Abstract
The Transient Receptor Potential Vanilloid type-2 (TRPV2) channel is expressed in both the central and peripheral nervous systems. As a cation (calcium) and mechanosensitive channel, TRPV2 plays a role in neuronal development and in maintaining the blood-brain barrier (BBB). Brain tumour cell survival and proliferation involve calcium-dependent mechanisms, and loss or inactivation of TRPV2 promoted glioblastoma cell proliferation and increased resistance to CD95-induced apoptotic cell death. The TRPV2 channel was proven as a modulator of brain tumorigenesis and glioma progression by regulating GSCs differentiation. TRPV2 oncogenicity in brain cancer is driven by alteration of its expression profile. TRPV2 activation was reported to promote GSCs differentiation and inhibit gliomagenesis. The TRPV2 agonist, Cannabidiol (CBD), suppresses GSCs’ clonogenic capacity by upregulating genes involved in either apoptosis or autophagy. Therefore, TRPV2 may be considered a prime candidate biomarker and future therapeutic target in brain cancer.
Keywords: Brain cancer (tumor), Glioma, Transient Receptor Potential Vanilloid type-2 (TRPV2, VRL, VRL-1, VRL1), Cannabidiol (CBD), Neurogenesis, Neurons, Glioblastoma stem/progenitor- like cells (GSCs).
Introduction
Malignant brain tumours are known for their high degree of invasiveness, aggressiveness, and lethality despite the availability of a variety of current standard and optimized treatments. Patients with brain tumours could benefit clinically and therapeutically from the identification of molecular targets that play a crucial role in tumour growth and survival, leading to a process that brings accuracy and specificity in patient medication. Nowadays, the concept of precision medicine, which consists of identifying the molecular signature of individual tumours that can be selected for the most appropriate therapeutic approach, has become the pivot of contemporary oncology. Indeed, the discovery of new brain cancer therapies or improvement of current ones focuses on the identification of biomarkers that are causally connected to, instead of merely related to, the disease process. On this basis, these biomarkers must undergo alteration by effective therapeutic interventions to be validated for certain populations of patients who will probably benefit from precision medicine.
The TRPV2 channel has attracted attention as a biomarker in several types of cancers1‘2‘3. Notably, in brain cancer, TRPV2 exhibits oncogenic capacity linked to brain tumour growth and survival (metastasis). In this review, we will focus on demonstrating the pathophysiological significance of the TRPV2 channel in brain cancer and provide evidence of how the measurable molecular changes in TRPV2 could validate its quality as a brain cancer biomarker and potential therapeutic target.
Search strategy and data sources
A literature search was conducted using only the peer-reviewed database available in “PubMed Central”. We used a combination of the following keywords “Brain cancer (tumor)”, “Glioma”, “Transient Receptor Potential Vanilloid type-2 (TRPV2, VRL, VRL-1, VRL1)”, “Cannabidiol (CBD)”, “Cannabinoids”, “Neurons”, “Neurogenesis”, “Glial cells”, “Glioblastoma stem/progenitor-like cells (GSCs)” to identify and select all published papers that are written in English and directly related to the focus of this review article. We reviewed both pre-clinical and clinical studies as well as their experimental methodologies, tools, and techniques that were used to generate data. Here, we reported figures and tables from studies that are relevant to the reader and align with the overall structure and goal of this paper as well.
What we know about brain tumours
Malignant gliomas, including glioblastoma (GBM), are the most widespread and devastating group of primary brain tumours in adults, with an incidence of 8.9 cases per 100,000 persons/year in the US3‘4‘5. The median survival of GBM patients ranges from 12 to 17 months under current standard treatments such as surgical resection, radiation, and chemotherapy3‘6‘7‘8‘9.
Glial tumours (gliomas) refer to a wide spectrum of malignancies, including grades II and III oligodendroglioma, grades II and III astrocytoma, and glioblastoma from initial classification based on anatomocytopathological criteria linked to the morphotypic characteristics and numerous cytonuclear atypologies, accompanied by anaplasia for high-grade glioma (GBM, grade IV)3‘10‘11. GBM is characterized by cellular heterogeneity due to the creation of tumour micro-environments resulting from glioma cell invasion. The development of abnormal angiogenesis (vascular micro-proliferation) in hypoxic and necrotic tumour areas often leads to resistance to conventional treatments and cancer recurrence3‘12‘13‘14. Microglial cells and astrocytes play a direct and critical role in promoting GBM. During GBM progression, the blood-brain barrier (BBB) becomes compromised due to a cascade of chronic inflammatory reactions, ultimately leading to dysregulation of the immune response and abnormal crosstalk between tumor cells and microglial/astrocyte cells15. This complex process involves multiple steps (e.g., chemotaxis, glial cells activation, and phenotype change), molecular factors (e.g., cytokines and chemokines secretion), and cellular components (e.g., extracellular matrix (ECM) and cytoskeletal rearrangements) that together alter the interaction between microglia/astrocytes and GBM and induce the breakdown of BBB15. The transition of microglial cells into M2 macrophages has been strongly associated with aggressive GBM and poor prognosis in GBM patients. M2 macrophages, also known as tumor-associated macrophages (TAMs), promote tumor growth, angiogenesis, and an immunosuppressive environment within the tumor microenvironment15. Moreover, extracellular vesicles (EVs) and the ATP Binding Cassette transporters (ABC transporter proteins) are the main mechanisms by which the tumor cell communicates with astrocytes/microglial cells16. Gliomas were always considered to originate from the neoplastic transformation of differentiated glial cells17. However, recent and remarkable progress made in the understanding of the pathophysiology of brain cancer brought us much-needed evidence that stem cells are directly implicated in the development of glioblastoma malignancy9. Indeed, Glioblastoma stem/progenitor-like cells (GSCs) are identified by several markers, including GFAP, and have been isolated from both human brain tumours and several glioma cell lines9.
Brain cancer involves complex mechanisms and requires a focus on potential new molecular actors to consider new treatment options, promoting cell differentiation to eliminate the tumor-driving cell population involved in glioma genesis and the acquisition of chemoresistance9. Notably, TRPV2 is expressed in both the central and peripheral nervous systems, and also expressed in the human neural progenitor cells (hNPCs) and normal human astrocytes (NHA)9. TRPV2 was localized at both mRNA and protein levels in the cerebral cortex, hypothalamus, hippocampus, caudate, and cerebellum9. It was suggested that TRPV2 plays a role in axon outgrowth because its expression is mainly concentrated in myelinated A- and C-fiber sensory neurons9. Especially, in developing growth cones, there is evidence of TRPV2 activation and an increase in intracellular Ca2+ concentration due to mechanical membrane stretch during axonal outgrowth9‘18. NGF was considered necessary for axon outgrowth mediated by TRPV2 activation through membrane stretch, since no axon outgrowth was observed in developing neurons upon removal of NGF from the culture medium18. Later on, it was demonstrated that neurite outgrowth involves upregulation of TRPV2 protein content and intracellular Calcium levels in developing neurons by NGF via the mitogen-activated protein kinase (MAPK) signaling pathway19. This process begins by binding NGF to TrkA at the end of neurites and forming the NGF/TrkA complex that will be trafficked to the nucleus via the signalling endosomes. Then, a cascade of events at the nucleus triggers phosphorylation of TRPV2 by Erk2 phosphorylation sites located at N and C termini of TRPV2, an increase in TRPV2 expression and Ca2+ activity, leading to neurite outgrowth enhancement19.
TRPV2 is among emerging and underexplored targets, belonging to a superfamily of non-selective cation channels which are involved in various physiological functions, and for the most part sustain calcium homeostasis and calcium signalling. Calcium-dependent mechanisms regulate brain tumour cell survival, proliferation, and invasion, making the TRPV2 channel a putative potent modulator of brain tumorigenesis and glioma progression by possibly regulating GSCs differentiation1‘20.
TRPV2 identity and structure Other names: VRL, VRL-1, VRL1 HGNC (Hugo): TRPV2
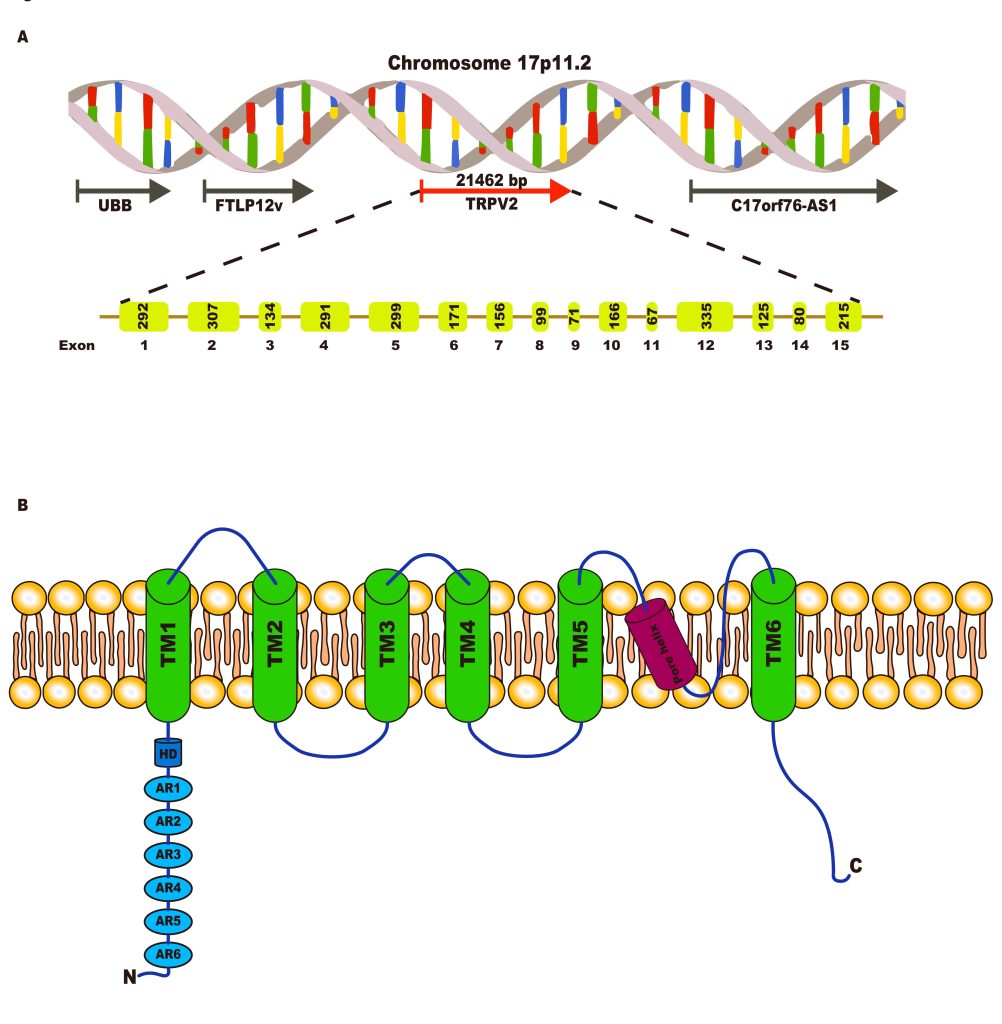
Chromosome location The gene encoding TRPV2 is located on chromosome 17 (17p11.2), located ![]() 10 Mb apart from TRPV1, and colocalizes upstream with UBB (ubiquitin B) and FTLP12 (ferritin, light polypeptide pseudogene 12) and downstream with C17orf76-AS1 (C17orf76 antisense RNA 1) (non-protein coding) (Figure 1A).
10 Mb apart from TRPV1, and colocalizes upstream with UBB (ubiquitin B) and FTLP12 (ferritin, light polypeptide pseudogene 12) and downstream with C17orf76-AS1 (C17orf76 antisense RNA 1) (non-protein coding) (Figure 1A).
DNA/RNA The TRPV2 gene consists of 15 exons and 14 introns spanning 21462 bp of DNA and includes a 5′-/3′-non-coding regions. The TRPV2 mRNA product length is 2829 bp, and analysis of its alternative splice variants showed the existence of a larger 2.3-kbp product as the full-length TRPV2 (f-TRPV2) and a smaller 1.9-kbp product as a novel splice-variant of TRPV2 (s-TRPV2), lacking exons 10 and 11, corresponding to the pore region and the fifth and sixth transmembrane domains21.
Protein TRPV2 functions physiologically as a tetrameric protein, with each monomer containing 761 residues, with a large N-terminal region of 389 amino acids, a smaller 250-residue trans-membrane (TM) domain, and a 122-residue C-terminal region. The channel structure, recently resolved by cryo-electron microscopy22‘23, shows six transmembrane spanning domains, a putative highly conserved pore-loop region located between transmembrane domains 5 and 6, and cytoplasmic amino- and carboxy termini24‘25. The transmembrane segment 6 (S6) is involved in gate opening with a rotation of the ankyrin-repeat domain coupled with pore opening via the TRP domain23‘25. The 3D structure of TRPV2 reveals “hanging gondola architecture” with a calmodulin binding site present in the C-terminal of TRPV2 (654-683) and six ankyrin repeats present in the N-terminal tail, and may play a role in the interaction between subunits of TRPV226 (Figure 1B). f-TRPV2 and s-TRPV2 protein products were recently characterized by the immunoblot technique2.
Regulation TRPV2 is a homotetrameric N-glycosylated protein that is largely located in the endoplasmic reticulum compartment under unstimulated conditions. However, a ligand (mechanical stress) that stimulates the activity of phosphatidylinositol 3-kinase (PI3K) triggers TRPV2 translocation to the plasma membrane, where it functions as a cation channel (Figure 2). The putative (exogenous) activators and inhibitors of TRPV2 are listed in Table Patch-clamp electrophysiology studies showed a non-selective permeability (P) of TRPV2 to Ca2+ > Mg2+ > Na+ ![]() Cs+
Cs+ ![]() K+ (PCa2+ / PNa+ = 2.94; PMg2+ / PNa+ = 2.40)24. The ratio of the functional full-length TRPV2 isoform (f-TRPV2) and the short splice variant (s-TRPV2) that is poreless and non-functional appears to control the biological function and oncogenicity of TRPV22‘25‘27. Especially, TRPV2 was identified as an IGF-regulated channel that translocates from intracellular compartments to the plasma membrane in response to IGF-I using green fluorescent protein (GFP)–tagged TRPV2 (TRPV2-GFP)28. Nevertheless, it was reported elsewhere that IGF-1 had little to no effect on trafficking and plasma membrane expression of TRPV2 using custom TRPV2 antibodies29. These contradictory findings might be due to the use of different detection methods and or different cell systems.
K+ (PCa2+ / PNa+ = 2.94; PMg2+ / PNa+ = 2.40)24. The ratio of the functional full-length TRPV2 isoform (f-TRPV2) and the short splice variant (s-TRPV2) that is poreless and non-functional appears to control the biological function and oncogenicity of TRPV22‘25‘27. Especially, TRPV2 was identified as an IGF-regulated channel that translocates from intracellular compartments to the plasma membrane in response to IGF-I using green fluorescent protein (GFP)–tagged TRPV2 (TRPV2-GFP)28. Nevertheless, it was reported elsewhere that IGF-1 had little to no effect on trafficking and plasma membrane expression of TRPV2 using custom TRPV2 antibodies29. These contradictory findings might be due to the use of different detection methods and or different cell systems.
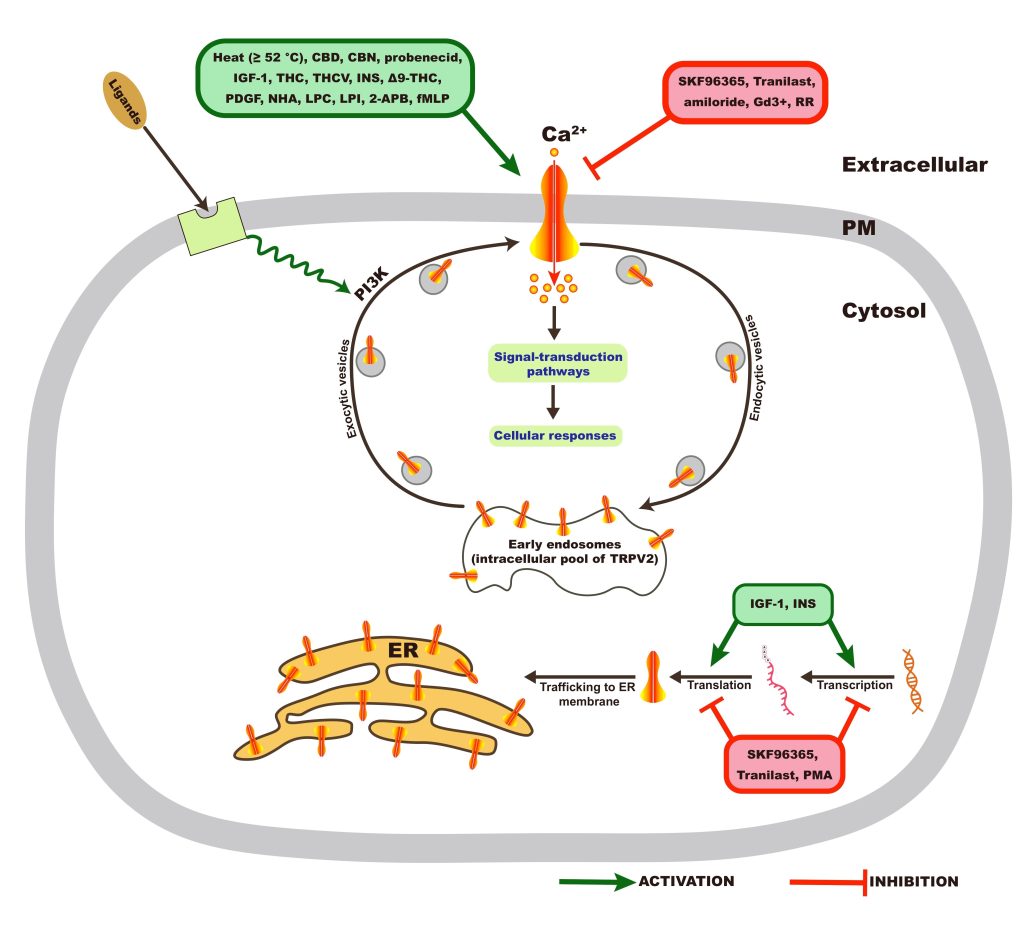
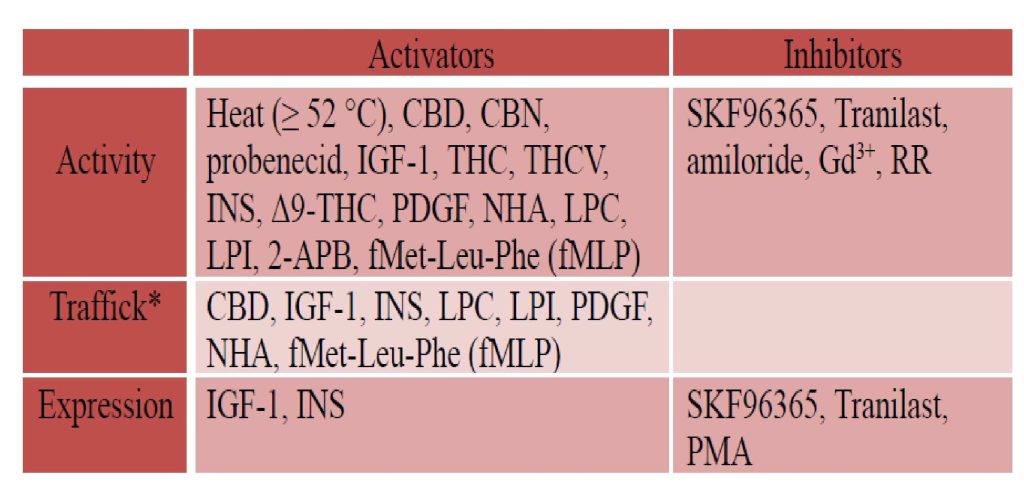
*Translocation of TRPV2 from cytoplasm to plasma membrane, CBD: Cannabidiol, THC: (-)-trans-∆9–tetrahydrocannabidol, CBN: Cannabinol, THCV: ∆9–tetrahydro- cannabivarin, NHA: neuropeptide head activator, PDGF: Platelet-derived growth factor, IGF-1: insulin-like growth factor-1, PMA: Phorbol-12-myristate-13-acetate, RR: Ruthenium red RR, Gd3+: Gadolinium, LPC: Lysophosphatidylcholine, LPI: Lysopho- sphatidylinositol, 2-APB: 2-Aminoethoxydiphenyl borate, fMLP: fMet-Leu-Phe, INS: Insulin, Tranilast: N-[3,4-dimethoxycinnamonyl]-anthranilic acid, SKF96365: 1-[2-(4-Methoxyphenyl)-2-[3-(4-methoxyphenyl)propoxy]ethyl-1H-imidazole. The information reported in this table is from reference1.
TRPV2 oncogenicity in brain cancer is driven by alteration of its expression profile
Rising evidence points to negative control of the survival and proliferation of glioblastoma (GBM) by TRPV230‘31‘32. A progressive decline in TRPV2 expression was observed as the histological stage of the disease increased30‘32. TRPV2 silencing increased proliferation and resistance to apoptosis in the high-grade U87MG glioma cell line, which displays a predominant mesenchymal stem cell (MSC) phenotype30, whereas overexpression of TRPV2 enhanced differentiation of glioblastoma stem-like cells (GSCs) and reduced tumour size and viability33‘34. Indeed, the control of GSCs’ phenotype by TRPV2-mediated Ca2+ activity was shown to determine the fate of GBM. TRPV2 silencing or inhibition with RR markedly reduced the expression of differentiation markers glial fibrillary acidic protein (GFAP) and class III ![]() -tubulin in GSCs. Similarly, PMA treatment reduced TRPV2 expression levels, inhibited astroglial differentiation, and promoted GSC proliferation. On the other hand, overexpression of TRPV2 promoted the differentiation and inhibited the proliferation of GSCs33‘34. The injection of TRPV2-overexpressing GSCs in a xenograft mouse model reduced tumour growth due to cell cycle arrest and increased glial differentiation33‘34. The Acute Myeloid Leukemia Transcription Factor Aml1/Runx1, which directly interacts with TRPV2, was shown to regulate the Proliferation and Differentiation of GSCs35. In this regard, upregulation of spliced variant Aml-1a was observed during GSC differentiation. Notably, knockdown of AML-1a by RNA interference during GSC differentiation reduced the expression of differentiation markers (e.g., GFAP,
-tubulin in GSCs. Similarly, PMA treatment reduced TRPV2 expression levels, inhibited astroglial differentiation, and promoted GSC proliferation. On the other hand, overexpression of TRPV2 promoted the differentiation and inhibited the proliferation of GSCs33‘34. The injection of TRPV2-overexpressing GSCs in a xenograft mouse model reduced tumour growth due to cell cycle arrest and increased glial differentiation33‘34. The Acute Myeloid Leukemia Transcription Factor Aml1/Runx1, which directly interacts with TRPV2, was shown to regulate the Proliferation and Differentiation of GSCs35. In this regard, upregulation of spliced variant Aml-1a was observed during GSC differentiation. Notably, knockdown of AML-1a by RNA interference during GSC differentiation reduced the expression of differentiation markers (e.g., GFAP, ![]() -III-tubulin) and triggered stemness, pluripotency, and proliferation35.
-III-tubulin) and triggered stemness, pluripotency, and proliferation35.
Effect of CBD-induced TRPV2 activation on Brain cancer
The cannabis plant (i.e., Cannabis sativa L.) contains various chemicals that are known to exert a pharmacological effect on the brain. The most notable category of bioactive molecules found in this plant includes the cannabinoids, which interact directly with cannabinoid receptors, G-protein coupled receptors (cannabinoid receptor 1, CB1, and cannabinoid receptor 2, CB2), ligand-gated ion channels (i.e. vanilloid cell surface channels) and nuclear receptors (i.e. peroxisome proliferator-activated receptor gamma, PPAR![]() ) comprising the endogenous endocannabinoid system (ECS)36‘37. The three major classes of cannabinoids are Phytocannabinoids (plant-based), such as ∆9-Tetrahydrocannabinol (∆9-THC) and Cannabidiol (CBD), Endocannabinoids (or endogenous cannabinoids) which include Anandamide (AEA) and 2-arachidonoylglycerol (2-AG), and synthetic cannabinoids that mimic the cannabinoid groups (1) and (2) (see diagram below).
) comprising the endogenous endocannabinoid system (ECS)36‘37. The three major classes of cannabinoids are Phytocannabinoids (plant-based), such as ∆9-Tetrahydrocannabinol (∆9-THC) and Cannabidiol (CBD), Endocannabinoids (or endogenous cannabinoids) which include Anandamide (AEA) and 2-arachidonoylglycerol (2-AG), and synthetic cannabinoids that mimic the cannabinoid groups (1) and (2) (see diagram below).

Among more than 120 Phytocannabinoids isolated from Cannabis sativa L., Cannabidiol (CBD) is the most abundant non-psychoactive compound that can stimulate TRPV2 channel activity37‘38.
Since CBD interacts with a wide variety of molecular targets in the brain, its therapeutic potential has been investigated in several neuropsychiatric diseases, including anxiety and mood disorders. Specifically, CBD has received growing attention due to its anxiolytic and antidepressant properties39. Given its safety profile, CBD is considered a promising new agent in the treatment of anxiety and mood disorders. In the present review, we provide a summary of cellular and animal-based studies that support the use of CBD as an anti-brain cancer agent through activation of the TRPV2 channel.
TRPV2 activation was reported to promote GSCs differentiation and inhibition of gliomagenesis9‘40‘37. For example, the TRPV2 agonist CBD was shown to induce GSC differentiation by inhibiting their clonogenic capacity40‘37‘38‘41. In fact, treatment of GSCs with CBD triggered downregulation of genes involved in chemoresistance (e.g., BCL-XL, CTDS) and upregulation of genes involved in apoptosis (e.g., BAD and BAX)37‘42. Moreover, CBD induced glioma cell chemosensitivity and apoptosis by increasing uptake of the cytotoxic drug doxorubicin43‘40‘42. CBD was also found to increase GSC chemosensitivity to other conventional anti-cancer drugs such as carmustine (BCNU)44. Combining Temozolomide (TMZ) with THC or with THC plus CBD was reported to substantially reduce tumour growth in glioma xenografts44. The pore region of TRPV2, critical for its Ca2+ activity, was required for boosting glioma chemosensitivity to cytotoxic drugs44. In patients with high-grade glioma, CBD has been administered in conjunction with chemoradiation therapy to improve outcome and survival45. In this regard, CBD was found to upregulate the expression of the transcription factor acute myeloid leukemia-1 (AML-1A) isoform, which is involved in GSC differentiation. Interestingly, the downregulation of AML-1A, which binds TRPV2 gene promoters, led to the restoration of the undifferentiated (immature) phenotype of mature GSCs39‘37. Further evidence linking TRPV2 to AML-1A in controlling the fate of GSC differentiation was provided by the elegant study of Nabissi and his co-workers (Int J Cancer. 2015). They demonstrated that CBD treatment induced TRPV2 activation and binding of AML-1A to TRPV2 promoters. Next, upregulation of AML-1A inhibited GSC proliferation and promoted differentiation and autophagy through a PI3K/AKT-dependent pathway46. Herein, they established a mechanism involving TRPV2, AML-1A, and autophagy in eliminating GSCs’ proliferation and clonogenic capacity. We summarized in Table 2 the effects of CBD and other structurally similar compounds on brain cancer cells reported in various pre-clinical studies37.
TRPV2 is highly expressed in neighboring cells of the BBB. The TRPV2 agonist, CBD, was shown to increase the transendothelial electrical resistance (TEER) of the BBB and prevent the increase in its permeability, thereby exhibiting a protective role of the BBB likely through direct TRPV2 activation47. Recently, CBD was shown to stimulate microglial phagocytosis through TRPV2 activation and increased expression48‘49.
Moreover, a possible relationship between Microglia phagocytic capacity and TRPV2 channel expression and activity was also reported elsewhere50. Thus, TRPV2 activation and overexpression may play a protective role for the BBB in gliomagenesis and during GBM progression, and this is worth further investigation as a therapeutic strategy.
| Cancer Cell line | Cannabinoids | Inhibitory concentrations | In vitro actions | References |
| Human Glioblastoma, U878MG, U373MG | CBD | 5–40 | Antiproliferative effects correlated to induction of apoptosis | 51 |
| Rat C6 glioma cells | ∆9-THC, CBD, CBDA, CBG, CBC, AM251, JWH-133, AM630, SR141716A, SR144528 | 0–50 | CBD most potent. CBD, CBG and CBD-A activated TRPV1 | 52 |
| Human Glioma cancer,U251, F126, U87 | ∆9-THC, CBD | 0.1–10 | Inhibition of cell proliferation, apoptosis | 53. |
| Human Glioblastoma, U87-MG, T98G | CBD | 0–20 | Decrease in cell Invasion via MMP-9, TIMP-1, TIMP-4, u-PA, PAI-1, VEGF | 54 |
| Human Glioma, T98G, U87MG, Murine Glioma, GL261 | CBD, ∆9-THC (Pure and BDS) | 0–20 | Increase in radiosensitivity associated with increase in apoptosis and autophagy | 55 |
| Human Glioblastoma, U251, 3832, 387 Primary glioma Stem cells (GSC) lines | CBD | 0–5 | Activation of p-p38 pathway, downregulation of key stem cell regulators; Sox2, Id1 and p-STAT3 | 56 |
| Human Neuroblastoma, SK-N-SH, IMR-32, NUB-6 and LAN-1 | ∆9-THC, CBD | 0–50 | Cell viability reduction and apoptosis | 57 |
| Human Glioblastoma, U87MG, Glioblastoma patient derived stem cell like cells (GIC) | ∆9-THC, CBD | 0–5 | Very significant reduction of the GIC population, induction of apoptosis | 58 |
| Human Glioma cells, U87MG (U87), A172,SW1783, U373MG (U373), T98G (T98),SW1088, and LN405 | ∆9-THC, CBD, SR141716, SR144528 | 0.9–3 | Reduction in cell viability and induction of apoptosis and autophagy | 59 |
Relationship between TRPV2 interactome expression and Brain tumour progression
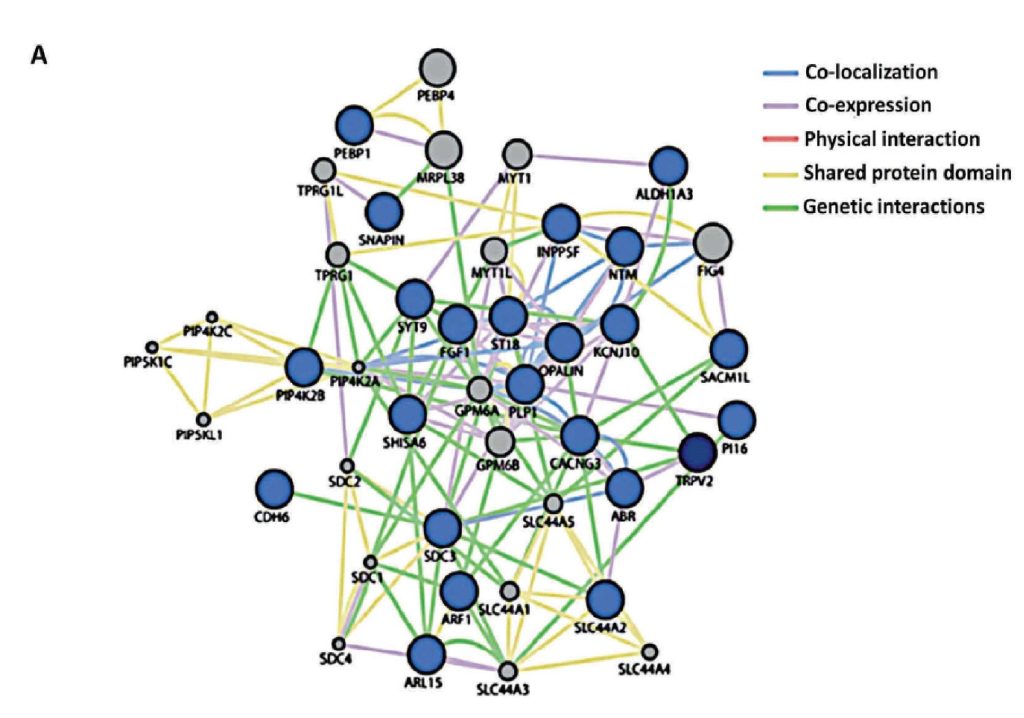
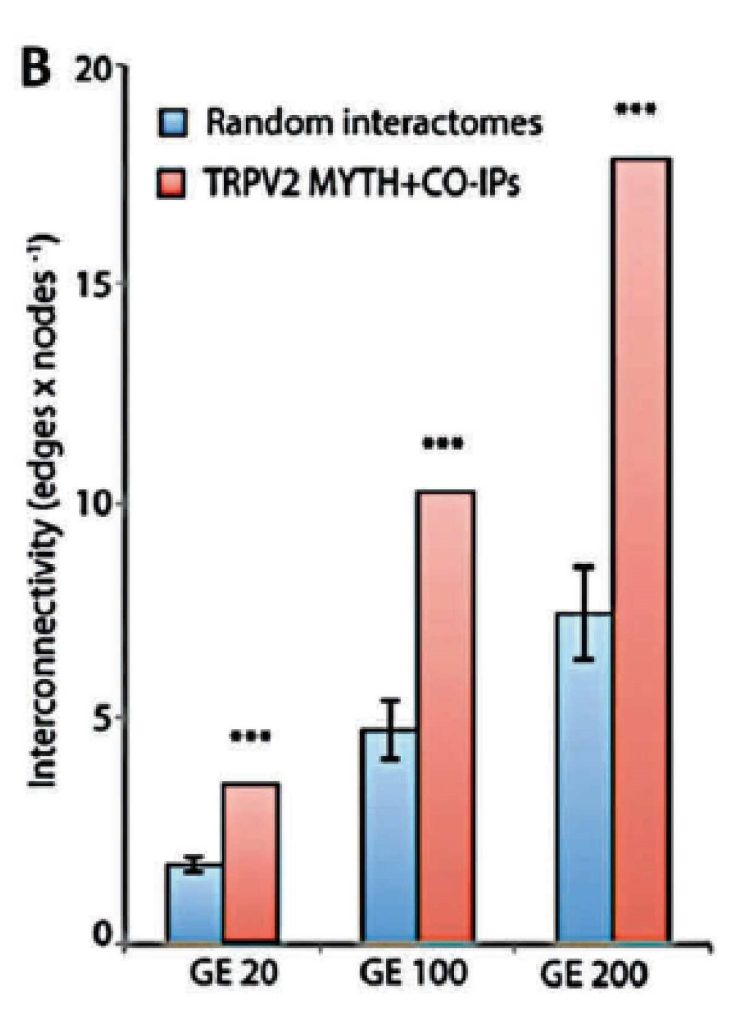
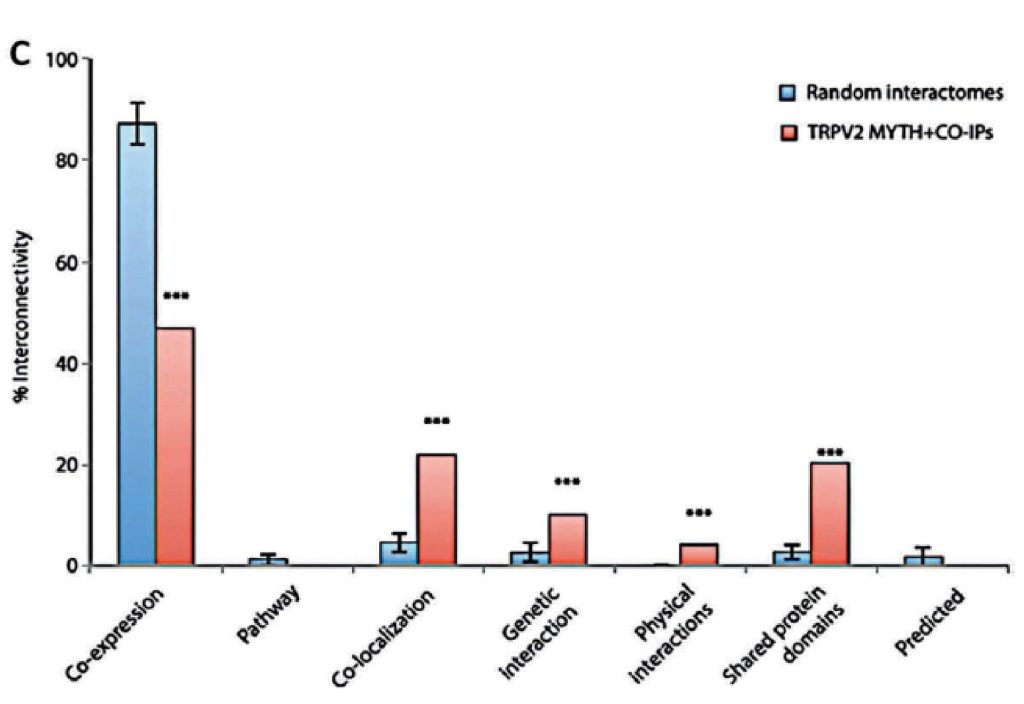
The establishment of the TRPV2 interactome (Figure 3)60. raised questions and issues but also vopened opportunities to discover new biomarkers and therapeutic targets in GBM. The strongest interactors with TRPV2 were ABR, ARL15, NTM, Opalin, SACM1L, and ST18 proteins60. A high TRPV2 interactome protein expression correlated with greater tumour progression, recurrence, TMZ-resistance, and poor prognosis of GBM patients61. Therefore, the TRPV2 interactome-based signature permits discrimination between high- and low-risk GBM, in terms of overall survival, with less survival in GBM patients expressing the TRPV2 protein interactome.
.
Conclusions
The oncogenic activity of TRPV2 in brain cancer is mainly associated with deregulation of its expression profile. Though the possibility of using TRPV2 in cancer therapy is still in its infancy, TRPV2 represents a novel, promising pharmacologic/molecular target, especially in the management of high-grade glioma development. Besides being a potential anti-tumour target, the TRPV2 channel may play a role as a “drug carrier” in cancer therapy, facilitating via the central pore chemotherapy drug uptake, thus improving the efficacy of cancer therapies. CBD is in the list of new promising anti-cancer compounds, since it has been shown to inhibit GBM growth by stimulating glial differentiation and decreasing the GSCs’ efficiency in glioma formation. In fact, CBD, via TRPV2 activation, triggers the GSC differentiation by activating an autophagic process, inhibiting the GSCs’ clonogenic capability, and thereby promoting cell death and enhancement of chemosensitivity in human GBM. Clinical studies are needed to identify cancer populations that may benefit from TRPV2 as a molecular biomarker and perhaps will open new prospects to explore TRPV2 as a novel therapeutic target in brain cancer and metastasis.
References
- K.S. Siveen, P.B. Nizamuddin, S. Uddin, M. Al-Thani, M.P. Frenneaux, I.A. Janahi, M. Steinhoff, F. Azizi. TRPV2: A Cancer Biomarker and Potential Therapeutic Target. Dis Markers Dec 10, (2020). [↩] [↩] [↩] [↩] [↩]
- K.S. Siveen, K.S. Prabhu, A.S. Parray, M. Merhi, A. Arredouani, M. Chikri, S. Uddin, S. Dermime, R.M. Mohammad, M. Steinhoff, I.A. Janahi, F. Azizi. Evaluation of cationic channel TRPV2 as a novel biomarker and therapeutic target in Leukemia-Implications concerning the resolution of pulmonary inflammation. Sci Rep 9, 1554 (2019). [↩] [↩] [↩]
- Chinigò G, Castel H, Chever O, Gkika D. TRP Channels in Brain Tumors. Front Cell Dev Biol 9, 617801 (2021). [↩] [↩] [↩] [↩] [↩]
- Q.T. Ostrom, H. Gittleman, P. Liao, T. Vecchione-Koval, Y. Wolinsky, C. Kruchko, J.S. Barnholtz-Sloan. CBTRUS statistical report: primary brain and other central nervous system tumors diagnosed in the United States in 2010–2014. Neuro Oncol 19 (suppl_5), v1-v88 (2017). [↩]
- Q.T. Ostrom, H. Gittleman, J. Fulop, M. Liu, R. Blanda, C. Kromer, Y. Wolinsky, C. Kruchko, J.S. Barnholtz-Sloan. CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2008-2012. Neuro Oncol 17, Suppl 4 (Suppl 4), iv1–iv62 (2015). [↩]
- P.Y. Wen, A.A. Brandes. Treatment of recurrent high-grade gliomas. Curr Opin Neurol 22, 657–664 (2009). [↩]
- R. Stupp, M.E. Hegi, W.P. Mason, M.J. van den Bent, M.J.B. Taphoorn, R.C. Janzer, S.K. Ludwin, A. Allgeier, B. Fisher, K. Belanger, P. Hau, A.A. Brandes, J. Gijtenbeek, C. Marosi, C.J. Vecht, K. Mokhtari, P. Wesseling, S. Villa, E. Eisenhauer, T. Gorlia, M. Weller, D. Lacombe, J.G. Cairncross, RO. Mirimanoff. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomized phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol 10, 459–466 (2009). [↩]
- R. Stupp, W.P. Mason, M.J. van den Bent, M. Weller, B. Fisher, M.J.B. Taphoorn, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, R.C. Janzer, S.K. Ludwin, T. Gorlia, A. Allgeier, D. Lacombe, J.G. Cairncross, E. Eisenhauer, RO. Mirimanoff. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352, 987–996 (2005). [↩]
- G. Santoni, C. Amantini. The transient receptor potential vanilloid type-2 (TRPV2) ion channels in neurogenesis and gliomagenesis: crosstalk between transcription factors and signalling molecules. Cancers 11, p11-E322 (2019). [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- D.N. Louis, H. Ohgaki, O.D. Wiestler, W.K. Cavenee, P.C. Burger, A. Jouvet, B.W. Scheithauer, P. Kleihues. The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol 114, 97–109 (2007). [↩]
- C.R. Miller, A. Perry. Glioblastoma. Arch Pathol Lab Med 131, 397–406 (2007). [↩]
- A. Prabhu, P. Kesarwani, S. Kant, S.F. Graham, P. Chinnaiyan. Histologically defined intratumoral sequencing uncovers evolutionary cues into conserved molecular events driving gliomagenesis. Neuro Oncol. 19, 1599–1606 (2017). [↩]
- C. Alifieris, D.T. Trafalis. Glioblastoma multiforme: pathogenesis and treatment. Pharmacol. Ther 152, 63–82 (2015). [↩]
- M. Karsy, M. Gelbman, P. Shah, O. Balumbu, F. Moy, E. Arslan. Established and emerging variants of glioblastoma multiforme: review of morphological and molecular features. Folia Neuropathol 50, 301–321 (2012). [↩]
- D. Matias, J. Balça-Silva, G.C. da Graça, C.M. Wanjiru, L.W. Macharia, C.P. Nascimento, N.R. Roque, J.M. Coelho-Aguiar, C.M. Pereira, M.F. Dos Santos, L.S. Pessoa, F.R.S. Lima, A. Schanaider, V.P. Ferrer; Tania Cristina Leite de Sampaio e Spohr; V. Moura-Neto. Microglia/Astrocytes-Glioblastoma Crosstalk: Crucial Molecular Mechanisms and Microenvironmental Factors. Front Cell Neurosci 12, 235 (2018). [↩] [↩] [↩]
- D. Matias, J. Balça-Silva, G.C. da Graça,C.M. Wanjiru, L.W. Macharia, C.P. Nascimento, N.R. Roque, J.M. Coelho-Aguiar, C.M. Pereira, M.F. Dos Santos, L.S. Pessoa, F.R.S. Lima, A. Schanaider, V.P. Ferrer; Tania Cristina Leite de Sampaio e Spohr; V. Moura-Neto. Microglia/Astrocytes-Glioblastoma Crosstalk: Crucial Molecular Mechanisms and Microenvironmental Factors. Front Cell Neurosci 12, 235 (2018). [↩]
- G. Santoni, C. Amantini. The transient receptor potential vanilloid type-2 (TRPV2) ion channels in neurogenesis and gliomagenesis: crosstalkbetween transcription factors and signalling molecules. Cancers 11, p11-E322 (2019). [↩]
- K. Shibasaki, N. Murayama, K. Ono, Y. Ishizaki, M. Tominaga. TRPV2 enhances axon outgrowth through its activation by membrane stretch in developing sensory and motor neurons. J Neurosci 30, 4601-4612 (2010). [↩] [↩]
- M.R. Cohen, W.M. Johnson, J.M. Pilat, J. Kiselar, A. DeFrancesco-Lisowitz, R.E. Zigmond, V.Y. Moiseenkova-Bell. Nerve Growth Factor Regulates Transient Receptor Potential Vanilloid 2 via Extracellular Signal-Regulated Kinase Signaling To Enhance Neurite Outgrowth in Developing Neurons. Mol Cell Biol 35, 4238-4252 (2015). [↩] [↩]
- G. Chinigò, H. Castel, O. Chever, D. Gkika. TRP Channels in Brain Tumors. Front Cell Dev Biol 9, 617801 (2021). [↩]
- S. Caprodossi, R. Lucciarini, C. Amantini, M. Nabissi, G. Canesin, P. Ballarini, A. Di Spilimbergo, M.A. Cardarelli, L. Servi, G. Mammana, G. Santoni. Transient receptor potential vanilloid type 2 (TRPV2) expression in normal urothelium and in urothelial carcinoma of human bladder: correlation with the pathologic stage. Eur Urol 54, 612–620 (2008). [↩]
- K.W. Huynh, M.R. Cohen, J. Jiang, A. Samanta, D.T. Lodowski, Z.H. Zhou, V.Y. Moiseenkova-Bell. Structure of the full-length TRPV2 channel by cryo-EM. Nat Commun 7, 11130 (2016). [↩]
- L. Zubcevic, M.A. Herzik, B.C. Chung, Z. Liu, G.C. Lander, S.Y. Lee. Cryo-electron microscopy structure of the TRPV2 ion channel. Nat Struct Mol Biol 23, 180–186 (2016). [↩] [↩]
- M.J. Caterina, T.A. Rosen, M. Tominaga, A.J. Brake, D.A. Julius. Capsaicin-receptor homologue with a high threshold for noxious heat. Nature 398, 436–441 (1999). [↩] [↩]
- A. Perálvarez-Marín, P. Doñate-Macian, R. Gaudet. What do we know about the transient receptor potential vanilloid 2 (TRPV2) ion channel? FEBS J 280, 5471–5487 (2013). [↩] [↩] [↩]
- K.W. Huynh, M.R. Cohen, S. Chakrapani, H.A. Holdaway, P.L. Stewart, V.Y. Moissenkova-Bell. Structural insights into the assembly of TRPV channels. Structure 22, 1–9 (2014). [↩]
- G. Santoni, G. Farfariello, C. Amantini. Chapter 49: TRPV channels in tumor growth and progression. (ed. Islam, M. S.). Transient receptor potential channels. Adv Exp Med Biol 704 (2011). [↩]
- M. Kanzaki, Y.Q. Zhang, H. Mashima, L. Li, H. Shibata, I. Kojima. Translocation of a calcium-permeable cation channel induced by insulin-like growth factor-I. Nat Cell Biol 1, 165-170 (1999). [↩]
- M.R. Cohen, K.W. Huynh, D. Cawley, V.Y. Moiseenkova-Bell. Understanding the cellular function of TRPV2 channel through generation of specific monoclonal antibodies. PLoS One 8, e85392 (2013). [↩]
- M. Nabissi, M.B. Morelli, C. Amantini, V. Farfariello, L. Ricci-Vitiani, S. Caprodossi, A. Arcella, M. Santoni, F. Giangaspero, R. De Maria, G. Santoni. TRPV2 channel negatively controls glioma cell proliferation and resistance to Fas-induced apoptosis in ERK-dependent manner. Carcinogenesis 31, 794-803 (2010). [↩] [↩] [↩]
- M. Alptekin, S. Eroglu, E. Tutar, S. Sencan, M.A. Geyik, M. Ulasli, A.T. Demiryurek, C. Camci. Gene expressions of TRP channels in glioblastoma multiforme and relation with survival. Tumour Biol 36, 9209–13 (2015). [↩]
- J. Zheng, F. Liu, S. Du, M. Li, T. Wu, X. Tan, W. Cheng. Mechanism for regulation of melanoma cell death via activation of thermo-TRPV4 and TRPV2. J Oncol 2019, 7362875 (2019). [↩] [↩]
- M.B. Morelli, M. Nabissi, C. Amantini, V. Farfariello, L. Ricci-Vitiani, S. di Martino, R. Pallini, L.M. Larocca, S. Caprodossi, M. Santoni, R. De Maria, G. Santoni. The transient receptor potential vanilloid-2 cation channel impairs glioblastoma stem-like cell proliferation and promotes differentiation. Int J Cancer 131, E1067-E1077 (2012). [↩] [↩] [↩]
- R.C. Gimple, S. Bhargava, D. Dixit, J.N. Rich. Glioblastoma stem cells: lessons from the tumor hierarchy in a lethal cancer. Genes Dev 33, 591-609 (2019). [↩] [↩] [↩]
- G. Santoni, C. Amantini, F. Maggi, O. Marinelli, M. Santoni, M. Nabissi, M.B. Morelli. The TRPV2 cation channels: from urothelial cancer invasiveness to glioblastoma multiforme interactome signature. Lab Invest 100, 186-198 (2020). [↩] [↩]
- P.A. Melas, M. Scherma, W. Fratta, C. Cifani, P. Fadda. Cannabidiol as a Potential Treatment for Anxiety and Mood Disorders: Molecular Targets and Epigenetic Insights from Preclinical Research. Int J Mol Sci 22, 1863 (2021). [↩]
- N. Mangal, S. Erridge, N. Habib, A. Sadanandam, V. Reebye, M.H. Sodergren. Cannabinoids in the landscape of cancer. J Cancer Res Clin Oncol 147, 2507-2534 (2021). [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- T. Aguado, A. Carracedo, B. Julien, G. Velasco, G. Milman, R. Mechoulam, L. Alvarez, M. Guzmán, I. Galve-Roperh. Cannabinoids induce glioma stem-like cell differentiation and inhibit gliomagenesis. J Biol Chem 282, 6854–6862 (2007). [↩] [↩]
- P.A. Melas, M. Scherma, W. Fratta, C. Cifani, P. Fadda. Cannabidiol as a Potential Treatment for Anxiety and Mood Disorders: Molecular Targets and Epigenetic Insights from Preclinical Research. Int J Mol Sci 22, 1863 (2021). [↩] [↩]
- M.B. Morelli, M. Nabissi, C. Amantini, V. Farfariello, L. Ricci-Vitiani, S. di Martino, R. Pallini, L.M. Larocca, S. Caprodossi, M. Santoni, R. De Maria, G. Santoni. The transient receptor potential vanilloid-2 cation channel impairs glioblastoma stem-like cell proliferation and promotes differentiation. Int J Cancer 131, E1067–E1077 (2012). [↩] [↩] [↩]
- M. Nabissi, M.B. Morelli, M. Santoni, G. Santoni. Triggering of the TRPV2 channel by cannabidiol sensitizes glioblastoma cells to cytotoxic chemotherapeutic agents. Carcinogenesis 34, 48–57 (2013). [↩]
- X. Gong, P.H. Schwartz, M.E. Linskey, D.A. Bota. Neural stem/progenitors and glioma stem-like cells have differential sensitivity to chemotherapy. Neurology 76, 1126–1134 (2011). [↩] [↩]
- A. Prabhu, P. Kesarwani, S. Kant, S.F. Graham, P. Chinnaiyan. Histologically defined intratumoral sequencing uncovers evolutionary cues into conserved molecular events driving gliomagenesis. Neuro Oncol 19, 1599–1606 (2017). [↩]
- S. Torres, M. Lorente, F. Rodríguez-Fornés, S. Hernández-Tiedra, M. Salazar, E. García-Taboada, J. Barcia, M. Guzmán, G. Velasco. A combined preclinical therapy of cannabinoids and temozolomide against glioma. Mol Cancer Ther 10, 90–103 (2011). [↩] [↩] [↩]
- P.B. Dall’Stella, M.F.L. Docema, M.V.C. Maldaun, O. Feher, C.L.P. Lancellotti. Case Report: Clinical Outcome and Image Response of Two Patients with Secondary High-Grade Glioma Treated with Chemoradiation, PCV, and Cannabidiol. Front Oncol 8, 643 (2019). [↩]
- M. Nabissi, M.B. Morelli, C. Amantini, S. Liberati, M. Santoni, L. Ricci-Vitiani, R. Pallini, G. Santoni. Cannabidiol stimulates Aml-1a-dependent glial differentiation and inhibits glioma stem-like cells proliferation by inducing autophagy in a TRPV2-dependent manner. Int J Cancer 137, 1855-1869 (2015). [↩]
- H. Luo, X. Declèves, S. Cisternino. Transient Receptor Potential Vanilloid in the Brain Gliovascular Unit: Prospective Targets in Therapy. Pharmaceutics 13,334 (2021). [↩]
- S. Yang, Y. Du, X. Zhao, Q. Tang, W. Su, Y. Hu, P. Yu. Cannabidiol Enhances Microglial Beta-Amyloid Peptide Phagocytosis and Clearance via Vanilloid Family Type 2 Channel Activation. Int J Mol Sci 23, 5367 (2022). [↩]
- S. Hassan, K. Eldeeb, P.J. Millns, A.J. Bennett, S.P. Alexander, D.A. Kendall. Cannabidiol enhances microglial phagocytosis via transient receptor potential (TRP) channel activation. Br J Pharmacol 171, 2426-2439 (2014). [↩]
- M.J.E. Maksoud, V. Tellios, D. An, Y.Y. Xiang, W.Y. Lu. Nitric oxide upregulates microglia phagocytosis and increases transient receptor potential vanilloid type 2 channel expression on the plasma membrane. Glia 67, 2294-2311 (2019). [↩]
- E. Singer, J. Judkins, N. Salomonis, L. Matlaf, P. Soteropoulos, S. McAllister, L. Soroceanu. Reactive oxygen species-mediated therapeutic response and resistance in glioblastoma. Cell Death Dis 6, e1601 (2015). [↩]
- A. Ligresti, A.S. Moriello, K. Starowicz, I. Matias, S. Pisanti, L. De Petrocellis, C. Laezza, G. Portella, M. Bifulco, V. Di Marzo. Antitumor activity of plant cannabinoids with emphasis on the effect of cannabidiol on human breast carcinoma. J Pharmacol Exp Ther 318, 1375–1387 (2006). [↩]
- Z. Qamri, A. Preet, M.W. Nasser, C.E. Bass, G. Leone, S.H. Barsky, R.K. Ganju. Synthetic cannabinoid receptor agonists inhibit tumor growth and metastasis of breast cancer. Mol. Cancer Ther. 8, 3117–3129 (2009). [↩]
- M. Solinas, P. Massi, V. Cinquina, M. Valenti, D. Bolognini, M. Gariboldi, E. Monti, T. Rubino, D. Parolaro. Cannabidiol, a nonpsychoactive cannabinoid compound, inhibits proliferation and invasion in U87-MG and T98G glioma cells through a multitarget effect. PLoS One 8, e76918 (2013). [↩]
- K.A. Scott, A.G. Dalgleish, W.M. Liu. The combination of cannabidiol and D 9-tetrahydrocannabinol enhances the anticancer effects of radiation in an orthotopic murine glioma model. Mol Cancer Ther 13, 2955-2967 (2014). [↩]
- E. Singer, J. Judkins, N. Salomonis, L. Matlaf, P. Soteropoulos, S. McAllister, L. Soroceanu. Reactive oxygen species- mediated therapeutic response and resistance in glioblastoma. Cell Death Dis 6, e1601 (2015). [↩]
- T. Fisher, H. Golan, G. Schiby, S. PriChen, R. Smoum, I. Moshe, N. Peshes-Yaloz, A. Castiel, D. Waldman, R. Gallily, R. Mechoulam, A. Toren. In vitro and in vivo efficacy of non- psychoactive cannabidiol in neuroblastoma. Curr Oncol 23, S15-22 (2016). [↩]
- I. Lopez-Valero, C. Saiz-Ladera, S. Torres, S. Hernandez-Tiedra, E. Garcia-Taboada, F. Rodriguez-Fornes, M. Barba, D. Davila, N. Salvador-Tormo, M. Guzman, J.M. Sepulveda, P. Sanchez-Gomez, M. Lorente, G. Velasco. Targeting glioma initiating cells with A combined therapy of cannabinoids and temozolomide. Biochem Pharmacol 157, 266–274 (2018). [↩]
- S. Torres, M. Lorente, F. Rodríguez-Fornés, S. Hernández-Tiedra, M. Salazar, E. García- Taboada, J. Barcia, M. Guzmán, G. Velasco. A combined preclinical therapy of cannabinoids and temozolomide against glioma. Mol Cancer Ther 10, 90–103 (2011). [↩]
- P. Donate-Macian, A. Gomez, I.R. Degano, A. Peralvarez-Marin. A TRPV2 interactome-based signature for prognosis in glioblastoma patients. Oncotarget 9, 18400–18409 (2018). [↩] [↩] [↩]
- P. Donate-Macian, A.Gomez,I.R.Degano,A.Peralvarez-Marin. A TRPV2 interactome-based signature for prognosis in glioblastoma patients. Oncotarget 9, 18400–18409 (2018). [↩]