fogassi, G. Rizzolatti. Action recognition in the premotor cortex. Brain 119(2), 593-609 (1996). [↩] [↩]
- G. Rizzolatti, C. Sinigaglia. The functional role of the parieto-frontal mirror circuit: interpretations and misinterpretations. Nature Reviews Neuroscience 11(4), 264-274 (2010). [↩]
- V. Gallese. The ‘shared manifold’ hypothesis: from mirror neurons to empathy. Journal of Consciousness Studies 8(5-7), 33-50 (2001). [↩]
- V. Gallese. Embodied simulation theory: imagination and narrative. Neuropsychoanalysis 13(2), 196-200 (2011). [↩]
- G. Hickok. Eight problems for the mirror neuron theory of action understanding in monkeys and humans. Journal of Cognitive Neuroscience 21(7), 1229-1243 (2009). [↩]
- A. Lingnau, B. Gesierich, A. Caramazza. Asymmetric fMRI adaptation reveals no evidence for mirror neurons in humans. Proceedings of the National Academy of Sciences of the United States of America 106(24), 9925-9930 (2009). [↩]
- M. Iacoboni, I. Molnar-Szakacs, V. Gallese, G. Rizzolatti. Cortical mechanisms of human imitation. Science 286(5449), 2526-2528 (1999). [↩]
- G. Gimpl, F. Fahrenholz. The oxytocin receptor system: structure, function, and regulation. Physiological Reviews 81(2), 629-683 (2001). [↩]
- K. Uvnäs Moberg, M. Handlin, L. Petersson. Maternal plasma levels of oxytocin during breastfeeding – a systematic review. Psychoneuroendocrinology 121, 104843 (2020). [↩]
- H. J. Lee, A. H. Macbeth, J. H. Pagani, W. S. Young. Oxytocin: the great facilitator of life. Progress in Neurobiology 88(2), 127-151 (2009). [↩] [↩]
- C. S. Carter. Neuroendocrine perspectives on social attachment and love. Psychoneuroendocrinology 23(8), 779-818 (1998). [↩] [↩]
- R. Feldman. Oxytocin and social affiliation in humans. Hormones and Behavior 61(3), 380-391 (2012). [↩] [↩] [↩]
- N. L. Ostrowski. Oxytocin receptor mRNA expression in the rat brain: implications for behavioral roles. The Journal of Comparative Neurology 398(2), 206-220 (1998). [↩]
- H. E. Ross, M. L. Cole, L. J. Smith. Variation in oxytocin receptor density in the nucleus accumbens. The Journal of Neuroscience 29(5), 1312-1321 (2009). [↩] [↩]
- M. Iacoboni. Imitation, empathy, and mirror neurons. Annual Review of Psychology 60, 653-670 (2009). [↩]
- V. A. van Ast, S. Cornelisse, M. F. Marin, S. Ackermann, S. N. Garfinkel, H. C. Abercrombie. Modulatory mechanisms of cortisol effects on emotional learning and memory: novel perspectives. Psychoneuroendocrinology 38(9), 1874–82 (2013). [↩] [↩]
- D. P. Crowne, D. Marlowe. A new scale of social desirability independent of psychopathology. Journal of Consulting Psychology 24(4), 349-354 (1960). [↩]
- H. G. Hanstock, J. P. Edwards, N. P. Walsh. Tear lactoferrin and lysozyme as clinically relevant biomarkers of mucosal immune competence. Frontiers in Immunology 10, 1558 (2019). [↩] [↩]
- J. Peng, D. Feinstein, S. DeSimone, P. Gentile. A review of the tear film biomarkers used to diagnose sjogren’s syndrome. International Journal of Molecular Sciences 25(19), 10380 (2024). [↩] [↩]