Abstract
This review examines calcium looping (CaL) as a post-combustion CO2 capture and thermochemical energy storage technology, with a focus on its solar-driven integration and experimental methods. Key quantitative findings are: typical initial sorbent capture capacities are in the range of 0.4-0.6 g CO2 per g sorbent for fresh limestone; practical cyclic retention often falls to 20-40% after 20-30 cycles for untreated limestone due to sintering and attrition. Published techno-economic studies estimate that CaL integration would lead to a reduction of energy efficiency by 6-8% and show a wide range of cost estimates (US$20 to US$160) per tonne CO2, avoided. Recent solar-driven materials report high photothermal absorption (>80-90%) and energy densities up to 1.55 MJ/kg for modified calcium oxide (CaO) particles under laboratory testing. Machine-learning (ML) studies have accelerated screening of doped CaO formulations, predicting capture capacity with ±0.05 g mean-absolute error for compositions within the training domain, but they still underestimate small dopant concentrations (<1 moldm-3) and cannot yet link atomic-level sintering kinetics to macroscopic particle lifetime. This review (i) compares sorbents for practical use, (ii) summarises pilot-scale findings, (iii) evaluates machine learning applications for sorbent design, and (iv) provides specific recommendations for future research in CaL.
Introduction
Fossil-fuel combustion in power plants and industry produces flue gases containing CO2 alongside other pollutants. Carbon capture technologies play a crucial role in reducing these emissions. Specifically, these technologies can be categorised into three categories, all with the same goal of achieving high efficiency and low pollution: pre-combustion capture, oxy-fuel combustion, and post-combustion capture1 Calcium looping (CaL) is a post-combustion approach using the reversible reaction between calcium oxide (CaO) and CO2 to form calcium carbonate (CaCO3), offering the advantages of low-cost natural sorbents due to the abundance of natural ores such as dolomite and limestone, and integration with thermochemical energy storage.
However, CaL is not the only new idea for post-combustion CO2 capture. Solid-sorbent direct-air-capture (DAC), metal-organic-frameworks (MOFs), and amine-functionalised hybrids all exhibit >90% capture, but they still cost far more. A techno-economic study compared these four options on the same coal-plant flue gas. and found that untreated CaL has the lowest equipment cost (US $15–20 per tonne CO₂ avoided) and the smallest energy penalty (6–8 % of plant output)2. MOFs and amine hybrids need 30–50 % more steam or electricity to run pumps and temperature-swing cycles, pushing their avoided-cost above US $60 t⁻¹ even in optimistic projections3. DAC, because it works at 400 ppm CO₂, needs even more energy and is designed for atmospheric capture4. CaL’s cheap limestone sorbent (≈ US $10 t⁻¹) can also be re-used for 300–500 cycles if steam re-activation is added, while MOFs and amines lose >20 % capacity after only 100 cycles1. This positions CaL as a more promising solution compared to other emerging technologies.
This review will first cover the fundamentals of CaL, followed by an introduction to the concepts behind solar integrated CaL. Section 4 will then review experimental and modelling approaches, Section 5 provides a summary of current research and materials innovations, and Section 6 discusses techno-economic, environmental, scale-up issues and pioritises research gaps. The conclusion emphasises CaL’s potential and outlook.
Fundamentals of Calcium Looping (CaL)
CaL was first proposed as a post-combustion carbon capture technology by Shimizu et al. in 19995. The process utilises the reversible reaction of calcium oxide (CaO) and CO2 to result in CaCO3 (Figure 1).
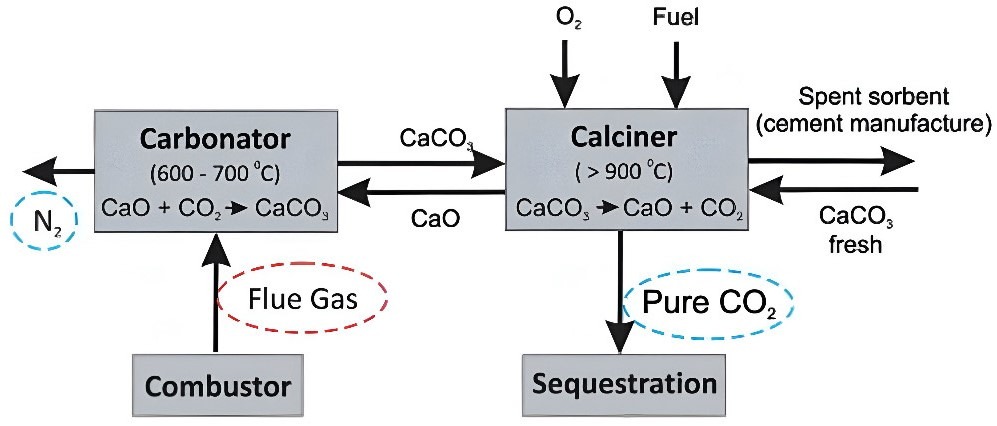
In this process, CaO serves as the sorbent in the carbonator, capturing CO₂ to form solid CaCO₃:While its main role is CO₂ capture, CaO can also react with SO₂, forming calcium sulfate (CaSO₄)6. After CO₂ is captured, the remaining flue gas, which consists primarily of nitrogen, is released into the atmosphere. The process then shifts to the calciner, where the reaction is reversed at higher temperatures7. Since calcination is an endothermic reaction, heat is applied by burning a fuel in the presence of pure oxygen, resulting in pure CO₂ gas that can be captured and stored, thereby helping to reduce carbon emissions and combat climate change8. The regenerated CaO is then sent back to the carbonator to continue the cycle. Whereas, the spent sorbent—CaO that has already reacted with CO₂ and can no longer efficiently capture more—can then be repurposed for cement manufacturing9.
Sorbents: Types, Strengths and Weaknesses
Common sorbents include natural limestones (CaCO₃ → CaO), dolomites (CaMg(CO₃)₂), bio‑derived CaO (eggshells), and synthetic structured pellets such as CaO/Al₂O₃ composites. Fresh limestone shows high initial uptake (~0.4–0.6 g CO₂/g sorbent) but suffers from progressive loss of reactivity due to sintering and attrition, often retaining only 20–40% of initial capacity after tens of cycles without reactivation or doping10. Dolomite offers somewhat lower initial capacity but improved thermal stability, exhibiting stable performance even after 20-30 cycles11. Synthetic CaO/Al2O3 pellets are designed to resist attrition and maintain surface area, trading higher processing costs for better mechanical durability12.
Deactivation Mechanisms: Sintering and Attrition
Sintering is the thermally induced reduction of surface area and pore volume caused by particle coarsening at higher temperatures; it is accelerated at typical calcination temperatures (around 900 °C) and is a primary driver of cyclic capacity loss13‘14. Attrition is the mechanical breakage and generation of fines during particle handling and fluidisation, reducing active sorbent mass over time15. Both effects lead to the need for continuous sorbent make-up or reactivation methods such as hydration.
Role of Dopants and Stabilisers
One effective strategy to reduce sintering is to dope CaO with inert or thermally stable materials. Metal oxides such as MgO, Al₂O₃, and ZrO₂ can be incorporated into the sorbent structure to act as structural stabilisers16. These dopants create a high-melting-point framework that hinders the accumulation of CaO particles, even at high calcination temperatures. For instance, MgO-doped CaO exhibits enhanced resistance to sintering due to the formation of a mixed oxide (e.g., CaMgO₂) that prevents the reduction of sorbent surface area17. Additionally, the listed metal oxide dopants can also improve mechanical strength, reducing attrition during the carbonation-calcination process.
Methodology
To gather information for this literature review, I followed a step-wise search plan. First, I built a short, consistent set of keywords—“calcium looping”, “solar calcination”, “CO₂ capture”—and to ensure the credibility and quality of the information included, I relied on peer-reviewed journals and verified databases such as Google Scholar, ScienceDirect, PubMed and Scopus. Each source had to contain data on sorbent capacity or pilot performance, and the full text had to be accessible. I deliberately prioritised recent publications, typically from the last 10 years, to ensure that the information reflected the latest advances in experimental methods, reactor design, and solar integration. Older publications were generally avoided unless they provided essential background or introduced widely cited foundational concepts in the field.
Solar Integration of CaL Systems
Rationale
Calcination in CaL is energy-intensive because it requires high temperature to heat. This positions concentrated solar power as a feasible solution to supply energy without burning fossil fuels, enabling low-carbon regeneration and the storage of energy as chemical potential in CaO18. Additionally, by directly utilising solar radiation, it can solve challenges faced by traditional CaL, such as the excessively high wall temperature and large thermal resistance. This would lead to a reduction in heat losses and improve the overall efficiency of the system. Laboratory studies of doped CaO particles report high photothermal absorption (>80%) and retained thermochemical energy densities in the range of hundreds of kJ/kg to 1.55 MJ/kg for specially engineered particles; however, these values are laboratory-scale and measured as energy density under controlled conditions rather than continuous CO2 capture performance in a full CaL plant. A pilot study showed a 200kW solar rotary-kiln reaching only 72% calcination,compared with 95% in a conventional 1 MW fluidised-bed calculator, while using 1.4-times more energy per kg CO2 released19.
Solar Receiver and Calciner Concepts
Two broad concepts exist: (i) indirect heating, where heliostats mirrors concentrate sunlight on a receiver that transfers heat (via a heat transfer fluid or medium) to the calciner; and (ii) direct absorbers, where sorbent particles are doped or structured to directly absorb solar radiation and undergo calcination in a calciner. Direct absorption reduces loss of heat in the transfer but requires active sorbents and careful reactor design20.
Reactor Designs Suitable for Solar CaL
Common reactor designs for CaL calcination include bubbling fluidised beds, circulating fluidised beds, entrained flow reactions, rotary kilns and fixed beds. For solar calcination, entrained flow reactors and direct volumetric receivers are ideal due to the uniform high-temperature exposure and short duration. Where, for carbonation, fluidised beds are ideal as they exhibit excellent gas-solid contact but require materials and designs that minimise attrition under heliostat receiver conditions20‘21.
Challenges
Still, there are major challenges involved in this integration, preventing its widespread usage.
Firstly, the scaling up of pilot-scale setups to industrial-scale operations becomes more complex and expensive, as there is a need to incorporate heliostat fields and modify reactors. It is difficult to ensure that these systems maintain or even improve their efficiency and reliability at larger scales, especially as they undergo extensive modifications22. The reactor must be capable of handling much higher energy and feedstock. Scaling up would also require alterations to optimise heat transfer, flow dynamics, and material handling23.
Secondly, the impact of weather fluctuations becomes more pronounced, potentially affecting continuous system operations24. To combat this issue, backup power solutions or thermal energy storage systems would be required to provide a consistent energy supply. Adding to the financial burden, reactors may require heat-resistant alloys or specialised ceramics to withstand extreme conditions25.
Furthermore, integrating these systems into industrial processes typically involves comprehensive environmental assessments and compliance with safety standards, which can add time and cost to the implementation processes26. While advances in research and technology may help mitigate some of these issues over time, the current financial challenges require careful consideration when planning for large-scale industrial operations.
Experimental Approaches
Experimental evaluation of CaL materials and processes uses different tools: (a) thermogravimetric analysis (TGA) for a small sample of the material (usually a few milligrams) and to measure how its mass changes as it reacts with gases such as CO227, (b) pilot-scale fluidised beds (bubbling or circulating) for realistic mixing and heat transfer used in industrial processes, and (c) entrained flow or larger rigs to approximate industrial conditions. Process modelling (Aspen Plus, IECM) and techno-economic analysis translate experimental performance to plant-level impacts28.
Thermogravimetric Analysis (TGA)
With this method, scientists can study how much CO₂ the material can absorb and release, how fast the reactions happen, and how many times the material can repeat the cycle before it loses effectiveness. The main advantage of TGA is that it provides highly accurate data and allows fine control over temperature and gas conditions. However, because the sample size is small, this method does not fully reflect the behaviour of CaL systems at larger, industrial scales10.
Fluidised bed Reactors and Pilot Rigs
For studies that involve larger amounts of material and more realistic conditions, researchers often use fluidised bed reactors. In these reactors, gas is passed upwards through a bed of solid particles, such as limestone or calcium oxide, causing the particles to behave like a fluid. When the gas flow rate is carefully controlled, bubbles form in the bed, resulting in what is called a bubbling fluidised bed29. These systems allow much better mixing and heat transfer, making them more similar to the reactors used in real industrial applications. However, these experiments are more complex to set up, require specialised equipment, and are more difficult to analyse compared to simpler methods like TGA.
Pilot rigs provide the most relevant data for scale-up; several research groups have operated 0.5-2 MWth CaL pilots and reported steady CO2 capture and attrition behaviour under varied feed conditions10.
Entrained Flow Rigs
Entrained flow rigs are used to simulate high-temperature, short-residence calcination and to study sorbent activation and reactivation pathways. They are useful for validating reactivation concepts, e.g. Ca(OH)2 routes, and for conditions closer to industrial calcination conditions10.
Machine Learning
Process models (Aspen Plus, IECM) are used to estimate energy penalties, integration opportunities, and costs. Machine learning (ML) tools are beginning to be applied to discover sorbent formulations and predict cyclic performance from synthesis conditions; ML can accelerate screening but must be used alongside mechanistic understanding and careful cross-validation30.
Table 1 is a comparative table provided to analyse the reliability of fluidised bed studies and TGA in researching different sorbents in calcium looping.
| Sorbent | Findings from FB (Fluidised Bed Studies) | Findings from TGA (Thermogravimetric Analysis) | Practical Implications – Scaling Challenges |
| Limestone (CaCO3) | Initial capture capacity of 0.4-0.6 g CO2/g Rapid capacity decay after 10-20 cyclesSevere performance drop under high calcination temperature (>900 °C)10 | Initial uptake of 0.5-0.6 g CO2/gRetains 20-30% of initial capacity after 20+ cycles31 | High initial capacity but needs stabilisation or doping for long-term use |
| Dolomite (CaMg(CO3)2) | Initial capacity 0.3–0.4 g CO2/g.Better cycle stability and mechanical strength than limestone.More resistant to sintering and attrition.13 | Capture capacity of 0.4-0.5g CO2/gRetention is better than CaCO3 across cycles.Stable microstructure seen, but attrition not evaluated.32 | Thermally stable; lower theoretical capacity but promising for multi-cycle applications |
| Eggshell -derived CaO | Shows initial uptake of 0.4–0.5 g CO2/g.Prone to mechanical degradation and particle breakage.33 | Initial uptake of 0.5–0.6 g CO2/g. Moderate stability over cycles.Porous structure aids reactivity, but sintering still occurs at high temperatures.33 | Low-cost reuse of waste shells; requires shaping/pelleting to limit attrition |
| CaO/Al₂O₃ Structured Pellets | Initial uptake of 0.35-0.4 g CO2/gImproved attrition resistance and mechanical strengthStable cyclic performance5 | Stable CO2 uptake over multiple cyclesLess sintering compared to pure CaO5 | Higher manufacturing cost but better practical durability; good candidate for industrial trials |
However, these results obtained from using TGA or FB analysis have been seen to differ when implemented in the calcium looping scale and conditions. To address the limitation of TGA usage, Mader et al. employed an entrained flow test rig at the University of Stuttgart to simulate industrial conditions. Their work compared the CO2 capture efficiency of calcium-based sorbents immediately after production and following a full CaL cycle under more realistic flow conditions. They found that when the same limestone was tested in a 30kWth fluidised-bed pilot at 850-950°C, the CO2 capture capacity measured under real flue gas was only 0.17 g CO₂ per sorbent gram, which is 43% lower than the TGA tests at the same temperature and cycle length34.
Furthermore, Ortiz et al. introduced a novel integrated CaL simulation designed for coal-fired power plants, where the heat needed to regenerate calcium-based sorbents was supplied by oxygen-fuel combustion. Their simulation of this integrated system showed a modest efficiency penalty of 4 to 7%, which is relatively low given the complexity of carbon capture. Their trial also reached > 95 % calcination in < 5 s time at 900 °C, confirming that short-residence solar receivers can match conventional fluidised-bed conversion if particle temperature is kept above 880 °C; the corresponding TGA test needed 60 s to reach the same conversion35. However, these conclusions were based on an idealised reactor model, and extending the time in an industrial-scale reactor presents significant practical challenges, such as ensuring consistent flow and avoiding agglomeration.
Table 2 provides a comparison of sorbents under a practical criteria. Relative cost is included instead of cost per tonne as the range of sorbents vary widely.
| Sorbent | Raw material availability / relative cost | Cyclic retention (after 20-30 cycles) | Attrition resistance (qualitative) | Processing requirements |
| Limestone (CaCO3) | High/low cost (widely available locally) | 20-40% (untreated) | Moderate, fines generation is common | Common feedstock; needs stabilisation or replacement |
| Dolomite (CaMg(CO3)2) | Medium/abundant regionally | 40-60% | Higher resistance | Promising for high-temperature stability |
| Eggshell -derived CaO | Low / Waste reuse | 30-50% | Low unless pelletised | Good circular economy; requires pelletising and quality control |
| CaO/Al₂O₃ Structured Pellets | Medium-high (is manufactured) | 50-80% | High (engineered stability) | Higher CAPEX for pellets but lower sorbent make-up and better lifetime |
Current Research Trends and Innovations
Optical Doping and Direct Photothermal Sorbents
Recent work focuses on doping CaCO₃/CaO with transition metal oxides (Mn–Fe, Ce/Co/Mn multi‑doping) or adding components that produce negative thermal expansion to improve direct solar absorption and mechanical stability37. Specifically, Teng et al. (2020) fabricated CaCO3-based composites with doping Mn–Fe oxides to enhance the ability of capturing sunlight, and results showed that the light absorption was 90.15% and the heat storage density only decreased 3.31% after 60 cycles38. Whereas Da et al. (2020) demonstrated that multi-doping with cerium, cobalt, and manganese produced composites with a light absorption efficiency of 83%, maintaining over 64% energy storage efficiency after 20 cycles. In addition, Liu et al. (2023) introduced a novel approach by incorporating a negative thermal expansion component into CaO particles. This strategy resulted in a solar absorption of 90.8%, energy densities up to 1554 kJ/kg, and excellent cyclic stability over 260 cycles39.
Machine Learning
Calcium looping research is moving toward using data-driven methods like machine learning to speed up the development of sorbents and improve the overall process. Techniques such as linear regression, decision trees, and logistic regression can help predict important properties of materials and how well they capture CO₂ without relying on complicated mathematical models40. One of the key benefits of machine learning is its ability to find statistically significant correlations between materials (e.g., synthesis methods and operational conditions) and performance metrics (e.g., CO₂ capture capacity and long-term stability)41. By studying large amounts of data from experiments and simulations, machine learning can uncover patterns that might not be obvious otherwise, helping scientists design better CaO-based sorbents and fine-tune the process for greater efficiency and longer-lasting materials. This approach can reduce the amount of trial-and-error testing needed and bring CaL technology closer to real-world use.
For example, a model trained on 1000 experimental cycles (10 CaO chemistries, 4 solar-flux levels, 20–900 °C) predicted residual conversion after 30 cycles with a mean-absolute-error of 0.05 g CO₂ g⁻¹ sorbent; the same model identified that ≥ 3.2 wt % Al₂O₃ is required to keep conversion above 0.20 g g⁻¹ under 1 MW m⁻² solar flux, a threshold later confirmed in a 10 kWth solar drop-tube reactor42.
Techno-Economic and Environmental Considerations
Efficiency penalty and cost ranges
Multiple techno-economic studies find that CaL imposes a modest efficiency penalty compared with a base power plant: commonly reported estimates are ~6-8 percentage penalty from the plant efficiency (versus ~ 9.5-12.5% for amine scrubbing in some studies)43. Costs of capture depend strongly on assumptions (plant size, fuel, sorbent lifetime, integration). Reported values in the literature range from relatively low figures (~US$20–50/tCO₂ avoided in some early or optimistic studies) to higher estimates (>US$100/tCO₂) for conservative assumptions or early‑stage systems; uncertainty is large and sensitivity analysis is essential44.
Environmental and Lifecycle Impacts
Lifecycle assessment must consider the extraction and processing of raw carbonates, sorbent make-up rates, fines handling, and potential utilisation in cement feedstock. Early lifecycle assessment indicates tradeoffs: CaL may reduce fuel combustion if solar or low-carbon heat is used for calcination, but can shift environmental burdens to mineral extraction and sorbent processing if make-up rates are high45. For example, supplying 1.3–1.5 wt % fresh limestone make-up per cycle adds about 0.18 t CO₂-eq upstream per tonne of CO₂ captured, while recycling the spent sorbent as 0.45 t clinker substitute can avoid a further 0.33 t CO₂, keeping the net avoidance cost near 55 € t⁻¹ CO₂46.
Prioritised Research Needs
As CaL continues to gain attention as a carbon capture technology, several research gaps remain to be addressed. A notable gap lies in the lack of pilot-scale testing for many promising sorbents. Much of the current research has been carried out at the laboratory scale using techniques such as TGA, but it does not consider the key factors in experimental conditions. Moreover, the physical wear and attrition of sorbents during extended use in large reactors remain unclear, making it difficult to predict operational challenges at a large scale. To advance CaL technology toward commercial use, more pilot-scale experiments are needed to validate lab-scale findings and assess sorbent behaviour under conditions that resemble industrial processes.
Advancing CaL technology will require stronger interdisciplinary collaboration across fields such as materials science, environmental science, and economics. From a materials science perspective, further work is needed to engineer sorbents with improved resistance to sintering, better mechanical strength, and higher CO₂ capture efficiency. At the same time, environmental scientists play an important role in assessing the impact of CaL deployment, including emissions of pollutants, energy consumption, and waste generation throughout the process. Integrating calcium looping into existing industrial systems—such as power plants or cement factories—also demands detailed techno-economic analysis to evaluate operational costs, energy penalties, and lifecycle benefits. Such studies are essential to identify the most cost-effective sorbents and process designs while balancing environmental goals with economic feasibility.
Looking ahead, advanced tools such as machine learning and multi-scale simulations are likely to become more prominent in CaL research. These methods can improve the discovery of new sorbent materials and optimise reactor designs by identifying key performance patterns from large datasets. By combining experimental work, process modelling, and data-driven approaches, researchers can address the current gaps and bring calcium looping closer to large-scale implementation as an efficient and sustainable carbon capture solution. As the field evolves, it is worth asking—how might the integration of automation reshape the operation and scalability of calcium looping systems in the future?
The following is a list to summarise which research gaps would benefit from increased attention:
- Pilot-scale validation of solar-driven calcination integrated with carbonators.
- Mechanical studies into the photothermal conversion behaviour of sorbents.
- Standardised attrition testing and multi-cycle pilots with industrially relevant sorbents.
- Techno-economic studies with uncertainty quantification (Monte Carlo, parametric sensitivity) and lifecycle assessments across deployment conditions
- Open datasets for Machine Learning model development and cross-lab evaluation.
Conclusion
Calcium looping is a versatile carbon capture and thermochemical storage approach with clear advantages: low raw sorbent cost, compatibility with fluidised‑bed reactors, and the potential to store solar energy as chemical potential. However, limitations remain: sorbent deactivation through sintering and attrition, scale‑up costs, and the need for robust techno‑economic validation. Solar integration leads to low-carbon calcination but adds complexity in receivers and materials. This review has summarised data on sorbent performance, pilot operations and solar-reactor concepts to provide a critical overview of CaL’s status and future research priorities.
Author’s Note
My interest in carbon capture technologies began with a general interest in the field of chemical engineering, and I aimed to read more about the field. This interest deepened during my internship at a waste-to-energy facility, where I observed firsthand how flue gas is treated to remove pollutants. I was also exposed to the challenges that the plant faced regarding environmental regulations, limitations of specific materials and the cost of production. I chose to investigate CaL because it is a newer, potentially more sustainable solution with a wide range of applications beyond just carbon capture, such as energy storage and fuel production. Its ability to serve multiple roles made it especially appealing for me to explore in greater depth.
This literature review holds personal relevance for me because it allows me to explore a topic that connects both my academic interests and real-world observations from my internship. Ultimately, this project has shown me that even as a student, I can meaningfully engage with complex scientific topics and connect them to real-world environmental challenges.
References
- Tiwary, S., Bhaumik K.S., 2022, Journal of CO2 Utilisation [↩] [↩]
- Rao, G., Senthilkumar P., Ratchagaraja D., Rajendran, S., 2024, Global NEST Journal [↩]
- Yang, Z., Boshi, C., Chen, H., Li, H., 2023, Frontiers in Energy Research [↩]
- Realmonte, G., Drouet, L., Gambhir, A., Glynn, J., Hawkes, A., Köberle, A.C., Tavoni, M., 2019, Nature Communications [↩]
- Valverde, J.M., 2013, Journal of Materials Chemistry [↩] [↩] [↩] [↩]
- Hu, Y., Liu, W., Peng, Y., Yang, Y., Sun, J., Chen, H., Zhou, Z. and Xu, M., 2017, Fuel Processing Technology [↩]
- Li, X., Zhao, K., Li, Z., Li, X. Peng K., 2024, Journal of Cleaner Production [↩]
- Perejón, A., Luis, M.R., Yolanda, L., Pilar, L., Ana M., and Jose, M.W.. 2016, Journal of Cleaner Production [↩]
- Valverde, J.M., 2013, Journal of Materials Chemistry [↩]
- R. T. Symonds, D. Y. Lu, V. Manović, E. J. Anthony, 2012, Industrial & Engineering Chemistry Research [↩] [↩] [↩] [↩] [↩]
- Perejón, A., Luis, M.R., Yolanda, L., Pilar, L., Ana M., and Jose, M.W.. 2016, Journal of Cleaner Production [↩]
- Tiwary, S., Bhaumik K.S., 2022, Journal of CO2 Utilisation [↩]
- Coppola, A., Fabio, M., Piero, S., Fabrizio S., 2012., Chemical Engineering Journal [↩] [↩]
- Chen, J., Lunbo, D., Zhenkun S.., 2020. Energy & Fuel [↩]
- Xu, Z., Tao, J., Hao Z., Yujun, Z., Xinbin, M., Shengping W., 2021, Frontiers of Chemical Science and Engineering [↩]
- Han, R., Yang, W., Shuang, X., Caihong, P., Yang, H., Chunfeng ,S., Qingling, L.. 2022, Chemical Engineering Journal [↩]
- Tian, X. K., S. C. Lin, J. Yan, C. Y. Zhao, 2022, Chemical Engineering Journal [↩]
- Jiang, D., Shouzhuang, Li., Annukka S., Mika, J., 2024, Energy Conversion and Management [↩]
- Alvarez, M., Rodrigues, D., Pinheiro, C.I.C., Cardoso, J.P. Mendes, L.F., 2022, Renewable and Sustainable Energy [↩]
- Chacartegui, R., A. Alovisio, Ortiz, C., Verda, V. Becerra, J.J., 2016, Applied Energy [↩] [↩]
- Lisbona, P., Bailera, M., Hills, T., Sceats, M., Díez, L.I. and Romeo, L.M., 2020, Renewable Energy [↩]
- Sansaniwal, S.K., 2019, International Journal of Ambient Energy [↩]
- Saghafifar, M., Gabra, S., 2020, International Journal of Greenhouse Gas Control [↩]
- Mahmudul, H.M., Rasul, M.G., Akbar, D., Narayanan, R. and Mofijur, M., 2021, Science of The Total Environment [↩]
- Teng, L., Xuan, Y., Da, Y., Liu, X., Ding, Y., 2020, Energy Stoarge Materials [↩]
- Da, Y., Zhou, J. 2022, Solar Energy Materials and Solar Cells [↩]
- Fennell, P., Anthony, B., 2015, Woodhead Publishing [↩]
- Mahmudul, H.M., Rasul, M.G., Akbar, D., Narayanan, R. and Mofijur, M., 2021, Science of The Total Environment [↩]
- Fennell, P., Anthony, B., 2015, Woodhead Publishing [↩]
- Jiang, D., Shouzhuang, Li., Annukka S., Mika, J., 2024, Energy Conversion and Management [↩]
- Borgwardt, R.H., 1989, Chemical Engineering Science [↩]
- Grasa, G., Abanades, J.C.,Anthony, E.J., 2009, Industrial & Engineering Chemistry Research [↩]
- Blamey, J., Anthony, E.J., Wang, J. and Fennell, P.S., 2010, Progress in Energy and Combustion Science [↩] [↩]
- Mader, N., Rastogi, M., Maier, J.,Scheffknecht, G. (2024, Electronic Journal [↩]
- Ortiz, C., Chacartegui, R., Valverde, J.M. Becerra, J.A., 2016, Applied Energy [↩] [↩]
- Wang, Z., Ma, C., Harrison, A., Khulud Alsouleman, Gao, M., Huang, Z., Chen, Q., Nie, B., 2025, Small [↩]
- Erans, M., Manovic, V., Anthony, E.J., 2016, Applied Energy [↩]
- Teng, L., Xuan, Y., Da, Y., Liu, X., Ding, Y., 2020, Energy Storage Materials [↩]
- Liu, J., Xuan, Y., Teng, L., Sun, C., Zhu, Q., Liu, X., 2023, Energy Advances [↩]
- Wang, Z., Ma, C., Harrison, A., Khulud Alsouleman, Gao, M., Huang, Z., Chen, Q., Nie, B. (2025, Small [↩]
- Wang, Z., Ma, C., Harrison, A., Khulud Alsouleman, Gao, M., Huang, Z., Chen, Q., Nie, B. (2025, Small [↩]
- Theodoros Papalas, Antzaras, A.N., Lemonidou, A.A., 2024, Energy & Fuels [↩]
- Rao, G., Senthilkumar P., Ratchagaraja D., Rajendran, S., 2024, Global NEST Journal [↩]
- Hanak, D.P., Biliyok, C., Anthony, E.J., Manovic, V., 2015, International Journal of Greenhouse Gas Control [↩]
- Ortiz, C., Chacartegui, R., Pérez-Maqueda, L.A., Pau Gimenez-Gavarrell, 2021, Kona Powder and Particle Journal [↩]
- Hanak, D.P., Biliyok, C., Anthony, E.J., Manovic, V., 2015, International Journal of Greenhouse Gas Control [↩]