Abstract
Immunotherapy including immune checkpoint inhibitors and CAR-T cell therapy has transformed the treatment of certain cancers in the past decade. Therapeutic cancer vaccines are emerging as the next significant advance in cancer immunotherapy, as they offer a precise and personalized approach by training one’s own immune system to attack cancer cells by recognizing tumor-specific neoantigens. Improved understanding of tumor biology coupled with recent advancements in multi-omics, artificial intelligence/machine learning, and vaccine delivery technologies, are helping overcome many of the past barriers to the development of effective cancer vaccines. This paper reviews the state-of-art on therapeutic cancer vaccines, including the underlying science, challenges to their development, and the recent technological advances that help overcome these challenges. Finally, it describes a recent successful clinical trial to illustrate the promise of this exciting new treatment modality for cancer patients.
Keywords: Immunotherapy, therapeutic cancer vaccines, tumor-specific neoantigens.
Introduction
Cancer treatment has made huge strides in the last decade or so, esp. with the advent of immunotherapy, which harnesses the body’s own immune system to fight cancer. Numerous immunotherapeutic agents spanning from monoclonal antibodies including immune checkpoint inhibitors (ICI), to adoptive cell transfer (ACT) therapies including chimeric antigen receptor modified T (CAR-T) cell therapy, are currently approved by the US-FDA, and have proved transformative for the management of certain cancers1.
Despite these advances, an unmet treatment need remains. Not all patients, and not all cancers, respond to ICIs. Some cancers like pancreatic and prostate cancer often exhibit primary resistance to ICIs, while others like melanoma and non-small cell lung cancer (NSCLC) may develop acquired resistance following an initial response2. The success of CAR-T cell therapies remains confined mainly to hematological malignancies1.Hence there is a need for new treatment modalities with different mechanisms of actions, that may overcome some of the limitations of the currently approved therapies and add to the list of tractable cancers.
Cancer vaccines are also a type of immunotherapeutic agents that can potentially be used to treat cancers. While they are still largely in the research and development space, they are generating a lot of scientific interest owing to their potential to transform cancer treatment in the coming years. The objective of this scientific review is to understand the state of art, as well as the challenges and opportunities that lie ahead of us, as we strive to realize the promise of this new modality of cancer treatment.
Methodology
We conducted our literature search in 4 steps as described below. We used two databases – NCBI PubMed and clinicaltrials.gov, and confined our search to English language publications.
Cancer vaccine landscape
We began with a search for existing literature reviews on this topic, to help us define the scope of our review, using the following search strategy:
Database: NCBI PubMed
Key words: Therapeutic AND cancer AND vaccines
Filters: Article type – Review; Publication date – 2023/01/01 to 2024/12/31; Article language – English; Species – Human
This search retrieved 983 abstracts. We excluded reviews restricted to a particular cancer type or vaccine technology, to identify 5 most recent broad-based reviews that critically analyzed the totality of the information, including the concepts, reasons for the failure of first-generation vaccines, and the recent technological advances enabling the current generation.
Completed cancer vaccine trials
Then, we searched for randomized controlled trials that have assessed and reported on the efficacy of therapeutic cancer vaccines, using the following search strategy:
Database: NCBI PubMed
Key words: Therapeutic AND cancer AND vaccines
Filter: Article type – Clinical trial, Phase III
This search retrieved 164 abstracts. We reviewed each of these abstracts, and excluded the ones that did not meet the following inclusion/exclusion criteria for our review:
Inclusion Criteria: Randomized controlled trial assessing Disease-free survival (DFS)/Relapse- or Recurrence-free survival (RFS)/Progression-free survival (PFS)/Overall survival (OS) as the primary endpoint.
Exclusion Criteria: Study intervention does not target tumor-associated antigen/s, e.g., studies assessing use of antimicrobial vaccines (HPV, COVID-19, Influenza, BCG, etc.)in cancer patients.
Application of the above inclusion/exclusion criteria yielded 24 high-quality trials that reported a definitive assessment of the clinical benefit and risk of a cancer vaccine versus a comparator.
Ongoing cancer vaccine trials
Next, we searched for currently ongoing clinical trials on cancer vaccines using the following search strategy:
Database: clinicaltrials.gov
Condition/disease: Cancer OR Carcinoma OR malignant
Other terms: Vaccine
Study type: Interventional
Study status: Looking for participants (Recruiting/Not yet recruiting)
This search retrieved 342 ongoing studies. We then narrowed down our search by applying the following inclusion/exclusion criteria, which yielded 17 ongoing Phase II/III RCTs assessing long-term outcomes.
Inclusion Criteria: Study Phase – Phase II/Phase III; Allocation – Randomized; Outcome Measures – DFS/RFS/PFS/OS
Exclusion Criteria: Study intervention does not target tumor-associated antigen/s, e.g., studies assessing use of antimicrobial vaccines (HPV, COVID-19, Influenza, BCG, etc.) in cancer patients, 2. Trial does not have a standard-of-care comparator arm.
Ongoing trials on mRNA-4157/V940/Intismeran autogene vaccine
Finally, we searched for currently ongoing Phase II/III clinical trials on mRNA-4157/V940/Intismeran autogene using the following search strategy:
Database: clinicaltrials.gov
Intervention/treatment: mRNA-4157 OR V940 OR Intismeran autogene
Study type: Interventional
Study phase: Phase II and Phase III
This search yielded 8 ongoing Phase III studies on this promising cancer vaccine.
We intentionally restricted our searches for completed and ongoing trials to randomized controlled trials assessing a long-term outcome measure, because these provide a definitive assessment of whether the vaccine in question provides a clinically meaningful benefit compared to placebo or standard of care. The field of cancer vaccines is unfortunately littered with numerous examples of vaccines that showed some benefit in small trials assessing immunological or clinical response, but failed subsequently in the larger trials assessing long-term outcomes. We did not want to base our conclusions on findings from such trials, that may not be confirmed subsequently.
What are cancer vaccines?
Cancer vaccines are similar to the more commonly used vaccines against infectious diseases, in that they prime the body’s own immune system to mount an attack on cells bearing antigens of interest. In case of anti-infective vaccines, the antigen of interest is a viral or bacterial protein, while in case of cancer vaccines, it is a protein present on cancer cells, also referred to as tumor-specific antigen.
Cancer cells bear unique antigens that are not present on the body’s normal cells. These unique antigens, also referred to as ‘neoantigens’, arise as a result of genetic mutations that transform normal body cells into cancerous cells. In addition to conferring hallmark functional capabilities that are characteristic of cancer cells, these mutations may also code for altered or new proteins that are not recognized by the immune system as ‘self’. Thus, cancer cells produce ‘neoantigens’ that are antigenically novel for the host, and can therefore be targeted by cancer vaccines to target those cells3. Some cancer vaccines may target tumor associated antigens (TAA), which are autoantigens expressed in normal tissues, but overexpressed in various cancers4.
When a cancer vaccine containing the selected tumor-specific antigen is administered to a patient, the antigen is taken up by the circulating antigen presenting cells (APCs; typically, dendritic cells) to be processed and bound to major histocompatibility complex (MHC) molecules. The dendritic cells circulate to the lymph nodes to present the MHC-bound antigen to T lymphocytes, leading to the activation of T helper and T cytotoxic cells. The activated T cells then circulate back through the blood to the tumor, where they attack and destroy the tumor cells4.
Off-the-shelf vs personalized vaccines
Off-the-shelf vaccines
As the term suggests, these vaccines are manufactured and kept ready for use whenever patients require them. They use ‘shared antigens’ that arise from mutations in genes like tumor protein 53 (TP53) and Kirsten rat sarcoma (KRAS), which are common across different cancer types. Since these mutations are common, shared antigens are likely to be expressed in tumors across many patients. Vaccines directed against these mutations can be used in any patient who has the mutation in question1.
Personalized vaccines
These vaccines use unique neoantigens that are produced by unique genetic mutations in an individual patient tumor. Each vaccine is bespoke, produced for a given patient following genetic sequencing of his/her tumor1 (Fig. 1).
Vaccine delivery platforms
mRNA vaccines
mRNA encoding tumor-specific antigens is delivered to cell cytoplasm using various methods including viral vectors and nanocarriers. The mRNA instructs the ribosomes to produce the neoantigens, which are presented to the immune system to trigger an immune response. These vaccines have numerous advantages including1:
- Ease, rapidity and low cost of mRNA production
- Single stand of mRNA can target multiple neoantigens
- Efficient delivery to cell cytoplasm by encapsulating in lipid nanoparticles
- Lower risk of insertional mutagenesis and undesired immune responses
- Rapid degradation limits sustained exposure
- Robust and enduring immune response
A number of different routes of administration have been explored for mRNA vaccines, but intramuscular and intravenous routes remain the most widely used5.
This is a relatively new technology that came of age during the Covid pandemic. Our literature search did not yield any completed Phase III studies with mRNA cancer vaccines. However, two mRNA vaccines have recently demonstrated promising results in early phase trials. In a Phase I trial, adjuvant therapy of pancreatic ductal adenocarcinoma (PDAC) with autogene cevumeran (personalized mRNA vaccine) in combination atezolizumab (checkpoint inhibitor) led to impressive recurrence-free survival rates in responding patients6. mRNA-4157 is the other mRNA vaccine that has produced impressive results in the Phase II KEYNOTE-942 trial. We have reviewed this trial in detail in the following sections.
While the experience with these vaccines for cancer treatment is still limited, the early phase trials have not generated any platform-specific safety signals. There is also extensive experience from the use of mRNA vaccines for mass vaccination against COVID-19. In June 2025, the US-FDA added a safety warning to the COVID-19 mRNA vaccine labels for an estimated unadjusted incidence of myocarditis and/or pericarditis of approximately 8 cases per million doses in general population, and a higher incidence of 27 cases per million doses in young males7. The rapid and cost-effective manufacturing process with a potential for short turn-around time from biopsy to vaccine delivery (needle-to-needle time) lends mRNA vaccines well to the personalized vaccine paradigm. At least three mRNA vaccines, mRNA-4157, BNT-111 and autogene cevumeran, are currently in Phase II/III trials.
Dendritic cell vaccines
Patient’s dendritic cells are harvested, loaded with neoantigens, and transfused back into the patient’s body. Neoantigen could be loaded into the cells in the form of peptides, DNA or mRNA, using a variety of methods like pulsing with peptides or cell lysates, fusing with whole tumor cells, or transfection with viral vectors. These vaccines are mainly administered as intravenous infusions or subcutaneous injections. Since dendritic cells are specialized antigen presenting cells with ability to initiate and modulate immune response, these vaccines tend to have high and durable immunogenicity, and specificity1. Cytokines like interleukin-2 (IL-2), interferon-alpha (IFN-α), etc. may be added to the vaccine to further enhance the immune response8.
A number of dendritic cell vaccines have been studied in clinical trials, and while there is some indication of clinical activity, there haven’t been any particular safety concerns to date. Sipuleucel-T, which is approved in the US for prostate cancer, is a dendritic cell vaccine. Patient’s dendritic cells are activated ex vivo by recombinant fusion protein consisting of a prostate antigen (prostatic acid phosphatase) fused to an immune-cell activator (GM-CSF), and then infused back into the patient9. However, optimizing the vaccine regimen for individual patients is a challenge, as is the complexity and cost of manufacture1. Indeed, even though Sipuleucel-T is approved, it is not available in the US, owing to high price, modest clinical benefit, complex manufacturing and administration, and lack of reimbursement. Nevertheless, we found at least 4 ongoing randomized controlled trials of dendritic vaccine, indicating continued interest in this platform.
Peptide vaccines
Amino acid (AA) chains corresponding to peptide sequences of tumor-specific neoantigens are presented to the immune system to elicit a response10. These include 8-12 AA-long MHC I-binding short peptides, which have the advantage of a simple manufacturing process and highly targeted immune response. Challenges specific to peptide vaccines include limited immunogenicity, a short half-life, and potential development of immune tolerance11. They are generally administered subcutaneously or intradermally, often with adjuvants such as granulocyte monocyte colony stimulating factor (GM-CSF) to enhance the immune response12.
Synthetic long peptide (SLP) vaccines are typically 25-35 AA-long, encompassing multiple epitopes or larger portions of the target protein.SLP vaccines have an advantage over the short peptide ones in terms of broader and more diverse immune responses, enhanced peptide stability, and efficient antigen delivery. However, they still have challenges in terms of complex preparation, rapid degradation, and potential for HLA restriction13.
Peptide vaccines are generally considered to be safe. Our literature search retrieved one RCT in which a peptide vaccine showed a modest benefit, but otherwise the clinical outcomes haven’t shown much promise yet14. Nevertheless, peptide vaccines were the ones that came up most frequently in our searches for completed and ongoing randomized controlled studies, indicting that this is a popular platform for cancer vaccine development.
DNA vaccines
Synthetic DNA fragments encoding tumor-specific antigens are introduced into cellular DNA using plasmid- or viral vector-based delivery systems. The cell then starts producing the tumor-specific antigens, which are presented to the immune system to trigger an immune response. These vaccines are primarily administered as intramuscular injections. They can simultaneously deliver multiple neoepitopes coding for multiple neoantigens, but the need to deliver DNA into the cell nucleus is a challenge. Physical methods such as electroporation, gene gun, and sonoporation have been used, but these will be difficult to implement in clinical practice4.
While these vaccines have shown promise in preclinical studies, clinical efficacy has thus far been generally limited due to weak and short-lived immune response. Concerns regarding the potential for immune reactions to viral vector and integration of the DNA into host genome will have to be addressed before these vaccines become mainstream1. Indeed, our literature search did not retrieve a single completed Phase III RCT with a DNA vaccine, and just one RCT is currently ongoing with a DNA vaccine.
Anti-idiotypic vaccines
These vaccines use anti-idiotypic antibodies, which mimic the structure of tumor antigens and elicit antigen-specific immune responses. They can be used to elicit immune responses against carbohydrate and lipid antigens, which are less immunogenic than protein antigens15.
Racotumumab, which is approved in Cuba and Argentina for advanced or recurrent NSCLC, is an anti-idiotypic antibody against Neu-glycolyl-containing gangliosides, sulfatides, and other antigens expressed in tumors15.
Whole tumor cell and cell lysate vaccines
Some of the earlier vaccines used killed tumor cells or tumor cell lysates to stimulate anti-cancer immune responses. They have the advantage of inducing immune response to multiple tumor antigens, without the need to prospectively identify the antigens15.
Tumor cells that are genetically engineered to secret immunomodulatory cytokines, such as GM-CSF, in order to promote DC activation and enhance antigen presentation and immune activation, are also being studied15.
Route of administration
The route of administration for a cancer vaccine may depend upon the delivery platform used, as well as several other factors. Each route has its own advantages and disadvantages.
Intravenous
Intravenous route allows for easy administration of large volumes. Nucleic acids undergo rapid degradation in the blood, but packaging in lipid nanoparticles allows for DNA and mRNA vaccines also to be administered intravenously. There is a risk of toxicity from systemic distribution. For instance, predominant homing of mRNA vaccines to the liver leading to liver toxicity has been observed following IV administration16.
Intramuscular
Intramuscular route is convenient to administer, and is associated with minimal side effects at injection site. Since the skeletal muscle contains few immune cells, intramuscular injections require adjuvants to induce an inflammatory response at the injection site, to draw in the antigen presenting cells4. Another challenge is the specific requirement for particle size and charge16.
Subcutaneous
Subcutaneous route is also convenient, and relatively large volumes can be injected. However, this route is limited by relatively few antigen presenting cells in the subcutaneous tissue, and low absorption rates16.
Intra-dermal
Intra-dermal injections facilitate efficient antigen uptake and presentation by the skin’s resident dendritic cells, the Langerhans cells in the epidermis, and mesenchymal dendritic cells in the dermis4. However, the amount of vaccine that can be injected in the skin is limited16.
Intra-nodal and intra-tumoral
These routes have also been explored, and have an advantage in terms of targeted delivery to the site of action, but are more complicated to administer16.
Development of cancer vaccines: Challenges, opportunities, and trends
Historical context
The earliest form of vaccination to treat cancer can be traced back to Coley’s toxins, which were unfiltered bacterial mixtures used by Dr. William Coley in the late 19th century to treat inoperable tumors. Coley started using these mixtures in his patients following an observation that cancer patients who experienced severe post-operative infections sometimes experienced long-lasting remissions. It is now known that the bacterial infections activated the patients’ immune systems, which in turn destroyed the tumors10. Thus, Coley’s toxins were in a way, doing exactly what vaccines do – stimulate the immune system to eradicate cancer cells and protect from recurrence.
In 1988, Mitchell et al successfully induced anti-melanoma immune response with allogeneic melanoma lysate. The first cancer vaccine to be approved by the US-FDA was Sipuleucel-T. It was approved in April 2010 for treatment of advanced prostate cancer17. In a double-blind, randomized, placebo-controlled trial, Sipuleucel-T significantly reduced the odds of death (overall survival) by 22%, representing an increase of 4.1 months in median survival9. Thus, while statistically significant, the clinical benefit was modest. Several other cancer vaccines developed during the same period managed to elicit an immune response in early phase trials, but that failed to translate into a clinical benefit on disease-free or overall survival in subsequent randomized controlled trials18‘19‘20‘21‘22‘23‘24‘25‘26‘27‘28‘29‘30‘31‘32‘33‘34‘35‘36. For the few that did demonstrate a statistically significant benefit, the clinical benefit remained modest37‘38‘39‘40‘41‘42. Most of these were off-the-shelf vaccines delivering protein/s known to be associated with the tumor.
Notably, the early vaccines failed primarily due to lack of efficacy, and none of these trials have generated any safety signals18‘43‘44‘45‘46‘47‘24‘48‘49‘50‘51‘52‘53‘54‘55‘56‘57‘58‘59‘60’61‘62‘63‘64‘65. In most trials, across the different vaccine platforms, the safety profile of the vaccine in terms of type, frequency and severity of the AEs was comparable to that of the placebo or standard of care comparator. Commonly reported adverse events in the vaccine arm included mainly local injection site reactions and manageable constitutional symptoms like headache, pain, fatigue and weakness. It is important to acknowledge that most patients studied were followed up for short durations owing to lack of efficacy, and long-term effects of these vaccines have therefore not been studied. Hence the risks associated with chronic immune stimulation, including cross-reactivity with self-antigens having structurally similar epitopes leading to autoimmune conditions, or chronic low-grade inflammation leading to tissue injury, fibrosis, and theoretically even secondary malignancies, or T cell exhaustion and tolerance leading to loss of efficacy, are yet to be discharged66‘67‘68.
| Publication | Cancer type | Target antigen | Vaccine type | Study Design | Primary Endpoint | # of patients | Efficacy Outcomes |
| Kantoff PV. N Engl J Med 2010; 363: 411. | Prostate – Metastatic castration-resistant | Recombinant fusion prostate antigen protein PA2024 | Personalized Dendritic cell | Double-blind RCT Sipuleucel T vs Placebo | Overall Survival (OS) | 512 | Sipuleucel T prolonged survival [Median OS (Sipuleucel T vs Placebo): 25.8 vs 21.7 mths; HR (95CI): 0.78 (0.61-0.98); P=0.03] |
| Jocham D. Lancet 2004; 363: 594. | Renal Cell Carcinoma (RCC) – Resected | Tumor cell | Personalized tumor cell | RCT Adjuvant treatment with vaccine vs No adjuvant treatment | Progression Rate | 379 | Vaccine reduced progression rate [Progression Rate (Vaccine vs. No vaccine): 0.26 vs 0.39; HR (95CI): 1.58 (1.05-2.37); P=0.02] |
| Wood C. Lancet 2008; 372: 145. | RCC – Localized | HSP glycoprotein 96 | Personalized Peptide | Observer-blind RCT Vitespen vs Observation | Recurrence-free survival (RFS) | 818 | No difference on RFS between arms [P=0.126] |
| Freeman AJ. J Clin Oncol 2009; 27: 3036. | Follicular Lymphoma – Post-rituximab | Tumor-specific idiotype | Personalized Anti-idiotype | Mitumprotimut-T + GM-CSF vs Placebo + GM-CSF | Time to progression (TTP) | 364 | Worse in vaccine arm [Median TTP (Mitumprotimut-T vs Placebo): 9 vs 12.6 mths; HR (95CI): 1.384 (1.053-1.819); P=0.019] |
| Schulze T. Cancer Immunol Immunother 2009; 58: 61. | Colorectal – with liver metastases | Irradiated tumor cell | Personalized tumor cell | Placebo-controlled RCT ATV-NDV vs. Placebo | Overall Survival (OS) | 51 | No difference on OS between arms [P=0.597] |
| Amato RJ. Clin Cancer Res 2010; 16; 5539. | RCC – Clear cell, metastatic | 5T4 oncofetal antigen | Off-the-shelf Peptide | Double-blind RCT MVA-5T4 vs. Placebo | Overall Survival (OS) | 733 | No difference between arms [Median OS (MVA-5T4 vs. Placebo): 20.1 vs 19.2 mths; HR (95CI): 1.07 (0.86-1.32); P=0.55] |
| Schuster SJ. J Clin Oncol 2011; 29: 2787. | Follicular Lymphoma – Post-chemotherapy | Tumor-specific idiotype | Personalized Hybridoma-derived Anti-idiotype | Double-blind RCT Id-KLH + GM-CSF vs KLH + GM-CSF | Disease-Free Survival (DFS) | 234 | Longer DFS in vaccine arm [DFS (Id-KLH vs KLH): 23 vs 20.6 mths; HR (95CI): 0.81 (0.56-1.16); P=0.256] |
| Schwartzentruber DJ. N Engl J Med 2011; 364: 2119. | Melanoma – Advanced | Glycoprotein 100:209-217(210M) | Off-the-shelf Peptide | Observer-blind RCT gp100:209-217(210M) vs IL-2 | Clinical Response Rate | 479 | Higher response rate & longer PFS in vaccine arm [Response Rate (Vaccine vs IL-2): 16% vs 6%; P=0.03] [PFS (Vaccine vs IL-2): 2.2 vs 1.6 mths; P=0.008] |
| Miles D. | Breast – Metastatic | sialyl-Tn | Off-the-shelf Peptide | Double-blind RCT | Time to progression (TTP) | 1028 | No difference between arms [Median TTP (STn-KLH vs KLH): 3.4 vs 3.0 mths; P=0.353] [Median OS (STn-KLH vs KLH): 23.1 vs 22.3 mths; P=0.916] |
| Oncologist 2011; 16: 1092. | (STn) | STn-KLH vs KLH | OS | ||||
| glycoprotein | |||||||
| Dillman RO. J | Melanoma – Metastatic | Tumor cells | Personalized Tumor cell Dendritic cell | Phase II RCT | OS | 42 | Survival benefit in DC arm [OS (DC vs TC): NR vs 15.9 mths; HR (95CI): 0.27 (0.1-0.73); P=0.007] |
| Immunother 2012; 35: 641. | Tumor Cell (TC) vaccine vs Dendritic Cell (DC) vaccine | ||||||
| Middleton G. | Pancreatic – Locally advanced or metastatic | Human telomerase reverse transcriptase catalytic subunit | Off-the-shelf Peptide | Open-label RCT | OS | 1062 | No difference between arms [Median OS (CT vs Seq): 7.99 vs 6.94; HR (95CI): 1.19 (0.97-1.48); P=0.05] [Median OS (CT vs Con): 7.99 vs 8.36 mths; HR (95CI): 1.05 (0.85-1.29); P=0.64] |
| Lancet Oncol 2014 | (hTERT) class II 16mer | Chemotherapy (CT) vs Sequential CT + GV1001 (Seq) vs Concurrent CT + GV1001 (Con) | |||||
| Alfonso S. | NSCLC – Advanced, post-chemotherapy | NeuGcGM3 tumor-associated ganglioside | Off-the-shelf Anti-idiotype | Double-blind Phase I/II RCT | OS | 176 | Survival benefit in racotumumab arm [Median OS (Racotumomab vs. Placebo): 8.23 vs 6.8 mths; HR (95CI): 0.63 (0.46-0.87); P=0.004] |
| Clin Cancer Res 2014; 20: 3660. | Racotumomab vs Placebo | ||||||
| Butts C. | NSCLC – Unresectable | Mucin-1 (MUC1) glycoprotein | Off-the-shelf Peptide | Double-blind RCT | OS | 1513 | No difference between arms [Median OS (Tecemotide vs Placebo): 25.6 vs 22.3 mths; HR (95CI): 0.88 (0.75-1.03); P=0.123] |
| Lancet Oncol 2014; 15: 59 | Tecemotide vs Placebo | ||||||
| Levy R. | Follicular Lymphoma – Advanced | Tumor-specific idiotype | Personalized Anti-idiotype | Double-blind RCT | Progression-Free Survival (PFS) | 287 | No difference between arms [PFS (MyVax vs. Placebo) HR (95CI): 0.98 (0.72-1.33); P=0.89] |
| J Clin Oncol 2014; 32: 1797. | Id-KLH (MyVax) vs KLH | ||||||
| Giaccone G. | NSCLC – Stage III/IV | Whole tumor cells | Off-the-shelf Tumor cell | Double-blind RCT | Overall Survival (OS) | 532 | No difference between arms [Median OS (Belagenpumatucel-L vs. Placebo): 20.3 vs 17.8 mths; HR (95CI): 0.94 (0.73-1.20); P=0.594] |
| Eur J Cancer 2015; 51 : 2321. | Belagenpumatucel-L vs Placebo | ||||||
| Lawson DH. J Clin Oncol 2015; 33: 4066. | Melanoma – Resected high-risk | Multi-epitope peptides | Off-the-shelf Peptide | RCT | Relapse-Free Survival (RFS) | 456 | No difference between arms [Median RFS (PV vs Placebo): 11.5 vs 9.8 mths; HR (95CI): 0.96 (0.74-1.23); P=0.708] [Median OS (PV vs Placebo): 68.6 vs 63.3 mths; HR (95CI): 0.93 (0.71-1.21); P=0.598] |
| Peptide vaccine (PV) +/- GM-CSF vs Placebo | Overall Survival (OS) | ||||||
| Vansteenkiste JN. Lancet Oncol 2016; 17: 822. | NSCLC – Resected | MAGE-A3 | Off-the-shelf Peptide | Double-blind RCT MAGE-A3 immunotherapeutic vs Placebo | Disease-Free Survival (DFS) | 2312 | No difference between arms [Median DFS (MAGE-A3 vs. Placebo): 60.5 vs 57.9 mths; HR (95CI): 1.02 (0.89-1.18); P=0.74] |
| Quoix E. Lancet Oncol 2016; 17: 212. | NSCLC – Advanced, 1st line therapy | MUC1 | Off-the-shelf Viral | Double-blind Phase 2b RCT TG4010 vs Placebo | PFS | 222 | TG4010 improved PFS [Median PFS (TG4010 vs. Placebo): 5.9 vs 5.1 mths; HR (95CI): 0.74 (0.55-0.98); P=0.019] |
| Rini BI. Lancet Oncol 2016 | RCC – Advanced or metastatic, 1st line therapy | 10 different tumor-associated peptides | Off-the-shelf Peptide | Open-label RCT IMA901 + Sunitinib vs Sunitinib | Overall Survival (OS) | 339 | No difference between arms [Median OS (IMA901 vs. Control): 33.17 mths vs NR; HR (95CI): 1.34 (0.96-1.86); P=0.087] |
| Faries MB. Ann Surg Oncol 2017; 24: 3991. | Melanoma – Resected Stage IV | Whole tumor cells | Off-the-shelf Tumor cell | RCT | Overall Survival (OS) Canvaxin + BCG vs Canvaxin + Placebo | 496 | No difference between arms [Median OS (Canvaxin vs. Placebo): 38.6 vs 34.9 mths; HR (95CI): 1.04 (0.8-1.35); P=0.773] |
| Weller M. Lancet Oncol 2017; 18: 1373. | Glioblastoma – Newly diagnosed | EGFRvIII | Off-the-shelf Peptide | Double-blind RCT Rindopepimut + Temozolomide vs Control + Temozolomide | Overall Survival (OS) | 745 | No difference between arms [Median OS (Rindopepimut vs. Control): 17.4 vs 17.4 mths; HR (95CI): 0.89 (0.75-1.07); P=0.22] |
| Narita Y. Neuro-Oncol 2019; 21: 348. | Glioblastoma – Recurrent | Tumor-associated peptides | Personalized Peptide | Double-blind RCT | Overall Survival (OS) | 88 | No difference between arms [Median OS (PPV vs. BSC): 8.4 vs 8.0 mths; HR (95CI): 1.13 (0.6-1.9); P=0.621] |
| Mittendorf EA. Clin Cancer Res 2019; 25: 4248 | Breast – Resected | HER-2 | Off-the-shelf Peptide | Double-blind RCT Nelipepimut-S + GM-CSF vs Placebo + GM-CSF | Disease-Free Survival (DFS) | 758 | No difference between arms [DFS (Nelipepimut-S vs. Placebo) HR (95CI): 1.564 (0.96-2.55); P=0.07] |
| Noguchi M. Oncol Rep 2021; 45: 159. | Prostate – Castration-resistant | Tumor-associated peptides | Personalized Peptide | Double-blind RCT | Overall Survival (OS) | 310 | No difference between arms [Median OS (PPV vs. Placebo): 16.1 vs 16.9 mths; HR (95CI): 1.04 (0.8-1.37); P=0.77] |
| Vreeland TJ. Ann Surg Oncol 2021; 28: 6126. | Melanoma – Resected Stage III/IV | Tumor lysate | Personalized Dendritic cell | Double-blind RCT TDPLDC vaccine vs Placebo | Disease-Free Survival (DFS) Overall Survival (OS) | 144 | No difference between arms [36-mth DFS (TDPLDC vaccine vs. Placebo) 34.2% vs 21.6%; p=0.889] [36-mth OS (TDPLDC vaccine vs. Placebo): 67.8% vs 49.3% mths; P=0.16] |
| Hewitt DB. Ann Surg 2022; 275: 45. | PDAC – Locally Advanced Unresectable | Genetically modified allogenic tumor cells | Off-the-shelf Tumor cell | Open-label RCT Algenpantucel-L vs Standard of Care (SoC) | Overall Survival (OS) | 303 | No difference between arms [Median OS (Algenpantucel-L vs. SoC): 14.3 vs 14.9 mths; HR (95CI): 1.02 (0.66-1.58); P=0.98] |
Challenges
Several challenges have hampered the development of an effective cancer vaccine in the past (Table 2). Some of these stem from the hallmark characteristics of cancer cells, and are common to all cancer therapeutics. For e.g., the genomic instability of cancer cells results in the evolution of numerous distinct clones of cells within the tumor. Several researchers have demonstrated genetic and epigenetic heterogeneity within tumors by performing multi-region genomic and epigenomic profiling using techniques like next generation sequencing, single cell transcriptomics, and DNA methylation analysis69‘70’71‘72‘73.
Thus, most tumors are heterogenous at genetic, epigenetic, phenotypic and functional levels. It follows that a single therapeutic may not be able to hit all the clones and subclones, allowing the resistant ones to persist and continue to grow, undermining the treatment efficacy74. Secondly, tumors can evolve mechanisms to evade detection and destruction by the immune system. Mechanisms of immune evasion include loss of immunogenic antigens, disruption of antigen presentation, expression of inhibitory surface molecules, secretion of inhibitory cytokines, inhibition of immune cells, recruitment of inhibitory cells, etc.75‘76’77‘78‘79. The inhibitory surface molecules that prevent T cell activation are also referred to as immune checkpoints, and include Cytotoxic T Lymphocyte Antigen 4 (CTLA-4), Programmed death 1 (PD-1) and Lymphocyte Activation Gene 3 (LAG-3). These mechanisms create a tumor micro-environment (TME) that suppresses the anti-tumor immune cells, thereby protecting the tumor from the immune system74. Some tumors are particularly notorious for low levels of immune cell infiltration in the TME, and are referred to as ‘cold tumors’, in contrast to highly infiltrated and inflamed ‘hot tumors’80. Immunotherapies including cancer vaccines may be expected to have limited efficacy against cold tumors. Any of these mechanisms may lead to either primary or acquired resistance to an immunotherapeutic agent like a cancer vaccine. Finally, continuous antigenic stimulation of T cells results in T cell exhaustion and acquired resistance, characterized by reduced effector function associated with expression of immune checkpoints and reduced production of inflammatory proteins81‘82‘83‘84‘85.
Then there are challenges that are unique to the neoantigen vaccine platform. For instance, identifying neoantigens capable of inducing adequate T cell response, while critical to the clinical success of the vaccine, is not straightforward. It includes predicting the ability of the antigen presenting cells to process the neoantigen into peptides that can bind to the MHC molecules to be presented to the T cells as MHC-peptide complex, and further, the ability of T cell receptors to recognize and bind to the peptide presented on MHC molecules4. Developing the vaccines that can deliver the active component, whether DNA or RNA or peptide, to its respective site of action within the cell is another challenge. Finally, manufacturing the vaccines at scale adds another layer of complexity, particularly so for the personalized neoantigen vaccines.
Opportunities
A variety of wide-ranging breakthrough technological advances in the past decade are now making it possible to overcome at least some of the challenges. Several ICIs are now available to block the immune checkpoints, esp. CTLA-4, PD-1 and Programmed Death-Ligand 1 (PD-L1). CTLA-4 and PD-1 are expressed on T cells, while PD-L1 is the ligand expressed on tissue cells for binding to PD-186. These ICIs enhance the anti-tumor immune responses, leading to a durable anti-tumor activity and improved survival. These are approved for use in numerous cancers including melanoma, lung cancer, breast cancer, and renal cancer to name just a few, often in combination with conventional chemotherapy, and have greatly improved the prognosis of these cancers87. Cancer vaccines are now being tested in combination with ICIs, as the two have complementary mechanisms – cancer vaccines activate the immune system, while ICIs help to avoid or overcome TME-induced suppression of the immune system.
The development of personalized vaccines has received a boost from the advances in multi-omics techniques that enable rapid and precise profiling of a tumor by generating DNA sequencing, RNA sequencing, and protein expression data. Artificial intelligence (AI) techniques including machine learning (ML) help to analyze and interpret this data by applying statistical models and algorithms11‘88. The end-result is rapid identification of unique immunogenic neoantigens associated with the tumor, in time to produce a bespoke vaccine to treat the patient. Rapid development of mRNA vaccine technology during the COVID-19 pandemic has given us a suitable delivery platform to administer the immunogenic neoantigens.
For off-the-shelf vaccines targeting shared antigens, predictive biomarkers will be used to assess the level of antigen expression (e.g., KRAS mutational status) before making a treatment decision89. This will improve success rates by helping to identify and treat only those patients who are likely to respond to the cancer vaccine. Additionally, given that cancer vaccines like other immunotherapies, are less effective against cold tumors, an assessment of the composition and density of immune cells within the tumor microenvironment using tumor biopsy samples may also help select the right patients90. Assessment of immune checkpoint expression levels will help determine the need for combining a vaccine with ICI.
Liquid biopsies can now be used to monitor the tumors for response to the vaccine as well as for emergence of resistance and recurrence, by longitudinally analyzing non-invasive biomarkers like circulating tumor cells (CTC) and circulating tumor DNA (ctDNA). They employ technologies such as digital PCR (dPCR) for identifying new mutations in pre-defined cancer genes, and next generation sequencing (NGS) for whole exome or genome sequencing. Single cell RNA sequencing can help generate transcriptomics data from CTCs. Machine learning approaches can help analyze the vast amounts of sequencing data generated over time to track the genetic evolution of the tumor91. This information could potentially be used to change the composition of the vaccine in response to the changes in the tumor mutational burden.
These advances in our understanding of the interaction between the cancer cells and immune cells in TME coupled with technological breakthroughs in multi-omics techniques, mRNA and other vaccine platforms, and AI/ML tools, have given us the means to overcome some of the roadblocks that precluded the development, manufacture and delivery of efficacious cancer vaccines in the past (Table 2)11‘2‘92‘13‘10‘93‘94.
| Identification & characterization of immunogenic neoantigens | |
| Since only a minority of neoantigens have the capacity to elicit an immune response, selection of the right (most immunogenic) neoantigen is critical for the clinical success of the vaccine | Integration of genomic, transcriptomic and proteomic data to increase the efficiency of immunogenic neoantigen identification: Use of novel multi-omics techniques like high throughput DNA sequencing, single cell sequencing, and spatial transcriptomics, accompanied by advanced bioinformatic algorithms and prediction pipelines, enhances the precision of potential target identification, ultimately leading to improved clinical outcomes Computational prediction of neoantigen immunogenicity: Immunogenicity of the neoantigen depends upon its ability to engage with the T cell receptors (TCR). This can now be investigated using deep sequencing of TCR genes and prediction algorithms to identify neoantigens that complement the patient’s TCR sequences. In vitro validation of immunogenic neoantigens: Patient-derived organoids (PDOs) are three-dimensional cell culture models that closely recreate the architecture, characteristics, and functionality of original tumors and mimic the tumor microenvironment (TME). These have recently emerged as a valuable tool for validating and screening neoantigens. |
| Optimization of scalable vaccine manufacturing & delivery platform | |
| The process for production of neoantigen vaccines is intricate and expensive. | Rapid development of mRNA vaccine technology during the COVID-19 pandemic: Technological advances have optimized the stability, backbone structure, delivery methods, and cost-effectiveness of mRNA-based vaccines, solving at least one problem associated with cancer vaccine development. |
| T cell suppression by tumor microenvironment | |
| Even if the vaccine elicits a T cell response, several factors within the tumor microenvironment (TME) can impair their function. These include accumulation of immune suppressive cells as well as tumor cells themselves, which can suppress the functioning and proliferation of T cells, either through direct contact or via secretion of soluble inhibitors. | Combination with immune checkpoint inhibitors (ICI): Administration of ICIs concomitantly with vaccination blocks the checkpoint-mediated inhibition of T cells, which in turn may help them avoid or overcome TME-induced suppression. Use of adjuvants: Adjuvants are substances that are added to the vaccine to promote, expedite or lengthen the immune response to the vaccine. They are required for certain types of vaccines like the peptide vaccines. |
| T cell exhaustion | |
| Even when a therapeutic cancer vaccine activates T cells, these T cells will eventually become dysfunctional and exhausted in face of high tumor burden and persistent TCR stimulation by the antigen. T cell exhaustion is characterized by loss of function, reduced proliferative capacity, increased inhibitory receptor expression, and propensity to undergo cell death | Combination with immune checkpoint inhibitors: As above, administration of ICIs concomitantly or after vaccination may help reinvigorate T cells rendered exhausted by TME due to the blockade of checkpoint-mediated inhibitory signals on T cells. |
| Emergence of resistant clones | |
| Tumors can acquire resistance to immunotherapies by adapting to evade immune system through various mechanisms | Liquid biopsies: Sequencing data from non-invasive biomarkers like ctDNA and single cell sequencing of CTCs can be used to monitor for emergence of clones resistant to the vaccine, and modify vaccine composition in response to changes in the tumor mutational burden. |
Trends
One of the first indications that these technological advances may finally help us translate the scientific promise of therapeutic cancer vaccines into clinical medicines, came in the form of KEYNOTE-942 trial. This head-to-head randomized controlled trial demonstrated that addition of a vaccine (mRNA-4157) to standard treatment with a checkpoint inhibitor can meaningfully improve patient outcomes compared to standard treatment alone95. This trial is described in detail in the following section.
A renewed interest in the promise of therapeutic cancer vaccines is also borne out by the number of ongoing clinical trials with different vaccine candidates. A search on clinicaltrials.gov per search strategy described in the Methodology section revealed 342 ongoing clinical trials testing different types of therapeutic cancer vaccines (mRNA, dendritic cell, peptide, etc.) in various phases of clinical development (Phase I/II/III) for solid and hematological malignancies affecting a wide range of organs (brain, blood, breast, pancreas, lungs, colon, liver, etc.). Application of inclusion/exclusion criteria as described in the Methodology section narrowed down our search to 17 ongoing Phase II/III randomized controlled trials assessing long-term outcomes compared to standard of care (Table 3).
| NCT06749925 | Glioblastoma – Post- standard treatment | III | Dendritic cell | DC vaccine + Pembrolizumab vs. DC vaccine vs. Placebo | 186 | Jan 2026 |
| NCT06790966 | Head & Neck– Recurrent / Metastatic Squamous Cell Carcinoma | III | Peptide | PDS0101 + Pembrolizumab vs. Pembrolizumab | 351 | 05/30/2025 |
| NCT04534205 | Head & Neck– Recurrent / Metastatic – HPV16+ Squamous Cell Carcinoma | II/III | mRNA | BNT111 + Pembrolizumab vs. Pembrolizumab | 350 | 01/07/2021 |
| NCT06472245 / ATALANTE-1 | NSCLC – Metastatic | III | Peptide | OSE2101 vaccine vs. Docetaxel | 363 | 03/12/2024 |
| NCT06314087 / iNATURE | Solid tumors – Advanced | II | Peptide | Individualized neoantigen vaccine + Radiotherapy vs Placebo + Radiotherapy | 154 | 01/31/2024 |
| NCT06675201 | Esophageal – Locally advanced, unresectable, squamous cell carcinoma | II | Dendritic cell | NeoDC-Vac + ICI vs ICI | 165 | 10/01/2024 |
| NCT05344209 / LUNGVAC | NSCLC – Stage IIIB/IIIC/IV | II | Peptide | UV1 vaccine + anti-PD-1/PD-L1 vs anti-PD-1/PD-L1 | 138 | 08/12/2022 |
| NCT06157151 | Cervical cancer – Recurrent or metastatic | II | Peptide | PRGN-2009 + Pembrolizumab vs Pembrolizumab | 46 | 03/21/2025 |
| NCT05968326 | PDAC – Resected | II | mRNA | Autogene cevumeran + Atezolizumab + mFOLFIRINOX vs mFOLFIRINOX | 260 | 18/10/2023 |
| NCT06534983 | Urothelial – High-Risk Muscle-Invasive Carcinoma (MIUC) | II | mRNA | Autogene cevumeran + Nivolumab vs Nivolumab | 362 | 09/12/2024 |
| NCT06636682 | Prostate – Post-prostatectomy | II | Tumor cell | FK-PC101 vs Follow-up | 100 | 10/14/2024 |
| NCT06805305 | Glioblastoma – Newly diagnosed | II | Dendritic cell | DOC1021 + peg-IFN + SoC vs SoC | 180 | 03/17/2025 |
| NCT05794659 | Ovarian – Post debulking surgery | II | DNA | AST-201 + GM-CSF + CT vs Placebo + GM-CSF + CT | 98 | 11/15/2023 |
| NCT06639074 / FAROUT | Ovarian/Fallopian tube/Primary peritoneal – Stage III/IV | II | Dendritic cell | FRαDC vs Placebo | 78 | 11/08/2024 |
| NCT03606967 | Breast – Triple negative metastatic | II | Peptide SLP | Vaccine + nab-paclitaxel + durvalumab + tremelimumab vs. nab-paclitaxel + durvalumab + tremelimumab | 70 | 04/13/2021 |
| NCT05528952 | Hepatocellular carcinoma – Unresectable | II | Peptide | UCPVax + Atezoliumab + Bevacizumab vs. Atezoliumab + Bevacizumab | 105 | 09/27/2022 |
| NCT06223568 | Head & Neck – HPV-associated squamous cell carcinoma | II | Viral | PRGN-2009 + Docetaxel + Cisplatin vs. Docetaxel + Cisplatin | 70 | 06/10/2024 |
mRNA-4157 vaccine for melanoma: A potential breakthrough in the endeavor to develop a successful cancer vaccine
mRNA-4157 (also referred to as V-940 or intismeran autogene) is an individualized neoantigen mRNA vaccine that can encode up to 34 neoantigens in a single synthetic mRNA molecule encapsulated in lipid nanoparticles95. This vaccine was granted a ‘breakthrough therapy’ designation by the US-FDA following a successful outcome in a randomized controlled clinical trial in melanoma, which compared mRNA-4157 given in combination with a checkpoint inhibitor, pembrolizumab to pembrolizumab monotherapy only96.
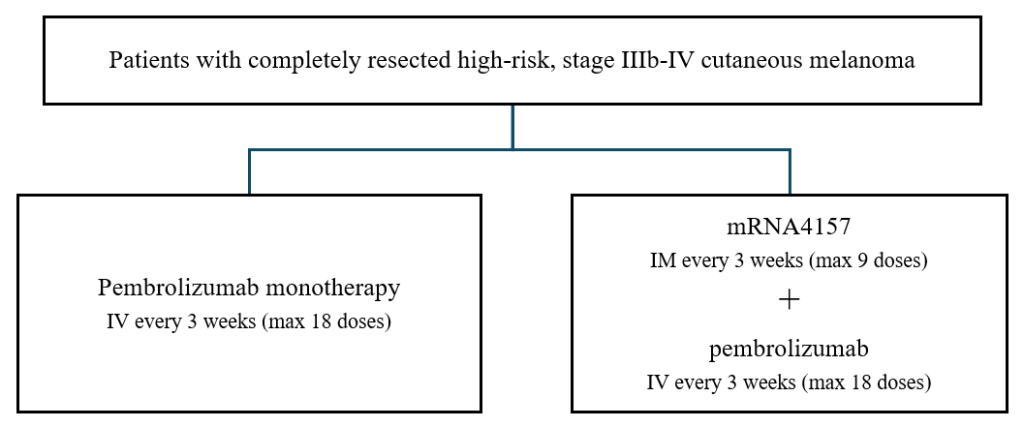
The trial was conducted in patients with high-risk, stage IIIb-IV cutaneous melanoma, a type of skin cancer. Following the complete surgical resection of the melanoma, the patients received 3-weekly doses of either mRNA-4157 in combination with pembrolizumab, or pembrolizumab alone95. (Fig 2). Pembrolizumab inhibits the checkpoint PD-1, thereby disrupting a pathway that suppresses immune cells, and allowing the immune cells activated by the vaccine to attack cancer cells88. It is currently the standard treatment given to such patients.
The mRNA sequence to be used for the vaccine was individually determined for each patient following an analysis of the resected melanoma tissue as well as blood samples. This involved assessment of the patient’s mutanome (all somatic mutations in the tumor) by whole exome sequencing (WES), transcriptome by RNA sequencing, and HLA type by next generation sequencing. The patient-specific data was then fed into an automated bioinformatics system to obtain the optimal mRNA sequence for the given patient. The vaccine was manufactured using Moderna’s mRNA platform95.
A total of 157 patients were enrolled in this study, of which 107 were randomized to mRNA-4157 + pembrolizumab group, and 50 were randomized to pembrolizumab group (2:1 randomization). The primary endpoint was recurrence-free survival (RFS), which is the time from the first dose of pembrolizumab till the first date of melanoma recurrence, or new melanoma, or death from any cause.
At the end of 2 years, RFS events (recurrent or new melanoma, or death) occurred in 22% of patients who received the vaccine in combination with pembrolizumab, compared to 40% in patients who received pembrolizumab only. The hazard ratio (HR), which is the ratio of the probability of experiencing an RFS event in the two groups, was 0.56. A HR<1 indicates a reduction in the probability of experiencing the event; the HR of 0.56 in this case indicates that the addition of vaccine to pembrolizumab cut the probability of melanoma recurrence or death by 44%. This magnitude of reduction is highly clinically meaningful, though it narrowly missed achieving statistical significance. The 95% confidence interval (95% CI), which is the interval within which the value of HR will lie for 95% of the times the study is repeated with different samples from the whole population, ranged from 0.3 to 1.01. The outcome is not considered statistically significant since the outer bound of confidence interval for HR crosses 1, and the p value, which denotes the probability that the observed outcome could be due to chance, is 0.053 (5.3%); p<0.05 (5%) is generally considered as statistically significant in medical research (Table 4). The side effect profile of the vaccine was unremarkable95.
| Endpoint | Pembro n=50 | mRNA-4157 + pembro N=107 |
| Median Follow-up, months | 24 | 23 |
| Recurrence-Free Survival (RFS) | ||
| Events, n (%) | 20 (40) | 24 (22) |
| Hazard Ratio (95CI) | 0.56 (0.3-1.01) | |
| P value | 0.053 | |
| 12-month RFS, % | 77 | 83 |
| 18-month RFS, % | 62 | 79 |
| Safety | ||
| Any Adverse Event (AE), n (%) | 47 (94) | 104 (100) |
| Severe* AE, n (%) | 18 (36) | 36 (35) |
* Graded as Grade 3 or worse on National Cancer Institute (NCI) Common Toxicity Criteria Adverse Event (CTCAE) scale
In summary, the KEYNOTE-942 trial provides the proof of concept (PoC) for addition of mRNA-4157 cancer vaccine to current standard treatment to provide added benefit over and above the current treatment. A larger Phase III trial is now required to confirm this finding. In order to approve any new treatment for general use in patients, the US-FDA and other regulatory agencies across the world require that the benefit of the new treatment be shown to be clinically meaningful and statistically significant compared to current standard of care treatment in an adequately powered Phase III randomized controlled trial. The V940-001 trial is currently underway to generate this confirmatory evidence to pave the way for the approval of mRNA-4157 cancer vaccine for melanoma patients97.97. The breakthrough therapy designation granted by the FDA may help accelerate this process, by allowing for a closer interaction between the FDA and the vaccine manufacturer, as well as the potential for an accelerated approval based on a surrogate endpoint like RFS, without having to wait for demonstration of survival benefit.
Importantly, the clinically meaningful benefit seen in KEYNOTE-942 demonstrates that with modern technology, it is possible to personalize cancer treatment by using the unique mutational signature of a patient’s tumor to stimulate a targeted immune response against the tumor. mRNA-4157 vaccine is also being tested for treatment of other cancers. Current ongoing Phase II/III RCTs for mRNA-4157, retrieved using search strategy as described in the methodology section, are listed in Table 5.
| NCT Number / Trial Name | Condition | Study Phase | Treatment Interventions | Estimated enrolment | Start Date | Estimated Primary Completion Date |
| NCT05933577 / V940-001 | Melanoma – High-Risk Stage II-IV, Adjuvant treatment | III | V940 + Pembrolizumab vs Placebo + Pembrolizumab | 1089 | 07/19/2023 | 10/26/2029 |
| NCT06961006 / V940-012 / INTerpath-012 | Melanoma – Advanced, First-line treatment | II | V940 + Pembrolizumab vs Placebo + Pembrolizumab | 160 | 05/29/2025 | 07/22/2028 |
| NCT06077760 / INTerpath-002 | NSCLC – Resected Stage II-IIIB (N2) | III | V940 + Pembrolizumab vs Placebo + Pembrolizumab | 868 | 12/6/2023 | 6/25/2030 |
| NCT06623422 / INTerpath-009 | NSCLC – Resected Stage II-IIIb (N2) | III | V940 + Pembrolizumab vs Placebo + Pembrolizumab | 680 | 10/21/2024 | 5/16/2033 |
| NCT07221474 / INTerpath-13 | NSCLC – Metastatic | II | V940 + Pembrolizumab + Chemotherapy vs Placebo + Pembrolizumab + Chemotherapy | 180 | 12/02/2025 | 07/02/2029 |
| NCT06295809 / INTerpath-007 | Cutaneous SCC –Locally Advanced Resectable | II/III | V940 + Pembrolizumab + SoC vs Pembrolizumab + SoC vs SoC | 1012 | 4/18/2024 | 03/05/2026 |
| NCT06307431 / INTerpath-004 | RCC – Post-resection | II | V940 + Pembrolizumab vs Placebo + Pembrolizumab | 272 | 10/04/2024 | 08/01/2028 |
| NCT06833073 / V940-011 / INTerpath-011 | Bladder cancer – High-risk non-muscle invasive (HR-NMIBC) | II | V940 + BCG vs V940 | 308 | 03/11/2025 | 03/09/2031 |
Regulatory considerations
Personalized cancer vaccines are manufactured as bespoke products for each patient. Scaling up of this technology for routine treatment of cancer patients in the real-world will pose peculiar challenges in terms of development of robust manufacturing and quality assurance processes that maintain standardization and quality control across the individually manufactured products. Whether these medicines should have different preclinical testing requirements, and whether the clinical testing could be performed in smaller than usual patient samples are the other questions for regulatory agencies to consider98. Ultimately this will involve development of analytical assays, process control measures, clinical trial designs, and a fit-for-purpose regulatory framework.
Currently however, personalized therapies in the field of oncology are using conventional drug development pathways. The CAR-T cell therapies are highly personalized therapies that require bespoke manufacturing just like cancer vaccines, and their experience has shown that the current clinical development paradigm of moving from proof-of-concept in smaller early-phase clinical studies to confirmation of benefit-vs-risk in large, phase III randomized controlled trials can be applied to personalized medicines as well. As of today, at least six CAR-T cell therapies are FDA-approved using the conventional regulatory pathway and are commercially available (Tisagenlecleucel, Axicabtagene Ciloleucel, Brexucabtagene Autoleucel, Lisocabtagene Maraleucel, Idecabtagene Vicleucel, Ciltacabtagene Autoleucel). Indeed, personalized cancer vaccines development is also following this same established drug development paradigm, and results from ongoing clinical programs will indicate whether this established pathway will work for personalized vaccines as well.
Comparison with currently available immunotherapeutic agents
Finally, it will be pertinent to assess how cancer vaccines compare with currently available immunotherapeutic agents. Different immunotherapies act with different mechanisms, and have their unique strengths and limitations. We believe that if successful, cancer vaccines will have a place in the therapeutic armamentarium alongside other immunotherapies like ICI and CAR-T cell therapies.
| Mechanism of Action | Cancer cells evade immune surveillance by increasing the expression of surface receptors that dampen immune activation within the tumor microenvironment. ICIs inhibit these inhibitory checkpoints, thereby allowing the immune system to attack cancer cells. They are off-the-shelf monoclonal antibodies targeting immune checkpoints like CTLA-4, PD-L1 and PD-199‘100. | T cells are genetically engineered to express receptors that recognize and bind to the antigen of interest expressed on cancer cells, leading to activation of the T cell and destruction of the cancer cell. Currently available CAR-T cell therapies are mostly autologous, meaning that the patient’s own T cells are used. The patient undergoes leukapheresis to obtain the T cells, which are then modified in the lab to CAR-T cells. The CAR-T cells are expanded ex vivo in the lab, and infused back in the patient101. | Cancer vaccines present tumor-specific antigens to the patient’s immune system, triggering a T cell mediated immune response against cancer cells102. |
| Usage & impact | Have revolutionized the treatment of several cancers by demonstrating durable responses.At least 15 different ICIs are currently approved for a broad range of tumors.These are off-the-shelf therapies, hence easier to manufacture, supply, and use. | Have revolutionized the treatment of certain B cell malignancies, but haven’t yet been successful in solid tumors.Six different CAR-T cell therapies are currently approved for B cell malignancies including multiple myeloma and certain lymphomas. Autologous CAR-T cell therapy is personalized, and requires lead time for manufacturing, which is complex and costly. Research is currently ongoing to produce allogenic CAR T cell therapies, which may be used off-the-shelf. | Have shown promising efficacy in early phase studies in solid tumors like melanoma and pancreatic cancer, but a meaningful impact on patients in real-world remains to be seenThe only FDA-approved cancer vaccine is currently not available in the US. Personalized cancer vaccines also require lead time for manufacturing, which can be complex and costly depending on the vaccine platform. However, off-the-shelf vaccines targeting common antigens are also being researched. |
| Potential to develop resistance | These are off-the-shelf therapies targeting a single checkpoint or its ligand. Once a tumor develops mutations that confer resistance, they lose effectivenessICIs can be combined to target 2 antigens at once to reduce the chances of immune escape. | Currently available CAR-Ts target a single antigen (e.g., CD19, BCMA). Once the tumor develops mutations that confer resistance to the CAR construct (e.g., loss of target antigen), the therapy loses effectiveness.Research is ongoing to produce dual-targeting CAR-T therapies to reduce the chances of immune escape.Since it is one-time treatment, failure to develop immunological memory can also lead to loss of effectiveness | Can be designed to target multiple antigens at once. Theoretically, this may help address tumor heterogeneity and reduce the probability of immune escape.It may be possible to monitor the tumor mutational burden longitudinally and change the vaccine composition accordingly, to preempt the development of resistance. |
| Safety profile | The release of inhibition on immune system can lead to immune-related adverse events affecting various organ systems including skin, gastrointestinal, endocrine, lungs, liver, heart, etc. These are generally managed by early recognition and prompt glucocorticoid therapy99‘100. | Potential for severe adverse events like cytokine release syndrome (CRS), immune effector cell associated neurotoxicity syndrome (ICANS), delayed neurotoxicity (e.g., parkinsonism), etc.103. | Reported safety profile appears to be benign, with injection-site reactions being the main side effect. However, long-term risks of chronic immune stimulation with these vaccines haven’t been assessed yet. |
| Impact on Health-Related Quality of Life (HR-QoL) | Require regular administration in treatment cycles. This means that the patient is at risk of adverse events during each cycle, which could impact the (HR-QoL). | May induce long-term immunological memory leading to long term remission with a single infusion, such that some patients may not need further treatment. Long treatment-free intervals can positively impact HR-QoL104. | Require regular administration in treatment cycles. The patient is at risk of AEs during each cycle impacting the HR-QoL, though AE profile appears to be generally favorable. |
Conclusion
An increased understanding of the interaction between cancer cells and tumor microenvironment, availability of checkpoint inhibitors, and simultaneous advances in diverse technologies such as multi-omics, bioinformatics, artificial intelligence, and vaccine delivery platforms, have created a perfect melting pot that should hopefully lead to the development of individualized cancer vaccines as the next frontier of cancer immunotherapy. A literature search reveals frenetic research activity on this topic, both in preclinical space as well as in terms of ongoing clinical trials in actual cancer patients. It is hoped that this novel approach of training the body’s own immune system to attack and destroy cancer will mark a significant advance in humankind’s conquest against the emperor of all maladies.
References
- X. Li, J. You, L. Hong, W. Liu, P. Guo, X. Hao. Neoantigen cancer vaccines: A new star on the horizon. Cancer Biology and Medicine. 21, 274-311 (2024). [↩] [↩] [↩] [↩] [↩] [↩] [↩] [↩]
- M. R. Zamani, P. Sacha. Immune checkpoint inhibitors in cancer therapy: what lies beyond monoclonal antibodies? Medical Oncology. 42, 273 (2025). [↩] [↩]
- P. D. Katsikis, K. J. Ishii, C. Schliehe. Challenges in developing personalized neoantigen cancer vaccines. Nature Reviews Immunology. 24, 213–227 (2024). [↩]
- T. Fan, M. Zhang, J. Yang, Z. Zhu, W. Cao, C. Dong. Therapeutic cancer vaccines: advancements, challenges, and prospects. Signal Transduction and Targeted Therapy. 450, https://doi.org/10.1038/s41392-023-01674-3 (2023). [↩] [↩] [↩] [↩] [↩] [↩]
- Q. Fu, X. Zhao, J. Hu, Y. Jiao, Y. Yan, X. Pan, X. Wang, F. Jiao. mRNA vaccines in the context of cancer treatment: from concept to application. Journal of Translational Medicine. 23, 12 (2025). [↩]
- L. A. Rojas, Z. Sethna, K. C. Soares, C. Olcese, N. Pang, E. Patterson, J. Lihm, N. Ceglia, P. Guasp, A. Chu, R. Yu, A. K. Chandra, T. Waters, J. Ruan, M. Amisaki, A. Zebboudj, Z. Odgerel, G. Payne, E. Derhovanessian, F. Müller, I. Rhee, M. Yadav, A. Dobrin, M. Sadelain, M. Łuksza, N. Cohen, L. Tang, O. Basturk, M. Gönen, S. Katz, R. K. Do, A. S. Epstein, P. Momtaz, W. Park, R. Sugarman, A. M. Varghese, E. Won, A. Desai, A. C. Wei, M. I. D’Angelica, T. P. Kingham, I. Mellman, T. Merghoub, J. D. Wolchok, U. Sahin, Ö. Türeci, B. D. Greenbaum, W. R. Jarnagin, J. Drebin, E. M. O’Reilly, V. P. Balachandran. Personalized RNA neoantigen vaccines stimulate T cells in pancreatic cancer. Nature. 618, 144-150 (2023). [↩]
- FDA Safety communication. FDA Approves Required Updated Warning in Labeling of mRNA COVID-19 Vaccines Regarding Myocarditis and Pericarditis Following Vaccination. US FDA. Accessed from URL: FDA Approves Required Updated Warning in Labeling of mRNA COVID-19 Vaccines Regarding Myocarditis and Pericarditis Following Vaccination | FDA on 02-November-2025. [↩]
- P. M. Muehlbauer, D. J. Schwartzentruber. Cancer vaccines. Seminars in Oncology Nursing. 19, 206-216 (2003). [↩]
- P. W. Kantoff, C. S. Higano, N. D. Shore, E. R. Berger, E. J. Small, D. F. Penson, C. H. Redfern, A. C. Ferrari, R. Dreicer, R. B. Sims, Y. Xu, M. W. Frolich, P. F. Schellhammer (IMPACT Study Investigators). Sipuleucel-T Immunotherapy for Castration-Resistant Prostate Cancer. New England Journal of Medicine. 363, 411-22 (2010). [↩] [↩]
- P. M. Muehlbauer, D. J. Schwartzentruber. Cancer vaccines. Seminars in Oncology Nursing. 19, 206-216 (2003). [↩] [↩] [↩]
- X. Li, J. You, L. Hong, W. Liu, P. Guo, X. Hao. Neoantigen cancer vaccines: A new star on the horizon. Cancer Biology and Medicine. 21, 274-311 (2024). [↩] [↩] [↩]
- D. Liu, L. Liu, X. Li, S. Wang, G. Wu, X. Che. Advancements and Challenges in Peptide-Based Cancer Vaccination: A Multidisciplinary Perspective. Vaccines. 12, 950 (2024). [↩]
- T. Fan, M. Zhang, J. Yang, Z. Zhu, W. Cao, C. Dong. Therapeutic cancer vaccines: advancements, challenges, and prospects. Signal Transduction and Targeted Therapy. 450, https://doi.org/10.1038/s41392-023-01674-3 (2023). [↩] [↩]
- D. J. Schwartzentruber, D. H. Lawson, J. M. Richards, R. M. Conry, D. M. Miller, J. Treisman, F. Gailani, L. Riley, K. Conlon, B. Pockaj, K. L. Kendra, R. L. White, R. Gonzalez, T. M. Kuzel, B. Curti, P. D. Leming, E. D. Whitman, J. Balkissoon, D. S. Reintgen, H. Kaufman, F. M. Marincola, M. J. Merino, S. A. Rosenberg, P. Choyke, D. Vena, P. Hwu. gp100 Peptide Vaccine and Interleukin-2 in Patients with Advanced Melanoma. New England Journal of Medicine. 364,2119-27 (2011). [↩]
- E. Grimmett, B. Al‑Share, M. B. Alkassab, R. W. Zhou, A. Desai, M. M. A. Rahim, I. Woldie. Cancer vaccines: past, present and future. Discover Oncology. 13, 31 (2022). [↩] [↩] [↩] [↩]
- Q. Fu, X. Zhao, J. Hu, Y. Jiao, Y. Yan, X. Pan, X. Wang, F. Jiao. mRNA vaccines in the context of cancer treatment: from concept to application. Journal of Translational Medicine. 23, 12 (2025). [↩] [↩] [↩] [↩] [↩]
- C. S. Higano, E. J. Small, P. Schellhammer, U. Yasothan, S. Gubernick, P. Kirkpatrick, P. W. Kantoff. Sipuleucel-T. Nature Reviews Drug Discovery. 9, 513-514 (2010). [↩]
- C. Wood, P. Srivastava, R. Bukowski, L. Lacombe, A. I. Gorelov, S. Gorelov, P. Mulders, H. Zielinski, A. Hoos, F. Teofilovici, L. Isakov, R. Flanigan, R. Figlin, R. Gupta, B. Escudier (C-100-12 RCC Study Group). An adjuvant autologous therapeutic vaccine (HSPPC-96; vitespen) versus observation alone for patients at high risk of recurrence after nephrectomy for renal cell carcinoma: A multicentre, open-label, randomised phase III trial. Lancet. 372, 145–54 (2008). [↩] [↩]
- A. Freedman, S. S. Neelapu, C. Nichols, M. J. Robertson, B. Djulbegovic, J. N. Winter, J. F. Bender, D. P. Gold, R. G. Ghalie, M. E. Stewart, V. Esquibel, P. Hamlin. Placebo-Controlled Phase III Trial of Patient-Specific Immunotherapy With Mitumprotimut-T and Granulocyte-Macrophage Colony-Stimulating Factor After Rituximab in Patients With Follicular Lymphoma. J Clin Oncol. 27, 3036-3043 (2009). [↩]
- T. Schulze, W. Kemmner, J. Weitz, K. D. Wernecke, V. Schirrmacher, P. M. Schlag. Efficiency of adjuvant active specific immunization with Newcastle disease virus modified tumor cells in colorectal cancer patients following resection of liver metastases: results of a prospective randomized trial. Cancer Immunology Immunotherapy. 58, 61–69 (2009). [↩]
- R. J. Amato, R. E. Hawkins, H. L. Kaufman, J. A. Thompson, P. Tomczak, C. Szczylik, M. McDonald, S. Eastty, W. H. Shingler, J. deBelin, M. Goonewardena, S. Naylor, R. Harrop. Vaccination of metastatic renal cancer patients with MVA-5T4: A randomized, double-blind, placebo-controlled Phase III study. Clinical Cancer Research. 16, 5539-47 (2010). [↩]
- D. Miles, H. Roche, M. Martin, T. J. Perren, D. A. Cameron, J. Glaspy, D. Dodwell, J. Parker, J. Mayordomo, A. Tres, J. L. Murray, N. K. Ibrahim (Theratope Study Group). Phase III Multicenter Clinical Trial of the Sialyl-TN (STn)-Keyhole Limpet Hemocyanin (KLH) Vaccine for Metastatic Breast Cancer. The Oncologist. 2011;16, 1092–1100 (2011). [↩]
- G. Middleton, P. Silcocks, T. Cox, J. Valle, J. Wadsley, D. Propper, F. Coxon, P. Ross, S. Madhusudan, T. Roques, D. Cunningham, S. Falk, N. Wadd, M. Harrison, P. Corrie, T. Iveson, A. Robinson, K. McAdam, M. Eatock, J. Evans, C. Archer, T. Hickish, A. Garcia-Alonso, M. Nicolson, W. Steward, A. Anthoney, W. Greenhalf, V. Shaw, E. Costello, D. Naisbitt, C. Rawcliffe, G. Nanson, J. Neoptolemos. Gemcitabine and capecitabine with or without telomerase peptide vaccine GV1001 in patients with locally advanced or metastatic pancreatic cancer (TeloVac): An open-label, randomised, phase 3 trial. Lancet Oncology. 15, 829-840 (2014). [↩]
- C. Butts, M. A. Socinski, P. L. Mitchell, N. Thatcher, L. Havel, M. Krzakowski, S. Nawrocki, T. Ciuleanu, L. Bosquée, J. M. Trigo, A. Spira, L. Tremblay, J. Nyman, R. Ramlau, G. Wickart-Johansson, P. Ellis, O. Gladkov, J. R. Pereira, W. E. E. Eberhardt, C. Helwig, A. Schröder, F. A. Shepherd (START trial team). Tecemotide (L-BLP25) versus placebo after chemoradiotherapy for stage III non-small-cell lung cancer (START): a randomised, double-blind, phase 3 trial. Lancet Oncology. 15, 59–68 (2014). [↩] [↩]
- R. Levy, K. N. Ganjoo, J. P. Leonard, J. M. Vose, I. W. Flinn, R. F. Ambinder, J. M. Connors, N. L. Berinstein, A. R. Belch, N. L. Bartlett, C. Nichols, C. E. Emmanouilides, J. M. Timmerman, S. A. Gregory, B. K. Link, D. J. Inwards, A. S. Freedman, J. V. Matous, M. J. Robertson, L. A. Kunkel, D. E. Ingolia, A. J. Gentles, C. L. Liu, R. Tibshirani, A. A. Alizadeh, D. W. Denney Jr. Active Idiotypic Vaccination Versus Control Immunotherapy for Follicular Lymphoma. Journal of Clinical Oncology. 32, 1797-1803 (2014). [↩]
- G. Giaccone, L. A. Bazhenova, J. Nemunaitis, M. Tan, E. Juhasz, R. Ramlau, M. M. van den Heuvel, R. Lal, G. H. Kloecker, K. D. Eaton, Q. Chu, D. J. Dunlop, M. Jain, E. B. Garon, C. S. Davis, E. Carrier, S. C. Moses, D. L. Shawler, H. Fakhrai. A phase III study of belagenpumatucel-L, an allogeneic tumour cell vaccine, as maintenance therapy for non-small cell lung cancer. European Journal of Cancer. 51, 2321– 2329 (2015). [↩]
- D. H. Lawson, S. Lee, F. Zhao, A. A. Tarhini, K. A. Margolin, M. S. Ernstoff, M. B. Atkins, G. I. Cohen, T. L. Whiteside, L. H. Butterfield, J. M. Kirkwood. Randomized, Placebo-Controlled, Phase III Trial of Yeast-Derived GM-CSF Versus Peptide Vaccination Versus GM-CSF Plus Peptide Vaccination Versus Placebo in Patients With No Evidence of Disease After Complete Surgical Resection of Locally Advanced and/or Stage IV Melanoma: A Trial of the ECOG–ACR Imaging Network Cancer Research Group (E4697). Journal of Clinical Oncology. 33, 4066-4076 (2015). [↩]
- J. F. Vansteenkiste, B. C. Cho, T. Vanakesa, T. de Pas, M. Zielinski, M. S. Kim, J. Jassem, M. Yoshimura, J. Dahabreh, H. Nakayama, L. Havel, H. Kondo, T. Mitsudomi, K. Zarogoulidis, O. A. Gladkov, K. Udud, H. Tada, H. Hoffman, A. Bugge, P. Taylor, E. E. Gonzalez, M. L. Liao, J. He, J. Pujol, J. Louahed, M. Debois, V. Brichard, C. Debruyne, P. Therasse, N. Altorki. Efficacy of the MAGE-A3 cancer immunotherapeutic as adjuvant therapy in patients with resected MAGE-A3-positive non-small-cell lung cancer (MAGRIT): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncology. 17, 822–35 (2016). [↩]
- B. I. Rini, A. Stenzl, R. Zdrojowy, M. Kogan, M. Shkolnik, S. Oudard, S. Weikert, S. Bracarda, S. J. Crabb, J. Bedke, J. Ludwig, D. Maurer, R. Mendrzyk, C. Wagner, A. Mahr, J. Fritsche, T. Weinschenk, S. Walter, A. Kirner, H. Singh-Jasuja, C. Reinhardt, T. Eisen. IMA901, a multipeptide cancer vaccine, plus sunitinib versus sunitinib alone, as first-line therapy for advanced or metastatic renal cell carcinoma (IMPRINT): a multicentre, open-label, randomised, controlled, phase 3 trial. Lancet Oncology. 17, 1599–611 (2016). [↩]
- M. B. Faries, N. Mozzillo, M. Kashani-Sabet, J. F. Thompson, M. C. Kelley, R. C. DeConti, J. E. Lee, J. F. Huth, J. Wagner, A. Dalgleish, D. Pertschuk, C. Nardo, S. Stern, R. Elashoff, G. Gammon, D. L. Morton, (MMAIT-IV Clinical Trial Group). Long-Term Survival after Complete Surgical Resection and Adjuvant Immunotherapy for Distant Melanoma Metastases. Annals of Surgical Oncology. 24, 3991–4000 (2017). [↩]
- M. Weller, N. Butowski, D. D. Tran, L. D. Recht, M. Lim, H. Hirte, L. Ashby, L. Mechtler, S. A. Goldlust, F. Iwamoto, J. Drappatz, D. M. O’Rourke, M. Wong, M. G. Hamilton, G. Finocchiaro, J. Perry, W. Wick, J. Green, Y. He, C. D. Turner, M. J. Yellin, T. Keler, T. A. Davis, R. Stupp, J. H. Sampson (ACT IV Trial Investigators). Lancet Oncology. 18, 1373–85 (2017). [↩]
- Y. Narita, Y. Arakawa, F. Yamasaki, R. Nishikawa, T. Aoki, M. Kanamori, M. Nagane, T. Kumabe, Y. Hirose, T. Ichikawa, H. Kobayashi, T. Fujimaki, H. Goto, H. Takeshima, T. Ueba, H. Abe, T. Tamiya, Y. Sonoda, A. Natsume, T. Kakuma, Y. Sugita, N. Komatsu, A. Yamada, T. Sasada, S. Matsueda, S. Shichijo, K. Itoh, M. Terasaki. A randomized, double-blind, phase III trial of personalized peptide vaccination for recurrent glioblastoma. Neuro-Oncology. 21, 348–359 (2019). [↩]
- E. A. Mittendorf, B. Lu, M. Melisko, J. P. Hiller, I. Bondarenko, A. M. Brunt, G. Sergii, K. Petrakova, G. E. Peoples. Efficacy and Safety Analysis of Nelipepimut-S Vaccine to Prevent Breast Cancer Recurrence: A Randomized, Multicenter, Phase III Clinical Trial. Clin Cancer Res. 25, 4248–54 (2019). [↩]
- M. Noguchi, K. Fujimoto, G. Arai, H. Umera, K. Hashine, H. Matsumoto, S. Fukasawa, Y. Kohkimoto, H. Nakatsu, A. Takenaka, M. Fujisawa, H. Uemura, S. Naito, S. Egawa, H. Fujimoto, S. Hinotsu, K. Itohi. A randomized phase III trial of personalized peptide vaccination for castration‑resistant prostate cancer progressing after docetaxel. Oncology Reports. 45, 159-168 (2021). [↩]
- T. J. Vreeland, G. T. Clifton, D. F. Hale, R. C. Chick, A. T. Hickerson, J. L. Cindass, A. M. Adams, P. M. K. Bohan, R. H. I. Andtbacka, A. C. Berger, J. W. Jakub, J. J. Sussman, A. M. Terando, T. Wagner, G. E. Peoples, Mark B. Faries. A Phase IIb Randomized Controlled Trial of the TLPLDC Vaccine as Adjuvant Therapy After Surgical Resection of Stage III/IV Melanoma: A Primary Analysis. Annals of Surgical Oncology. 28, 6126–6137 (2021). [↩]
- D. B. Hewitt, N. Nissen, H. Hatoum, B. Musher, J. Seng, A. L. Coveler, R. Al-Rajabi, C. J. Yeo, B. Leiby, J. Banks, L. Balducci, G. Vaccaro, N. LoConte, T. J. George, W. Brenner, E. Elquza, N. Vahanian, G. Rossi, E. Kennedy, C. Link, H. Lavu. A Phase 3 Randomized Clinical Trial of Chemotherapy With or Without Algenpantucel-L (HyperAcute-Pancreas) Immunotherapy in Subjects With Borderline Resectable or Locally Advanced Unresectable Pancreatic Cancer. Annals of Surgery. 275, 45-53 (2022). [↩]
- D. J. Schwartzentruber, D. H. Lawson, J. M. Richards, R. M. Conry, D. M. Miller, J. Treisman, F. Gailani, L. Riley, K. Conlon, B. Pockaj, K. L. Kendra, R. L. White, R. Gonzalez, T. M. Kuzel, B. Curti, P. D. Leming, E. D. Whitman, J. Balkissoon, D. S. Reintgen, H. Kaufman, F. M. Marincola, M. J. Merino, S. A. Rosenberg, P. Choyke, D. Vena, P. Hwu. gp100 Peptide Vaccine and Interleukin-2 in Patients with Advanced Melanoma. New England Journal of Medicine. 364,2119-27 (2011). [↩]
- S. J. Schuster, S. S. Neelapu, B. L. Gause, J. E. Janik, F. M. Muggia, J. P. Gockerman, J. N. Winter, C. R. Flowers, D. A. Nikcevich, E. M. Sotomayor, D. S. McGaughey, E. S. Jaffe, E. A. Chong, C. W. Reynolds, D. A. Berry, C. F. Santos, M. A. Popa, A. M. McCord, L. W. Kwak. Vaccination With Patient-Specific Tumor-Derived Antigen in First Remission Improves Disease-Free Survival in Follicular Lymphoma. Journal of Clinical Oncology. 29, 2787-2794 (2011). [↩]
- R. O. Dillman, A. N. Cornforth, C. DePriest, E. F. McClay, T. T. Amatruda, C. de Leon, R. E. Ellis, C. Mayorga, D. Carbonell, J. M. Cubellisz. Journal of Immunotherapy. 35, 641–649 (2012). [↩]
- S. Alfonso, A. Valdes-Zayas, E. R. Santiesteban, Y. I. Flores, F. Areces, M. Hernandez, C. E. Viada, I. C. Mendoza, P. P. Guerra, E. García, R. A. Ortiz, A. V. de la Torre, M. Cepeda, K. Perez, E. Chong, A. M. Hernandez, D. Toledo, Z. Gonzalez, Z. Mazorra, T. Crombet, R. Perez, A. M. Vazquez, A. E. Macías. A Randomized, Multicenter, Placebo-Controlled Clinical Trial of Racotumomab-Alum Vaccine as Switch Maintenance Therapy in Advanced Non–Small Cell Lung Cancer Patients. Clin Cancer Res. 20, 3660–71 (2014). [↩]
- E. Quoix, H. Lena, G. Losonczy, F. Forget, C. Chouaid, Z. Papai, R. Gervais, C. Ottensmeier, A. Szczesna, A. Kazarnowicz, J. T. Beck, V. Westeel, E. Felip, D. Debieuvre, A. Madroszyk, J. Adam, G. Lacoste, A. Tavernaro, B. Bastien, C. Halluard, T. Palanché, J. Limacher. TG4010 immunotherapy and first-line chemotherapy for advanced non-small-cell lung cancer (TIME): results from the phase 2b part of a randomised, double-blind, placebo-controlled, phase 2b/3 trial. Lancet Oncology. 17, 212–23 (2016). [↩]
- D. Jocham, A. Richter, L. Hoffmann, K. Iwig, D. Fahlenkamp, G. Zakrzewski, E. Schmitt, T. Dannenberg, W. Lehmacher, J. von Wietersheim, C. Doehn. Adjuvant autologous renal tumour cell vaccine and risk of tumour progression in patients with renal-cell carcinoma after radical nephrectomy: phase III, randomised controlled trial. Lancet. 363, 594–99 (2004). [↩]
- A. Freedman, S. S. Neelapu, C. Nichols, M. J. Robertson, B. Djulbegovic, J. N. Winter, J. F. Bender, D. P. Gold, R. G. Ghalie, M. E. Stewart, V. Esquibel, P. Hamlin. Placebo-Controlled Phase III Trial of Patient-Specific Immunotherapy With Mitumprotimut-T and Granulocyte-Macrophage Colony-Stimulating Factor After Rituximab in Patients With Follicular Lymphoma. J Clin Oncol. 27, 3036-3043 (2009). [↩]
- T. Schulze, W. Kemmner, J. Weitz, K. D. Wernecke, V. Schirrmacher, P. M. Schlag. Efficiency of adjuvant active specific immunization with Newcastle disease virus modified tumor cells in colorectal cancer patients following resection of liver metastases: results of a prospective randomized trial. Cancer Immunology Immunotherapy. 58, 61–69 (2009). [↩]
- R. J. Amato, R. E. Hawkins, H. L. Kaufman, J. A. Thompson, P. Tomczak, C. Szczylik, M. McDonald, S. Eastty, W. H. Shingler, J. deBelin, M. Goonewardena, S. Naylor, R. Harrop. Vaccination of metastatic renal cancer patients with MVA-5T4: A randomized, double-blind, placebo-controlled Phase III study. Clinical Cancer Research. 16, 5539-47 (2010). [↩]
- D. Miles, H. Roche, M. Martin, T. J. Perren, D. A. Cameron, J. Glaspy, D. Dodwell, J. Parker, J. Mayordomo, A. Tres, J. L. Murray, N. K. Ibrahim (Theratope Study Group). Phase III Multicenter Clinical Trial of the Sialyl-TN (STn)-Keyhole Limpet Hemocyanin (KLH) Vaccine for Metastatic Breast Cancer. The Oncologist. 2011;16, 1092–1100 (2011). [↩]
- G. Middleton, P. Silcocks, T. Cox, J. Valle, J. Wadsley, D. Propper, F. Coxon, P. Ross, S. Madhusudan, T. Roques, D. Cunningham, S. Falk, N. Wadd, M. Harrison, P. Corrie, T. Iveson, A. Robinson, K. McAdam, M. Eatock, J. Evans, C. Archer, T. Hickish, A. Garcia-Alonso, M. Nicolson, W. Steward, A. Anthoney, W. Greenhalf, V. Shaw, E. Costello, D. Naisbitt, C. Rawcliffe, G. Nanson, J. Neoptolemos. Gemcitabine and capecitabine with or without telomerase peptide vaccine GV1001 in patients with locally advanced or metastatic pancreatic cancer (TeloVac): An open-label, randomised, phase 3 trial. Lancet Oncology. 15, 829-840 (2014). [↩]
- R. Levy, K. N. Ganjoo, J. P. Leonard, J. M. Vose, I. W. Flinn, R. F. Ambinder, J. M. Connors, N. L. Berinstein, A. R. Belch, N. L. Bartlett, C. Nichols, C. E. Emmanouilides, J. M. Timmerman, S. A. Gregory, B. K. Link, D. J. Inwards, A. S. Freedman, J. V. Matous, M. J. Robertson, L. A. Kunkel, D. E. Ingolia, A. J. Gentles, C. L. Liu, R. Tibshirani, A. A. Alizadeh, D. W. Denney Jr. Active Idiotypic Vaccination Versus Control Immunotherapy for Follicular Lymphoma. Journal of Clinical Oncology. 32, 1797-1803 (2014). [↩]
- G. Giaccone, L. A. Bazhenova, J. Nemunaitis, M. Tan, E. Juhasz, R. Ramlau, M. M. van den Heuvel, R. Lal, G. H. Kloecker, K. D. Eaton, Q. Chu, D. J. Dunlop, M. Jain, E. B. Garon, C. S. Davis, E. Carrier, S. C. Moses, D. L. Shawler, H. Fakhrai. A phase III study of belagenpumatucel-L, an allogeneic tumour cell vaccine, as maintenance therapy for non-small cell lung cancer. European Journal of Cancer. 51, 2321– 2329 (2015). [↩]
- D. H. Lawson, S. Lee, F. Zhao, A. A. Tarhini, K. A. Margolin, M. S. Ernstoff, M. B. Atkins, G. I. Cohen, T. L. Whiteside, L. H. Butterfield, J. M. Kirkwood. Randomized, Placebo-Controlled, Phase III Trial of Yeast-Derived GM-CSF Versus Peptide Vaccination Versus GM-CSF Plus Peptide Vaccination Versus Placebo in Patients With No Evidence of Disease After Complete Surgical Resection of Locally Advanced and/or Stage IV Melanoma: A Trial of the ECOG–ACR Imaging Network Cancer Research Group (E4697). Journal of Clinical Oncology. 33, 4066-4076 (2015). [↩]
- J. F. Vansteenkiste, B. C. Cho, T. Vanakesa, T. de Pas, M. Zielinski, M. S. Kim, J. Jassem, M. Yoshimura, J. Dahabreh, H. Nakayama, L. Havel, H. Kondo, T. Mitsudomi, K. Zarogoulidis, O. A. Gladkov, K. Udud, H. Tada, H. Hoffman, A. Bugge, P. Taylor, E. E. Gonzalez, M. L. Liao, J. He, J. Pujol, J. Louahed, M. Debois, V. Brichard, C. Debruyne, P. Therasse, N. Altorki. Efficacy of the MAGE-A3 cancer immunotherapeutic as adjuvant therapy in patients with resected MAGE-A3-positive non-small-cell lung cancer (MAGRIT): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncology. 17, 822–35 (2016). [↩]
- B. I. Rini, A. Stenzl, R. Zdrojowy, M. Kogan, M. Shkolnik, S. Oudard, S. Weikert, S. Bracarda, S. J. Crabb, J. Bedke, J. Ludwig, D. Maurer, R. Mendrzyk, C. Wagner, A. Mahr, J. Fritsche, T. Weinschenk, S. Walter, A. Kirner, H. Singh-Jasuja, C. Reinhardt, T. Eisen. IMA901, a multipeptide cancer vaccine, plus sunitinib versus sunitinib alone, as first-line therapy for advanced or metastatic renal cell carcinoma (IMPRINT): a multicentre, open-label, randomised, controlled, phase 3 trial. Lancet Oncology. 17, 1599–611 (2016). [↩]
- M. B. Faries, N. Mozzillo, M. Kashani-Sabet, J. F. Thompson, M. C. Kelley, R. C. DeConti, J. E. Lee, J. F. Huth, J. Wagner, A. Dalgleish, D. Pertschuk, C. Nardo, S. Stern, R. Elashoff, G. Gammon, D. L. Morton, (MMAIT-IV Clinical Trial Group). Long-Term Survival after Complete Surgical Resection and Adjuvant Immunotherapy for Distant Melanoma Metastases. Annals of Surgical Oncology. 24, 3991–4000 (2017). [↩]
- M. Weller, N. Butowski, D. D. Tran, L. D. Recht, M. Lim, H. Hirte, L. Ashby, L. Mechtler, S. A. Goldlust, F. Iwamoto, J. Drappatz, D. M. O’Rourke, M. Wong, M. G. Hamilton, G. Finocchiaro, J. Perry, W. Wick, J. Green, Y. He, C. D. Turner, M. J. Yellin, T. Keler, T. A. Davis, R. Stupp, J. H. Sampson (ACT IV Trial Investigators). Lancet Oncology. 18, 1373–85 (2017). [↩]
- Y. Narita, Y. Arakawa, F. Yamasaki, R. Nishikawa, T. Aoki, M. Kanamori, M. Nagane, T. Kumabe, Y. Hirose, T. Ichikawa, H. Kobayashi, T. Fujimaki, H. Goto, H. Takeshima, T. Ueba, H. Abe, T. Tamiya, Y. Sonoda, A. Natsume, T. Kakuma, Y. Sugita, N. Komatsu, A. Yamada, T. Sasada, S. Matsueda, S. Shichijo, K. Itoh, M. Terasaki. A randomized, double-blind, phase III trial of personalized peptide vaccination for recurrent glioblastoma. Neuro-Oncology. 21, 348–359 (2019). [↩]
- E. A. Mittendorf, B. Lu, M. Melisko, J. P. Hiller, I. Bondarenko, A. M. Brunt, G. Sergii, K. Petrakova, G. E. Peoples. Efficacy and Safety Analysis of Nelipepimut-S Vaccine to Prevent Breast Cancer Recurrence: A Randomized, Multicenter, Phase III Clinical Trial. Clin Cancer Res. 25, 4248–54 (2019). [↩]
- M. Noguchi, K. Fujimoto, G. Arai, H. Umera, K. Hashine, H. Matsumoto, S. Fukasawa, Y. Kohkimoto, H. Nakatsu, A. Takenaka, M. Fujisawa, H. Uemura, S. Naito, S. Egawa, H. Fujimoto, S. Hinotsu, K. Itohi. A randomized phase III trial of personalized peptide vaccination for castration‑resistant prostate cancer progressing after docetaxel. Oncology Reports. 45, 159-168 (2021). [↩]
- T. J. Vreeland, G. T. Clifton, D. F. Hale, R. C. Chick, A. T. Hickerson, J. L. Cindass, A. M. Adams, P. M. K. Bohan, R. H. I. Andtbacka, A. C. Berger, J. W. Jakub, J. J. Sussman, A. M. Terando, T. Wagner, G. E. Peoples, Mark B. Faries. A Phase IIb Randomized Controlled Trial of the TLPLDC Vaccine as Adjuvant Therapy After Surgical Resection of Stage III/IV Melanoma: A Primary Analysis. Annals of Surgical Oncology. 28, 6126–6137 (2021). [↩]
- D. B. Hewitt, N. Nissen, H. Hatoum, B. Musher, J. Seng, A. L. Coveler, R. Al-Rajabi, C. J. Yeo, B. Leiby, J. Banks, L. Balducci, G. Vaccaro, N. LoConte, T. J. George, W. Brenner, E. Elquza, N. Vahanian, G. Rossi, E. Kennedy, C. Link, H. Lavu. A Phase 3 Randomized Clinical Trial of Chemotherapy With or Without Algenpantucel-L (HyperAcute-Pancreas) Immunotherapy in Subjects With Borderline Resectable or Locally Advanced Unresectable Pancreatic Cancer. Annals of Surgery. 275, 45-53 (2022). [↩]
- D. J. Schwartzentruber, D. H. Lawson, J. M. Richards, R. M. Conry, D. M. Miller, J. Treisman, F. Gailani, L. Riley, K. Conlon, B. Pockaj, K. L. Kendra, R. L. White, R. Gonzalez, T. M. Kuzel, B. Curti, P. D. Leming, E. D. Whitman, J. Balkissoon, D. S. Reintgen, H. Kaufman, F. M. Marincola, M. J. Merino, S. A. Rosenberg, P. Choyke, D. Vena, P. Hwu. gp100 Peptide Vaccine and Interleukin-2 in Patients with Advanced Melanoma. New England Journal of Medicine. 364,2119-27 (2011). [↩]
- S. J. Schuster, S. S. Neelapu, B. L. Gause, J. E. Janik, F. M. Muggia, J. P. Gockerman, J. N. Winter, C. R. Flowers, D. A. Nikcevich, E. M. Sotomayor, D. S. McGaughey, E. S. Jaffe, E. A. Chong, C. W. Reynolds, D. A. Berry, C. F. Santos, M. A. Popa, A. M. McCord, L. W. Kwak. Vaccination With Patient-Specific Tumor-Derived Antigen in First Remission Improves Disease-Free Survival in Follicular Lymphoma. Journal of Clinical Oncology. 29, 2787-2794 (2011). [↩]
- R. O. Dillman, A. N. Cornforth, C. DePriest, E. F. McClay, T. T. Amatruda, C. de Leon, R. E. Ellis, C. Mayorga, D. Carbonell, J. M. Cubellisz. Journal of Immunotherapy. 35, 641–649 (2012). [↩]
- S. Alfonso, A. Valdes-Zayas, E. R. Santiesteban, Y. I. Flores, F. Areces, M. Hernandez, C. E. Viada, I. C. Mendoza, P. P. Guerra, E. García, R. A. Ortiz, A. V. de la Torre, M. Cepeda, K. Perez, E. Chong, A. M. Hernandez, D. Toledo, Z. Gonzalez, Z. Mazorra, T. Crombet, R. Perez, A. M. Vazquez, A. E. Macías. A Randomized, Multicenter, Placebo-Controlled Clinical Trial of Racotumomab-Alum Vaccine as Switch Maintenance Therapy in Advanced Non–Small Cell Lung Cancer Patients. Clin Cancer Res. 20, 3660–71 (2014). [↩]
- E. Quoix, H. Lena, G. Losonczy, F. Forget, C. Chouaid, Z. Papai, R. Gervais, C. Ottensmeier, A. Szczesna, A. Kazarnowicz, J. T. Beck, V. Westeel, E. Felip, D. Debieuvre, A. Madroszyk, J. Adam, G. Lacoste, A. Tavernaro, B. Bastien, C. Halluard, T. Palanché, J. Limacher. TG4010 immunotherapy and first-line chemotherapy for advanced non-small-cell lung cancer (TIME): results from the phase 2b part of a randomised, double-blind, placebo-controlled, phase 2b/3 trial. Lancet Oncology. 17, 212–23 (2016). [↩]
- D. Jocham, A. Richter, L. Hoffmann, K. Iwig, D. Fahlenkamp, G. Zakrzewski, E. Schmitt, T. Dannenberg, W. Lehmacher, J. von Wietersheim, C. Doehn. Adjuvant autologous renal tumour cell vaccine and risk of tumour progression in patients with renal-cell carcinoma after radical nephrectomy: phase III, randomised controlled trial. Lancet. 363, 594–99 (2004). [↩]
- K. Palucka, J. Banchereau. Cancer immunotherapy via dendritic cells. Nature Reviews Cancer. 12, 265–277 (2012). [↩]
- O. J. Finn. Cancer vaccines: between the idea and the reality. Nature Reviews Immunology. 3, 630–641 (2003). [↩]
- I. Melero, G. Gaudernack, W. Gerritsen, A. Horel, A. P. Griset, S. van der Burg, C. Chalouni. Therapeutic vaccines for cancer: an overview of clinical trials. Nature Reviews Clinical Oncology. 12, 384–396 (2015). [↩]
- A. Sottoriva, I. Spiteri, S. G. M. Piccirillo, A. Touloumis, V. P. Collins, J. C. Marioni, C. Curtis, C. Watts, S. Tavaré. Intratumor heterogeneity in human glioblastoma reflects cancer evolutionary dynamics. Proceedings of the National Academy of Sciences. 110, 4009–4014 (2013). [↩]
- D. A. Landau, S. L. Carter, P. Stojanov, A. McKenna, K. Stevenson, M. S. Lawrence, C. Sougnez, C. Stewart, A. Sivachenko, L. Wang, Y. Wan, W. Zhang, S. A. Shukla, A. Vartanov, S. M. Fernandes, G. Saksena, K. Cibulskis, B. Tesar, S. Gabriel, N. Hacohen, M. Meyerson, E. S. Lander, D. Neuberg, J. R. Brown, G. Getz, C. J. Wu. Evolution and impact of sub-clonal mutations in chronic lymphocytic leukemia. Cell. 152, 714-26 (2013). [↩]
- J. Peng, B. Sun, C. Chen, J. Zhou, Y. Chen, H. Chen, L. Liu, D. Huang, J. Jiang, G. Cui, Y. Yang, W. Wang, D. Guo, M. Dai, J. Guo, T. Zhang, Q. Liao, Y. Liu, Y. Zhao, D. Han, Y. Zhao, Y. Yang, W. Wu. Single-cell RNA-seq highlights intra-tumoral heterogeneity and malignant progression in pancreatic ductal adenocarcinoma. Cell Research. 29, 725–738 (2019). [↩]
- Z. Zhang, X. Sun, Y. Liu, Y. Zhang, Z. Yang, J. Dong, N. Wang, J. Ying, M. Zhou, L. Yang. Spatial Transcriptome-Wide Profiling of Small Cell Lung Cancer Reveals Intra-Tumoral Molecular and Subtype Heterogeneity. Advanced Science. 11, 2402716 (2024). [↩]
- L. Mamanova, J. B. Neves, L. Bolt, E. S. Fasouli, A. R. J. Lawson, M. D. Young, Y. Hooks, T. R.W. Oliver, T. M. Butler, J. N. Armitage, T. Aho, A. C. P. Riddick, V. Gnanapragasam, S. J. Welsh, K. B. Meyer, A. Y. Warren, M. G. B. Tran, G. D. Stewart, I. Cortes-Ciriano, S. Behjati, M. R. Clatworthy, P. J. Campbell, S. A. Teichmann, T. J. Mitchell. Mapping single-cell transcriptomes in the intra-tumoral and associated territories of kidney cancer. Cancer Cell. 40, 1583–1599 (2022). [↩]
- P. V. Sonar, A. K. Singh, S. Mandadi, N. K. Sharma. Expanding horizons of cancer immunotherapy: Hopes and hurdles. Frontiers of Oncology. 15, doi: 10.3389/fonc.2025.1511560 (2025). [↩] [↩]
- N. P. Restifo, F. M. Marincola, Y. Kawakami, J. K. Taubenberger, J. R. Yannelli, S. A. Rosenberg. A. Loss of functional beta 2–microglobulin in metastatic melanomas from five patients receiving immunotherapy. Journal of Experimental Medicine. 183, 977–985 (1996). [↩]
- C. Uyttenhove, L. Pilotte, I. Théate, V. Stroobant, D. Colau, N. Parmentier, T. Boon, B. J. Van den Eynde. Evidence for a tumoral immune resistance mechanism based on tryptophan degradation by indoleamine 2,3-dioxygenase. Nature Medicine. 9, 1269–1274 (2003). [↩]
- H. Dong, S. E. Strome, D. R. Salomao, H. Tamura, F. Hirano, D. B. Flies, P. C. Roche, J. Lu, G. Zhu, K. Tamada, V. A. Lennon, E. Celis, L. Chen. Tumor-associated B7-H1 promotes T-cell apoptosis: A potential mechanism of immune evasion. Nature Medicine. 8, 793–800 (2002). [↩]
- J. M. Zaretsky, A. Garcia-Diaz, D. S. Shin, H. Escuin-Ordinas, W. Hugo, S. Hu-Lieskovan, D. Y. Torrejon, G. Abril-Rodriguez, S. Sandoval, L. Barthly, J. Saco, B. H. Moreno, R. Mezzadra, B. Chmielowski, K. Ruchalski, I. P. Shintaku, P. J. Sanchez, C. Puig-Saus, G. Cherry, E. Seja, X. Kong, J. Pang, B. Berent-Maoz, B. Comin-Anduix, T. G. Graeber, P. C Tumeh, T. N. Schumacher, R. S. Lo, A. Ribas. Mutations Associated with Acquired Resistance to PD-1 Blockade in Melanoma. New England Journal of Medicine. 375, 819–829 (2016). [↩]
- S. Spranger, R. Bao, T. F. Gajewski. Melanoma-intrinsic β-catenin signaling prevents anti-tumor immunity. Nature. 523, 231–235 (2013). [↩]
- M. Saxena, S. H. van der Burg, C. J. M. Melief, N. Bhardwaj. Therapeutic cancer vaccines. Nature Reviews Cancer. 21, 360-378 (2021). [↩]
- A. Chow, K. Perica, C. A. Klebanoff, J. D. Wolchok. Clinical implications of T cell exhaustion for cancer immunotherapy. Nature Reviews Clinical Oncology. 19, 775-790 (2022). [↩]
- D. Fourcade, Z. Sun, M. Benalla, P. Guillaume, I Luescher, C. A. Sander, J. M. Kirkwood, H. M. Zarour. Upregulation of Tim-3 and PD-1 expression is associated with tumor antigen–specific CD8+ T cell dysfunction in melanoma patients. Journal of Clinical Investigation. 122, 235–244 (2012). [↩]
- D. S. Thommen, V. H. Koelzer, P. Herzig, A. Roller, M. Trefny, S. Dimeloe, R. Kiessling, V. Sintchenko, B. Becher. A transcriptionally and functionally distinct PD-1(+) CD8(+) T cell pool with predictive potential in non–small-cell lung cancer treated with PD-1 blockade. Nature Medicine. 24, 994–1004 (2018). [↩]
- C. Zheng, L. Zheng, J. K. Yoo, H. Guo, Y. Zhang, X. Guo, B. Kang, R. Hu, J. Y. Huang, Q. Zhang, Z. Liu, M. Dong, X. Hu, W. Ouyang, J. Peng, Z. Z. Zhang. Landscape of Infiltrating T Cells in Liver Cancer Revealed by Single-Cell Sequencing. Cell. 169, 1342–1356 (2017). [↩]
- M. Sade-Feldman, K. Yizhak, S. L. Bjorgaard, J. P. Ray, C. G. de Boer, R. W. Jenkins, D. J. Lieb, J. H. Chen, D. T. Frederick, M. Barzily-Rokni, S. S. Freeman, A. Reuben, P. J. Hoover, A. Villani, E. Ivanova, A. Portell, P. H. Lizotte, A. R. Aref, J. Eliane, M. R. Hammond, H. Vitzthum, S. M. Blackmon, B. Li, V. Gopalakrishnan, S. M. Reddy, Z. A. Cooper, C. P. Paweletz, D. A. Barbie, A. Stemmer-Rachamimov, K. T. Flaherty, J. A. Wargo, G. M. Boland, R. J. Sullivan, G. Getz, N. Hacohen. Defining T Cell States Associated with Response to Checkpoint Immunotherapy in Melanoma. Cell. 175, 998–1013 (2018). [↩]
- P. V. Sonar, A. K. Singh, S. Mandadi, N. K. Sharma. Expanding horizons of cancer immunotherapy: Hopes and hurdles. Frontiers of Oncology. 15, doi: 10.3389/fonc.2025.1511560 (2025). [↩]
- M. R. Zamani, P. Sacha. Immune checkpoint inhibitors in cancer therapy: what lies beyond monoclonal antibodies? Medical Oncology. 42, 273 (2025). [↩]
- P. D. Katsikis, K. J. Ishii, C. Schliehe. Challenges in developing personalized neoantigen cancer vaccines. Nature Reviews Immunology. 24, 213–227 (2024). [↩] [↩]
- D. Lucchetti, I. V. Zurlo, F. Colella, C. Ricciardi‑Tenore, M. Di Salvatore, G. Tortora, R. De Maria, F. Giuliante, A. Cassano, M. Basso, A. Crucitti, I. Laurenzana, G. Artemi, A. Sgambato. Mutational status of plasma exosomal KRAS predicts outcome in patients with metastatic colorectal cancer. Scientific Reports. 11, 22686 (2021). [↩]
- J. Galon, F. Pagès, F. M. Marincola, H. K. Angell, M. Thurin, A. Lugli, I. Zlobec, A. Berger, C. Bifulco, G. Botti, F. Tatangelo, C. M. Britten, S. Kreiter, L. Chouchane, P. Delrio, H. Arndt, M. Asslaber, M. Maio, G. V. Masucci, M. Mihm, F. Vidal-Vanaclocha, J. P. Allison, S. Gnjatic, L. Hakansson, C. Huber, H. Singh-Jasuja, C. Ottensmeier, H. Zwierzina, L. Laghi, F. Grizzi, P. S. Ohashi, P. A. Shaw, B. A. Clarke, B. G. Wouters, Y. Kawakami, S. Hazama, K. Okuno, E. Wang, J. O’Donnell-Tormey, C. Lagorce, G. Pawelec, M. I. Nishimura, R. Hawkins, R. Lapointe, A. Lundqvist, S. N. Khleif, S. Ogino, P. Gibbs, P. Waring, N. Sato, T. Torigoe, K. Itoh, P. S. Patel, S. N. Shukla, R. Palmqvist, I. D. Nagtegaal, Y. Wang, C. D’Arrigo, S. Kopetz, F. A. Sinicrope, G. Trinchieri, T. F. Gajewski, P. A. Ascierto, B. A. Fox. Cancer classification using the Immunoscore: a worldwide task force. Journal of Translational Medicine. 10, 205 (2012). [↩]
- H. Yin, M. Zhang, Y. Zhang, X. Zhang, X. Zhang, B. Zhang. Liquid biopsies in cancer. Molecular Biomedicine. 6, 18 (2025). [↩]
- P. D. Katsikis, K. J. Ishii, C. Schliehe. Challenges in developing personalized neoantigen cancer vaccines. Nature Reviews Immunology. 24, 213–227 (2024). [↩]
- M. Saxena, S. H. van der Burg, C. J. M. Melief, N. Bhardwaj. Therapeutic cancer vaccines. Nature Reviews Cancer. 21, 360-378 (2021). [↩]
- H. Yin, M. Zhang, Y. Zhang, X. Zhang, X. Zhang, B. Zhang. Liquid biopsies in cancer. Molecular Biomedicine. 6, 18 (2025). [↩]
- J. S. Weber, M. S. Carlino, A. Khattak, T. Meniawy, G. Ansstas, M. H. Taylor, K. B. Kim, M. McKean, G. V. Long, R. J. Sullivan, M. Faries, T. T. Tran, C. L. Cowey, A. Pecora, M. Shaheen, J. Segar, T. Medina, V. Atkinson, G. T. Gibney, J. J. Luke, S. Thomas, E. I. Buchbinder, J. A. Healy, M. Huang, M. Morrissey, I. Feldman, V. Sehgal, C. Robert-Tissot, P. Hou, L. Zhu, M. Brown, P. Aanur, R. S. Meehan, T. Zaks. Individualised neoantigen therapy mRNA-4157 (V940) plus pembrolizumab versus pembrolizumab monotherapy in resected melanoma (KEYNOTE-942): a randomised, phase 2b study. Lancet. 403, 632–644 (2024). [↩] [↩] [↩] [↩] [↩]
- Moderna. Moderna and Merck Announce mRNA-4157/V940, an Investigational Personalized mRNA Cancer Vaccine, in Combination With KEYTRUDA(R) (pembrolizumab), was Granted Breakthrough Therapy Designation by the FDA for Adjuvant Treatment of Patients With High-Risk Melanoma Following Complete Resection. News Release (2024). [↩]
- Merck. Merck and Moderna Initiate Phase 3 Study Evaluating V940 (mRNA-4157) in Combination with KEYTRUDA® (pembrolizumab) for Adjuvant Treatment of Patients with Resected High-Risk (Stage IIB-IV) Melanoma. News Release (2023). [↩] [↩]
- US FDA. Focus area: Individualized therapeutics and precision medicine. Accessed on 31-Oct-2025 from URL: Focus Area: Individualized Therapeutics and Precision Medicine | FDA. [↩]
- M. R. Zamani, P. Sacha. Immune checkpoint inhibitors in cancer therapy: what lies beyond monoclonal antibodies? Medical Oncology. 42, 273 (2025). [↩] [↩]
- P. V. Sonar, A. K. Singh, S. Mandadi, N. K. Sharma. Expanding horizons of cancer immunotherapy: Hopes and hurdles. Frontiers of Oncology. 15, doi: 10.3389/fonc.2025.1511560 (2025). [↩] [↩]
- M. R. Zamani, P. Sacha. Immune checkpoint inhibitors in cancer therapy: what lies beyond monoclonal antibodies? Medical Oncology. 42, 273 (2025). [↩]
- T. Fan, M. Zhang, J. Yang, Z. Zhu, W. Cao, C. Dong. Therapeutic cancer vaccines: advancements, challenges, and prospects. Signal Transduction and Targeted Therapy. 450, https://doi.org/10.1038/s41392-023-01674-3 (2023). [↩]
- J. G. Berdeja, D. Madduri, S. Z. Usmani, A. Jakubowiak, M. Agha, A. D. Cohen, A. K. Stewart, P. Hari, M. Htut, A. Lesokhin, A. Deol, N. C. Munshi, E. O’Donnell, D. Avigan, I. Singh, E. Zudaire, T. Yeh, A. J. Allred, Y. Olyslager, A. Banerjee, C. C. Jackson, J. D. Goldberg, J. M. Schecter, W. Deraedt, S. H. Zhuang, J. Infante, D. Geng, X. Wu, M. J. Carrasco-Alfonso, M. Akram, F. Hossain, S. Rizvi, F. Fan, Y. Lin, T. Martin, S. Jagannath. Ciltacabtagene autoleucel, a B-cell maturation antigen-directed chimeric antigen receptor T-cell therapy in patients with relapsed or refractory multiple myeloma (CARTITUDE-1): A phase 1b/2 open-label study. Lancet. 398, 314-324 (2021). [↩]
- S. Jagannath, T. G. Martin, Y. Lin, A. D. Cohen, N. Raje, M. Htut, A. Deol, M. Agha, J. G. Berdeja, A. M. Lesokhin, J. J. Liegel, A. Rossi, A. Lieberman-Cribbin, S. Z. Usmani, B. Dhakal, S. Parekh, H. Li, F. Wang, R. Montes de Oca, V. Plaks, H. Sun, A. Banerjee, J. M. Schecter, N. Lendvai, D. Madduri, T. Lengil, J. Zhu, M. Koneru, M. Akram, N. Patel, O. Costa Filho, A. J. Jakubowiak, P. M. Voorhees. Long-Term (≥5-Year) Remission and Survival After Treatment With Ciltacabtagene Autoleucel in CARTITUDE-1 Patients With Relapsed/Refractory Multiple Myeloma. Journal of Clinical Oncology. 43, 2766-2771 (2025). [↩]