Abstract
The efficiency of in vitro micropropagation of Vitis vinifera is crucial for clonal expansion and germplasm preservation. However, the effects of varying Murashige and Skoog (MS) salt concentrations on the growth and viability of softwood grapevine explants have yet to be fully quantified. To this end, this study aims to evaluate various MS salt strengths on the success of producing Vitis vinifera ‘Thompson Seedless’ explants and to identify nutrient conditions that achieve optimal growth while maintaining long-term health. Softwood shoots were first harvested, surface-sterilized, and cultured in vitro under controlled environmental conditions. Explants were grown on media with concentrations of 0 MS, ½ MS, 1 MS, and 2 MS. After a two-week period for growth, each culture was evaluated using metrics including shoot proliferation, leaf development, and tissue viability. 0 MS retained limited growth with moderate survival. ½ MS maintained healthy shoot multiplication with consistent viability and minimal tissue stress. 1 MS displayed a balance between growth rate and survivability. 2 MS underwent rapid early growth and increased size among surviving explants, but this was accompanied by the highest level of necrosis and the lowest overall shoot proliferation rate, resulting in a reduction of overall propagation efficiency. Thus, the findings of this study demonstrate that successful micropropagation of softwood grapevine explants is contingent upon an appropriate nutrient balance rather than maximal MS salt concentration. Reduced-strength media remain better suited for in vitro culture across an extended period of time. Future studies evaluating “cycling” between MS concentrations would help to refine protocols regarding grapevine micropropagation.
Keywords: Murashige and Skoog; salt concentration; Vitis vinifera; in vitro micropropagation; tissue viability
Introduction
Vitis vinifera, the common grapevine, is a fruit crop grown for human consumption, wine production, and other economic uses1,2. This plant has many uses in food, cosmetic, and pharmaceutical industries3,4. Extracts from Vitis vinifera have antioxidant and antimicrobial properties, which are attributed to their phytochemical composition5,6,7. Given its commercial value and the need to preserve specific genetic characteristics, in vitro plant tissue propagation is used to multiply these plants8,9. In vitro propagation allows for crop improvement through genetic editing and enables the production of secondary metabolites, which are used for industries including food and pharmaceuticals, allowing independence from external environmental factors10,4,11. Conventional propagation methods typically use woody cuttings in order to reproduce plants. However, conventional propagation methods are constrained by seasonal factors and the risk of disease transmission12,13. In order to address these issues, the biotechnological approach of plant tissue culture has emerged as a way to allow for the mass production of disease-free plants9,14. Plant tissue culture involves growing harvested plant cells or tissues (known as explants) in a controlled and sterile environment, with these plant cells or cuttings placed on an artificial nutrient medium that has a specific composition15. This micropropagation technique enables the clonal propagation of a plant from small tissue samples, allowing for consistent production quality, independence from seasonal and geographic variations, and shorter growth cycles9,16.
The Murashige and Skoog medium is a standard nutrient formulation for plant tissue culture that provides essential inorganic salts, a carbon source, vitamins, and a mix of plant hormones and growth regulators17. Plant growth regulators are vital to directing plant development during tissue culture as they influence root proliferation, organogenesis, and shoot development18,19. Auxins such as IAA, IBA, and NAA all promote root and callus formation. Cytokinins such as BAP and Kinetin encourage shoot formation and cell division20. Whether or not an in vitro culture is successful depends on the correct concentrations of these regulators, which vary depending on the plant and depending on the morphology of the given plant tissue21,22. As a whole, optimization of culture media for Vitis vinifera growth and other plants has been an ongoing research focus23,24,25.
While general protocols for Vitis vinifera micropropagation have been established, most studies focus on hardwood or dormant tissues cultured in Woody Plant Medium (WPM), which was specifically formulated for woody species and is favored for its lower salt content and reduced incidence of hyperhydricity26. In contrast, Murashige and Skoog (MS) medium, characterized by higher nutrient and nitrogen content, is more commonly employed for herbaceous tissues and actively growing softwood material, where rapid cell division and shoot multiplication are prioritized27. Despite its widespread use, MS medium is often applied at full or reduced strength without systematic evaluation of how salt concentration influences growth-stress tradeoffs in softwood grapevine explants28. For commercially important table grape cultivars such as Vitis vinifera ‘Thompson Seedless,’ the concentration-dependent effects of MS salts on softwood shoot micropropagation remain under-characterized3. Existing studies frequently report successful regeneration under specific media formulations or hormone regimes but rarely assess how incremental changes in MS salt strength alter shoot proliferation, leaf development, and tissue viability within a unified experimental framework27,29. As a result, the threshold at which MS salts transition from promoting rapid growth to inducing physiological stress in softwood grapevine tissue has not been quantified. The objective of this study was to systematically evaluate the dose-dependent effects of MS salt concentration (0, ½, 1, and 2 MS) on shoot proliferation, leaf production, and tissue viability in Vitis vinifera ‘Thompson Seedless’ softwood explants under standardized in vitro conditions. We tested whether intermediate MS concentrations would optimize the balance between rapid multiplication and tissue health, and whether elevated salt strength would elicit measurable stress responses. Defining these concentration-specific effects is essential for developing efficient MS-based propagation strategies for genetic modification, germplasm preservation, and rapid clonal multiplication of this economically significant cultivar, particularly in contexts where accelerated growth is prioritized over long-term maintenance.
Methods
Explant Collection and Initial Sterilization
Vitis vinifera ‘Thompson Seedless’ explants were harvested from actively growing grapevines in a 9b/10a USDA hardiness zone location in California during the months of June and July. Single-node segments, approximately 10 cm in length, were excised from the actively-growing ends of current-season softwood vines. Each nodal segment included one axillary bud with minimal leaf tissue retained. Explants were immediately transported to the laboratory and stored at 4°C for no more than 24 hours prior to sterilization. Initial sterilization was performed by subjecting shoots to continuous water flow with laboratory-grade detergent for 2 hours, followed by sequential rinsing with tap water (5×) and deionized water (5×). Surface sterilization was achieved using 1% sodium hypochlorite solution with manual agitation for 20-30 minutes under aluminum foil coverage. The sterilized specimens were transferred to a laminar flow hood following external decontamination with 70% ethanol, where they underwent five sequential rinses with sterile deionized water15,30.
Media Preparation
Varying concentrations of Plant Cell Technology’s Modification 9 Murashige & Skoog Basal Salts were prepared (0 MS, ½ MS, 1 MS, and 2 MS) and supplemented with a constant set of ingredients. For each liter of media, constant ingredients included 30.0 g sucrose, 10.0 g agar, 1.0 mg BAP, and 1.0 mg IBA21,31. BAP and IBA were respectively prepared as 1.0 mg/mL and 0.1 mg/mL stock solutions in DMSO (dimethyl sulfoxide), and 1.0 mL of each stock solution was added to the media prior to autoclaving.
The tested MS concentrations were 0.000 g/L (0 MS), 2.215 g/L (½ MS), 4.430 g/L (1 MS), and 8.860 g/L (2 MS)25,32. All ingredients were combined with deionized water and mixed using a magnetic stirrer. pH was adjusted to 5.8 ± 0.2 using 1.0 and 0.1 N HCl and NaOH solutions. The prepared media was autoclaved at 121°C at 18-20 PSI for a 20-minute period. Prior to autoclaving, media containers were wrapped in aluminum foil and indicator tape. The laminar flow hood was sterilized via 20-minute UV light and wiping down with 70% ethanol. Following autoclave sterilization, containers were transferred to the laminar flow hood and then poured into autoclaved flasks under aseptic conditions and labeled. Media solidified within 24 hours and was stored at room temperature for up to 3 days to monitor for potential contamination before use in culturing.
| Supplemented Ingredients (constant across 0, ½, 1, and 2 MS concentration flasks) | Amount for creation of 1 Liter of MS Media |
| Deionized Water | 1.0 L |
| Sucrose | 3% (30.0 g L -1) |
| Agar | 1% (10.0 g L -1) |
| 6-Benzylaminopurine (BAP) | 4.41 μM (1 mg L -1) |
| Indole-3-butyric Acid (IBA) | 4.92 μM (1 mg L -1) |
| Type of Media | Concentration of MS Powder in g/L |
| 0 MS Media | 0.000 g/L of DI water |
| ½ MS Media | 2.215 g/L of DI water |
| 1 MS Media | 4.430 g/L of DI water |
| 2 MS Media | 8.860 g/L of DI water |
Flow Hood Culture Preparation
All tools were wrapped and sterilized by autoclaving before use. Prior to placement in the laminar flow hood, tools and their aluminum foil covering were surface sterilized with 70% ethanol. Within the hood, tools underwent additional sterilization using a Bunsen burner and ethanol, with particular attention paid to surfaces that would contact plant tissue. A sterilized tool stand was used to hold implements when not in use. During culture work, tools were re-sterilized between each task by immersion in pure ethanol for 10 seconds followed by flame sterilization for 10-15 seconds29,33.
Culture Protocol
For initial cultures, sterilized explants were transferred to sterile petri dishes in the laminar flow hood. For reculturing, existing shoots were removed from their previous media into sterile petri dishes. All flasks were surface sterilized with 70% ethanol before placement in the hood. Flask openings were briefly flame-sterilized upon opening. Dead tissue and leaves were removed using sterile instruments, and small sections were trimmed from shoot ends to further prevent infection. For recultures, new growth was separated as it was not included in the main shoot measurements. Each flask contained 4-6 shoots; to account for this variation, shoot number was normalized across flasks by equalizing both total biomass and the number of apical buds present per shoot, ensuring comparable developmental potential across all experimental units regardless of absolute shoot count. Processed shoots were collected in a secondary sterile petri dish until all material from the original flask was prepared. For recultures, shoots were transferred to new media matching their previous MS concentration. Initial cultures were randomly assigned to different MS concentrations. Flasks were sealed with vented caps containing gas-permeable membrane filters to allow gas exchange while maintaining sterility. Cap edges were sealed with parafilm to prevent contamination through the cap-flask junction. Flasks were labeled to track dates before being placed in the greenhouse.
Storage Maintenance
All cultures were maintained in a greenhouse under controlled conditions. Temperature was maintained at 25°C ± 2°C with a 16/8 hour photoperiod light regimen. Flasks were positioned under light banks and monitored regularly for contamination or signs of stress. Flasks exhibiting visible microbial contamination were excluded immediately upon detection to prevent cross-contamination, due to the fact that pathogen stress can confound normal developmental responses31.
Data Collection and Analysis
Data were collected from multiple independent culture trials conducted over an 8-week period. Each trial consisted of 2 weeks of culture, with each trial initiated at staggered time points to account for temporal variation. Data from all trials were pooled for analysis, with each flask serving as the experimental unit (total N=43 flasks: 0 MS n=19, ½ MS n=9, 1 MS n=9, 2 MS n=6). These sample sizes reflect flasks that remained uncontaminated throughout the culture period. Flasks that exhibited visible microbial contamination were excluded upon detection and are not represented in the final counts, because infection artificially accelerates plant decay and stunts growth, which would prevent an accurate assessment of the experimental variables. While contaminated flasks were removed to ensure data integrity, flasks exhibiting abiotic necrosis were retained in the final analysis to accurately reflect the physiological response to the treatment conditions. For each of the 4 concentration groups, we evaluated relative performance using 3 metrics: shoot proliferation rate, leaf production rate, and necrosis rate. Shoot proliferation rate can be defined as the number of new shoots divided by the number of original shoots, and this metric gauges the efficiency of new shoot generation per initial explant. Leaf production rate can be defined as the number of leaves observed divided by the number of original shoots, and this metric measures overall growth and photosynthetic potential per initial explant. Necrosis rate can be defined as the number of dead shoots divided by the number of original shoots, and this metric measures the rate of treatment-induced shoot mortality. The average (mean) and standard deviation (SD) were calculated for each respective metric. However, prior to group comparisons, assumptions of normality and homogeneity of variance were assessed using Shapiro-Wilk tests and Levene’s and Brown-Forsythe tests, respectively. For shoot proliferation and leaf production, one-way analysis of variance (ANOVA) with Tukey’s honestly significant difference (HSD) post-hoc tests was used, with Welch’s ANOVA conducted as a robustness check. For necrosis, where variance heterogeneity was detected, Welch’s ANOVA with Games-Howell post-hoc comparisons was applied. All statistical analyses were conducted in Python (version 3.10.9) using the NumPy (1.23.2), SciPy (1.9.0), and statsmodels (0.14.0) libraries, with significance evaluated at α=0.05.
Results
Three response variables were analyzed to assess culture performance across MS salt concentrations (0, ½, 1, and 2): shoot proliferation rate (new shoots per original shoot), leaf production rate (leaves per original shoot), and necrosis rate (dead shoots per original shoot). Together, these metrics capture vegetative multiplication efficiency, leaf development, and explant tissue health at the flask level, which served as the experimental unit.
For shoot proliferation and leaf production, group variances were sufficiently homogeneous to justify the use of standard one-way ANOVA followed by Tukey’s HSD for pairwise comparisons; results were consistent with variance-robust alternatives. In contrast, necrosis exhibited pronounced variance heterogeneity and was therefore analyzed using Welch’s ANOVA with Games-Howell post-hoc tests. Detailed statistical outcomes for each response variable are presented below.
Shoot Proliferation
Shoot proliferation differed significantly among MS salt concentrations (Figure 1). A one-way ANOVA revealed a significant effect of treatment on proliferation rate, F(3, 39)=4.72, p=0.0066, η²=0.266. Post-hoc Tukey HSD comparisons showed that explants cultured on ½ MS medium exhibited significantly higher shoot proliferation than those grown on 0 MS (mean difference=0.32, 95% CI [0.03, 0.61], p=0.027) and 2 MS medium (mean difference=0.40, 95% CI [0.02, 0.78], p=0.038). No other pairwise comparisons were statistically significant.
Because variance homogeneity was slightly violated, Welch’s ANOVA was performed to assess the robustness of the result. Welch’s test also indicated a significant effect of MS concentration on shoot proliferation, Fw(3, 15.19)=7.53, p=0.0026, confirming the overall ANOVA findings.
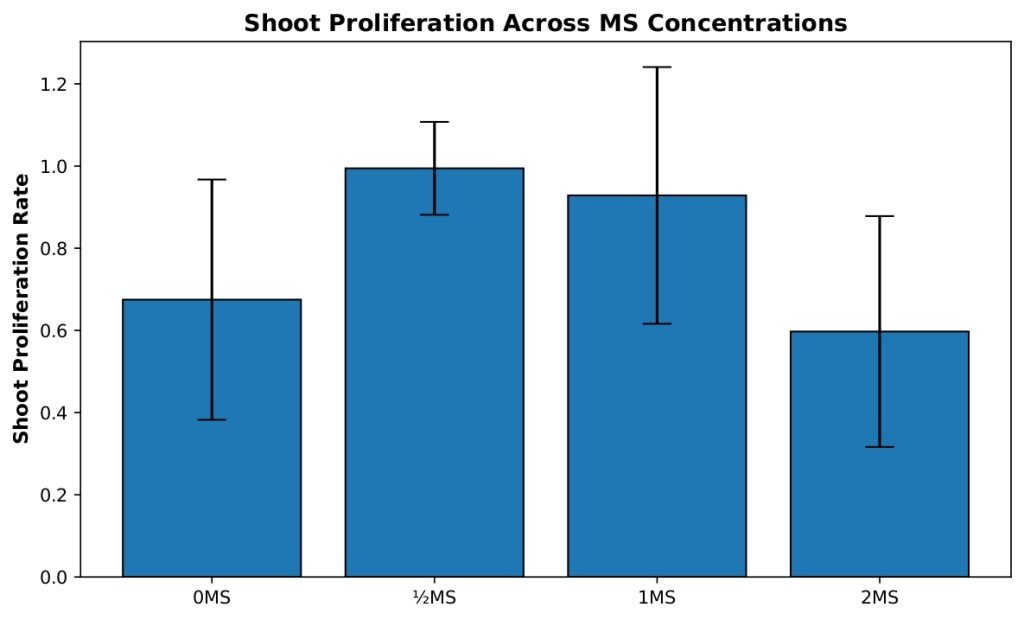
The 0 MS treatment group (n=19) exhibited a mean proliferation rate of 0.675, indicating that, on average, shoots produced approximately two-thirds of a new shoot per original shoot. This group showed moderate variability with a standard deviation of 0.292 and a standard error of 0.067. The ½ MS concentration (n=9) demonstrated the highest proliferation rate with a mean of 0.994, nearly achieving one new shoot per original shoot, and showed the most consistent results with the lowest standard deviation of 0.113 and standard error of 0.038. The 1 MS treatment (n=9) displayed a mean of 0.929, performing nearly as well as ½ MS but with considerably higher variability (SD=0.312, SE=0.104), suggesting more heterogeneous responses among replicates. The 2 MS concentration (n=6) showed the poorest performance with a mean of 0.597, producing fewer than two-thirds of a new shoot per original shoot, with moderate variability (SD=0.281, SE=0.115). These results suggest that ½ MS concentration provides optimal conditions for shoot proliferation with the most reliable outcomes.
Leaf Production
Leaf production varied across MS salt concentrations (Figure 2), but these differences were not statistically significant. A one-way ANOVA did not find a significant effect of MS concentration on leaf growth, F(3, 39)=2.35, p=0.087, η²=0.153. Accordingly, none of the Tukey HSD pairwise comparisons reached significance at α=0.05. The largest observed difference occurred between the 0 MS and 1 MS treatments (mean difference=1.62 leaves, 95% CI [−0.13, 3.36], p=0.078), but this did not meet the threshold for significance.
Welch’s ANOVA was also performed and similarly indicated no significant effect of MS concentration on leaf production, Fw(3, 15.31)=2.29, p=0.119, confirming the non-significant outcome of the standard ANOVA.
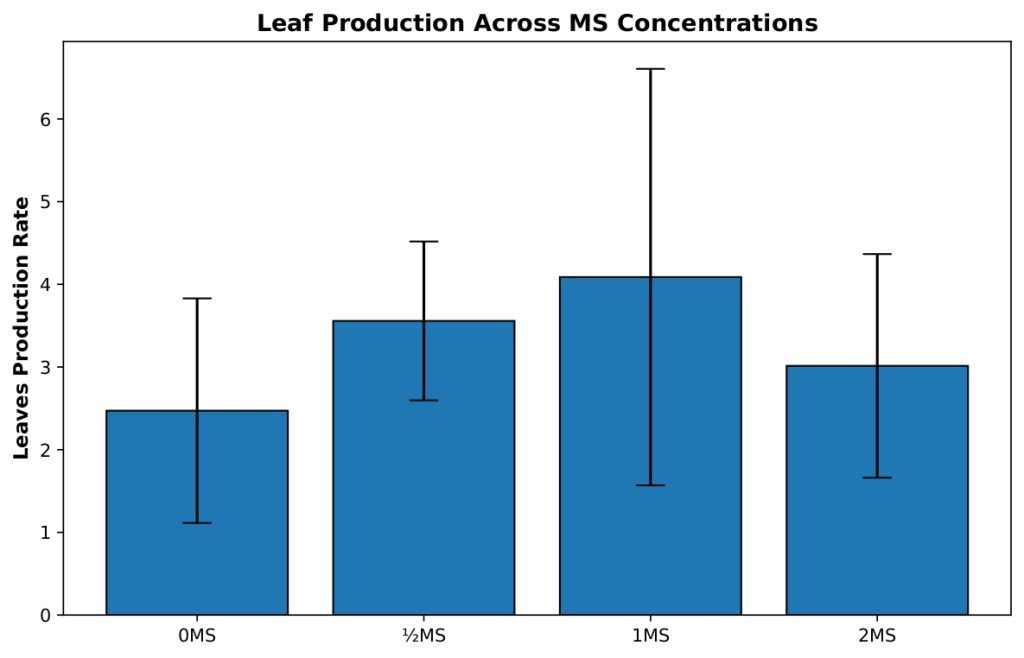
The 0 MS group (n=19) produced a mean of 2.472 leaves per original shoot with considerable variability (SD=1.358, SE=0.312), indicating inconsistent leaf development in the absence of MS nutrients. The ½ MS treatment (n=9) enhanced leaf production to a mean of 3.557 leaves per shoot with reduced variability (SD=0.961, SE=0.320), demonstrating more uniform and improved leaf development. The 1 MS concentration (n=9) achieved the highest mean leaf production at 4.087 leaves per shoot, representing a 65% increase over the 0 MS control; however, this treatment exhibited the greatest variability among all groups (SD=2.519, SE=0.840), suggesting that while some replicates responded very favorably, others did not. The 2 MS treatment (n=6) showed a mean of 3.014 leaves per shoot with moderate variability (SD=1.352, SE=0.552), performing better than the control but substantially lower than the 1 MS treatment. These findings indicate that while 1 MS maximizes leaf production potential, ½ MS provides a more consistent and reliable response, making it potentially more suitable for standardized propagation protocols.
Necrosis
Necrosis differed markedly across MS salt concentrations (Figure 3). Because variance heterogeneity was detected for this metric (Brown-Forsythe test, p < 0.05), Welch’s ANOVA with Games-Howell post-hoc comparisons was used as the primary analytical approach. Welch’s ANOVA revealed a highly significant effect of MS concentration on necrosis, Fw(3, 15.70)=14.34, p < 0.001.
Games-Howell post-hoc analyses showed that the 2 MS treatment resulted in significantly higher necrosis than the ½ MS (p < 0.001), 1 MS (p=0.022), and 0 MS (p=0.049) treatments. In addition, the 0 MS group exhibited significantly greater necrosis than the ½ MS treatment (p < 0.001). The difference between the ½ MS and 1 MS treatments approached, but did not reach, statistical significance (p=0.063). Together, these comparisons indicate a strong increase in necrosis at higher MS concentrations, with the lowest necrosis observed under reduced-strength medium.
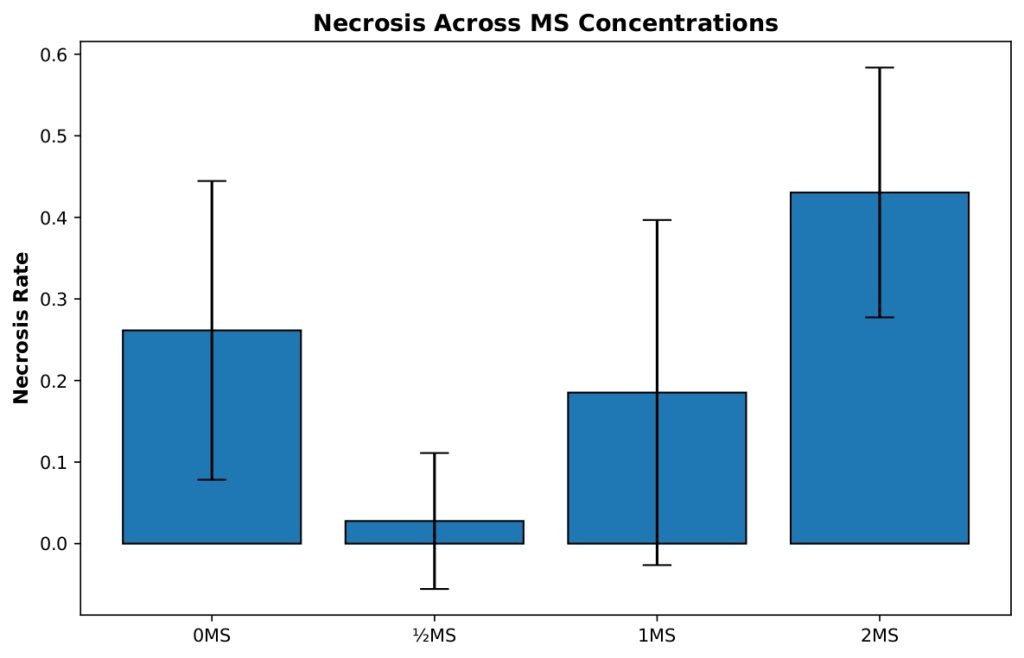
The 0 MS control group (n=19) displayed a mean necrosis rate of 0.261, indicating that approximately one-quarter of shoots experienced tissue death, with moderate variability (SD=0.183, SE=0.042). The ½ MS treatment (n=9) demonstrated remarkably low necrosis with a mean of only 0.028, representing an almost ten-fold reduction compared to the control, and showed low variability (SD=0.083, SE=0.028), with most replicates experiencing minimal to no tissue death. The 1 MS concentration (n=9) exhibited an intermediate necrosis rate with a mean of 0.185, performing better than the control but substantially higher than ½ MS, with moderate variability (SD=0.212, SE=0.071). Most notably, the 2 MS treatment (n=6) showed the highest necrosis rate with a mean of 0.431, indicating that nearly half of the shoots experienced tissue death, with moderate variability (SD=0.153, SE=0.062). This pattern clearly demonstrates that excessive MS concentration is detrimental to tissue health, while ½ MS provides optimal conditions for minimizing necrosis and maintaining healthy shoot cultures.
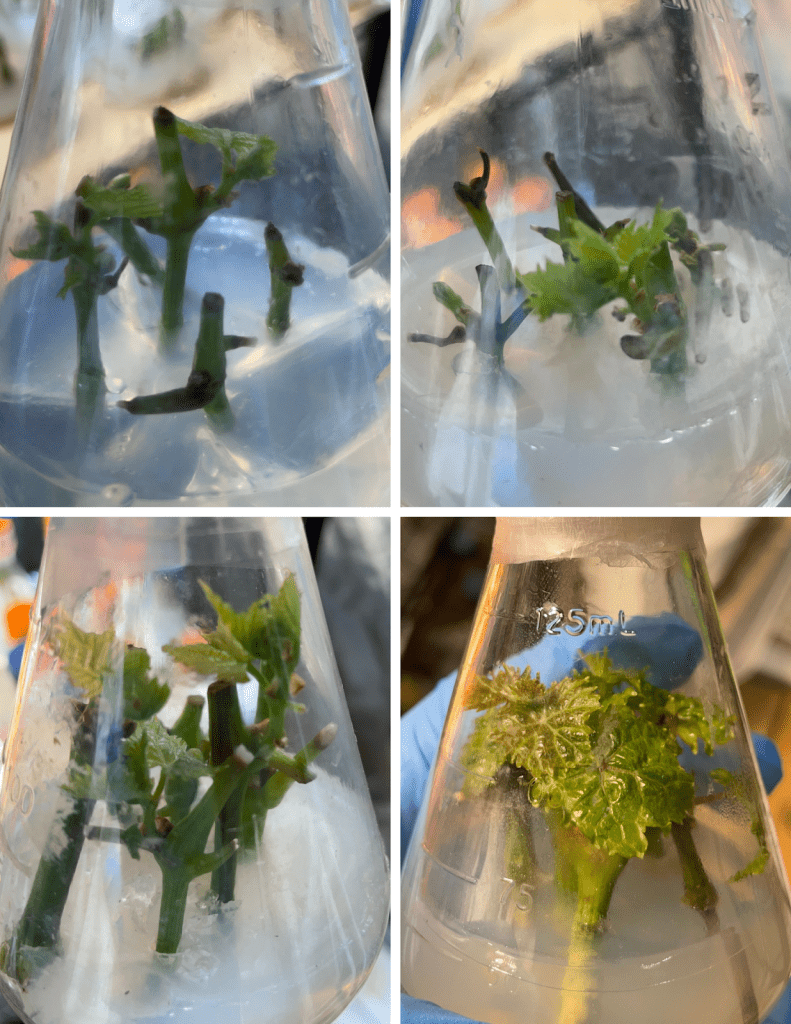
Discussion
Growth Response Patterns Across MS Concentrations
Growth responses across MS concentrations revealed a clear tradeoff between rapid biomass accumulation and tissue viability in softwood Vitis vinifera explants. Reduced-strength MS (½ MS) supported efficient shoot proliferation with minimal necrosis, indicating favorable conditions for sustained propagation. Full-strength MS (1 MS) produced comparable shoot and leaf development while maintaining overall tissue health, suggesting a balance between nutrient availability and physiological tolerance. In contrast, elevated MS concentration (2 MS) promoted rapid early growth in surviving explants but was consistently associated with increased necrosis and reduced shoot proliferation, indicating the onset of nutrient-induced stress. Together, these results demonstrate that increasing MS salt strength does not linearly improve propagation performance and that intermediate concentrations provide the most reliable outcomes for softwood grapevine culture.
Comparison with Previous Research
Our findings align with established patterns in prior plant tissue culture research. Asmono, Rahmawati & Sjamsijah (2021) found similar trends with Stevia rebaudiana, where ¼ MS plantlet length was lower than ½ MS and full MS concentrations, though they observed no statistically significant difference between ½ MS and full MS growth rates34. Our study revealed more pronounced visual differences in biomass between ½ MS, 1 MS, and 2 MS concentrations in Vitis vinifera, though quantitative analyses showed that apparent size increases in 2 MS were offset by elevated necrosis. Similarly, Shohael, Khatun, Alam & Paek (2013) reported comparable results with Eleutherococcus senticosus embryos in bioreactors. After 45 days, 1 MS produced the highest biomass, while 2 MS showed no significant advantage over ½ MS concentrations28. This pattern suggests that the “fall-off” in 2 MS performance observed in our study may represent a general response to nutrient overload across different species. Beyond cultivar-dependent differences, several studies have also emphasized how the ionic composition and osmotic potential of MS medium directly shape morphogenic outcomes. Mederos-Molina reported that adjusting nitrogen source ratios significantly altered shoot vigor in Vitis vinifera, paralleling our observation that elevated total salts accelerated early biomass but reduced long-term viability35. Similarly, Mozafari et al. demonstrated that full-strength MS supplemented with benzyl adenine enhanced initial shoot induction but ultimately promoted vitrification, again mirroring the short-term–long-term tradeoff seen here36. Notably, no hyperhydricity (vitrification) was observed in any treatment in the present study, suggesting that the tested MS concentrations remained within physiologically tolerable ranges for softwood explants during the 2-week culture period. Later formulations, such as the WPBS medium by Dalton, confirmed that re-balancing macronutrients and reducing ammonium toxicity could sustain longer morphogenic potential. Together, these reports reinforce the conclusion that nutrient balance, rather than absolute strength, governs growth in in vitro tissue culture23,37.
Affirmative Findings and Practical Applications
Explants cultured in 2 MS media for the initial 2-week period and subsequently transferred to ½ MS media appeared to retain their initial biomass while avoiding stress responses and necrosis. This observation supports the idea that higher concentration media can be used for rapid, initial-growth phases, followed by transfer to lower concentrations (½ MS) for more stable, long-term maintenance. Adopting this “cycling” approach could potentially maximize explant biomass accumulation while avoiding the detrimental effects of prolonged nutrient stress.
Study Limitations and Future Direction
While this study provides robust quantitative data on Vitis vinifera growth, several limitations must be acknowledged. First, contamination was assessed qualitatively by visual inspection, and infected flasks were discarded upon detection. A Fisher’s Exact Test indicated a statistical association between treatment and observed contamination (exact p=0.0004), but this result should be interpreted cautiously and considered exploratory. The assessment was not blinded to treatment group, cultures were not subjected to microbial isolation or molecular identification, and additional uncontrolled factors (such as variation in initial endophyte load among explants, minor handling or sterilization inconsistencies, or stochastic contamination events) could have contributed to the observed pattern. Although redefining the flask as the experimental unit corrected for pseudoreplication, contamination disproportionately reduced sample sizes at higher MS concentrations (e.g., n=6 for 2 MS). Because MS concentration was the only experimentally manipulated variable, it is the only factor that can be statistically associated with differential contamination; however, the available evidence does not establish a causal mechanism. Future studies should increase initial replication, implement blinded and semi-quantitative contamination scoring, and include targeted microbial assays or controlled inoculation experiments to determine whether and how MS salt concentration influences contaminant survival or proliferation.
Although increasing MS concentration was associated with greater apparent explant size and leaf expansion (Figure 4), these visual differences did not correspond to improved propagation performance: quantitative analyses (Figures 1-3) show that higher MS, particularly 2 MS, was accompanied by elevated necrosis rates and reduced shoot proliferation. Consequently, the larger tissues visible in some 2 MS flasks reflect growth among a smaller subset of surviving explants rather than overall culture health or superior propagation. It should be noted that Figure 4 lacks scale bars, meaning these visual size comparisons cannot be objectively verified and should therefore be interpreted with caution as qualitative observations only. Biomass assessments were qualitative and based on visual observation rather than quantitative measurement. While 2 MS cultures often appeared to produce larger individual shoots and leaves, we did not collect fresh weight, dry weight, or morphometric data (e.g., shoot length, leaf area). As a result, we cannot definitively quantify whether the apparent size increase in 2 MS compensated for the significantly higher necrosis rate observed in this treatment. Future work should incorporate standardized biomass measurements (fresh/dry weight, shoot length, leaf area) to comprehensively characterize growth-stress tradeoffs across MS concentrations.
Implications for Vitis vinifera Micropropagation
The results establish important groundwork for optimizing Vitis vinifera micropropagation protocols. The identification of concentration-specific growth/stress responses provides a foundation for developing targeted propagation strategies. For commercial applications, ½ MS appears optimal for routine maintenance cultures, while 1 MS offers the best balance of growth and sustainability.
An unexpected preliminary observation was that shoots transferred from 2 MS to ½ MS after two weeks appeared to retain their enhanced biomass while avoiding further necrosis, suggesting that cycling shoots from high to low strength MS may accelerate short-term growth while reducing the risk of later tissue health decline. While this observation was not part of our experimental design and lacks statistical validation, it suggests a potential avenue for future research into sequential medium strategies. Dedicated experiments with appropriate controls, replication, and quantitative biomass measurements would be required to test this hypothesis.
Conclusion
To conclude, ANOVA revealed significant effects of MS salt concentration on shoot proliferation (F(3,39)=4.72, p=0.0066) and necrosis (Fw(3,15.70)=14.34, p < 0.001), but not on leaf production (F(3,39)=2.35, p=0.087). Post-hoc analyses demonstrated that ½ MS medium supported significantly greater shoot proliferation than both 0 MS and 2 MS, while 2 MS exhibited the highest necrosis rate. Together, these findings indicate that reduced-strength MS medium provides a more favorable balance between vegetative multiplication and tissue viability than either nutrient deprivation or nutrient excess. Overall, this study underscores the importance of nutrient balance rather than maximal nutrient strength in in vitro grapevine propagation. The results support the use of ½ MS for routine micropropagation and highlight the limitations of using visual biomass alone as an indicator of culture success. Future work incorporating quantitative biomass measurements, longer culture durations, and sequential medium strategies may further refine optimized protocols for Vitis vinifera micropropagation.
Acknowledgments
We would like to thank our advisor, Dr. Bharat Kumar Poudyal, for his continuous support and guidance throughout this project. We would also like to thank the Aspiring Scholars Directed Research Program for providing the facilities to perform this research.
References
- B. B. Bigger. Micropropagation and acclimatization of ‘Norton’ grapevine (Vitis aestivalis). (M.S. thesis, University of Nebraska) (2010). [↩]
- G. Melyan, A. Sahakyan, A. Barsegyan, K. Dangyan, N. Sahakyan, K. Sargsyan, Y. Martirosyan. Micropropagation of (Vitis vinifera L.) cultivar ‘Sev Khardji’ using biotechnological approaches and its impact on leaf quality. Functional Food Science. 4, 277-291 (2024). [↩]
- A. Giancaspro, A. Mazzeo, A. Carlomagno, A. Gadaleta, S. Somma, G. Ferrara. Optimization of an in vitro embryo rescue protocol for breeding seedless table grapes (Vitis vinifera L.) in Italy. Horticulturae. 8, 12 (2022). [↩] [↩]
- A. Hasnain, S. A. H. Naqvi, S. I. Ayesha, F. Khalid, M. Ellahi, S. Iqbal, M. Z. Hassan, A. Abbas, R. Adamski, D. Markowska, A. Baazeem, G. Mustafa, M. Moustafa, M. E. Hasan, M. M. A. Abdelhamid. Plants in vitro propagation with its applications in food, pharmaceuticals and cosmetic industries; current scenario and future approaches. Frontiers in Plant Science. 13, 1009395 (2022). [↩] [↩]
- S. Parihar, D. Sharma. A Breif Overview on Vitis vinifera. Scholars Academic Journal of Pharmacy. 10, 231-239 (2021). [↩]
- M. Sharafan, M. A. Malinowska, M. Kubicz, P. Kubica, M. P. Gémin, C. Abdallah, M. Ferrier, C. Hano, N. Giglioli-Guivarc’h, E. Sikora, A. Lanoue, A. Szopa. Shoot cultures of Vitis vinifera different cultivars as a promising innovative cosmetic raw material — Phytochemical profiling, antioxidant potential, and whitening activity. Molecules. 28, (2023). [↩]
- C. A. Espinosa-Leal, C. A. Puente-Garza, S. García-Lara. In vitro plant tissue culture: means for production of biologically active compounds. Planta. 248, (2018). [↩]
- N. V. Romadanova, M. M. Aralbayeva, A. S. Zemtsova, A. M. Alexandrova, S. Z. Kazybayeva, N. V. Mikhailenko, S. V. Kushnarenko, J. C. Bettoni. In vitro collection for the safe storage of grapevine hybrids and identification of the presence of Plasmopara viticola resistance genes. Plants. 13, (2024). [↩]
- B. Loberant, A. Altman. Micropropagation of plants. (2010). [↩] [↩] [↩]
- R. Dev, S. K. Singh, V. Dayal, K. Kumar, T. Singh. Standardization of in vitro hardening strategies for tissue cultured wine grape (Vitis vinifera L.) genotypes. International Journal of Current Microbiology and Applied Sciences. 8, 2108-2117 (2019). [↩]
- M. S. Hussain, S. Fareed, S. Ansari, M. A. Rahman, I. Z. Ahmad, M. Saeed. Current approaches toward production of secondary plant metabolites. Journal of Pharmacy and Bioallied Sciences. 4, 10-20 (2012). [↩]
- M. B. Pithiya, S. K. Sharma, M. Sharma, M. Sharma, N. Kotwal, J. Bhaderwah. Advancements and challenges in plant tissue culture: a comprehensive overview. Journal of Plant Biota. (2022). [↩]
- A. T. Sharma, M. S. Diksha Jadhav, R. R. Dachawar, D. V. Pawde, S. S. Zunzulde. Plant tissue culture: a review. IJPPR.Human. 30, (2024). [↩]
- M. G. Mullins. Applications of tissue culture to the genetic improvement of grapevines. (1990). [↩]
- F. Shan, K. Seaton. Semi-sterilized tissue culture for rapid propagation of grapevines (Vitis vinifera L.) using immature cuttings. HortScience. 49, (2014). [↩] [↩]
- L. C. Carvalho, S. Amâncio. Cutting the Gordian knot of abiotic stress in grapevine: from the test tube to climate change adaptation. Physiologia Plantarum. 165, 330-342 (2019). [↩]
- T. Murashige, F. Skoog. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant. 15, 473-497 (1962). [↩]
- Y. Long, Y. Yang, G. Pan, Y. Shen. New insights into tissue culture plant-regeneration mechanisms. Frontiers in Plant Science. 13, 926752 (2022). [↩]
- K. A. Roubelakis-Angelakis, S. B. Zivanovitc. A new culture medium for in vitro rhizogenesis of grapevine (Vitis spp.). HortScience. (1991). [↩]
- M. Mhatre, C. K. Salunkhe, P. S. Rao. Micropropagation of Vitis vinifera L: towards an improved protocol. Scientia Horticulturae. 84, 357-363 (2000). [↩]
- N. Khan, M. Ahmed, I. Hafiz, N. Abbasi, S. Ejaz, M. Anjum. Optimizing the concentrations of plant growth regulators for in vitro shoot cultures, callus induction and shoot regeneration from calluses of grapes. J. Int. Sci. Vigne Vin. (2015). [↩] [↩]
- M. Zhan. Role of plant hormones in tissue culture. 9, 1-2 (2022). [↩]
- R. Hameg, T. A. Arteta, M. Landin, P. P. Gallego, M. E. Barreal. Modeling and optimizing culture medium mineral composition for in vitro propagation of Actinidia arguta. Frontiers in Plant Science. 11, 554905 (2020). [↩] [↩]
- A. Rebrov. Influence of concentration and ratio of macro elements in culture medium on the regeneration of grape meristems in vitro. BIO Web of Conferences. 25, 05005 (2020). [↩]
- M. DOĞAN. Influence of different concentrations of Murashige and Skoog medium on multiple shoot regeneration of Staurogyne repens (Nees) Kuntze. Journal of Engineering Technology and Applied Sciences. 7, 61-67 (2022). [↩] [↩]
- S. Osama. MICROPROPAGATION OF GRAPEVINE (VITIS VINIFERA L.) CVS. RED GLOBE AND SUPERIOR. Iraqi Journal of Agricultural Sciences. (2022). [↩]
- T. S. Pedro, R. Peiró, J. Villanova, A. Olmos, C. Gisbert. In vitro propagation of Vitis vinifera L. cv. ‘Monastrell’. Electronic Journal of Biotechnology. 27, 80-83 (2017). [↩] [↩]
- A. Shohael, S. Khatun, M. Alam, K. Paek. Effects of Murashige and Skoog medium strength on germination and secondary metabolites production of Eleutherococcus senticosus somatic embryos in bioreactor. International Journal of Biosciences. 3, 155-163 (2013). [↩] [↩]
- K. Beza, F. Tileye, B. Girma. In vitro micropropagation of grape vine (Vitis vinifera L.) from nodal culture. African Journal of Biotechnology. 16, 2083-2091 (2017). [↩] [↩]
- S. H. Kim, M. Zebro, D. C. Jang, J. E. Sim, H. K. Park, K. Y. Kim, H. M. Bae, S. Tilahun, S. M. Park. Optimization of plant growth regulators for in vitro mass propagation of a disease-free ‘Shine Muscat’ grapevine cultivar. Current Issues in Molecular Biology. 45, 7721-7733 (2023). [↩]
- N. A. L. Ghasheem, R. A. Al-Saeed, L. T. Fadala. In vitro effect of sucrose concentration and type of culture medium on fungal colonies infections on Murashige and Skoog medium. Journal of Survey in Fisheries Sciences. 10, (2023). [↩] [↩]
- J. Groll. Effect of medium salt concentration on differentiation and maturation of somatic embryos of cassava (Manihot esculenta Crantz). Annals of Botany. 89, 645-648 (2002). [↩]
- W. S. Faizy, A. Z. A. Kassab Bashi, R. S. Toma. In vitro propagation of two grapes, Vitis vinifera L. cvs. Superior and Red Globe. NTU Journal of Agricultural and Veterinary Sciences. 4, 99-107 (2024). [↩]
- S. L. Asmono, S. Rahmawati, N. Sjamsijah. The effect of Murashige and Skoog (MS) modified medium and several types of auxins on the growth of stevia (Stevia rebaudiana Bertoni) in vitro. IOP Conference Series: Earth and Environmental Science. 672, 012001 (2021). [↩]
- S. Mederos-Molina. Culture medium requirements for micropropagation of Vitis vinifera. Acta Horticulturae. (2007). [↩]
- A.-A. Mozafari, O. Ghoraishi, N. Ghaderi, T. Javadi. Micropropagation of grape cultivars (Vitis vinifera L.) on different basal media supplemented with benzyl adenine. Agriculturae Conspectus Scientificus. 81, (2016). [↩]
- S. J. Dalton. A reformulation of Murashige and Skoog medium (WPBS medium) improves embryogenesis, morphogenesis and transformation efficiency in temperate and tropical grasses and cereals. Plant Cell, Tissue and Organ Culture. 141, 257-273 (2020). [↩]