Abstract
Glutathione (GSH) is one of the most important intracellular substances in mammalian cells, over 100,000 scientific studies have been published regarding its biological functions. GSH is a multifunctional molecule, it plays a central role in the antioxidant cascade, it is the key detoxifier in cells, and it can regulate the immune system. Although the importance of GSH has been extensively studied by scientists in various fields, information on how to safely and consistently increase intracellular GSH is limited. The purpose of this review is to introduce the three main functions of GSH, discuss the importance of GSH in professional athletes, and present recent studies that demonstrate the correlation between low GSH concentrations and chronic diseases. Finally, since elevated GSH concentrations have been suggested to be associated with improved human health, several potential methods to increase intracellular GSH levels—such as through chemical compounds, natural foods, and scientifically supported food supplements—will be introduced.
Keywords and Common Abbreviations: glutathione (GSH), superoxide dismutase (SOD), reactive oxygen species (ROS), antioxidant, detoxification, heavy metals, immune regulation, neurodegenerative disease, professional athletes, aging, ME-3, probiotic, Immunocal® whey protein isolate.
Introduction
Through natural biological processes in our bodies, such as digesting food, breathing air, converting fat into energy, and metabolizing alcohol and drugs, we not only gain nutrients but also produce harmful substances called free radicals. Free radicals are usually destroyed by the body’s antioxidant system, which involves the sequential work of antioxidants like superoxide dismutase (SOD), GSH, and catalase. If the system does not function properly, free radicals can trigger a chain reaction in the body, one that can damage cell membranes, block the activity of major enzymes, cause protein misfolding, attack DNA, and prevent normal cell division1. When there are too many free radicals in the cells, oxidative stress increases. Many diseases have been reported to be associated with oxidative stress, such as metabolic diseases, chronic disorders, neurodegenerative diseases, and cancer2.
Antioxidants play important role in scavenging free radicals and maintaining the cells in balanced redox state. GSH is the antioxidant that has been intensively studied.
GSH is a tripeptide composed of three amino acids, namely glutamic acid, cysteine, and glycine. Among them, cysteine is the rate-limiting factor3. Cysteine is a sulfur-containing amino acid and is considered semi-essential. The supply of cysteine comes from food or other amino acids. Glutamic acid and glycine, on the other hand, are non-essential amino acids. Under normal circumstances, they are abundant in the human body. Glutamic acid becomes conditionally essential in individuals under certain physical conditions, such as cancer patients. Glutamic acid is closely associated with mucositis, which is often observed in head and neck cancer patients during the course of radiation treatment4. The synthesis of these amino acids takes place inside almost all cells, mostly in liver and kidney cells. GSH plays a pivotal role in numerous cellular functions, such as scavenging excessive reactive oxygen species (ROS), detoxifying endogenous and exogenous substances — including free radicals and drugs — and balancing the immune system (Figure 1). After antioxidant reactions, reduced GSH is oxidized to glutathione disulfide or GSSG. GSSG is the oxidized form that results from two GSH molecules form a disulfide bridge. The GSH:GSSG ratio is commonly used as an index of cellular oxidative status5. This ratio tracks the balance between reducing and oxidizing forces and is considered more sensitive and informative than simply measuring total GSH levels6. It is often used as biomarker under various physiological conditions.
The purpose of this study is to review the important cellular functions of GSH and to present and discuss updated research on the correlations between GSH, athletic performance, and chronic diseases. Finally, potential strategies to increase intracellular GSH levels — including supplementation, pharmaceutical compounds, probiotics, dietary sources, and whey protein isolates — are investigated.
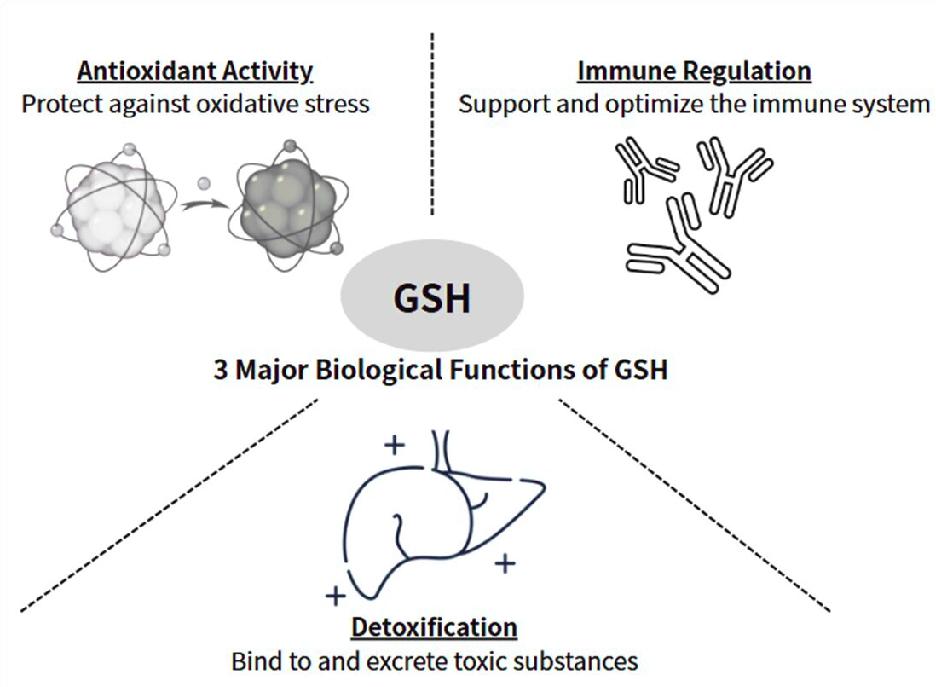
GSH as Master Antioxidant
Most life forms face challenges from ROS generated by respiration and cellular metabolism7,8,9. ROS molecules include singlet oxygen (![]() ), superoxide anions (
), superoxide anions (![]() ), hydrogen peroxides (
), hydrogen peroxides (![]() ), and hydroxyl radicals (
), and hydroxyl radicals (![]() ). These highly active species can easily interact with various components of the human body, such as lipids, proteins, and DNA, leading to numerous diseases and disorders, including cardiovascular diseases, protein denaturation, genetic mutations, and possibly an accelerated aging process10. To combat ROS, numerous antioxidants are involved, namely SOD, GSH, and catalase. Among them, GSH is known for its powerful antioxidant activity11. GSH plays a key role in neutralizing ROS and reducing oxidative stress in the cells; it’s the major player in maintaining the redox balance12. Furthermore, GSH is also capable of regenerating other antioxidants – vitamin C (ascorbic acid)13, and vitamin E (
). These highly active species can easily interact with various components of the human body, such as lipids, proteins, and DNA, leading to numerous diseases and disorders, including cardiovascular diseases, protein denaturation, genetic mutations, and possibly an accelerated aging process10. To combat ROS, numerous antioxidants are involved, namely SOD, GSH, and catalase. Among them, GSH is known for its powerful antioxidant activity11. GSH plays a key role in neutralizing ROS and reducing oxidative stress in the cells; it’s the major player in maintaining the redox balance12. Furthermore, GSH is also capable of regenerating other antioxidants – vitamin C (ascorbic acid)13, and vitamin E (![]() -tocopherol)14. These antioxidants work synergistically to maintain the body’s redox balance and defend against oxidative stress.
-tocopherol)14. These antioxidants work synergistically to maintain the body’s redox balance and defend against oxidative stress.
GSH as Key Detoxifier
We inhale and ingest natural or synthetic products that contain toxins in our everyday life; it is almost impossible to avoid them. These toxins include the chemical additives in food, cigarette smoke, air and water pollution, pesticide residues in fruits and vegetables, heavy metals and drug residues. Here is a short list of everyday toxins to which we may all be exposed (Table 1).
When these toxins accumulate in our body up to a certain level, they begin to damage our cells and organs, and various illness may occur. According to a team of biochemists – D. P. Jones, L. A. Brown and P. Sternberg from Emory School of Medicine in Atlanta – “GSH has multiple functions in detoxification, and its depletion has been associated with an increased risk of chemical toxicity”. In conclusion, they suggested that by monitoring GSH levels we can assess an individual’s risk of toxification from environmental pollutants15.
The accumulation of heavy metals in the human body is also a serious threat to our health. Cadmium, for example, is found almost everywhere including in water, fruits, cereals, seafood, tobacco, and air. It can cause damage to organs such as the gastrointestinal tract, kidneys, liver, lungs, and blood. GSH binds directly to toxins through enzymatic reactions. The sulfhydryl groups (![]() ) in the cysteine residues of GSH bind to heavy metals, such as aluminum (Al), cadmium (Cd), arsenic (As), mercury (Hg), lead (Pb), and chromium (Cr)16, forming a water-soluble GSH-toxin complex. This complex is then excreted via bile or urine. The liver and kidneys are the major organs responsible for detoxification and elimination. Laboratory animal studies have demonstrated that the liver and kidneys contain the highest levels of intracellular GSH in the body17. GSH exerts its detoxification ability by conjugating with endogenous and exogenous harmful substances. Glutathione transferase, a detoxification enzyme, catalyzes this conjugation process18. Therefore, the higher the intracellular GSH concentration, the greater the protection and the better the overall health status. Other toxins, such as the painkiller acetaminophen19, carcinogens like diethylnitrosamine20, and various other heavy metals, have been documented to be detoxified by GSH to some extent21.
) in the cysteine residues of GSH bind to heavy metals, such as aluminum (Al), cadmium (Cd), arsenic (As), mercury (Hg), lead (Pb), and chromium (Cr)16, forming a water-soluble GSH-toxin complex. This complex is then excreted via bile or urine. The liver and kidneys are the major organs responsible for detoxification and elimination. Laboratory animal studies have demonstrated that the liver and kidneys contain the highest levels of intracellular GSH in the body17. GSH exerts its detoxification ability by conjugating with endogenous and exogenous harmful substances. Glutathione transferase, a detoxification enzyme, catalyzes this conjugation process18. Therefore, the higher the intracellular GSH concentration, the greater the protection and the better the overall health status. Other toxins, such as the painkiller acetaminophen19, carcinogens like diethylnitrosamine20, and various other heavy metals, have been documented to be detoxified by GSH to some extent21.
| Toxin Name | Type/Category | Common Sources |
| Acetaminophen (Tylenol, Atasol, others) | Pharmaceutical | Over-the-counter pain reliever Toxic in high doses |
| Other pharmaceuticals (e.g., Adriamycin) | Chemotherapeutics | Used in cancer treatment Known for organ toxicity |
| Acetone | Solvent, Industrial agent | Used in cleaning agents and industry |
| Aflatoxin B₁ | Natural toxin | Moldy nuts, grains Potent carcinogen |
| Aliphatic hydrocarbons | Industrial chemicals | Vinyl chloride, hexachlorohexane Used in plastic production |
| Aromatic hydrocarbons | Solvent/fuel compounds | Benzene derivatives Found in fuels, solvents, etc. |
| Nitrosamines | Food-derived toxins | Smoked meats, preserved foods like salami and hot dogs |
| Benzopyrenes | Polycyclic aromatic hydrocarbons (PAHs) | Barbecued foods, fuel exhaust, cigarette smoke |
| Heavy metals | Metals | Lead, mercury, cadmium, cobalt, copper Toxic to multiple organs |
| Organophosphate pesticides | Pesticides | Examples: Parathion Neurotoxic agents |
| Peroxides | Reactive oxygen species (ROS) | Includes lipid and cholesterol peroxides Oxidative stress agents |
| Isothiocyanates | Naturally occurring compounds | Found in cruciferous vegetables Can be toxic in high concentrations |
| Carbamates, thiocarbamates | Pesticides/chemicals | Used in agriculture Can inhibit cholinesterase |
| Arylamines, aryl halides | Industrial chemicals | Used in dye production, plastics, and more |
| Sulfates, nitrocompounds | Industrial chemicals | Found in various manufacturing processes |
| Naphthalene | Aromatic hydrocarbon | Found in fuels, mothballs, and fuel by-products |
Reference: J. Gutman. The Comprehensive Guide to Glutathione (5th edition). GSH Books Inc. Montreal (2019).
GSH Regulates Immune Function
It is surprising that many people still believe catching a cold is caused by sitting in cold air or wandering around with wet hair. Perhaps this misconception comes from the term “catch a cold”. Another common belief is that people get sick when they are overworked, stressed out, or after excessive exercise. This belief is, in fact, closer to the truth. When people stay up all night, are under pressure, or exercise too much, their metabolism becomes imbalanced, excessive free radicals are produced, as the result that the levels of GSH are depleted. As a result, immune function becomes temporarily compromised, making them more vulnerable to viral infection.
GSH plays a crucial role in immune regulation. It is vital for the proper function of white blood cells. Studies have shown that even a slight reduction in GSH levels can lead to a notable decrease in immune system function22. The human immune system is a complex mechanism that protects the body from both endogenous and exogenous attacks. Endogenous attacks from ROS and exogenous attacks from bacteria and viruses result in inflammation. In response to inflammation, the body produces antibody, natural killer (NK) cells, macrophages, T cells, and B cells. The effectiveness of these immune response is closely associated with intracellular GSH concentrations23.
In a study published in Nature Communications in 2022, compelling evidence was provided regarding the critical role of GSH in humoral immunity, especially in B cell function and antibody production. The research demonstrated that GSH levels are essential for the proper functioning of different B cell subsets. It also showed that when GSH production is inhibited, by removing the glutamate-cysteine synthesis enzyme, antibody responses are significantly reduced. The mice in the study failed to produce adequate levels of both IgM and IgG antibodies upon immunization and viral infection24. Besides humoral immunity, GSH also exerts an importance role in cell-mediated immunity. Several studies have indicated that GSH levels are essential for T cell activation, cytokine production, antitumor activity of CD8+ T cells25, and the cytotoxicity of NK cells26.
In a recent clinical study published in 2025, it was reported that GSH supplementation enhances NK cell function and metabolic activity in patients with acute leukemia. Although the mechanism remains unclear, patients with acute leukemia are often observed to have impaired NK cell function and weakened antitumor immune responses. In the study, GSH reduced ethyl ester, a GSH precursor, was administered to leukemia patients to restore their GSH levels. As a result, mitochondrial activity, oxidative phosphorylation, ATP production, and NK cell-mediated cytotoxicity were all enhanced. NK cell function and proliferation were also promoted27. This is a very interesting finding, perhaps in the future, GSH-based interventions could be considered as a potential immunotherapy strategy for treating leukemia patients.
Impact of GSH on Professional Athletes
Professional athletes are continuously under attack from ROS because they frequently participate in competitive events, they undergo intensive training and must exercise regularly to stay in peak physical condition. Intensive physical activity increases oxygen consumption and accelerates the production of metabolites such as ROS or free radicals. As a result, oxidative breakdown products quickly accumulate, increasing oxidative stress in the body. This is one reason why athletes may experience fatigue, physical weakness, and immune suppression. Although it is well known that moderate exercise can strengthen the immune system, excessive exercise may have the opposite effect28.
Detoxification may also be important for professional athletes. Swimmers, for example, are exposed to excessive chlorine, the chemical used in swimming pools to keep the water clean.
Therefore, it seems reasonable and beneficial for athletes to supplement with antioxidants. According to a study conducted in 2023, several antioxidants showed positive effects on athletic performance. GSH, vitamin C, vitamin E, resveratrol, coenzyme Q10, selenium, and curcumin were shown to improve physical performance, shorten recovery time from fatigue, and reduce inflammation29. In a double-blinded, placebo-controlled trial in 202330 and a study conducted in 201931, GSH was shown to improve the sports performance and physical condition of athletes. Similar results were observed in triathletes and swimmers32. In one study, oral supplementation with 450 mg of GSH per day for three weeks improved antioxidant status33. On the other hand, another study showed that intravenous administration of a single acute dose of 3000 mg per day improved total antioxidant capacity34. Additionally, a study using a daily dose of 1000 mg of GSH for three weeks found a reduction in serum levels of oxidative stress markers35. A specific cysteine-rich whey protein isolate was even shown to deliver GSH precursors to cells, increasing intracellular GSH levels in healthy individuals by 35.5% and enhancing their muscular performance by 13%36.
Although numerous studies have demonstrated that the use of antioxidants, especially GSH, may have positive effects on the athletic performance, more research is needed to determine the optimal dose of GSH, the timing of supplementation, and the duration of treatment. Also, the sample sizes in most of these studies are relatively small, and some of the results are inconsistent, large-scale and well-controlled experiments are warranted.
Here is a summary of potential benefit and impact of GSH on athletes (Table 2).
| Benefit | Impact on Athletes |
| Reduces oxidative stress | Minimizes muscle damage and inflammation after intense exercise |
| Enhances recovery | Speeds up post-exercise muscle repair and reduces fatigue |
| Improves mitochondrial function | Supports sustained energy production during performance |
| Boosts immune defense | Lowers risk of infection from overtraining |
| Regulates cortisol | Helps maintain hormonal balance and reduce overtraining symptoms |
| Enhances aerobic metabolism | Promotes efficient oxygen use and lactate clearance |
Reference: M. Sadowski, E. Zawieja, A. Chmurzynska. The impact of N‐acetylcysteine on lactate, biomarkers of oxidative stress, immune response, and muscle damage: A systematic review and meta‐analysis. J Cell Mol Med 28, 1-17 (2024)
Diseases Associated with Low GSH
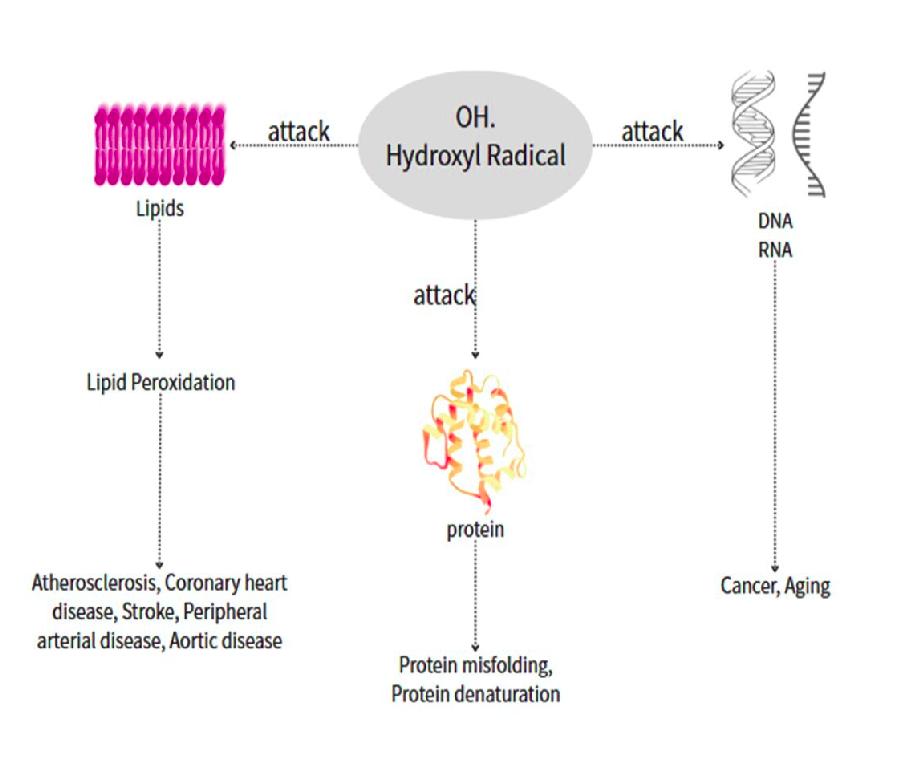
Most common chronic or age-related diseases are associated with low GSH levels and ROS attacks. ROS can cause cellular damage (Figure 2) and subsequent problems such as cardiovascular, inflammatory, immune, and metabolic disorders, as well as cancer37,38. Excessive ROS, such as hydroxyl radicals (![]() ), attack lipids, resulting in peroxidation, which leads to various cardiovascular diseases, including atherosclerosis, coronary heart disease, stroke, peripheral arterial disease, and aortic disease. When proteins are attacked by ROS, they may undergo denaturation. Protein denaturation is a potential causal mechanism for many diseases (Table 3)39,40,41,42,43. ROS can also damage DNA, leading to mutations and contributing to various diseases such as neurodegenerative diseases, genetic disorders, cancer, and premature aging.
), attack lipids, resulting in peroxidation, which leads to various cardiovascular diseases, including atherosclerosis, coronary heart disease, stroke, peripheral arterial disease, and aortic disease. When proteins are attacked by ROS, they may undergo denaturation. Protein denaturation is a potential causal mechanism for many diseases (Table 3)39,40,41,42,43. ROS can also damage DNA, leading to mutations and contributing to various diseases such as neurodegenerative diseases, genetic disorders, cancer, and premature aging.
| Disease | Mechanism of ROS-Induced Protein Denaturation | Key Proteins Affected |
| Alzheimer’s Disease (AD) | ROS oxidize and misfold proteins, forming amyloid-beta plaques | Amyloid-beta, tau |
| Parkinson’s Disease (PD) | Oxidative damage to α-synuclein, promoting aggregation | α-synuclein |
| Amyotrophic Lateral Sclerosis (ALS) | ROS-induced SOD1 oxidation, leading to motor neuron death | SOD1, TDP-43, FUS |
| Huntington’s Disease (HD) | Oxidative stress exacerbates polyglutamine protein misfolding | Huntingtin |
Reference: M. T. Lin, M. F. Beal. Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature 443, 787-795 (2006).
Neurodegenerative diseases have attracted considerable attention from researchers due to the rapidly rising number of cases. GSH is well known for its neuroprotective role44. Low GSH levels have been found in the brains of people with Parkinson’s disease, Alzheimer’s disease, schizophrenia, bipolar disorder, depression, ADHD, and other neurological disorders45,46,47. Among neurodegenerative diseases, PD and AD have been shown altered GSH in patients compared with controls, the evidence is more robust than for diseases like ALS and HD. Depleted GSH levels are consistently observed in both brain and blood during the early stage of PD48 and AD49, with imaging and biomarker date providing direct evidence. However, inconsistent findings have been reported in ALS50 and HD51 patients, likely due to early stage and smaller sample sizes.
Some infectious diseases are also associated with low GSH levels as well, such as those observed in HIV-positive individuals52,53,54 and patients with hepatitis55. Chronic Obstructive Pulmonary Disease (COPD) is characterized by chronic progressive airway obstruction. It is a common condition among cigarette smokers, and its progression is irreversible. GSH concentrations are also reduced in patients with COPD56.
Regarding GSH levels in cancer patients, the situation is quite complex. While GSH levels are commonly low in cancer patients overall, they are often elevated in tumor cells57. Studies have shown that GSH levels are higher in breast, ovarian, head and neck, and lung cancer cells compared to adjacent healthy tissues58. Elevated GSH levels may contribute to the strong survival and rapid proliferation of the tumor cells. In addition, since GSH is a detoxifying agent, it may protect tumor cells from chemotherapy by conjugating with xenobiotics, and with the help of glutathione transferase, the resulting toxic compounds are exported and eliminated. Because of the increased detoxification ability, cancer cells become more tolerant to drugs, which makes cancer treatment more difficult. A new therapeutic approach that combines GSH depletion or inhibition of its synthesis with standard cancer treatment has been reported to improve the effectiveness chemotherapies by inducing ferroptosis (a type of programmed cell death that is iron-dependent). The concept is to deplete intracellular GSH in tumor cells, making them more vulnerable and sensitive to drugs, followed by chemotherapy59. This new therapeutic strategy may offer oncologists more treatment options in the future.
Some people describe aging philosophically as a chronic disease that begins from day one. Aging is a law of nature, and we all wish to experience a healthy aging journey. In an in vivo experiment, mosquitos with augmented GSH levels (increased by 50 — 100% through dietary supplementation with the GSH precursor MgTC — magnesium thiazolidine-4-carboxylic acid) showed a 30—38% increase in lifespan compared with the control group60. In another study conducted at the Ageing Research Center, Odense University in Denmark, blood samples were collected from 41 centenarians aged 100 —105 years and 52 community-dwelling control subjects aged 60 — 79 years. The enzyme activities of SOD, glutathione peroxidase, catalase, and glutathione reductase (GR) in erythrocytes were measured. The researchers found that the mean GR activity was significantly higher in centenarians than in elderly individuals. The range of GR activity was also shifted toward higher values in the centenarian group compared to the younger group. Furthermore, centenarians with the best functional capacity tended to have the highest GR activity61. Aging in humans is a long and complex process, and there are limitations to conducting clinical trials on the correlation between GSH levels and aging. Such studies are not only impractical but also unethical, therefore most research investigating GSH levels and longevity in human relies on the results of animal or insect experiments.
Although GSH appears to be crucial for maintaining health and promoting longevity, a study published in 2025 reported contradictory results. In that study, mice with lifelong GSH deficiency exhibited an increased lifespan and delayed age-related motor decline compared with control mice62. The concept of hormesis was introduced in the study. “In the fields of biology and medicine, hormesis is defined as an adaptive response of cells and organisms to a moderate (usually intermittent) stress”63. Although the authors used hormesis theory to explain their findings, further investigations and research are needed.
To achieve healthy aging, a combined approach involving the administration of antioxidant compounds, antioxidant-rich dietary patterns, and regular physical exercise would be ideal. This comprehensive strategy is currently being investigated in human studies64,65.
Methods to Boost GSH
GSH is synthesized intracellularly through the sequential enzymatic actions of glutamylcysteine ligase and GSH synthetase. This process occurs inside the cells, primarily in the liver66. Among the three amino acids, cysteine, a sulfur-containing amino acid, is responsible for the biological activity of GSH. To elevate GSH concentrations, several methods have been proposed, such as administrating chemically synthesized GSH, pharmaceutical compounds that act as prodrugs of GSH, natural foods rich in GSH or GSH precursors, and commercially available dietary supplements.
These approaches are introduced below:
Oral GSH
Oral GSH supplements are commercially available in pill or powder form and are produced by many pharmaceutical and chemical companies. However, consuming GSH directly is not an effective way to raise GSH levels because it does not survive the digestive process; it is broken down into amino acids by intestinal enzyme ![]() -glutamyl transpeptidase. After being broken down into free amino acids, these components are easily oxidized along the digestive tract before reaching the liver, where most GSH is synthesized. Therefore, consuming GSH itself provides poor bioavailability66,67,68. Research has shown that its bioavailability is less than 1%69.
-glutamyl transpeptidase. After being broken down into free amino acids, these components are easily oxidized along the digestive tract before reaching the liver, where most GSH is synthesized. Therefore, consuming GSH itself provides poor bioavailability66,67,68. Research has shown that its bioavailability is less than 1%69.
Pharmaceutical Compounds
Many of the studies described in this paper were carried out using pharmaceutical compounds to raise GSH levels in test subjects. These drugs included NAC (N-acetylcysteine), S-adenosylmethionine, ornithine decarboxylase, and oxothiazolidine carboxylate. Among them, NAC is the most commonly used compound in GSH research, especially in human trials. However, all drugs have contraindications, dosage limitations, and potential toxic side effects. Therefore, their use is usually temporary, either for treating illnesses or for experimental purposes in laboratory settings. The safety of long-term use needs to be carefully evaluated.
Natural Foods
There are several natural foods that contain GSH. Spinach, avocados, asparagus, cucumbesr, and green beans are among the richest dietary sources of GSH70. Foods high in the amino acids cysteine and methionine may also help boost GSH levels. Cysteine and methionine are sulfur-containing amino acids, and sulfur is essential for the synthesis of GSH71. Dietary proteins from beef, fish, and poultry are good sources of cysteine and methionine. Rich sources of sulfur can also be found in vegetables like broccoli, Brussels sprouts, cauliflower, kale, watercress, mushrooms, and allium vegetables such as garlic, shallots, and onions72.
Although these food sources are rich in GSH or building blocks of GSH, concentration and bioavailability of GSH may differ depending on the method of cooking the foods. For example, asparagus is one of the vegetables with the highest GSH content73, according to a study that examined high-temperature cooking methods (baking, frying, and grilling), more than 40% of GSH was lost from asparagus74. The researchers concluded that high temperatures accelerate GSH decomposition, including oxidation to the disulfide form (GSSG) or further degradation. This finding is consistent with a previous study, which reported losses of GSH during any type of heat-based food processing75. The health benefits of GSH derived from natural sources such as fruits and vegetables are still under debated.
The digestive system may also pose a challenge for the delivery of GSH in humans. As a small peptide, it may not survive the acidic environment of the stomach and could be degraded by the digestive enzymes. Thus, the efficiency of obtaining GSH from natural foods warrants further investigation.
Milk Thistle
This herbal supplement is extracted from milk thistle plants. It contains an active compound known as Silybum marianum, which is well known for antioxidant properties76, and it has been shown to increase and help maintain GSH levels77.
Turmeric Extract
Turmeric is a yellow-orange herb commonly used as a spice in Indian cuisine. It has been used as traditional medicine in India as well since ancient times. Its medicinal properties are believed to be related to its main compound, curcumin78. Numerous animal and test-tube studies have shown that turmeric and curcumin extract have the ability to increase GSH levels79,80. Researchers concluded that curcumin may help restore adequate GSH levels and improve the activity of GSH related enzymes.
Probiotics
Probiotics are known to modulate the gut microbiota, and a healthy gut microbiota composition can support better nutrient absorption and reduce oxidative stress. Lactobacillus, Bifidobacterium, Lactococcus, Streptococcus, and Enterococcus species are the probiotic microorganisms mainly used in humans81,82,83. More than 500 different bacteria species reside in an adult human gastrointestinal tract serving as a natural source of probiotics, and many of the probiotic species used today have been isolated from the human gut. Some probiotic strains can produce or enhance the availability of amino acids such as cysteine, glycine, and glutamate. Lactobacillus fermentum ME-3 (often referred to as ME-3) for example, is a specific strain of bacteria isolated from the intestinal tract of a healthy 1-year-old child in 1995. Studies have showed that ME-3 exhibits high antioxidant activity84. Further research has indicated that the antioxidant activity of ME-3 is due to its ability to synthesize GSH85. Results from a clinical trial of 21 volunteers suggested that this particular bacterial strain could lower cardiovascular risk by reducing LDL-cholesterol, increasing GSH/GSSG ratio, and enhancing total antioxidant capacity86.
Whey protein isolate
Whey protein refers to a group of different types of proteins derived from the liquid portion of raw milk. When these proteins are concentrated to higher than 90%, it is called whey protein isolate or WPI. WPI is rich in proteins such as lactoferrin, serum albumin, and immunoglobulins, which contain significant amounts of cysteine residues and ![]() -glutamylcysteine peptides. The amino acid cysteine and the
-glutamylcysteine peptides. The amino acid cysteine and the ![]() -glutamylcysteine are both building blocks of GSH, providing an excellent source for GSH synthesis87. In an open-label dose-response study, 36 healthy individuals were administrated 15, 30, or 45 g/day pressurized WPI for 14 days. As a result, there was an increase of lymphocyte GSH by 24% in the 45 g/day group88. Another open-label study was conducted on 38 individuals with nonalcoholic steatohepatitis (NASH), daily dose of 20 g WPI was given for 12 weeks, at the end of study as the report showed that there were improvements in liver biochemistry, and plasma GSH and total antioxidant capacity were increased (53
-glutamylcysteine are both building blocks of GSH, providing an excellent source for GSH synthesis87. In an open-label dose-response study, 36 healthy individuals were administrated 15, 30, or 45 g/day pressurized WPI for 14 days. As a result, there was an increase of lymphocyte GSH by 24% in the 45 g/day group88. Another open-label study was conducted on 38 individuals with nonalcoholic steatohepatitis (NASH), daily dose of 20 g WPI was given for 12 weeks, at the end of study as the report showed that there were improvements in liver biochemistry, and plasma GSH and total antioxidant capacity were increased (53 ![]() 11 vs 68
11 vs 68 ![]() 11 and 1.26
11 and 1.26 ![]() 0.10 vs 2.03
0.10 vs 2.03 ![]() 0.10)89.
0.10)89.
Immunocal® is an undenatured WPI, in which the proteins remain in their native forms, preserving the dipeptides cystine (two cysteine molecules linked by a disulfide bond) and glutamyl-cysteine in the proteins are intact. These dipeptides are enzyme-resistant and serve as the precursors for GSH synthesis. Several studies have been published — including laboratory animal experiments, blinded human trials, and review papers — have supported its GSH boosting effects. In a 1999 study, it was demonstrated that taking Immunocal® (20 g/day for three months), GSH levels were augmented in lymphocytes of healthy individuals by 35.5% compared to the control group90. Immunocal® has also been shown to increase GSH concentrations in various tissues, enhance immune responses, and potentially inhibit tumor development91. Additionally, it has been found to preserve brain GSH92. In the most recent study published in 2025, Immunocal® was shown to promote Reelin expression in vitro (in hippocampal — entorhinal cortex slices from rat brain), and to sustain brain GSH and Reelin expression while reducing amyloid plaque load in the entorhinal cortex and hippocampus of J20 mice93.
The main concept of boosting GSH levels through natural foods involving consuming those that are rich in GSH or its precursors. However, the quantities of GSH and its precursors are difficult to determine and can vary greatly depending on factors such as food types, species, seasonal variation, storage conditions, preparation methods, and manufacturing process. There are also limitations in determining whether any observed increase in GSH levels is directly associated with the food intake, and the stability and sustainability of GSH are also difficult to measure scientifically. Currently, there is insufficient evidence to confirm that human intracellular GSH can be increased through simply dietary intake, and further research is warranted.
Discussion and Future Perspectives
Undoubtedly, GSH continues to attract researchers’ attention because of the multiple and critical roles it plays. The depletion of GSH has been observed in athletes and has been shown to be associated with many chronic diseases.
In this review, we presented the results from recent studies showing that supplementing athletes with GSH or its precursors in athletes, improved antioxidant status, increased total antioxidant capacity, and reduced oxidative stress markers in serum. However, the modes of GSH delivery varied among different studies. Some used oral administration, while others employed intravenous delivery. In addition, the dosages of GSH administered ranged widely from 450 mg to 1000 mg per day. Furthermore, the timing of supplementation and durations of treatment are also important factors to consider. The results among the studies were inconsistent, and the sample sizes of most experiments were relatively small. Therefore, further studies are needed to determine the optimal GSH dosage, timing of supplementation, duration of treatment, and the differences among various administration methods for improving athletic performance.
The growing prevalence of chronic diseases and premature aging has led to increased research interest in GSH due to its neuroprotective role. Several studies have shown that GSH levels are low in the brains of patients with PD and AD, and the most recent clinical trial even provided robust imaging evidence demonstrating significant GSH depletion in substantia nigra region of PD patients. GSH depletion has also been reported in other neurodegenerative diseases such as ALS and HD, but the associations with GSH depletion remain preliminary, and further investigation are expected.
In normal cells, GSH protects against carcinogenesis by neutralizing mutagenic ROS and supporting DNA repair. However, GSH also performs this role in cancer cells, shielding them from chemotherapy-induced cytotoxicity. This paradox—where GSH protects both healthy and malignant cells—complicates therapeutic interventions. Strategies to selectively deplete tumor GSH while preserving GSH in normal cells have been proposed by researchers, but more research is needed to validate this concept.
During this review, an important observation is that although GSH is essential for cellular function, identifying a safe and effective method to increase and maintain GSH levels in humans remains challenging. Oral GSH, available in pill or powder form, is easily purchased in supermarkets in the United States but is classified as a drug in most other countries. Regardless of its classification, oral GSH has poor bioavailability because it is broken down into amino acids during digestion, and the intracellular reassembly of those components is highly inefficient. NAC, the prodrug of GSH, has been widely used as a means to increase GSH levels in human trials. However, to achieve clinically beneficial GSH levels, high doses of NAC are required, which may lead to toxic side effects. This may explain why among all the clinical studies that we’ve reviewed, most experimental periods involving NAC lasted between three and six weeks. Another important issue worth discussing is the sustainability of GSH levels, even when GSH can be raised. In the clinical studies that we’ve reviewed, most the experiment periods ranged from three weeks to three months. However, no publications providing clear and quantifiable evidence on the long-term sustainability of GSH levels have been identified. Most studies demonstrated only short-term biochemical changes.
Diet and food, on the other hand, are natural, safe and enjoyable. Spinach, avocados, asparagus, cucumbers, and green beans are rich dietary sources of GSH. Beef, fish, and poultry are good source of the sulfur-containing amino acids cysteine and methionine. Vegetable and fruits such as broccoli, Brussels sprouts, cauliflowers, and kale also provide abundant sulfur. However, the quantity of GSH or its precursors in food and their actual efficacy in raising intracellular GSH levels remain subjects of ongoing debate.
Food supplements, on the other hand are more scientifically supported and generally considered safe for long-term use. Both the probiotic L. fermentum ME-3 and the whey protein isolate Immunocal® are backed by research. Numerous studies have demonstrated a wide range of health benefits, including reduced cardiovascular risk, lower oxidative stress and inflammation, and elevated GSH levels. Although results from both supplements appear promising, the sample sizes in current studies remain small. Further large-scale clinical research is expected.
It remains necessary to determine whether there is adequate evidence that raising glutathione (GSH) levels leads to better health outcomes, offers protection against disease, or enhances athletic performance. So far, it seems possible that GSH concentrations can be raised in humans, however, several translational barriers remain difficult to overcome.
In this research, several translational limitations should be noted. 1. Limited bioavailability. Oral GSH undergoes rapid breakdown in the gastrointestinal tract, and systemic uptake efficacy varies greatly among individuals. Although NAC shows better absorption, the dosage and duration of treatment remain inconsistent across studies. 2. Sustainability of GSH elevation. Almost all clinical trials indicate that increases in GSH are transient; GSH tend to return to baseline within several weeks after supplement is discontinued. 3. Lack of long-term human trials. In most human studies, the duration of treatment ranged from 6 to 24 weeks, focusing primarily on biochemical changes rather than clinical outcomes. 4. Quantification challenging. The accurate measurement of GSH and GSSG in vivo remains difficult. Standardized analytical protocols should be developed to improve measurement reliability.
As for future directions, strategies should be explored to deliver GSH or its precursors directly to target organs such as the brain and liver, for example, through nanocarriers or exosome-based formulations, in order to minimize systemic interference. In addition, more large-scale, long-term randomized controlled trials are needed. The integration of pharmacological, nutritional, and lifestyle approaches may help determine whether GSH modulation can successfully transition from an experimental concept to a clinical therapy.
Methods
This review summarizes the fundamental concepts of GSH, including its structure, synthesis, and biological functions. It then outlines recent research findings on the role of GSH in chronic diseases and among athletes. Finally, it presents several potential methods for increasing GSH levels and discusses the more feasible and scientifically supported approaches. Based on the aforementioned topics, a systematic literature search was conducted using Google Scholar, ScienceDirect, and PubMed to identify relevant studies, journal articles, research papers, and clinical trials. Search terms combined the keywords “GSH functions”, “GSH synthesis”, “antioxidant”, “detoxification”, “immune function”, “chronic diseases”, “professional athletes”, and “GSH supplementation”. Boolean operators (e.g., AND, OR) were applied to refine results and retrieve publications relevant to the focus of this research.
Inclusion criteria were applied as studies were considered eligible if they were directly related to at least one of the topics in antioxidant defense mechanisms, detoxification pathway, immune system, association between GSH concentrations and chronic diseases, GSH status and athletic performance, or GSH supplementation. In addition, randomized controlled trials, systematic reviews, human trials and animal studies were prioritized. Timeframe preference was set between 2015 and 2025 to ensure the most recent findings, unless they were landmark studies or foundational to the field. As for exclusion criteria, publications in language other than English were not included. Case reports, editorials, commentaries, and studies with very small sample sizes or lack of control groups were excluded. To ensure the reliability, key factors such as publication date, peer-review status, author credentials, institutional affiliations, and diversity in referenced sources were evaluated. Also, only articles from reputable journals indexed in major databases were considered.
This methodology provides a transparent, systematic, and evidence-based approach to screen and select literatures, ensuring the most updated, relevant, and valuable studies are included in this review.
Acknowledgments
I would like to thank Dr. Arri Esien, a professor in the Department of Biology at Emory University, for his valuable mentorship and guidance throughout the research. I am also grateful to Jerry Lu, Chris Wang, and Rita for their feedback and suggestions.
References
- E. B. Kurutas. The importance of antioxidants which play the role in cellular response against oxidative/nitrosative stress: current state. Nutr J 15, 71 (2015). [↩]
- T. Finkel, N. J. Holbrook. Oxidants, oxidative stress and the biology of ageing. Nature 408, 239–247 (2000). [↩]
- S. C. Lu. Glutathione synthesis. Biochim Biophys Acta 5, 3143-53 (2013). [↩]
- F. E. A. Hassanein, C. Mikhail, S. Elkot, A. Abou-Bakr. L-arginine vs. L-glutamine oral suspensions for radiation-induced oral mucositis: a triple-blind randomized trial. J Cancer Res Clin Oncol 151, 198 (2025). [↩]
- O. Zitka, S. Skalickova, J. Gumulec, M. Masarik, V. Adam, J. Hubalek, L. Trnkova, J. Kruseova, T. Eckschlager, R. Kizek. Redox status expressed as GSH:GSSG ratio as a marker for oxidative stress in paediatric tumour patients. Oncol Lett 4, 1247-1253 (2012). [↩]
- F. Nuhu, A. Gordon, R Sturmey, A. M. Seymour, S. Bhandari. Measurement of Glutathione as a Tool for Oxidative Stress Studies by High Performance Liquid Chromatography. Molecules 25, 4196 (2020). [↩]
- S. Vaish, D. Gupta, R. Mehrotra, S. Mehrotra, M. K. Basantani. Glutathione S-transferase: A versatile protein family. 3 Biotech 10, 321 (2020). [↩]
- J. A. Imlay. The molecular mechanisms and physiological consequences of oxidative stress: Lessons from a model bacterium. Nat Rev Microbiol 11, 443–454 (2013). [↩]
- M. Deponte. Glutathione catalysis and the reaction mechanisms of glutathione-dependent enzymes. Biochim Biophys Acta Gen Subj 1830, 3217–3266 (2013). [↩]
- R. Pelton. The glutathione theory of aging. Altern Ther Health Med 30, 54-58 (2024). [↩]
- J. R. Arthur. The glutathione peroxidases. Cell Mol Life Sci 57, 1825-1835 (2000). [↩]
- J. D. Hayes, J. R. Flanagan, I. R. Jowsey. Glutathione transferase. Annu Rev Pharmacol Toxicol 45, 51-88 (2005). [↩]
- P. Biswas, C. Dellanoce, A. Vezzoli, S. Mrakic-Sposta, M. Malnati, A. Beretta, R. Accinni. Antioxidant activity with increased endogenous levels of vitamin C, E and A following dietary supplementation with a combination of glutathione and resveratrol precursors. Nutrients 12, 3224 (2020). [↩]
- J. Pizzorno . Glutathone!. Integr Med (Encinitas) 13, 8-12 (2014). [↩]
- D. P. Jones, L. A. Brown, P. Sternberg. Variability in glutathione-dependent detoxication in vivo and its relevance to detoxication of chemical mixtures. Toxicology 105, 267-74 (1995). [↩]
- K. Jomova, S. Y. Alomar, E. Nepovimova, K. Kuca, M. Valko. Heavy metals: toxicity and human health effects. Toxicology 99, 153-209 (2025). [↩]
- J. Gutman. Glutathione – Your Key to Health. 4th edition, (2008). [↩]
- Li. Xianchun. Glutathione and Glutathione-S-Transferase in Detoxification Mechanisms. General and Applied Toxicology, 1-13 (2009). [↩]
- A. Ramachandran, J. Y. Akakpo , S. C. Curry, B. H. Rumack, H. Jaeschke. Clinically relevant therapeutic approaches against acetaminophen hepatotoxicity and acute liver failure. Biochem Pharmacol 228, 116056 (2024). [↩]
- Y. F. Hsiao, S. C. Huang, S. B. Cheng, C. C. Hsu, Y. C. Huang. Glutathione and selenium supplementation attenuates liver injury in diethylnitrosamine-induced hepatocarcinogenic mice by enhancing glutathione-related antioxidant capacities. Int J Mol Sci 25, 11339 (2024). [↩]
- R. Teschke. Aluminum, arsenic, beryllium, cadmium, chromium, cobalt, copper, iron, lead, mercury, molybdenum, nickel, platinum, thallium, titanium, vanadium, and zinc: molecular aspects in experimental liver injury. Int J Mol Sci 23, 12213 (2022). [↩]
- W. Dröge, K. Schulze-Osthoff, S. Mihm, D. Galter, H. Schenk, H. P. Eck, S. Roth, H. Gmünder. Functions of glutathione and glutathione disulfide in immunology and immunopathology. FASEBJ 8, 1131-1138 (1994). [↩]
- G. Morris, M. Gevezova, V. Sarafian, M. Maes. Redox regulation of the immune response. Cellular & Molecular Immunology 19, 1079–1101 (2022). [↩]
- D. G. Franchina, H. Kurniawan, M. Grusdat, C. Binsfeld, L. Guerra, L. Bonetti, L. Soriano-Baguet, A. Ewen, T. Kobayashi, S. Farinelle, A. R. Minafra, N. Vandamme, A. Carpentier, A. K. Borgmann, C. Jäger, Y. Chen, M. Kleinewietfeld, V. Vasiliou, M. Mittelbronn, K. Hiller, P. A. Lang, D. Brenner. Glutathione-dependent redox balance characterizes the distinct metabolic properties of follicular and marginal zone B cells. Nat Commun 13,1789 (2022). [↩]
- S. Chen, J. Fan, P. Xie, J. Ahn, M. Fernandez, L. K. Billingham, J. Miska, J. D. Wu, D. A. Wainwright, D. Fang, J. A. Sosman, Y. Wan, Y. Zhang, N. S. Chandel, B. Zhang. CD8+ T cells sustain antitumor response by mediating crosstalk between adenosine A2A receptor and glutathione/GPX4. J Clin Invest 5, 134 (8) (2024). [↩]
- A. C. Millman, M. Salman, Y. K. Dayaram, N. D. Connell, V. Venketaraman. Natural killer cells, glutathione, cytokines, and innate immunity against mycobacterium tuberculosis. Journal of Interferon & Cytokine Research 28; 3 (2008). [↩]
- Y. Zhao, Y. Wang, T. Liang, X. Song, Y. Zhu, X. Liu, M. Lv, C. Zheng, F. Ni. Dysregulated glutathione metabolism impairs natural killer cell function in patients with acute leukemia. International Immunopharmacology 8, 154 (2025). [↩]
- H. M. Alessio. Exercise-induced oxidative stress. Med Sci Sports Exerc 25, 218-224 (1993). [↩]
- V. J. C. Suarez, A. B. Sanchez, J. M. Ayuso, I. M. Guardado, A. M. Rodriguez, J. F. T. Aguilera. Antioxidants and sports performance. Nutrients 15, 2371 (2023). [↩]
- E. Lee, H. Y. Park, S. W. Kim, Y. Sun, J. H. Choi, J. Seo, Y. P. Jung, A. J. Kim, J. Kim, K. Lim. Enhancing supplemental effects of acute natural antioxidant derived from yeast fermentation and vitamin c on sports performance in triathlon athletes: a randomized, double-blinded, placebo-controlled, crossover trial. Nutrients 15, 3324 (2023). [↩]
- K. Grucza, P. Chołbiński, D. Kwiatkowska, M. Szutowski. Effects of supplementation with glutathione and its precursors on athlete performance. Biomed J Sci & Tech Res 12, 4 (2019). [↩]
- L. Petrov, A. Alexandrova, M. Kachaunov, R. Penov, T. Sheytanova, S. Kolimechkov. Effect of glutathione supplementation on swimmers’ performance. Pedagogy of Physical Culture and Sports 25(4), 215-224 (2021). [↩]
- B. Schmitt, M. Vicenzi, C. Garrel, F. M. Denis. Effects of N-Acetylcysteine, Oral Glutathione (GSH) and a Novel Sublingual Form of GSH on Oxidative Stress Markers: A Comparative Crossover Study. Redox Biol 6, 198–205 (2015). [↩]
- T. Saitoh, H. Satoh, M. Nobuhara, M. Machii, T. Tanaka, H. Ohtani, M. Saotome, T. Urushida, H. Katoh, H. Hayashi. Intravenous Glutathione Prevents Renal Oxidative Stress after Coronary Angiography More Effectively than Oral N-Acetylcysteine. Heart Vessel 26, 465–472 (2011). [↩]
- S. D. Søndergård, I. Cintin, A. B. Kuhlman, T. H. Morville, M. L. Bergmann, L. K. Kjær, H. E. Poulsen, D. Giustarini, R. Rossi, F. Dela. The Effects of 3 Weeks of Oral Glutathione Supplementation on Whole Body Insulin Sensitivity in Obese Males with and without Type 2 Diabetes: A Randomized Trial. Appl. Physiol. Nutr. Metab. 46, 1133–1142 (2021). [↩]
- L. C. Lands, V. L. Grey, A. A. Smountas. Effect of supplementation with a cysteine donor on muscular performance. J Appl Physiol (1985) 87, 1381-5 (1999). [↩]
- N. Ballatori, S. M. Krance, S. Notenboom, S. Shi, K. Tieu, C. H. Hammond. Glutathione dysregulation and the etiology and progression of human diseases. Biol Chem 390, 191-214 (2009). [↩]
- C. A. Lang, B. J. Mills, W. Mastropaolo, M. C. Liu. Blood glutathione decreases in chronic diseases. J Lab Clin Med 135, 402-5 (2000). [↩]
- M. T. Lin, M. F. Beal. Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature 443, 787–795 (2006). [↩]
- B. Mu, Y. Zeng, L. Luo, K. Wang. Oxidative stress-mediated protein sulfenylation in human diseases: Past, present, and future. Redox Biology 76, October (2024). [↩]
- C. A. Juan, J. M. Pérez de la Lastra, F. J. Plou, E. Pérez-Lebeña. The chemistry of reactive oxygen species (ROS) revisited: outlining their role in biological macromolecules (DNA, lipids and proteins) and induced pathologies. Int J Mol Sci 22, 4642 (2021). [↩]
- A. Phaniendran, D. B. Jestadi, L. Periyasamy. Free radicals: properties, sources, targets, and their implication in various diseases. Ind J Clin Biochem 30,11–26 (2015). [↩]
- T. Rahman, I. Hosen, M. M. T. Islam, H. U. Shekhar. Oxidative stress and human health. Advances in Bioscience and Biotechnology 3, No.7A, November (2012). [↩]
- E. Sofic, K. W. Lange, K. Jellinger, P. Riederer. Reduced and oxidized glutathione in the substantia nigra of patients with Parkinson’s disease. Neurosci Lett 142, 128-30 (1992). [↩]
- I. Y. Iskusnykh, A. A. Zakharova, D. Pathak. Glutathione in brain disorders and aging. Molecules 27, 324 (2022). [↩]
- E. Lorenc-Koci. Chapter: Dysregulation of Glutathione Synthesis in Psychiatric Disorders in Studies on Psychiatric Disorder. Humana Press (2015). [↩]
- J. W. Gawryluk, J. F. Wang, A. C. Andreazza, L. Shao, L. T. Young. Decreased levels of glutathione, the major brain antioxidant, in post-mortem prefrontal cortex from patients with psychiatric disorders. International Journal of Neuropsychopharmacology 14, 123–130 (2011). [↩]
- D. Shukla, A. Goel, P. K. Mandal, S. Joon, K. Punjabi, Y. Arora, R. Kumar, V. S. Mehta, P. Singh, J. C. Maroon, R. Bansal, K. Sandal, R. G. Roy, A. Samkaria, S. Sharma, S. Sandhilya, S. Gaur, S. Parvathi, M. Joshi. Glutathione depletion and concomitant elevation of susceptibility in patients with Parkinson’s Disease: state-of-the-art MR spectroscopy and neuropsychological study. ACS Chemical Neuroscience 14, 4383-4394 (2023). [↩]
- J. J. Chen, M. Thiyagarajah, J. Song, C. Chen, N. Herrmann, D. Gallagher, M. J. Rapoport, S. E. Black, J. Ramirez, A. C. Andreazza, P. Oh, S. Marzolini, S. J. Graham, K. L. Lanctôt. Altered central and blood glutathione in Alzheimer’s disease and mild cognitive impairment: a meta-analysis. Alzheimer’s Research & Therapy, 14:23 (2022). [↩]
- T. K. Pham, N. Verber, M. R. Turner, A. Malaspina, M. O. Collins, R. J. Mead, P. J. Shaw. Glutathione oxidation in cerebrospinal fluid as a biomarker of oxidative stress in amyotrophic lateral sclerosis. Translational Neurodegeneration. 14, 36 (2025). [↩]
- A. J. Lowe, F. B. Rodrigues, M. Arridge, E. De Vita, E. B. Johnson, R. I. Scahill, L. M. Byrne, R. Tortelli, A. Heslegrave, H. Zetterberg, E. J. Wild. Longitudinal evaluation of proton magnetic resonance spectroscopy metabolites as biomarkers in Huntington’s disease. Brain Communications 4, 1-17 (2022). [↩]
- G. Bounous, S. Baruchel, J. Falutz, P. Gold. Whey protein as a food supplement in HIV-seropositive individuals. Clinical investigative medicine 16, 204-209 (1993). [↩]
- S. Baruchel, G. Bounous, P. Gold. Place for an Antioxidant Therapy in Human Immunodeficiency Virus (HIV) Infection. Oxidative Stress, Cell Activation and Viral Infection. Birkhauser Verlag Basel/Switzerland (1994). [↩]
- S. Baruchel, G Viau, R. Oliver, G. Bounous, M. A. Wainberg. Nutriceutical Modulation of Glutathione with a Humanized Native Milk Serum Protein Isolate, Immunocal
 : Application in AIDS and Cancer. Oxidative stress in cancer, aids, and neurodegenerative diseases. Luc Montagnier et al., (ed.) Marcel Dekker Inc. 447-461, (1998). [↩]
: Application in AIDS and Cancer. Oxidative stress in cancer, aids, and neurodegenerative diseases. Luc Montagnier et al., (ed.) Marcel Dekker Inc. 447-461, (1998). [↩] - A. Watanabe, K. Higuchi, K. Okada, Y. Shimizu, Y. Kondo, H. Kohri. Treatment of chronic hepatitis using whey protein (non-heated). Journal of medicine 31, 283-302 (2000). [↩]
- T Rangasamy, C. Y. Cho, R. K. Thimmulappa, L. Zhen, S. S. Srisuma, T. W. Kensler, M. Yamamoto, I. Petrache, R. M. Tuder, S. Biswal. Genetic ablation of Nrf2 enhances susceptibility to cigarette smoke–induced emphysema in mice. J Clin Invest 114, 1248–1259 (2004). [↩]
- L. Kennedy, J. K. Sandhu, M. E. Harper, M. Cuperlovic-Culf. Role of glutathione in cancer: From mechanisms to therapies. Biomolecules 10, 1429 (2020). [↩]
- M. P. Gamcsik, M. S. Kasibhatla, S. D. Teeter, O. M. Colvin. Glutathione levels in human tumors. Biomarkers 17, 671–691 (2012). [↩]
- J. Li, F. Cao, H. L. Yin, Z. J. Huang, Z. T. Lin, N. Mao, B. Sun, G. Wang. Ferroptosis: past, present and future. Cell Death & Disease 11:88 (2020). [↩]
- J. P. Jr. Richie, B. J. Mills, C. A. Lang. Correction of a glutathione deficiency in the aging mosquito increases its longevity. Proc Soc Exp Biol Med 184, 113-117 (1987). [↩]
- H. R. Andersen, B. Jeune, H. Nybo, J. B. Nielsen, K. Andersen-Ranberg, P. Grandjean. Low activity of superoxide dismutase and high activity of glutathione reductase in erythrocytes from centenarians. Age Ageing 27, 643-648 (1998). [↩]
- J. T. Mock, P. Mensah-Kane, D. L. Davis, J. M. Wong, P. H. Vann, M. J. Forster, N. Sumien. Lifelong glutathione deficiency in mice increased lifespan and delayed age-related motor declines. Aging and Disease 16, 6 (2025). [↩]
- M. P. Mattson. Hormesis Defined. Ageing Res Rev 7, 1–7 (2008). [↩]
- R. Gabbianelli, E. Damiani. Epigenetics and neurodegeneration: role of early-life nutrition. J Nutr Biochem 57: 1-13, (2018). [↩]
- A. L. Cremonini, I. Caffa, M. Cea, A. Nencioni, P. Odetti, F. Monacelli. Nutrients in the prevention of Alzheimer’s disease. Oxid Med Cell Longev 2019, Sep 4 (2019). [↩]
- M. Turner, N. A. Mantick, G. P. Carlson. Comparison of the depletion of glutathione in mouse liver and lung following administration of styrene and its metabolites styrene oxide and 4-vinuylphenol. Toxicology 206, 383-388 (2005). [↩] [↩]
- S. Liu, X. Wen, X. Zhang, S. Mao. Oral delivery of biomacromolecules by overcoming biological barriers in the gastrointestinal tract: an update. Expert Opin Drug Deliv 20, 1333-1347 (2023). [↩]
- X. Guan. Glutathione and glutathione disulfide—their biomedical and pharmaceutical applications. Med Chem Res 32, 1972-1994 (2023). [↩]
- N. Yin, P. W. R. Harris, M. Liu, J. Sun, G. Chen, J. Wen, M. A. Brimble. Enhancing the oral bioavailability of glutathione using innovative analogue approaches. Pharmaceutics 17, 385 (2025). [↩]
- A. A. Al-Temimi, A. E. B. Al-Mossawi, S. A. Al-Hilifi, S. A. Korma, T. Esatbeyoglu, J. M. Rocha, V. Agarwal. Glutathione for food and health applications with emphasis on extraction, identification, and quantification methods: a review. Metabolites 13, 465 (2023). [↩]
- R. F. Grimble. The effects of sulfur amino acid intake on immune function in humans. J Nutr 136, 1660S-1665S (2006). [↩]
- F. Bianchini, H. Vainio. Allium vegetables and organosulfur compounds: do they help prevent cancer? Environ Health Perspect 109, 893-902 (2001). [↩]
- D. P. Jones, R. J. Coates, E. W. Flagg, J. W. Eley, G. Block, R. S. Greenberg, E. W. Gunter, B. Jackson. Glutathione in foods listed in the National Cancer Institute’s health habits and history foods frequency questionnaire. Nutr. Cancer 17, 57-75 (1992). [↩]
- J. M. Drinkwater, R. Tsao, R. Liu, C. Defelice, D. J. Wolyn. Effects of cooking on rutin and glutathione concentrations and antioxidant activity of green asparagus (Asparagus officinalis) spears. Journal of Functional Foods 12, 342-353 (2015). [↩]
- L. Tesoriere, M. Fazzari, M. Allegra, M. A. Livrea. Biothiols, taurine, and lipid-soluble antioxidants in the edible pulp of Sicilian cactus pear (Opuntia ficus-indica) fruits and changes of bioactive juice components upon industrial processing. Journal of Agriculture and Food Chemistry 53, 7851-5 (2005). [↩]
- L. Abenavoli, R. Capasso, N. Milic N, F. Capasso. Milk thistle in liver diseases: past, present, future. Phytother Res 24, 1423-32 (2010). [↩]
- P. Muriel, T. Garciapiña, V. Perez-Alvarez, M. Mourelle. Silymarin protects against paracetamol-induced lipid peroxidation and liver damage. J Appl Toxicol 12(6), 439-42 (1992). [↩]
- M. Nagpal, S. Sood. Role of curcumin in systemic and oral health: An overview. J Nat Sci Biol Med 4, 3–7 (2013). [↩]
- S. K. Biswas, D. McClure, L. A. Jimenez, I. L. Megson, I. Rahman. Curcumin induces glutathione biosynthesis and inhibits NF-kappaB activation and interleukin-8 release in alveolar epithelial cells: mechanism of free radical scavenging activity. Antioxid Redox Signal 7, 32-41 (2005). [↩]
- I. A. Donatus, Sardjoko, N. P. Vermeulen. Cytotoxic and cytoprotective activities of curcumin. Effects on paracetamol-induced cytotoxicity, lipid peroxidation and glutathione depletion in rat hepatocytes. Biochem Pharmacol 39, 1869-75 (1990). [↩]
- R. Dudek-Wicher, A. Junka, J. Paleczny, M. Bartoszewicz. Clinical trials of probiotic strains in selected disease entities. International Journal of Microbiology, https://doi.org/10.1155/2020/8854119 (2020). [↩]
- Y. Nami, B. Haghshenas, A. Y. Khosroushahi. Molecular Identification and Probiotic Potential Characterization of Lactic Acid Bacteria Isolated from Human Vaginal Microbiota. Adv Pharm Bull 8, 683-695 (2018). [↩]
- Y. Nami, B. Haghshenas, A. Y. Khosroushahi. Effect of psyllium and gum Arabic biopolymers on the survival rate and storage stability in yogurt of Enterococcus durans IW3 encapsulated in alginate. Food Sci Nutr 5, 554-563 (2017). [↩]
- M. Mikelsaar, M. Zilmer. Lactobacillus fermentum ME-3 – an antimicrobial and antioxidative probiotic. Microb Ecol Health Dis 21, 1-27 (2009). [↩]
- T. Kullisaar, E. Songisepp, M. Aunapuu, K. Kilk, A. Arend, M. Mikelsaar, A. Rehema, M. Zilmer. Complete glutathione system in probiotic Lactobacillus fermentum ME-3. Prikl Biokhim Mikrobio 46, 527-31 (2010). [↩]
- T. Kullisaar, E. Songisepp, M. Mikelsaar, K. Zilmer, T. Vihalemm, M. Zilmer. Antioxidative probiotic fermented goats’ milk decreases oxidative stress mediated atherogenicity in human subjects. Br J Nutr 90, 449-456 (2003). [↩]
- C. V. Morr, E. Y. Ha. Whey protein concentrates and isolates: processing and functional properties. Crit Rev Food Sci Nutr 33,431-76 (1993). [↩]
- G. S. Zavorsky, S. Kubow, V. Grey, V. Riverin, L. C. Lands. An open-label dose-response study of lymphocyte glutathione levels in healthy men and women receiving pressurized whey protein isolate. Int J Food Sci Nutr 58, 429-36 (2007). [↩]
- T. Chitapanarux, P. Tienboon, S. Pojchamarnwiputh, D. Leelarungrayub. Open-labeled pilot study of cysteine-rich whey protein isolate supplementation for nonalcoholic steatohepatitis patients. J Gastroenterol Hepatol 24, 1045-50 (2009). [↩]
- L. C. Lands, V. L. Grey, A. A. Smountas. The Effect of Supplementation with a Cysteine Donor on Muscular Performance. Journal of applied physiology 87, 1381-1385, (1999). [↩]
- G. Bounous, G. Batist, P. Gold. Whey proteins in cancer prevention. Cancer Lett 57, 91-4 (1991). [↩]
- E. Ignownski, A. N. Winter, N. Duval, H. Fleming, T. Wallace, E. Manning, L. Koza, K. Huber, N. J. Serkova, D. A. Linseman. The cysteine-rich whey protein supplement, Immunocal®, preserves brain glutathione and improves cognitive, motor, and histopathological indices of traumatic brain injury in a mouse model of controlled cortical impact. Free Radic Biol 124, 328-341 (2018). [↩]
- A. Sandberg, S. Puttagunta, N. Duval, H. Fleming, L. Koza, K. Hieber, J. Holsopple, M. Reyna, D. Paredes, D. A. Linseman. Immunocal®, a cysteine-rich whey protein, rescues reelin and reduces amyloid plaque burden in a transgenic amyloid-β protein precursor (hAβPPSweInd) mouse model of Alzheimer’s disease. J Alzheimers Dis 106, 94-110 (2025). [↩]