Abstract
Research has shown that an increase of cortisol levels can increase drug use in both females and males. Recent studies have focused on how sex differences play a role in addiction, and specifically how they could be affected by stress through the HPA axis, a part of the endocrine system that plays a key role in our body’s response to stress. This literature review, using articles sourced from Google Scholar and Pub Med, reviews how alcohol, tobacco, and the HPA axis are affected by sex differences and stress. Studies were used based on relevance to HPA dysregulation, cortisol, and sex differences, with inclusion criteria of peer-reviewed, randomized trials with both sexes. Overall, females had higher levels of cortisol after withdrawals from alcohol and nicotine, which led to females relapsing faster than males, who had more stress when under the influence of the drug. Hormones such as estrogen and progesterone also played a role. Women have a more sensitive HPA response when in withdrawal, highlighting the necessity for hormone-based therapies which can be used to reduce stress-induced cravings. These findings show a need for sex-specific treatments which can counteract addiction. Some overarching limitations of the existing literature were a lack of longitudinal data and no distinguishment between females and males. Future research can include the effect of opioids and other stimulants on the HPA, as well as the effect of age on stress levels.
Introduction
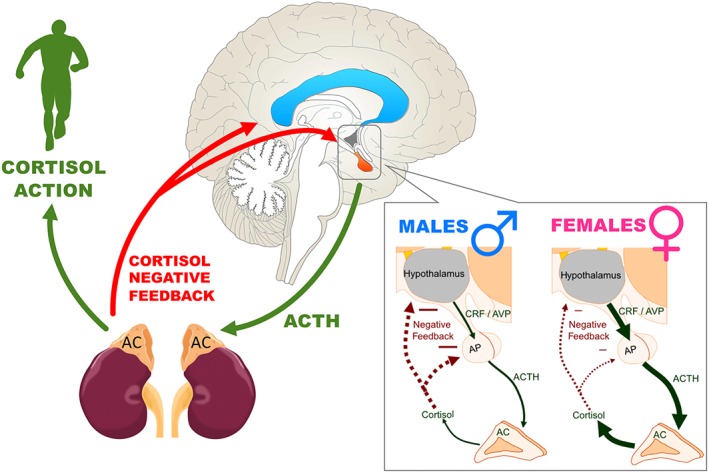
The hypothalamic-pituitary-adrenal axis, or HPA system, plays an important role in regulating the body’s homeostasis by using a cascade of signals in response to stress. Stress triggers the release of corticotropin releasing factor (CRF), a hormone that stimulates the anterior pituitary gland. This gland then releases adrenocorticotropin hormone (ACTH), which travels through the bloodstream to the adrenal cortex and increases the rate of production for cortisol1. Repeated drug use alters HPA axis function, which increases or decreases cortisol levels based on substance type and context. This stress, combined with the rewarding effects of drugs, reinforces drug-seeking behavior and perpetuates repeated use2.
Using animal models, researchers found that a dysfunctional HPA response to stress is associated with an increased rate in self-administering addictive-like substances3. Repeated drug use also contributes to an unregulated cortisol production. Chronic exposure to substances, such as cocaine and alcohol, can overactivate the HPA axis and disrupt the body’s homeostasis4. Individuals with alcohol dependence can also create a blunted cortisol response to stress over time, reflecting in a desensitized HPA5. These findings are significant because they show how HPA axis dysregulation can perpetuate a cycle of addiction by impairing the body’s natural stress mechanisms. Results of this review can be used to better understand relapse rates between sexes and how they can be prevented to maintain sobriety.
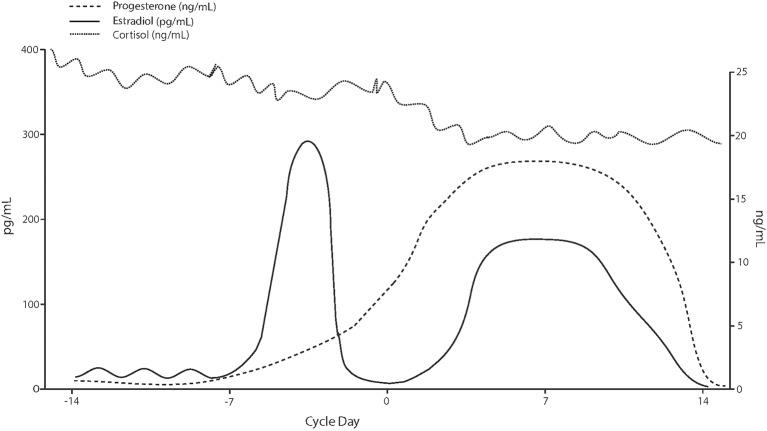
Sex differences significantly change the HPA’s response to stress and substance use, influencing heart rate and cortisol excretion significantly6. Estrogen and progesterone are two key hormones to the female reproductive system and menstrual cycle (Fig. 2). Depending on the phase of the menstrual cycle, females can have different HPA sensitivity to drugs during the withdrawal period7.
Previous literature reviews focusing on the HPA axis and stress have not investigated how sex differences play a role. It is important to research this because it can help with our understanding of why some individuals may be more vulnerable to relapse quicker than others. The present literature review examines how sex differences play a role in how the HPA Axis is being affected by drug use.
Methods
This literature review consists of peer-reviewed journals, pre-clinical studies, and clinical studies. With Google Scholar and PubMed the key words are HPA Axis, relapse, sex differences, various drug names, self-administration, cue-induced, withdrawal, drug-seeking, reinstatement. Google Scholar and PubMed were the primary database as it provided for a wide range of articles that related to the biomedical sciences. Most studies were from within the last twenty years were primarily used with the oldest source discussing nicotine use, with filters for peer-reviewed articles. Older studies were included as they discussed core principals, which this research paper was built upon, in depth. Almost thirty studies were selected for use based on the relevance to HPA axis dysregulation, cortisol responses, and sex differences in contexts of drug use. Inclusion criteria contained randomized controlled trials, studies over longer periods of time, and recent publication dates. Exclusion criteria were non-peer-reviewed sources, unrelated topics, and studies that did not specify or include both sexes. Over fifty articles with a variety of data were screened which included research designs, preclinical and clinical findings, and the result of cortisol and ACTH levels after a stimulus during the experiment. The information was categorized into key themes such as repeated use of alcohol and tobacco, relapse, and HPA axis involvement. These groupings were then analyzed to identify overarching patterns and inform the review’s conclusions.
Results
Alcohol
Acute alcohol consumption stimulates the HPA axis, increasing ACTH and cortisol levels in both females and males, yet the magnitude of these responses differ9. Studies show that men exhibit a higher cortisol increase following alcohol intake when compared to women. However, chronic alcohol exposure and withdrawal reveal a contrasting pattern: Women have greater hyperactivity in the HPA axis during withdrawal, with elevated cortisol levels reflecting greater stress sensitivity9. Women with longer alcohol use had an increased cortisol awakening response (CAR) compared to moderate drinkers, an indication of a dysregulated HPA axis as CAR measures the rise in cortisol when waking up (Fig. 3)10. This leads to a chronic alcohol induced desensitization of the HPA axis that makes females more vulnerable which can lead to cravings and relapse risk11. Adolescent rats who were exposed to social isolation stress, a type of chronic stress, had an increased alcohol intake during adulthood, particularly females, which indicated an increased vulnerability to self-administration of alcohol because of an altered HPA axis during development12.
Tobacco/Nicotine
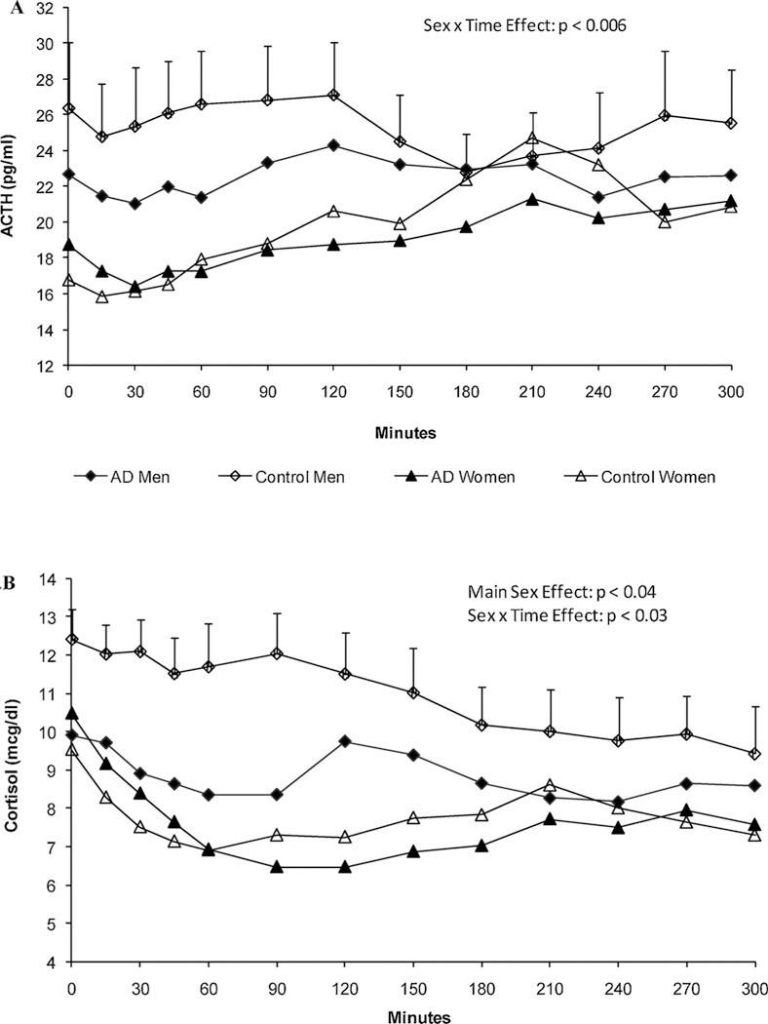
Nicotine, the primary component of tobacco, stimulates the HPA axis and leads to significant increases in cortisol levels13. This response is more noticeable in chronic smokers, suggesting that repeated nicotine use can sustain HPA axis hyperactivity14,13. Compared to men, women exhibit a blunted cortisol response to nicotine-induced stress, which increases their reliance on it. This response is also accompanied with lower ACTH levels, which correlated with a higher risk of relapse14. However, when conducting a preclinical study with the estrous cycle kept in mind, female rats had a greater ACTH and corticosterone response to nicotine than males15. For human males, high-nicotine cigarettes also increased ACTH significantly which was later strongly correlated to an increase of nicotine in the plasma16. During early periods of cessation, men had cravings for nicotine earlier than women did, but did not end up leading to relapse17. In preclinical studies researching behavior after chronic nicotine use, female rats had the greatest effects with decreased feeding and body weight and an increase in stress. Chronic use had little to no of the same effects on males18.
Estrogen/progesterone
Estrogen and progesterone play important roles in changing HPA activity, influencing stress-induced cortisol production and susceptibility to drug use19. Estrogen enhances HPA axis reactivity by increasing corticotropin-releasing hormone (CRH) in the hypothalamus, leading to higher ACTH and cortisol levels during stress, particularly in high-estrogen states, like the follicular phase of the menstrual cycle (Fig. 3). This response amplifies stress sensitivity, increasing drug-seeking behavior20. Progesterone however, shows the opposite effect and is associated with decreased stress (Fig. 3). Previous studies have shown that high progesterone is associated with decreased drug cue-induced cravings, cue-induced anxiety, and blood pressure21. When testing if naltrexone, a medication used to treat alcohol use disorder, has a different response on the HPA axis in both sexes, results showed that it significantly increased ACTH and cortisol levels in women, especially when in a “high estrogen” phase of their menstrual cycle (Fig. 3), but not as much in men. A later analysis also showed that smoking one cigarette led to ACTH increases when using naltrexone22. Women with substance use disorders exhibit higher cortisol levels during the follicular phase, where estrogen levels are the highest, correlating with increased drug-seeking behavior compared to the luteal phase, when progesterone levels are higher (Fig. 3)23.
Relapse
The amount of cortisol produced by the HPA Axis is associated with the ability for an addicted individual to stay in a long-term treatment facility. The less cortisol produced, the longer one can abstain from an addictive substance3. Additionally, heightened HPA activity during early abstinence predicts risk of relapsing. Higher cortisol levels to drug-related cues and stress in those with substance use disorders are associated with more frequent cravings and a higher likelihood of relapse within a couple weeks of initiating treatment24. Women with cocaine dependence exhibited a blunted cortisol response and a more pronounced blunted ACTH response when compared to men. This, which can be triggered by negative emotions or conflicts, can contribute to higher relapse rates in females25. When researching if stress-induced or drug cue-induced had the greater effect on cocaine cravings after relapse, after 30 days of abstinence, stressed males were more likely to have cocaine-induced cravings when compared to non-stressed males. However, females had no differences in results between the two, an outlier according to the current literature26. Other researchers found that stress-induced cues had a greater effect on relapse time and the subsequent cortisol responses predicted a greater cocaine use after 90 days. Additionally, treatment centers that addressed stress-induced cocaine cravings could improve relapse outcomes24.
Discussion
The HPA axis plays a critical role in substance use disorders and significantly regulates stress responses. When these substances are abused, this system is dysregulated and can lead to elevated levels of ACTH and cortisol, which can reinforce addiction through reward pathways and stress. Chronic alcohol and nicotine use, however, can result in a blunted HPA response, which leads to tolerance towards the substance and an increased chance of relapse. Sex differences can add to this, with preclinical and clinical evidence showing that estrogen and progesterone can change the HPA’s reactivity. Females generally exhibit greater HPA sensitivity, especially during periods of high-estrogen.
Females were generally more negatively affected from the HPA axis after periods of substance use, particularly from alcohol and tobacco. During withdrawal periods, females had a greater HPA response, alongside a blunted cortisol response during specific stress tasks, such as cue-induced scenarios, possibly because of an adaptive desensitization from one’s hormonal fluctuations27. This increased both the rate and chance of relapse. Females were found to have a higher base level of cortisol, which only increased with the use of drugs. With alcohol dependence, males had higher levels of stress and stronger cortisol increases when compared to females. This may have created tolerance and contribute to the a lower relapse risk by decreasing their stress cues during abstinence, despite males having a higher vulnerability during use9. For chronic nicotine use, exposure sustained HPA activation in females while females had a more pronounced cortisol and ACTH response, which correlated with greater substance dependence.
Hormonal factors also played a role in the HPA’s effect on both sexes as estrogen levels lead to a release of CRH which causes higher ACTH and stress levels. Progesterone, however, causes an opposite reaction and is associated with stress and drug-induced cravings. During the estrogen phase, women had an increase in drug-seeking behaviors when compared to progesterone, which decreased cue-induced anxiety and substance cravings. Understanding how these hormones affect drug seeking behaviors in females, can help us further understand why or how these sex differences are occurring.
Despite these findings, gaps remain in the literature, particularly due to the limited exploration of other substances such as opioids or stimulants. Additionally, age-related effects, especially during adolescence and puberty, remain unexplored. Through this, we could discover if hormones truly do play a role in stress levels by comparing adolescents who have not gone through puberty yet. Social factors further these biological differences as societal stigma and gender norms have historically framed addiction as a predominantly male issue. This has led men to be more vocal and likely to seek help, while women may internalize their struggles, face greater shame from their families and communities to get help, and use more substances to cope27. Some limitations that were present in the studies were a few number of clinical trials and insufficient longitudinal data, especially in studies on relapse. Many studies also did not distinguish between male and female participants, leading to no tracking of the menstrual cycle in females as a variable. This would have been especially important in studies on the hormonal effects on stress and when researching why females and males have different relapse rates. As many of the cited sources were conducted on animals, it is important to recognize that the fundamental physiological and neurological differences between animals and humans can contribute to limitations in translating the results between species.
This research can be used to create personalized prevention and relapse treatment that is sex-specific. If hormones like estrogen and progesterone are influencing the HPA Axis that deeply, then medications or treatments that specifically target them would be more beneficial and effective than more uniform treatments. For those with hormonal disorders or imbalances, such as those with estrogen, progesterone, testosterone, or cortisol, they may show signs of amplified addiction behaviors. These conditions can disrupt the HPA axis function, increase stress sensitivity, and change reward pathways, creating an overall increased vulnerability to addiction4. Further research can include the effect of opioids and age on stress levels, particularly the role of puberty. Sex differences profoundly influence how alcohol and nicotine affect the HPA axis, with females often showing greater vulnerability to stress-related dysregulation and relapse. Currently in the literature, there is little research on the effect that sex-differences have on relapse. In this review, some studies found a correlation between sex-differences, drug use, and relapse while other studies showed no clear relationship. Additionally, research can be done to determine why these sex differences exist through a biological and evolutionary lens and if that can affect other stress-related disorders. Further studies need to be conducted to understand how and if sex differences and stress play a role in drug use. Using these findings, this literature review highlights the need for a more personalized approach in addiction management, ultimately breaking cycles of dependence and increasing long-term sobriety.
References
- LaFond, M., DeAngelis, B., & al’Absi, M. (2024). Hypothalamic Pituitary Adrenal and Autonomic Nervous System Biomarkers of Stress and Tobacco Relapse: Review of the Research. Biological Psychology, 108854. [↩]
- Fox HC, Sinha R. Sex differences in drug-related stress-system changes: implications for treatment in substance-abusing women. Harv Rev Psychiatry. 2009;17(2):103-119. [↩]
- Daughters, S. B., Richards, J. M., Gorka, S. M., & Sinha, R. (2009). HPA axis response to psychological stress and treatment retention in residential substance abuse treatment: a prospective study. Drug and alcohol dependence, 105(3), 202-208. [↩] [↩]
- Koob. G., & Kreek, M. J. (2007). Stress, dysregulation of drug reward pathways, and the transition to drug dependence. American journal of psychiatry, 164(8), 1149-1159. [↩] [↩]
- Lovallo, W. R., Cohoon, A. J., Acheson, A., Sorocco, K. H., & Vincent, A. S. (2019). Blunted stress reactivity reveals vulnerability to early life adversity in young adults with a family history of alcoholism. Addiction, 114(5), 798-806. [↩]
- Collins, A., & Frankenhaeuser, M. (2010). Stress Responses in Male and Female Engineering Students. Journal of Human Stress, 4(2), 43–48. [↩]
- Goletiani, N. V., Siegel, A. J., Lukas, S. E., & Hudson, J. I. (2015). The effects of smoked nicotine on measures of subjective states and hypothalamic-pituitary-adrenal axis hormones in women during the follicular and luteal phases of the menstrual cycle. Journal of addiction medicine, 9(3), 195-203. [↩]
- Kokras, N., Hodes, G. E., Bangasser, D. A. & Dalla, C. (2019). Sex differences in the hypothalamic-pituitary-adrenal axis: an obstacle to antidepressant drug development?. British journal of pharmacology, 176(21), 4090-4106. [↩]
- Anthenelli, R. M., Heffner, J. L., Blom, T. J., Daniel, B. E., McKenna, B. S., & Wand, G. S. (2018). Sex differences in the ACTH and cortisol response to pharmacological probes are stressor-specific and occur regardless of alcohol dependence history. Psychoneuroendocrinology 94, 72-82. [↩] [↩] [↩] [↩]
- Badrick, E., Bobak, M., Britton, A., Kirschbaum, C., Marmot, M., & Kumari, M. (2008). The relationship between alcohol consumption and cortisol secretion in an aging cohort. The Journal of Clinical Endocrinology & Metabolism, 93(3), 750-757. [↩]
- Georgakouli, K., Manthou, E., Fatouros, I. G., Deli, C. K., Koutedakis, Y., Theodorakis, Y., & Jamurtas, A. Z. (2022). HPA-Axis Activity and Nutritional Status Correlation in Individuals with Alcohol Use Disorder. Nutrients, 14(23), 4978. [↩]
- Burke, A. R., & Miczek, K. A. (2014). Stress in adolescence and drugs of abuse in rodent models: role of dopamine, CRF, and HPA axis. Psychopharmacology, 231(8), 1557-1580. [↩]
- Rohleder, N., & Kirschbaum, C. (2006). The hypothalamic–pituitary–adrenal (HPA) axis in habitual smokers. International Journal of Psychophysiology, 59(3), 236-243. [↩] [↩]
- al’Absi, M., Hatsukami, D., & Davis, G. L. (2005). Attenuated adrenocorticotropic responses to psychological stress are associated with early smoking relapse. Psychopharmacology, 181, 107-117. [↩] [↩]
- Rhodes, M. E., Kennell, J. S., Belz, E. E., Czambel, R. K., & Rubin, R. T. (2004). Rat estrous cycle influences the sexual diergism of HPA axis stimulation by nicotine. Brain research bulletin, 64(3), 205-213. [↩]
- Mendelson, J. H., Sholar, M. B., Goletiani, N., Siegel, A. J., & Mello, N. K. (2005). Effects of low-and high-nicotine cigarette smoking on mood states and the HPA axis in men. Neuropsychopharmacology, 30(9), 1751-1763. [↩]
- al’Absi, M., Nakajima, M., Allen, S., Lemieux, A., & Hatsukami, D. (2015). Sex differences in hormonal responses to stress and smoking relapse: a prospective examination. Nicotine & Tobacco Research, 17(4), 382-389. [↩]
- Faraday, M. M., Blakeman, K. H., & Grunberg, N. E. (2005). Strain and sex alter effects of stress and nicotine on feeding, body weight, and HPA axis hormones. Pharmacology Biochemistry and Behavior, 80(4), 577-589. [↩]
- Hudson, A., & Stamp, J. A. (2011). Ovarian hormones and propensity to drug relapse: a review. Neuroscience & Biobehavioral Reviews, 35(3), 427-436. [↩]
- Ter Horst, G. J., Wichmann, R., Gerrits, M., Westenbroek, C., & Lin, Y. (2009). Sex differences in stress responses: focus on ovarian hormones. Physiology & behavior, 97(2), 239-249. [↩]
- Sinha, R., Fox, H., Hong, K. I., Sofuoglu, M., Morgan, P. T., & Bergquist, K. T. (2007). Sex steroid hormones, stress response, and drug craving in cocaine-dependent women: implications for relapse susceptibility. Experimental and clinical psychopharmacology, 15(5), 445. [↩]
- Roche, D. J., Childs, E., Epstein, A. M., & King, A. C. (2010). Acute HPA axis response to naltrexone differs in female vs. male smokers. Psychoneuroendocrinology, 35(4), 596-606. [↩]
- Hamidovic, A., Karapetyan, K., Serdarevic, F., Choi, S., Eisenlohr-Moul, T., Pinna, G. (2020). Higher Circulating Cortisol in the Follicular vs. Luteal Phase of the Menstrual Cycle: A Meta-Analysis. Frontiers in Endocrinology, 2(11), 311. [↩] [↩]
- Sinha, R., Garcia, M., Paliwal, P., Kreek, M. J., & Rounsaville, B. J. (2006). Stress-induced cocaine craving and hypothalamic-pituitary-adrenal responses are predictive of cocaine relapse outcomes. Archives of general psychiatry, 63(3), 324–331. [↩] [↩]
- Waldrop, A. E., Price, K. L., Desantis, S. M., Simpson, A. N., Back, S. E., McRae, A. L., Spratt, E. G., Kreek, M. J., & Brady, K. T. (2010). Community-dwelling cocaine-dependent men and women respond differently to social stressors versus cocaine cues. Psychoneuroendocrinology, 35(6), 798–806. [↩]
- Morales-Silva, R. J., Perez-Perez, Y., Alvarado-Torres, J., Rivera-Aviles, N., Rodriguez-Torres, G., Gelpi-Dominguez, U., … & Sepulveda-Orengo, M. T. (2024). Sex-specific effects of chronic stress prior to cocaine exposure on cue-vs drug-induced relapse after prolonged abstinence. Behavioural Brain Research, 474, 115197. [↩]
- Waldrop, Angela. Price, Kimber. Desantis, Stacia. Simpson, Annie…. (2010). Community-dwelling cocaine-dependent men and women respond differently to social stressors versus cocaine cues. Psychoneuroendocrinology, 35(6), 798-806. [↩] [↩]